Effect of group rearing on the life table parameters of the two-spotted spider mite Tetranychus urticae Koch (Acari: Tetranychidae)
Hayder, Tasfia  1
; Meem, Mst. Masuma Momtaj
1
; Meem, Mst. Masuma Momtaj  2
; Tonni, Farhana Akter
2
; Tonni, Farhana Akter  3
; Nikson, A. K.
3
; Nikson, A. K.  4
; Gotoh, Tetsuo
4
; Gotoh, Tetsuo  5
and Ullah, Mohammad Shaef
5
and Ullah, Mohammad Shaef  6
6
1Laboratory of Applied Entomology and Acarology, Department of Entomology, Bangladesh Agricultural University, Mymensingh 2202, Bangladesh.
2Laboratory of Applied Entomology and Acarology, Department of Entomology, Bangladesh Agricultural University, Mymensingh 2202, Bangladesh.
3Laboratory of Applied Entomology and Acarology, Department of Entomology, Bangladesh Agricultural University, Mymensingh 2202, Bangladesh.
4Laboratory of Applied Entomology and Acarology, Department of Entomology, Bangladesh Agricultural University, Mymensingh 2202, Bangladesh.
5Faculty of Economics, Ryutsu Keizai University, Ryugasaki, Ibaraki 301-8555, Japan.
6✉ Laboratory of Applied Entomology and Acarology, Department of Entomology, Bangladesh Agricultural University, Mymensingh 2202, Bangladesh.
2026 - Volume: 66 Issue: 1 pages: 118-133
https://doi.org/10.24349/uhbs-wb0jOriginal research
Keywords
Abstract
Introduction
The two-spotted spider mite, Tetranychus urticae Koch (Acari: Tetranychidae), an acarine of the Tetranychidae family, is a significant polyphagous and cosmopolitan pest posing a great threat to various crops in fields and greenhouses, with more than 1500 host plant species worldwide (Migeon and Dorkeld 2024). Consequently, much of the research effort has been directed toward managing this species, often overlooking its behavioral aspects, particularly its social behavior.
Tetranychus urticae is widely used as a model species in laboratory studies of arthropod life history and population dynamics, including life table analyses (Shih et al. 1976). Group living in arthropods offers numerous evolutionary benefits for both individuals and the collective, such as improved access to diet and mates, as well as enhanced protection from predators (Ranta et al. 1993; Dyer 2000; Wertheim et al. 2005; Costa 2006; Mori and Saito 2006; Sato and Saito 2006; Saemi et al. 2017; Jin et al. 2024). Furthermore, group formation has been shown to provide a significant advantage in terms of protection against predation for individuals (Wertheim et al. 2005). However, cooperative behavior also presents drawbacks, including the necessity to share resources such as diet, mates, and living space, which can potentially lead to conflicts among individuals (Ranta et al. 1993).
The creation of a shared, spider-like web illustrates the specific advantages of group living. In T. urticae, collective web spinning results in denser structures compared to those produced by solitary individuals. A dense silk web provides better defense against environmental factors such as rain (Davis 1952), predator attacks (McMurtry et al. 1970), and pesticides (Ashley et al. 2006). Although T. urticae is a well-established model species for life-history and population biology studies, research explicitly addressing the effects of group rearing or conspecific density on individual performance remains relatively limited. Previous research has demonstrated a decrease in male development periods with increasing individual density (Van Impe 1984, 1985), suggesting possible group effects. However, a comprehensive understanding of the implications of social organization in T. urticae populations is still lacking.
Life tables serve as a crucial instrument for collecting comprehensive data on insect and mite population dynamics, including growth, survival, reproduction, and the intrinsic rate of increase as they provide fundamental insights into population dynamics and aid in developing more targeted and effective management strategies (Bayu et al. 2017; Ullah et al. 2020; Chi et al. 2023). However, conventional female age-specific life tables miss critical aspects, such as the male population, stage differentiation, and individual variations in juvenile development, leading to inaccuracies in population descriptions (Chi 1988; Huang and Chi 2013). To address these shortcomings, the age-stage, two-sex life table model has become the preferred method. This advanced approach incorporates data from both sexes, reducing inherent errors and offering a more comprehensive understanding of population dynamics (Sugawara et al. 2017; Rismayani et al. 2021; Chi 2024).
Although T. urticae is a major prey item for a wide range of natural enemies, it is also a widely used model species in laboratory life-history studies, where individuals are commonly reared singly under enemy-free conditions. However, the effects of conspecific density and group living on life-history traits and population growth parameters under such controlled conditions remain poorly quantified. This study addresses this gap by experimentally comparing single and group rearing to evaluate density-dependent effects on the biology of T. urticae. We hypothesized that individuals residing in groups would exhibit altered developmental patterns, reproductive rates, and survival probabilities compared to solitary individuals. Specifically, we expected that group living would lead to accelerated development, increased fecundity, and enhanced survival rates among T. urticae individuals. By employing the age-stage, two-sex life table methodology, we aimed to obtain a better understanding of the population dynamics of T. urticae and elucidate the role of group living in shaping its life history traits. Through this study, we aimed to enhance the fundamental understanding of the ecological dynamics and adaptive strategies of T. urticae under conditions devoid of external selective pressures, focusing specifically on group effects and their role in shaping population behavior.
Materials and methods
Rearing and experimental conditions
The green form of the two-spotted spider mite, T. urticae, was initially collected from Dahlia (Dahlia × hortensis) in Boilor, Mymensingh, Bangladesh (24°62′N, 90°41′E) on 25 March 2022. The species was identified morphologically under a compound microscope using the standard taxonomic keys provided by Meyer (1989), based on the shape and size of aedeagus: the terminal knob is ca. 2.5-2.6 µm diameter, and approximately as long as 1/4 the length of the dorsal margin of the shaft. The mites were maintained under laboratory conditions on fresh leaf discs (approximately 16 cm²) of hyacinth bean (Dolichos lablab L.), which served as the host plant. The bean plants were grown from certified seeds sourced from the Bangladesh Agricultural Research Institute (BARI) in plastic pots (25 cm diameter) containing a sterilized soil-sand-compost mixture (2:1:1) in a greenhouse. Plants were grown under natural light with periodic irrigation, and no pesticides were applied. Fully expanded, untreated leaves from 3–4-week-old plants were harvested and cut into discs for experimental use. The leaf discs of 16 cm2 were placed on water-saturated sponges in plastic Petri dishes (90 mm diameter, 20 mm depth). The conditions were kept at 25 ± 1 °C, 70 ± 10% relative humidity, and 16L: 8D photoperiod. To ensure a consistent water supply and prevent mite escape, the edges of the bean leaves were enclosed with moist tissue paper.
Life table experiment


For experiments with T. urticae with one, group rearing of 2, group rearing of 4, and group rearing of 6 individuals, the leaf discs diameters were 8, 11, 16, and 20 mm, respectively, to provide a density of approximately 1 female per 50 mm2 on each disc (Le Goff et al. 2010). This enabled us to assess the influence of conspecific presence separately from density-related factors such as food availability and living space per individual. The leaf discs with mites were held at 25 °C under a 16L: 8D photoperiod with 70 ±10% RH. The leaf discs were replaced every 5-7 days to ensure a continuous fresh food supply and optimal conditions for mite development and reproduction. One, group rearing of 2, group rearing of 4 and group rearing of 6 newly laid eggs were placed on fresh 8, 11, 16, and 20 mm bean leaf discs, respectively. Developmental stages were noted at every 12-hour intervals until all individuals reached adulthood. The sex of the spider mites was determined at the teleiochrysalis stage [this stage does not move], prior to adult emergence beforereaching maturity, and an adult male was introduced with each teleiochrysalis female to ensure timely mating. At this stage, females were distinguished from males based on their larger body size, and more rounded opisthosoma (abdomen), whereas males were smaller, and more tapered posteriorly. The male remained on the disc all throughout the experiment and was replaced with a fresh one if it died. Developmental data for T. urticae males and females were recorded at 12-hour intervals, with the stage structure (i.e., the number of individuals in each stage) tracked separately for each mite. Because immature stages of T. urticae are relatively sedentary and disc sizes were adjusted to maintain low crowding, individuals could be reliably distinguished throughout the experiment. Developmental stage transitions, survival, and reproductive events were recorded separately for each mite, ensuring that individual life-history data did not overlap. This procedure followed established methodologies for monitoring individually identifiable mites under group-rearing conditions (Jin et al., 2024). In the group-rearing treatments, one adult male was paired with each teleiochrysalis female to ensure successful and standardized mating. Although natural populations of T. urticae typically exhibit a female-biased sex ratio, a 1:1 operational sex ratio is commonly used in laboratory life table studies to prevent male limitation and to allow unbiased estimation of female life-history and demographic parameters. Each male remained on the disc with the corresponding female throughout her reproductive period. In the event of a male's death, he was promptly replaced with a new adult male from the mass culture, ensuring the continuous availability of mates. This practice was implemented to minimize disruptions in mating and ensure consistent reproductive conditions throughout the experiment. The number of individuals tested was 68 for single rearing, 68 for double rearing, 76 for quadruple rearing, and 84 for sextuple rearing.
