Taxonomic contributions to the genus Protogamasellopsis Evans & Purvis (Acari: Mesostigmata: Rhodacaridae), with supplementary descriptions of P. denticus (Nasr) and P. corticalis Evans & Purvis
Mohammadi, Leila  1
; Hajizadeh, Jalil
1
; Hajizadeh, Jalil  2
and Kazemi, Shahrooz
2
and Kazemi, Shahrooz  3
3
1Department of Plant Protection, Faculty of Agricultural Sciences, University of Guilan, Rasht, Iran.
2✉ Department of Plant Protection, Faculty of Agricultural Sciences, University of Guilan, Rasht, Iran.
3✉ Department of Biodiversity, Graduate University of Advanced Technology, Kerman 7631885356, Iran & Department of Evolution, Ecology and Organismal Biology, Ohio State University, Columbus OH 43212, USA.
2026 - Volume: 66 Issue: 1 pages: 92-112
https://doi.org/10.24349/8xf3-20fmZooBank LSID: 84A9B901-D09F-48B9-B72A-81790A178BE9
Original research
Keywords
Abstract
Introduction
The family Rhodacaridae Oudemans comprises free-living, cosmopolitan, soil-dwelling mites that occur predominantly in the upper soil layer. Although most records are from soil, they are also found in mosses, lichens, leaf litter, and other organic substrates (Lindquist et al. 2009; Castilho et al. 2012). The family currently includes more than 160 described species in 15 genera (Braga & Castilho 2024; Anonymous 2025).
The genus Protogamasellopsis Evans & Purvis, 1987 represents a small assemblage of rhodacarid mites. It was originally assigned to the family Ascidae but was later transferred to Rhodacaridae by Karg (1994a). The genus presently comprises 10 described species: P. dioscorus Manson, 1972; P. denticus (Nasr, in Zaher 1986) (originally described as Protogamasellus denticus); P. corticalis Evans & Purvis, 1987; P. posnaniensis Wiśniewski & Hirschmann, 1991; P. granulosus Karg, 1994b; P. leptosomae Karg, 1994b; P. praeendopodalis Karg, 1994a; P. transversus Karg, 2000; P. zaheri Abo-Shnaf et al., 2013; and P. rhizoglyphusi Amin & Khanjani, 2022. Additionally, Rhodacarella cavernicola Moraza, 1994 was synonymized with P. posnaniensis by Castilho et al. (2012). Most species appear to have localized distributions, seemingly restricted to the regions from which they were originally collected and described. Among these, P. corticalis and P. posnaniensis have been recorded from Iran (Kazemi & Rajaei 2013) and P. rhizoglyphusi was originally described from this country (Amin & Khanjani 2022).
Recent studies have investigated the biological potential of several Protogamasellopsis species including P. posnaniensis, P. zaheri, P. denticus, and P. rhizoglyphusi as potential biological control agents (Castilho et al. 2009, 2015; Momen et al. 2020; Rueda-Ramirez et al. 2022; Rossini et al. 2022; Prado et al. 2024; Amin & Khanjani 2024). Although these investigations indicate promising predatory roles, evaluations have so far been limited to laboratory and greenhouse conditions, and no field assessments have been undertaken.
The present study (1) provides supplementary descriptions of P. corticalis and P. denticus based on material collected from northern Iran and the United States, together with comparative information derived from type material and original descriptions of P. posnaniensis and P. denticus; (2) re-evaluates and supports the synonymy of P. posnaniensis and Rhodacarella cavernicola with P. denticus, and discusses the status of P. rhizoglyphusi as a putative synonym based on available evidence; and (3) clarifies several inconsistently interpreted morphological characters within Protogamasellopsis and resolves discrepancies in previous diagnoses and identification keys.
Material and methods
Mite specimens collected in northern Iran were extracted from soil, manure, stored rice, and rice husks samples using Berlese-Tullgren funnels, then cleared in Nesbitt's fluid and mounted in Hoyer's medium on microscope slides. Additional specimens from the United States were examined from the Acarology Collection, Ohio State University (OSU).
Comparative information on type and non-type specimens of P. posnaniensis and P. denticus was obtained from colleagues in Poland and Egypt who examined these materials. Attempts to obtain additional details from the authors of P. rhizoglyphusi were unsuccessful; therefore, the present study relies solely on the published description of that species.
Morphological observations and measurements were made using an Olympus BX51 microscope (Olympus Optical Co., Ltd., Tokyo, Japan) equipped with differential interference contrast and phase-contrast optics, as well as a drawing tube. Final illustrations were prepared using Adobe Illustrator. All measurements are given in micrometers. The lengths and widths of the shields were taken from the anterior to posterior margins along the midline and at the broadest levels, respectively. The fixed cheliceral digit was measured from the anterior margin of the dorsal poroid to the digit apex, and the movable digit from the base to the apex. Leg length was measured from the base of the coxa to the apex of the tarsus, excluding the ambulacrum (stalk, claws, and pulvillus). For each character, the mean is given, followed by the range in parentheses.
The notation for idiosomal setae follows that of Lindquist & Evans (1965), as adapted by Lindquist (1994); notation for leg and palp setae follows Evans (1963a, 1963b); and that for idiosomal pore-like structures (gland pores and poroids), follows primarily Athias-Henriot (1975) and Johnston & Moraza (1991).
Results
Family Rhodacaridae Oudemans
Genus Protogamasellopsis Evans & Purvis
Protogamasellopsis Evans & Purvis, 1987: 855.
Protogamasellopsis Karg, 1994b: 207; Halliday et al., 1998: 2; Castilho et al., 2012: 17.
Type species: Protogamasellopsis corticalis Evans & Purvis, 1987, by original designation.
Rhodacarella Moraza, 2004: 2 (synonymy by Castilho et al. 2012).
Type species: Rhodacarella cavernicola Moraza, 2004, by monotypy.
Detailed diagnosis
Dorsal shields smooth or lightly punctate. All idiosomal setae acicular. Podonotal shield with narrow desclerotized and usually granular band behind setae j6 and z6; scleronoduli and transverse line behind setae j4 absent; with 16 pairs (P. transversus with 19 pairs) of moderately short setae, setae j2 almost in transverse line with j1 and z1. Opisthonotal shield moderately narrow, considerably longer than wide, usually without anterior granular band (except in P. praeendopodalis), and a pair of eye-shaped carinae on anterior margin (present in P. corticalis); bearing 15 pairs of setae, Z5 longer than others. Dorsal idiosomal soft cuticle with a pair of subtriangular plates extending from anterior level of podonotal shield to level of j4; with 5–6 pairs of setae lateral to podonotal shield and usually 9–10 pairs lateral to opisthonotal shield (5 pairs in P. granulosus). Ventral shields usually lightly punctate. One pair of transverse plates located laterad tritosternum, between anterior level of coxae I. Tritosternal base columnar, elongated; laciniae pilose, basally fused. Presternal region lightly sclerotized and granulated, bearing setae st1, with or without 3–4 pairs of narrow transverse platelets, each comprising 1–3 elements. Sternal shield with anterior margin somewhat truncate, posterior margin concave; bearing setae st2–4 and poroids iv1–3, iv1 and iv3 situated on anterior and posterior margins, and usually with a pair of small, rounded, densely punctate areas medially at level of st2 or slightly posterior. Epigynal shield subrectangular, posterior aera granular; anterior hyalin area convex, usually partially covering rounded sclerotized endogynal plate, or tongue-shaped and extending to posterior level of st2. Setae st5 on lateral margins of epigynal shield. Usually with four small transverse postgenital platelets, median pair sometimes fused. Ventrianal shield markedly longer than wide; bearing 1–2 pairs of preanal setae, post-anal seta longest. Peritrematal shield free, not developed anterior to peritreme and narrowly developed alongside peritreme; poststigmatal section narrowly extended to posterior level of coxa IV. Peritreme short, anteriorly reaching posterior to mid-level of coxae II. Opisthogastric integument with 6–8 pairs of setae, two pairs of subrectangular or suboval platelets flanking anterior area of ventrianal shield, and a pair of narrow longitudinal metapodal platelets (original description of P. leptosomae indicates absence of platelets and nine pairs of setae on integument). Male sternogenital shield with somewhat truncate posterior margin; bearing four pairs of setae st2–5. One suboval platelet and one pair of setae present on soft integument between sternigenital and ventrianal shields. Male ventrianal shield free, very large, reaching lateral and posterior margins of opisthogaster; bearing 5–6 pairs of ventral setae in addition to circumanals. Gnathotectum subtriangular, with a median simple projection (laterally serrate in P. granulosus); anterior margin of lateral shoulders serrates. Fixed digit of chelicera in female with 6–8 teeth in addition to apical hook, 5–6 teeth in male; movable digit bidentate in female, unidentate in male; spermadactyl short, straight, with recurved tip. Corniculi subparallel, elongated, extending beyond anterior level of palp trochanter; internal malae laterally pilose in basal half, longer than cuniculi. Deutosternal groove with anterior ridge followed by seven denticulate rows, anterior one Λ-shaped. Palp tarsus apotele tri-tined. Legs shorter than idiosoma length; leg IV in both sexes thicker than other legs; leg I with well-developed paired claws and rounded pulvilli; coxa I bearing a dorsomedian spine. Setation of legs I–IV: coxae 2, 2, 2, 1; trochanters 6, 5, 5, 5; femora 12 (2 2/1 3/2 2), 11 (2 3/1 2/2 1), 6 (1 2/1 1/0 1), 6 (1 2/1 1/0 1); genua 13 (2 3/2 3/1 2), 11 (2 3/1 2/1 2), 8 (2 2/1 2/0 1), 9 (2 2/1 3/0 1); tibiae 13 (2 3/2 3/1 2), 10 (2 2/1 2/1 2), 8 (2 1/1 2/1 1), 9 (2 1/1 3/1 1); tarsi II–IV each with 18 setae.
Remarks on the generic characters
Evans and Purvis (1987) established the genus Protogamasellopsis based on two closely related species, P. corticalis and P. dioscorus. Several additional species have subsequently been described in this genus. Although Castilho et al. (2012) provided a definition of the genus, their account did not encompass several key diagnostic features characteristic of these mites. In the present study, we provide a revised and detailed diagnosis of Protogamasellopsis, based on the original descriptions of the included species and on examination of specimens collected from northern Iran (by the senior authors, LM and JH) and the United States (by the third author, SK). Additional comparative data on the type and non-type specimens of P. posnaniensis and P. denticus were obtained through personal communications with specialists who examined the relevant materials (LM pers. com. with Dr. D. Gwiazdowicz; SK pers. com. with Dr. A. Nasr), and these data were incorporated into the present evaluation of the genus.
