Damaeid mite species (Acari, Oribatida, Damaeidae) and their occurrence in subterranean habitats. A case study from the Romanian Carpathians
Nae, Ioana  1
; Miko, Ladislav
1
; Miko, Ladislav  2
; Nae, Augustin
2
; Nae, Augustin  3
; Băncilă, Raluca Ioana
3
; Băncilă, Raluca Ioana  4
and Maraun, Mark
4
and Maraun, Mark  5
5
1✉ The Romanian Academy, "Emil Racovitza" Institute of Speleology, 13 September Road, No. 13, 050711, Sector 5, Bucharest, Romania.
2Faculty of Science, Institute of Environmental Studies, Charles University, Benátská 2, Prague 128 00, Czech Republic.
3The Romanian Academy, "Emil Racovitza" Institute of Speleology, 13 September Road, No. 13, 050711, Sector 5, Bucharest, Romania.
4The Romanian Academy, "Emil Racovitza" Institute of Speleology, 13 September Road, No. 13, 050711, Sector 5, Bucharest, Romania.
5J.F. Blumenbach Institute of Zoology and Anthropology, University of Göttingen, 37073, Göttingen, Germany.
2026 - Volume: 66 Issue: 1 pages: 207-223
https://doi.org/10.24349/64gw-w5w3Original research
Keywords
Abstract
Introduction
Oribatid mites (Acari: Oribatida) are among the most diverse and ecologically significant groups of soil microarthropods, occurring in almost all terrestrial ecosystems – from forest litter and moss to alpine soils (Maraun and Scheu 2000). They play a fundamental role in decomposition and nutrient cycling by feeding on fungi, lichens, plant detritus and animal remains (Walter and Proctor, 1999; Schneider et al. 2004; Maraun et al. 2023). Although oribatid mite diversity and abundance vary considerably among habitats (Schatz and Behan-Pelletier 2008), the ecological and environmental factors shaping their spatial distribution remain poorly understood (Maraun and Scheu 2000; Caruso et al. 2019).
The Mesovoid shallow substratum (MSS), part of the Superficial Underground Compartment (SUC) or, in French, ′Milieu Souterrain Superficiel′ (Juberthie et al. 1980; Juberthie and Delay 1981; Juberthie and Decu 1994; Decu et al. 2006), represents an intermediate environment between the soil base and the bedrock. The MSS is composed of a network of small voids and cracks between rock fragments, generally situated in the lower levels of scree slopes (Juberthie et al. 1980). It develops mostly on limestone mountain slopes, either exposed (''nude'' screes) or stabilized by a thin layer of soil and vegetation, and is characterized by the absence of light, minimal temperature variation, and high relative humidity (Culver and Pipan 2019). Another essential characteristic is its ecological connection with the deep subterranean environment, evidenced by the presence of troglobitic species inside scree habitats.
Because of its structure and depth, the MSS is difficult to access and requires specific sampling techniques. Drillings are currently the only efficient method for collecting invertebrates from MSS, as traditional arthropod sampling techniques are not suitable for this environment (López and Oromí 2010). MSS habitats play an important ecological role, providing refuge and stable microclimatic conditions for various arthropod groups as shown by Nitzu et al. (2014). The oribatid material analyzed in the present study was collected as part of a biospeological research plan for a PhD thesis (Nae 2010). Although not meant for oribatid mites, their significant presence in the samplings and the scarce scientific papers on the topic, determined us to publish our findings.
In the Piatra Craiului National Park, a region characterized by well-developed limestone screes and caves, the diversity of oribatid mites was first investigated in edaphic habitats (Honciuc and Stănescu 2003). Beginning in 2015, subsequent studies have expanded this research to include both MSS and edaphic habitats, providing new insights into oribatid diversity and community composition (Nae and Ivan 2015; Nae and Băncilă 2017; Nae and Nae 2018; Nae et al. 2021; Nae and Maraun 2023).
The family Damaeidae Berlese, 1896 is diverse, being represented by more than 250 described species worldwide (Subías 2004). Grandjean (1954a) included them in the ′Euphérédermes', one of the five taxa within Brachypylina, which is characterised namely by the nymphs (less often also by the adults) carrying gastronotic exuviae of previous stages (''scalps'') and by the reduction of the notogastral setae f1, da, dm, dp (the sete da, dm, dp are present only in the larvae) (Miko and Mourek 2008). However, recent studies have shown that Damaeoidea are paraphyletic and are phylogenetically associated with some taxa of Gustavioidea, Ameroidea and Polypterozetoidea (Cordes et al. 2024). Damaeid mites are mostly long-legged, middle sized to large forms (rarely smaller than 500 μm, some species exceeding 1,500 μm belong to largest oribatid mites) with roughly triangular prodorsum, which is separated from usually circular or ovoid notogaster by a deep dorsosejugal furrow. The nymphs and partly also adults, carry gastronotic exuviae. Some species are remarkable by carrying a bulk of adherent dirt on the notogaster, serving probably as a kind of camouflage against predators (Luxton 1981; Miko and Mourek 2008; Miko 2021). With its high morphological variability and due to its old age, any study regarding the Damaeidae has the potential of bringing new ways of understanding their phylogeny, evolution, functional traits and habitat preferences.
The aims of our study are: a) to investigate and compare the diversity and community structure of Damaeidae from two types of MSS (nude and fixed) in a sub-alpine region in the Piatra Craiului National Park, Southern Carpathians, Romania; b) to assess the relationship between the type of MSS studied (nude and fixed) and the diversity and community structure of Damaeidae mites. This represents the first ecological study focusing on Damaeidae from a subterranean environment in Romania.
Material and methods
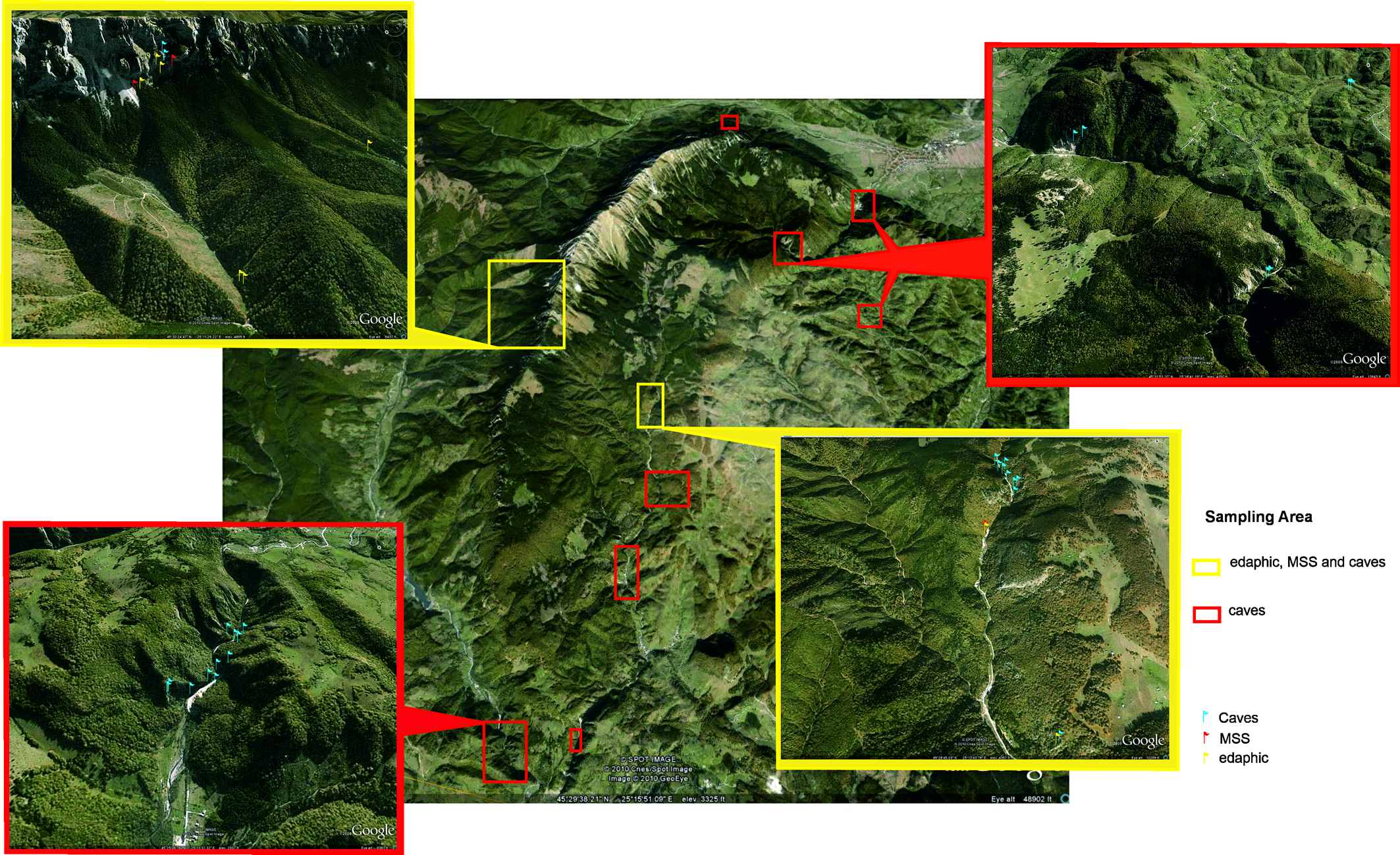


The study area is located in the Piatra Craiului National Park, Southern Carpathians, one of the most important karst areas in Romania (Nitzu et al. 2014). Piatra Craiului Massif is a 20 km2 large limestone ridge with more than 500 caves and diverse types of talus and scree slopes, both covered and open (Nae and Băncilă 2017). Three sampling sites were selected (Fig. 1):
- Cerdacul Stanciului (CS) (Figure 2) - a mobile nude limestone scree situated near Stanciului Cave; geographical coordinates: 45°30′21″N, 25°11′39″E.
