The first record of a scabies mite of the family Sarcoptidae (Acari, Sarcoptiformes) associated with Chiroptera in Russia
Kolesnikov, Vasiliy B.  1
and Orlova, Maria V.
1
and Orlova, Maria V.  2
2
1Papanin Institute for Biology of Inland Waters, Russian Academy of Sciences, 152742 Borok,Yaroslavl Region, Russia.
2Tyumen State Medical University, 625023 Tyumen, Russia.
2026 - Volume: 66 Issue: 2 pages: 296-306
https://doi.org/10.24349/kzn1-ogpvZooBank LSID: 9DAFA973-BB80-41AC-BC41-E11C9A1152B4
Original research
Keywords
Abstract
Introduction
Bats (Chiroptera) are the second largest order of mammals after rodents (Rodentia) currently comprising over 1400 species that is about 20% of all classified mammal species worldwide (Arnaout et al. 2022; Simmons and Cirrannello 2024). The ability to fly enabled bats to occupy the most diverse roosts, including tree-holes and crowns, caves, crevice, dens of larger mammals, human buildings and other natural and artificial shelters. The body of bats themselves is a habitat for a large number of parasites and commensals, among which mites and ticks (Acari) represent one of the most diverse group of arthropods associated with these hosts. A great diversity among the mites belong to astigmatid mites (Psoroptidia). These mites associated with bats live on or in the skin, in hair follicles, or in the nostrils and mouth of bats (Baker and Craven 2003).
Among Psoroptidia, scabies mites of the family Sarcoptidae are one of the most highly specialized taxonomic group of parasites associated with mammals (Fain 1978; Andrews 1983; Lange and Sokolova 1992; Klompen 1992, Ansari et al. 2024). This family is a large taxon of parasitic mites (including 117 species and 15 genera), all members of which are obligate, permanent parasites of mammals (Bochkov 2010). Some species of Sarcoptidae have gained a significant notoriety by being dangerous parasites of humans and domestic animals, e.g. Sarcoptes scabiei (Linnaeus, 1758) and Notoedres (Notoedres) cati (von Hering, 1838) (Klompen 1992). Among sarcoptids, a large number of species from the subfamily Teinocoptinae Fain, 1959 are parasites of bats (OConnor 2009). The genus Notoedres Railliet 1893 comprises about 45 species in two subgenera (Klompen 1992) parasitizing various mammal hosts, but bats (Chiroptera) harbor over the two thirds of species recognized in this genus (Ah 1975). It is hypothesized that Notoedres originated as a parasite of bat, however its crown group contains species associated with Rodents (e.g. N. musculi (Kramer, 1865), N. muris Megnin, 1877) and Carnivora (N. cati) that is apparently due to host shifts from bats to members of these mammalian host orders (Klimov and He 2024). According to previous data (Klompen 1992, Bochkov 2010), chiropteran mange mites Notoedres spp. have worldwide distribution and parasitize bat hosts belonging to families Vespertilionidae (14 species), Miniopteridae (1 species), Emballonuridae (3 species), and Molossidae (15 species). Klompen (1992) made the latest revision of the genus, also providing a key to all species.
Previously, only three Notoedres species have been reported from the territory of the former USSR: Notoedres (N.) miniopteri Fain, 1959 (from Miniopterus schreibersi (Kuhl, 1819), Chiroptera: Miniopteridae) and N. (N.) pseudomuris Lavoipierre, 1968 (from Microtus transcaspicus Satunin, 1905, Rodentia: Cricetidae) in Turkmenistan, N. (N.) myoticola (Fain, 1959) in Uzbekistan (from Myotis blythii (Tomes, 1857), Chiroptera: Vespertilionidae), (Klompen 1992). To date, no findings of Notoedres species from chiropterans had reported from the territory of Russia.
Investigation of bat's ectoparasites in the Western and especially Eastern Siberia (Russia) started not long ago because of the hard accessibility of many territories in this regions, and so far it has only covered the Mesostigmata (Orlova et al. 2014; Orlova and Zhigalin 2015; Orlova et al. 2015; Orlova et al. 2017). During the study of ectoparasites of Chiroptera in the southern part of Eastern Siberia (Russia), a new Notoedres species was found on the Ikonnikov's bat M. ikonnikovi and is described herein from the female and larva.
Material and methods
Bats were collected using mist nets and Borissenko mobile traps (Borissenko 1999) near bat roosts (Orlova et al. 2021). After capture, each animal was placed in a separate cotton bag to prevent sample cross-contamination. Bats were morphologically identified using the taxonomic descriptions of Ditz et al. (2009). Bat classification and nomenclature follow Wilson & Mittermeier (2019). Material collection was carried out in accordance with the requirements of the Ministry of Natural Resources and Ecology of Russia.