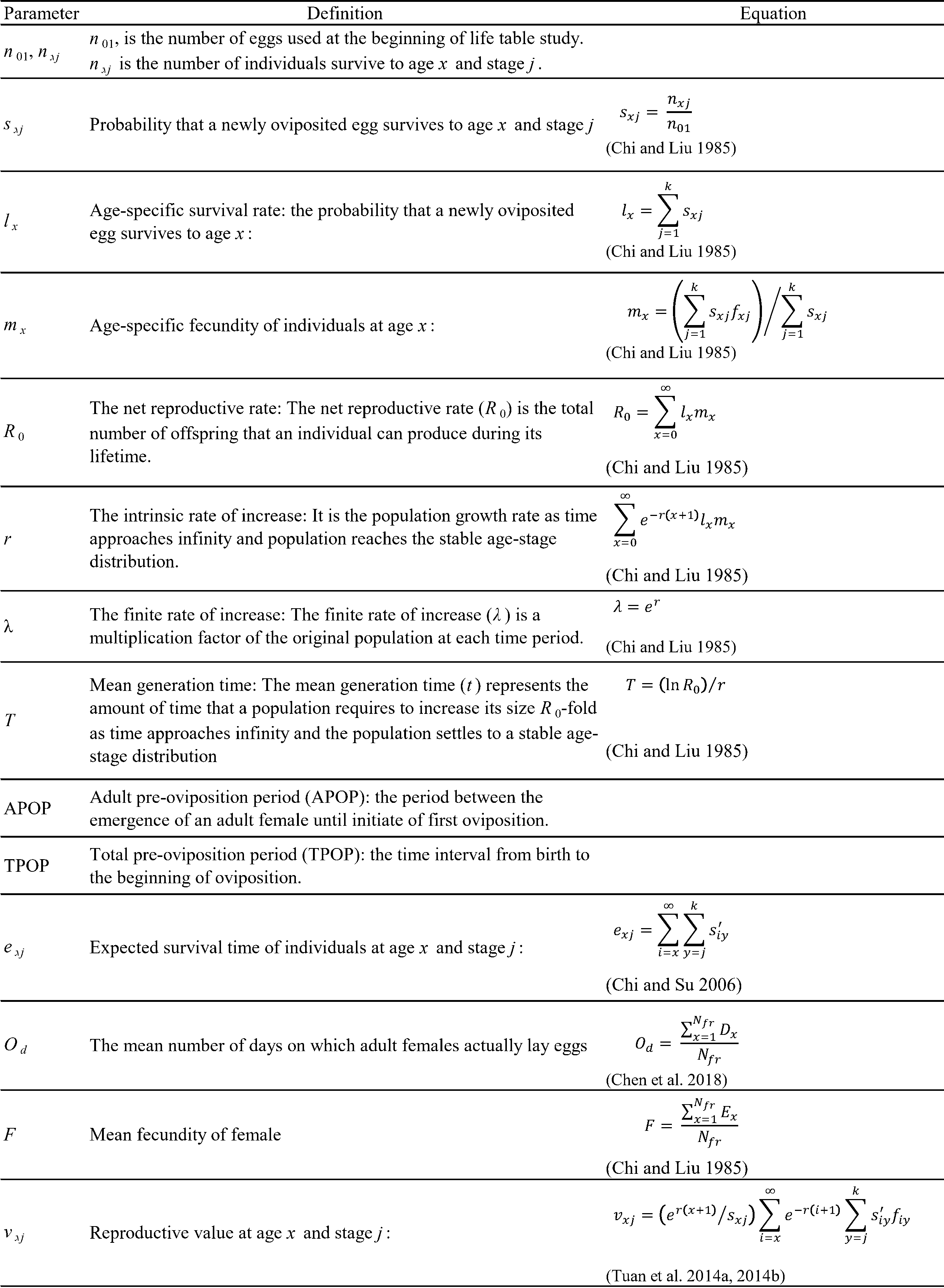
Females and males that emerged from the previously described experiments under similar environmental conditions were selected to assess the immature development, reproductive characteristics and longevity. Twice per day observations were made under a stereomicroscope to record the number of eggs laid by each female T. urticae. This data was used to assess the oviposition period, daily laid eggs, total eggs laid per female, and the longevity of both females and males until all mites died. Since the number of females on each disc (one, two, four, or six) is known, the average number of eggs per female was counted though dividing the total egg by the number of females on that disc (Chi et al. 2023). Survival and fecundity data were used to assess the life table parameters according to the age-stage two-sex life table theory (Table 1) (Chi 2024).
Statistical analysis
The age-stage survival rates and fecundity of different spider mite numbers (1, 2, 4, and 6) were assessed, and the life table parameters (Table 1) under each rearing condition were analyzed by using the TWOSEX-MS Chart computer program (Chi 2024). Bootstrap analysis was used to estimate the variances and standard errors of the population parameters . Given that bootstrap analysis relies on random resampling, results from a limited number of replications may show fluctuations in means and standard errors. To minimize such variability, we conducted 100,000 bootstrap iterations. Paired bootstrap test (Efron and Tibshirani 1993) were used to examine the differences among the rearing number treatments.
Results
Immature development
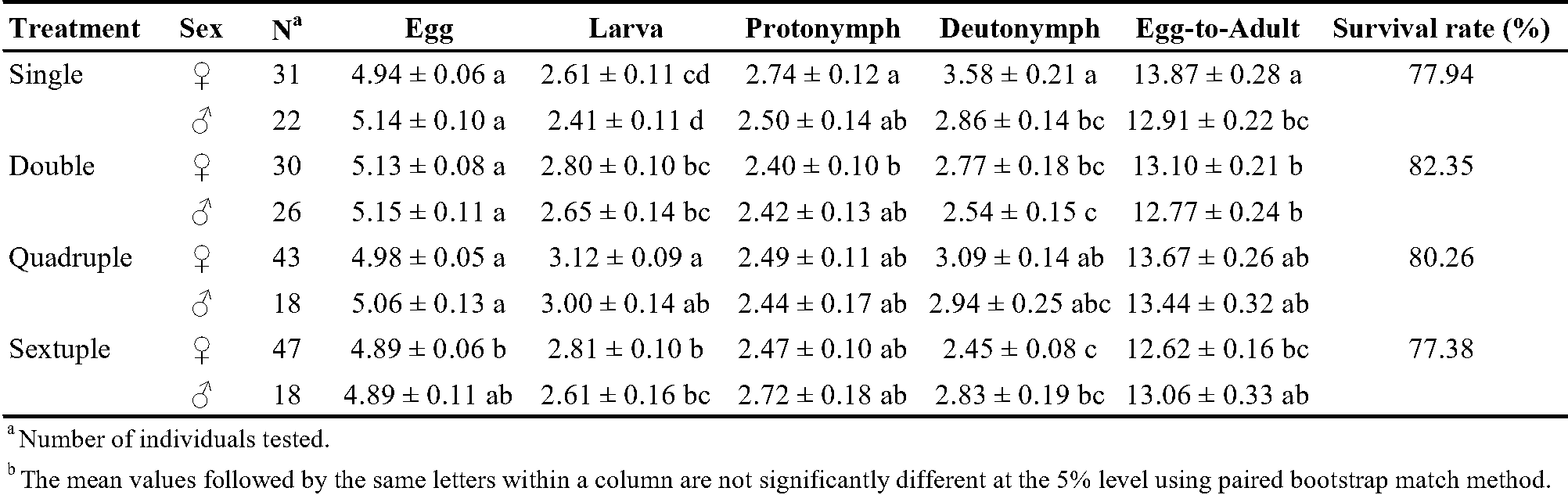


Both male and female T. urticae successfully completed development under all rearing densities at 25 °C, confirming that the experimental conditions were suitable for evaluating density-dependent effects on life-history traits and life table parameters. The duration of the immature development period for T. urticae was influenced by both the number of individuals in the rearing unit and the sex of the mites. Egg duration varied significantly with group rearing number but without a trend. The larval duration varied significantly with group rearing number but without a trend. The deutonymph period was highest in single rearing and lowest in sextuple rearing. Egg-to-adult female duration was lower in group rearing than in single rearing (Table 2).