Protogamasellopsis corticalis Evans & Purvis, 1987
Diagnosis (adults)
Dorsal setae mostly short, not reaching the base of the subsequent seta in the same series; setae Z5 4.3–4.7 times longer than J5; setae J3 and Z3–5 slightly barbed. Presternal region heavily granulated, without transverse groups of platelets. Unsclerotized cuticle between genital and ventrianal shields in female bearing three transversely oval platelets, and between sternogenital and ventrianal shields in male with a transverse spindle-shaped platelet. A small triangular endopodal platelet present between coxae III–IV. Peritreme extending anteriorly to slightly beyond mid-level of coxa II, and its length shorter than width of posterior margin of podonotal shield. Anterior margin of ventrianal shield in male straight. In male, trochanter IV without spur-like projection; seta pv2 on tarsus IV modified to a moderately short and thick spur.
Supplementary description
Adult female (Figures 1–4) — Five specimens measured.
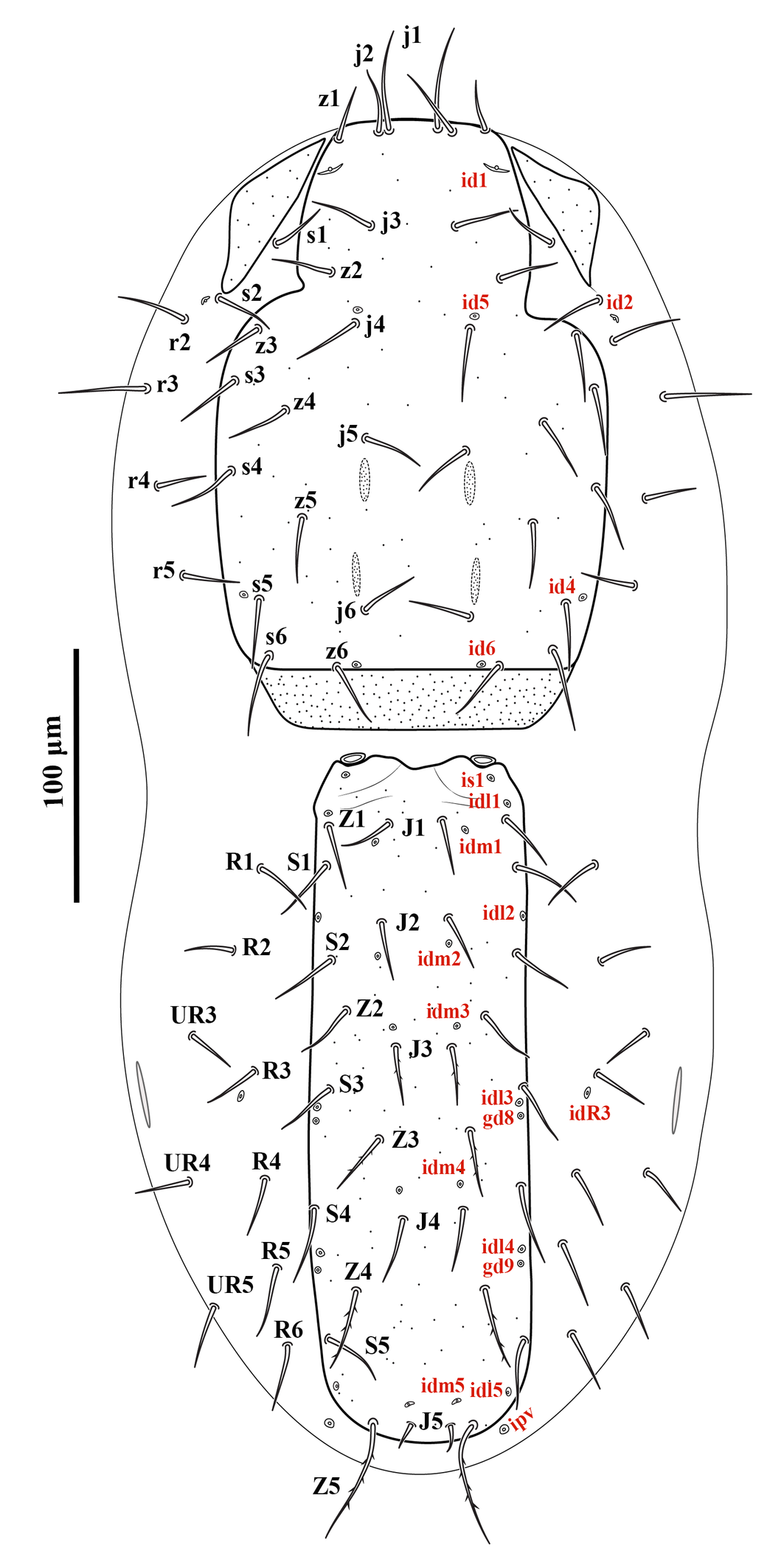


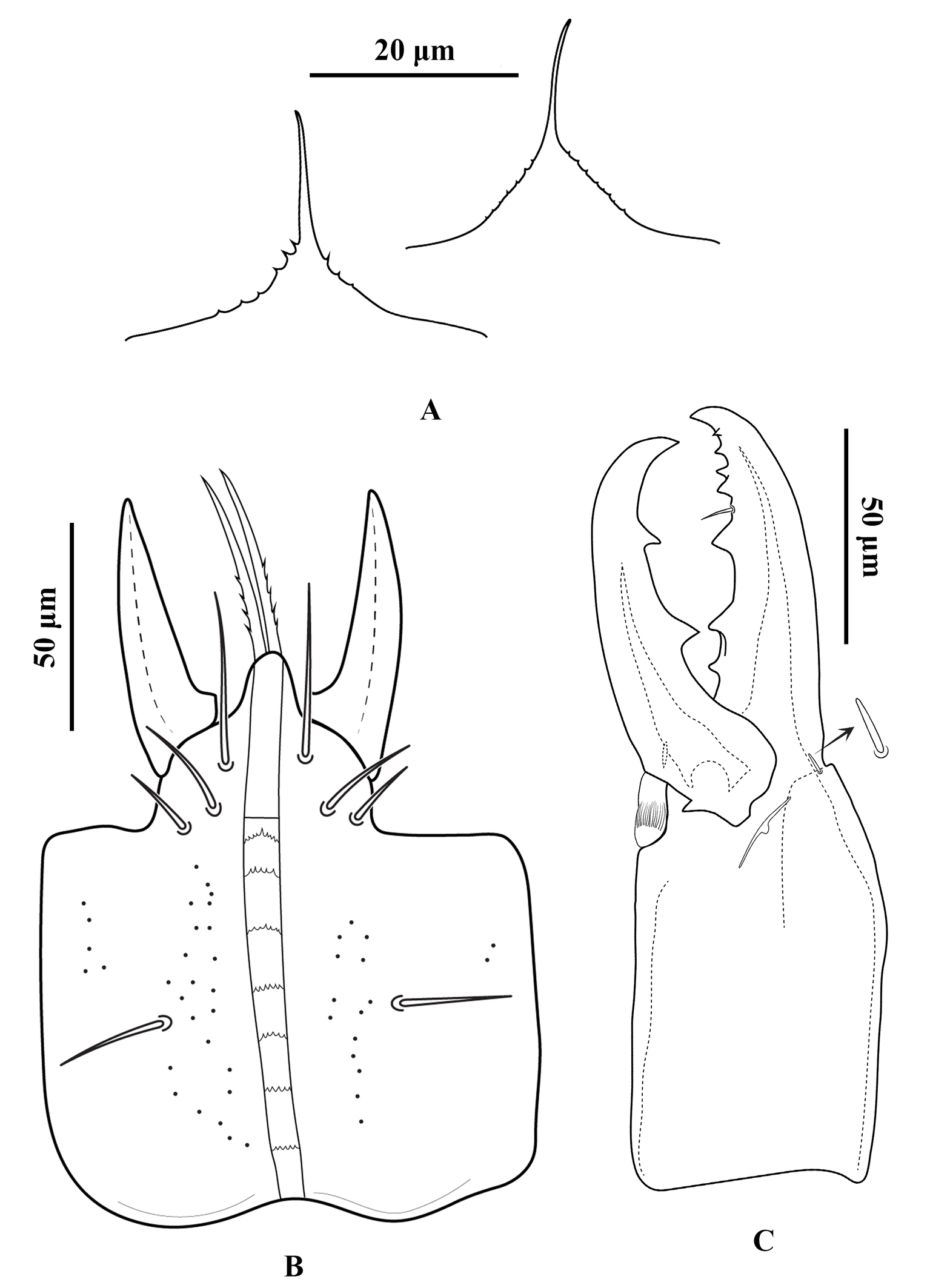
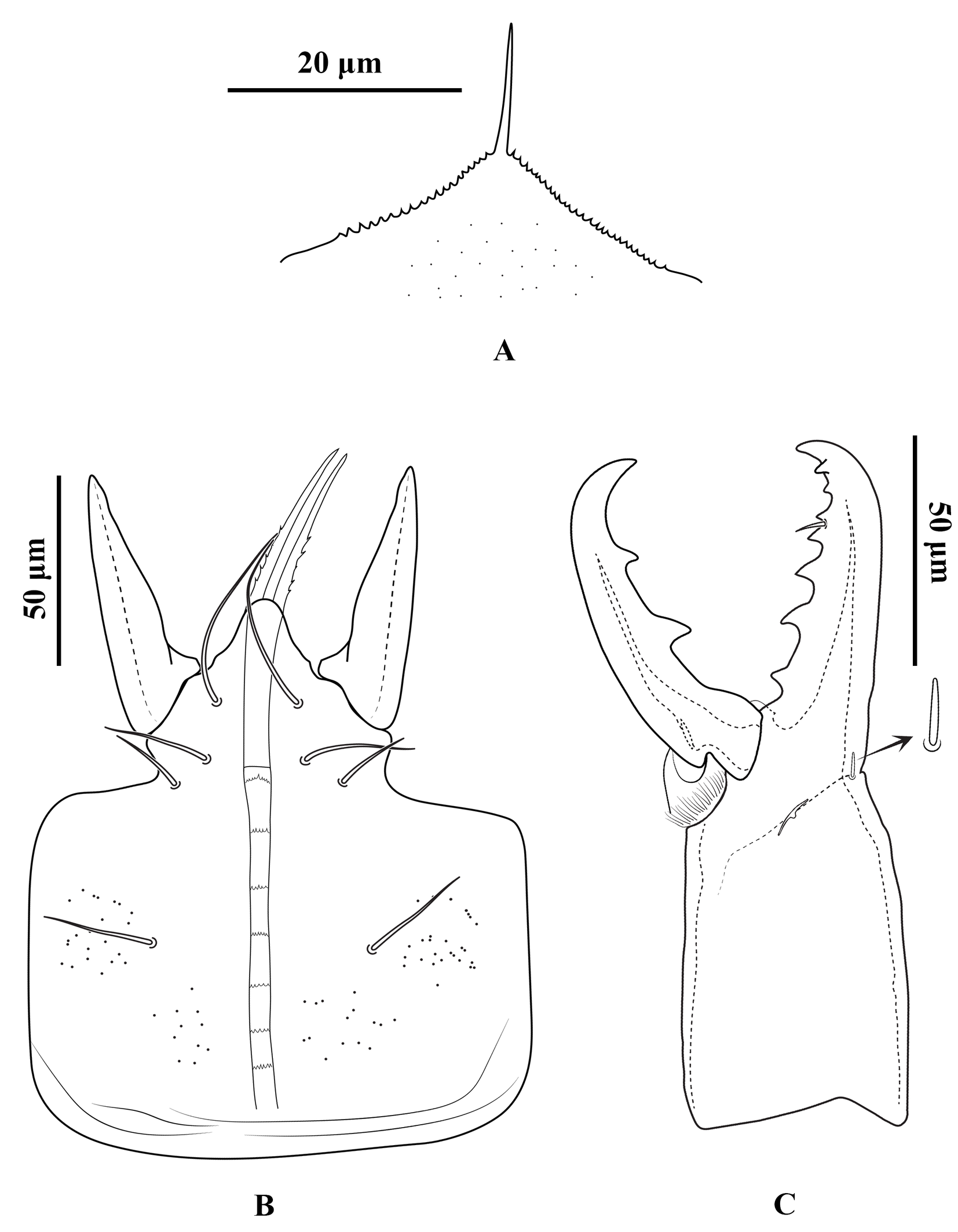
Gnathosoma — Gnathotectum subtriangular, with anterior margin finely denticulate, and a narrow, smooth and pointed median projection (Figure 3A). Internal mala with two elongate projections, the outer margins densely barbed in the basal half and smooth distally. Corniculi horn-like, slightly divergent, 51 (50–54) long, 17 (14–18) wide at base. Deutosternum with a smooth anterior ridge followed by seven transverse denticulate rows, delimited by lateral lines. Hypostomal setae acicular and smooth, h1 38 (36–40), h2 15 (13–16), h3 25 (23–26), sc 23 (20–26) (Figure 3B). Fixed cheliceral digit 72 (70–76) long, with seven teeth in addition to apical hook, and a setiform pilus dentilis (four small teeth, followed by pilus dentilis, two larger teeth, and a small basal tooth); movable digit 73 (70–76) long, with two teeth; dorsal and antiaxial lyrifissures distinct; dorsal cheliceral seta small, apically blunt (Figure 3C). Palp setae aciculate and smooth, except for chisel-shaped anterolateral setae on palp genu and palp femur; number of setae on palp trochanter–tarsus, 2-5-6-14-15; apotele 3-tined.