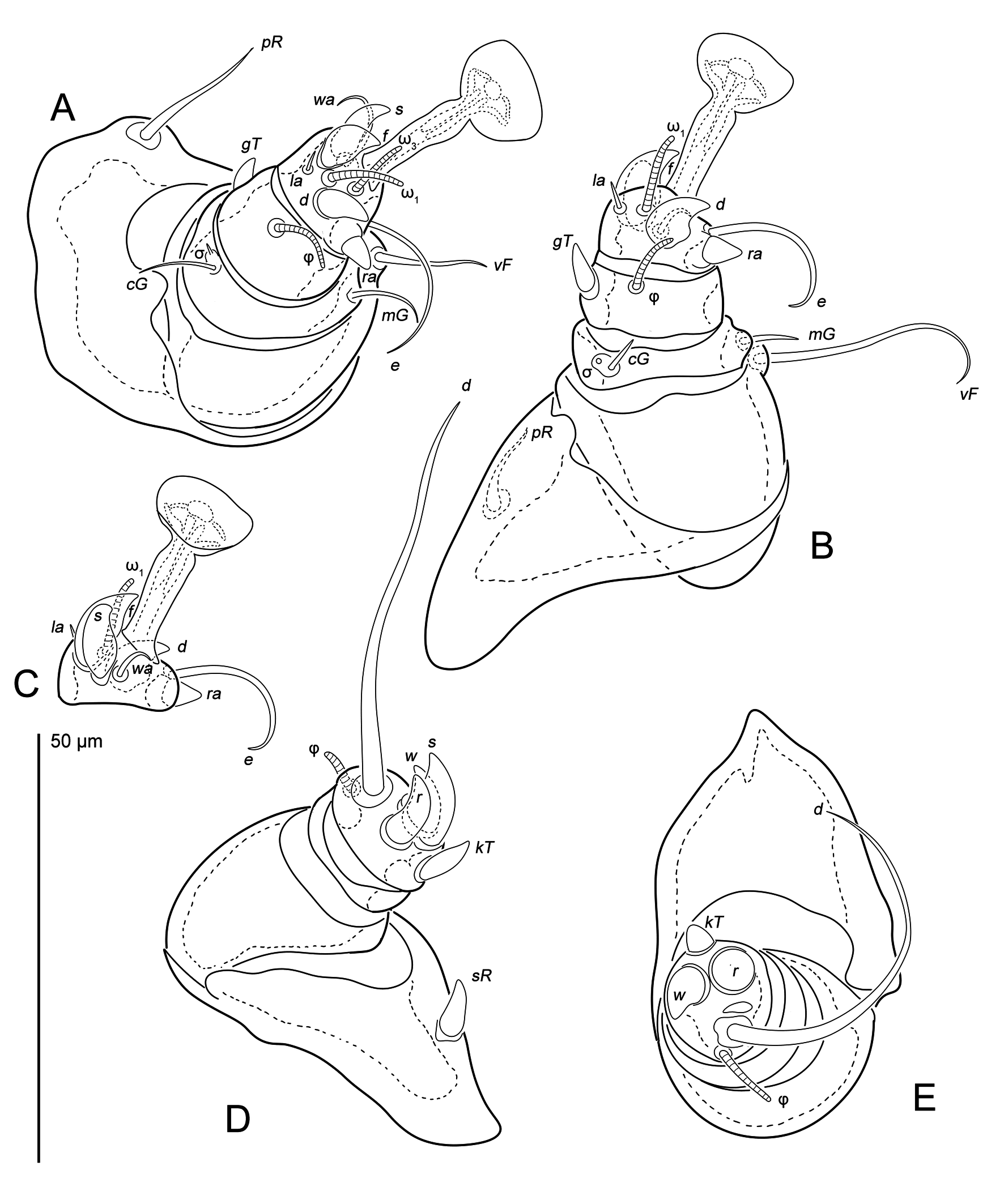
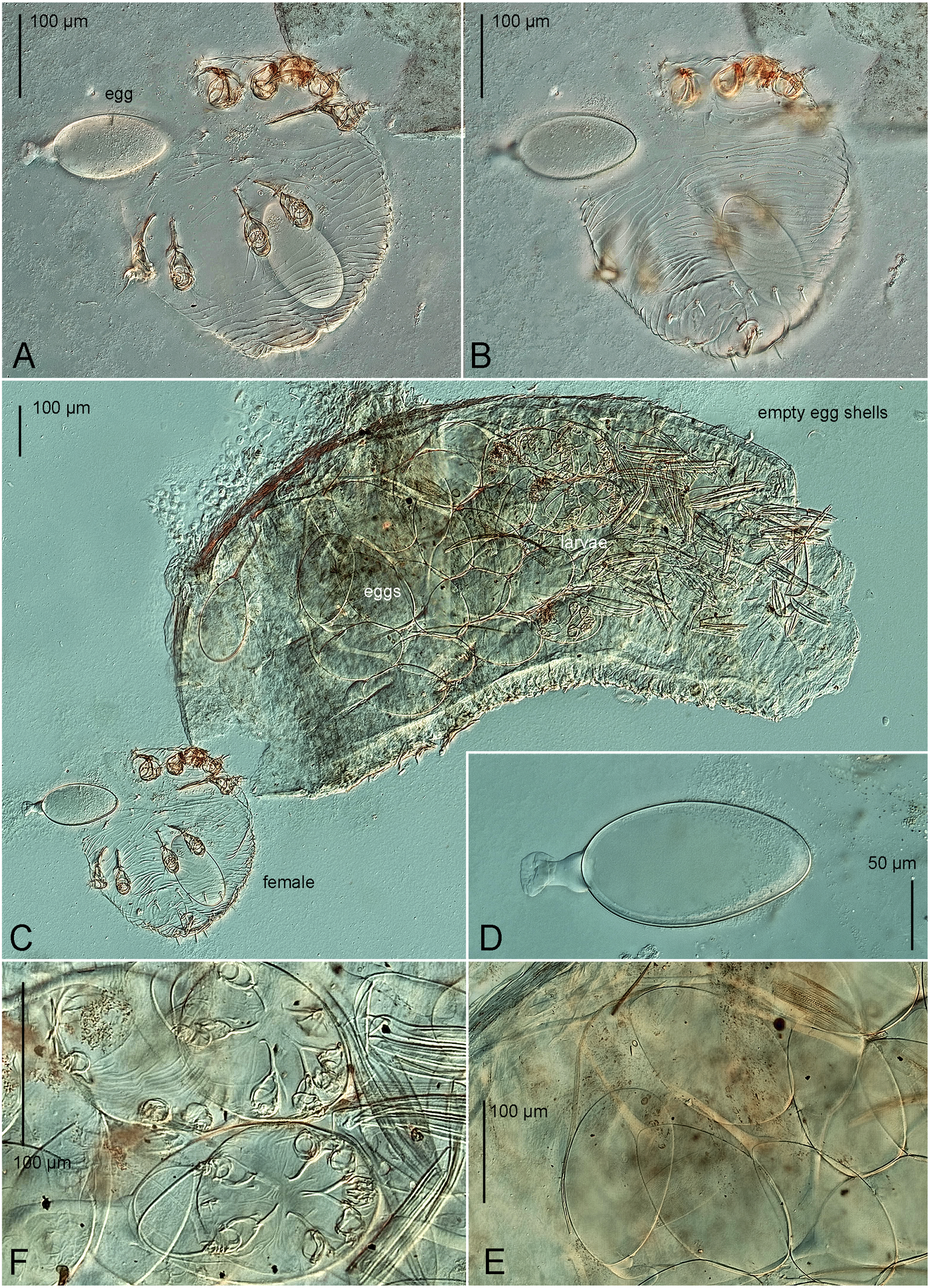
One female, a mane of eggs and larvae discovered in a bean-shaped shell (''trunk-like structure'' by Klompen 1992) projecting from the skin (Fig. 5C), typical for some sarcoptic mites (Roesler, 1932; Hedeen, 1953; Fain & Aellen, 1961; Klompen 1992). This shell with mites was removed from bat with forceps and dissecting needles and fixed in 70% ethanol, after which the host bat was released. The mites were mounted on slides in Faure-Berlese medium. The idiosomal chaetotaxy follows Griffiths et al. (1990) with modifications by Klompen (1992) and Norton (1998) for coxal-genital setae. The leg chaetotaxy follows Grandjean (1939) and Griffiths (1977) (see Klimov et al. (2026) for homologies of leg setae and solenidia). Host systematics follows the checklist of Wilson and Reeder (2005). All measurements are given in micrometers (μm). Mite morphology was studied using a Bioptic C-400 (Bioptic, Moscow, Russian Federation) microscope equipped with bright field and differential interference contrast optics (DIC).
Systematics
Family Sarcoptidae Murray, 1877
Subfamily Teinocoptinae Fain. 1959
