Mideopsis milankovici sp. nov. a new water mite from Montenegro based on morphological and molecular data (Acariformes, Hydrachnidia, Mideopsidae)
Pešić, Vladimir1 and Smit, Harry2
1✉ Department of Biology, University of Montenegro, Cetinjski put b.b., 81000 Podgorica, Montenegro.
2Naturalis Biodiversity Center, P.O. Box 9517, 2300 RA Leiden, the Netherlands.
2020 - Volume: 60 Issue: 3 pages: 566-575
https://doi.org/10.24349/acarologia/20204387ZooBank LSID: 1627D1BA-DBD7-4FFE-820A-AB390A3B5FFE
Original research
Keywords
Abstract
Introduction
Water mites of the genus Mideopsis Neuman, 1880 are known from the Holarctic and Neotropical regions (Cook 1974; Pešić et al. 2013). Recently, the genus was revised by Pešić et al. (2013). The distribution is disjunct, with one group of species with a Holarctic distribution extending with a few species into the Oriental region, and a few species extending into Costa Rica in the New World, and another group limited to South America. According to Pešić et al. (2013) it is likely that the South American taxa which are characterized by extensive setal patches in the male genital field area could represent a distinct genus.
So far, only five species of the genus Mideopsis, i.e., M. crassipes Soar, 1904, M. orbicularis (Müller, 1776), M. roztoczensis Biesiadka & Kowalik, 1979, M. rossicus Tuzovskij, 2002 and M. persicus Pešić & Saboori, 2015 have been reported from the Western Palearctic (Tuzovskij 2002; Pešić and Saboori 2015; Gerecke et al. 2016).
The present study, from a methodological point of view, is based on techniques established in the past decade (e.g., Fisher 2015, Pešić et al. 2019b and bibliography cited therein). Thus, with the exception of Xystonotus willmanni (K. Viets, 1920) (see Blattner et al. 2019) so far molecular methods have not been used in studies of Palaearctic mideopsid mites.
In this paper, Mideopsis milankovici sp. nov. is described from Montenegro. In order to gain insight into the position of the new species in relation to other Mideopsis species from the Palaearctic region, we combined morphological and molecular analyses to infer molecular similarities among the studied species.
Materials and methods
Sampling
Water mites were collected by hand netting, sorted live in the field, and immediately preserved in 96% ethanol. Specimens for molecular analysis were examined without dissecting under a compound microscope in ethanol, using a cavity well slide with a central depression. After DNA extraction, some specimens were dissected and slide mounted in Hoyer's medium. DNA sequences prepared in the course of this study were deposited in Bold with voucher codes and accession numbers indicated in Table 1. The holotype and paratypes of the new species will be deposited in Naturalis Biodiversity Center in Leiden (RMNH).



All measurements are given in µm. The following abbreviations are used: Ac-1 = first acetabulum; Cx-I = first coxae; COI = cytochrome c oxidase subunit I; dL = dorsal length; H = height; I-L-4-6 = fourth-sixth segments of first leg; L = length; mL = medial length; P-1-P-5 = palp segment 1-5; RMNH = Naturalis Biodiversity Center, Leiden; W = width.
Molecular analysis
For the methods used for COI gene amplification and sequencing see Pešić et al. (2017, 2019a). For this study, DNA was extracted from 16 specimens of the genus Mideopsis from Montenegro and The Netherlands (Table 1). Xystonotus willmanni (K. Viets, 1920) from The Netherlands was used as an outgroup.
Phylogenetic analysis
Sequences were aligned by MUSCLE 3.8.425 algorithm as implemented in Geneious Prime 2020.1.1 (Biomatters Ltd.). The neighbor-joining (NJ) and maximum-likelihood (ML) were constructed using the MEGA X software (Kumar et al. 2018). Pairwise distance calculations between nucleotide sequences were computed using Kimura's 2-parameter (K2P) distance model (Kimura 1980) for all codon positions and transition/transversion ratio was calculated using MEGA X (Kumar et al. 2018). The evolutionary history was inferred by using the Maximum Likelihood method based on the Tamura-Nei model (Tamura and Nei 1993). We used ML analyses with 500 bootstrap replicates using GTR+I as the most appropriate model of sequence evolution for our data set based on the Bayesian (BIC) and corrected Akaike Information Criterion (AICc) in the ML model selection feature of MEGA X. The tree is drawn to scale, with branch lengths measured in the number of substitutions per site.
