Abundance and distribution of Aculus schlechtendali on apple orchards in Southern of Brazil
Moreira do Nascimento, Joseane1 ; Evangelho Silva , Darliane2 ; Pavan, Aline Marjana3 ; Liberato Costa Corrêa, Luiz4 ; Schussler, Matheus5 ; Johann, Liana6 and Juarez Ferla, Noeli7
1✉ Laboratório de Acarologia, Tecnovates, Universidade do Vale do Taquari – Univates, Rua Avelino Talini, 171, 95914-014, Lajeado, RS, Brazil & Programa de Pós-Graduação em Biotecnologia; Universidade do Vale do Taquari – Univates, Rua Avelino Talini, 171, 95914-014, Lajeado, RS, Brazil.
2Laboratório de Acarologia, Tecnovates, Universidade do Vale do Taquari – Univates, Rua Avelino Talini, 171, 95914-014, Lajeado, RS, Brazil.
3Laboratório de Acarologia, Tecnovates, Universidade do Vale do Taquari – Univates, Rua Avelino Talini, 171, 95914-014, Lajeado, RS, Brazil & Programa de Pós-Graduação em Sistemas Ambientais Sustentáveis; Universidade do Vale do Taquari – Univates, Rua Avelino Talini, 171, 95914-014, Lajeado, RS, Brazil.
4Laboratório de Acarologia, Tecnovates, Universidade do Vale do Taquari – Univates, Rua Avelino Talini, 171, 95914-014, Lajeado, RS, Brazil.
5Laboratório de Acarologia, Tecnovates, Universidade do Vale do Taquari – Univates, Rua Avelino Talini, 171, 95914-014, Lajeado, RS, Brazil.
6Laboratório de Acarologia, Tecnovates, Universidade do Vale do Taquari – Univates, Rua Avelino Talini, 171, 95914-014, Lajeado, RS, Brazil & Programa de Pós-Graduação em Sistemas Ambientais Sustentáveis; Universidade do Vale do Taquari – Univates, Rua Avelino Talini, 171, 95914-014, Lajeado, RS, Brazil.
7Laboratório de Acarologia, Tecnovates, Universidade do Vale do Taquari – Univates, Rua Avelino Talini, 171, 95914-014, Lajeado, RS, Brazil & Programa de Pós-Graduação em Biotecnologia; Universidade do Vale do Taquari – Univates, Rua Avelino Talini, 171, 95914-014, Lajeado, RS, Brazil & Programa de Pós-Graduação em Sistemas Ambientais Sustentáveis; Universidade do Vale do Taquari – Univates, Rua Avelino Talini, 171, 95914-014, Lajeado, RS, Brazil & CNPq Researcher.
2020 - Volume: 60 Issue: 4 pages: 659-667
https://doi.org/10.24349/acarologia/20204394Original research
Keywords
Abstract
Introduction
The presence of pathogens and organisms considered as invasive pests has been a major threat to food security worldwide, with losses in quality and reduction in production yield estimated at 30-40% (Fisher et al. 2012; Flood 2010; Strange and Scott 2005). The analysis of the propagation of these organisms in agriculture must consider where they are found, where they occur and how they are spreading (Savary et al. 2019).
Dispersal of insects and mites through international and regional borders can be favored by the increase in circulation of plant materials. There's a scarcity of information about the loss in revenue suffered by the affected countries (Bebber et al. 2014; Sileshi et al. 2019). Phytophagous mites, specially eriophyoids, have a greater potential as adventive mite species (AMS) (Navia et al. 2010), which include many exotic species that can cause economic and ecological problems around the world (Wheeler and Hoebeke 2009).
These mites can become invasive, as they are hard to be detected, easily adapting to new host plants, have favorable reproductive characteristics and disperse by wind (Navia et al. 2007). They can also survive in adverse conditions during the dispersion, like solar radiation, high water transpiration, low temperatures/concentrations of oxygen and the absence of food (Valenzano et al. 2019). Two species of phytophagous mites, previously considered quarantine pests, were recently introduced in Brazil: Raoiella indica Hirst (Tenuipalpidae, Red Palm Mite - RPM) (Navia et al. 2010) and Aculus schlechtendali (Nalepa) (Eriophyidae, Apple Rust Mite - ARM) (Ferla et al. 2018).