Genus Notoedres Railliet, 1893
Subgenus Notoedres Railliet, 1893
Type species: Sarcoptes cati Hering, 1838
Notoedres (Notoedres) alinae sp. n.
ZOOBANK: 10B4C47D-65A1-41E6-843A-BF92E2848FB1 ![]()
(Figures 1–6)
Type material
Holotype (female), slide, Khongor-Uula, Tunkinsky National Park, Republic of Buryatia, Russia, 06.08.2016, ex. Myotis ikonnikovi, (Chiroptera: Vespertilionidae), leg. D.V. Kazakov. Paratypes (6 larvae), same data as holotype.
Depository
Holotype and all addition material deposited in the Zoological Institute, Russian Academy of Sciences, St. Petersburg, Russia.
Diagnosis
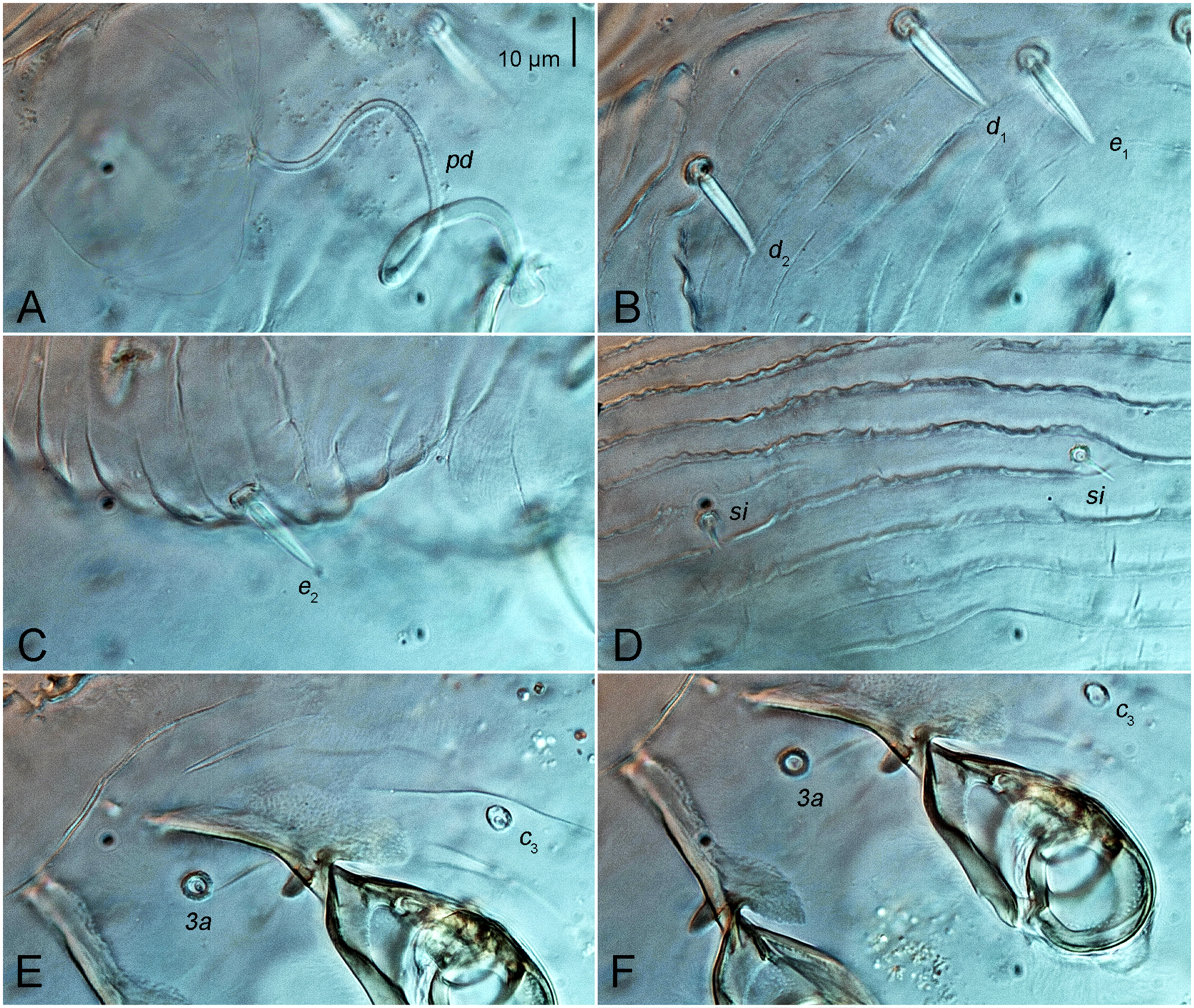
Female. Idiosoma rounded, dorsal entirely striated with irregular and often interrupted striae. Setae d1, d2, e1, e2, spine-like, with pointed tip, approximately equal in length (19–20) and width (2.5–3). Preanal setae h1, h2 spine-like, 22–23 long. Setae se, si, c1, c2 and cp well developed short spine (5–7 long). Prodorsal shield absent. Coxal apodemes I merged in a Y. Setae 4a present. Palpal seta a (11 long) with inflated base (2 wide). Legs seta d III 50 length. Trochanteral setae pR I, II with inflated base, sR III spiniform.