Results and discussion
COI sequences analysis
The analysis involved 18 nucleotide sequences. The nucleotide sequences could be translated into amino acid sequences without any stop codons. The final alignment for the species delimitation using COI sequence data comprised 668 nucleotide positions (nps) for five species including Xystonotus willmanni to root the tree. In the analysis of our COI data, the neighbor-joining (NJ) and maximum-likelihood (ML) result trees gave similar results. Other than minor differences in bootstrap support values, no notable differences were found. The ML result tree is presented in Figure 1.
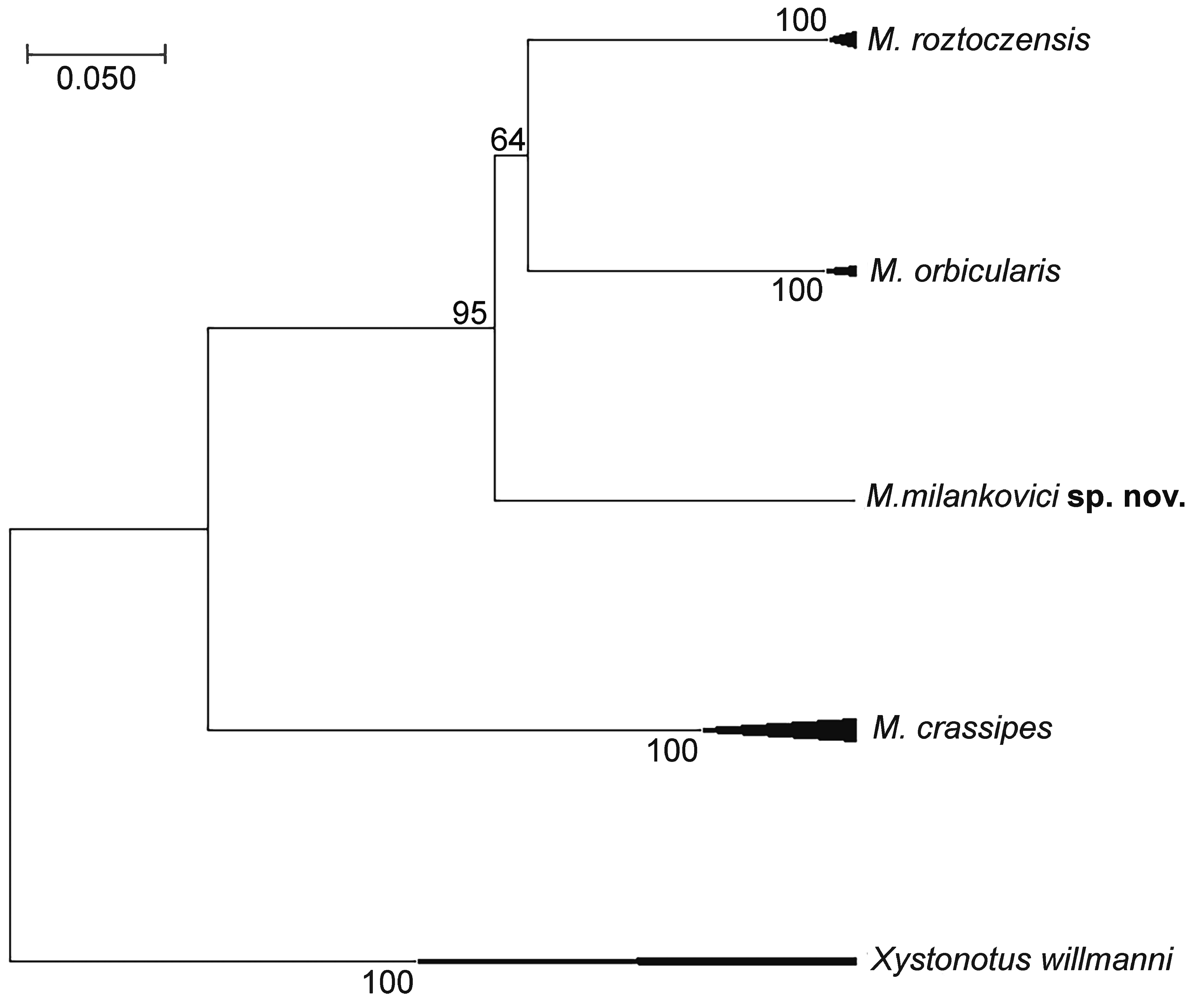


The COI sequence found in the Mideopsis specimen collected in Montenegro was recovered as a sister clade of the clade formed by Mideopsis orbicularis and M. roztoczensis (Figure 1). The genetic distance between the COI sequence of the specimen from Montenegro here assigned to M. milankovici sp. nov. and M. orbicularis was 18.8%. Only one specimen of the new species could be acquired for use in molecular analysis, so intraspecific differences in COI sequence could not be investigated.