Adult longevity and reproduction
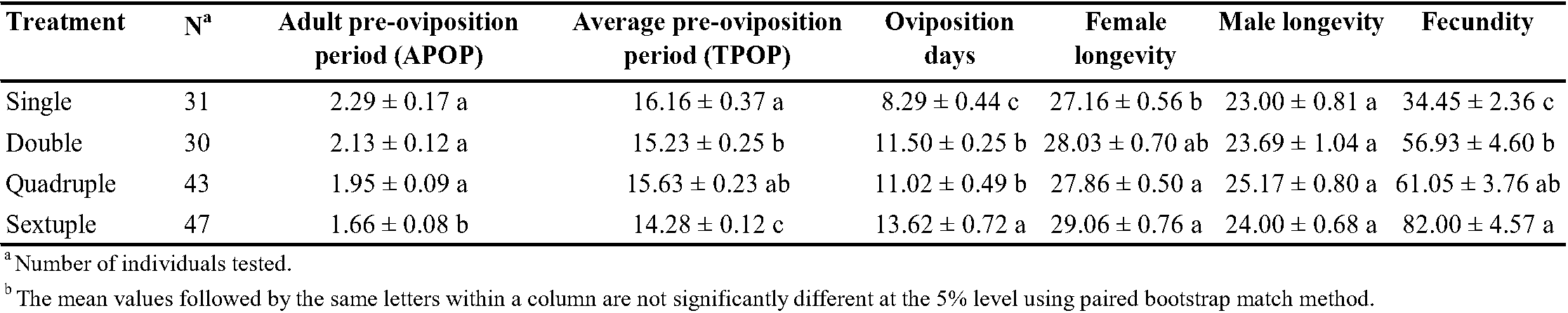


Rearing number significantly affected the pre-oviposition, oviposition, female adult longevity, fecundity, and daily egg production/female (Table 3). Tetranychus urticae had a shorter pre-oviposition period in sextuple rearing than from single, double, and quadruple rearing. The oviposition days showed a similar trend; oviposition days were highest in sextuple rearing, and the eggs per female increased with the increasing rearing number. Female longevity was also higher in group rearing than in single rearing (Table 3). Rearing number had no significant effect on male longevity, but it did significantly affect the number of eggs per female. The highest number of eggs per female (82 eggs/female) was found in sextuple rearing, followed by quadruple, double, and single rearing (Table 3).
Age stage and life cycle
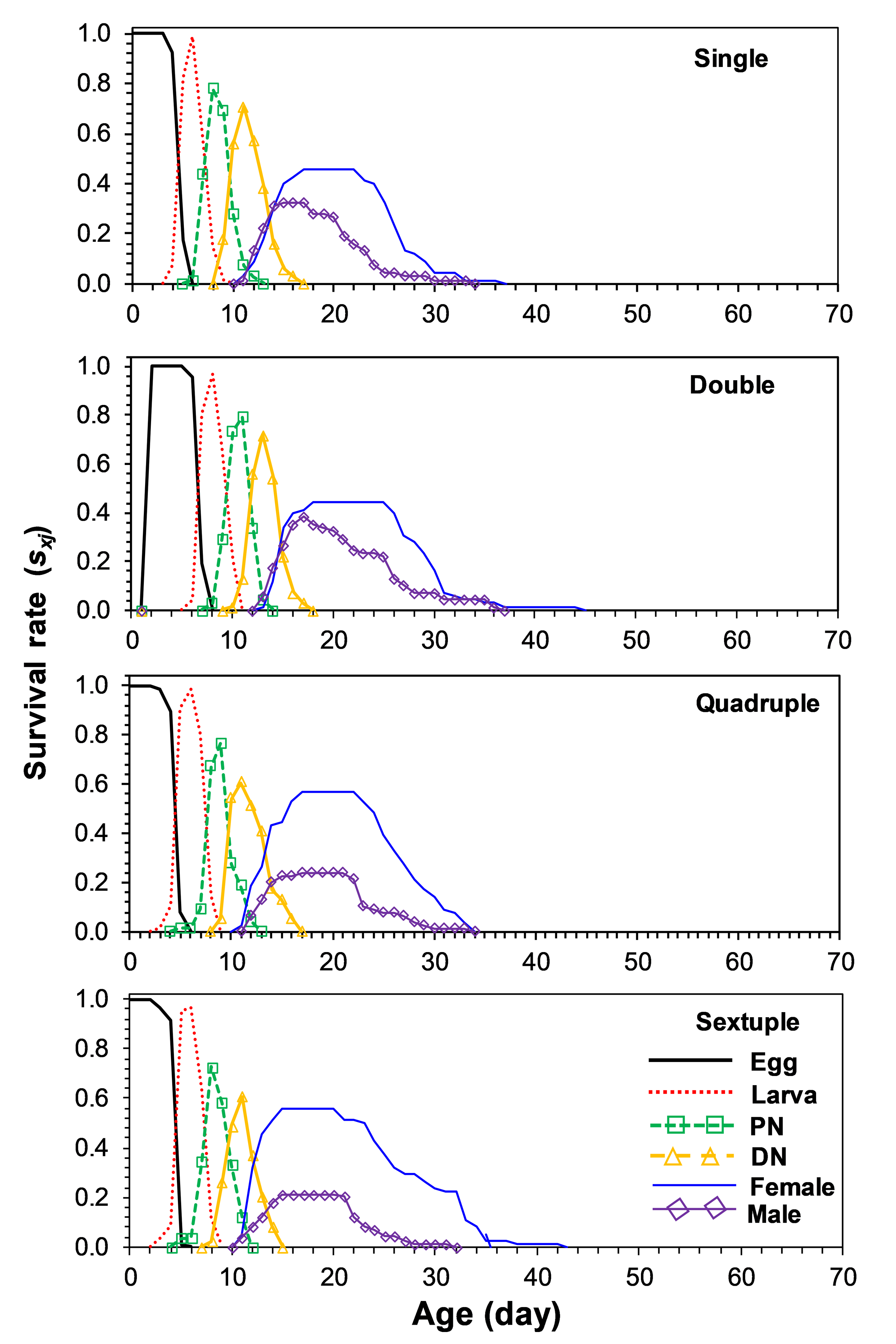


The age-stage specific survivorship (sxj) curves are presented in Figure 1. The overlapping of the curves is a result of the difference in developmental rates amongst individuals.