Larva. Dorsal surface striated transversely except for large glabrous oval area around anus. Setae d1, d2, e1, e2, h1 spine-like, with pointed tip. Coxal apodemes I fused at posterior halves. Seta 3a spine-like, with a pointed tip. Legs seta d III 11 length.
Descriptions
Female — (Figures 1, 2, 5A–C, 6). Idiosoma ball shaped (Figures 1, 5A, B), 343 × 330.
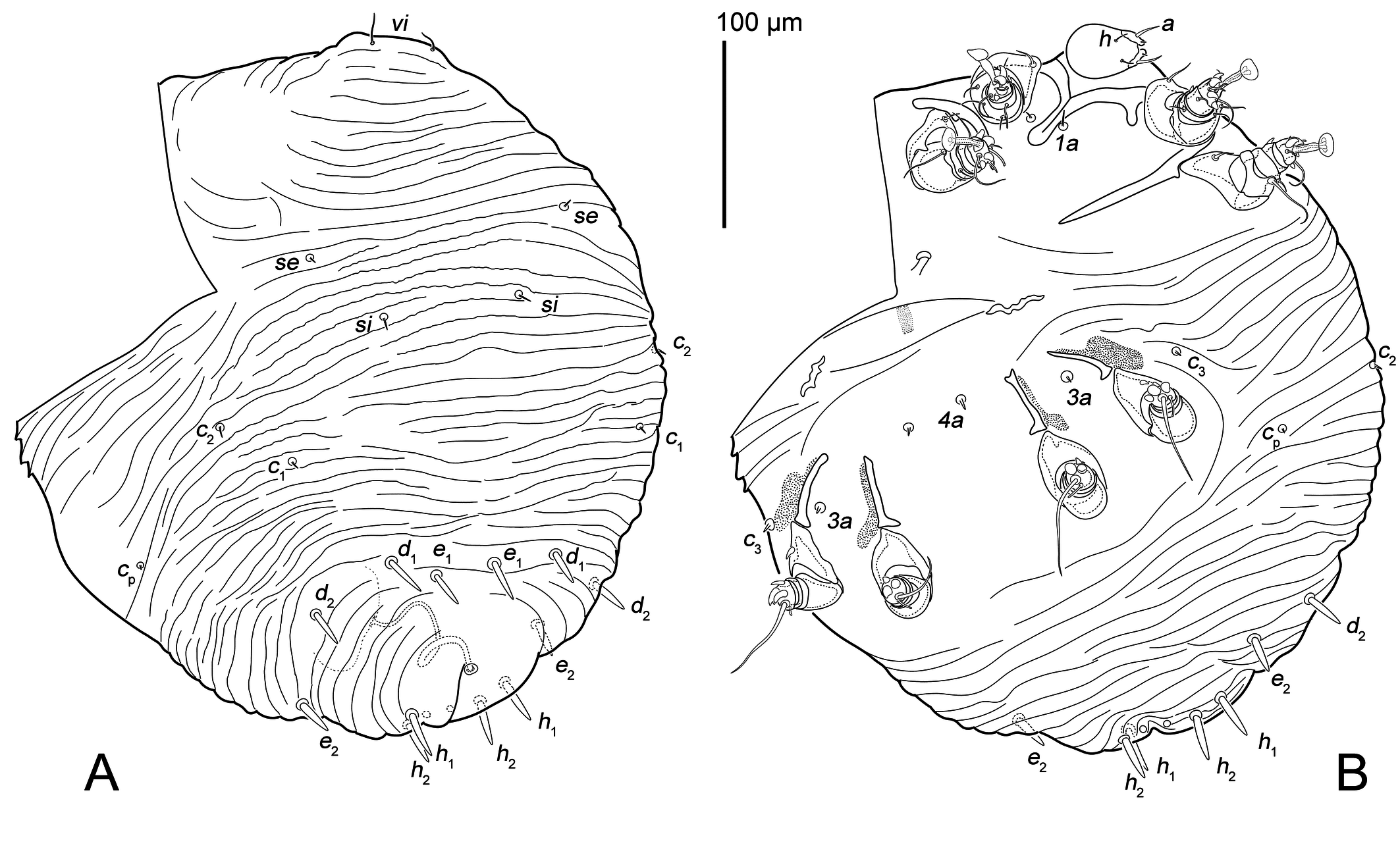




Gnathosoma (Figure 1B) short (48 × 30). Subcapitular seta h (6) thin, filiform. Palpal seta a (11) with inflated base (2 wide).
