A new species and new records of Tetranychidae (Acari: Trombidiformes) from Saudi Arabia, with a key to world species of Mixonychus Ryke and Meyer
Khan, Eid Muhammad1 ; Kamran, Muhammad2 and Alatawi, Fahad Jaber3
1Acarology Laboratory, Department of Plant Protection, College of Food and Agriculture Sciences, King Saud University, 11451, P.O. Box 2460, Riyadh, Saudi Arabia.
2Acarology Laboratory, Department of Plant Protection, College of Food and Agriculture Sciences, King Saud University, 11451, P.O. Box 2460, Riyadh, Saudi Arabia.
3✉ Acarology Laboratory, Department of Plant Protection, College of Food and Agriculture Sciences, King Saud University, 11451, P.O. Box 2460, Riyadh, Saudi Arabia.
2019 - Volume: 59 Issue: 4 pages: 492-506
https://doi.org/10.24349/acarologia/20194349ZooBank LSID: 3302B394-5608-4184-8307-D6F66B951196
Original research
Keywords
Abstract
Introduction
Mixonychus Ryke and Meyer and Neotetranychus Trägårdh are small spider mite genera consisting of nineteen and ten species, respectively (Migeon and Dorkeld, 2006−2018). The economic importance of these two genera is not known yet.
Ryke and Meyer (1960) erected the genus Mixonychus based on type species Mixonychus acaciae Ryke and Meyer from South Africa. The genus Mixonychus is morphologically close to Brevinychus Meyer and Evertella Meyer. It can be differentiated from Brevinychus by longer empodial claws as compared to the pads of the true claw, whereas in the later empodial claws are short, and about as long as the pads of true claws. Mixonychus is different from Evertella by having 10 pairs of dorsal body setae as compared to nine pairs setae in latter (Meyer, 1987; Bolland et al., 1998).
Chaudhri (1971) erected the genus Bakerina with the following characters states; the dorsal propodosoma medially with reticulations and hysterosoma with broken and irregular striations. Gutierrez (1985) treated Bakerina as a subgenus of genus Mixonychus and divided Mixonychus into three subgenera on the basis of dorsal integument ornamentation, namely Mixonychus Ryke and Meyer, Bakerina Chaudhri and Tylonychus Miller. Species in the subgenus Mixonychus have a dorsum with protuberances that form a reticulate pattern (four species); species of the subgenus Tylonychus have a striate dorsum with spinules, and lacking protuberances (four species); and species of the subgenus Bakerina have a dorsum with simple striations, lacking protuberances and spinules (eleven species) (Bolland et al., 1998; Migeon and Dorkeld, 2006−2018). Most of the species belonging to the genus Mixonychus have been reported from Asia, Africa, Australia and the Americas. Recently, Mixonychus (B.) murrayae (Gao and Ma), M. (B.) nanchangensis (Ma and Yuan) and M. (B.) ganjuis (Qian, Yuan and Ma), from China, were re-described and illustrated (Yi and Jin, 2017). To date, there is no key for the species of Mixonychus in the world.
The genus Neotetranychus is characterized by having the empodium split distally, long dorsal body setae set on tubercles, cuticle of the female opisthosoma with varying morphology including patterns of strong striae or clusters of stellate or reticulate protuberances, female with two pairs of pseudoanal setae (ps1-2) (Meyer, 1987; Bolland et al., 1998). Not including the new species proposed here, the genus is comprised of ten nominal species, five of them reported from Central or South America (N. peniculus Aranda, N. raphidoseta Aranda, N. granifer Feres and Flechtmann, N. asper Feres and Flechtmann, N. gloriosus Estebanes and Baker), two from Europe (the type species N. rubi Tragardh, and N. rubicola Bagdasarian), one species each from Africa (N. decorus Meyer and Bolland), Australia (N. victoriae Davis) and Thailand (N. lek Flechtmann). A key to the all previously known species of Neotetranychus was provided by Flechtmann (2013).
Thirty tetranychid mite species belonging to twelve genera have been reported previously from SA (Alatawi and Kamran, 2018; Kamran et al., 2018). In the present study two genera; Mixonychus (Bakerina Chaudhri), and Neotetranychus Trägårdh, also three species M. (B.) aculus (Chaudhri), Paraplonobia (Anaplonobia) harteni (Meyer) and Sonotetranychus daleae (Tuttle and Baker) are recorded for the first time from SA. Neotetranychus longisetus n. sp., is described and illustrated based on adult females collected from a native species of Heliotropium arbainense (Boraginaceae). In addition, Mixonychus (B.) aculus (Chaudhri), is re-described and illustrated based on specimens collected from an exotic mesquite species (native to Mexico, South America and the Caribbean), Prosopis juliflora (Sw.) DC. (Fabaceae). Evertella orissaensis (Prasad) is n. comb., transferred from Mixonychus to the genus Evertella Meyer as opisthosomal setae f1 absent, and Mixonychus (B.) ganjuis (Qian, Yuan and Ma) n. comb., is moved to the subgenus Bakerina (from subgenus Mixonychus) as it lacks clusters of protuberances on the dorsal integument. A key and details of the legs chaetotaxy, is provided for the world species of the genus Mixonychus.