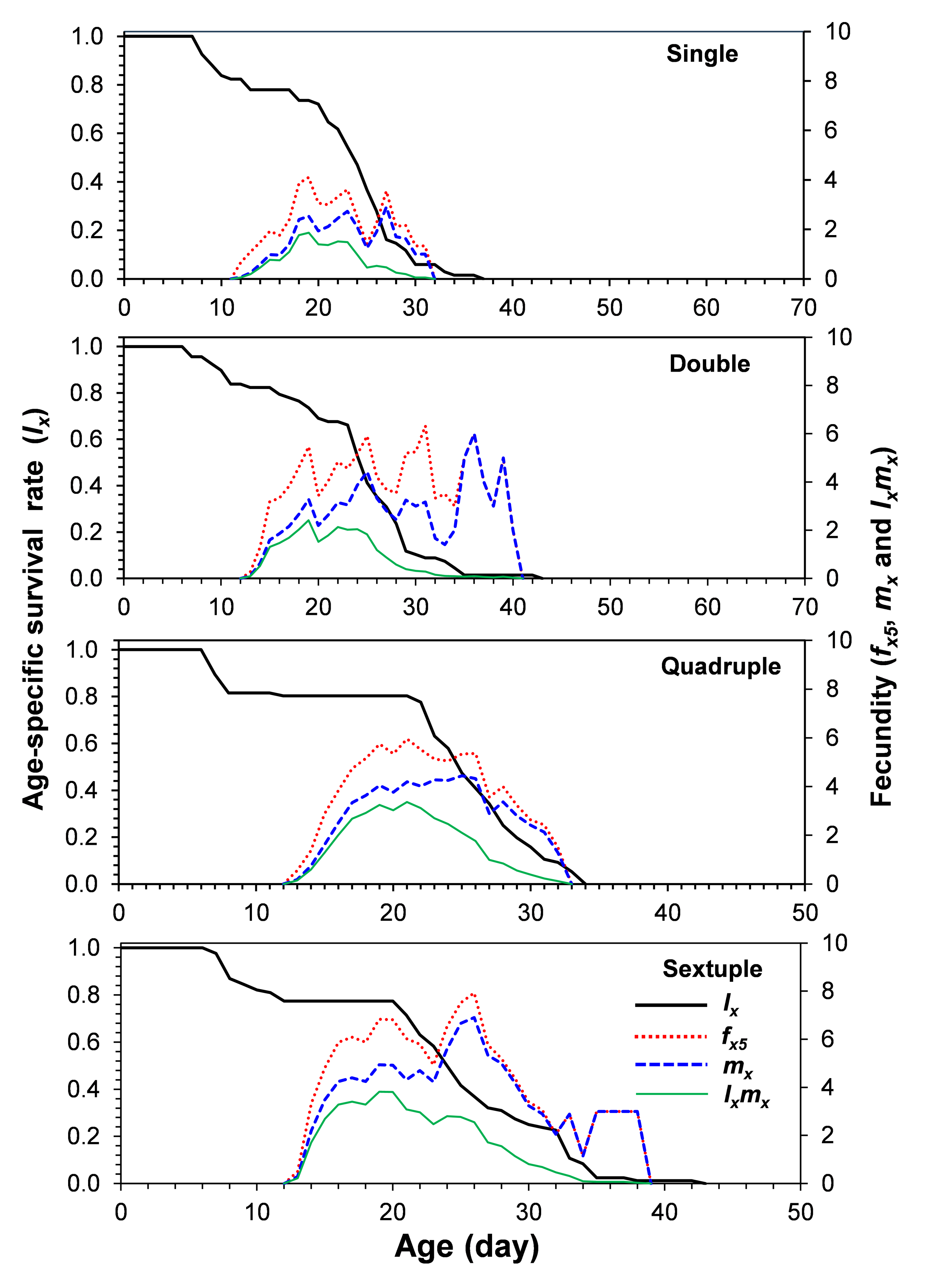


Figure 2 shows the age-specific survival rate (lx), age-specific fecundity (mx), and age-specific maternity (lxmx) of T. urticae on each rearing number. Adult females in the single, double, quadruple, and sextuple rearing begin to die on days 8, 7, 7, and 7, respectively. The age-specific fecundity (mx) of T. urticae on different rearing numbers fluctuated all over the oviposition period, showing an irregular pattern. Oviposition in T. urticae females started on days 12, 13, 13, and 13 on single, double, quadruple, and sextuple rearing, respectively.
Life table parameters
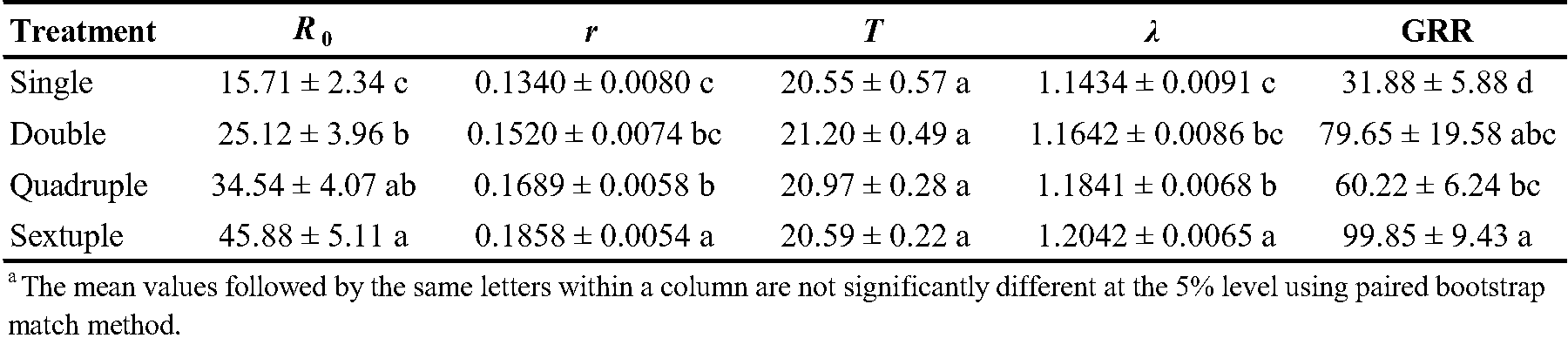


The life table parameters of T. urticae were significantly influenced by the rearing number (Table 4). Key metrics such as the net reproduction rate (R0), intrinsic rate of increase (r), finite rate of increase (λ), and gross reproduction rate (GRR) all exhibited significant variations with different rearing numbers. The net reproduction rate (R0) increased significantly with increasing rearing number. Intrinsic rate of increase (r) showed a similar trend, being highest in sextuple rearing and lowest in single rearing. Rearing number had no significant effect on mean generation time (T). The finite rate of increase (λ) increased with increasing rearing number. Gross reproduction rate (GRR) values showed a similar trend, being highest in sextuple rearing and lowest in single rearing (Table 4).