Aculus schlechtendali, considered an important apple pest in many countries, feeds in flowers, fruits and leaves, using a stylet that penetrates the plant's epidermal cells (Duso et al. 2010; Krantz and Lindquist 1979). Light-colored spots appear in the infested leaves and end up merging, causing a rusty aspect in the attacked spots (Danelski et al. 2015; Hoy 2011). When the population reaches more than 50 mites/cm2, it can reduce photosynthesis, therefore setting the control risk threshold for this species at 10-40 individuals/cm2 of leaf surface (Hoy 2011). The infested leaves can wither and fall with severe mite damage, which can also inhibit plant growth and causing brown spots in the fruits' peel, known as 'apple russeting' (Alford 2014; Danelski et al. 2015), affecting the physiological activities of plants and the aesthetical quality of the fruits (Easterbrook 1996; Hoyt 1969; Spieser et al. 1998; Walde et al. 1997).
Among the reasons for the success of apple culture in Brazil, centered in the highest regions of the three Southern states, are good environmental conditions for fruit production with high acceptance by consumers and the constant search for better production alternatives (Kist 2019a). The 2016/2017 crop year registered an apple production record in Southern Brazil, with the highest volume being produced in Santa Catarina (SC), followed by Rio Grande do Sul (RS) and Paraná (PR) (Kist 2019b). According to the data provided by Epagri (Empresa de Pesquisa Agropecuária e Extensão Rural de Santa Catarina), the Gala cultivar represents 56% of production in the last five years in Brazil, followed by Fuji, with 39%, and other cultivars with 6% (Assmann 2019). The cultivar Eva, the product of the cross between the cultivars Anna and Gala (Hauagge and Tsuneta 1999), is within 6% of the production of other cultivars (IAPAR 1999).
Considering that ARM is a recently reported species in Brazil, with an unknown population abundance and distribution, we hypothesize that ARM is present in the three Southern Brazil states, and its abundance varies amongst the different cultivars. The aim of this exploratory analysis was to carry out a survey of abundance and distribution of ARM in the three apple producing states in Southern Brazil, within cultivars produced in these regions. Additionally, we checked for spatial autocorrelation (dependence) between the sampling points, to determine if there is an ARM infestation hotspot.
Materials and methods
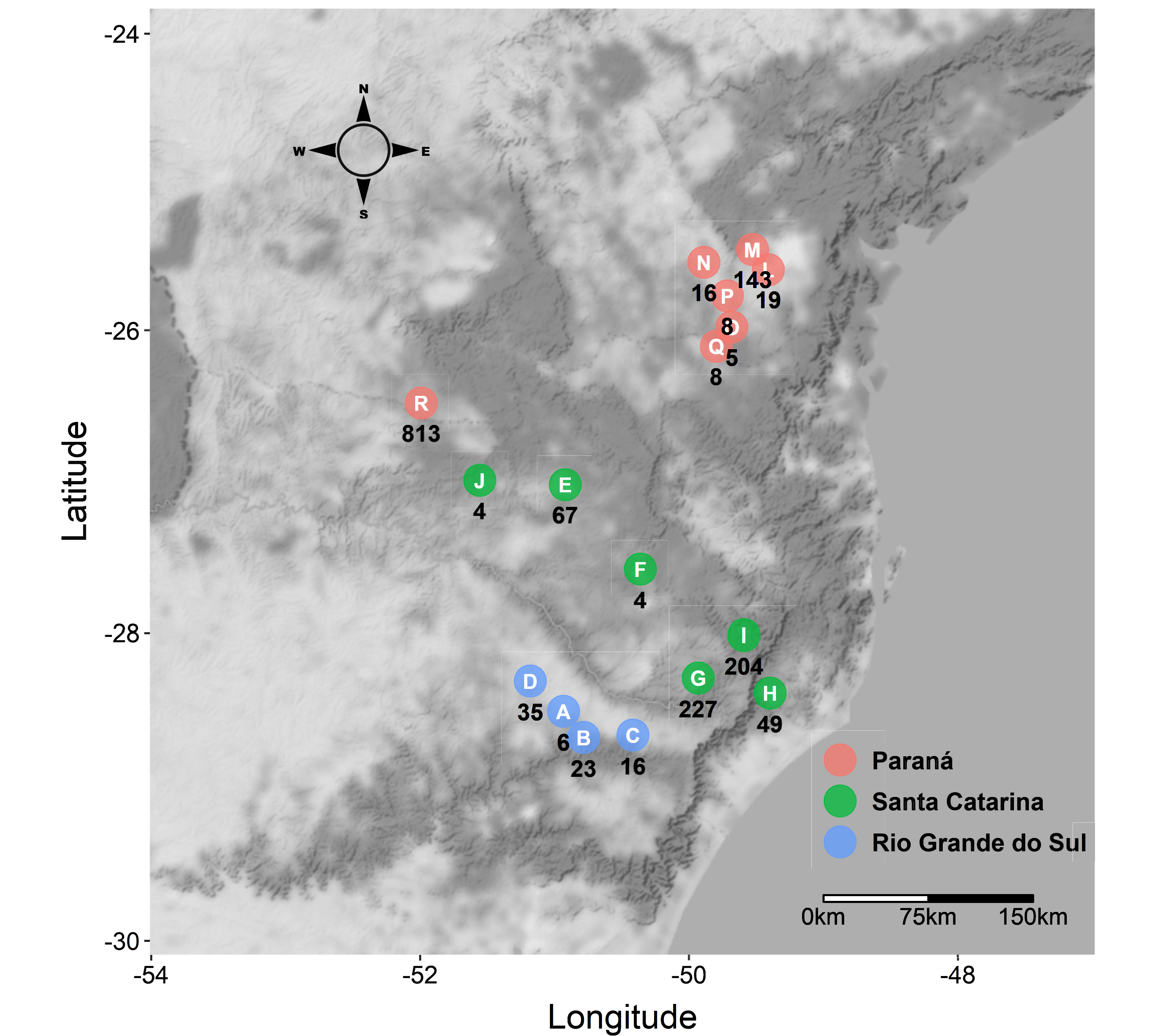
The study was carried out in the three apple producing states in Southern Brazil, Rio Grande do Sul (RS), Santa Catarina (SC) and Paraná (PR). A total of 94 orchards were sampled in 19 municipalities. Specifically, 27 orchards were located in four municipalities in RS, 39 in eight municipalities in SC, and 28 in seven municipalities in PR (Figure 1). Regarding cultivars, there were 40 orchards of Fuji cultivar, 43 of Gala and 11 of Eva (Table S1 – Supplementary material).