Materials and methods
Spider mites were collected from a diversity of host plants in different localities across Saudi Arabia during 2016−2018. The mite specimens were collected by shaking the aerial parts over a piece of white paper. The mites moving on paper were collected with the help of camel hair brush and preserved in small vials containing 70% ethanol, then mounted in Hoyer's medium under a stereomicroscope (SZX10, Olympus®, Tokyo, Japan). The specimens were examined and identified under a phase contrast microscope (BX51, Olympus®, Japan) using diagnostic keys and available literature. Different mite body parts were imaged using an auto-montage software system (Syncroscopy, Cambridge, UK) attached with phase contrast microscope (DM2500, Leica®, Germany) and then drawn with Adobe Illustrator (Adobe System Inc., San Jose, CA, USA). All measurements are given in micrometers (μm). The morphological terminology used in this study followed Lindquist (1985). Legs setal counts are presented as the number of tactile setae followed by the number of solenidia and eupathidia in parentheses. The body measurements are presented for the holotype followed by the range for the paratypes in parentheses when types were examined. All collected specimens, including type specimens of the new species have been deposited at King Saud University Museum of Arthropods (KSMA, Acarology section), Department of Plant Protection, College of Food and Agriculture Sciences, King Saud University, Riyadh, Saudi Arabia.
Results
Family Tetyranychidae Donnadieu
Subfamily Tetranychinae Berlese
Tribe Tetranychini Reck
Genus Neotetranychus Trägårdh
Type species: Neotetranychus rubi Trägårdh, 1915: 33.
Diagnosis — Based on the Pritchard and Baker (1955) and Meyer, 1987.
Female: Dorsal body setae long and set on strong tubercles; opisthosomal integument mostly with transverse striae, wrinkled or reticulated clusters; legs empodia curved, uncinate and undivided along most of its length, with distal tip divided into pairs of fine hairs; two pairs of pseudoanal setae (ps1−2 ).
Neotetranychus longisetus n. sp.
(Figures 1−4)
ZOOBANK: 553E8F35-7EF4-46F7-B567-DB5CF26F0093 ![]()



Diagnosis (Based on adult female) — Propodosoma medially with longitudinal striae; hysterosoma medially with transverse striae and laterally with longitudinal to oblique striae; striae encircling bases of each dorsal seta, forming concentric rings; cuticle with rows of regular, shallow, scallop-shaped projections; dorsal body setae acicular, of even thickness along length, finely serrated, inserted on distinct tubercle; most of the dorsal setae twice as long as the distance to base of setae next consecutive row, dorsocentral setal pairs c1, d1, e1 and f1 subequal distance apart.