Life expectancy
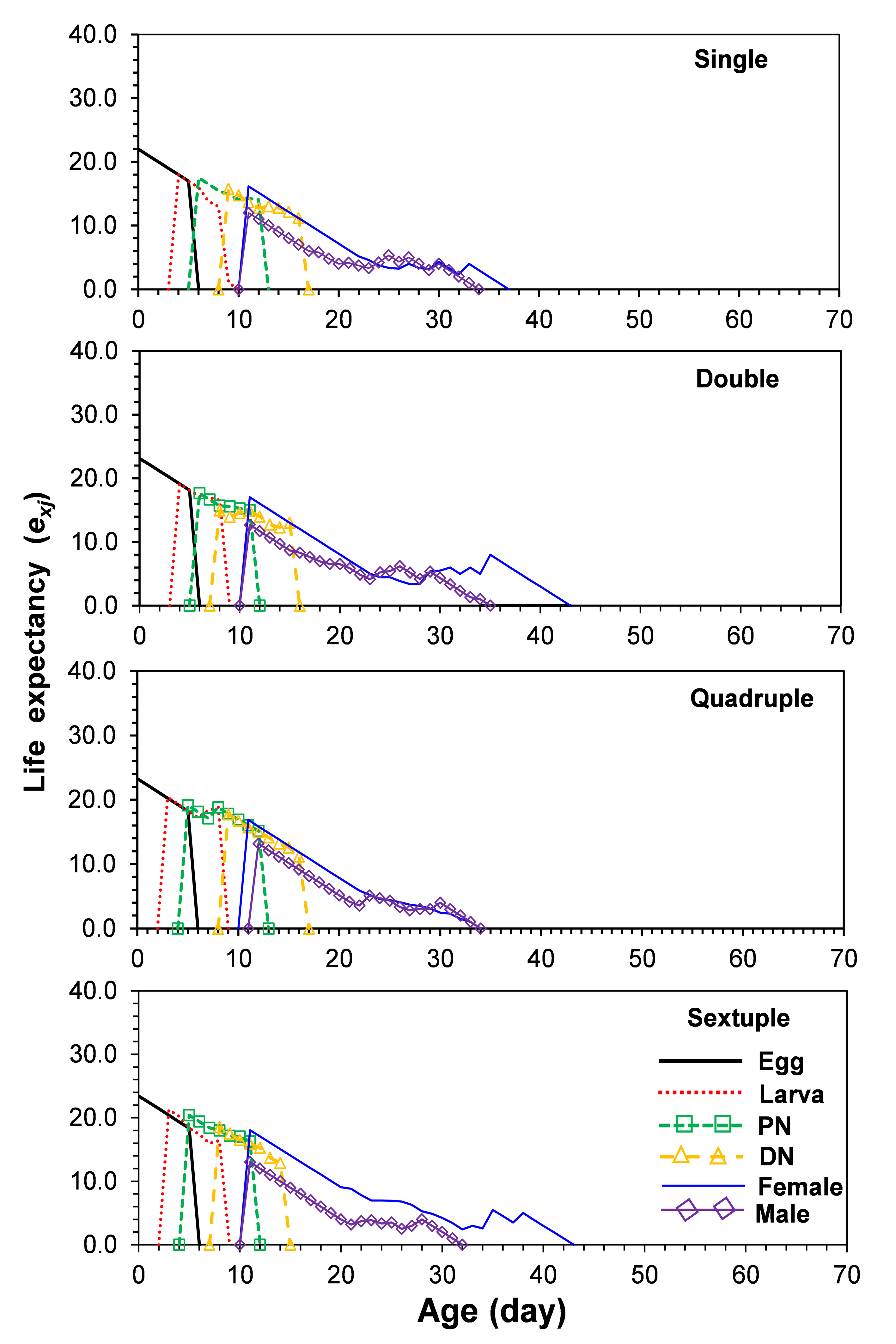


The life expectancies of a newborn egg reared on single, group rearing of 2, group rearing of 4, and group rearing of 6 individuals rearing were 21.9 23.1, 23.3, and 23.4 days, respectively (Figure 3). The highest life expectancy of adults reared on single, group rearing of 2, group rearing of 4, and group rearing of 6 individuals rearing at 25 °C was 11 days for females, and 11, 11, 12, and 11 days, respectively, for males (Figure 3).
Reproductive values
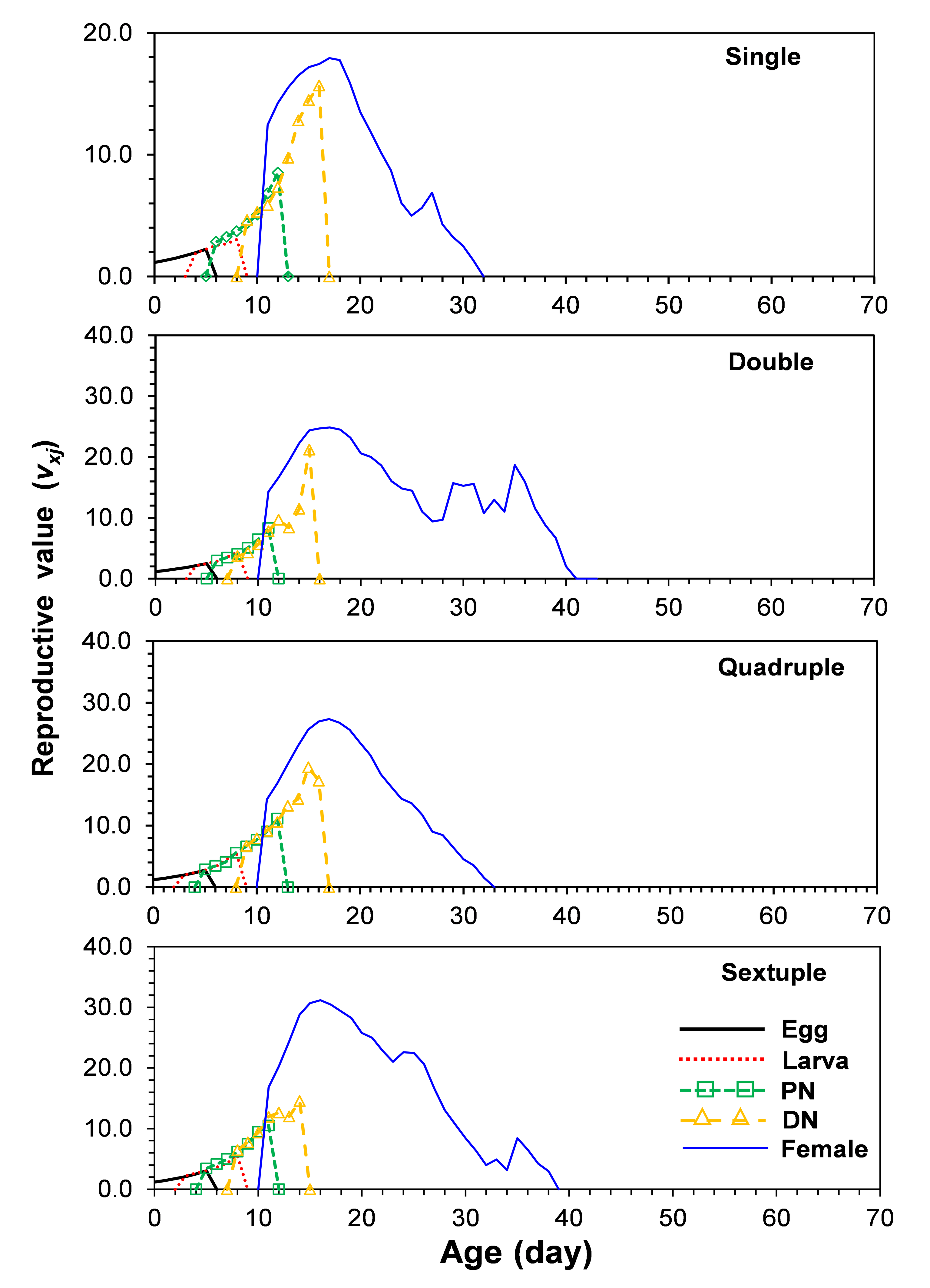


The reproductive values (vxj) of T. urticae individuals at age and stage are illiustrated in Figure 4. The vxj of a neonatal egg on single, double, quadruple, and sextuple rearing were 1.1, 1.2, 1.2, and 1.2, respectively (Figure 4). Adult females near the top of reproduction contribute more to the population than those of females at other ages and stages. The peak vxj of adult females on single, group rearing of 2, group rearing of 4, and group rearing of 6 individuals rearing were 17.9, 24.9, 27.3, and 31.2 days, respectively.
Discussion
The social environment has a significant influence on an individual's behavior and physiology across various species, including both vertebrates and invertebrates (Chen and Sokolowski 2022). These effects, commonly referred to as ''group effects,'' involve the costs and benefits associated with living in a group. These influences can stem from numerical effects and synergistic interactions among individuals. This study investigated the population dynamics of spider mites, which can be useful in the management of spider mites by providing a clear understanding of ecological adaptation without adversely affecting the environment.
Present studies showed that the number of individuals reared together significantly influenced various aspects of T. urticae development, such as the duration of developmental stages, egg-laying period, lifespan, reproductive output, and key life table parameters. The intrinsic rate of increase was notably higher in sextuple rearing compared to quadruple, double, and single rearing, indicating that sextuple rearing enhanced population growth of T. urticae over other rearing densities. These findings underscore the impact of rearing number on development, reproductive capacity, and the population dynamics of T. urticae. They also highlight the need to consider rearing conditions in assessing the ecological traits and management strategies of this species.
