A quantitative method for collecting water mites in lotic, riffle-run habitats for water quality biomonitoring
Stenger, Logan R.1
and Skvarla, Michael J.  2
2
1✉ Department of Entomology, Penn State University, University Park, Pennsylvania, U.S.A.
2Department of Entomology, Penn State University, University Park, Pennsylvania, U.S.A.
2022 - Volume: 62 Issue: 3 pages: 621-636
https://doi.org/10.24349/05qz-joakOriginal research
Keywords
Abstract
Introduction
Approximately 85% of all known freshwater species (~126,000) are invertebrates, with the majority comprised of insects (60%), crustaceans (10%), and arachnids (5%), (Balian et al. 2008). While arachnids are not commonly associated with aquatic systems, some of the more well-recognized arachnids, such as spiders, live at the transitional edge between water and land, with some species even capable of submerging for short periods of time (Voshell 2002). However, the only true aquatic group of arachnids are water mites (Trombidiformes: Parasitengona: Hydrachnidia) (Voshell 2002; Smith et al. 2010). Despite comprising only a small percent of the global freshwater biodiversity, water mites are among the most diverse and abundant groups of benthic macroinvertebrates in the world.
While nearly 7,500 species, representing more than 300 genera, 59 families and 8 superfamilies, have been described, acarologists conservatively estimate that another 4,000 species have yet to be discovered (Smit 2020). Water mites can be found in nearly every known lentic or lotic freshwater environment, as well as most saltwater environments (Smith et al. 2010; Fisher 2016). In lotic systems, water mites are commonly found in fast-flowing, riffle-run stream habitats, where one square meter of substrate can contain as many as 5,000 individual mites representing more than 50 species, 30 genera, and all 8 superfamilies (Smith et al. 2010). In lentic systems, as many as 2,000 individual water mites representing 75 species and more than 30 genera have been collected from a single square meter of substrate in littoral weed beds (Smith et al. 2010). In addition, water mites have a close connection with benthic macroinvertebrate communities as their parasitic larvae are reliant on various aquatic invertebrates as hosts while deutonymphs and adults fill a predatory role (Di Sabatino et al. 2000; Smith et al. 2010).
Given their diversity and abundance across aquatic environments, previous literature has primarily emphasized improving our understanding of water mite taxonomy, distribution, and life history. However, several studies have examined whether water mites might be used as bioindicators of water quality conditions (e.g., Gerecke and Schwoerbel 1992; Growns 2001; Lombardo et al. 2013; Goldschmidt et al. 2016). Despite their relative diversity, abundance, and connection with benthic macroinvertebrate communities, water mites are widely neglected in freshwater biomonitoring strategies (Goldschmidt 2016). In fact, in most bioassessment protocols water mites are either ignored completely or identified collectively as ''Hydracarina'' or ''Hydrachnidia,'' an artificial grouping of several water mite superfamilies (Goldschmidt 2016). Two exceptions include the Canadian CABIN protocols (Environment and Climate Change Canada 2020) and Australian River Assessment System (Australian Government 1994), which require the identification of water mites to at least family level (Goldschmidt 2016).
Until now, most studies have relied on qualitative sampling methods, such as those described by Smith et al. (2010), as the primary means for collecting water mites for taxonomic and life history purposes (e.g., Di Sabatino et al. 2004; Matsumoto et al. 2005; Radwell et al. 2011; Fisher et al. 2015). However, to further examine water mites and their potential role as bioindicators, future studies need to be able to compare water mite abundance and diversity across numerous samples collected from both impacted and unimpacted aquatic environments. Therefore, acaralogists have acknowledged the need for a standardized, quantitative sampling method (Smith et al. 2010). We propose such a standardized sampling protocol herein.
Methods
The collection protocol given here is modified from the water mite collecting methods proposed by Smith et al (2010) and incorporates standardizations included in the Wadable Freestone Riffle-Run Stream Macroinvertebrate Assessment Method used by the Pennsylvania Department of Environmental Protection (Chalfant 2018).