Description of the Adult female (n = 12)
Length of idiosoma (excluding gnathosoma) 331 (315−345), (including gnathosoma) 404 (390−420), maximum width at the level of row c setae 263 (255−270).
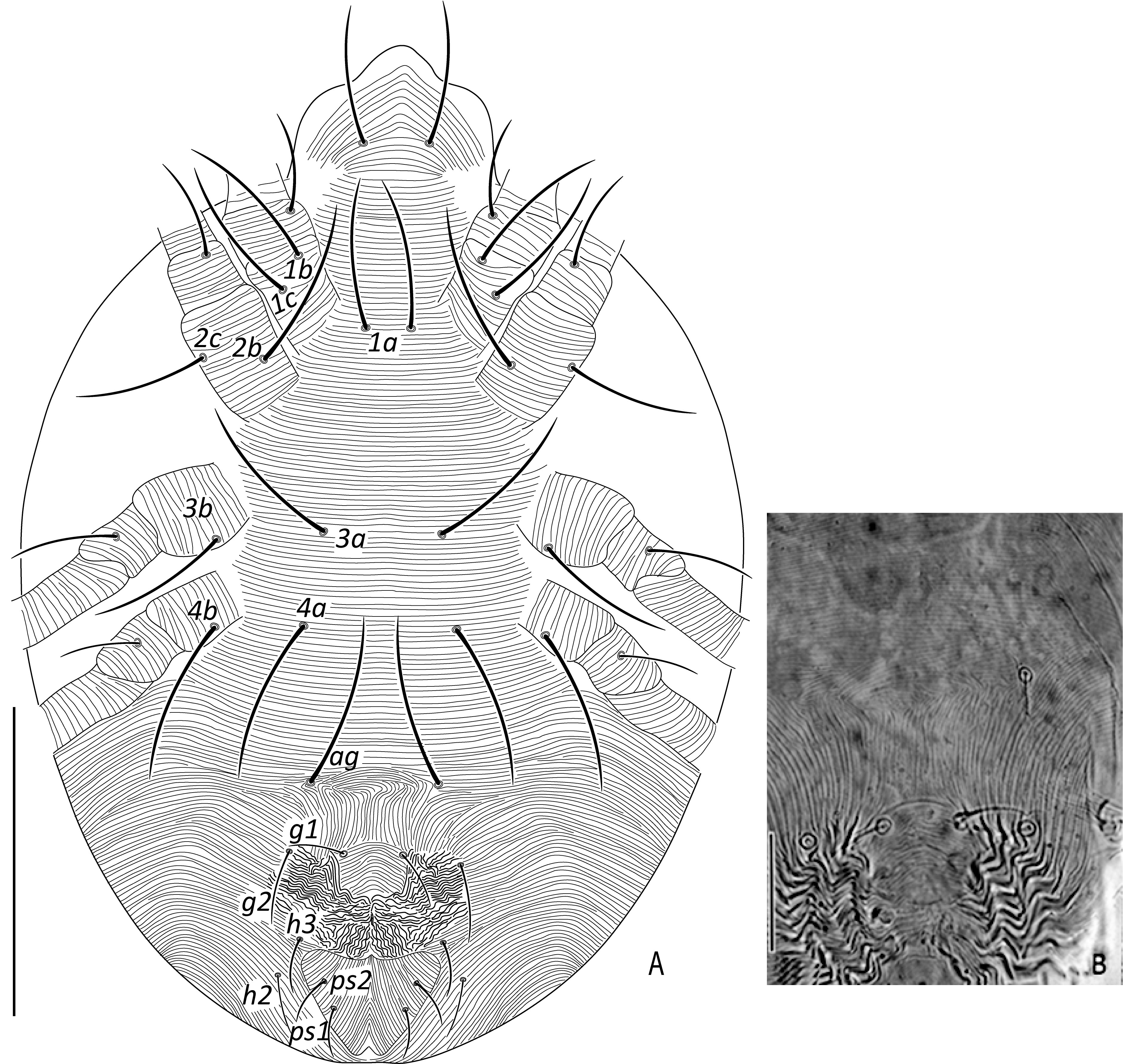


Dorsum (Figures 1A-C) — Dorsal integument entirely striated, striae longitudinal on propodosoma, striae transverse medially and longitudinal to oblique laterally on hysterosoma; cuticle with rows of regular, shallow, scallop-shaped projections (Figures 1A-B); dorsum with 13 pairs of setae; seta c3 inserted at lateral margin of body, often situated ventrally; setae v2, sc2, c3 and h1 shorter than other dorsal setae, almost equal in length; dorsocentral setal pairs c1, d1, e1 and f1 subequal distance apart. Length of dorsal setae: v2 63 (60−65), sc1 110 (107−114), sc2 63 (60−65), c1 142 (135−148), c2 126 (120−130), c3 65 (62−68), d1 142 (138−150), d2 129 (120−135), e1 89 (84−93), e2 123 (120−128), f1 105 (100−120), f2 84 (80−90), h1 63 (60−68); distance between dorsal setae: v2−v2 53 (50−55), sc1−sc1 68 (65−70), sc2−sc2 205 (200−210), c1−c1 42 (39−43), c2−c2 126 (120−135), c3−c3 263 (250−280), d1−d1 42 (39−45), d2−d2 147 (140−155), e1−e1 37 (35−42), e2−e2 131 (127−140), f1−f1 37 (35−40), f2−f2 108 (104−115), h1−h1 27 (25−30), v2−sc1 23 (21−25), sc1−sc2 63 (60−65), sc2−c3 74 (70−80), sc2−c2 53 (50−58), sc2−c1 84 (81−90), c1−c2 42 (38−45), c2−c3 71 (68−74), c1−d1 54 (50−58), c2−d2 58 (56−61), d1−e1 68 (66−72), d2−e2 68 (65−74), e1−f1 58 (53−60), e2−f2 74 (72−78), f1−h1 42 (39−45), f2−h1 55 (52−58), h2 21 (19−22), h3 26 (24−28), h2−h2 25 (23−26), h3−h3 45 (40−45).
Venter (Figures 2A-B) — Ventral idiosoma with transverse striations from setae 1a to just anterior to ag, pregenital area with transverse to arching striation (Figure 2B); striations on venter without lobes. Length of ventral setae: 1a 53 (51−55), 3a 55 (52−58), 4a 53 (50−55), 1b 63 (60−65), 1c 53 (51−56), 2b 58 (55−60), 2c 42 (39−44), 3b 50 (50−52), 4b 53 (50−55); distance between inter-coxal and coxae setae: 1a−1a 26 (25−28), 1b−1c 11 (10−12), 3a−3a 53 (50−55), 4a−4a 63 (60−65); aggenital setae: ag 55 (53−57), ag−ag 62 (60−65); two pairs of genital setae: g1 27 (25−29), g2 32 (31−34), g1−g1 26 (23−28), g2−g2 70 (65−74); two pairs of pseudoanal setae: ps1 16 (15−18), ps2 15 (13−16), ps1−ps2 6 (5−7), ps1−ps1 32 (30−35), ps2−ps2 21 (20−23).
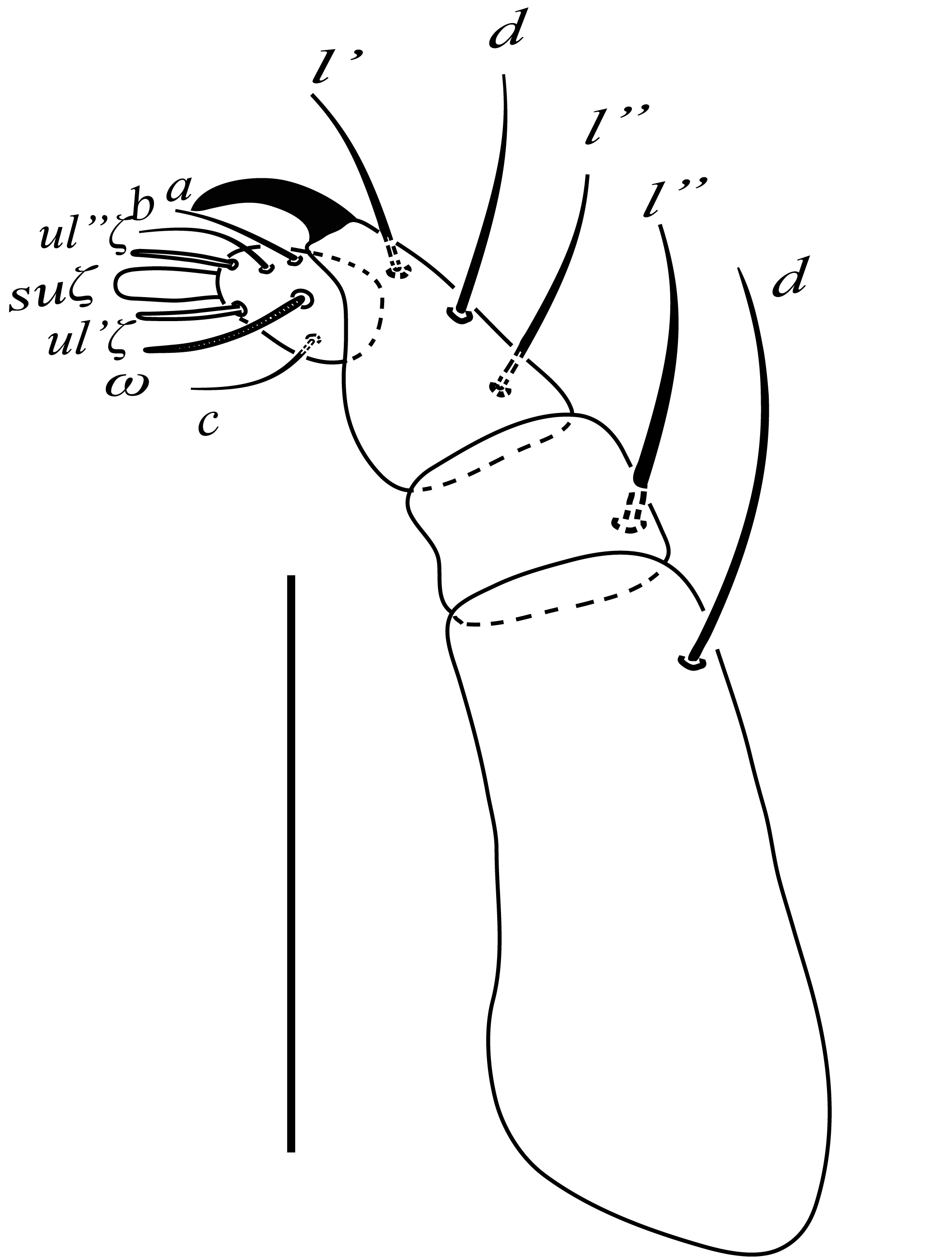