In spider mites, males typically complete development faster than females, a pattern widely reported in T. urticae and other tetranychid species and generally attributed to the smaller size and lower developmental investment required for males (Sabelis 1981, Razmjou et al. 2009). Consistent with this expectation, males exhibited shorter egg-to-adult developmental times than females in the single, double, and quadruple rearing treatments in the present study. However, this pattern was reversed under sextuple rearing conditions, where females developed slightly faster than males, although there was no statistical difference between males and females. This shift suggests that developmental rates in T. urticae are not fixed but can respond plastically to conspecific density. Higher local densities may differentially affect the sexes through mechanisms such as sex-specific sensitivity to chemical cues, altered feeding dynamics, or interference among individuals during immature stages. Females, which benefit more directly from rapid maturation under favorable conditions, may accelerate development in response to group-associated cues, whereas males may experience greater developmental costs or interference at higher densities. Although survival remained high across treatments, these subtle shifts in developmental timing highlight the complexity of density-dependent effects on sex-specific life-history traits in T. urticae. Although spider mite embryos are physically isolated within the egg, density-dependent differences in egg development may arise through indirect mechanisms rather than direct conspecific interactions. One possible explanation is maternal effects, whereby females exposed to higher conspecific densities adjust egg provisioning, hormonal signaling, or egg physiology prior to oviposition, resulting in faster embryonic development. Maternal environment–dependent effects on offspring development have been documented in arthropods and may represent an adaptive response to perceived population conditions.
In addition, group rearing may modify the microenvironment of the leaf disc, for example through localized changes in humidity, temperature, or plant tissue condition caused by collective feeding activity. Such microenvironmental changes could influence embryonic development rates despite the physical isolation of the embryo within the egg. While these mechanisms cannot be disentangled with the present experimental design, the observed reduction in egg-stage duration under higher rearing densities suggests that embryonic development in T. urticae may be indirectly sensitive to conspecific density via maternal or environmental pathways rather than direct social interactions.
Group rearing has also been shown to have positive effects on various aspects of T. urticae behavior and physiology. For example, Le Goff et al. (2010) demonstrated that females exhibited significantly higher daily reproduction and web production when reared in groups of two to six individuals compared to solitary rearing conditions, and Yano (2008) showed that this species can form colonies either by dispersing individually or in groups of two or more, which suggests a propensity for group living behavior in this species.
The presence of conspecifics in T. urticae colonies has been proposed to influence life history traits compared to solitary individuals. Our study corroborates these findings, as we observed increased fecundity, oviposition days, female longevity, and survival in T. urticae reared in groups under experimental conditions. This indicates that individuals are able to sense the presence of others of the same species through either direct or indirect interactions, potentially mediated by chemical cues, such as pheromones or tactile perception (Prokopy and Duan 1998; Prokopy and Reynolds 1998).
Furthermore, a study by Lihoreau and Rivault (2008) found that group living enhances ootheca production and nymphal development in species that aggregate, such as the cockroach Blattella germanica (L.). In contrast, non-aggregating cockroaches like B. lituricollis (Walker) and B. biligata (Walker) were unaffected by group living. Similar trends have been reported in other taxa. For example, Krause and Ruxton (2002) discussed how sociality can increase reproductive success in fish and insect groups via stress reduction and enhanced resource access. In spider mites, Schausberger et al. (2021) found that group size influences feeding behavior and dispersal in Tetranychus spp., with potential effects on fitness outcomes through cooperative behaviors and local aggregation in response to both direct and indirect cues. Likewise, Clotuche et al. (2011) showed that aggregation behavior in T. urticae promotes synchronized development and influences dispersal strategies. Conversely, not all group-living species benefit equally from conspecific presence. For instance, Strodl and Schausberger (2012) demonstrated that in the predatory mite Phytoseiulus persimilis Athias-Henriot, the advantages of group living – such as enhanced predator evasion – depend on social familiarity between individuals. These findings collectively suggest that the effects of group living are context-dependent and shaped by aggregation tendency, social interaction, and resource distribution. Additionally, studies on social spider mites have demonstrated that larger colony sizes enhanced the likelihood of offspring reaching maturity and successfully reproducing (Avilés and Tufino 1988; Salomon and Lubin 2007). These findings collectively highlight the importance of group dynamics in influencing the behavior and fitness of T. urticae populations.
Our study revealed a significant positive impact of group rearing on the fertility of T. urticae. The higher fecundity observed under group-rearing conditions cannot be explained by increased mating opportunities, as a 1:1 male-to-female ratio was maintained across all treatments. Instead, the results suggest that conspecific presence influences reproductive output through other density-dependent mechanisms. Group living may enhance fecundity via chemical or tactile cues, such as aggregation pheromones or other social signals, which have been reported in spider mites and may affect reproductive physiology. Additionally, the presence of conspecifics may reduce stress associated with isolation or modify the microenvironment on the leaf disc, thereby promoting oviposition. Notably, increased egg production in the sextuple group occurred despite the potential for interference among individuals, including possible disruption of mating or oviposition due to crowding. This indicates that any negative effects of increased conspecific density were outweighed by positive group-related effects. Together, these findings demonstrate that group rearing influences reproduction in T. urticae through mechanisms beyond mating frequency alone. In the present study, the highest number of eggs per female (82 eggs) was observed under sextuple rearing conditions, whereas egg production decreased with decreasing group size and was lowest under single rearing. Similar density-dependent effects on fecundity have been reported previously in T. urticae (Oku et al. 2009). The increased egg production in group-reared T. urticae individuals can be attributed to various factors. For instance, the presence of conspecifics may induce life history trait modifications, facilitating investment in egg production (number and volume). Individuals of group-living can advantage from the presence of conspecifics species (i.e. Allee effect by gaining easier access to food, defense against predators, improvement of the environment) (Courchamp et al. 2008; Clotuche et al. 2013). Additionally, the energy saved by not needing to construct a web, due to the presence of an existing one, may be redirected towards reproductive activities such as egg laying (Holbrook et al. 2000).
Studies on other arthropods have provided further insights into the physiological mechanisms underlying group effects on reproductive output. For instance, group raising in B. germanica was found to enhance oocyte development by reducing the brain's control of juvenile hormone synthesis (Regev and Cone 1976; Thind and Edwards 1990). While our findings highlight the positive influence of group rearing on T. urticae fertility, it's worth noting that group effects may vary among species. Saemi et al. (2017) reported that group rearing did not affect the life table and predation rate of predatory mites. However, our study demonstrated significant effects of group rearing on life table parameters in T. urticae. These differences could stem from variations in experimental setups and mite behavior, which emphasizes the importance of species-specific investigations.
Jin et al. (2024) recently used an age-stage, two-sex life table approach to examine development of the papaya mealybug, Paracoccus marginatus Williams and Granara de Willink (Hemiptera: Pseudococcidae), on jatropha plants, Jatropha integerrima Jacq. (Euphorbiaceae), using both individual and group rearing methods. They noted that the developmental durations of individually-reared insects were significantly longer than those reared in groups. Although the mean fecundity and net reproductive rate of individually-reared insects were notably higher compared to group-reared insects, there were no significant differences in population growth rate parameters, i.e., the intrinsic and finite rates of increase (r and λ, respectively). Understanding the effects of group living is crucial for pest control, as emphasized by Myllymäki (1975). Social behaviors drive the spatial distribution, dispersal movement, and life history features of pests. These behaviors can affect the instant outcome of control measures, as individuals in groups may respond quickly to induced mortality. Disrupting the usual behavioral mechanisms of social pest species, such as by luring with aggregative or dispersal pheromones, could be a viable control strategy. A key limitation of group-rearing approaches is that individual-level life-history data cannot be tracked throughout the entire life cycle with the same precision as in individually reared treatments, because observations are typically based on stage structure or averaged across multiple individuals within a replicate (Chi et al., 2023; Jin et al., 2024). Despite this limitation, group-reared life tables provide valuable insights into population-level characteristics and may better reflect natural population dynamics than individual-based designs (Fazel-Dinan et al., 2022; Li et al., 2021; Wang et al., 2022).
Controlling mites in greenhouses and fields poses challenges due to their complex nests and high reproductive rates. One potential approach is to deploy traps or baits infused with mite pheromones to lure adults, nymphs, and larvae into an area devoid of protective webs, thereby effectively trapping them. Our results indicate that increased local density can enhance reproductive output in T. urticae, suggesting that management strategies that inadvertently promote aggregation without effective removal could risk increasing population growth. In contrast, pheromone-based approaches, when used for attraction followed by containment or mortality, are intended to reduce population size rather than alter reproductive performance. Dispersing or exploiting aggregation pheromones as part of an effective trapping or removal strategy, as discussed by Dicke (2008), may therefore offer a promising component of integrated spider mite management.