To determine that the rinsing method proposed below successfully removed the majority of water mites from a final composited sample, we retained the contents from rinsed composited samples for three study sites. These samples underwent the entire rinse and seine process described below, however, instead of discarding the final composited sample contents afterwards, we retained them to determine if any water mites remained in the sample. These samples were taken to the lab and picked with the same method described a below. Hereafter, these samples are referred to as ''unfiltered'' samples.
All water mite collections were made under Pennsylvania Fish and Boat Commission Type I Scientific Collector Permit Number 2021-01-0017.
Collection protocol. Water mite collection begins by delineating a 100-meter reach along the stream of interest from which collections will take place. The proposed protocol targets riffle habitats (Figure 1), so the 100-meter reach should contain enough riffles to be sampled. If enough riffles are not present, another method may need to be employed. Within the selected reach, the collector will sample four separate one square meter areas of riffle-run habitat. All four individual samples are combined to create a final composited sample for each stream reach that is assessed. If possible, the collector should attempt to distribute these samples across a variety of riffle habitats (e.g., fast-flowing, shallow riffles and slow-flowing, deeper riffles) as well as across the stream channel (e.g., left, middle, and right areas of the stream). This is done to ensure the final, composited sample provides an accurate representation of the riffle-run water mite community throughout the stream reach. Samples should be collected in a downstream to upstream direction to prevent the downstream drift of dislodged water mites, silt, and detritus from upstream to downstream sites.



The proposed method for collecting water mites is a two-person process but can be accomplished by one person if needed. One individual will place and hold a 12-inch-wide (31 cm) × 10-inch-high (25 cm) non-truncated D-frame net with 250 μm mesh flat against the stream bottom (Figure 2). The net should not be placed in riffles where the water depth exceeds the net height (25cm). This net was custom made as D-frame nets with 250 μm mesh are not commercially available. To create this net, we purchased a truncated, D-frame net with 500 μm mesh (Figure 2), removed the mesh netting, and replaced the mesh with a hand sewn, non-truncated pattern created from 250 μm fabric. If large rocks are in the square meter immediately upstream of the net, these can be moved to allow access to the finer substrate beneath. These rocks and others from nearby can be placed at either side of the net to help focus the flow of water through the net (Figure 3). The second collector will then use a spade shovel to disturb a one square meter area immediately upstream of the net for three minutes (Figures 3, 4), which the net-holder keeps time of with a stopwatch. Collectors should be cognizant to not disturb the substrate upstream of the net until the collector is ready to begin the sample collection. Since the area of disturbance is larger than the sampling net being used, it is highly likely that some water mites will drift past the net and not be collected. However, the goal of this protocol is to collect a representative sample of the riffle-run water mite community, not every water mite dislodged by shoveling, so missing mites is acceptable. Indeed, is it widely acknowledged that benthic macroinvertebrate collection methods do not capture every single specimen dislodged during sampling, which is why current protocols include guidelines for reducing the number of specimens missed (e.g., not completely submerging the D-net so specimens flow over as well as around it). If just one person is collecting, the net can be held upright in place by piling rocks around it, although this can be difficult or impossible in fast-flowing streams.









After the three minute shoveling period ends, the contents collected in the net are placed into a single plastic sample container. After the second, third, and fourth collections are made, they are also placed into the same sample container as the first to make a single, composited sample. It is recommended that the collectors place this container within a large bucket (we used a five-gallon bucket; a 20 liter or other large plastic bucket should work as well) as they traverse the stream from the downstream sites to the upstream sites. The bucket allows easier transportation of the sample container and helps catch any contents that fall out of the net and but into the sample container (Figure 5). Any spilled contents can then be scooped by hand or using a spoon back into the sample container. An ideal sample container is four-sided with a screw on lid (e.g., reused plastic cat treat or mixed nut containers). Depending on the amount of fine sediment within the stream, it is recommended that sample containers be no less than 1.5 liters and/or that two collection containers be used. After the collection is finished, the final composited sample is now ready to be sieved.