The mean genetic distance between congeneric COI sequence groups recovered in the molecular analysis ranged from 18% between M. orbicularis and M. roztoczensis, to 29% between M. orbicularis and M. crassipes (Table 2). The intraspecific distance of M. orbicularis was 1% whereas the other two species, i.e., M. crassipes and M. roztoczensis showed no intraspecific variation.



Systematics
Family Mideopsidae Koenike, 1910
Genus Mideopsis Neuman, 1880
Mideopsis milankovici sp. nov.
ZOOBANK: B619AD87-81F8-4156-B86E-3360053AF3F8 ![]()
Figures 2–3, 4a–b, e–g






Material examined — Holotype ♂ (RMNH), sequenced [22. M19_24_2_E12], dissected and slide mounted, Montenegro, Bar, Međurječka rijeka stream, downstream, between villages of Pečurice and Krute, 42°01'21.21''N, 19°13'11.92''E, 26.vi.2019, leg. Pešić. Paratype: 5♂♂, 17♀♀, Bar, Međurječka rijeka stream, upstream, 42°2'10.80''N, 19°13'4.30''E, 16.vi.2020, leg. Pešić, 1♂, 1♀ dissected and slide mounted (RMNH).
Other material — Montenegro, Bar, Stari Bar old town, Rikavac steam, 42°6'1.72''N, 19°8'33.03''E, 3.vi.2021, leg. Pešić 1♀.
Diagnosis — Dorsal shield flattened, in the centre slightly elevated, V-shaped area formed by anteriorly diverging lines of fine porosity little evident. Ejaculatory complex with well sclerotized anterior keel, anterior ramus wedge-shaped. Postgenital area short (males 116–125 µm, 18% dorsal shield L; females 85 µm, 12% dorsal shield L), excretory pore closer to posterior idiosoma margin (distance 25–35 µm).
Description — Idiosoma rounded; colour dark yellowish to brown. Dorsal shield in the centre slightly elevated, with anteriorly diverging lines of particularly fine porosity forming a V-shaped area sligthly evident (Figures 4a–c). Postgenital area short (about 12–18% dorsal shield L), excretory pore close to posterior idiosoma margin (distance 25–35 µm). Ejaculatory complex with a strongly sclerotized anterior keel, anterior ramus wedge-shaped (Figures 2c–d). Palp: P-1 with a seta in it's proximal part; P-2 ventral margin almost straight; P-3 ventral margin concave, distal margin convex; P-4 ventral projection directed ventrally, with two tips, both flanked by a fine subterminal seta; distal part of P-4 slightly narrower than basal part, with 3 subapical setae, one ventrally, slightly thicker and stiff, and two fine, located dorsally and laterally (Figures 2e and 3b). Male – P-2 L/H ratio 2.5. Female – P-2 more slender than in male, L/H ratio 2.7.
Measurements — Male (Holotype; in parentheses paratype, n = 1) – Dorsal shield L 684 (641), W 606 (575), L/W ratio 1.13 (1.12). Ventral shield L 800 (772), W 800 (747); gnathosomal bay L 149 (141), Cx-III W 416 (378), distance between IV-L insertions 466 (419). Genital field: gonopore L/W 153/50 (156/50), ratio 3.1 (3.1), L Ac-1-3: 44 (41), 44 (45), 44 (41). Distance genital field-excretory pore 77 (75), excretory pore-caudal idiosoma margin 34 (30). Ejaculatory complex L 244 (275). Capitulum vL 130; chelicera: total L (163), claw L (92). Palp: total L 240 (227), dL/H, dL/H ratio: P-1, 28/31, 0.92 (30/30, 1.0); P-2, 67/45, 1.48 (59/42, 1.41); P-3, 31/37, 0.84 (31/34, 0.93); P-4, 73/29, 2.5 (69/28, 2.46); P-5, 41/16, 2.65 (38/16, 2.4); L ratio P-2/P-4 0.93 (0.86). dL of I-L-1-6: 61 (61), 63 (68), 65 (62), 77 (67), 103 (98), 127 (128); I-L-6 H 36 (36); dL/H I-L-6 ratio 3.5 (3.6). dL of IV-L-1-6: 84 (77), 106 (91), 84 (78), 113 (116), 138 (132), 131 (134).