At each orchard, 20 plants were sampled once, in March 2019 at harvest point, with four leaves collected from each one, totaling 80 leaves/orchard. The first sampled plant was the sixth plant of the 6th row counted from the orchard edge, with an interval of two plants between them, totaling four plants/row in five selected rows.
The leaves were packaged in polyethylene bags, properly labeled and stored under low temperatures with reusable rigid ice (Gelox®), until they were sent to the Laboratório de Acarologia of the Universidade do Vale do Taquari – Univates, in Lajeado (RS), and screening was performed under a stereomicroscope (40x). The ARM specimens found were mounted on microscope slides using modified Berlese's medium (Amrine and Manson 1996) and kept at 50-60°C, to clarify the specimens and dry the medium. Mites were identified using optical microscope with phase contrast and the identification were made according to Amrine and Manson (1996), Baker et al. (1996) and de Lillo et al. (2010).
Data analysis
Average number of ARM per states and apple cultivars
It was compared if the average number of ARM would be different between the three states (Rio Grande do Sul, Santa Catarina and Paraná), as well as between the three apple cultivars (Eva, Fuji and Gala). Therefore, two generalized linear mixed models (GLMMs) with Poisson or negative binomial distribution were fitted. Both GLMMs had the average number of ARM as a response variable, while the states and the apple cultivars were included as fixed factors (predictor variable). Additionally, the municipalities and sampling places were considered as random effects. The GLMMs were carried out using the `glmer' and `glmer.nb' functions from the lme4 package (Bates et al. 2015). After that, the best-fitted LMM was chosen according to the smallest score of the Akaike Information Criterion (AIC) that was computed and compared using the `AICctab' function from the bbmle package (Bolker and R Core Team 2016).
After choosing GLMMs with Poisson distribution, multiple comparisons were made among the three states and the three apple cultivars using the `glht' function, with differences tested with Tukey (Holm = p-value adjustment method) in multcomp package (Hothorn et al. 2008). All analyses were performed in R (Ihaka and Gentleman 1996; R Core Team 2018).
Analysis of spatial autocorrelation (Moran's I)
Since our variable of interest (number of ARM) could be correlated with geographic distances, for example, higher values of mites if nearer other sampling locations and small values if far from it, a spatial autocorrelation analysis was performed (Global Moran's I). Such analysis evaluates whether the pattern expressed in our data is clustered, dispersed or random. Thus, it was evaluated how related would be the values of the number of ARM based on the locations where they were sampled.
