The natural history of the sea snake tick Amblyomma nitidum Hirst and Hirst, 1910 (Ixodidae: Amblyomminae) and its first definitive country record in the Philippines
Amarga, Ace Kevin S.  1
; Robbins, Richard G.
1
; Robbins, Richard G.  2
and Chiu, Ming-Chung
2
and Chiu, Ming-Chung  3
3
1✉ Department of Entomology, National Taiwan University, Taipei 106, Taiwan & International Union for Conservation of Nature Species Survival Commission (IUCN SSC) Parasite Specialist Group & Department of Biology, College of Science, De La Salle University, Taft Avenue, Manila 1004, Philippines.
2Department of Entomology, National Museum of Natural History, Smithsonian Institution, Washington, DC 20560, U.S.A.
3✉ Department of Entomology, National Taiwan University, Taipei 106, Taiwan.
2026 - Volume: 66 Issue: 1 pages: 271-281
https://doi.org/10.24349/cn91-ic1rOriginal research
Keywords
Abstract
Introduction
Sea snakes (Reptilia: Elapidae) inhabit marine ecosystems ranging from mangrove swamps to the shallow regions of the epipelagic zone. These snakes are classified in two subfamilies: Hydrophiinae, which include all ''true'' sea snakes, and Laticaudinae, which include only sea kraits (Rasmussen et al. 2011). Most sea snakes are venomous (Mackessy & Tu, 1993), with the exception of three species in the genus Emydocephalus Krefft, 1869, which are oophagous, exhibiting greatly reduced dentition and a vestigial venom gland (Shine et al. 2004). Among the sea snake genera, only members of the amphibious genus Laticauda Laurenti, 1768 (sea kraits) are able to enter littoral zones, as evidenced by their retention of large ventral scales that aid in navigating terrestrial environments (Bonnet et al. 2005). Sea kraits are widely distributed, ranging from the eastern Indian Ocean to the western Pacific and extending to subtropical coastal waters of Australasia (Heatwole et al. 2005). Of the eight recognized extant species of sea kraits (Uetz et al. 2025), three are known to occur within the tropical waters of the Philippine archipelago: L. colubrina (Schneider, 1799), L. laticaudata (Linnaeus, 1758), and L. semifasciata (Reinwardt, 1837) (Leviton et al. 2018; Weinell et al. 2019).
Despite its wide geographic range, the genus Laticauda is relatively understudied in terms of its associated endo- and ectoparasites (Amarga et al. 2025b). Parasitic taxa reported on sea kraits include some species of haemogregarines (Qiu et al. 2021b), trematodes (Yamaguti 1933; Coil & Kuntz 1960; Choe et al. 2020), nematodes (Schmidt & Kuntz 1973; Chinnadurai et al. 2008; Moravec & Justine 2019), and a few specialized trombiculids (Southcott 1957; Takahashi et al. 2012). Interestingly, there is only one species of tick known to occur on sea kraits. Amblyomma nitidum Hirst and Hirst, 1910 is a seldom-collected tick exclusively ectoparasitic on members of the genus Laticauda. Due to its rarity, there are few published geographic records of A. nitidum. In the Philippines, A. nitidum has never been formally reported, and it is absent from Philippine tick faunistic accounts, including the works of Kohls (1950) and Velasquez & Eduardo (1994). Here, we present the first definitive record of A. nitidum in the Philippines. Additionally, we provide an updated summary of geographic records for A. nitidum as well as an overview of its natural history.
Material and methods
During July 2025, while conducting research as a visiting scientist at the American Museum of Natural History, the first author examined several species of Amblyomma Koch, 1844. Among these, a single specimen of A. nitidum was found and photographed under a Leica MZ12 stereomicroscope. Names of host species follow Uetz et al. (2025). Additional specimens from different institutions were examined as supplementary material. Abbreviations used in this study are as follows: AMNH (American Museum of Natural History, New York, New York, U.S.A.), NMNS (National Museum of Natural Science, Taichung, Taiwan), NTNU (National Taiwan Normal University, Taipei, Taiwan), and RML (Rocky Mountain Laboratories, now U.S. National Tick Collection, Statesboro, Georgia, U.S.A).
Results and discussion
Amblyomma nitidum Hirst and Hirst, 1910
Amblyomma nitidum Hirst and Hirst, 1910: 304. Type locality: Solomon Islands. Type host: Unspecified sea snake. Repository: Natural History Museum, London.
Amblyomma laticaudae Warburton, 1933: 558.
Material examined
PHILIPPINES — On Laticauda colubrina: 1♂ (RML 65917), Gigantes, Iloilo, IX.1975, leg. S.A. Minton (Figures 1a-b).