- Marele Grohotis (MG) (Figure 3) - the largest mobile nude limestone scree accumulation from Piatra Craiului Massif; geographical coordinates: 45°30′34″N, 25°11′35″E.
- Valea Seacă (VS) (Figure 4) - a stabilized type of MSS, covered by spruce and beech forest; geographical coordinates: 45°28′48″N, 25°13′28″E.









CS and MG are sub-alpine habitats, classified as ''calcareous and calcashist screes of the montane to alpine levels - Thlaspietea rotundifolii'' (Doniță et al. 2005, pg.327) and are listed in the 8210 habitat types following Natura 2000 habitats classification. VS is a R6111 type of habitat - Carpathian South-East communities of fixed screes with Geranium macrrorhizum, Sedum fabaria and Geranium lucidum (Doniță et al. 2005). All Oribatida material used in this study was collected as a part of a biospeological research plan for a PhD thesis focusing on Araneae (Nae, 2010). Pitfall traps (plastic containers, with ethanol, buried with the top edge at ground level, covered with a flat rock, as in Barber 1931) were used to collect invertebrates from the edaphic habitat (EDAF), and drillings, plastic containers with ethanol and antifreeze agent, inserted in PVC tubes with perforations at the base, covered by a plastic lid, as in Gers (1992), modified as in Nae (2010) and Nitzu et al. (2010) for MSS (López and Oromi 2010). The CS and MG MSS was sampled at two depths: 0.5 m - the superficial MSS (SMSS) and 0.75 m - the deep MSS (DMSS), and two altitudes: CS at 1637 m and 1672 m, MG at 1579 m and 1580 m. The VS MSS sampling was done only at 0.5 m depth, at 1087 m altitude (the second drilling was lost due to bear interference). Sampling was done monthly, from April to October 2008, and from April to November 2009. To collect mites with drillings, we used two drillings per sampling site, placed at different altitudes. Inside of each drilling, we placed a trap half filled with 70% ethanol. The upper part of the drilling was covered with a plastic lid to prevent the debris and rocks from falling inside. Each trap was emptied once a month. To collect mites from EDAF, we selected four plots, one in CS, two at MG and one plot in VS. In each plot we placed five pitfall traps, with 70% ethanol, buried at a depth of 9 cm, in a perimeter of 25 m2, (forming a square, with four traps at each corner, and one in the middle). This sampling design made a total of 120 sampling units (3 sites x 1 habitat type (edaphic) x 1 year (2008) x 7 months =21; 3 sites x 1 habitat type (edaphic) x 1 year (2009) x 8 months =24; 2 sites x 1 habitat type (MSS) x 1 year (2008) x 7 months x 2 depths = 28; 2 sites x 1 habitat type (MSS) x 1 year (2009) x 8 months x 2 depths = 32; 1 sites x 1 habitat type (MSS) x 1 year x 7 months x 1 depth = 7; 1 sites x 1 habitat type (MSS) x 1 years (2009) x 8 months x 1 depth = 8).
The collected mites (only adults were included in the study) were sorted and identified to genus and species level. We used the systematic ranking as presented in Miko (2006) and Behan-Pelletier and Lindo (2023) with additions and modifications published Iater (Miko and Mourek 2008; Miko 2010, 2015, 2021; Miko et al. 2022; Kolesnikov and Miko 2022; Miko et al. 2024). After identification the material was preserved in 70% ethanol and stored in the collection of ''Emil Racoviță'' Institute of Speleology, Bucharest, Romania.
Oribatid mites belonging to family Damaeidae were dried at room temperature, mounted on aluminium stubs and sputter-coated with a 15 nm thick layer of gold-palladium. Subsequently, samples were observed in the scanning electron microscope (SEM) at University of Göttingen, Germany.
Data analysis
All statistical analyses were conducted in R (version 4.0.3, R Core Team, 2020). To assess diversity patterns, we visualized species abundance distributions using abundance distribution curves generated with rankabuncomp function from the BiodiversityR package. To examine the influence of habitat type (MSS versus EDAF), site (CS, MG and VS) and season (spring, summer, fall) on mite species composition, we applied Redundancy Analysis (RDA) using the ''vegan'' package (Oksanen et al. 2020). A separate RDA was performed to assess the influence of MSS type (nude versus fixed) and depth (0.5 versus 0.75 m) on mite species composition, using only data collected from MSS samples. Prior to analysis, species abundance data were Hellinger-transformed.
Results
We identified a total of 15 damaeid species, with 151 adult individuals (Table 1).
Download as
Species
Species abbreviations
Sampling stations
CS
MG
VS
MSS
Edaphic
MSS
Edaphic
MSS
Edaphic
Damaeus sp.
Damasp
-
-
-
-
7
-
Damaeus (D.) crispatus (Kulczynski, 1902)
Damacris
-
1
1
-
-
Damaeus (D.) firmus Kunst, 1958
Damafirm
-
-
2
-
-
-
Damaeus (D.) gracilipes (Kulczynski, 1902)
Damagrac
4
5
4
-
8
-
Damaeus (D.) riparius Nicolet, 1855
Damaripa
-
-
-
-
-
2
Damaeus (Adamaeus) onustus Koch, 1844
Damaonus
-
-
-
-
8
-
Kunstindamaeus sp.
Kunssp
-
-
1
-
-
-
Kunstidamaeus lengersdorfi (Willmann, 1932)
Kunsleng
23
-
59
-
-
-
Kunstidamaeus tecticola Michael, 1888
Damtect
2
-
3
-
-
-
Belba corynopus (Hermann,1804)
Belbacory
-
3
-
-
-
-
Metabelba (M.) pulverosa Strenzke, 1953
Metapulv
-
-
-
-
-
1
Trimetabelba propexa (Kulczynski, 1902)
Metaprop
-
3
-
-
-
-
Metabelbella sp.
Metasp
1
-
-
-
-
-
Metabelbella macerochaeta (Bulanova-Zachvatkina, 1967)
Allomace
1
-
-
-
-
Parabelbella clavigera (Willmann, 1954)
Paraclav
7
2
3
-
-
-
Family Damaeidae Berlese, 1896
Genus Damaeus (Damaeus) Koch, 1835 (= Hypodamaeus Bulanova-Zachvatkina, 1957)
Damaeus sp
We found seven specimens in the MSS at VS, at the altitude of 1087 m.
Damaeus (D.) crispatus (Kulczynski, 1902)
Remarks — Forest species (Bernini et al. 1986).
Distribution — Palearctic - Europe, Caucasus, Central Asia (Schatz 2015). We identified the species in pitfall traps from CS (one individual) at an altitude of 1526 m and in MSS at MG (one individual), at 1579 m.