In overcrowded situations, conspecifics may negatively impact population dynamics, leading to a negative group effect. Further research is needed to unravel the underlying physiological and behavioral mechanisms contributing to group effects. Insights into these mechanisms could speed up the development of more targeted and effective pest management strategies (Grassé 1946; Prokopy and Duan 1998; Prokopy and Reynolds 1998; Holbrook et al. 2000).
This study highlights the influence of rearing numbers on the developmental and life table parameters of T. urticae. Specifically, it demonstrates that sextuple rearing conditions result in notably higher intrinsic rates of increase compared to single, double, and quadruple rearing. These findings underscore the importance of considering rearing conditions – particularly conspecific density and group structure – when studying the population biology of T. urticae. While this study was conducted under controlled laboratory conditions and does not directly evaluate pest management outcomes, the observed effects of group size on development and reproduction highlight potential mechanisms that could influence population build-up in agricultural settings. As such, this research provides a biological basis for future investigations into how group dynamics might interact with pest control strategies. Further applied studies, including field validation, will be essential to translate these insights into effective management practices for T. urticae and related pest species.
Acknowledgments
Tasfia Hayder was awarded the National Science and Technology Fellowship from the Ministry of Science and Technology (MoST), Government of the People's Republic of Bangladesh.
Financial interests
The authors have no relevant financial or non-financial interests to disclose.
Author contribution statements
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Tasfia Hyder and Mohammad Shaef Ullah. The first draft of the manuscript was written by Mohammad Shaef Ullah and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Conflicts of interest
The authors declared no conflict of interest.
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
- Ashley J.L., Herbert D.A., Lewis E.E., Brewster C.C., Huckaba R. 2006. Toxicity of three acaricides to Tetranychus urticae (Tetranychidae: Acari) and Orius insidiosus (Anthocoridae: Hemiptera). J. Econ. Entomol., 99: 54-59. https://doi.org/10.1093/jee/99.1.54
- Avilés L., Tufino P. 1998. Colony size and individual fitness in the social spider Anelosimus eximius. Am. Nat., 152: 403-418. https://doi.org/10.1086/286178
- Bayu M.S.Y.I., Ullah M.S., Takano Y., Gotoh T. 2017. Impact of constant versus fluctuating temperatures on the development and life history parameters of Tetranychus urticae (Acari: Tetranychidae). Exp. Appl. Acarol. 72: 205-227. https://doi.org/10.1007/s10493-017-0151-9
- Chen G.M., Chi H., Wang R.C., Wang Y.P., Xu Y.Y., Li X.D., Yin P., Zheng F.Q. 2018. Demography and uncertainty of population growth of Conogethes punctiferalis (Lepidoptera: Crambidae) reared on five host plants with discussion on some life history statistics. J. Econ. Entomol., 111: 2 143-2152. https://doi.org/10.1093/jee/toy202
- Chen M., Sokolowski M.B. 2022. How social experience and environment impacts behavioural plasticity in Drosophila. Fly (Austin), 16: 68-84. https://doi.org/10.1080/19336934.2021.1989248
- Chi H. 1988. Life table analysis incorporating both sexes and variable development rate among individuals. Environ. Entomol., 17: 26-34. https://doi.org/10.1093/ee/17.1.26
- Chi H. 2024. TWOSEX-MSChart: a computer program for the age stage, two sex life table analysis National Chung Hsing University, Taichung, Taiwan. http://140.120.197.174/Ecology/download/TWOSEX-MSChart.rar
- Chi H., Liu H. 1985. Two new methods for the study of insect population ecology. Bull. Inst. Zool. Acad. Sin., 24(2): 225-240.
- Chi H., Kavousi A., Gharekhani G., Atlihan R., Özgökçe M.S., et al. 2023. Advances in theory, data analysis, and application of the age-stage, two-sex life table for demographic research, biological control, and pest management. Entomol. Gen. 43: 705-732. https://doi.org/10.1127/entomologia/2023/2048
- Chi H., Su H.-Y. 2006. Age-stage, two-sex life tables of Aphidius gifuensis (Ashmead) (Hymenoptera: Braconidae) and its host Myzus persicae (Sulzer) (Homoptera: Aphididae) with mathematical proof of the relationship between female fecundity and the net reproductive rate. Environ. Entomol., 35: 10-21. https://doi.org/10.1603/0046-225X-35.1.10
- Clotuche G., Mailleux A.C., Astudillo Fernández A., Deneubourg J.L., Detrain C., Hance T. 2011. The formation of collective silk balls in the spider mite Tetranychus urticae Koch. PLoS One, 6(4): e18854. https://doi.org/10.1371/journal.pone.0018854
- Clotuche G., Mailleux A.C., Yano S., Detrain C., Deneubourg J.L., Hance T. 2013. Settlement decisions by the two-spotted spider mite Tetranychus urticae. C. R. Biol., 336: 93-101. https://doi.org/10.1016/j.crvi.2013.02.006
- Costa J.T. 2006. The other insect societies. Harvard University Press, Cambridge, MA. 767 pp. https://doi.org/10.4159/9780674271616
- Courchamp F., Berec L., Gascoigne J. 2008. Allee effects in ecology and conservation. Oxford University Press, New York. https://doi.org/10.1093/acprof:oso/9780198570301.001.0001
- Davis D.W. 1952. Influence of population density on Tetranychus multisetis. J. Econ. Entomol. 45: 652-654. https://doi.org/10.1093/jee/45.4.652
- Dicke M. 2008. Volatile spider-mite pheromone and host-plant kairomone, involved in spaced-out gregariousness in the spider mite Tetranychus urticae. Physiol. Entomol., 11: 251-262. https://doi.org/10.1111/j.1365-3032.1986.tb00412.x
- Dyer F.C. 2000. Group movement and individual cognition: lessons from social insects. In: On the move: how and why animals travel in groups. University of Chicago Press, Chicago, pp. 127-164
- Efron B., Tibshirani R.J. 1993. An introduction to the bootstrap. Chapman & Hall, New York, pp. 456 https://doi.org/10.1007/978-1-4899-4541-9
- Fazeli-Dinan M., Azarnoosh M., Özgökçe M., Chi H., et al. 2022. Global water quality changes posing threat of increasing infectious diseases, a case study on malaria vector Anopheles stephensi coping with the water pollutants using age-stage, two-sex life table method. Malar. J., 21: 178. https://doi.org/10.1186/s12936-022-04201-x
- Grassé P.P. 1946. Sociétés animales et effet de groupe. Experientia, 2: 77-116. https://doi.org/10.1007/BF02172562
- Holbrook G.L., Armstrong E., Bachmann J.A., Deasy B.M., Schal C. 2000. Role of feeding in the reproductive ′group effect′ in females of the German cockroach Blattella germanica (L.). J. Insect. Physiol., 46: 941-949. https://doi.org/10.1016/S0022-1910(99)00201-2
- Huang Y.B., Chi H. 2013. Life tables of Bactrocera cucurbitae (Coquillett) (Diptera: Tephritidae): with an invalidation of the jackknife technique. J. Appl. Entomol., 137: 321-400. https://doi.org/10.1038/npre.2012.7070.1
- Jin Y., Wang J., Huang D.-L., Shi M.-Z., Chi H., Rostami E., Kavousi A., Fu J.-W. 2024. Comparative demography of group- and individually-reared life tables of papaya mealybug with an innovative life table analysis for species in which females and males have a different number of stages. Entomol. Gen. 44: 727-735. https://doi.org/10.1127/entomologia/2024/2332
- Krause J., Ruxton G.D. 2002. Living in groups. Oxford University Press. 210 pp. https://doi.org/10.1093/oso/9780198508175.001.0001
- Le Goff G.J., Mailleux A.C., Detrain C., Deneubourg J.L., Clotuche G., Hance T. 2010. Group effect on fertility, survival and silk production in the web spinner Tetranychus urticae (Acari: Tetranychidae) during colony foundation. Behaviour, 147: 1169-1184. https://doi.org/10.1163/000579510X510980
- Li X.H., Wang D.J., Lei Z.R., Wang H.H. 2021. Comparison of life tables for experimental populations of individual rearing and group-rearing Frankliniella occidentalis. Sci. Agric. Sin., 54: 659-968.