Female (paratype, n = 1) – Dorsal shield L 708, W 625, L/W ratio 1.13. Ventral shield L 750, W 775; gnathosomal bay L 153, Cx-III W 391, distance between IV-L insertions 447. Genital field: gonopore L/W 141/86, ratio 1.6; L Ac-1-3: 47, 47, 38. Distance genital field-excretory pore 44, excretory pore-caudal idiosoma margin 25. Chelicera total L 164, claw L 84. Palp: total L 242, dL/H, dL/H ratio: P-1, 34/30, 1.16; P-2, 59/44, 1.36; P-3, 31/36, 0.88; P-4, 77/28, 2.73; P-5, 41/15, 2.65; L ratio P-2/P-4 0.78. dL of I-L-1-6: 52, 61, 63, 70, 97, 122; dL of IV-L-1-6: 77, 106, 82, 125, 141, 138.
Etymology — Named after Prof Milutin Milanković (1879–1958), the eminent Serbian astrophysicist best known for developing one of the most significant theories relating to earth movements and long-term climate change.
Discussion — The phylogenetic analysis based on COI data reveals that M. milankovici sp. nov. is most similar to M. orbicularis. The high distance between these two species (18.8% K2P) suggests a long independent history of these two species. The relatively high K2P distance seems to be typical for water mites (Blattner et al. 2019) and the obtained data of our study are comparable with the genetic distance between cryptic species of other water mite clades (see Stålstedt et al. 2013, Martin et al. 2010, Pešić et al. 2017, 2019a). Mideopsis orbicularis is widely distributed in the Palaearctic, inhabiting various types of standing waters such as lakes and canals, occasionally also lowland streams (Gerecke et al. 2016), but never recorded from sites with a seasonal flow. From a morphological point of view, M. orbicularis can be separated from the new species only in the male sex, based on the shape of the ejaculatory complex (anterior ramus regularly rounded, anterior keel narrow and weakly sclerotized – see Figure 3c). Mideopsis roztoczensis, a species widely distributed in running waters in Europe, is characterized by a more elevated dorsal shield, a larger postgenital area (140–190 µm), the excretory pore more distanced from the posterior margin of the idiosoma (distance in general 40–90 µm), and the anterior ramus of the male ejaculatory complex (wedge-shaped as in M. milankovici sp. nov.) being wider, with a characteristic arrow-shaped delimited area – see Figure 3d).
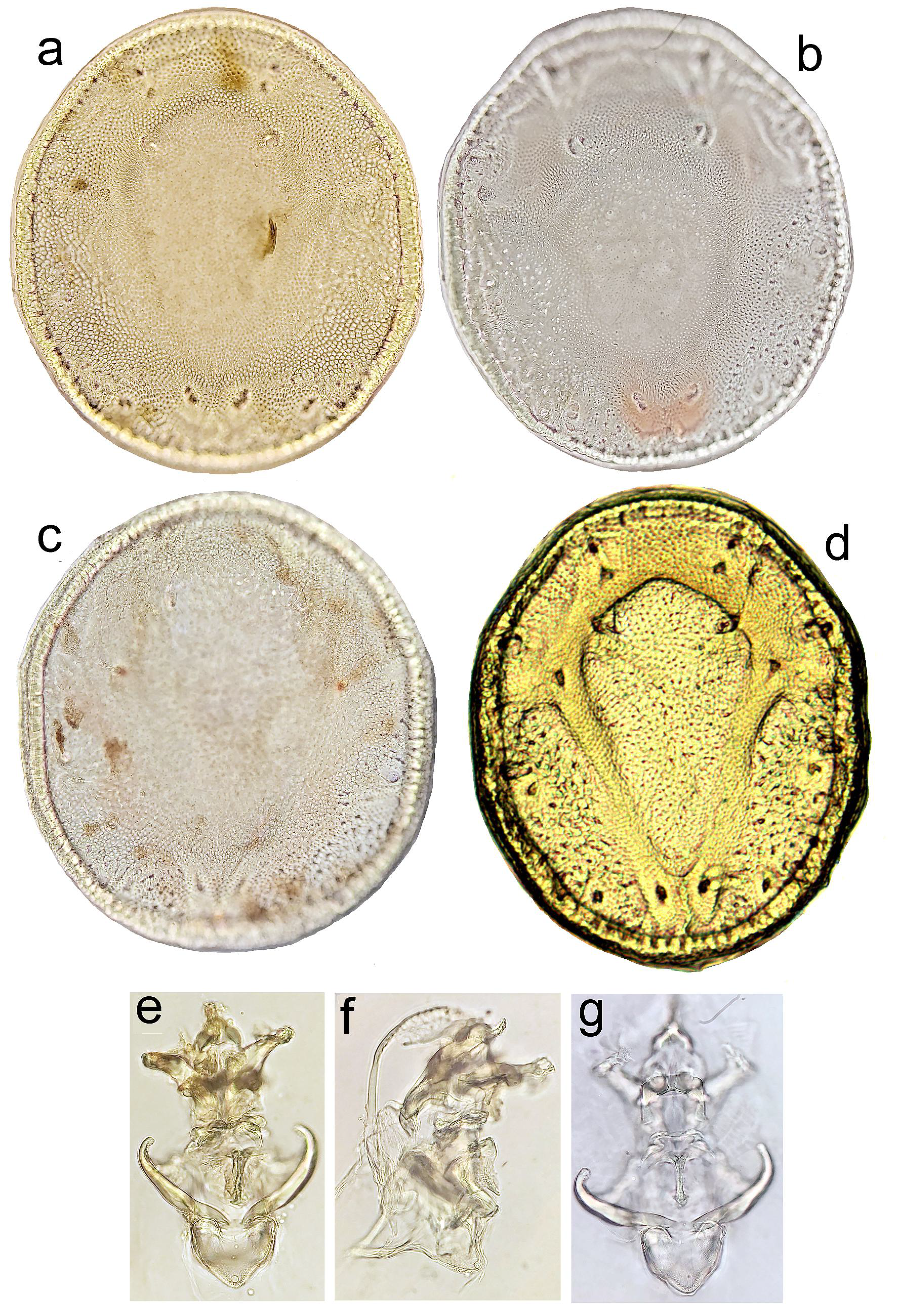