Damaeus (D.) firmus Kunst, 1958 (= Damaeus longipes Mihelčič, 1957, nom. praeoc. by Willmann, 1940)
Remarks — The species was described and recorded from the mountains of south-west Bulgaria (Belite Brezi, Vitoša Mountains and Bistrica, Rila planina Mountains) (Miko 2015), the Iberian Peninsula (Balazantegui et al. 2016) and it is found, for the first time, in the Romanian fauna. It appears to live on the soil surface, dwelling in forest and shrubland litter, at higher elevations (Miko 2015).
Distribution — One specimen was collected from pitfall traps from MG, the largest limestone scree accumulation from Piatra Craiului Mountains, Central Carpathians, at a depth of 0.5 m, at an altitude of 1579 m, in MSS.
Damaeus (D.) gracilipes (Kulczynski, 1902)
Remarks — This species, common in forest litter and other epiedaphic habitats, was recorded in caves in Europe (L'uptáčik and Miko 2003; Mock et al. 2005; Skubała et al. 2013). Recently, it was found in Korea (Bayartogtokh and Bae 2023).
Distribution — D. gracilipes is known from the Holarctic region (Subías 2004). We identified the species from all three sampling stations: CS (four individuals in MSS and five in edaphic), MG (four individuals in MSS) and VS (eight individuals in MSS), at altitudes of 1087m, 1343 m, 1578m, 1637 m.
Damaeus (D.) riparius Nicolet, 1855
Remarks — The species is - together with D. (A.) onustus - one of the most abundant species of the genus in temperate forests, it was previously reported also from Scandinavian caves (Hippa et al. 1988).
Distribution — It is a frequent species in the Palaearctic (Subías 2004). We identified two individuals from VS, at an altitude of 1085 m, in edaphic samples.
Subgenus Damaeus (Adamaeus) Norton, 1977
Damaeus (Adamaeus) onustus Koch, 1844
Remarks — The species was considered cavernicolous by Skubała et al. (2013), but it is a very common species of the soil surface, living in soil, litter and even in grasses (Miko 2015). It was first cited for the Romanian fauna in 2006, in Maramureș (Mahunka 2006).
Distribution — Palaearctic and Ethiopia (Subías 2004). We identified eight individuals from VS, in MSS, at the depth of – 0.5 m, 1087 m altitude.
Genus Kunstidamaeus Miko, 2006
Kunstidamaeus sp
One specimen was identified in MSS, at MG, at an altitude of 1579 m.
Kunstidamaeus lengersdorfi (Willmann, 1932) (Figure 5)
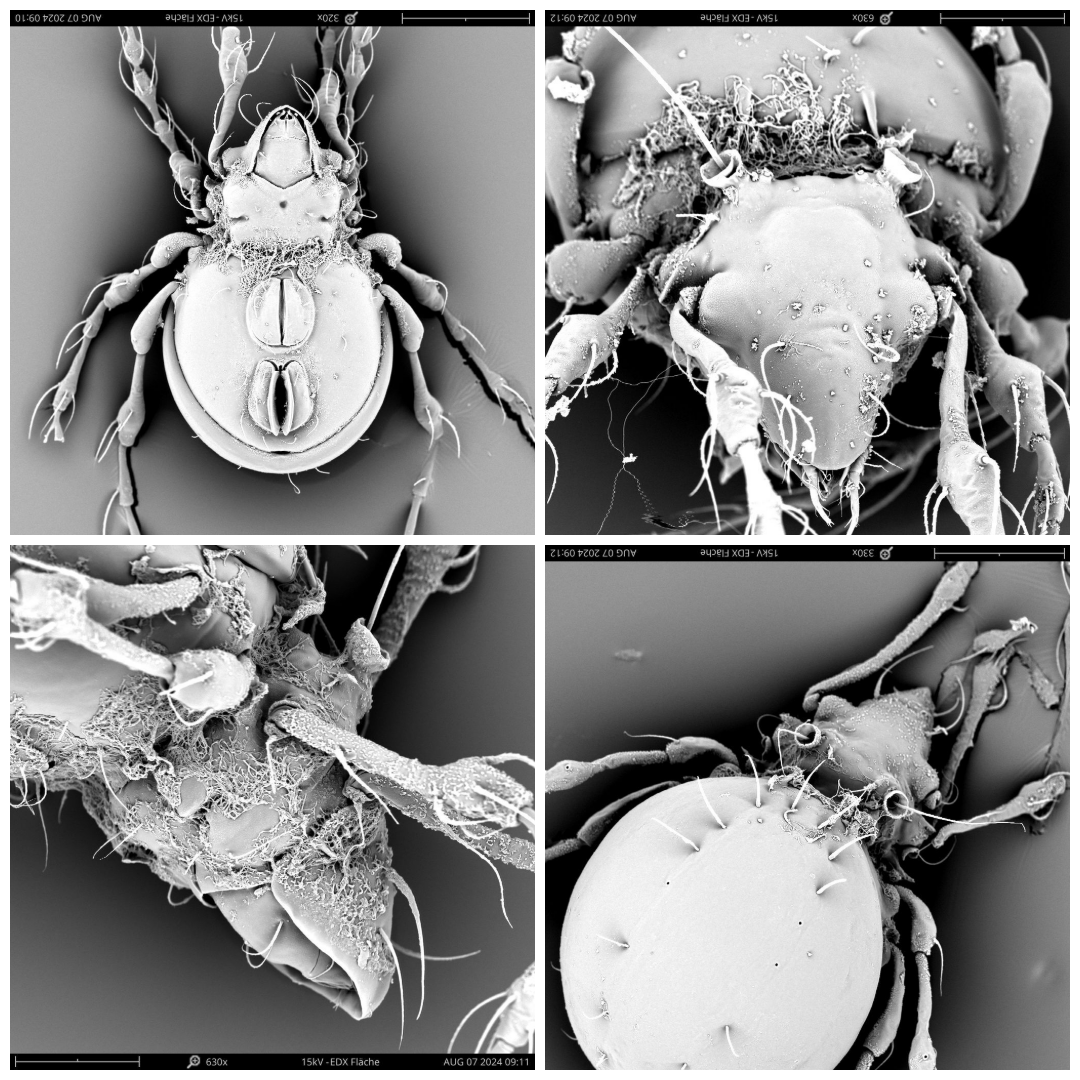


Remarks — The species was first described from the cave ''Iburger Tropfsteinhöhle'' in the Harz, Germany (Willmann 1932) and subsequently recorded from caves in Slovak Karst, Slovakia (L'uptáčik and Miko 2003), Belgium (Willmann 1935, L'uptácik and Miko 2003), French Jura, France (Willmann 1938), Germany (Griepenburg 1939) and Austria (Bruckner 1995). It was recorded from Russia in 2017 (Ignatov et al. 2017). Specimens have been found only in caves or in cave entrances, usually on organic debris such as bat guano, rotten wood or even old candle paraffine (L'uptáčik and Miko 2003). It may be a true troglobiont (Lebrun 1967) or at least a troglophile (L'uptáčik and Miko 2003).
Distribution — The species is distributed in Central Europe (Subías 2004), Russia (Ignatov et al. 2017). We collected 23 individuals from CS and 59 from MG, at altitudes of 1578 m, 1637 m and 1672 m, only from MSS.
Kunstidamaeus tecticola Michael, 1888
Remarks — The species has been recorded throughout Europe and Iceland (Luxton 1989). In Poland it was recorded only from southern parts of country (Olszanowski et al. 1996). The species occurs usually in mosses, and sometimes in dry habitat (Miko 2006), while Mahunka and Mahunka-Papp (2006) found it in beech litter. In the Botanical Garden on the campus of the University of Life Sciences in Ås (Norway) it was rather abundant in spruce litter (Seniczak and Seniczak 2011). Schatz (1983) considered the species myrmecophilous and xerophilous. It was also collected from caves in Poland (Maślak and Barczyk 2011).
Distribution — Kunstidamaeus tecticola is considered a European species (Subías 2004; Miko, 2006). Two individuals were identified from CS and three from MG, only in MSS samples, at altitudes of 1579 m and 1637 m.