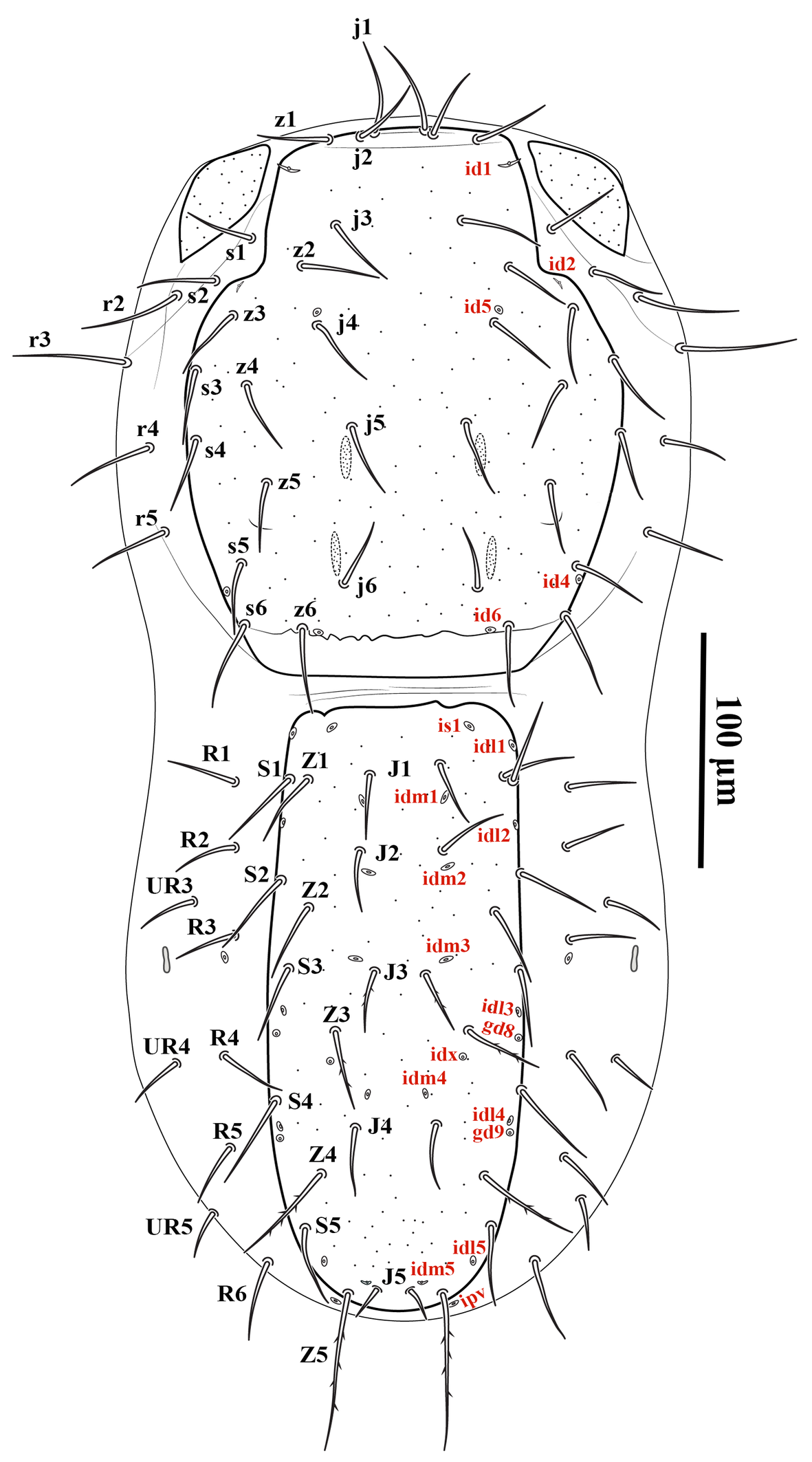
Dorsal idiosoma — (Figure 1). Idiosoma 518 (504–528) long and 223 (200–244) wide at the level of r3. Podonotal shield 227 (224–232) long and 159 (140–176) wide at the level of s3; shield surface mostly smooth, with faint punctuation over most of the surface, and a punctate band along the posterior margin; with 16 pairs of setae (j1–j6, z1–z6, s3–s6) and four pair of distinguishable poroids (id1, id4, id5 and id6); scleronoduli absent. Unsclerotized cuticle laterad podonotal shield with a pair of subtriangular plates, extending from the level of z1 to s2, six pairs of setae (s1, s2 and r2–5) and one pair of poroids (id2). Opisthonotal shield 246 (228–256) long and 89 (80–96) wide at the level of S3; shield surface mostly smooth, with faint punctuation over the entire surface, and two eye-shaped carinae on the anterior margin; bearing 15 pairs of setae (J1–5, Z1–5 and S1–5), 11 pairs of poroids (idm1, idm2, idm3, idm4, idm5, is1, idl1, idl2, idl3, idl4 and idl5) and two pairs of gland pores (gd8 and gd9). Unsclerotized cuticle laterad opisthonotal shield with nine pairs of setae (R1–6 and UR3–5), one pair of poroids (idR3) posteriad R3, and two pairs of poroids, idR3 posteriad seta R3 and ipv posterior to opisthonotal shield, one pair of narrow platelets flanking opisthonotal shield at level of setae R3. Measurements of setae: j1 39 (36–40), j2 26 (24–28), j3 25 (24–26), j4 25 (20–28), j5 25 (24–26), j6 20 (19–21), z1 20 (20), z2 23 (22–24), z3 26 (22–28), z4 23 (22–24), z5 23 (22–24), z6 26 (26), s1 21 (20–23), s2 23 (20–26), s3 26 (24–30), s4 25 (23–26), s5 25 (24–26), s6 31 (30–32), r2 28 (26–30), r3 35 (32–38), r4 20 (18–22), r5 20 (18–21), J1 21 (20–22), J2 21 (20–22), J3 21 (20–22), J4 23 (22–24), J5 10 (8–12), Z1 22 (20–24), Z2 23 (22–24), Z3 24 (22–26), Z4 30 (30), Z5 47 (40–50), S1 26 (24–28), S2 25 (24–26), S3 24 (22–26), S4 29 (27–30), S5 25 (23–26), R1 21 (20–24), R2 20 (18–21), R3 21 (20–23), R4 20, R5 22 (20–24), R6 22 (18–24), UR3 19 (16–20), UR4 20, UR5 21 (20–24); all dorsal setae aciculate and smooth, except J3 and Z3–5, aciculate and slightly barbed.