Gnathosoma (Figure 3) — Ventral infracapitular setae m 50 (48−53), m−m 35 (33−37). Palp femur and genu each with one seta, d 28 (26−29), l'' 18 (16−19); palp tibia with three setae, d 22 (19−22), l'' 16 (15−18), l' 15 (15−16) and a palp tibial claw; palp tarsus 14 long, 11 wide, with three simple setae a 10 (9−11), b 7 (7−9), c 8 (7−10), three eupathidia suζ 7 (6), ul''ζ = ul'ζ 6 (5−7), a solenidion ω 5 long; stylophore with weak anterior notch; peritremes terminating in short hook, like a golf-club (Figure 1C); terminal sensillum of palp tarsus conical, long, pointed about three times as long as wide (Figure 3).



Legs (Figures 4A-E) — Length of legs I−IV (without coxae): 184 (175−190), 173 (165−180), 191 (184−199), 200 (190−210) respectively; leg I 184 (175−190): trochanter 16 (14−17), femur 58 (55−61), genu 37 (35−38), tibia 37 (35−40), tarsus 37 (35−42); leg II 173 (165−180): trochanter 16 (15−17), femur 53 (51−58), genu 37 (34−39), tibia 42 (39−45), tarsus 42 (38−44); leg III 191 (184−199): trochanter 24 (23−26), femur 47 (45−49), genu 34 (31−36), tibia 42 (40−45), tarsus 42 (40−45); leg IV 200 (190−210): trochanter 21 (20−22), femur 47 (43−50), genu 37 (35−39), tibia 42 (40−45), tarsi 53 (50−55). Chaetotaxy on legs I−IV (solenidia in parentheses): coxae 2−1−1−1, trochanter 1−1−1−1; femora 9−7−4−4; genua 5−5−3−3; tibiae 9(1ω)−7(1ω)−6−6; tarsi 11(3ζ, 2ω)−9 (3ζ, 2ω)−8(1ω)−8(1ω).



Male and immature stages Unknown.
Etymology — The specific epithet refers to the length of dorsocentral setae which are comparatively longer than that of other species.
Material examined — Holotype female and 11 paratypes females, Heliotropium arbainense (Boraginaceae), Taif, 21°21.151ʹN, 040°19.515'E, elevation 1843 m, 11 September 2017, coll. E. M. Khan and M. U. Rehman. One Holotype female (KSMA-ARL -19/5) and 11 paratypes female (KSMA-ARL-19/6-16) has been deposited at King Saud University Museum of Arthropods (KSMA, Acarology section), Department of Plant Protection, College of Food and Agriculture Sciences, King Saud University, Riyadh, Saudi Arabia.
Remarks — Neotetranychus longisetus n. sp., is different from all other species of Neotetranychus by entire dorsum striated and with scallop-shaped structures. However, the new species resembles N. lek Flechtmann 2013. Neotetranychus lek has longitudinal striae between opisthosomal setae e1−e1 vs. transverse striae on N. longisetus n. sp., femur and genu III each 4 setae vs. 4 and 3 on N. longisetus and femur and genu IV each with 3 setae vs. 4 and 3 setae on N. longisetus n. sp..
New records for Saudi Arabia
Genus Mixonychus Ryke and Meyer
Mixonychus (Bakerina) aculus (Chaudhri)
Bakerina aculus Chaudhri, 1971: 197−199.
Re−description of adult female (n = 6)
(Figures 5−8)
Idiosoma oval—Length of body (excluding gnathosoma) 340−353, (including gnathosoma) 407−428, width of idiosoma at the level of setae c series 202−215.