Sieve protocol. The contents of the final composited sample often contain a mix of various size substrate (e.g., silt, sand, gravel, and cobble) and organic matter (e.g., leaves, sticks, or algae). The purpose of the sieving process is to concentrate the water mites into a smaller sample by removing materials ≥3mm from the sample. This makes it easier to pick the water mites from the final, condensed sample in the lab. The sieve process proposed here requires two sieves, a 3mm mesh sieve and a 250-micron mesh sieve. It is important that both sieves are the same dimensions so they can be stacked on top of one another. The sieves used in his study were 9 cm (3½ inches) high with a 31 cm (12-inch) diameter.
It is easiest to complete the sieve process at the sample site along a stream bank, gravel bar, or other nearby flat area (Figure 6). To begin the sieve process, stack the 3mm sieve on top of the 250-micron sieve. Then, take the composited sample container and fill the container approximately three-quarters of the way full with water. This can be done by carefully placing the container into the stream to allow water to enter the mouth of the container. Caution should be taken not to allow the container to overflow as this could remove water mites from the sample. Alternatively, one can collect stream water using another empty container and pour the water directly into the composited sample container. Since stream water is to be utilized to rinse the sample, it is recommended that the sieve process be completed at the upstream end of the sample reach to avoid collecting water that could harbor drifting water mites that were dislodged during the previous shoveling collection efforts. Preliminary data not presented here suggests that a negligible number of water mites are collected from undisturbed, flowing water so using unfiltered stream water should not affect the collections.



With the composited sample container filled three-quarters of the way with water, secure the lid back onto the container. Next, vigorously shake the container for approximately 5–10 seconds. This dislodges the water mites from the substrate and temporarily suspends them in the water column. Remove the lid and dump the water into the two stacked sieves. Repeat this rinsing process ten times. While some sediment or organic matter may fall out of the container into the sieves during each pour, care should be taken to minimize the amount falling into the sieves. If larger stones, leaves, or sticks in the composited sample container prevent the contents in the container from being thoroughly shaken, one can manually remove and place them into the 3mm sieve. If multiple sample containers were used due to large amounts of sediment in the net, then this process should be applied to each container individually. Once the sample container has been shaken and poured ten times, the remaining substrate can be returned to the stream. Fill the empty container with stream water and pour the water over the 3mm sieve to provide a final rinse of the larger materials in the sieve. Repeat this final rinse two or three times as needed.
Remove the 3mm sieve from the 250 μm sieve. The contents in the 3mm sieve can be returned to the stream. Collectors should observe that the 250 μm sieve has only accumulated fine sediment (\textless3 mm) (Figure 7). Using a spoon, carefully scoop and transfer the sediment from the sieve into a clean, empty sample container. If needed, one can gently pour water into the sieve to push all the sediment towards one end of the sieve, making it easier to scoop. After most of the sediment has been transferred from the sieve, you can fill a clean container with water, tip the 250 μm sieve vertically, and gently pour water against the back/underside to wash the remainder of the sediment into the final collection container. Be sure not to overfill the collection container while washing this remaining sediment. If there is still a lot of air space in the collection container after the 250 μm sieve has been rinsed, top off the container with water. After all of the contents have been transferred from the sieve to the sample container (Figure 8), label the inside and outside of the container with the date, time, location, and identity of the collectors using a graphite pencil or water-proof pen on cardstock, waterproof paper, or other sturdy material (standard printer paper dissolves into pulp when submerged in water for any period of time and labels on such paper may be destroyed). If multiple samples are collected in the same day, it is recommended that the samples be placed in a cooler with or without ice to prevent the sample from overheating or freezing, depending on the conditions and time of year.






Picking and preservation protocol. Upon completion of the field sampling, the water mite samples will need to be returned to the lab and should be prepped for picking on the same day they are collected. In the lab, prepare a single, white sorting tray (photography development trays with rounded corners are ideal) for each final composited sample. It is highly recommended that all sorting trays be white as this makes it easier for the collector to observe the water mites swimming in the sorting tray. Clear and dark colored sorting trays should be avoided. In addition, there are no specific size requirements for the sorting trays, however, the trays need to be large enough to hold all the sediment from the sample while it is completely submerged in water. For this study, the dimensions of the sorting trays were 17 inches long (43 cm) x 14 inches wide (36 cm) x 2 inches high (5 cm). The sediment from the sample should be spread out across the tray in a manner that a one-to-two-inch perimeter of the tray is left free of sediment as the water mites tend to accumulate along the edges and in the corners of the sorting tray.