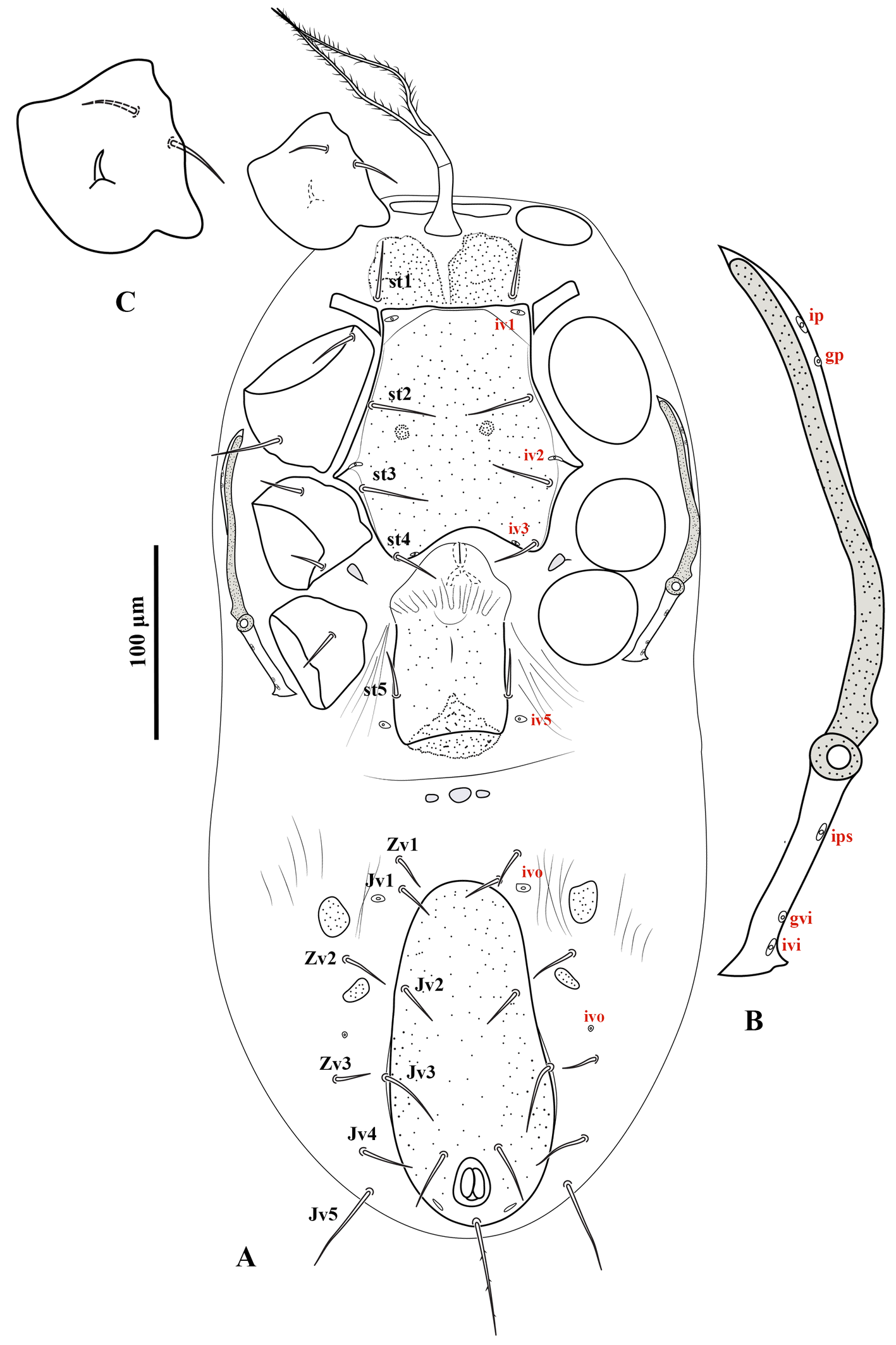


Ventral idiosoma — (Figure 2A). Base of tritosternum columnar, 60 (56–64) long and 13–15 wide proximally; laciniae pilose, 80–100 long, separated for about 80–85% of their total length. With one pair of elongate transverse plates anteriad base of tritosternum. Presternal region heavily granulated, bearing setae st1. Sternal shield 100–104 long and 122–126 wide at level between coxae II–III, lightly punctate, with one pair of median small circular areas with dense punctation; anterior margin somewhat straight, posterior margin concave; with three pairs of setae (st2–4) and three pairs of poroids (iv1–3). Endopodal platelets between coxae I–II adjacent, but free from the sternal shield; with a pair of small, free subtriangular endopodal platelets between coxae III–IV. Endogynal structure heart-shaped, covered by hyaline area of epigynal shield. Epigynal shield approximately rectangular, extending posteriorly well beyond the level of coxa IV; posterior margin slightly concave with a punctate conical area slightly extending beyond the shield; distance between st5-st5 58 (50–60). Poroids iv5 on unsclerotized cuticle, posterolaterad st5. Postgenital area with three transversely suboval platelets, middle one larger than the other two. Ventrianal shield pear-shaped, slightly punctate; 166–176 long and 80–82 wide at broadest level; with one pair of setae (Jv2) in addition to circumanals, and with a pair of distinguishable pore-like structures posterolaterad paranal setae; Jv3 on lateral margins of shield. Unsclerotized cuticle around ventrianal shield with six pairs of setae (Jv1, Jv4, Jv5, Zv1–Zv3), two pairs of platelets (anterior pair larger), and two pairs of distinguishable poroids (posterolaterad Jv1 and anterior Zv3). Peritreme short, extending anteriorly to slightly beyond mid-level of coxa II (posterior level of r3). Peritrematic shield narrow, confined to region along peritreme, with one gland pore and one poroid on lateral margin; poststigmatic region of shield as wide as peritreme, extending to posterior level of coxa IV, bearing two poroids and one gland pore (Figure 2B). Measurements of setae: st1 28 (26–30), st2 31 (30–32), st3 30 (28–32), st4 25 (24–28), st5 20 (18–22), Jv1 21 (20–24), Jv2 22 (20–26), Jv3 28 (26–30), Jv4 23 (20–24), Jv5 41 (40– 45), Zv1 18 (16–20), Zv2 21 (20–23), Zv3 15 (14–16), paranal 28 (25–30), postanal 51 (46–55); all setae aciculate and smooth, except postanal, slightly barbed.


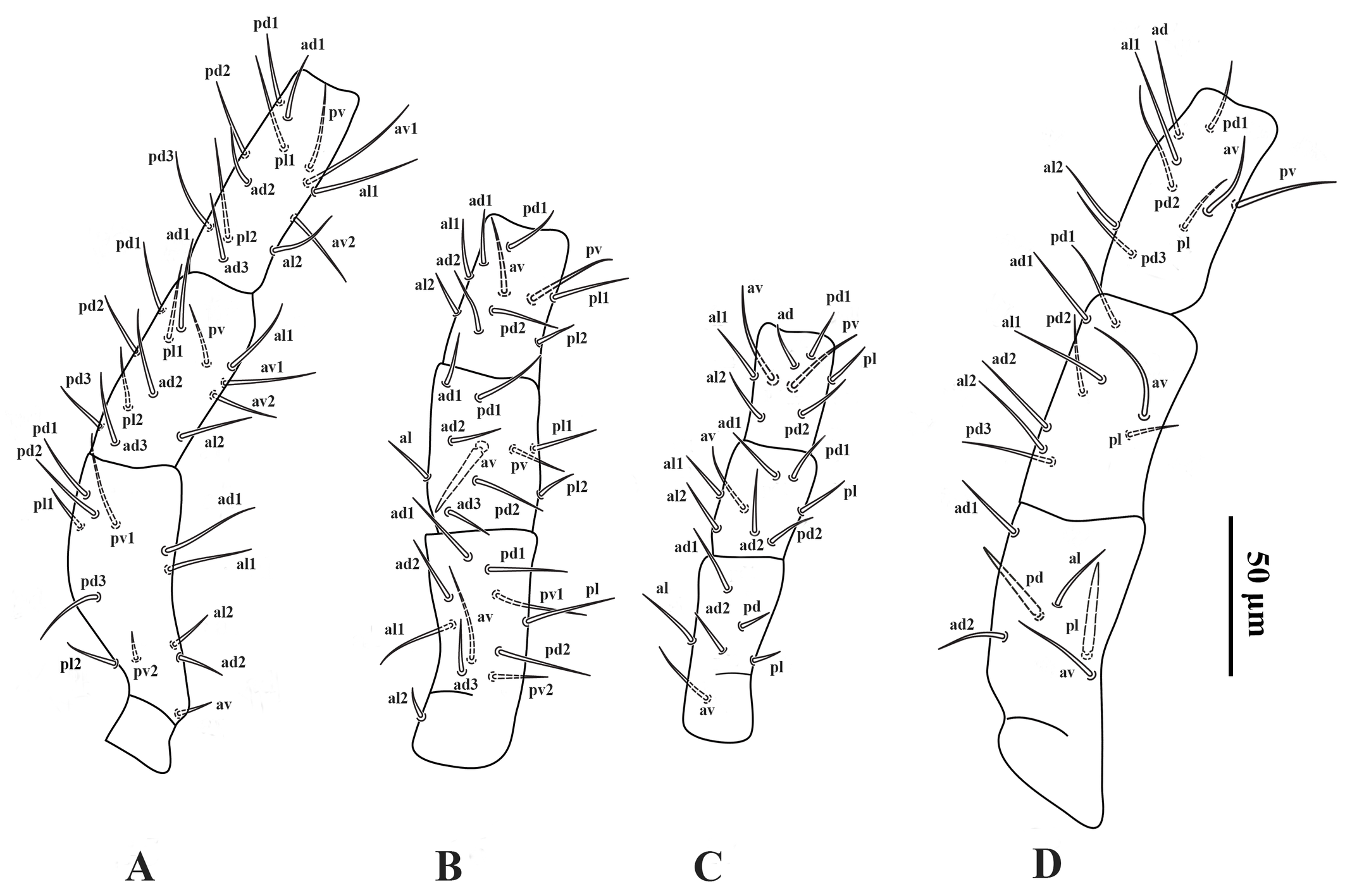
Legs — (Figures 4A–D). Lengths of legs I–IV 374 (360–380), 268 (260–272), 242 (232–252) and 366 (350–380), respectively. Leg IV thicker than legs I–III. Setation of legs I–IV as described for the genus; coxa I with a small dorsal median spine (Figure 2C); setae pd and pl on femur IV spine-like; seta av on genu II spine-like; setae av and pv on tibiae II–IV slightly thickened.