Genus Belba Heyden, 1826
Belba corynopus (Hermann, 1804) (Figure 6)
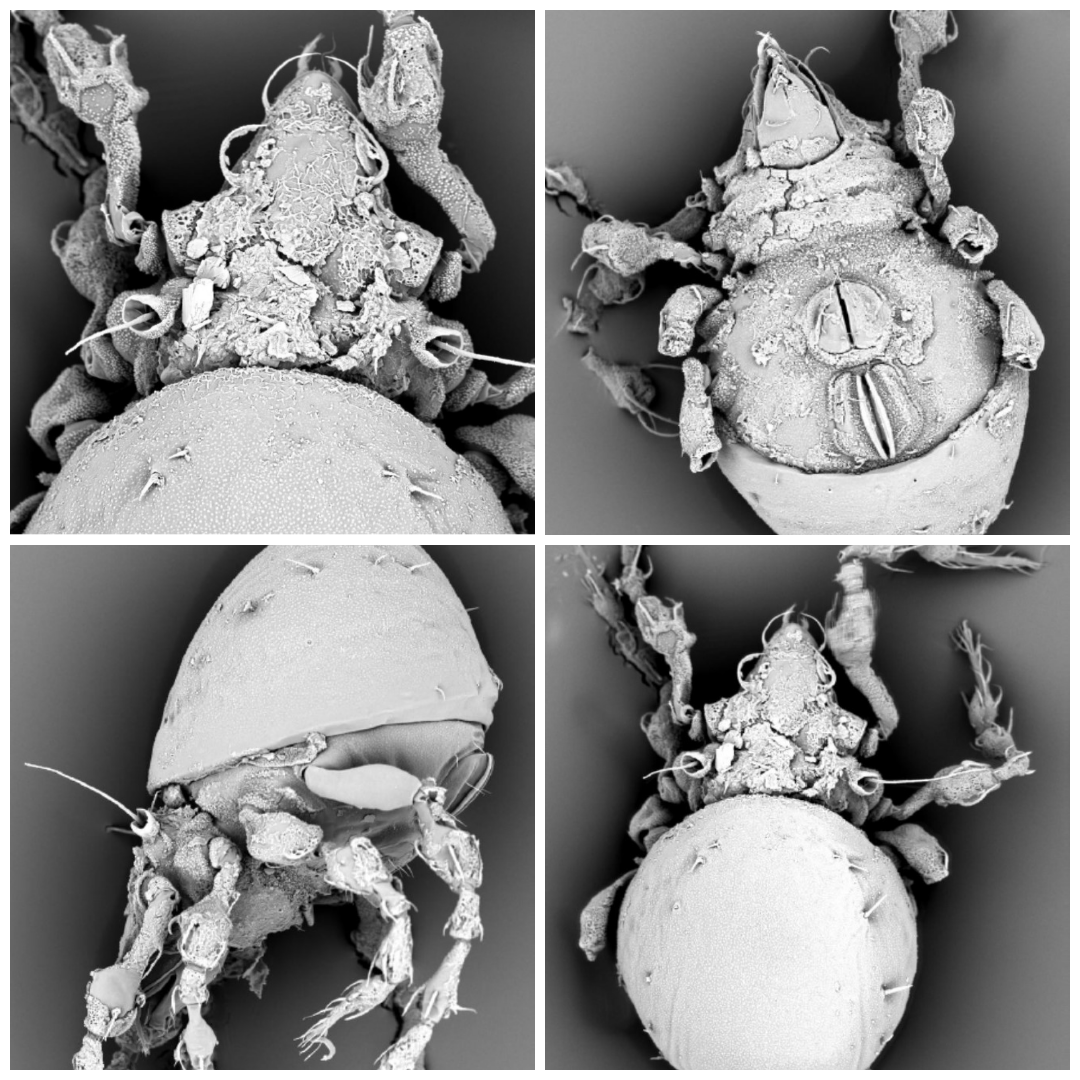


Remarks — Common European species in various elevations, found also in mountain forests and subalpine habitats in South Tyrol in Italy (Schatz 2016), mountain areas of Austria (Leiblachtal, Ried near Hohenweiler), Slovakia (Tomanova dolina Valley (1350 m), Západné Tatry Mountains), Czech Republic (Bohemia and Moravia) (Miko 2016) and Poland (Reserve ''Pod Rysianka'' (1100m) in Beskid Zywiecki Mountains) (Miko 2021); in Vietnam it was recorded from forest, grassland and around human habitation areas (Vu 2020). Present in the Romanian fauna (Hunedoara County, Valea Rea; Maramureș County, Baia Mare; Neamț County, Agapia) (Vasiliu et al. 1993).
Distribution — Holarctic and Oriental distribution (Subías 2004). We collected three individuals of the species in CS, in edaphic samples, at 1343 m altitude.
Genus Metabelba (Metabelba) Grandjean, 1936
Metabelba pulverosa Strenzke, 1953
Remarks — Ubiquitous species, that prefers forest ecosystems (Vasiliu et al. 1993). It was also collected from caves in Poland (Maślak and Barczyk 2011).
Distribution — Holarctic, frequent species in Palearctic and Virginia, U.S.A. (Subías 2004). We identified one individual in edaphic samples from VS, at an altitude of 1085 m.
Genus Trimetabelba Miko, 2024
Trimetabelba propexa (Kulczynski, 1902)
Remarks — The original type material of Kulczynski was lost or destroyed during the Second World War, therefore new material was collected in the locus typicus area: Western Tatras main ridge on Polish – Slovak border south of Zakopane, in the vicinity of Kluczynski's collection sites, within the same elevation and habitat type by Ladislav Miko (Miko et al. 2024), who redescribed the species.
Distribution — Central Europe distribution (Subías 2004; Miko et al. 2024). We identified three individuals in edaphic samples at CS site, at 1526 m.
Genus Metabelbella Bulanova-Zachvatkina, 1967
Metabelbella sp
We found one specimen at MSS, in edaphic samples, at an altitude of 1672 m.
Metabelbella macerochaeta (Bulanova-Zachvatkina, 1967)
Remarks — Bulanova-Zachvatkina (1967) mentiones the species distribution as U.S.S.R. (Caucasus and The Black Sea coast). It was previously recorded from Romania in alpine in mountain habitats and in drillings near Movile Cave (Ivan and Vasiliu 2010), near the Black Sea coast. Ivan and Vasiliu (2010) consider it silvicolous. It was identified in other cave studies in Georgia (at the entrance zone of a cave) (Murvanidze 2014).
Distribution — The species is considered oriental Mediterranean by Subías (2004). We identified one individual in MSS at CS site, at an altitude of 1672 m.
Genus Parabelbella Bulanova-Zachvatkina 1972
Parabelbella clavigera (= Belba clavigera) Willmann, 1954
Remarks — The species was collected from mosses on a rock wall from Balcarova (Czech Republic). Willmann (1954) suggests the possibility that, due to its morphology, the species could be troglophile or even truly troglobiont, and is considered so by Bernini (1980).
Distribution — The species is recorded from Central Europe: Czech Republic (Subías 2004), Slovakia (Mock et al. 2005). We identified seven individuals in CS, in MSS samples, two in edaphic samples at the same site and three at MG site, also in MSS, at altitudes of 1343 m, 1526 m and 1672 m. Our finding represents the first record of the species in Romania.
Data analysis
Eleven species of Damaeidae were found in MSS, at the three sampling stations (CS, MG and VS) and only four species were found in edaphic habitats, Damaeus (D.) riparius, Metabelba pulverosa, Belba corynopus and Trimetabelba propexa. The species with the highest abundance was Kunstidamaeus lengersdorfi with 59 individuals collected from the MSS at MG. Two species of damaeid mites were identified from VS, from covered MSS, fixed by a mixed forest, Damaeus sp. and Damaeus (Adamaeus) onustus.
Parabelbella clavigera, K. lengersdorfi, Metabelbella macerochaeta are considered troglophile and cavernicolous species of damaeid mites, some others occur frequently in these conditions, even if they are similarly abundant outside of caves and MSS (such as D. (D.) riparius, D. (A.) onustus). Thus, the Family Damaeidae has a good representation in MSS, the sites CS and MG having the highest abundance of species.