Moran's I was calculated after generating a matrix of inverse distance weights (latitude, longitude). On this matrix, entries for pairs of geographic coordinates that are close together are higher than for pairs of geographic coordinates that are far apart. Then, we tested if the quantity of ARM was positively (clustered) or negatively (dispersed) spatially autocorrelated. In other words, it was tested whether municipalities with similar values should be closer to each other or if high values and low values in our data would be more spatially dispersed than random. To estimate the Moran's I, the `Moran.I' function was used in the ape library (Paradis et al. 2004). Furthermore, the geographic distance matrix (straight-line distance) among pairs of municipalities sampled was calculated using the `GeoDistanceInMetresMatrix' function for R (https://eurekastatistics.com/calculating-a23distance-matrix-for-geographic-points-using-r/ ![]() ).
).
Results
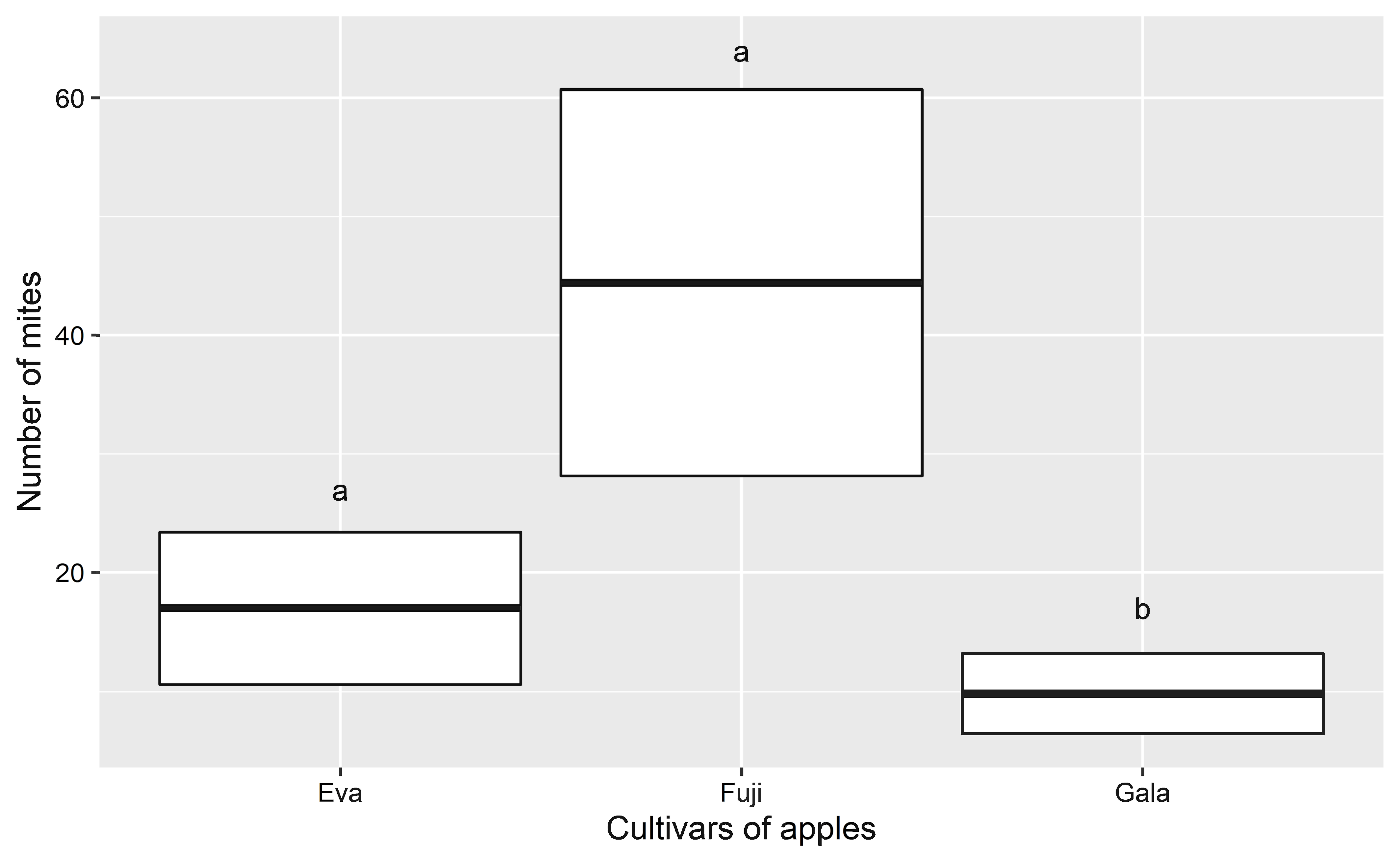
A total of 1,647 specimens of ARM were found in 66 of the 94 orchards evaluated (70%) and in 17 of the 19 municipalities sampled (89%). The average number (mean ± standard deviation) of mites/orchard was of 24 ± 55 in general. Cultivar specific means were 44 ± 83 in Fuji cultivar, 10 ± 19 in Gala and 17 ± 21 in Eva (Figure 2).