Adult male (Figure 5) — Four specimens measured.
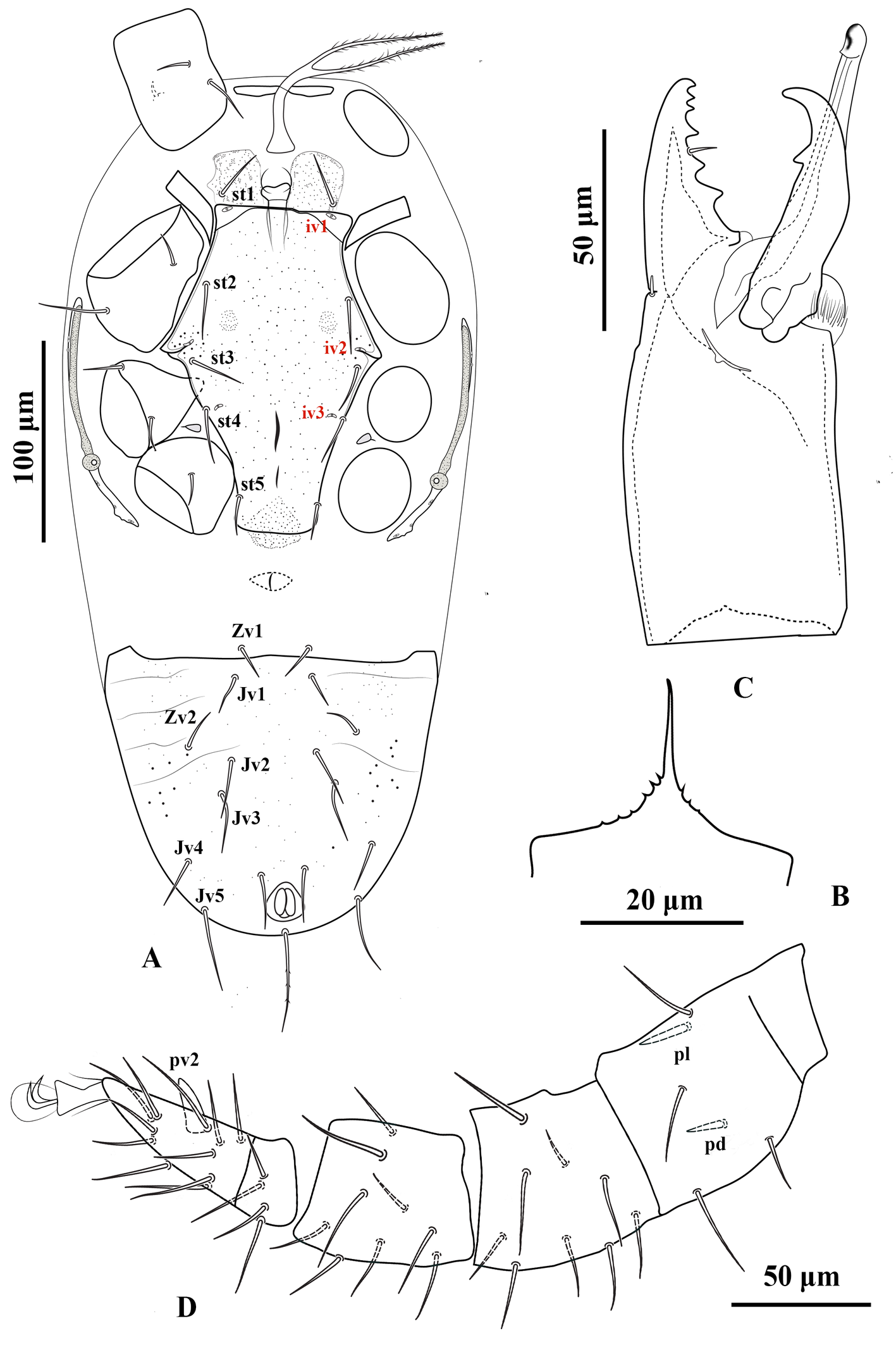


Gnathosoma — Fixed cheliceral digit 47 (44–50) long, with five teeth in addition to apical hook, and a setiform pilus dentilis; movable digit 47 (42–50) long, with one tooth; dorsal lyrifissure distinct; dorsal cheliceral seta blunt (Figure 5B); spermatodactyl 62 (60–64) long, with recurved hook-like tip. Gnathotectum triangular, with anterior margin finely denticulate, and a median smooth spine-like extension (Figure 5C). Corniculi 43 (40–46) long, about three times as long as its basal width 14. Measurements of setae: h1 28 (20–32), h2 12 (12), h3 20 (20), sc 20 (20); all setae aciculate and smooth. All other characters similar to female.
Dorsal idiosoma — Idiosoma 404 (392–424) long and 199 (172–216) wide at the level of r3. Podonotal and opisthonotal shields separate. Podonotal shield 193 (188–200) long and 141 (132–160) wide at the level of s3; opisthonotal shield 203 (196–208) long and 77 (72–80) wide at the level of S3. Chaetotaxy and sclerotization of the dorsum as in adult female, except for the absence of UR setae. Measurements of setae: j1 34 (32–36), j2 23 (20–24), j3 23 (22–24), j4 21 (20–23), j5 21 (21), j6 19 (18–20), z1 16 (12–18), z2 22 (20–24), z3 21 (18–24), z4 20 (20), z5 21 (20–22), z6 24 (23–26), s1 20 (20), s2 18 (16–20), s3 21 (18–24), s4 20 (17–22), s5 23 (22–24), s6 27 (26–30), r2 25 (22–26), r3 33 (30–36), r4 17 (16–18), r5 19 (18–20), J1 19 (18–20), J2 20 (20), J3 19 (18–20), J4 20 (20), J5 11 (10–12), Z1 20 (20), Z2 20 (20), Z3 21 (20–22), Z4 27 (24–28), Z5 44 (38–46), S1 25 (24–26), S2 22 (22), S3 21 (20–22), S4 24 (22–26), S5 23 (22–24), R1 16 (16), R2 17 (16–18), R3 17 (16–18), R4 18 (18), R5 18 (18), R6 10 (10). Shape of dorsal setae as in adult female.
Ventral idiosoma (Figure 5A). Base of tritosternum 47 (42–50) long and 13 wide proximally; laciniae 61 (50–74) long, separate for about 80% of their total length. Genital opening in the middle of this area. Sternogenital shield approximately 159 (154–166) long, and 113 (110–118) wide at level between coxae II–III, slightly punctate; with almost straight anterior and posterior margins; with four pairs of setae (st2–5) and three pairs of poroids (iv1–3). Unsclerotized cuticle between sternogenital and ventrianal shields with a transverse spindle-shaped platelet with a central longitudinal marking. Ventrianal shield lightly punctate; 137 (130–144) long and 152 (150–154) wide at the level of Zv2; with six pairs of setae (Jv1–5 and Zv2) in addition to circumanal setae, and without distinguishable pore-like structures. Seta Zv1 on unsclerotized cuticle. All other characters similar to female. Measurements of setae: st1 26 (24–28), st2 31 (30–32), st3 31 (30–32), st4 27 (26–28), st5 18 (18), Jv1 15 (12–16), Jv2 25 (24–26), Jv3 33 (30–36), Jv4 20 (16–22), Jv5 37 (34–40), Zv1 23 (21–26), Zv2 21 (20–22), para-anal 25 (24–26), post-anal (46–50); all setae aciculate and smooth, except post-anal, slightly serrate.
Legs — Lengths of legs I–IV 327 (320–332), 232 (224–248), 190 (180–200) and 320 (312–328), respectively. Numbers and shape of setae on legs I–IV as in female, except for a spur-like ventral process (pv2) on tarsus IV, spine-like setae pl and pd on the femur IV slightly thicker and shorter than those in the female (Figure 5D).
Material examined
Five females, and four males from chicken manure at Babol County (36°31′44.05″N, 52°39′39.08″E), Mazandaran Province, Iran, in 29 October 2021. Three females and four males are deposited in the Acarology Laboratory, Department of Plant Protection, Faculty of Agricultural Sciences, University of Guilan, Rasht, Iran; two females are deposited in Jalal Afshar Zoological Museum, Tehran, Iran.
Remarks
The original description of P. corticalis by Evans & Purvis (1987), based on three males and 10 females, is generally clear and well-illustrated. However, several measurements, particularly the lengths of the circumanal setae, were not provided. Although the authors stated that the circumanal setae of the female are nearly subequal, their illustration shows the post-anal seta only slightly longer than the paranal setae, with a post-anal/paranal ratio of approximately 1.25. This discrepancy suggests that the difference observed in the type material was considered minor by the original authors and may not have been interpreted as taxonomically significant at that time. In contrast, the male of P. corticalis in the original description was illustrated with a distinctly longer post-anal seta, a condition identical to that observed in our material.
The material examined in the present study exhibits a similar qualitative pattern in both sexes, with an elongate post-anal seta. However, the magnitude of this difference is more pronounced in our females, in which the post-anal seta consistently exceeds the paranal setae (post-anal 46–55; paranal 25–30), yielding higher post-anal/paranal ratios (1.5–2.2). This indicates the presence of appreciable variation in this character within the available material. Despite the high quality of the original description, the absence of measurements for many structures, combined with the clearer difference in post-anal/paranal proportions in our material, underscores the need for re-examination of the female type material of P. corticalis to determine the true range of variation.
Other minor differences observed in our specimens, such as the slightly barbed condition of setae J3 and Z3–5, are likely attributable to observational or terminological differences. The term ''simple» used by Evans & Purvis in the original description probably referred to a general hair-like or unmodified form rather than strictly smooth setae, and these features are therefore not interpreted as taxonomically meaningful discrepancies.
Given that P. corticalis has so far, based on the available information, been reported only from Iran after the original collection, obtaining additional material, ideally from localities closer to the type region, will be important for confirming whether the variation observed here falls within a single species or whether more than one closely related taxon may be involved. Until such material becomes available, we regard the specimens examined here as representing P. corticalis in the morphological sense of Evans & Purvis.