Due to similarity in shape of ejaculatory complexes (see Figure 3e), M. milankovici sp. nov. resembles M. persicus Pešić & Saboori, 2015. The latter species is known from a single male collected in a stream in Fars Province of South Iran (Pešić and Saboori 2015). Mideopsis persicus can be separated by the shape of the dorsal shield with distinct, anteriorly diverging lines of fine porosity forming a well visible V-shaped area. In the new species from Montenengro this V-shaped area is almost indistinguishable (compare Figures 4a–c with Figure 4d).
Mideopsis crassipes, a species widely distributed in the Holarctic (Gerecke et al. 2016) and M. rossicus, a species known from Russia (Tuzovskij 2002), can be separated from all above-mentioned species including M. milankovici sp. nov. in having an egg-shaped idiosoma and a ventral extension of P-4 strongly curved with anteriorly directed tips.
Habitat — Characteristics of sampling sites indicate a preference for intermittent habitats. Both streams in which M. milankovici sp. nov. was collected are located in the narrow coastal region of Montenegro, their middle and lower courses regularly dry up in summer (for an overview of the species and communities that inhabit intermittent rivers in the southern part of Montenegro see Pešić et al. 2020). The upper part of the Međurječka rijeka stream is perennial (Figure 5) but runs dry in its lower reach.



Distribution — Montenegro.
Acknowledgements
Molecular analysis was conducted in the Department of Invertebrate Zoology and Hydrobiology, University of Łódź, Poland. Special thanks to MSc Milica Jovanović and Ana Manović (Department of Biology, Podgorica) for their excellent laboratory work. This study is part of the ''DNA-Eco'' scientific project, supported by a grant of the Montenegrin Ministry of Science.
References
Biesiadka E., Kowalik, W. 1979. A new species of Mideopsis Neuman (Hydrachnellae, Acari) from Poland. Bull. Acad. Pol. Sci., Sér. Sci. Biol., Cl. II, 26: 695-702
Blattner L., Gerecke R., von Fumetti S. 2019. Hidden biodiversity revealed by integrated morphology and genetic species delimitation of spring dwelling water mite species (Acari, Parasitengona: Hydrachnidia). Parasites Vectors, 12: 492. doi:10.1186/s13071-019-3750-y ![]()
Cook, D. R. 1974. Water mite genera and subgenera. Mem. Amer. Ent. Inst., 21: 1-860.