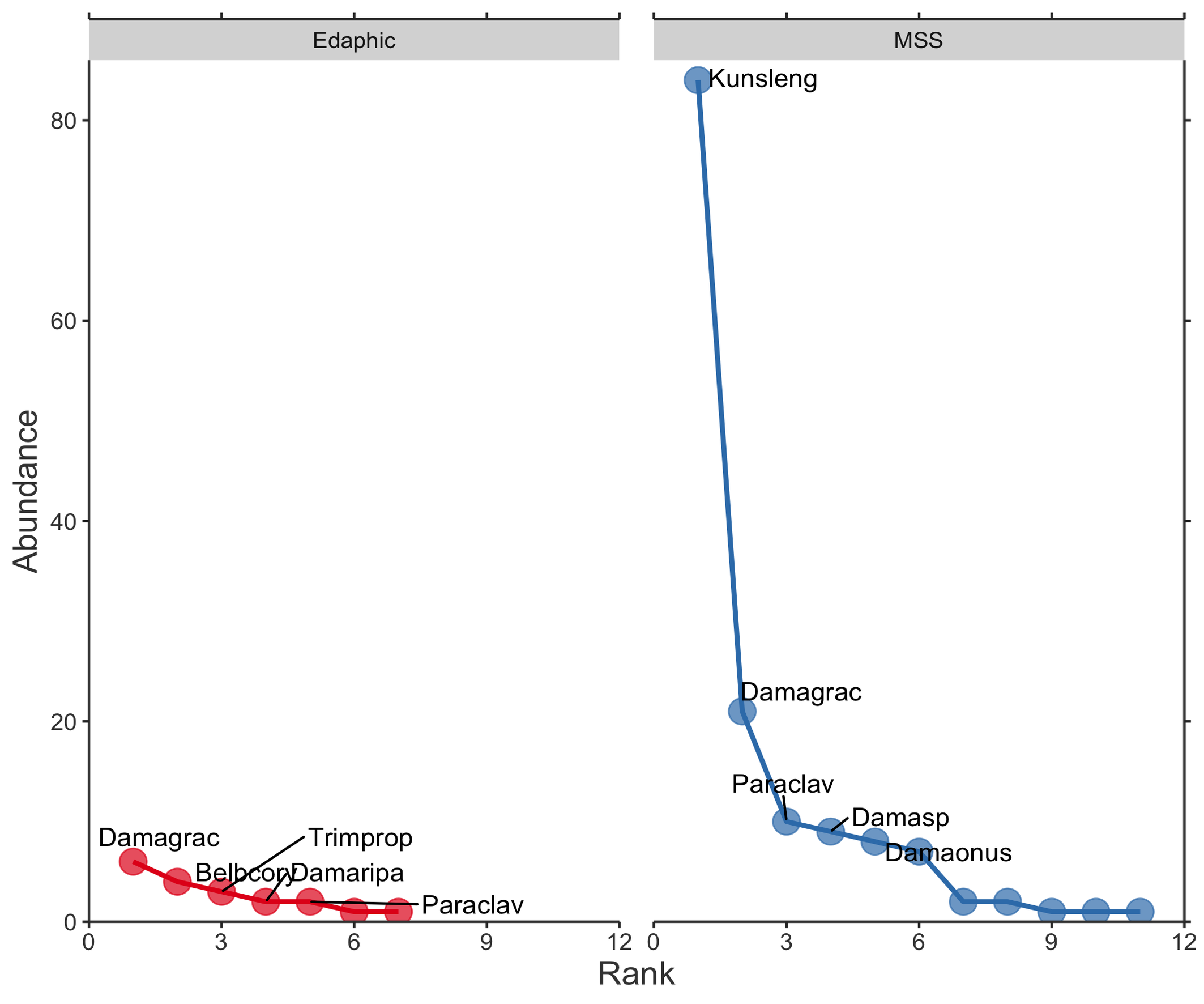


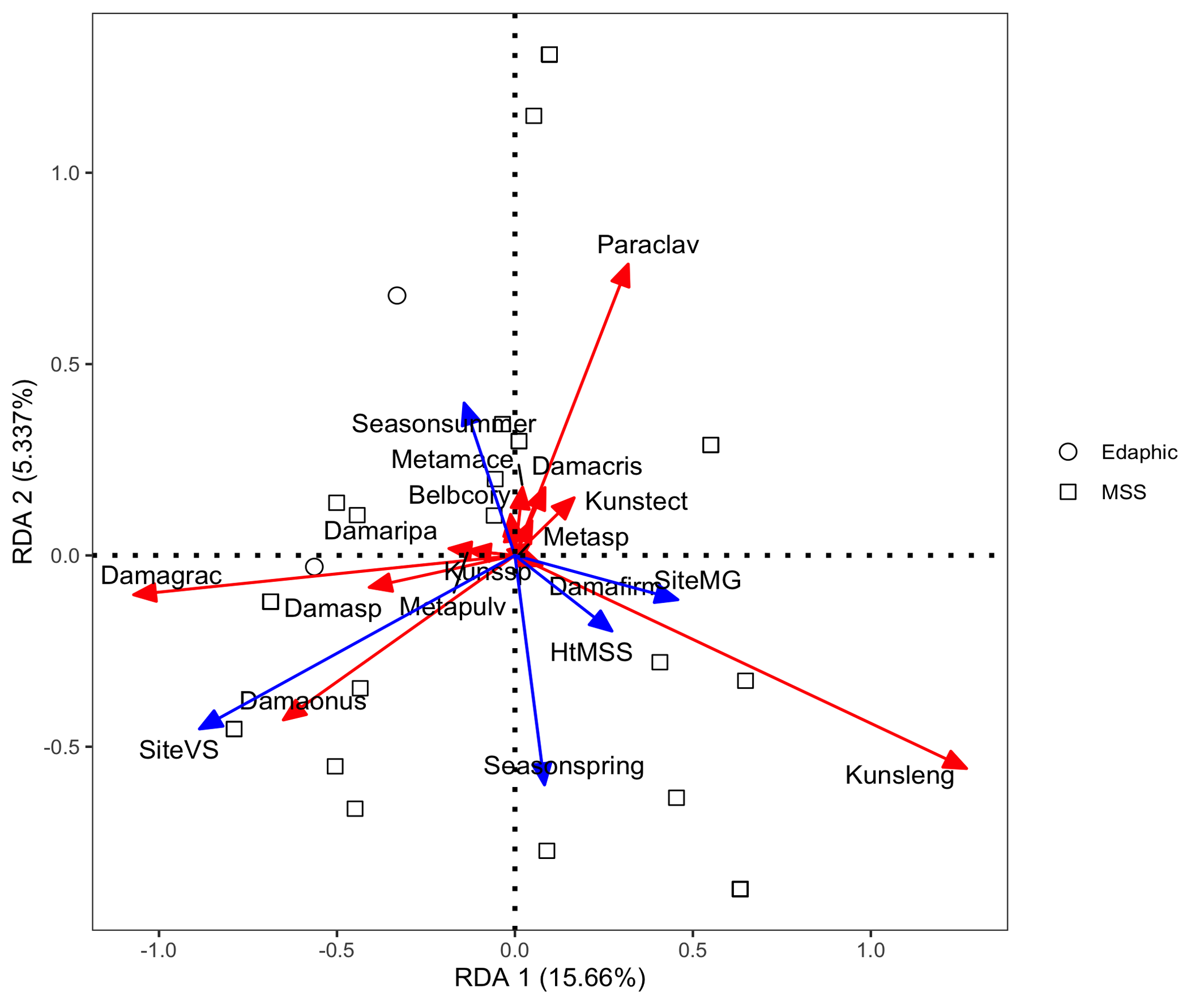


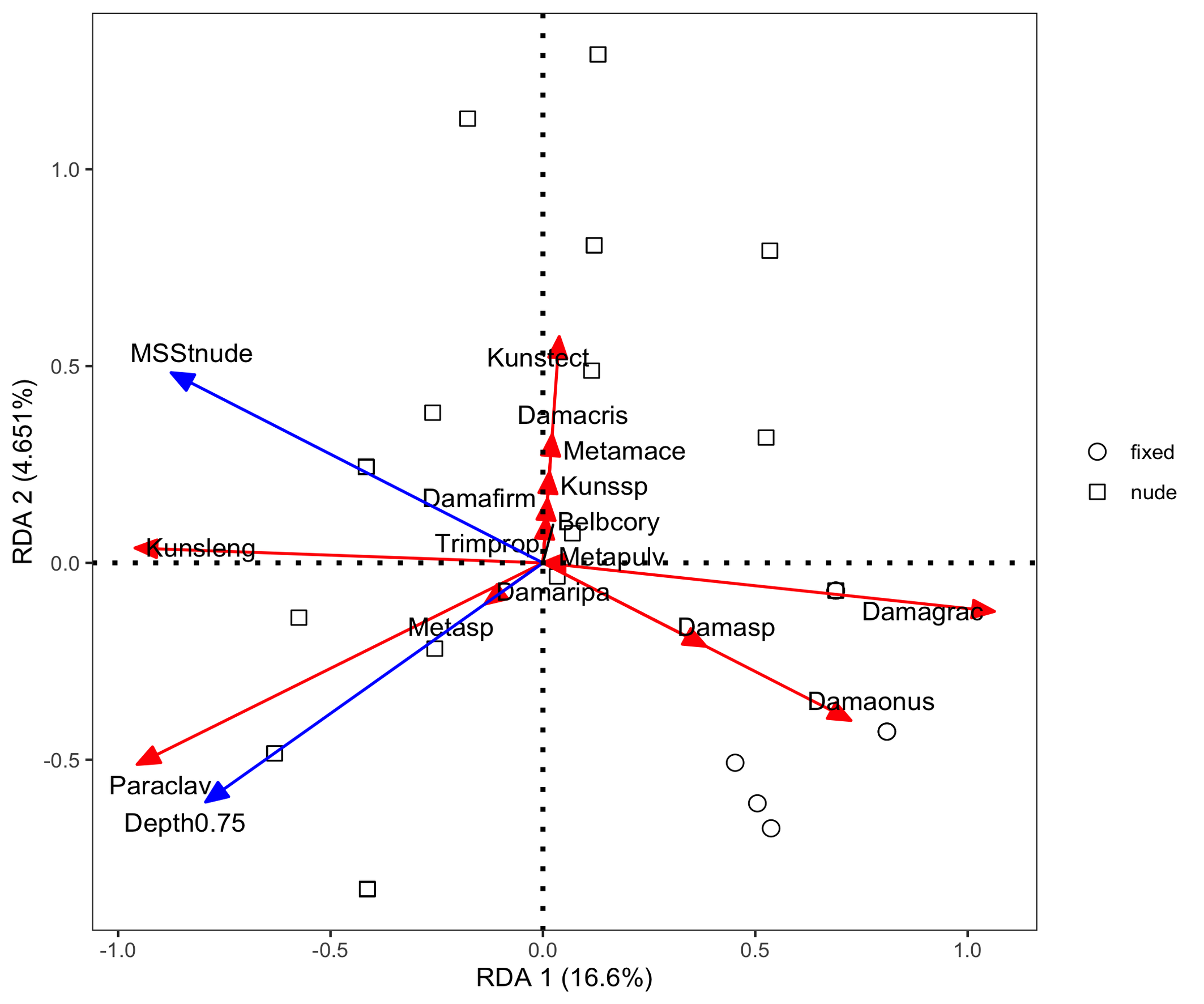
The abundance distribution patterns revealed differences in species composition between edaphic and MSS habitats (Figure 7). The first RDA model yielded an adjusted R² of 0.141, indicating that habitat type, site location and season together explained 14.1% of the total variation in the mite community (Figure 8). Among the explanatory factors, site VS had the most significant influence on species distribution, followed by season spring and season summer. Specifically, site VS showed a positive association with D. (A.) onustus and Damaeus sp., while Season spring was associated with K. lengersdorfi and Season summer with M. macerochaeta. The second RDA model yielded an adjusted R² of 0.168, indicating that MSS type and Depth together explained 16.8% of the total variation in the MSS mite community. The nude MSS showed a strong positive association with K. lengersdorfi and Depth 0.75 m with P. clavigera (Figure 9).


Discussion
Our study revealed distinct differences in the distribution and composition of Damaeidae between MSS and edaphic habitats in the Piatra Craiului Mountains. Eleven damaeid species were identified in MSS, while only four occurred in edaphic samples: D. (D.) riparius, M. pulverosa, B. corynopus, and T. propexa. The greater species richness and abundance in MSS suggests that these transitional subterranean systems provide stable microclimates and sufficient resources suitable for this group of mites.
The most abundant species was K. lengersdorfi (59 individuals in MSS at MG), followed by P. clavigera and M. macerochaeta. The association of K. lengersdorfi with the high-altitude, mobile scree MSS near Stanciului Cave supports its troglophilic or troglobiont status, consistent with morphological adaptations (elongated legs, reduced sclerotization) and its previous records from European cave systems (Arillo and Subías 2006; Miko and Mourek 2008; Ignatov et al. 2017).
Redundancy analysis (RDA) indicated that habitat type, site and season together explained 14.1% of community variation. The strongest effect was linked to site VS, which showed a positive association with D. (A.) onustus and Damaeus sp. Depth and MSS type explained a further 16.8% of variation, with K. lengersdorfi associated with nude MSS and P. clavigera with the deeper MSS (0.75 m). These results agree with the observations of Nae and Băncilă (2017), who found that species composition in MSS depends primarily on substrate stability and humidity rather than on vertical stratification.
Our data reinforce earlier acarological research in the same area (Nae and Ivan 2015; Nae and Băncilă 2017; Nae and Nae 2018; Nae et al. 2021; Nae and Maraun 2023). Nae and Ivan (2015) reported three oribatid species new to the Romanian fauna from Piatra Craiului MSS, emphasizing the role of these habitats in preserving rare and cryptic taxa. In the present study, D. (D.) riparius represents another first record for Romania, extending its known range into the Carpathians and demonstrating that MSS can harbor taxa with otherwise scattered distributions. Nae and Băncilă (2017) highlighted the diversity of oribatid mites in Piatra Craiului MSS and proposed these habitats as biodiversity hotspots for conservation priorities. The dominance of Damaeidae in our samples supports this conclusion