From a biological perspective, P. corticalis has been reported from geographically distant localities. While such a distribution does not by itself demonstrate intraspecific variation, it provides a broader biogeographical context within which the observed morphological and molecular differences should be interpreted (Shaw 1999; Young et al. 2019).
This species closely resembles P. dioscorus. However, females of the two species can be distinguished by the narrower ventrianal shield in P. dioscorus, the peritreme being longer than the width of the posterior margin of the podonotal shield in P. dioscorus, and the presence of a pair of eye-shaped carinae on the anterior margin of the opisthonotal shield in P. corticalis. These differences are more pronounced in males: the male of P. dioscorus possesses a ventrianal shield with an anterior margin extending anterolaterally on both sides and a large, spur-like projection on the ventral surface of trochanter IV, whereas the male of P. corticalis has a straight anterior margin of the ventrianal shield and lacks such spur on trochanter IV. The species P. granulosus and P. transversus, subsequently described by Karg (1994b, 2000), also resemble P. corticalis in lacking presternal platelets. Nevertheless, P. granulosus apparently differs from other members of this genus by having moderately larger punctate ornamentation on both sternal and ventrianal shields and bearing only five pairs of setae on the unsclerotized integument of the opisthonotal region, and P. transversus is distinct in having longer dorsal and opisthogastric setae than in the remaining congeners.
Protogamasellopsis denticus (Nasr, 1986)
Protogamasellus n. sp.1.—Nasr, 1978: 83 (Ph.D. dissertation, unpublished).
Protogamasellus denticus Nasr, in Zaher, 1986: 54 (original description).
Protogamasellopsis denticus (Nasr, 1986), new combination, Moraes et al., 2016: 250
Protogamasellopsis posnaniensis Wiśniewski & Hirschmann, 1991 (new synonymy)
Rhodacarella cavernicola Moraza, 2004 (new synonymy)
Protogamasellopsis rhizoglyphusi Amin & Khanjani, 2022 (putative synonymy)
Diagnosis (adult female)
Dorsal setae mostly short, not reaching the base of the subsequent seta in the same series; setae Z5 almost 4.3–4.7 times longer than J5; setae J3 and Z3–5 slightly barbed. Unsclerotized cuticle anterolateral to podonotal shield with a pair of subtriangular plates extending from the anterior level of podonotal shield to level of setae s2. Presternal area with four transverse pairs of presternal platelets; anteriormost pair usually subdivided into three platelets, two median pairs each usually subdivided into two elements, and basal pair generally undivided. Anterior hyaline margin of epigynal shield tongue-shaped, reaching to level of setae st3. Unsclerotized cuticle between genital and ventrianal shields with four transversely elongate platelets. Peritreme anteriorly reaching posterior level of coxae II, shorter than the posterior margin of podonotal shield. Fixed digit of chelicera bearing seven teeth.
Supplementary description
Adult female (Figures 6–11) — Five specimens measured.


Gnathosoma — (Figure 10). Gnathotectum triangular, with anterior margin finely denticulate, and a median smooth spine-like extension (Figure 10A). Internal malae totally separated from each other, the outer margins densely barbed in the basal half and smooth distally. Corniculi horn-like, slightly divergent, 62 (56–66) long, 22 (20–26) wide at base. Deutosternum with a smooth anterior ridge followed by seven denticulate transverse lines and, delimited by lateral lines (Figure 10B). Hypostomal setae acicular and smooth, h1 45 (36–50), h2 19 (15–24), h3 23 (20–28), sc 26 (24–28). Fixed cheliceral digit 85 (78–90) long, with seven teeth in addition to apical hook, and a setiform pilus dentilis (three small teeth, followed by pilus dentilis, and four large basal teeth); movable digit 80 (70–87) long, with two teeth; dorsal and antiaxial lyrifissures distinct, dorsal cheliceral seta small, apically blunt (Figure 10C). Palp setae aciculate and smooth, except for chisel-shaped anterolateral setae on palp genu and palp femur, number of setae on palp trochanter–tarsus, 2-5-6-14-15, apotele 3-tined.
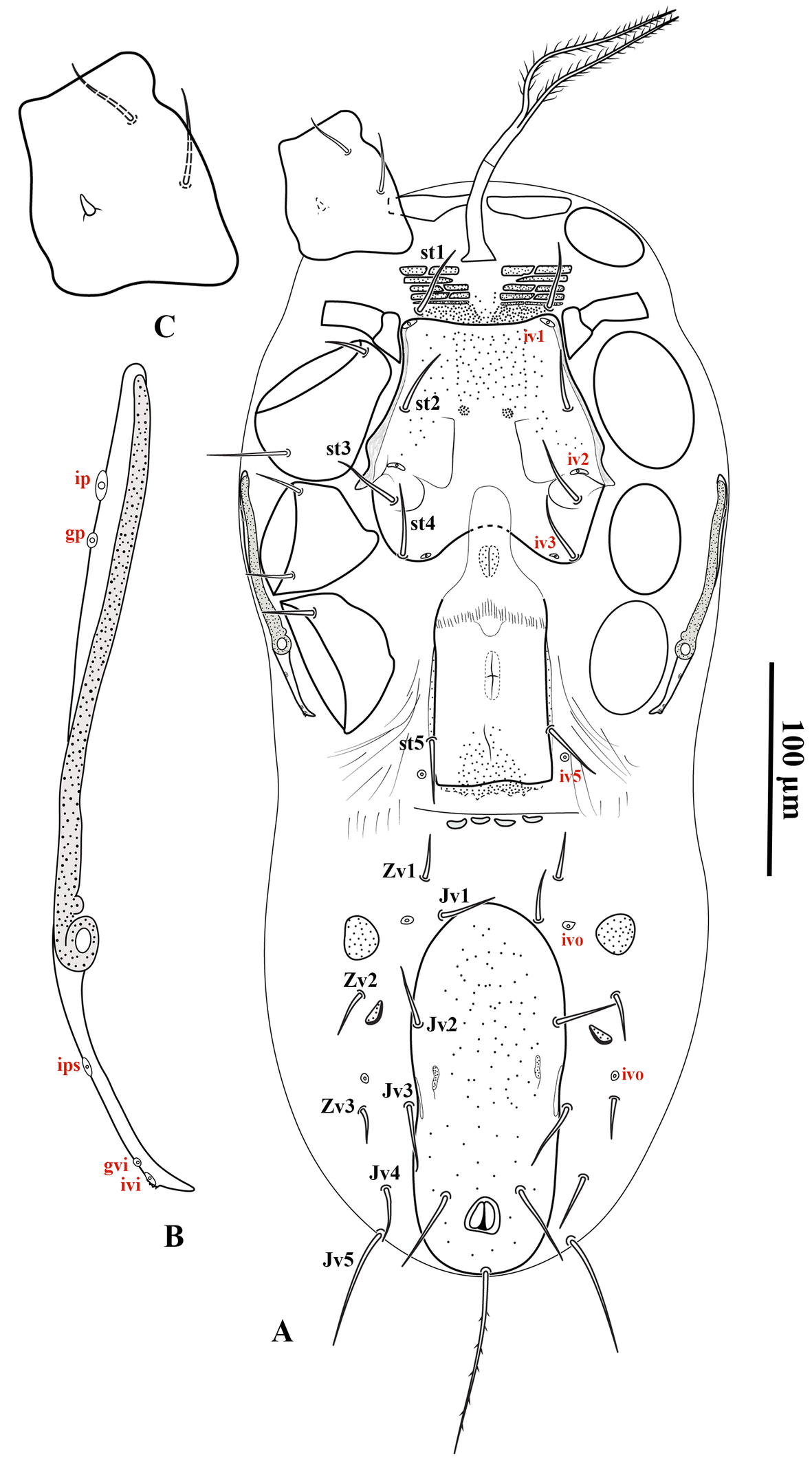


Dorsal idiosoma — (Figure 6, 8A). Idiosoma 510 (472–544) long and 250 (220–272) wide at the level of r3. Podonotal shield 210 (200–220) long and 171 (163–180) wide at the level of s3, usually with faint punctuation over most of the surface, and an unsclerotized band along the posterior margin, with 16 pairs of setae (j1–j6, z1–z6, s3–s6) and five pairs of distinguishable poroids (id1 posterlaterad z1, id2 anterolaterad z3, id4 posterolaterad s5, id5 anterolaterad j4 and id6 about in transverse line with z6); scleronoduli absent. Unsclerotized cuticle laterad podonotal shield with six pairs of setae (s1, s2 and r2–5); setae r1 absent. Unsclerotized cuticle anterolaterad podonotal shield with a pair of subtriangular plates extending from the level of setae z1 to s2 (in some specimens, lateral margin of subtriangular plates extends onto ventral surface between coxae I and II). Opisthonotal shield 230 (220–252) long and 90 (80–95) wide at the level of S3, usually with faint punctuation over the entire surface; bearing 15 pairs of setae (J1–5, Z1–5 and S1–5), 12 pairs of poroids (idm1, idm2, idm3, idm4, idm5, is1, Idl1, idl2, idl3, idl4, idl5, idx) and two pairs of glands (gd8 and gd9). Unsclerotized cuticle laterad opisthonotal shield with nine pairs of setae (R1–6 and UR3–5) and two pairs of poroids posteriad R3 (idR3) and posterior of opisthonotal shield (ipv), one pair of narrow platelets flanking opisthonotal shield at level of setae R3. Measurements of setae: j1 41 (38–42), j2 30 (26–34), j3 31 (29–34), j4 32 (30–34), j5 29 (27–30), j6 26 (25–28), z1 24 (22–26), z2 31 (30–32), z3 31 (28–36), z4 28 (24–32), z5 30 (27–32), z6 36 (35–38), s1 28 (26–30), s2 28 (26–30), s3 34 (30–34), s4 32 (30–34), s5 32 (30–34), s6 37 (34–40), r2 36 (33–40), r3 46 (43–50), r4 24 (22–26), r5 32 (30–34), J1 25 (24–26), J2 25 (25), J3 25 (24–26), J4 26 (25–28), J5 13 (10–14), Z1 27 (24–30), Z2 28 (25–30), Z3 34 (30–36), Z4 42 (38–44), Z5 58 (55–60), S1 36 (32–38), S2 33 (30–36), S3 33 (32–36), S4 36 (34–38), S5 34 (32–36), R1 26 (24–28), R2 25 (24–26), R3 25 (24–27), R4 25 (24–26), R5 29 (28–30), R6 33 (30–36), UR3 21 (20–22), UR4 19 (17–20), UR5 17; all dorsal setae aciculate and smooth, except J3 and Z3–5, aciculate and slightly serrate.