The AMNH specimen was diagnosed as a male A. nitidum by the following combination of morphological characters: scutum with shallow punctations; scutal ornamentation absent; cervical grooves short and convergent; festoons present, prominent and rectangular in outline; sclerotized plates absent on festoon posterior margins; eyes present, slightly bulging and situated on anterior lateral margins of idiosoma; genital aperture situated between coxae II; spiracular plates oval; palpi elongate, article II significantly longer than article III; hypostome denticulate with 4/4 dental formula; coxa I bearing two minute triangular spurs, external spur slightly larger than internal spur; coxae II-IV each bearing a single minute triangular spur; trochanteral spur absent.
TAIWAN — On L. laticaudata: 1♀ (NMNS), Gongliao District, New Taipei City (25.114193°N, 121.920320°E), 10.VI.2024, leg. C.-W. Chen; 4♂, 1♀, 1N (NTNU), Sanxiantai, 21.VII.2019, leg. local collector; on Laticauda sp.: 1♂ (NTNU), Sanxiantai, leg. local collector.
Taxonomic status
Amblyomma nitidum was first described in 1910 based on two specimens (a male and a female) collected on an unspecified sea snake in the Solomon Islands by Vidal Sharpe, an officer of the Royal Navy (Hirst & Hirst 1910). In the original description, no collection date was provided, but most probably the type specimens were collected during the British occupation of the Solomon Islands from 1893–1978. Nearly two decades after its original description, Mohammad Sharif, an Indian entomologist, provided the first description of the larval and nymphal stages of A. nitidum based on specimens collected at Port Blair (now Sri Vijaya Puram), Andaman Islands, on L. laticaudata (Sharif, 1928). In 1933, the British acarologist Cecil Warburton described A. laticaudae as a new species based on three specimens (1 male, 1 female, and 1 nymph) collected on a captive L. colubrina in Singapore (Warburton 1933). Although some authors have listed A. laticaudae as a valid species (e.g., Neveu-Lemaire et al. 1934; Rageau 1960; Camicas et al. 1998), Audy et al. (1960) regarded A. laticaudae as a synonym of A. nitidum, and this change has been accepted by several authors, including Voltzit & Keirans (2002), Kolonin (2009), Guglielmone et al. (2010, 2014), and Robbins et al. (2025).
Geographic distribution
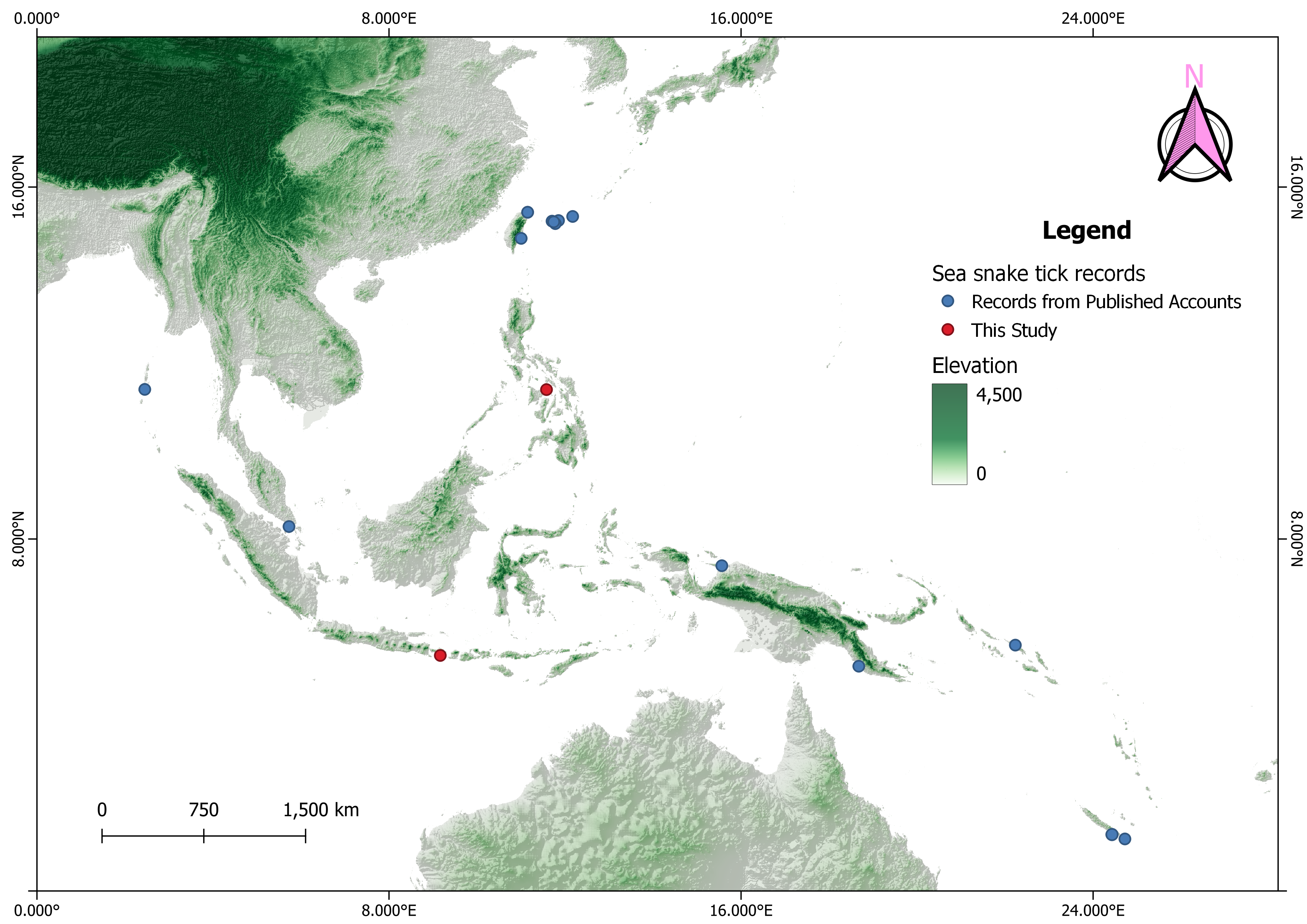