and confirms the group's ecological importance in subterranean habitats. Along an altitudinal gradient in the same Massif, Nae and Nae (2018) found that oribatid mite richness peaked at mid to high elevations, coinciding with the environmental range of our sampling sites (1300–1637 m). The high abundance of K. lengersdorfi at MG, the highest and coldest site, supports their conclusion that many troglophilic mites prefer cooler, MSS habitats near the upper forest limit. Nae et al. (2021) demonstrated, using stable isotopes, that oribatid communities in Piatra Craiului MSS span three trophic levels - lichen feeders, primary decomposers and secondary decomposers, and consist exclusively of sexual species. Our dominant taxa (K. lengersdorfi, P. clavigera, M. macerochaeta) belong to Damaeoidea, known to occupy higher trophic positions (Maraun et al. 2023), confirming the functional role of these mites as higher-level decomposers in MSS food webs. The most recent work by Nae and Maraun (2023) reported Jugatala cribelliger (Berlese, 1904) new for the Romanian fauna, again from the same massif, reinforcing the ongoing discovery of rare oribatid taxa in Piatra Craiului MSS. The presence of K. lengersdorfi and D. (D.) riparius in our samples underlines the taxonomic and ecological uniqueness of these habitats.
When compared with surface oribatid fauna inventories (Honciuc and Stănescu 2003), our findings confirm a distinct MSS assemblage. Those authors listed 27 oribatid species from Piatra Craiului soils and litter, including B. corynopus and Damaeus spp., but not K. lengersdorfi or P. clavigera. This difference indicates that many Damaeidae recorded here prefer subterranean habitats rather than the soil-litter layer.
At community level, our results parallel the broader patterns reported for arthropods in Romanian scree habitats by Nitzu et al. (2014), who described strong seasonal variation and microhabitat specialization within MSS arthropod fauna. Their study shows that the environmental conditions inside the screes are relatively constant and that it is permanently populated both by characteristic species and seasonal associations of soil-dwelling species.
Conclusion
Together, these results confirm that MSS in the Piatra Craiului Mountains harbors a diverse and functionally complex community of damaeid mites. Finding troglophilic and cavernicolous species across studies supports the view of MSS as a possible ecosystem connecting surface and cave environments, rather than a temporary refuge. The first Romanian record of D. (D.) riparius, the abundance of K. lengersdorfi and the coexistence of multiple trophic groups reinforce the conservation importance of these habitats for subterranean biodiversity in the Carpathians. Further research combining molecular, trophic and morphological approaches will be essential to clarify the evolutionary adaptations and ecological dynamics of oribatid mites in these understudied systems.
References
- Arillo A., Subías L.S. 1994. Oribatidos del ″M.S.S.'' de las Islas Canarias, Proscheloribatinae Subfam. n. (Acari, Oribatida). Mém. Biospéol. XXI: 1-6.
- Bayartogtokh B., Bae Y.-S. 2023. New findings of soil mites (Acari: Oribatida) from Korea. Systematic and Applied Acarology 28(10): 1597-1617. https://doi.org/10.11158/saa.28.10.3
- Barber H.S. 1931. Traps for cave inhabiting insects. Journal of the Elisha Michell Scientific Society. 46: 259 -266.