To begin the picking process, remove a sample container from the cooler and pour the contents into a sorting tray (Figure 9). It is important that all the contents from the container are transferred to the sorting tray. If needed, a spoon may be used to help remove all the sediment from the sample container. Using deionized water, the sample container can also be rinsed with water and poured into the sorting tray to ensure all contents are removed from the container. Once the contents are in the sorting tray, additional deionized water may need to be added to completely submerge all of the sediment. Repeat this process for each of the collected samples.



Once all the samples have been transferred into sorting trays, allow the trays to settle for approximately 20–30 minutes. This provides time for any suspended silt to settle and the water mites to emerge from the sediment and begin swimming around the sorting tray. If the collectors notice there is an excessive amount of fine silt suspended in the water that is impeding the ability to observe water mites, then the contents of the sample may need to be returned to a 250 μm sieve and gently rinsed under a sink. After rinsing, the sample contents should be returned and submerged back in the sorting tray. If this process needs to be repeated for multiple samples, either use a separate 250-micron sieve for each sample or ensure the sieve is thoroughly washed and cleaned in between samples to prevent cross-contamination between samples.
After the 20–30 minute settling period has ended, the collector will use a 10mL pipette to collect water mites from the sorting tray. All water mites should be transferred into a 4-dram glass vial with each sorting tray having its own respective vial. This process will continue for a six-hour period. The collector should check the sorting trays at regular 15–20 minute intervals for the first two hours of the picking process. Afterwards, the collector may extend this interval to 45–60 minutes for the final four hours if water mite abundance has decreased. In this study, researchers observed that the majority of a sample's water mites were collected within the first two to three hours of picking.
At the end of the six-hour picking period, water should be removed from the 4-dram vial using the pipette so that the vial is only ¼ of the way full or less. Care should be taken not to remove any water mites from the vial when removing the water. Place a label inside the vial that includes the date and time of collection, location, identity of the collectors, and collection code to link the vial to a more thorough collection data sheet (if such a sheet was kept). It is recommended that waterproof paper and ethanol-resistant ink or a graphite pencil be used to create the label. Fill the remainder of the vial with 80% ethanol (Figure 10) or GAW (also referred to as Koenike's solution; 50% glycerol, 10% glacial acetic acid, and 40% water).



The choice of preservative depends on the goals of the project. If DNA extraction is desired, ethanol is required. However, ethanol can make mites brittle and prone to leg and other appendage breakage; it also tends to make fine silt and other detritus stick to preserved mites. If mites are to be slide mounted without DNA extraction, they can be stored in GAW, which gently clears the mites and keeps them supple but destroys DNA (see Fisher et al 2015 for additional discussion about ethanol and GAW).
Once all of the vials have been labelled and preserved, the contents of the sorting trays can be discarded and the trays cleaned and dried. The vials now represent a sample of the water mite community from the targeted stream reach and are ready for identification.
Identification protocol. To begin identification, water mites are removed from the sample vial with a pipette and placed into a small, glass petri dish filled with 80% ethanol or GAW. A pipette is preferred rather than dumping the vial to ensure water mites are not accidentally left sticking to the sides of the vial. At no point should the water mites be exposed to air long enough to dry as this can cause the mites to become brittle and shriveled, rendering them unidentifiable.
Once the contents of the vial are removed, the petri dish can be placed under a microscope for identification and enumeration. During the previous picking process, it is possible that small amounts of fine organic matter or substrate was collected with the water mites. If large quantities of organic matter or substrate are present, the identifier may need to remove the water mites from the material before identification. This can be done by using a pair of fine-tipped forceps and the water mites can be placed into a clean petri dish.
Water mites can typically be identified to the family or genus level at magnifications ≤ 40 (Figure 11). However, smaller-sized taxa or specimens needing to be identified to species may need to be slide mounted and placed under higher magnification. A number of keys for identifying water mites to family and genus are available, including Smith et al. (2010) and Smit (2020). Once identification is complete, the individual taxa identified in the sample are returned to a labelled sample vial with 80% ethanol or GAW. Labels should now include the date and time of collection, location, identity of the collectors, collection code, and the specific taxa (e.g., family, genus, species) present within the vial.