Dorsum (Figures 5A-C) — Prodorsum bearing 3 pairs of lanceolate, finely barbed setae with blunt tips (v2, sc1, sc2). Propodosoma dorsomedially with lobed striae forming a strong reticulate pattern medially, with longitudinal laterally. Hysterosoma with 10 pairs of setae (c1−3, d1−2, e1−2, f1−2, h1), similar in shape to prodorsal setae. Opisthosoma medially with widely separated, lobed, transverse striae, laterally longitudinal to irregular. Dorsal idiosomal setae narrowly lanceolate, finely barbed (Figure 5C), just reaching the bases of setae next consecutive row, except setae c1 slightly shorter than distance to next setal row. Striae with irregularly shaped lobes. Length of dorsal body setae: v2 38−43, sc1 49−53, sc2 36−42, c1 37−46, c2 41−47, c3 40−45, d1 40−46, d2 44−50, e1 44−49, e2 49−53, f1 47−52, f2 38−41, h1 44−48; distance between dorsal setae: v2−v2 48−53, sc1−sc1 60−65, sc2−sc2 135−145, v2−sc1 20−22, c1−c1 50−53, c2−c2 95−100, c3−c3 178−190, d1−d1 42−46, d2−d2 106−117, e1−e1 33−39, e2−e2 92−99, f1−f1 44−47, f2−f2 70−77, h1−h1 15−16, v2−sc1 20−25, sc1−sc2 60−65, sc2−c3 50−56, sc2−c2 40−47, sc2−c1 53−59, c1−c2 19−23, c2−c3 48−54, c3−d2 51−55, c1−d1 49−52, c2−d2 54−59, d1−e1 45−50, d1−d2 31−35, d2−e2 49−53, d2−e1 46−50, e1−f1 46−53, e1−e2 32−36, e2−f2 62−68, f1−h1 40−47, f1−f2 25−27, f2−h1 33−38.



Venter (Figure 6) — Idiosoma ventrally with transverse striations from setae 1a to 4a, oblique or longitudinal in pre-genital area; striations on venter without lobes; length of inter-coxal and coxae setae, setae on coxae III and IV are serrate: 1a 43−47, 3a 41−44, 4a 31−34, 1b 51−55, 1c 33−37, 2b 41−46, 2c 19−23, 3b 27−30, 4b 30−34; distance between inter-coxal and coxae setae: 1a−1a 31−35, 1b−1c 10−12, 2b−2c 18−20, 3a−3a 50−55, 4a−4a 60−65, 1a−3a 81−88, 3a−4a 40−45, 4a−ag 47−52; aggenital setae: ag 33−37, ag−ag 42−49; two pairs of genital setae: g1 27−31, g2 24−26, g1−g1 28−30, g2−g2 46−53, g1−g2 14−16, ag−g1 20−22, ag−g2 27−29; two pairs of anal setae: ps1 11−13, ps2 10−12, ps1−ps2 11−13, ps1−ps1 15−17, ps2−ps2 16−18; para anal setae two pairs: h2 15−17, h3 16−18, h2−h2 10−12, h2−h3 33−38.



Gnathosoma (Figure 7) — Ventral infracapitular setae m 35−39, m−m 19−21. Palp femur and genu each with one seta, d 32−35, l'' 23−25; palp tibia with three setae, d 20−22, l'' 17−19, l' 15−16 and a palp tibial claw; palp tarsus 14 long, 11 wide, with three simple setae a 8−10, b 9−10, c 10−12, three eupathidia suζ 5−6, ul''ζ = ul'ζ 7−9, a solenidion ω 11 long; peritremes terminating in hooked; terminal sensillum of palp tarsus conical, long, pointed about 3 time long as wide to the base.


Legs (Figures 8A-D) — Length of legs I−IV (without coxae): 216−230, 165−180, 186−199, 224−240, respectively; leg I: trochanter 13−16, femur 75−83, genu 35−38, tibia 52−55, tarsus 40−45; leg II: trochanter 10−12, femur 55−59, genu 31−34, tibia 30−35, tarsus 38−44; leg III: trochanter 25−28, femur 49−53, genu 31−35, tibia 40−45, tarsus 45−48; leg IV: trochanter 20−22, femur 61−65, genu 35−39, tibia 50−55, tarsi 56−60; legs chaetotaxy on I−IV (solenidia in parenthesis): coxae; 2−2−1−1, trochanter 1−1−1−1; femora 8/7−6−4−4/3; genua 5−5−3−3; tibiae 9(1ω)−7−6−7; tarsus 11(3ζ, 2ω)−9 (3ζ, 2ω)−8(1ω)−8(1ω).



Male and immature stages Unknown.
Material examined — Four females, Prosopis juliflora (Fabaceae) (native to Mexico, South America, and the Caribbean), Alhasa 27°55.0143ʹN, 047°66.8970'E, 21 April 2018, coll. M. Kamran, J. H. Mirza and H. M. Saqib. Four females (KSMA-ARL-19/16-20) have been deposited at King Saud University Museum of Arthropods (KSMA, Acarology section), Department of Plant Protection, College of Food and Agriculture Sciences, King Saud University, Riyadh, Saudi Arabia.
Distribution — Pakistan (Chaudhri, 1971) and SA in present study.
Known hosts — Senegalia modesta (syn. Acacia modesta, Mimosaceae) (native to India, Pakistan and Afghanistan), Albizia lebbeck (paratypes host, Mimosaceae) (native to Indomalay region, Papua New Guinea and northern Australia), Platycladus orientalis (Cupressaceae) (native to Korea and northern China), Salvadora oleoides (Salvadoraceae) (paratypes host; native to India, Pakistan and southern Iran), Vachellia nilotica (syn. Acacia arabica) (Mimosaceae) holotype host (native to Africa, Middle East and India) (Chaudhri, 1971; Chaudhri et al., 1974).