In the state of RS, the average number of mites/orchard was of 5 ± 7, with 7 ± 9 in Fuji (51%) and 4 ± 7 in Gala (49%). ARM was found in all orchards evaluated in this state, relatively larger abundance was observed in Muitos Capões and smaller in Vacaria. In the state of SC, the average number of mites was of 20 ± 39, with 25 ± 50 in Fuji (64%) and 14 ± 26 in Gala (36%). Larger abundance was observed in São Joaquim and Ubirici, and smaller in Água Doce and Correia Pinto. No mites were found in orchards located in Lages or Painel. In the state of PR, a greater abundance of ARM was observed, with averages of 40 ± 79 mites/orchard, with 127 ± 131 in Fuji (75%), 8 ± 11 in Gala (6%) and 17 ± 21 in Eva (19%) (Table S2 – Supplementary material). In all orchards evaluated in this state, the presence of ARM was verified.
ARM populations were smaller in Gala cultivar (Figure 2) (GLMM Poisson, χ2 = 9,02, p = 0,01) and in the state of Rio Grande do Sul (Figure 3) (GLMM Poisson, χ2 = 7,46, p = 0,02) (Table S3 – Supplementary material).



Regarding the spatial scale, we sampled at municipalities as near as 18,219 Km (Campo do Tenente (PR) – Rio Negro (PR)) and as far as 378,330 Km (Monte Alegre dos Campos (RS) – Campo Largo (PR)), (Table S4 – Supplementary material) and (Figure 4). On the other hand, our analysis of spatial autocorrelation demonstrated that the Moran's I index was -0.04, while the expected index should be -0.06 (± 0.04 standard deviation). As a result, we did not find evidence that the number of ARM is spatially autocorrelated (p-value = 0.68, Figure 5). Therefore, there is no pattern related to the geographic distribution of ARM along their infestation within apple orchards in Southern Brazil, as well as there was no hotspot identified at this time.





Discussion
The first report of ARM in Brazil occurred in only one isolated point in the state of Rio Grande do Sul (Ferla et al. 2018) and the situation of orchards located in the states of SC and PR was unknown. Our results indicate the presence of ARM in all the producing regions of Southern Brazil, although not in all orchards. These results suggest that this mite can reach other places where apple production occurs. However, so far, there are no reports that this species has reached pest status in the regions or orchards evaluated. We strongly suggest monitoring the infestation and any possible damages, as well as identifying the associated natural enemies, which can carry out population control.
Factors such as cultivars, climatic factors (Danelski et al. 2015) and location of the orchard (Denizhan 2011), as well as occurrence of natural enemies (Maula and Khan 2015), may influence ARM populations. Differences among the investigated states were found, with a larger abundance of mites present in Santa Catarina and Paraná and a smaller in Rio Grande do Sul, even though the latter is the second largest producing state in volume of apples produced in Brazil. However, we suggest complementary studies in the evaluated states, in other phenological stages of apple trees, to expand the evaluation performed.
The introduction of this mite in Brazil may have occurred across borders, or due to the intense crossing of fruits from Argentina and Chile to Brazil. However, the possibility of this species being associated with traffic of vegetal material can't be excluded, despite the controls established in the Brazilian import system (Ferla et al. 2018). Besides that, long distance dispersal through air is an important survival strategy to many organisms, allowing them to swiftly colonize new territories or migrate between habitats (Gage et al. 1999). Some eriophyid species can travel short distances within the atmosphere layer close to the ground, while others can travel longer distances, being transported by air currents, reaching cloud stratification of where they are protected from drying or direct light action (Valenzano et al. 2019).