Results
In August 2021, the proposed collection method was utilized to collect 23 water mite samples from 12 agriculturally impaired (polluted) and 11 attaining (unpolluted) high-quality, coldwater streams (PADEP 2022) in central Pennsylvania, U.S.A. (Table 1). The described field protocol (i.e., collection and sieving but not picking mites) took approximately 45 to 60 minutes per site. In total, a combined 8,447 water mites were collected representing 16 genera and 10 families. Of the total water mites collected, less than 1% (n=73) have not yet been identified as they need to be slide mounted. Individual site abundance ranged from 0 to 2,473 water mites. All specimens have been or will be accessioned in the Frost Entomological Museum at the Pennsylvania State University in State College, PA, U.SA. for further bioindicator analyses and future taxonomic studies.
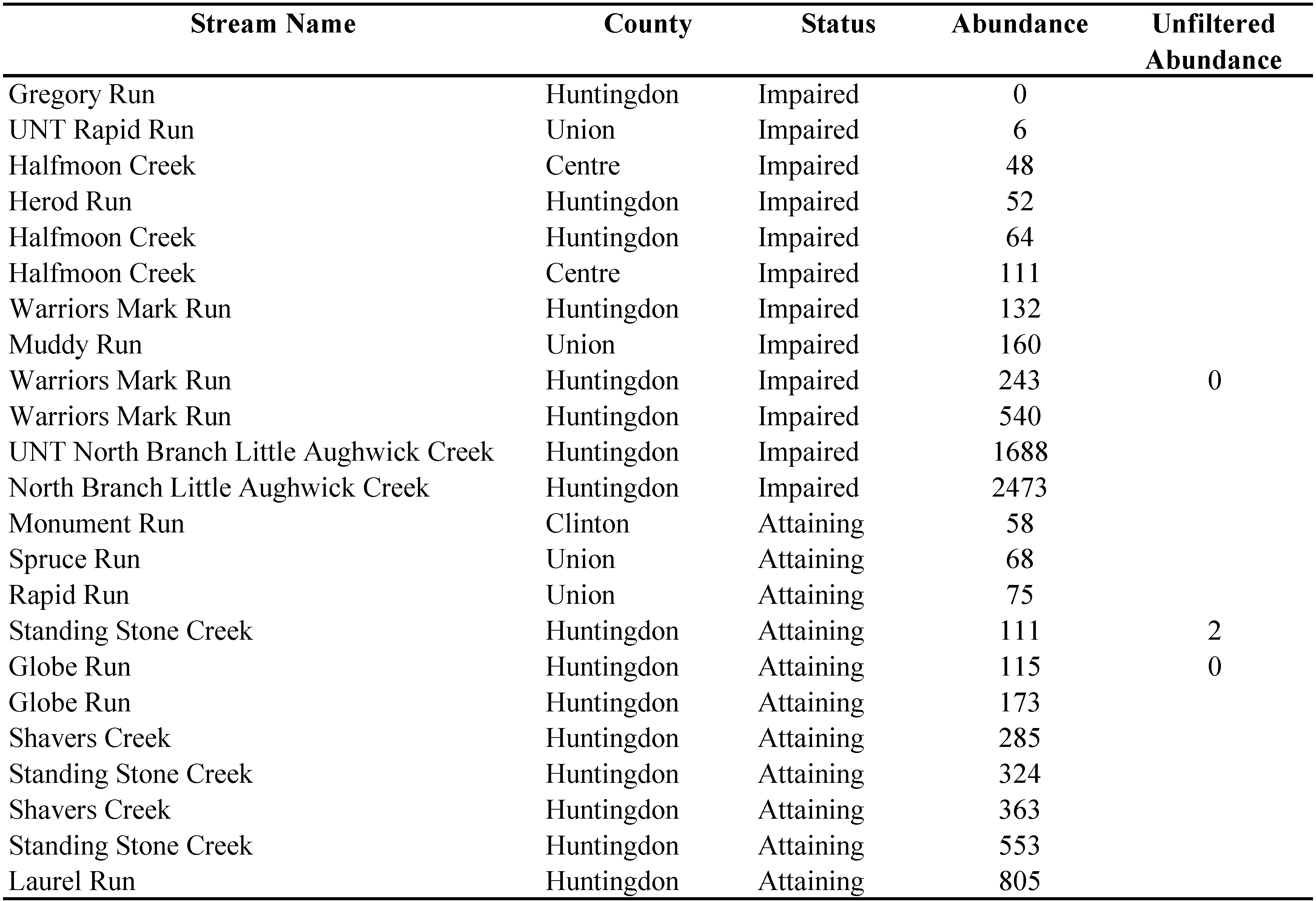


Two of the three unfiltered samples contained zero mites, while one unfiltered sample contained two mites (1.8% of the mites collected from the seined contents of the same sample). While this indicates that some water mites may remain in the composited sample after 10x rinses, it is a small amount that should be considered insignificant in comparison to the amount of water mites collected from the seined samples.