Despite the wide distribution of sea kraits within the tropical waters of the Oriental realm and Australasia (Heatwole et al. 2005), there are relatively few published reports of A. nitidum in the acarological literature. The westernmost record is that of Sharif (1928) from the Andaman Islands (now part of the Union Territory of India), and there are no published accounts of this species from the Indian coastline facing the Bay of Bengal despite the presence of sea kraits there (i.e., L. colubrina and L. laticaudata), as recorded by Ganesh et al. (2019). The northernmost record of A. nitidum is from Japan's Ryukyu archipelago, where it was first reported by Yamaguti et al. (1971) based on two female specimens collected in 1962 on L. semifasciata from Ishigaki Island. Subsequent published collections of A. nitidum in the Ryukyu archipelago include Hayashi & Masunaga (2001), Takano et al. (2014), and Qiu et al. (2021a, b). In addition, A. nitidum was recently reported from Taiwan by Kwak et al. (2020) and Amarga et al. (2025b). In mainland Southeast Asia, the only published record of A. nitidum is that of Audy et al. (1960) from Singapore, based on specimens collected from L. colubrina. However, it is expected that future collections of A. nitidum will be made in other mainland Southeast Asian countries, particularly along the rocky shorelines of Myanmar (Burmese coastlines facing the Bay of Bengal and Andaman Sea) and Thailand (Thai coastlines facing the Andaman Sea and Gulf of Thailand). Sea kraits, particularly L. colubrina and L. laticaudata, are known to occur in these countries based on the accounts of Chanhome et al. (1998), Leviton et al. (2003), and Das (2012).


In the Philippine archipelago, to date there have been no published accounts of A. nitidum, which is absent from Philippine tick faunistic records, including Kohls (1950) and Velasquez & Eduardo (1994), as well as the global study of ixodid tick distribution by Guglielmone et al. (2023). Here we provide the first definitive record of A. nitidum in the Philippines, based on a specimen collected on L. colubrina in the Gigantes Islands, a small island chain in the Visayan Sea under the jurisdiction of Iloilo Province (Figure 2). The Gigantes Islands are predominantly characterized by a karst landscape with patches of limestone vegetation. Additionally, these islands feature several limestone cliffs and lagoons that are suitable habitats for sea kraits (Figure 3). Since Laticauda species are widespread across the Philippine archipelago (Leviton et al. 2018), we expect that A. nitidum will eventually be found in coastal waters, along rocky shores, and in shoreline limestone caves of other Philippine islands, including Bohol, Cebu, Luzon, Marinduque, Mindanao, and Samar.



In Indonesia, only a single record of A. nitidum has been published to date. Wilson (1970) collected A. nitidum on L. laticaudata at Japen Island (now Yapen), West Papua (formerly West Irian or Irian Jaya Barat). Additionally, utilizing a citizen science platform, we here present a record for Indonesia west of Wallace's Line, an account based on a retrieved field record in iNaturalist from Kesiman, East Denpasar, Bali, observed in 2022.