- Lihoreau M., Rivault C. 2008. Tactile stimuli trigger group effects in cockroach aggregations. Anim. Behav., 75: 1965-1972. https://doi.org/10.1016/j.anbehav.2007.12.006
- McMurtry J.A., Huffaker C.B., Van de Vrie M. 1970. Ecology of Tetranychid mites and their natural enemies: a review I Tetranychidae enemies: their biological characters and the impact of spray practices. Hilgardia, 40: 331-390. https://doi.org/10.3733/hilg.v40n11p331
- Meyer M.K.P. 1987. African Tetranychidae (Acari: Prostigmata) with reference to the world genera. Entomol. Mem. Dep. Agric. Water. Supply. Repub. S. Afr., 69: 1-175.
- Migeon A., Dorkeld F. 2024. Spider Mites Web: a comprehensive database for the Tetranychidae. Available from https://www1.montpellier.inrae.fr/CBGP/spmweb (Accessed 30 March 2024)
- Mori K., Saito Y. 2006. Communal relationships in a social spider mite, Stigmaeopsis longus (Acari: Tetranychidae): an equal share of labor and reproduction between nest mates. Ethology, 112: 134-142. https://doi.org/10.1111/j.1439-0310.2006.01130.x
- Myllymäki A. 1975. Social mechanisms in the population ecology and population control of microtine rodents. Ecol. Bull. 19: 241-254.
- Oku K., Magalhaes S., Dicke M. 2009. The presence of webbing affects the oviposition rate of two-spotted spider mites, Tetranychus urticae (Acari: Tetranychidae). Exp. Appl. Acarol., 49: 167-172. https://doi.org/10.1007/s10493-009-9252-4
- Prokopy J.R., Duan J.J. 1998. Socially facilitated behaviour in Mediterranean fruit flies. Behav. Ecol. Sociobiol., 42: 117-122. https://doi.org/10.1007/s002650050419
- Prokopy J.R., Reynolds A.H. 1998. Ovipositional enhancement through socially faciliated behaviour in Rhagoletis pomonella flies. Entomol. Exp. Appl., 86: 281-286. https://doi.org/10.1046/j.1570-7458.1998.00290.x
- Ranta E., Rita H., Lindström H. 1993. Competition vs cooperation: success of individuals foraging alone and in groups. Am. Nat., 142: 42-58. https://doi.org/10.1086/285528
- Razmjou, J., Tavakkoli, H., Nemati, M. (2009). Life history traits of Tetranychus urticae Koch on three legumes (Acari: Tetranychidae). Mun. Ent. Zool., 4: 204-211.
- Regev G., Cone W.W. 1976. Evidence of gonadotropic effect of Farnesol in the two spotted spider mite, Tetranychus urticae. Environ. Entomol., 4: 517-519. https://doi.org/10.1093/ee/5.3.517
- Rismayani, Ullah M.S., Chi H., Gotoh T. 2021. Impact of constant and fluctuating temperatures on population characteristics of Tetranychus pacificus (Acri: Tetranychidae). J. Econ. Entomol., 114: 638-651. https://doi.org/10.1093/jee/toaa327
- Sabelis M.W. 1981. Biological control of two-spotted spider mites using phytoseiid predators. Agricultural Research Reports No. 910, Wageningen, The Netherlands, 242 pp.
- Saemi S., Rahmani H., Kavousi A., Chi H. 2017. Group-rearing did not affect the life table and predation rate of Phytoseiulus persimilis (Acari: Phytoseiidae) fed on Tetranychus urticae. Syst. Appl. Acarol., 22: 1698-1714. https://doi.org/10.11158/saa.22.10.11
- Salomon M., Lubin Y. 2007. Cooperative breeding increases reproductive success in the social spider Stegodyphus dumicola (Araneae: Eresidae). Behav. Ecol. Sociobiol., 61: 1743-1750. https://doi.org/10.1007/s00265-007-0406-2
- Sato Y., Saito Y. 2006. Nest sanitation in social spider mites: interspecific differences in defecation behaviour. Ethology, 112: 664-669. https://doi.org/10.1111/j.1439-0310.2005.01184.x
- Schausberger P., Yano S., Sato Y. 2021. Cooperative behaviors in group-living spider mites. Front. Ecol. Evol., 9: 745036. https://doi.org/10.3389/fevo.2021.745036
- Shih C., Poe S.L., Cromroy H.L. 1976. Biology, life table, and intrinsic rate of increase of Tetranychus urticae. Ann, Entomol. Soc. Am., 69: 362-364. https://doi.org/10.1093/aesa/69.2.362
- Strodl M.A., Schausberger P. 2012. Social familiarity reduces reaction times and enhances survival of group-living predatory mites under the risk of predation. PLoS One, 7: e43590. https://doi.org/10.1371/journal.pone.0043590
- Sugawara R., Ullah M.S., Ho C.-C., Gökçe A., Chi H., Gotoh T. 2017. Temperature-dependent demography of two closely related predatory mites Neoseiulus womersleyi and N. longispinosus (Acari: Phytoseiidae). J. Econ. Entomol., 110: 1533-1546. https://doi.org/10.1093/jee/tox177
- Thind B., Edwards J. 1990. Stimulation of egg production in the grain mite Acarus siro by the juvenile-hormone analogue fenoxycarb. Exp. Appl. Acarol., 9: 1-10. https://doi.org/10.1007/BF01198978
- Tuan S.J., Lee C.C., Chi H. 2014a. Population and damage projection of Spodoptera litura (F.) on peanuts (Arachis hypogaea L.) under different conditions using the age-stage, two-sex life table. Pest Manag. Sci., 70: 805-813. https://doi.org/10.1002/ps.3618
- Tuan S.J., Lee C.C., Chi H. 2014b. Erratum: Population and damage projection of Spodoptera litura (F.) on peanuts (Arachis hypogaea L.) under different conditions using the age-stage, two-sex life table. Pest Manag. Sci., 70: 1936. https://doi.org/10.1002/ps.3920
- Ullah M.S., Kamimura T., Gotoh T. 2020. Effects of temperature on demographic parameters of Bryobia praetiosa Koch (Acari: Tetranychidae). J. Econ. Entomol., 113: 211-221. https://doi.org/10.1093/jee/toz294
- Van Impe G. 1984. Influence of population density on the duration of immature development in Tetranychus urticae Koch (Acari: Tetranychidae). Acarology, 6: 617-621.
- Van Impe G. 1985. Contribution à la conception de stratégies de contrôle de l'acarien tisserand commun, Tetranychus urticae Koch (Acari: Tetranychidae). PhD thesis, Université catholique de Louvain, Louvain, pp. 283.
- Wang Y., Iqbal A., Mu M.-Y., Zang Z.-Y., Hou Y.-Y., Zang L.-S. 2022. Effect of carbohydrate nutrition on egg load and population parameters of four Trichogramma Species. Agronomy, 12: 3143. https://doi.org/10.3390/agronomy12123143
- Wertheim B., van Baalen E.J.A., Dicke M., Vet L.E.M. 2005. Pheromone-mediated aggregation in nonsocial arthropods: an evolutionary ecological perspective. Annu. Rev. Entomol., 50: 321-346. https://doi.org/10.1146/annurev.ento.49.061802.123329
- Yano S. 2008. Collective and solitary behaviors of two-spotted spider mite (Acari: Tetranychidae) are induced by trail following. Ann. Entomol. Soc. Am., 101: 247-252. https://doi.org/10.1603/0013-8746(2008)101[247:CASBOT]2.0.CO;2



2025-11-04
Date accepted:
2026-02-08
Date published:
2026-02-13
Edited by:
Marčić, Dejan

This work is licensed under a Creative Commons Attribution 4.0 International License
2026 Hayder, Tasfia; Meem, Mst. Masuma Momtaj; Tonni, Farhana Akter; Nikson, A. K.; Gotoh, Tetsuo and Ullah, Mohammad Shaef
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