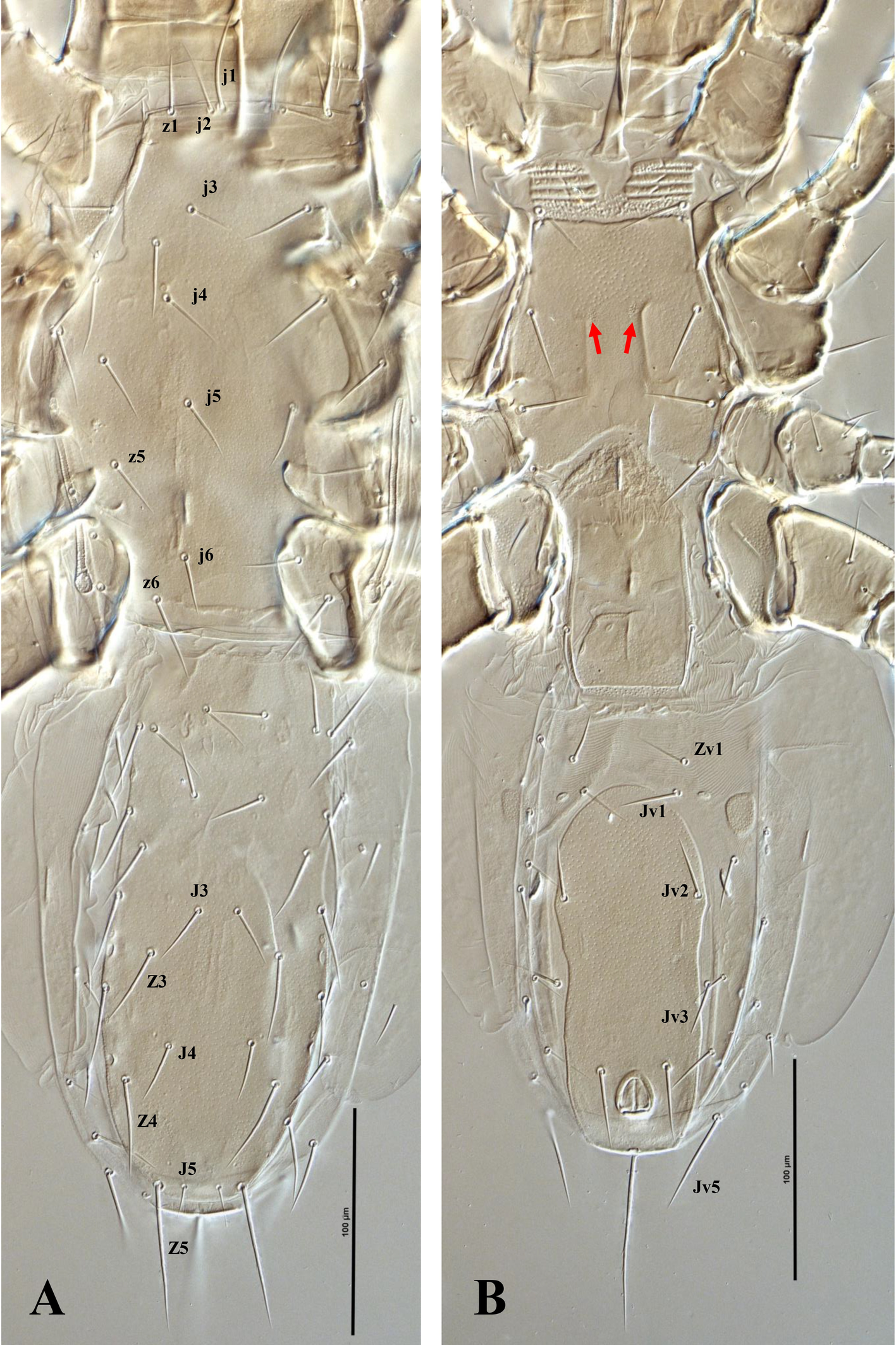


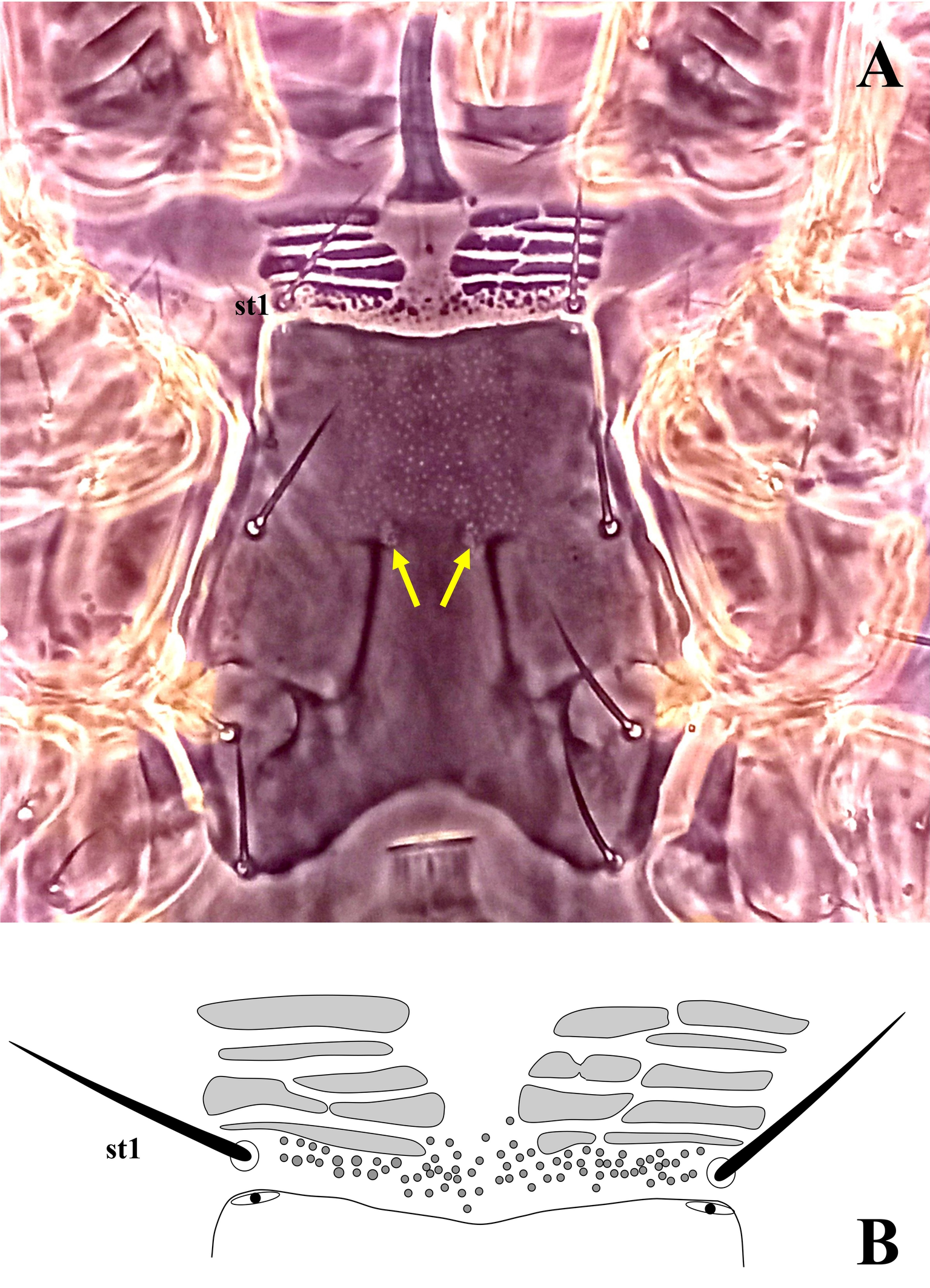


Ventral idiosoma — (Figure 7A–B, 8B, 9A–B). Base of tritosternum 74 (68–82) long and 15 (14–15) wide proximally; laciniae pilose, 102 (100–104) long, separated for about 80% of their total length. With one pair of elongate transverse plates anteriad base of tritosternum. Presternal region anteriad st1 (st1 on the granulated cuticle) with four transverse pairs of granulate presternal platelets, basal row narrower, and usually undivided, followed by two pairs of anterior rows, each usually subdivided into left and right elements, anteriormost pair larger than others, commonly subdivided into three platelets (Figure 10B). Sternal shield 100 (92–113) long, and 113 (103–120) wide at level between coxae II–III, with a punctate area on the anteromedian region and one pair of median small circular areas with dense punctation; with almost straight anterior margin; posterior margin concave; with three pairs of setae (st2–st4) and three pairs of poroids (iv1, iv2, and iv3). Endopodal plates between coxae I–II free from sternal shield. Epigynal shield long, extending posteriorly well beyond the level of coxa IV; anterior margin tongue-like, overlapping posterior margin of sternal shield; with heart-shaped structure; posterior margin slightly straight and punctate; distance between st5–st5 58 (50–60). Poroids iv5 on unsclerotized cuticle, posterolaterad st5. Unsclerotized cuticle between genital and ventrianal shields with four transversely narrow platelets. Ventrianal shield pill-shape, with faint punctuation; 157 (130–170) long and 70 (68–72) wide; with one pair of setae (Jv2) in addition to circumanals setae, without poroids, Jv3 on lateral margins of shield. Unsclerotized cuticle around ventrianal shield with six pairs of setae (Jv1, Jv4, Jv5, Zv1–Zv3), two pairs of platelets (anterior pair larger), and two pairs of poroids (posterolaterad Jv1 and anteriad Zv3). Peritreme short, extending anteriorly to posterior margin of coxa II (at level of r3). Peritrematic shield narrow, confined to region along peritreme, extending posteriorly to lateral of posterior margin of coxa IV; with a pair of distinguishable poroids and a pair of glands on lateral region of shield and two pairs of distinguishable poroids and a pair of glands posterior to stigma (Figure 7B). Measurements of setae: st1 31 (30–35), st2 32 (28–34), st3 31 (28–34), st4 30 (29–32), st5 24 (22–26), Jv1 24 (23–25), Jv2 28 (25–30), Jv3 30 (28–35), Jv4 26 (25–27), Jv5 47 (45–50), Zv1 22 (20–25), Zv2 23 (22–25), Zv3 17 (15–18), para-anal 35 (33–37), post-anal 75 (74–80); all setae aciculate and smooth, except post-anal, slightly serrate.