In the Australasian region, A. nitidum has been reported from New Caledonia, Papua New Guinea, and the Solomon Islands (Hirst & Hirst 1910; Rageau 1960, 1967; Guglielmone et al. 2023). Despite confirmed accounts of sea kraits in New Zealand (Gill 1997; Gill & Whitaker 2014), A. nitidum has never been documented there. In summary, there are currently nine countries or territories with confirmed, published records of A. nitidum: India, Indonesia, Japan (Ryukyu archipelago), New Caledonia, Papua New Guinea, the Philippines, Singapore, Solomon Islands, and Taiwan (Figure 4).


Host associations
Since its original description in 1910, A. nitidum has been reported on three Laticauda species: L. colubrina (Yellow-lipped sea krait), L. laticaudata (Blue-lipped sea krait), and L. semifasciata (Black-banded sea krait) (Table 1). To date, it has not been recorded on other sea krait species: L. frontalis (De Vis, 1905) (Dwarf sea krait), L. guineai Heatwole, Busack & Cogger, 2005 (Papuan sea krait), L. saintgironsi Cogger & Heatwole, 2006 (New Caledonian sea krait), and L. schistorhyncha (Günther, 1874) (Katuali). All feeding stages of A. nitidum (i.e., larvae, nymphs, and adults) may be found on sea krait hosts.
Download as Note: ª A field observation record retrieved on 16 July 2025 from iNaturalist authored by username “repski” [Bartosz Bajger] and documented on 16.XII.2022 from Kesiman, East Denpasar, Bali, Indonesia.
Country
Host(s)
References
India
L. laticaudata
Sharif 1928
Indonesia
L. laticaudata; Unspecified Laticauda
Wilson 1970; iNaturalist (2025)ª
Japan (Ryukyu archipelago)
L. colubrina; L. laticaudata; L. semifasciata
Yamaguti et al. 1971; Hayashi & Masunaga 2001; Qui et al. 2021a,b
New Caledonia
L. colubrina
Rageau 1960, 1967
Papua New Guinea
n/a
Guglielmone et al. 2023
Philippines
L. colubrina
This study
Singapore
L. colubrina
Audy et al. 1960
Solomon Islands
Unspecified sea snake
Hirst & Hirst 1910
Taiwan
L. colubrina, L. laticaudata
Kwak et al. 2020; Amarga et al. 2025b
Ecology
Ticks have evolved as ectoparasites of vertebrates, and no tick species are known to parasitize vertebrates inhabiting exclusively marine environments (e.g., whales, dolphins, and saltwater fishes). However, a few tick species are known to parasitize semi-marine vertebrates. These include A. nitidum (Amarga et al. 2025b); A. darwini Hirst and Hirst, 1910 and A. williamsi Banks, 1924, which parasitize marine iguanas (genus Amblyrhynchus Bell, 1825) (Wikelski 1999); Ixodes uriae White, 1852, which feeds on king penguins (Aptenodytes patagonicus J. F. Miller, 1778) (Mangin et al. 2003); and I. jacksoni Hoogstraal, 1967, which parasitizes the spotted shag (Phalacrocorax punctatus (Sparrman, 1786)) (Hoogstraal 1967).
Compared to ''true'' sea snakes (members of subfamily Hydrophiinae), which are fully aquatic and viviparous, sea kraits tend to move inland to rest and breed. When in the littoral zone, especially on rocky shorelines, sea kraits engage in gregarious mating behavior that provides opportunities for A. nitidum to access hosts (Audy et al. 1960; Kwak et al. 2020). Apart from rocky shorelines, sea krait mating groups can also be observed in hollow driftwood and mangrove tree holes (Audy et al. 1960; Tyabji et al. 2018). Additionally, in karst shoreline landscapes, sea kraits tend to utilize tidal caves as communal nesting sites (Bonnet et al. 2014). Such large host aggregations in a limited area help to ensure the success of host questing by A. nitidum (Figure 5).



On hosts, A. nitidum is usually observed attached between scales and is rarely encountered in large numbers on individual snakes. Additionally, this tick may attach at sites of traumatic injury (see Fig. 3A in Amarga et al. 2025b), a behavior that has also been observed in Amblyomma cordiferum Neumann, 1899, a reptile tick chiefly associated with terrestrial snakes (Amarga et al. 2024).
Several aspects of A. nitidum ecology and physiology are in need of further investigation. Since A. nitidum has co-evolved as an ectoparasite of semi-marine sea snakes, it may possess physiological mechanisms for adjusting to the salinity of sea water and perhaps retaining oxygen while its hosts are submerged and foraging for long periods of time. This tick may also be able to chemically track its hosts and adapt its life cycle to a challenging environment.