- Balazantegui I., Corral-Hernández E., Iturrondobeitia J. C. 2016. Systematic checklist of the oribatids (Acari: Oribatida) of beech forests (Fagus sylvatica L.) from the North-East of the Iberian Peninsula: updated known distributions and the first record of Damaeus (D.) firmus Kunst, 1957. Rev. Ibér. Aracnol. 28: 84-90.
- Behan Pelletier V., Lindo Z. 2023. Oribatid Mites: Biodiversity, Taxonomy and Ecology. CRC Press. pp. 508. https://doi.org/10.1201/9781003214649
- Bernini F. 1980. Notulae Oribatologicae XXIV. Gli Acari Oribatei di alcune piccole grotte del Senese. Redia LXIII: 359-405.
- Bernini F., Avanzati A.M., Bernini S. 1986. Notulae Oribatologicae XXXVII. Gli Acari Oribatei del Massiccio del Pollino (Italia Meridionale): Aspetti faunistici e biogeografici. Lavori della Società Italiana di Biogeografia. 10: 379-488. https://doi.org/10.21426/B610110297
- Bruckner A. 1995. Cave-dwelling oribatid mites (Acarina, Cryptostigmata) from East Austria. Vcrh. Zool.-Bot. Ges. Österreich. 132: 81-107.
- Caruso C. M., Eisen K. E., Martin R. A., Sletvold N. 2019. A meta-analysis of the agents of selection on floral traits. Evolution 73(1): 4-14. https://doi.org/10.1111/evo.13639
- Cordes P., Pan X., Murvanidze M., Seniczak A., Scheu S., Schaefer I., Maraun M., Heimburger B. 2024. Convergent evolution revealed by paraphyly and polyphyly of many taxa of oribatid mites: A molecular approach. Exp. Appl. Acarol. 93: 787-802. https://doi.org/10.1007/s10493-024-00960-1
- Culver D. C., Pipan T. 2009. The Biology of Caves and Other Subterranean Habitats. Oxford University Press. pp. 254.
- Culver D. C., Pipan T. 2014. Shallow Subterranean Habitats. Ecology, Evolution and Conservation. Oxford University Press. pp. 258. DOI: 10.1086/686831 https://doi.org/10.1086/686831
- Culver D. C., Pipan T. 2019. The Biology of Caves and Other Subterranean Habitats. Second Edition. Oxford University Press. pp. 254. DOI: 10.1093/oso/9780198820765.001.0001 https://doi.org/10.1093/oso/9780198820765.001.0001
- Doniță N., Popescu A., Paucă - Comănescu M., Mihăilescu S., Biriș, I. A. 2005. Habitatele din România (Habitats of Romania). Romania: Editura Tehnică Silvică. pp. 496.
- Gers C. 1992. Ecologie et Biologie des arthropods terrestres du Millieu Souterrain Superficiel. Fonctionnement et ecologie evolutive. [PhD Thesis]. Toulouse: Université Paul Sabatier. pp. 319.
- Gripenburg W. 1939. Die Tierwelt der Höhlen bei Kallenhardt. - Zeitschrift für Karst- und Höhlenforschung. 1: 17-26.
- Hippa H., Koponen S., Mannila R., Niemi R., Uusitalo M. 1988. Invertebrates of Scandinavian caves VII. Acari. Notulae Entomologicae. 68: 141-146.
- Honciuc V., Stãnescu M. 2003. Acarian fauna (Mesostigmata, Prostigmata, Oribatida) of Piatra Craiului National Park. In: Research in Piatra Craiului National Park, 1: 159-169.
- Ignatov M. S., Sidorchuk E. A., Ignatova E. A. 2017. A Schistostega pennata (Bryophyta) spore devourer in flagranti - a troglophyle mite Kunstidamaeus lengersdorfi (Acari, Oribatida: Damaeidae). Bryophyte Divers. Evol. 39(1): 059-068. https://doi.org/10.11646/bde.39.1.10
- Ivan O., Vasiliu N.A. 2010. Fauna of oribatid mites (Acari, Oribatida) from the Movile Cave area (Dobrogea, Romania). Trav. Inst. Spéol. «Émile Racovitza». XLIX: 29-40.
- Jiménez-Valverde A., Gilgado J. D., Sendra A., Pérez-Suárez G., Herrero-Borgoñón J. J., Ortuño V.M. 2015. Exceptional invertebrate diversity in a scree slope in Eastern Spain. J Insect Conserv. 19:713-728. https://doi.org/10.1007/s10841-015-9794-1
- Juberthie C., Delay D., Bouillon M. 1980. Extension du milieu souterrain en zone non calcaire: description d'un nouveau milieu et de son peuplement par les Coléoptères troglobies. Mém. Biospéol. 7: 19-52.
- Juberthie C. 1983. Le milieu souterrain: Étude et composition. Mém. Biospéol. 10: 17-65.
- Kolesnikov V. B., Miko L. 2022. Taxonomy of European Damaeidae X. Description of Coronabelba unicornis n. gen., n. sp. (Acari, Oribatida, Damaeidae) from Abkhazia, with comments on genus Metabelba Grandjean, 1936. Acarologia 62(2): 340-351. https://doi.org/10.24349/1khj-v25f
- Lebrun P. 1967. Note sur quelques oribates (Acarina: Oribatei) de la faune cavernicole de Belgique. Bull. Ann. Soc. R. Ent. Belg. 103: 183-188.
- Leruth, R. 1939. La biologie du domaine souterrain et la faune cavernicole de la Belgique. Ordre Collemboles. Mémoires du Musée Royal d'Histoire Naturelle de Belgique. Mémoire 87:194-201.
- López H., Oromi P. 2010. A pitfall trap for sampling the mesovoid shallow substratum (MSS) fauna. Speleobiology Notes 2: 7-11.
- L'uptáčik, P., Miko, L. 2003. Oribatid mites (Acarina, Oribatida) of Slovak caves. Subterranean Biology 1: 25-29.
- Luxton M. 1981. Studies on the oribatid mites of a Danish beech wood soil VI. Seasonal population changes. Pedobiologia 21(6): 387-409. https://doi.org/10.1016/S0031-4056(23)03600-4
- Luxton M. 1989. Michael's British Damaeids (Acari: Cryptostygmata). J. Nat. Hist. 23: 1367 - 1372. https://doi.org/10.1080/00222938900770731
- Mahunka S. 2006. Oribatids from Maramureș. Studia Univ. Vasile Goldiș, Seria Șt. Vieții. 17: 59-75.
- Mahunka, S., Mahunka-Papp, L. 2006. Faunistical and taxonomical studies on oribatids collected in Albania (Acari: Oribatida). I. Opusc. Zool. 37: 43-62.
- Maraun M., Scheu S. 2000. The structure of oribatid mite communities (Acari, Oribatida): patterns, mechanisms and implications for future research. Ecography 23(3): 374-382. https://doi.org/10.1111/j.1600-0587.2000.tb00294.x
- Maraun M., Thomas T., Fast E., Treibert N., Caruso T., Schaefer I., Lu J. Z., Scheu S. 2023. New perspectives on soil animal trophic ecology through the lens of C and N stable isotope ratios of oribatid mites. Soil Biol. Biochem. 177: 108890. https://doi.org/10.1016/j.soilbio.2022.108890
- Maślak M., Barczyk G. 2011. Oribatid mites (Acari, Oribatida) in selected caves of the Kraków-Wieluń Upland (southern Poland). Biological Lett. 48(1): 107-116. https://doi.org/10.2478/v10120-011-0011-y
- Miko, L. 2006. Damaeidae. In: Weigmann, G.: Hornmilben (Oribatida), Die Tierwelt Deutschlands, Teil 76. Dahl, F. (Ed.) Goecke and Evers, Keltern. p. 179-207.
- Miko L., Mourek J. 2008. Taxonomy of European Damaeidae (Acari: Oribatida) I. Kunstidamaeus Miko, 2006, with comments on Damaeus sensu lato. Zootaxa 1820: 1-26. https://doi.org/10.11646/zootaxa.1820.1.1
- Miko L. 2015. Taxonomy of European Damaeidae VIII. Contribution to classification of genus Damaeus C. L. Koch, 1835, with a review of Adamaeus Norton, 1978 and Paradamaeus Bulanova-Zachvatkina, 1957 and redescription of three species. Zootaxa 3980 (2): 151-188. https://doi.org/10.11646/zootaxa.3980.2.1
- Miko, L. 2016b. Oribatid mites (Acarina: Oribatida) of the Czech Republic. Revised check-list with a proposal for Czech oribatid nomenclature. Klapalekiana 52 (Suppl.): 1-302.