Remarks — We could not find the type specimens of M. (B.) aculus from the depository. The specimens of M. (B.) aculus collected from Saudi Arabia are morphologically similar to the original description except minor differences in length of dorsal and ventral body setae; v2 38−43 vs. 42, sc1 49−53 vs. 57 sc2 36−42 vs. 47, c1 37−46 vs. 44, c2 41−47 vs. 47 d1 40−46 vs. 52, d2 44−50 vs. 49, e1 44−49 vs. 52, e2 49−53 vs. 49, f1 47−52 vs. 52, f2 38−41 vs. 47, h1 44−48 vs. 49; 1a 43−47 vs. 39, 3a 41−44 vs. 42, 4a 31−34 vs. 36 and solenidia on both tarsi III and IV 10−12 vs. 16 in original description.
Genus Sonotetranychus Tuttle, Baker and Abbatiello
Sonotetranychus daleae (Tuttle and Baker)
Anatetranychus daleae Tuttle and Baker, 1968: 110.
Sonotetranychus daleae (Tuttle and Baker), Tuttle, Baker and Abbatiello, 1976: 70.
Material examined — Two females, Vachellia nilotica, Taif, 21°30.112'N, 040°29.118'E, elevation 1483 m, 11 September 2017, coll. E. M. Khan and M. U. Rehman; two females, Vachellia nilotica, Taif, 21°20.474ʹN, 040°19.325'E, elevation 1838 m, 11 September 2017, coll. E. M. Khan and M. U. Rehman; five females, Vachellia nilotica, Taif, 21°14.372ʹN, 040°31.135'E, elevation 1618 m, 13 September 2017, coll. E. M. Khan and M. U. Rehman. Nine females (KSMA-ARL-19/20-29) have been deposited at King Saud University Museum of Arthropods (KSMA, Acarology section), Department of Plant Protection, College of Food and Agriculture Sciences, King Saud University, Riyadh, Saudi Arabia.
Distribution — Mexico, Pakistan, Taiwan and United States of America (Migeon and Dorkeld, 2006−2018).
Known hosts — Dalea formosa (Fabaceae) (the type host, native to southwestern USA and Mexico), Psorothamnus emoryi (Fabaceae) (native to southern USA and Mexico) and Vachellia nilotica (Mimosaceae) (Migeon and Dorkeld, 2006−2018).
Subfamily Bryobiinae Berlese
Tribe Hystrichonychini Pritchard and Baker
Genus Paraplonobia Wainstein
Subgenus Anaplonobia Wainstein
Paraplonobia (Anaplonobia) harteni (Meyer)
Neopetrobia harteni Meyer, 1996: 9.
Material examined — Four females, Indigofera spinosa (Leguminosae) (native to the region and northeast Africa), Taif, 21°33.926'N, 040°07.260'E, elevation 643 m, 15 October 2016, coll. M. U. Rehman and M. Kamran. Four females (KSMA-ARL-19/29-33) have been deposited at King Saud University Museum of Arthropods (KSMA, Acarology section), Department of Plant Protection, College of Food and Agriculture Sciences, King Saud University, Riyadh, Saudi Arabia.
Distribution — Yemen (Meyer, 1996).
Known hosts — Unknown plant species (Meyer, 1996).
New combination
Evertella orissaensis (Prasad, 1975), n. comb.
Bakerina orissaensis Prasad, 1975: 18−20.
Mixonychus (Bakerina) orissaensis (Prasad): Bolland et al., 1998: 86.
Distribution — India.
Known hosts — Allium sp., (Amaryllidaceae), Artocarpus heterophyllus (Moraceae) (Prasad, 1975).
Remarks — Evertella was erected by Meyer (1987) to accommodate Mixonychus (Mixonychus) koegasensis (Meyer, 1974). Like Mixonychus (Mixonychus), Evertella is characterized by having the dorsal integument finely striate and with protuberances that produce a reticulate appearance, and a simple claw-like empodium. Evertella was separated from Mixonychus because of dorsal opisthosomal setae f1 are absent (present in Mixonychus).
Prasad 1975 described the dorsal integument of Bakerina orissaensis as striate with comparatively large, round punctations, and Bolland et al. (1998) placed B. orissaensis within Mixonychus (Bakerina) due to the striate dorsum and overlooked the absence of seta f1 in B. orissaensis which distinguish Evertella from Mixonychus. Therefore, due to absence of f1 seta, Mixonychus (Bakerina) orissaensis is transferred from Mixonychus (Bakerina) to Evertella.



Mixonychus (Bakerina) ganjuis (Qian, Yuan and Ma, 1980), n. comb.
Mixonychus (Mixonychus) ganjuis Qian, Yuan and Ma, 1980: 80.
Mixonychus (Mixonychus) ganjuis Qian, Yuan and Ma; Yi and Jin, 2017: 1296−1301.
Distribution — China.
Known hosts — Amelanchier sp., (Rosaceae), Citrus reticulata (Rutaceae) (Yi and Jin 2017).
Remarks — The species M. (B.) ganjuis having dorsal integument striated, without lumps and spinules, which was misplaced previously in the subgenus Mixonychus (Yi and Jin, 2017), is transferred from subgenus Mixonychus to subgenus Bakerina.
Key to world species of the genus Mixonychus Ryke and Meyer (Based on adult female)
1. Dorsal integument with small protuberances forming a reticulate pattern or producing the appearance of a reticulate pattern
...... subgenus Mixonychus Ryke and Meyer – 2
— Dorsal integument without protuberances; integument with/without spinules present
...... 4
2. Length of dorsal body setae less than half the longitudinal distance to the setae in the next row; only dorsal opisthosomal setae f2 and h1 set on tubercles
...... M. (M.) dulcis Meyer, South Africa
— Length of dorsal body setae more than half the longitudinal distance to the setae in the next row; almost all dorsal opisthosomal setae set on tubercles
...... 3
3. Femora I and II with 8 and 6 setae, respectively
...... M. (M.) acaciae Ryke and Meyer, South Africa
— Femora I and II with 7 and 5 setae, respectively
...... M. (M.) neoacaciae Smiley and Baker, Yemen
4. Dorsal integument with spinules
...... subgenus Tylonychus Miller – 5
— Dorsal integument without spinule
...... subgenus Bakerina Chaudhri – 8
5. Dorsal opisthosomal setae long, clearly longer than the bases of the setae in the next row
...... 6
— Dorsal opisthosomal setae short, not reaching the bases of the setae in the next row.