Phylogeny
In the phylogenetic assessment by Uribe et al. (2024), A. nitidum was recovered as a sister group to the clade giving rise to A. breviscutatum Neumann, 1899 and A. geoemydae (Cantor, 1847). The former is predominantly a parasite of mammals (Voltzit & Keirans 2002), whereas the latter is a reptile tick primarily associated with Oriental freshwater turtles and tortoises (Amarga et al. 2022, 2025a). However, the origin of host specialization by A. nitidum and the evolutionary scenario that drove it remain unknown. The genetic structure of A. nitidum populations also merits investigation across this tick's vast range because sea kraits are not constant transoceanic migrants across different countries and they also exhibit strong philopatric and homing behavior, indicating high site fidelity (Lane & Shine 2011).
Associated microbiota
Tick-borne microbes associated with A. nitidum have been under-examined. Qui et al. (2021a) reported the first microbes from this species based on tick specimens collected on sea kraits in Iriomote Island, Japan. These workers detected putative novel strains of the genera Rickettsia and Ehrlichia; both are groups of obligate intracellular alphaproteobacteria that are difficult to culture in laboratory settings. Subsequently, Qui et al. (2021b) detected an undescribed Hepatozoon in A. nitidum from Iriomote Island that is phylogenetically related to H. colubri, a species primarily associated with snakes.
Apart from Japan, no other published accounts describing microbiota associated with A. nitidum have appeared to date. It would be instructive to examine the genetic diversity of these microorganisms and their variation across this tick's vast geographic range, as this has not yet been attempted. Also, it would be worth examining the degree of pathogenicity of these tick-borne microbes and their impact on sea kraits, as well as the transmission risk of these microorganisms and their potential for cross-host infection.
Acknowledgements
The authors thank the following colleagues: Dr. Pio Colmenares and Dr. Lorenzo Perdini (American Museum of Natural History) for providing access and laboratory space; Dr. Jessica Ware and Erin Krichilsky for hosting the first author during his stay at the AMNH as a visiting researcher; Dr. Cristy Acabado (University of the Philippines Visayas) for providing the Gigantes landscape photo (Fig. 3); Marvin Jay Sarmiento (University of the Philippines Los Baños) for providing the photo of L. colubrina (Fig. 5); and Andrie Bon Flores (Academia Sinica, Taiwan) for aid in creating the tick locality map (Fig. 4).
This research was funded by a grant from the National Science and Technology Council, Taiwan (113-2621-B-002-002-MY3) and Ministry of Agriculture, Taiwan (114AS-1.3.2-AS-27) to Dr. Ming-Chung Chiu.
References
- Amarga A.K.S., Fu Y.-S., Chiu Y.-S. & Fang W.-H. 2024. Elaphe carinata (Taiwan stink snake). Ectoparasites. Herpetological Review 55(1): 111-112.
- Amarga A.K.S., Robbins R.G., Tseng H.-Y. & Lin S.-M. 2025a. New host and geographic records of Amblyomma geoemydae (Cantor) (Ixodidae: Amblyomminae) from southern China and Southeast Asia. Systematic & Applied Acarology 30(1): 7-12. https://doi.org/10.11158/saa.30.1.2
- Amarga A.K.S., Robbins R.G., Chen C.-W. & Lin S.-M. 2025b. First record of the ixodid tick Amblyomma nitidum on Laticauda laticaudata (Serpentes: Elapidae) in Taiwan. Phyllomedusa 24(1): 125-130. https://doi.org/10.11606/issn.2316-9079.v24i1p125-130
- Amarga A.K.S., Supsup C.E., Tseng H.-Y., Kwak M.L. & Lin S.-M. 2022. The Asian turtle tick Amblyomma geoemydae Cantor, 1847 (Acari: Ixodidae) in the Philippines: first confirmed local host and locality with a complete host index. Ticks and Tick-borne Diseases 13(4): 101958. https://doi.org/10.1016/j.ttbdis.2022.101958
- Audy JR, Nadchatram M, Lim B.-L. 1960. Malaysian parasites. XLIX. Host distribution of Malayan ticks (Ixodoidea). Studies from the Institute for Medical Research, Malaya 29: 225-246.
- Bonnet X., Ineich I., Shine R. 2005. Terrestrial locomotion in sea snakes: the effects of sex and species on cliff-climbing ability in sea kraits (Serpentes, Elapidae, Laticauda). Biological Journal of the Linnean Society 85: 433-441. https://doi.org/10.1111/j.1095-8312.2005.00505.x
- Bonnet X., Brischoux F., Bonnet C., Plichon P. & Fauvel T. 2014. Coastal nurseries and their importance for conservation of sea kraits. PLoS ONE 9(3): e90246. https://doi.org/10.1371/journal.pone.0090246
- Camicas J.L., Hervy J.P., Adam F., Morel P.C. 1998. Les tiques du monde. Nomenclature, stades décrits, hôtes, repartition (Acarida, Ixodida). Orstom, Paris, 233 pp.