There are examples of inadvertent introductions of eriophyid mites in new areas, where they found appropriate conditions to thrive in the absence of efficient natural enemies, resulting in marked damage to infested crops and consequent serious socioeconomic problems (Navia et al. 2010). Considering the example of occurrence and dispersal of Raoiella indica, the large distance between previously infested areas by RPM in Brazil (North and Southeast) and from this to the Northeast suggests that the propagation of RPM solely by natural dispersal is improbable, and that this mite has reached new regions by transporting host plants or infested vegetal material (Melo et al. 2018).
Aculus schlechtendali was present in all the evaluated cultivars, with larger populations in the Fuji and Eva, and smaller in Gala, despite being the most representative in Brazilian apple production. Duso et al. (2003) also related differences in ARM abundance among the studied cultivars, being larger in Florina and smaller in N.Y. 18491. The knowledge of the difference among cultivars being important to establish the damage threshold and the correct moment for the application of control measures (Angeli et al. 2007).
Our hypotheses were confirmed, as there was difference in abundance amongst the cultivars and due to the presence of this mite in the three Southern states. These results may be influenced by the favorable climate for apple cultivation, although no spatial autocorrelation between the sampled locations was found, considering that the distribution in these locations was related not only to geographical proximity, but also to other factors not evaluated in the present study. Also, there was no hotspot identified, confirming that the species is distributed throughout the region.
The presence of this species in the region comes as a warning to the apple production industry, in regards to the occurrence of a species formerly considered of quarantine status. Knowledge about the abundance, places of occurrence of these mites and the cultivars affected are important for the monitoring of the species and future management measures. Additionally, continued monitoring will help in the understanding of the distribution pattern and contribute to the comprehension of how the dispersal of this mite may have occurred in the evaluated places.
Acknowledgements
The first author thanks to Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brasil (CAPES) - Finance Code 001 for the doctoral scholarship. The authors are grateful to Ministério da Agricultura, Pecuária e Abastecimento (MAPA) and Embrapa - Estação Experimental de Fruticultura de Clima Temperado - Vacaria (RS) for lending assistance with samplings, and to the Universidade do Vale do Taquari – Univates for providing all necessary material during the study. We would also like to thank Charles Fernando dos Santos for assistance with the statistical analysis, Guilherme Garcia de Oliveira for preparing the map and Janaína Horn for helping with language revisions. Dr. Noeli Juarez Ferla is supported by CNPq productivity research scholarship (310035/2017-1).
Disclosure statement
The authors declare that they have no conflict of interest.
acarologia_4394_Supplementary-Table-S1.pdf
acarologia_4394_Supplementary-Table-S2.pdf
acarologia_4394_Supplementary-Table-S3.pdf
acarologia_4394_Supplementary-Table-S4.pdf
References
Alford D.V. 2014. Pests of fruit crops: A colour handbook. CRC press. Burlington, Massachusetts. USA. pp. 461.
Amrine J.W. Jr., Manson D.C.M. 1996. Preparation, mounting and descriptive study of eriophyoid mites. In: Lindquist, E. E.; Sabelis, M. W.; Bruin, J. (Eds.). World crop pests: Eriophyoid mites: their biology, natural enemies and control. Amsterdam: Elsevier, pp. 383-396. doi:10.1016/S1572-4379(96)80023-6
Angeli G., Rizzi C., Dorigoni A., Ioriatti C. 2007. Population injury levels of the apple rust mite Aculus schlechtendali (Nal.) on Golden Delicious and Red Delicious apple fruits. IOBC WPRS Bulletin, 30(4): 255-260.
Assmann, I. 2019. Atração à primeira vista. Anuário Brasileiro da Maçã. Editora Gazeta, Santa Cruz do Sul. pp.15.
Baker E. W., Kono T., Amrine Jr, J. W., Delfinado-Baker M., Stasny T. A. 1996. Eriophyoid mites of the United States. West Bloomfield, Indira Publishing House, p. 394.
Bates D., Maechler M., Bolker B., Walker S. 2015. lme4: Linear mixed-effects models using Eigen and S4. doi:10.18637/jss.v067.i01
Bebber D.P., Holmes T., Smith D., Gurr S.J. 2014. Economic and physical determinants of the global distributions of crop pests and pathogens. New Phytologist, 202(3): 901-910. doi:10.1111/nph.12722
Bolker B., R Core Team. 2016. Tools for general maximum likelihood estimation.