Discussion
Using the method proposed here, we successively collected water mites from riffle-run habitats. Future papers will explore whether the mites collected are accurate bioindicators that can be used to measure water quality. Modifications may be necessary for quantitative sampling in other habitats such as deeper, slower-flowing areas like pools and backwater habitats, larger habitats such as river, and of course lentic habitats like lakes and ponds.
Because the goal of our method is to determine stream quality using mites found in riffle-run habitats, the proposed method does not collect all water mite diversity. For example, Growns (2001) found that, in unpolluted streams, edge habitat samples harbored a greater diversity of water mites than riffle habitat samples. In addition, species overlap between edge and riffle habitats was minimal. By not including multiple habitats in our collection method, it is likely our samples failed to include ''edge'' preferring water mite taxa, which limits our interpretations of the entire water mite community.
However, the majority of the world's recognized benthic macroinvertebrate bioassessment protocols utilize the ''single, most productive habitat'' approach (Barbour et al. 1999). In other words, samples are collected from habitats that harbor the greatest number of pollution sensitive species, including mayflies, stoneflies, and caddisflies. In most cases, riffle-run habitats are generally recognized as the most productive habitat due to their lotic-erosional state and high dissolved oxygen concentrations. Pennsylvania streams are primarily dominated by cobble substrate and riffle-run habitats and therefore utilize this ''single, most productive habitat'' biomonitoring approach (Chalfant 2018). However, edge habitats have been shown to harbor a greater diversity of water mites compared to riffles elsewhere in the world (Growns 2001). Future studies should examine whether this is generally applicable worldwide by comparing riffle and edge habitats across a range of stream orders (e.g., headwater streams vs large rivers).
One reason water mites are often excluded from biomonitoring strategies is the conception that water mites are too difficult to sample and identify (Goldschmidt 2016). While the method proposed here uses some equipment that is different from sampling benthic macroinvertebrates (e.g., a 250 μm mesh D-net), the additional equipment is not cost prohibitive (although constructing a custom D-net will require some extra labor as there are currently no commercial options available). The described field protocol takes a similar amount of time (approximately 45-60 minutes per sample site) compared to standard methods used to collect a wadeable, riffle-run benthic macroinvertebrate sample (Chalfant 2018) and is no more physically rigorous, so collecting water mites using this protocol is comparable to methods already used to estimate water quality. This is important as water mite sampling methods that take more time or effort compared to current benthic macroinvertebrate sampling methods are not likely to be adopted when more efficient methods are available.
More than 99% of the specimens were identified to genus using a stereomicroscope that is appropriate for identifying other benthic macroinvertebrates and without slide mounting, so the relatively small size (compared to most benthic macroinvertebrates) should not be an issue for those equipped to identify benthic macroinvertebrates. That said, most keys to water mites rely on characters that are best seen on slide-mounted specimens and there is therefore a need for field guide-style resources for identifying water mites using a stereomicroscope.