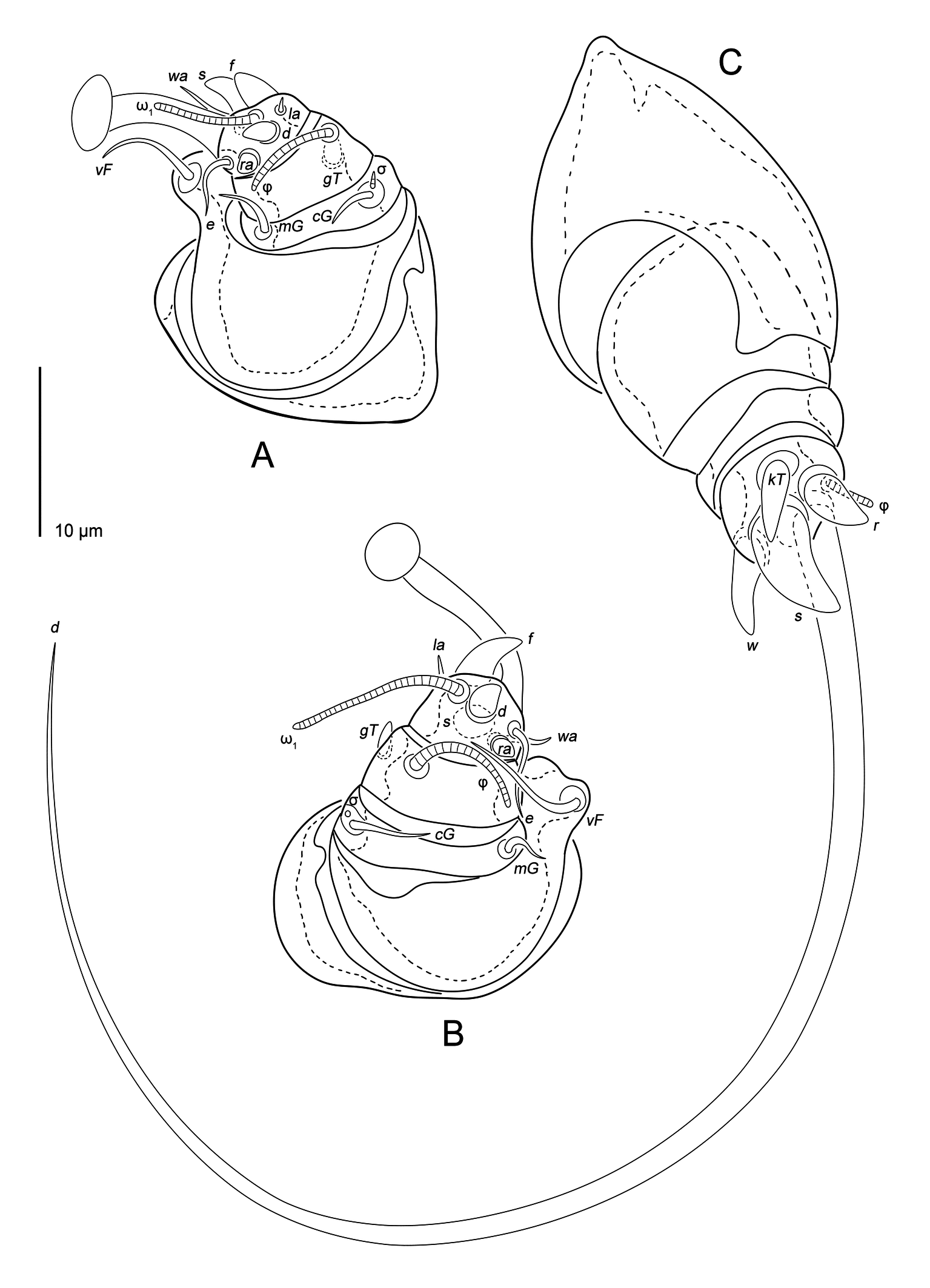
Dorsal surface completely striated transversely except for glabrous, oval area around the anus, dorsal striae very irregular, often interrupted (Figures 1A, 5A, 6D), with 17 striae between setal pairs si and setae e1. Prodorsal shield absent. Dorsum with 12 pairs of setae: vi (13), se (6), si (7), c1 (5), c2 (6), cp (5), d1 (22), d2 (19), e1 (20), e2 (19), h1 (22) and h2 (23). Seta vi filiform, slightly widened at base. Setae se, si, c1, c2 and cp well developed short spine (Figure 6D). Setae d1, d2, e1, e2, h1, h2 spine-like, with pointened tip (Figures 6B, C). Anus dorsally. External copulatory opening above anus. Spermatheca rounded, without formed spermathecal head, secondary spermaducts indistinct. Primary spermaduct long, narrowed gradually towards spermatheca (Figure 6A).
Vental idiosoma striated in posterior part and between coxal apodemes II and III (Figures 1B, 5B). Coxal apodemes I (70), their apices completely merge along half their length to form a Y-shape (the apex is slightly expanded) (Figure 1B). Coxal apodemes II (64) straight (Figure 1B). Coxal apodemes III (37) and IV (36) not fused, with elongated sclerotized area with expansion in the basal part (Figs 6E, F). Four pairs of ventral idiosomal setae 1a (9), 3a (5), 4a (5), c3 (5) shaped as well developed short spine.