- Chanhome L., Cox M.J., Wilde H., Jintakoon P., Chaiyabutr N., Sitprija V. 1998. Venomous snakebite in Thailand I: Medically important snakes. Military Medicine 163: 310-317. https://doi.org/10.1093/milmed/163.5.310
- Chinnadurai S.K., Brown D.L., Van Wettere A., Tuttle A.D., Fatzinger M.H., Linder K.E., Harms C.A. 2008. Mortalities associated with sepsis, parasitism, and disseminated round cell neoplasia on yellow-lipped sea kraits (Laticauda colubrina). Journal of Zoo and Wildlife Medicine 39: 626-630. https://doi.org/10.1638/2008-0018.1
- Choe S., Kim I.-H., Kim M.-S., Lee H.R., Kim Y., Eom K.S. 2020. Descriptions of two digenean trematodes found from a Chinese sea snake, Laticauda semifasciata, in Republic of Korea. Korean Journal of Parasitology 58(3): 279-285. https://doi.org/10.3347/kjp.2020.58.3.279
- Coil W.H., Kuntz R.E. 1960. Three new genera of trematodes from Pacific sea serpents, Laticauda colubrina and L. semifasciata. Proceedings of the Helminthological Society of Washington 27: 147-150.
- Das I. 2012. A Naturalist's Guide to the Snakes of South-East Asia including Malaysia, Singapore, Thailand, Myanmar, Borneo, Sumatra, Java, and Bali. John Beaufoy Publishing Limited, United Kingdom, 176 pp.
- Ganesh S.R., Nandhini T., Samuel V.D., Sreeraj C.R., Abhilash K.R., Purvaja R., Ramesh R. 2019. Marine snakes of Indian coasts: Historical resume, systematic checklist, toxinology, status, and identification key. Journal of Threatened Taxa 11(1): 13132-13150. https://doi.org/10.11609/jott.3981.11.1.13132-13150
- Gill B.J. 1997. Records of turtles and sea snakes in New Zealand, 1837-1996. New Zealand Journal of Marine and Freshwater Research 31: 477-486. https://doi.org/10.1080/00288330.1997.9516781
- Gill B.J., Whitaker A.H. 2014. Records of sea-kraits (Serpentes: Laticaudidae: Laticauda) in New Zealand. Records of the Auckland Museum 49: 39-42.
- Guglielmone A.A., Nava S., Robbins R.G. 2023. Geographic distribution of hard ticks (Acari: Ixodida: Ixodidae) of the world by countries and territories. Zootaxa 5251(1): 1-275. https://doi.org/10.11646/zootaxa.5251.1.1
- Guglielmone A.A., Robbins R.G., Apanaskevich D.A., Petney T.N., Estrada-Peña A., Horak I.G., Shao R., Barker S.C. 2010. The Argasidae, Ixodidae and Nuttalliellidae (Acari: Ixodida) of the world: a list of valid species names. Zootaxa 2528(1): 1-28. https://doi.org/10.11646/zootaxa.2528.1.1
- Guglielmone A.A., Robbins R.G., Apanaskevich D.A., Petney T.N., Estrada-Peña A., Horak I.G. 2014. The Hard Ticks of the World (Acari: Ixodida: Ixodidae). Springer, Heidelberg, 738 pp. https://doi.org/10.1007/978-94-007-7497-1
- Hayashi F., Masunaga, G. 2001. Ecological notes on the tick, Amblyomma nitidum, and trombiculid mites infesting sea snakes in Japan. Acarological Society of Japan 10: 1-17. https://doi.org/10.2300/acari.10.1
- Heatwole H., Busack S., Cogger H. 2005. Geographic variations in sea kraits of the Laticauda colubrina complex (Serpentes: Elapidae: Hydrophiinae: Laticaudini). Herpetological Monographs 19: 1-136. https://doi.org/10.1655/0733-1347(2005)019%5B0001:GVISKO%5D2.0.CO;2
- Hirst S., Hirst, L.F. 1910. Descriptions of five new species of ticks (Ixodidae). Annals and Magazine of Natural History 8: 299-308. https://doi.org/10.1080/00222931008692856
- Hoogstraal H. 1967. Ixodes jacksoni n. sp. (Ixodoidea: Ixodidae), a nest parasite of the Spotted cormorant, Phalacrocorax punctatus (Sparrman), in New Zealand. Journal of Medical Entomology 4(1): 37-41. https://doi.org/10.1093/jmedent/4.1.37
- Kohls G.M. 1950. Ticks (Ixodoidea) of the Philippines. National Institutes of Health Bulletin, Washington, U.S.A., 28 pp.