...... 7
6. Dorsal body setae set on strong tubercles; female propodosoma integument with simple striations; dorsal opisthosomal setae f2 short, as compared to other dorsal setae
...... M. (T.) tasmaniensis (Miller), Australia
—Dorsal body setae not set on tubercles; female propodosoma integument with a reticulate pattern; dorsal opisthosomal setae f2 as long as other dorsal body setae
...... M. (T.) jayaraji (Karuppuchamy and Mohanasundaram), India
7. Dorsal body setae stout, rod shaped; genua III and IV with 3 setae; femora I−IV with 6−5−3−2 setae
...... M. (T.) transvaalensis (Meyer), South Africa
— Dorsal body setae oblong, lanceolate; genua III and IV with 4 setae; femora I−IV with 9−8−4−4 setae
...... M. (T.) cruzae (Corpuz-Raros), The Philippines
8. Dorsal opisthosomal setae d1, e1 as long as or longer than the distance to the bases of setae in the next row
...... 9
— Dorsal opisthosomal setae d1, e1 obviously shorter than the distance to the bases of the setae in the next row (approx. 2/3 the distance)
...... 14
9. Femora I and II with 9 and 7 setae, respectively; genua III and IV each with 4 setae
...... 10
— Femora I and II with 8 and 6 setae, respectively; genua III and IV each with 3 setae
...... 11
10. Medial integument of propodosoma striated; dorsal setae gradually expanded distally (weakly clavate)
...... M. (B.) ganjuis (Qian, Yuan and Ma), China
— Medial integument of propodosoma reticulated; dorsal setae not expanded distally, of even thickness along length
...... M. (B.) nanchangensis (Ma and Yuan), China
11. Dorsal body setae lanceolate; tibia I without lanceolate seta; tarsi III and IV with long solenidion (16 μm long)
...... 12
— Dorsal body setae of even thickness along length, weakly to not tapering, with broadly blunt tips; tibia I with 3 lanceolate setae; tarsi III and IV with short solenidion (5 μm long)
...... 13
12. Dorsal opisthosomal setae c1 (44 μm) shorter than the distance c1−d1
...... M. (B.) aculus (Chaudhri), Pakistan
— Dorsal opisthosomal setae c1 (60 μm) reaching the base of setae d1
...... M. (B.) lepidus (Chaudhri), Pakistan
13. Peritremes ending in simple unexpanded bulb; dorsal opisthosomal f1 70 μm long and f2 44 μm long
...... M. (B.) carracis (Chaudhri), Pakistan
— Peritremes ending in an expanded bulb; dorsal opisthosomal setae f1 83μm long and f2 47 μm long
...... M. (B.) normalis (Chaudhri), Pakistan
14. Dorsal body setae gradually expanding along length, elongate obovate; femur III with 3 or 4 setae
...... 15
— Dorsal body setae of even thickness along length; femur III with 5 setae
...... M. (B.) ziolanensis (Lo and Ho), Taiwan
15. Femur II with 6 setae; tibia III with 5 setae
...... 16
— Femur II with 7 setae; tibia III with 6 setae
...... 17
16. Femur III with 3 setae; tibia II with 5 setae
...... M. (B.) citraeus Flechtmann and Mesa, Colombia
— Femur III with 4 setae; tibia II with 8 setae
...... M. (B.) aestiva (Tseng), Taiwan
17. Tibia IV with 7 setae
...... M. (B.) murrayae (Gao and Ma), China
—Tibia IV with 6 setae
...... M. (B.) thailandicus (Tangkanasing), Thailand
Acknowledgments
The authors wish to thank the Deanship of Scientific Research at the King Saud University, Riyadh for providing facilities and funds to complete this research work through the research project [RG– 1437–043]. We are also grateful to Dr. Carlos H.W. Flechtmann (University of Sao Paulo, Departamento de Entolomogia e Acarologia, Brazil) for providing literature.
References
Alatawi F.J., Kamran M. 2018. Spider mites (Acari: Tetranychidae) of Saudi Arabia: two new species, new records and a key to all known species. J. Nat. Hist., 52(7-8): 429-455. doi:10.1080/00222933.2018.1434251 ![]()
Bolland H.R., Gutierrez J., Flechtmann C.H.W. 1998. World catalogue of the spider mite family (Acari: Tetranychidae). Leiden, Brill Boston Koln. p. 1-392.
Chaudhri W.M. 1971. A new spider mite genus, Bakerina, (Acarina, Tenuipalpoidini), with the description of four new species. Pak. J. Zool., 3(2): 195-202.
Chaudhri W.M., Akbar, S., Rasool A. 1974. Taxonomic studies of the mites belonging to the families Tenuipalpidae, Tetranychidae, Tuckerellidae, Caligonellidae, Stigmaeidae and Phytoseiidae. Uni. of Agric., Lyallpur, Pak., PL-480. Project on mites. p. 1-250.
Corpuz-Raros L.A. 1978. New Philippine Tetranychoidea. Kalikasan, Philippine Journal of Biology, 7: 211-230.
Flechtmann C.H. 2013. A new species of Neotetranychus Trägårdh (Acari, Prostigmata, Tetranychidae) from Thailand with a key to world species. Persian J. Acarol., 2(1): 35-40.