Legs (Figures 2) short: I and II about 60, III and IV about 50. All segments of legs I and II free, legs III and IV with merged tibia and tarsus. All segments without cuticular ridges and denticles. Tarsi I and II with ambulacra (23 and 22 long, respectively), tarsi III and IV without ambulacra. Trochanters I and II with setae pR (17 and 12 long, respectively) with inflated base. Trochanter III with seta sR (7) spiniform. Trochanter IV without seta. Femora I and II with filiform setae vF (14 and 25, respectively), placed on small protrusions. Femora III and IV without seta. Genua I and II with filiform setae cG (8 and 5, respectively) and mG (10 and 8, respectively). Genua III and IV without setae. Tibiae I and II with spiniform gT (4 and 6, respectively). Tarsi I and II with filiform setae e (20), wa (10 and 8, respectively) and la (3); spiniform setae d (8), f (9 and 10, respectively), ra (4) and s (8). Tibiotarsus III with long and thick seta d (50) and spiniform setae kT (7), w (8), s (11) and r (9). Tibiotarsus IV with long and thick seta d (40) and spiniform setae kT (4), w (8) and r (9). Solenidia ω1 (9) and ω3 (10) on tarsus I and ω1 (10) on tarsus II bacilliforms. Solenidia φ I(10), φII (8), φIII (6) and φIV (8) bacilliforms. Solenidion σI short (2), σII minute.
Larva — (Figures 3, 4, 5F). Idiosoma rounded (Figures 3, 5F), 113 × 91 (n=1).
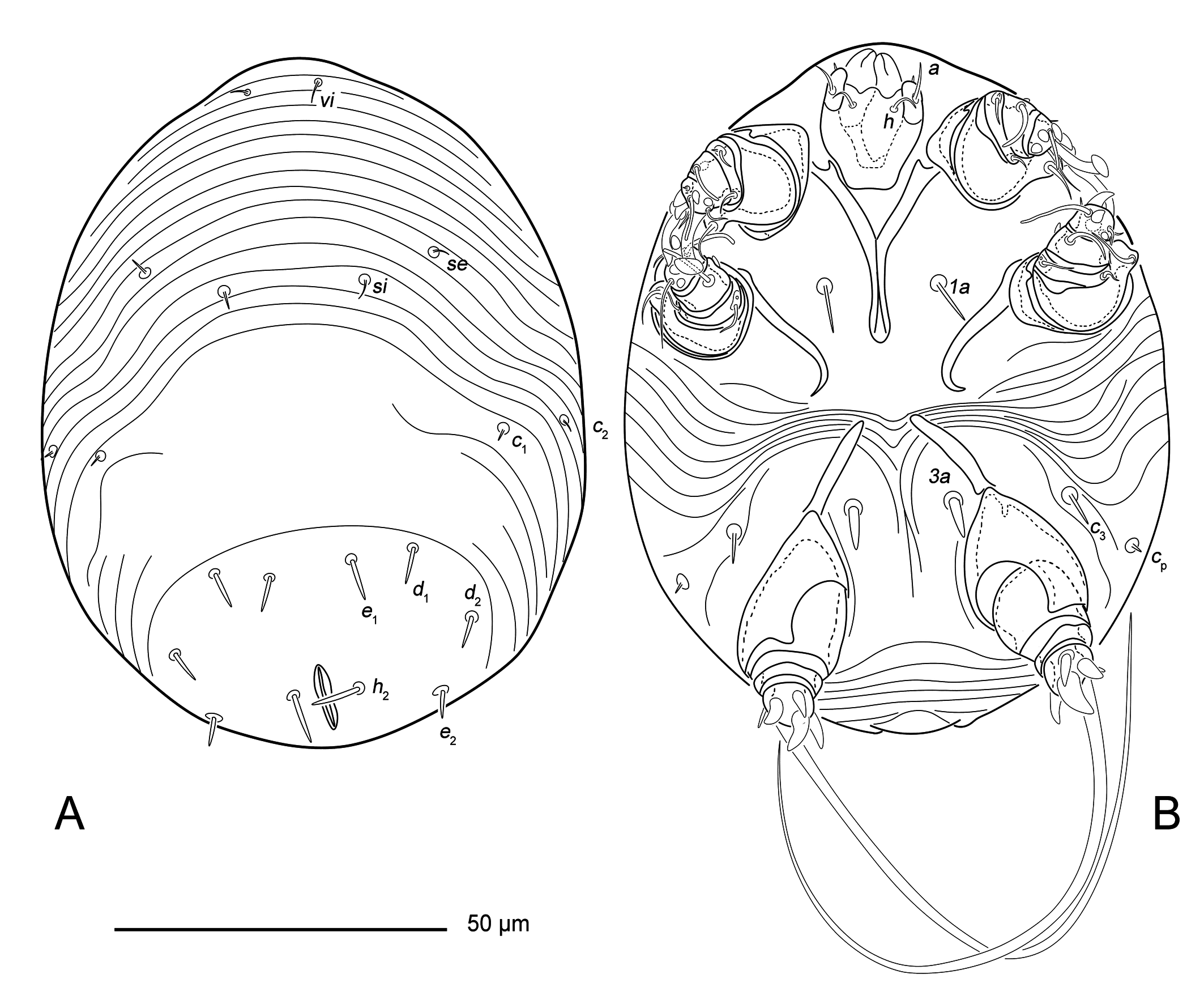




Gnathosoma short (18 × 15). Subcapitular seta h (5) thin, filiform. Palpal seta a (6) with slightly inflated base.
Dorsal surface striated transversely except for large glabrous, oval area around anus, dorsal striae in the anterior half of the idiosoma are not interrupted (Figure 4A). Prodorsal shield absent. Dorsum with 11 pairs setae: vi (4), se (3), si (3), c1 (2), c2 (2), cp (2), d1 (6), d2 (5), e1 (6), e2 (4) and h1 (7). Seta vi filiform. Setae se, si, c1, c2 and cp well developed short spine. Setae d1, d2, e1, e2, h2 spine-like, with pointed tip. Anus situated dorsally.