- Kolonin G.V. 2009. Fauna of ixodid ticks of the world. http://www.kolonin.org/. Accessed 21 July 2025.
- Kwak M.L., Kuo C.-C., Chu H.-T. 2020. First record of the sea snake tick Amblyomma nitidum Hirst & Hirst, 1910 (Acari: Ixodidae) from Taiwan. Tick and Tick-borne Diseases 11: 101383. https://doi.org/10.1016/j.ttbdis.2020.101383
- Lane A., Shine R. 2011. Intraspecific variation in the direction and degree of sex-biased dispersal among sea-snake populations. Molecular Ecology 20: 1870-1876. https://doi.org/10.1111/j.1365-294X.2011.05059.x
- Leviton A.E., Siler C.D., Weinell J.L., Brown R.M. 2018. Synopsis of the snakes of the Philippines. A synthesis of data from biodiversity repositories, field studies, and the literature. Proceedings of the California Academy of Sciences 64(14): 399-568.
- Leviton A.E., Wogan G.O.U., Koo M.S., Zug G.R., Lucas R.S., Vindum J.V. 2003. The dangerously venomous snakes of Myanmar. Illustrated checklist with keys. Proceedings of the California Academy of Sciences 54(24): 407-462.
- Mackessy S.P., Tu A.T. 1993. Biology of the sea snakes and biochemistry of their venom. In: Tu A.T. (ed.) Toxin-related Diseases: Poisons Originating from Plants, Animals and Spoilage. Oxford & IBH Publishing Co., New Delhi, pp. 305-351.
- Mangin S., Gauthier-Clerc M., Frenot Y., Gendner J.-P., Le Maho Y. 2003. Ticks Ixodes uriae and the breeding performance of a colonial seabird, king penguin Aptenodytes patagonicus. Journal of Avian Biology 34: 30-34. https://doi.org/10.1034/j.1600-048X.2003.02916.x
- Moravec F., Justine J.-L. 2019. New species and new records of camallanid nematodes (Nematoda, Camallanidae) from marine fishes and sea snakes in New Caledonia. Parasite 26: 66. https://doi.org/10.1051/parasite/2019068
- Neveu-Lemaire M., Dollfus R.-P., Galliard H. 1934. Répertoire des espèces et des genres nouveaux. Annales de Parasitologie Humaine et Comparée 12(1): 70-80. https://doi.org/10.1051/parasite/1934121070
- Qiu Y., Kidera N., Hayashi M., Fujishima K., Tamura H. 2021a. Rickettsia spp. and Ehrlichia spp. in Amblyomma ticks parasitizing wild amphibious sea kraits and yellow-margined box turtles in Okinawa, Japan. Ticks and Tick-borne Diseases 12: 101636. https://doi.org/10.1016/j.ttbdis.2020.101636
- Qiu Y., Kidera N., Thu M.J., Hayashi M., Fujishima K., Tamura H. 2021b. First molecular detection of Hemolivia and Hepatozoon parasites in reptile-associated ticks in Iriomote Island, Japan. Parasitology Research 120: 4067-4072. https://doi.org/10.1007/s00436-021-07345-y
- Rageau J. 1960. A propos d'Amblyomma laticaudae Warburton, 1933 (Acarien Ixodidae) en Nouvelle-Calédonie. Bulletin de la Société de Pathologie exotique 53(5): 831-833.
- Rageau, J. 1967. Observations biologiques sur les tiques (Acari: Argasidae et Ixodidae) des îles Françaises d'Océanie. Wiadomości Parazytologiczne 13: 547-553.
- Rasmussen A.R., Elmberg J., Gravlund P., Ineich I. 2011. Sea snakes (Serpentes: subfamilies Hydrophiinae and Laticaudinae) in Vietnam: a comprehensive checklist and an updated identification key. Zootaxa 2894: 1-20. https://doi.org/10.11646/zootaxa.2894.1.1
- Robbins, R.G., Nava S., Ronai I., Chong K.L., Guglielmone A.A. 2025. Type specimens of the world's hard tick species (Acari: Ixodida: Ixodidae): collection data and depositories for all valid names and the current status of invalid names. Zootaxa 5606: 1-287. https://doi.org/10.11646/zootaxa.5606.1.1
- Schmidt G.D., Kuntz R.E. 1973. Nematode parasites of Oceanica. XX. Paraheterotyphlum ophiophagos n. sp. (Heterocheilidae), from the Banded Yellow-lip sea snake, Laticauda colubrina. American Midland Naturalist 89(2): 481-484. https://doi.org/10.2307/2424056
- Sharif, M. 1928. A revision of the Indian Ixodidae with special reference to the collection in the Indian Museum. Records of Indian Museum 30: 217-334. https://doi.org/10.26515/rzsi/v30/i3/1928/162556
- Shine R., Bonnet X., Elphick M.J., Barrott E.G. 2004. A novel foraging mode in snakes: Browsing by the sea snake Emydocephalus annulatus (Serpentes, Hydrophiidae). Functional Ecology 18(1): 16-24. https://doi.org/10.1046/j.0269-8463.2004.00803.x
- Southcott R.V. 1957. Vatacarus ipoides n. gen., n. sp. (Acarina: Trombiculidae). Transactions of the Royal Society of Southern Australia 80: 165-176.