Fisher JR., Fisher DM., Nelson WA., O'Neill JC., Skvarla MJ., Radwell AJ., Ochoa R., Bauchan G., Dowling APG. 2015. An integrative description of Torrenticola trimaculata sp. n. (Parasitengona: Torrenticolidae), a three-spotted, riffle- dwelling mite from eastern North America: morphology, phylogenetics, and taxonomic history of the genus. Acarologia, 55: 71-116. doi:10.1051/acarologia/20152155 ![]()
Gerecke R., Gledhill T., Pešić V., Smit H. 2016. Chelicerata: Acari III. In: Gerecke R., ed. Süßwasserfauna von Mitteleuropa, Bd. 7/2-3. Springer-Verlag Berlin, Heidelberg, pp. 1-429 doi:10.1007/978-3-8274-2689-5 ![]()
Kimura M. 1980. A simple method for estimating evolutionary rate of base substitutions through comparative studies of nucleotide sequences. J. Mol. Evol., 16: 111-120. doi:10.1007/BF01731581 ![]()
Kumar S., Stecher G., Li M., Knyaz C., Tamura K. 2018. MEGA X: Molecular Evolutionary Genetics Analysis across computing platforms. Mol. Biol. Evol., 35: 1547-1549. doi:10.1093/molbev/msy096 ![]()
Martin P., Dabert M., Dabert J. 2010. Molecular evidence for species separation in the water mite Hygrobates nigromaculatus Lebert, 1879 (Acari, Hydrachnidia): evolutionary consequences of the loss of larval parasitism. Aquat. Sci., 72: 347-360. doi:10.1007/s00027-010-0135-x ![]()
Pešić, V., Saboori A. 2015. A new species of the water mite genus Mideopsis Neuman, 1880 from South Iran (Acari, Hydrachnidia). Ecol Monten., 2 (2): 112-116.
Pešić V., Cook D., Gerecke R., Smit H. 2013. The water mite family Mideopsidae (Acari: Hydrachnidia): a contribution to the diversity in the Afrotropical region and taxonomic changes above species level. Zootaxa, 3720: 1-75 doi:10.11646/zootaxa.3720.1.1 ![]()
Pešić V., Asadi M., Cimpean M., Dabert M., Esen Y., Gerecke R., Martin P., Savić A., Smit H., Stur E. 2017. Six species in one: Evidence of cryptic speciation in the Hygrobates fluviatilis complex (Acariformes, Hydrachnidia, Hygrobatidae). Syst. Appl. Acarol., 22: 1327-1377. doi:10.11158/saa.22.9.4 ![]()
Pešić V., Broda Ł., Dabert M., Gerecke R., Martin P., Smit H. 2019a. Re-established after hundred years: Definition of Hygrobates prosiliens Koenike, 1915, based on molecular and morphological evidence, and redescription of H. longipalpis (Hermann, 1804) (Acariformes, Hydrachnidia, Hygrobatidae). Syst. Appl. Acarol., 24: 1490-1511. doi:10.11158/saa.24.8.10 ![]()
Pešić V., Saboori A., Zawal A., Dabert M. 2019b. Hidden but not enough: DNA barcodes reveal two new species in Hygrobates fluviatilis complex from Iran (Acariformes, Hydrachnidia, Hygrobatidae). Syst. Appl. Acarol., 24: 2439-2459. doi:10.11158/saa.24.12.11 ![]()
Pešić V., Pavićević A., Savić A., Hadžiablahović S. 2020. The intermittent rivers of South Montenegro: ecology and biomonitoring. In: Pešić V., Paunović M., Kostianoy A. eds., The rivers of Montenegro. The handbook of environmental chemistry. Springer, Cham. doi:10.1007/698_2019_415 ![]()
Stålstedt J., Bergsten J., Ronquist, F. 2013. "Forms" of water mites (Acari: Hydrachnidia): intraspecific variation or valid species? Ecol. Evol., 3: 3415-3435. doi:10.1002/ece3.704 ![]()
Tamura K., Nei M. 1993. Estimation of the number of nucleotide substitutions in the control region of mitochondrial DNA in humans and chimpanzees. Mol. Biol. Evol., 10: 512-526.
Tuzovskij P. V. 2002. A new species of water mites of the genus Mideopsis Neuman (Acariformes, Mideopsidae) from Russia. Acarina, 10: 161-165.



2020-06-28
Date accepted:
2020-07-27
Date published:
2020-08-04
Edited by:
Mąkol, Joanna

This work is licensed under a Creative Commons Attribution 4.0 International License
2020 Pešić, Vladimir and Smit, Harry
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