Vental idiosoma striated in posterior part and between coxal apodemes II and III (Figures 4B, 5C). Coxal apodemes I (30), their apices completely merge along half their length to form a Y-shape (the apex is slightly expanded) (Figures 4B, 5C). Coxal apodemes II (20) with inner tips strongly curved (Figures 4B, 5C). Coxal apodemes III (15) without sclerotization area (Figures 4B, 5C). Four pairs of ventral idiosomal setae 1a (7), 3a (6) and c3 (5), 1a and c3 shaped as well developed thin spine, setae 3a spine-like, with a pointed tip.
Legs (Figures 4) short: I, II about 20, III 30. All segments of legs I and II free, leg III with merged tibia and tarsus. All segments without cuticular ridges and teeth. Tarsi I and II with ambulacra (10 and 16 length, respectively), tarsus III without ambulacrum. All trochanters without setae. Femur I and II with filiform setae vF (7 and 8, respectively), placed on gross protrusions. Genua I and II with filiform setae cG (3 and 5, respectively) and mG (4). Genua III without setae. Tibia I and II with spiniform gT (2). Tarsi I and II with filiform setae e (4 and 5, respectively), wa (4) and la (2); spiniform setae d (2), f (3 and 4, respectively), ra (2) and s (4 and 3, respectively). Tibiotarsus III with long and thick seta d (11) and spiniform setae kT (5), w (6), s (8) and r (5). Solenidion ω1 (7) on tarsus I and ω1 (10) on tarsus II strick-like. Solenidia φI (6), φII (7) and φIII (4) strick-like. Solenidion σI short (1), σ II minute.
Male, protonymph and tritonymph unknown. The eggs are shown in Figures 5D and E.




Localisation
On body skin of Myotis ikonnikovi.
Etymology
The new species is named after Alina Kharlamova, a family friend of the senior author.
Remarks
The female was slightly damaged (the idiosoma was ruptured) and the gnathosoma was slightly displaced. However, these deformations did not affect taxonomically important features. The larval description is based on specimens that had not yet hatched, but all structures were clearly visible.
The new species, N. (N.) alinae sp. n., belongs to a group of four Notoedres species associated with bats of the genus Myotis: N. (N.) nigricans Klompen, 1992, N. (N.) myoticola, N. (N.) myotis (Hedeen, 1953) and N. (N.) roesleri Vitzthum, 1932. All five species share the following characteristics: setae d1, e1, h1 are spine-like with a pointed tip, setae si, c1 and c2 are well developed short spines, setae 4a are present; base of palpal seta a is inflated, and seta sR III are spiniform.
Download as
Characters
N. alinae sp. n.
N. myotis
N. nigricans
N. myoticola
N. roesleri
Widthof setae e2
2.5–3
1.1–1.2
1.7–2.3
2.0–2.5
1,4–1.6
Ratio of setae e2 / h1, h2
4/5 length, (19–20 / 22–23)
1/4 length, (5–8 / 25–31)
1/2 length, (12–17 / 25–31)
1/2 length, (11–18 / 27–39)
1/3-1/2 length, (10–12 / 25–30)
Width of palpal seta a
2
3.8–5.0
3.7–4.2
2.3–2.9
3.4–4.1
Length of setae si, c1 and c2
5–7
4–8
10–19
5–15
5–9
Setae cG I-II
filiform
spiniform
spiniform
spiniform
spiniform
Length of Setae vi length
13
6–8
5–7
5–9
3–6
A set of differential characteristics that allows distinguishing all five Notoedres species from each other is given in Table 1. Data on four previously known species are from Klompen (1992: Table 14 and the key to Notoedres species, p. 145). The new species differs from all four closely related species in having longer setae vi (13), the length ratio of setae e2 / h1 and h2 about 4/5, and genual setae cGI, II setiform. In the compared Notoedres species, setae vi do not exceed 10, the ratio of setae e2 / h1 and h2 is within the limits 1/4–1/2, and genual setae cGI, II are spiniform.
Acknowledgements
The research by Vasiliy B. Kolesnikov was conducted in the framework of the State Assignment No. 124032500016-4 (Ministry of Science and Higher Education of the Russian Federation).
References
- Ah H-S. 1975. Notoedres (Bakeracarus) coreanus sp. n. (Acari: Sarcoptidae), a new intranasal sarcoptid mite from Pipistrellus savii (Ognev) from Korea. Journal of Parasitology, 61(4): 722-724. https://doi.org/10.2307/3279473
- Andrews J. R. N. 1983. The origin and evolution of host association of Sarcoptes scabiei and subfamily Sarcoptinae murray. Acarologia. 24(1): 85-94.
- Ansari S., Dabas R., Rajpurohit V., Yadav A., Dehru S. 2024. Notoedres cati infestation in a cat and its management: a case report. Uttar Pradesh Journal of Zoology, 45(17): 297-300. https://doi.org/10.56557/upjoz/2024/v45i174372
- Arnaout Y., Djelouadji Z., Robardet E., Cappelle J., Cliquet F., Touzalin F., Jimenez G., Hurstel S., Borel C., Picard-Meyer E. et al. 2022. Genetic identification of bat species for pathogen surveillance across France. PLoS ONE, 17: e026134 Baker A.S., Craven J.C. 2003. Checklist of the mites (Arachnida: Acari) associated with bats (Mammalia: Chiroptera) in the British Isles. Systematic & Applied Acarology, Vol 14: 1-20. https://doi.org/10.1371/journal.pone.0261344
- Bochkov A. V. 2010. A review of mammal-associated Psoroptidia (Acariformes: Astigmata). Acarina, 18 (2): 99-260.
- Borissenko A.V. 1999. [A mobile trap for capturing bats in flight.] Plecotus et al., 2: 10-19. [in Russian]
- Ditz C., Von Helversen O., Nill D. 2009. Bats of Britain, Europe & Northwest Africa. Black Publishers Ltd., London, 400 pp.