- Takahashi M., Misumi H., Takahashi Y. 2012. A new genus and two new species of chigger mites (Acari, Trombiculidae) collected from amphibious sea snakes of Japan. Bulletin of the National Museum of Natural Science Series A 38: 159-172.
- Takano A., Fijita H., Kadosaka T., Takahashi M., Yamauchi T., Ishiguro F., Takada N., Yano Y., Oikawa Y., Honda T., Gokuden M., Tsunoda T., Tsurumi M., Ando S., Andoh M., Sato K., Kawabata H. 2014. Construction of a DNA database for ticks collected in Japan: application of molecular identification based on the mitochondrial 16S rDNA gene. Medical Entomology and Zoology 65: 13-21. https://doi.org/10.7601/mez.65.13
- Tyabji Z., Mohanty N.P., Young E. & Khan T. 2018. The terrestrial life of sea kraits: insights from a long-term study on two Laticauda species (Reptilia: Squamata: Elapidae) in the Andaman Islands, India. Journal of Threatened Taxa 10(11): 12443-12450. https://doi.org/10.11609/jott.4311.10.11.12443-12450
- Uetz P., Freed P., Aguilar R., Reyes F., Hošek J. (eds.). 2025. The Reptile Database. https://reptile-database.reptarium.cz/. Accessed 21 July 2025.
- Uribe J.E., Kelava S., Nava S., Cotes-Perdomo A.P., Castro L.R., Rivera-Paéz F.A., Mans B.J., Gofton A., Teo E.J.M., Zardoya R. & Barker S.C. 2024. New insight into the molecular phylogeny, biogeographical history, and diversification of Amblyomma ticks (Acari: Ixodidae) based on mitogenomes and nuclear sequences. Parasites & Vectors 17: 139. https://doi.org/10.1186/s13071-024-06131-w
- Velasquez C.C., Eduardo S.V. 1994. Index Catalogue of Parasites of Philippine Vertebrates. National Academy of Science and Technology (Philippines), 391 pp.
- Voltzit O.V., Keirans J.E. 2002. A review of Asian Amblyomma species (Acari, Ixodida, Ixodidae). Acarina 10(2): 95-126.
- Warburton C. 1933. On five new species of ticks (Arachnida Ixodoidea): Ixodes petauristae, I, ampullaceus, Dermacentor imitans, Amblyomma laticaudae and Aponomma draconis, with notes on three previously described species, Ornithodoros franchinii Tonelli-Rondelli, Haemaphysalis cooleyi Bedford and Rhipicephalus maculatus Neumann. Parasitology 24: 558-568. https://doi.org/10.1017/S0031182000020953
- Wikelski M. 1999. Influences of parasites and thermoregulation on groping tendencies in marine iguanas. Behavioral Ecology 10(1): 22-29. https://doi.org/10.1093/beheco/10.1.22
- Wilson N. 1970. New distributional records of ticks from Southeast Asia and the Pacific (Metastigmata: Argasidae, Ixodidae). Oriental Insects 4(1): 37-46. https://doi.org/10.1080/00305316.1970.10433939
- Weinell J.L., Hooper E., Leviton A.E., Brown R.M. 2019. Illustrated key to the snakes of the Philippines. Proceedings of the California Academy of Sciences 66(1): 1-49.
- Yamaguti S. 1933. Studies on the helminth fauna of Japan. Part I. Trematodes of birds, reptiles and mammals. Japanese Journal of Zoology 5: 1-134.
- Yamaguti N., Tipton V.J., Keegan H.L., Toshioka S. 1971. Ticks of Japan, Korean, and the Ryukyu Islands. Brigham Young University Science Bulletin, Biological Series 15: 1-226. https://doi.org/10.5962/bhl.part.25691



2025-08-27
Date accepted:
2026-03-24
Date published:
2026-03-30
Edited by:
McCoy, Karen

This work is licensed under a Creative Commons Attribution 4.0 International License
2026 Amarga, Ace Kevin S.; Robbins, Richard G. and Chiu, Ming-Chung
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