- Miko L. 2021. Taxonomy of European Damaeidae IX. Contribution to the revision of the genus Belba von Heyden, 1826 (Acari, Oribatida). Syst. Appl. Acarol. 26(8): 1575-1613. https://doi.org/10.11158/saa.26.8.13
- Miko L., Kolesnikov V. B., Ermilov S. G., Klimov P. B. 2022. Taxonomy of European Damaeidae (Acari, Oribatida) XI. European species of the genus Piribelba Miko 2021: redescriptions of P. rossica (Bulanova-Zachvatkina, 1957) and P. piriformis (Mihelčič, 1964) using morphology and DNA sequence data. Zootaxa 5187 (1): 169-210. https://doi.org/10.11646/zootaxa.5187.1.11
- Miko L., Kolesnikov B. V., Murvanidze M. 2024. Taxonomy of European Damaeidae (Acari: Oribatida) XII. Trimetabelba Miko gen. nov. with redescription of T. propexa (Kulczynski, 1902) comb. nov. description of two new species and proposal of Prometabelba Miko gen. nov. Internat. J. Acarol. 50(8): 668-686. https://doi.org/10.1080/01647954.2024.2394244
- Mock A., L'uptáčik P., Fenda P., Svatoň, J. Horszagh, I., Krumpal M. 2005. Terrestrial arthropods inhabiting caves near Vel′ky Folkmar (Cierna Hora Mts.; Slovakia). In: Tajovsky, K., Schlaghamersky, J., Pizl, V. (Eds.). Contributions to Soil Zoology in Central Europe I. Institute of Soil Biology ASCR. Ceske Budejovice. p. 95-101.
- Murvanidze M. 2014. Oribatid mites of Georgian (Caucasus) caves including the description of a new species of Ghilarovus Krivolutsky, 1966. Internat. J. Acarol. 40(6): 463-472. https://doi.org/10.1080/01647954.2014.950604
- Nae A. 2010. Cercetări biospeologice în Masivul Piatra Craiului (Biospeleological research in Piatra Craiului Massif) [PhD thesys]. Bucharest: The Romanian Academy,″Emile Racovitza» Institute of Biospeleology. pp. 303.
- Nae I., Ivan O. 2015. First record of three oribatid mites species (Acari, Oribatida) in the Romanian fauna. Trav. Inst. Spéol. «Émile Racovitza». LIV: 33-40.
- Nae I., Băncilă R. 2017. Mesovoid shallow substratum as a biodiversity hotspot for conservation priorities: analysis of oribatid mite (Acari: Oribatida) fauna. Acarologia 57(4): 855-868. https://doi.org/10.24349/acarologia/20174202
- Nae I., Nae A. 2018. Oribatid mite (Acari: Oribatida) species inventory on an altitudinal gradient in the Piatra Craiului Mountains (Southern Carpathians, Romania). Trav. Inst. Spéol. «Émile Racovitza». LVI: 33-50.
- Nae I., Nae A., Scheu S., Maraun M. 2021. Oribatid mite communities in mountain scree: stable isotopes (15N, 13C) reveal three trophic levels of exclusively sexual species. Exp. Appl. Acarol. 83: 375-386. https://doi.org/10.1007/s10493-021-00597-4
- Nae I, Maraun M. 2023. First record of Jugatala cribelliger = Mycobates (Calyptozetes) cribelliger (Berlese, 1904) (Acari: Oribatida) in the Romanian fauna. Trav. Inst. Spéol. «Émile Racovitza». 62: 67-77. https://doi.org/10.59277/TISER.2023.05
- Nitzu E., Nae A., Băncilă R., Popa I., Giurginca A., Plăiaşu R. 2014. Scree habitats: ecological function, species conservation and spatial-temporal variation in the arthropod community. Syst. Biodiver. 12: 1-6. https://doi.org/10.1080/14772000.2013.878766
- Olszanowski, Z., Rajski, A., Niedbała, W. 1996. Catalogue of Polish fauna. Mites (Acari), moss mites (Oribatida). Sours Poznań, 34 (9): 1−243. (In Polish).
- Pipan T., López H., Oromi, P., Culver, D. 2010. Temperature variation and the presence of troglobionts in shallow subterranean habitats. J. Nat. Hist. 45(3): 253-273. https://doi.org/10.1080/00222933.2010.523797
- R Core Team. 2020. R: A language and environment for statistical computing. R Foundation for Statistical Computing. Vienna, Austria. Available at: https://www.R-project.org/
- Rendoš M., Mock A., Jászay, T. 2012. Spatial and temporal dynamics of invertebrates dwelling karstic mesovoid shallow substratum of Sivec National Nature Reserve (Slovakia), with emphasis on Coleoptera. Biologia 67: 1143-1151. https://doi.org/10.2478/s11756-012-0113-y
- Schatz, H. 1983. U. Ordn. Oribatei, Hornmilben. Catalogus faunae Austriae. Ein systematisches Verzeichnis aller auf österreichischem Gebiet festgestellten Tierarten, 9. Österreichische Akadademie der Wissenschaften. Wien. pp. 1-118.
- Schatz B, Behan-Pelletier V. 2008. Global diversity of oribatids (Oribatida: Acari: Arachnida). Hydrobiologia 595:323-328. https://doi.org/10.1007/s10750-007-9027-z
- Schatz H. 2015. Hornmilben (Acari, Oribatida) vom Fohramoos (Vorarlberg, Österreich). inatura - Forschung online. 18: 17 S.
- Schatz H. 2016. Oribatid mites (Acari, Oribatida) from the biodiversity days in South Tyrol (Prov. Bolzano, Italy). Gredleriana 16: 113-132.
- Schneider K., Renker C., Scheu S., Maraun M. 2004. Feeding biology of oribatid mites: a minireview. Phytophaga XIV: 247-256.
- Senickzak S., Senickzak A. 2011. Differentiation of external morphology of Damaeidae (Acari: Oribatida) in light of the ontogeny of three species. Zootaxa 2775: 1-36. https://doi.org/10.11646/zootaxa.2775.1.1
- Skubala P., Dethier M., Madej G., Solarz K., Mąkol J., Kaźmierschi A. 2013. How many mite species dwell in subterranean habitats? A survey of Acari in Belgium. Zool. Anz. 252: 307-318. https://doi.org/10.1016/j.jcz.2012.09.001
- Subías L.S. 2004. Listado sistemático, sinonímico y biogeográfico de los ácaros oribátidos (Acariformes: Oribatida) del mundo (excepto fósiles). Graellsia 60: 3-305. Internet update 2024. pp. 593, available from: http://escalera.bio.ucm.es/usuarios/bba/cont/docs/RO_1.pdf. https://doi.org/10.3989/graellsia.2004.v60.iExtra.218
- Vasiliu N., Ivan O., Vasiliu M. 1993. Conspectul faunistic al oribatidelor (Acarina: Oribatida) din România (The faunistic synopsis oribatids (Acarina: Oribatida) from Romania). Anuarul Muzeului Bucovinei, Fascicula Științele Naturii. Suceava. XII. pp. 82. (in Romanian).
- Vu Q.M. 2020. Oribatid mites (Acari: Oribatida) of Vietnam Species diversity and distribution characteristics. J. Sci. Nat. Sci. 65(6): 136-180. https://doi.org/10.18173/2354-1059.2020-0038
- Walter, D.E., Proctor, H.C. 1999. Mites: Ecology, Evolution, and Behaviour. Springer, Dordrecht, Netherlands. pp. 494. https://doi.org/10.1079/9780851993751.0000
- Willmann C. 1932. Milben aus Harzer Höhlen. Mitt. Höhlen- und Karstforsch. 1931-1932: 107-111.
- Willmann C. 1935. IV. Die Milbenfauna. 1. Oribatei. Zool. Jahr., Syst., Ökol. und Geogr. Tiere 66(5): 331-355.
- Willmann C. 1954. Mährische Acari hauptsächlich aus dem Gebiete des Mährischen Karstes. Československá parasitology 1: 213-272.



2025-07-02
Date accepted:
2026-03-09
Date published:
2026-03-18
Edited by:
Pfingstl, Tobias

This work is licensed under a Creative Commons Attribution 4.0 International License
2026 Nae, Ioana; Miko, Ladislav; Nae, Augustin; Băncilă, Raluca Ioana and Maraun, Mark
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