While we have demonstrated that the method proposed here successfully collects water mites and has the benefit that it is comparable in effort to benthic macroinvertebrate collection methods currently in use, there are other water mite collection protocols that have been proposed (e.g., Growns et al. 1997; Gerecke 2002; Smith et al. 2010). Future studies could compare the method proposed here to those proposed previously.
Conclusions
This method has proven effective in its ability to collect water mites from lotic, riffle-run environments. However, we acknowledge this method has its limitations. We encourage future studies to address these limitations and continue to refine this method. For example, future studies may address whether the proposed number of collection or rinse efforts is sufficient, or if more or less provide similar or improved results. We look forward to the continued advancement of this field of study.
Acknowledgements
First and foremost, we thank Francesca Ferguson, Ph.D. student in Ecology at Penn State, for her assistance with sampling efforts and her expert help sewing together our custom 250 μm mesh sampling net. In addition, we thank the Huntingdon County Conservation District for their technical assistance with landowner outreach and coordinating access to sample sites, Tim Charles Sr. for his assistance with field collection efforts, and J. Ray Fisher for his assistance in identifying water mites. This work was supported by the Sara Chinn Kalser Faculty Research Endowment through the Pennsylvania State University.
References
- Australian Government. 1994. Australian River Assessment System (AUSRIVAS). Department of Sustainability, Environment, Water, Populations and Communities. https://ausrivas.ewater.org.au/index.php/taxonomy

- Balian E.V., Segers H., Lévèque C., Martens K. 2008. The freshwater animal diversity assessment: an overview of the results. Hydrobiologia, 595: 627-637. https://doi.org/10.1007/s10750-007-9246-3
- Chalfant B. 2018. Wadable riffle-run stream macroinvertebrate data collection protocol. Shull D. (Chapter Ed). In: Shull D., Pulket M. (Eds), Office of Water Programs, Bureau of Clean Water, Water quality monitoring protocols for rivers and streams. 2-2-2-24.
- Di Sabatino A., Gerecke R., Martin P. 2000. The biology and ecology of lotic water mites (Hydrachnidia). Freshwater Biology, 44: 47-62. https://doi.org/10.1046/j.1365-2427.2000.00591.x
- Di Sabatino A., Smit H., Gerecke R., Goldschmidt T., Matsumoto N., Cicolani B. 2008. Global diversity of water mites (Acari, Hydrachnidia; Arachnida) in freshwater. Hydrobiologia, 595: 303-315. https://doi.org/10.1007/s10750-007-9025-1
- Di Sabatino A., Boggero A., Miccoli F.P., Cicolani B. 2004. Diversity, distribution and ecology of water mites (Acari: Hydrachnidia and Halacaridae) in high Alpine lakes (Central Alps, Italy). Experimental and Applied Acarology, 34: 199-210. https://doi.org/10.1007/978-94-017-0429-8_13
- Environment and Climate Change Canada. 2020. CABIN Laboratory Methods: processing, taxonomy, and quality control of benthic macroinvertebrate samples. Environment and Climate Change Canada, Gatineau, Quebec. https://publications.gc.ca/collections/collection_2021/eccc/En84-86-2021-eng.pdf

- Fisher J.R., Fisher D.M., Nelson W.A., O'Neill J.C., Skvarla M.J., Ochoa R., Bauchan G.R., Radwell A.J., Dowling A.P.G. 2015. Torrenticola trimaculata n. sp. (Parasitengona: Torrenticolidae), a three-spotted water mite from eastern North America: taxonomic history, species delimitation, and survey of external morphology. Acarologia, 55: 71-116. https://doi.org/10.1051/acarologia/20152155
- Fisher J.R. 2016. Integrative taxonomy of North American torrent mites (Parasitengona: Torrenticolidae: Torrenticola). Ph.D. dissertation, University of Arkansas.
- Gerecke R., Schwoerbel J. 1992. Water quality and water mites in the upper Danube region. 1959-1984. In: Dusbábek F., Bukva F. (Eds), Modern Acarology, Volume 1 (8th International Congress of Acarology, Budejovice): Academia, Prague, and SPB Academic Publishing, The Hague, 483-491 pp.