Danelski W., Badowska-Czubik T., Rozpara B. 2015. Occurrence of the apple rust mite Aculus schlechtendali (Nal.) in organic cultivation of apple. Journal of Research and Applications in Agricultural Engineering, 60(3): 25-27.
de Lillo E., Craemer C., Amrine J. W. Jr., Nuzzaci G. 2010. Recommended procedures and techniques for morphological studies of Eriophyoidea (Acari: Prostigmata). Experimental and Applied Acarology, 51(1-3): 283-307. doi:10.1007/s10493-009-9311-x
Denizhan E. 2011. Population development of Aculus schlechtendali (Nalepa) (Acarina: Eriophyidae) feeding on different apple varieties in Van province and effect of Zetzellia mali (Ewing) (Turkey). Bitki Koruma Bülteni, 51(3): 239-253.
Duso C., Castagnoli M., Simoni S., Angeli G. 2010. The impact of eriophyoids on crops: recent issues on Aculus schlechtendali, Calepitrimerus vitis and Aculops lycopersici. Experimental and Applied Acarology, (51): 151-168. doi:10.1007/s10493-009-9300-0
Duso C., Pasini M., Pellegrini M. 2003. Distribution of the predatory mite Typhlodromus pyri (Acari: Phytoseiidae) on different apple cultivars. Biocontrol Science and Technology, 13(7): 671-681. doi:10.1080/09583150310001606264
Easterbrook M.A. 1996. Damage and control of eriophyoid mites in apple and pear. In: Lindquist E.E., Sabelis M.W., Bruin J. (eds): Eriophyoid mites: their biology, natural enemies and control. Elsevier, The Netherlands, pp. 527-541. doi:10.1016/S1572-4379(96)80033-9
Ferla N.J., Silva D.E., Navia D., Do Nascimento J.M., Johann L., de Lillo E. 2018. Occurrence of the quarantine mite pest Aculus schlechtendali (Acari: Eriophyidae) in apple orchards of Serra Gaúcha, Rio Grande do Sul state, Brazil. Systematic and Applied Acarology, 23(6): 1190-1198. doi:10.11158/saa.23.6.14
Fisher M.C., Henk D.A., Briggs C.J., Brownstein J.S., Madoff L.C., McCraw S.L., Gurr S. J. 2012. Emerging fungal threats to animal, plant and ecosystem health. Nature, 484(7393): 186-194. doi:10.1038/nature10947
Flood J. 2010. The importance of plant health to food security. Food Security, 2(3), 215-231. doi:10.1007/s12571-010-0072-5
Gage S.H., Isard S.A., Colunga-G M. 1999. Ecological scaling of aerobiological dispersal processes. Agricultural and Forest Meteorology, 97(4): 249-261. doi:10.1016/S0168-1923(99)00070-2
Hauagge R., Tsuneta M. 1999. " IAPAR 75-Eva"," IAPAR 76-Anabela" e" IAPAR 77-Carícia"-Novas cultivares de macieira com baixa necessidade em frio. Revista Brasileira de Fruticultura, 21(3): 239-242.
Hothorn T., Bretz F., Westfall P. 2008. Simultaneous inference in general parametric models. Biometrical Journal: Journal of Mathematical Methods in Biosciences, 50(3): 346-363. doi:10.1002/bimj.200810425
Hoy M.A. 2011. Agricultural acarology: introduction to integrated mite management (Vol. 7). CRC press.
Hoyt S.C. 1969. Population studies on five mite species on apple in Washington. In: G.O. Evans (Editor), Proceedings of the 2nd International Congress of Acarology. Akadémia I Kiadó, Budapest, Hungary, pp. 117-133.
IAPAR - Instituto Agronômico do Paraná - Available from: http://www.iapar.br/pagina-1990.html
 . [16/03/2020].
. [16/03/2020].Ihaka R., Gentleman R. 1996. R: a language for data analysis and graphics. Journal of Computational and Graphical Statistics, 5(3): 299-314. doi:10.1080/10618600.1996.10474713
Kist, B.B. 2019a. O desafio de renovar. Anuário Brasileiro da Maçã. Editora Gazeta, Santa Cruz Sul, p. 20-21.
Kist, B.B. 2019b. Brilhando na rede. Anuário Brasileiro da Maçã. Editora Gazeta, Santa Cruz Sul, p. 25.