Flechtmann C.H.W., Mesa N.C. 2016. First record of the spider mite genus Mixonychus (Acari: Tetranychidae) from the Americas based on the description of a new species from Colombia. Persian J. Acarol., 2016, 5(4): 271-279. doi:/10.22073/pja.v5i4.25450 ![]()
Gutierrez J. 1985. Spider mites. Their Biology, Natural Enemies and Control. Vol. 1A. 1.1.4 systematics. Ed. By: Helle W. Sabelis M.W. Elsevier Sci. Publ. B.V., Amsterdam, pp. 75-90.
Hong X.Y., Zhang Z.Q., Lin G.Q. 2010. Tetranychidae of China: a review of progress, with a checklist. In: Zhang Z.Q., Hong X.Y., Fan, Q.H. (eds) Xin Jie-Liu Centenary: Progress in Chinese Acarology. Zoosymposia, 4: 133-150.
Kamran M., Khan E.M., Alatawi F.J. 2018. The spider mites of the genus Eutetranychus Banks (Acari, Trombidiformes, Tetranychidae) from Saudi Arabia: two new species, a re-description, and a key to the world species. ZooKeys, (799): 47-88. doi:10.3897/zookeys.799.25541 ![]()
Karuppuchamy P., Mohanasundaram M. 1987. New species and records of tetranychid mites (Tetranychidae: Acarina) from India. Entomon., 12: 89-94.
Lindquist E.E. 1985. Spider mites. Their Biology, Natural Enemies and Control. Vol. 1A.1.1.1 External anatomy. Ed. By: Helle W. Sabelis M.W. Elsevier Sci. Publ. B.V., Amsterdam, pp. 3-28.
Lo P.K.C., Ho C.C. 1989. A new species of spider mite from Taiwan (Acarina: Tetranychidae). J. Taiwan Museum, 42: 55-58.
Ma E.P., Yuan Y.L. 1981. A new species of the genus Bakerina from China (Acari: Tetranychidae). Entomotaxonomia, 3: 71-74.
Miller L.W. 1966. The tetranychid mites of Tasmania. Proceedings of the Royal Society of Tasmania, 100: 53-76.
Meyer M.K.P.S. 1974. A revision of the Tetranychidae of Africa (Acari) with a key to the genera of the world. Entomol. Mem. Dep. Agric. Tech. Serv., Repub. S. Afr., 36: 1-291.
Meyer M.K.P.S. 1987. African Tetranychidae (Acari: Prostigmata) with reference to the world genera. Entomol. Mem. Dep. Agric. Water Supply, Repub. S. Afr., 69: 1-175.
Meyer, M.K.P.S.1996. On some spider mites (Acari: Tetranychidae) of Yemen. Fauna of Saudi Arabia, 15: 5-19.
Migeon A., Dorkeld F. 2006-2018. Spider Mites Web: a comprehensive database for the Tetranychidae [Internet]. [25 Spetember, 2018] Available from http://www.montpellier.inra.fr/CBGP/spmweb ![]() .
.
Prasad V. 1975. Plant feeding mites of India (Acarina: Tetranychoidea): III. Genera Bakerina, Mesobryobia, Panonychus and Porcupinychus. Int. J. of Acarol., 1(2): 18-25. doi:10.1080/01647957508683745 ![]()
Pritchard A.E., Baker E.W. 1955. A revision of the spider mite family Tetranychidae. Te Pacifc Coast Entomological Society 2: 1-472. doi:10.5962/bhl.title.150852 ![]()
Qian Y.H., Yuan Y.L., Ma E.P. 1980. A new species of the genus Mixonychus from China (Acari: Tetranychidae). J. Shanxi Uni., 2(2): 80-82.
Ryke P.A.J., Meyer M.K.P.S. 1960. The parasitic and predacious mite fauna (Acarina) associated with Acacia karroo Hayne in the Western Transvaal. Libro Homenaje al Dr. Eduardo Caballero y Caballero, Mexico D. F.: 559-569.
Smiley R.L., Baker E.W. 1995. A report on some tetranychid mites (Acari: Prostigmata) from Yemen. Int. J. Acarol., 21(3): 135-164. doi:10.1080/01647959508684055 ![]()
Tangkanasing P. 1988. A new species of Bakerina Chaudhri from Thailand (Tetranychidae: Acari). Int. J. Acarol., 14(1): 9-11. doi:10.1080/01647958808683798 ![]()
Tseng Y.H. 1990. A monograph of the mite family Tetranychidae (Acarina: Tronbidiformes) from Taiwan. Taiwan Mus. special Publ., ser. n 9 Taipei, Rep. China, p. 1-226.
Tuttle D.M., Baker E.W. 1968. Spider mites of southwestern United States and a revision of the family Tetranychidae. Tuscon, Ariz., Uni. Ariz., Press, p. 1-143.
Tuttle D.M., Baker E.W., Abbatiello M.J. 1976. Spider mites of Mexico (Acari: Tetranychidae). Int. J. Acarol., 2 (2): 1-102. doi:10.1080/01647957608683760 ![]()
Yi T.C., Jin D.C. 2017. Re-description of three species of Mixonychus (Acari, Tetranychidae) from China. Syst. Appl. Acarol., 22(9): 1295-1315. doi:10.11158/saa.22.9.2 ![]()



2018-12-02
Date accepted:
2019-11-05
Date published:
2019-11-18
Edited by:
Migeon, Alain

This work is licensed under a Creative Commons Attribution 4.0 International License
2019 Khan, Eid Muhammad; Kamran, Muhammad and Alatawi, Fahad Jaber
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