- Gerecke R. 2002. The water mites (Acari, Hydrachnidia) of a little disturbed forest stream in southwest Germany - a study on seasonality and habitat preference, with remarks on diversity patterns in different geographical areas. In: Bernini F., Nannelli G., Nuzzaci G., de Lillo E. (Eds), Acarid Phylogeny and Evolution. Adaptations in mites and ticks. : Kluwer Academic Publishers, 69-89 pp. https://doi.org/10.1007/978-94-017-0611-7_9
- Goldschmidt T., Helson J.E., Williams D.D. 2016. Ecology of water mites in Panama - first data on water mites (Acari, Hydrachnidia) as bioindicators in the assessment of biological integrity of neotropical streams. Limnologica, 59: 63-77. https://doi.org/10.1016/j.limno.2016.03.007
- Goldschmidt T. 2016. Water mites (Acari, Hydrachnidia): a powerful but widely neglected bioindicator - a review. Neotropical Biodiversity, 2: 12-25. https://doi.org/10.1080/23766808.2016.1144359
- Growns J.E., Chessman B.C., Jackson J.E., Ross D.G. 1997. Rapid assessment of Australian rivers using macroinvertebrates: cost and efficiency of 6 methods of sample processing. Journal of North American Benthological Society, 16: 682-693. https://doi.org/10.2307/1468153
- Growns J.E. 2001. Aquatic mites as bioindicators, with an Australian example. In: Halliday R.B., Walter D.E., Proctor H.C., Norton R.A., Colloff M.J. (Eds), Acarology: Proceedings of the 10<sup>th</sup> International Congress; Melbourne: CSIRO Publishing. 136-142 pp.
- Lombardo P., Miccoli F.P., Cicolani B. 2013. Indicator value of lotic water mites (Acari: Hydrachnidia) and their use in macroinvertebrate-based indices for water quality assessment purposes. Knowledge and Management of Aquatic Ecosystems, 411: 1-28. https://doi.org/10.1051/kmae/2013075
- Matsumoto N., Dick M.H., Mawatari S.F. 2005. Water mites of the genus Hygrobates Koch, 1837 (Acari: Parasitengona: Hygrobatidae) from Hokkaido, northern Japan. Journal of Natural History, 39: 1893-1945. https://doi.org/10.1080/00222930400023727
- Pennsylvania Department of Environmental Protection (PADEP). 2022. Draft 2022 Pennsylvania integrated water quality monitoring and assessment report. Pennsylvania Department of Environmental Protection, Bureau of Clean Water, Harrisburg, Pennsylvania. https://storymaps.arcgis.com/stories/b9746eec807f48d99decd3a583eede12

- Radwell A.J., Dowling A.P.G., Smith I.M., Kaliki V. 2011. Kongsbergia robisoni, n. sp. (Acari: Hydrachnidiae: Aturidae) from the Interior Highlands of North America based on morphology and molecular genetic analysis. International Journal of Acarology, 37: 194-205. https://doi.org/10.1080/01647954.2010.548404
- Smith I.M., Cook D.R., Smith B.P. 2010. Water mites (Hydrachnidiae) and other arachnids. In: Thorp J., Covich A. (Eds), Ecology and classification of North American freshwater invertebrates. Massachusetts, Cambridge: Academic Press. 485-586 pp. https://doi.org/10.1016/B978-0-12-374855-3.00015-7
- Smit H. 2020. Water mites of the world with keys to the families, subfamilies, genera and subgenera (Acari: Hydrachnidia). Monografieën van de Nederlandse Entomologische Vereniging, no. 12. 774 pp.
- Viets K.O. 1987. Die Milben des Susswassers (Hydrachnellae und Halacaridae [part.]), Acari. 2. Katalog. Sonderbande Naturwissenschaftlichen Vereins Hamburg; 8:1012.
- Voshell J.R. 2002. A guide to common freshwater invertebrates of North America. Newark: The McDonald and Woodward Publishing Company. 442 pp.



2022-03-13
Date accepted:
2022-05-30
Date published:
2022-06-09
Edited by:
Mąkol, Joanna

This work is licensed under a Creative Commons Attribution 4.0 International License
2022 Stenger, Logan R. and Skvarla, Michael J.
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