- Fain A., V. Aellen. 1961. Les acariens psoriques parasites des chauves souris. XX. Un cas d'hyperparasitisme par Nycteridocoptes poppei. Nouvelles observations snr l'évolution cyclique de la gale sarcoptique chez les chiroptkres. Revue suisse de Zoologie, 68(31): 305-309. https://doi.org/10.5962/bhl.part.75087
- Fain A. 1978. Epidemiological problems of scabies. International Journal of Dermatology, 7(1): 20-30. https://doi.org/10.1111/j.1365-4362.1978.tb06040.x
- Grandjean F. 1939. La chaetotaxie des pattes chez les Acaridiae. Bulletin de la Société Zoologique de France, 64: 50-60.
- Griffiths D. A., Atyeo W. T., Norton R. A., Lynch C. A. 1990. The idiosomal chaetotaxy of astigmatid mites. Journal of Zoology (London), 220: 1-32. https://doi.org/10.1111/j.1469-7998.1990.tb04291.x
- Hedeen R.A. 1953. A new species of Sarcoptes (Acarina: Sarcoptidae) from the cave bat. Journal of Parasitology, 39(1-2): 334-335. https://doi.org/10.2307/3273959
- Klimov P.B., He Q. 2024. Predicting host range expansion in parasitic mites using a global mammalian-acarine dataset. Nature Communications, 15: 5431. https://doi.org/10.1038/s41467-024-49515-3
- Klimov P.B., Kolesnikov V.B., Oconnor B.M. 2026. Phylogeny and systematics of Astigmata with description of Lycoglyphidae fam. nov. and a comprehensive identification key to Acaroidea. Taxonomy, 6: 18. https://doi.org/10.3390/taxonomy6010018
- Klompen J.S.H. 1992. Phylogenetic relationships in the mite family Sarcoptidae (Acari: Astigmata). Miscellaneous Publications, Museum of Zoology, University of Michigan, 180: 1-154.
- Lange A.B., Sokolova T.V. 1992. Parasitism of the itch mite Sarcoptes scabiei (Acariformes: Sarcoptidae). Parasitologia, 26(4): 281-295
- Norton R. 1998. Morphological evidence for the evolutionary origin of Astigmata (Acari: Acariformes). Experimental and Applied Acarology, 22: 559-594. https://doi.org/10.1023/A:1006135509248
- OConnor B.M. 2009. Cohort Astigmatina. In A Manual of Acarology, 3rd ed.; Krantz, G.W., Walter, D.E., Eds.; Texas Tech University Press: Lubbock, TX, USA, pp. 565-657.
- Orlova M.V., Zhigalin A.V., Orlov, O.L. 2014. New records of ectoparasites of the eastern water bat Myotis petax Hollister, 1912 (Vespertilionidae, Chiroptera) and the revision of the material previously collected from Myotis daubentonii s. lato in the Eastern Palaearctic. Entomological Review, 94: 1306-1312. https://doi.org/10.1134/S0013873814090115
- Orlova M.V., Zhigalin A.V. 2015. Three new bat ectoparasite species of the genus Macronyssus from Western Siberia (with an identification key for females of the genus Macronyssus from the Palearctic boreal zone). Journal of Parasitology, 101: 314-319. https://doi.org/10.1645/14-609.1
- Orlova M.V., Zhigalin A.V., Khritankov A. M. 2015. New findings of bat ectoparasites (Chiroptera: Vespertilionidae) in Southern Siberia. Entomological Review, 95 (5), 681-686. https://doi.org/10.1134/S0013873815050127
- Orlova M.V., Kazakov D.V. Kravchenko L.B., Zhigalin A.V. 2017. Ectoparasite fauna of the Siberian bat Myotis sibiricus (Chiroptera: Vespertilionidae) with a revision of previous data on ectoparasites from Brandt's bat Myotis brandtii s. l. and the whiskered bat M. mystacinus s. l. of the Eastern Palaearctic. Entomological Review, 97: 1166-1173. https://doi.org/10.1134/S0013873817080164
- Orlova M.V., Klimov P.B., Moskvitina N.S., Orlov O.L. Zhigalin A.V., Smirnov D. G., Dzhamirzoyev H.S., Vekhnik V.P., Pavlov A.V., Emelyanova A.A., Khristenko E. 2021. New records of bat flies (Diptera: Nycteribiidae), with an updated checklist of the nycteribiids of Russia. Zootaxa, 4927: 410-430. https://doi.org/10.11646/zootaxa.4927.3.5
- Roesler R. 1932. Über eine durch Notoedres-Milben erzeugte Gallbildung (Thylacium) an einer Fledermaus. Zeitschrift für Parasitenkunde, 4(3): 407-408. https://doi.org/10.1007/BF02119992
- Simmons N. B., Cirranello A. L. 2024.. Bat species of the world: a taxonomicand geographic database. Available from URL: https://www.batnames.org/. Stanyukovich М.К. 1995. Spinturnix bregetovae sp. n. (Gamasina: Spinturnicidae), a new species of parasitic mites from bats. Acarina, 3(1-2): 105-122.
- Wilson D. E., Reeder D. M. 2005. Mammal Species of the World. A Taxonomic and Geographic Reference. 3rd ed. The Johns Hopkins University Press, Baltimore, USA. 2142 pp.
- Wilson D.E., Mittermeier R.A. 2019. Handbook of the Mammals of the World. Vol. 9. Bats. Lynx Edicions, Barcelona, 998 pp.



2025-11-12
Date accepted:
2026-04-07
Date published:
2026-04-09
Edited by:
Pfingstl, Tobias

This work is licensed under a Creative Commons Attribution 4.0 International License
2026 Kolesnikov, Vasiliy B. and Orlova, Maria V.
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