Krantz G.W., Lindquist E.E. 1979. Evolution of phytophagous mites (Acari). Annual Review of Entomology, 24(1): 121-158. doi:10.1146/annurev.en.24.010179.001005
Maula F., Khan I.A. 2015. Temporal response of different apple cultivars to different pest mite complex and their natural enemies in Swat valley of KPK province. Pakistan Entomology, 37(2): 155-161.
Melo J.W., Navia D., Mendes J.A., Filgueiras R.M., Teodoro A.V., Ferreira J.M., Guzzo E.C., Souza I.V. de, Mendonça R.S. de, Calvet E.C., Paz Neto A.A. Gondim Jr. M.G.C., Morais E.G.F. de, Godoy M.S., Santos J.R. dos, Silva R.I.R, Silva V.B. da, Norte R. F., Oliva A.B., Santos R.D.P. dos, Domingos C.A. 2018. The invasive red palm mite, Raoiella indica Hirst (Acari: Tenuipalpidae), in Brazil: range extension and arrival into the most threatened area, the Northeast Region. International Journal of Acarology, 44(4-5): 146-149. doi:10.1080/01647954.2018.1474945
Navia D., Moraes G.J. de, Flechtmann C.H.W. 2007. Phytophagous mites as invasive alien species: quarantine procedures. In: Morales-Malacara JB, Behan-Pelletier, V., Ueckermann, E., Perez, T.M., Estrada, E., Gispert, C., Badii, M. (eds) Acarology: Proceedings of the XI international congress. Merida, México, pp. 307-316.
Navia D., Ochoa R., Welbourn C., Ferragut F. 2010. Adventive eriophyoid mites: a global review of their impact, pathways, prevention and challenges. Experimental and Applied Acarology, 51(1-3): 225-255. doi:10.1007/s10493-009-9327-2
Paradis E., Claude J., Strimmer K. 2004. APE: analyses of phylogenetics and evolution in R language. Bioinformatics 20: 289-290. doi:10.1093/bioinformatics/btg412
R Core Team. 2018. R: A language and environment for statistical computing. The R Foundation for Statistical Computing, Vienna, Austria
Savary S., Willocquet L., Pethybridge S.J., Esker P., McRoberts N., Nelson A. 2019. The global burden of pathogens and pests on major food crops. Nature ecology & evolution, 3(3): 430-439. doi:10.1038/s41559-018-0793-y
Sileshi G.W., Gebeyehu S., Mafongoya P.L. 2019. The threat of alien invasive insect and mite species to food security in Africa and the need for a continent-wide response. Food Security: 1-13. doi:10.1007/s12571-019-00930-1
Spieser F., Graf B., Walther P., Noesberger J. 1998. Impact of apple rust mite (Acari: Eriophyidae) feeding on apple leaf gas exchange and leaf color associated with changes in leaf tissue. Environmental Entomology, 27(5): 1149-1156. doi:10.1093/ee/27.5.1149
Strange R.N., Scott P.R. 2005. Plant disease: a threat to global food security. Annual Review of Phytopathology, 43: 83-116. doi:10.1146/annurev.phyto.43.113004.133839
Valenzano D., Bari G., Valeria M., de Lillo E. 2019. Off-host survival of Eriophyoidea and remarks on their dispersal modes. Experimental and Applied Acarology, 79(1): 21-33. doi:10.1007/s10493-019-00417-w
Walde S.J., Hardman J.M., Magagula C.N. 1997. Direct and indirect species interactions influencing within season dynamics of apple rust mite, Aculus schlechtendali (Acari: Eriophyidae). Experimental and Applied Acarology, 21(9): 587-614. doi:10.1023/A:1018400500688
Wheeler, A.G., Hoebeke, E. R. 2009. Adventive (Non-Native) Insects: Importance to Science and Society. In Insect Biodiversity. p. 475-521. doi:10.1002/9781444308211.ch21



2020-04-13
Date accepted:
2020-09-22
Date published:
2020-09-28
Edited by:
Tsolakis, Haralabos

This work is licensed under a Creative Commons Attribution 4.0 International License
2020 Moreira do Nascimento, Joseane; Evangelho Silva , Darliane; Pavan, Aline Marjana; Liberato Costa Corrêa, Luiz; Schussler, Matheus; Johann, Liana and Juarez Ferla, Noeli
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