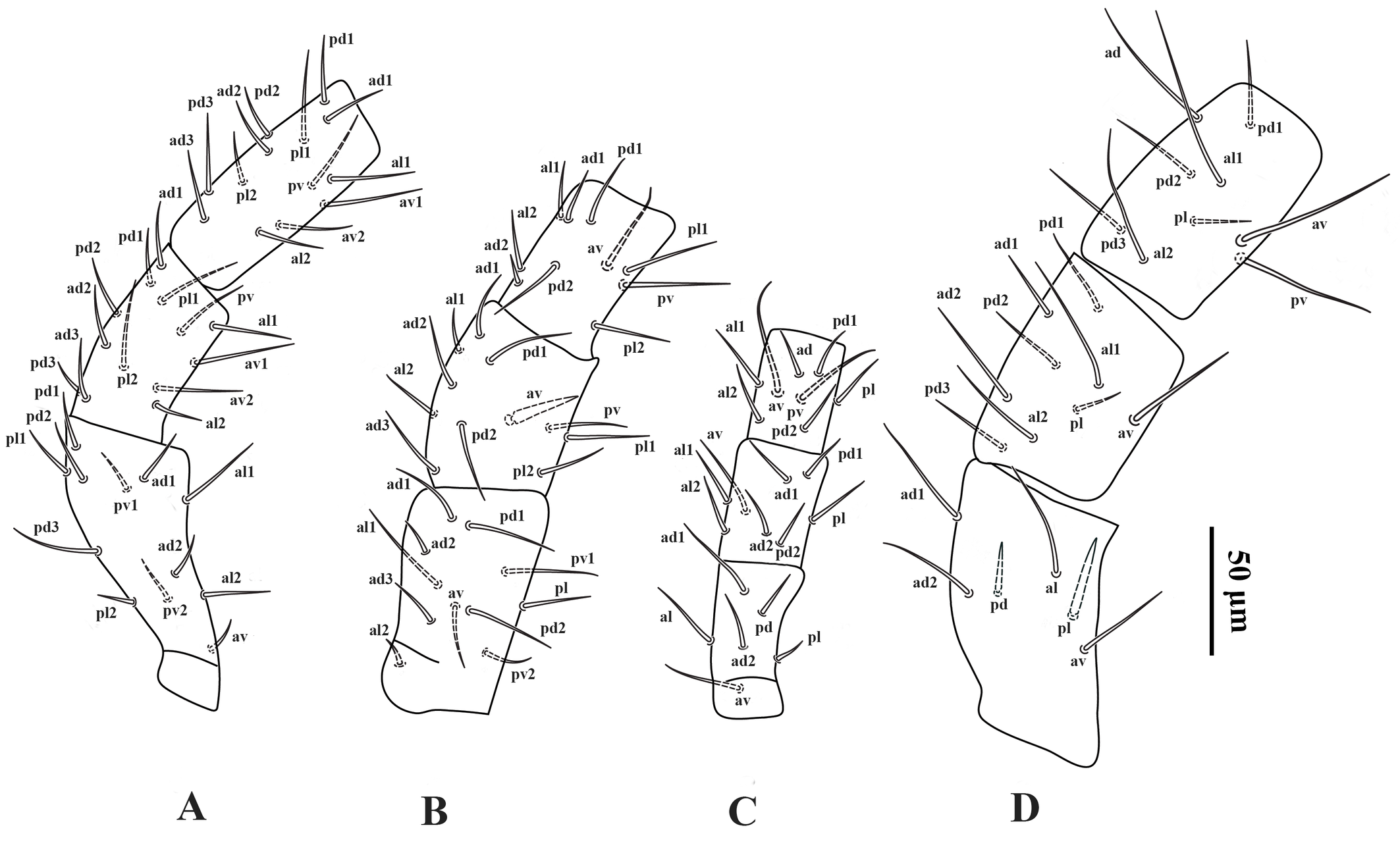


Legs — (Figure 11A–D). Lengths of legs I–IV: 418 (400–440), 294 (272–340), 260 (248–280), 402 (384–416), respectively. Leg IV thicker than legs I–III. Setation of legs I–IV standard for the genus, including 12 setae on genu II and 11 setae on tibia I; dorsal surface of coxa I with a short spine (Figure 5C); setae pd and pl on femur IV spine-like; seta av on genu II spine-like; seta av on genu IV slightly thickened; setae av and pv on tibia II–IV slightly thickened.
Male. Unknown
Material examined
Five females from rice husks at Sangar district (37°08′07.06''N, 49°41′07.08''E), Rasht County, Guilan Province, Iran, 16 June 2021; one female from weed at Rasht County (37°15′55.02''N, 49°35′29.07''E), 27 November 2018; three females from soil at Someh Sara County (37°18′18.06''N, 49°19′30.03''E), Guilan Province, Iran, 25 June 2022; two females from rice warehouse at Talesh County (37°47′58.06''N, 48°54′27.01''E), Guilan Province, Iran, 27 November 2018. Nine female specimens are deposited in the Acarology Laboratory, Department of Plant Protection, Faculty of Agricultural Sciences, University of Guilan, Rasht, Iran; two female specimens are deposited in Jalal Afshar Zoological Museum, Tehran, Iran.
Remark
After Rh. cavernicola was synonymized with P. posnaniensis by Castilho et al. (2012), the leg chaetotaxy proposed by Moraza (2004) for Rh. cavernicola was subsequently adopted as representative of P. posnaniensis, because leg setation had not been provided in the original description of the latter species. This substitution, however, unintentionally introduced a source of error, as Moraza's data indicated reduced numbers of setae on three leg segments compared with those reported by Evans & Purvis (1987): genu II 10 (vs. 11) and tibia I 12 (vs. 13). Re-examination of the type material of Rh. cavernicola (by SK) confirms that its leg chaetotaxy matches Evans & Purvis (1987) rather than the reduced counts given by Moraza (2004). Additional evidence (LM, pers. comm. with Dr. D. Gwiazdowicz) indicates that P. posnaniensis bears 11 and 13 setae on genu II and tibia I, respectively. The dorsal and ventral ornamentation of Rh. cavernicola also agrees with the original P. posnaniensis description.
Although the type specimens of P. posnaniensis were not examined directly by us, the oblique lines drawn in the anterolateral region of the podonotal shield in Figure 2 of its original description likely correspond to the unsclerotized median lines of the idiosoma shown in the same figure. These do not mark the true margins of the anterolateral shields, which in this genus partially surround the anterolateral area of the podonotal shield. On this basis, and in combination with the evidence discussed above, we confirm the synonymy of Rh. cavernicola and P. posnaniensis, and conclude that their leg chaetotaxy conforms to that presented by Manson (1972) and Evans & Purvis (1987).
Nasr (1978) described a new Protogamasellus species in his PhD dissertation as Protogamasellus sp. 1, providing detailed descriptions and illustrations of the species. However, measurements of the structures and leg setation, except for genua and tibiae I–IV, were overlooked. This species was later published as Protogamasellus denticus Nasr, in Zaher (1986), and subsequently transferred to Protogamasellopsis by Moraes et al. (2016). After careful re-evaluation of the original description and illustrations, together with supplementary information kindly provided by Dr. Nasr (SK, pers. comm.), we found that P. denticus is morphologically identical to P. posnaniensis and Rh. cavernicola. Consequently, based on the priority of description, P. denticus is considered the senior synonym of both species.
Amin & Khanjani (2022) described P. rhizoglyphusi as closely related to P. posnaniensis, distinguishing it primarily by the ornamentation of the sternal shield, leg chaetotaxy, and the reported absence of the dorsal cheliceral seta. Their interpretation appears to have been strongly influenced by Moraza's depiction of Rh. cavernicola, which shows an almost smooth sternal shield and reduced leg setation. However, the original P. posnaniensis description clearly shows a granulate anterior sternal region similar to that of P. rhizoglyphusi. The reported absence of the dorsal cheliceral seta is doubtful, as this seta in Protogamasellopsis is minute and somewhat laterally placed and can be easily overlooked if specimens are not properly mounted. The description of P. rhizoglyphusi also contains several inconsistencies. The opisthonotal shield is stated to be 273 (200–285) × 113 (90–110), but Figure 1 depicts it as approximately 293 × 122, exceeding the stated range; notably, the reported width of 113 already surpasses the maximum given. The 85 µm variation in shield length, based on the holotype and four paratypes, also warrants re-examination. Furthermore, Figure 7 shows coxa I dorsally without a spine, a diagnostic feature of the genus, which is not mentioned in the text. Figure 4 illustrates 12 and 10 setae on the palp tibia and tarsus, respectively, and the telotarsi of legs II–IV bear 9, 10, and 11 setae, contrary to the textual account. It is also worth noting that Amin & Khanjani (2022) placed P. denticus among the species of Protogamasellopsis lacking presternal platelets in their identification key. However, in the original description of P. denticus, these platelets are clearly illustrated in the presternal region. This discrepancy most likely resulted from an oversight in recognizing the presence of the platelets in the original figures, in which they are unambiguously visible. Taken together, the available evidence supports the interpretation that P. rhizoglyphusi is best treated as a putative junior synonym of P. posnaniensis (and therefore of P. denticus). Nevertheless, despite the strength and internal consistency of the morphological evidence discussed above, the absence of direct access to the type material of P. rhizoglyphusi currently precludes a formally definitive taxonomic conclusion. The type specimens of this species were not available for examination, and multiple email requests to the authors of the original description remained unanswered, preventing access to the material.
Abo-Shnaf et al. (2013) described P. zaheri from Egypt, the same country from which Protogamasellus denticus Nasr was originally described; however, they did not compare P. zaheri with the latter species, which was known from the same region. In their differential diagnosis, they compared P. zaheri with P. posnaniensis as the closest species and noted that the latter ''has the plate anterolaterad of the podonotal shield more elongate, extending from the level of z1 to the level of s5, bearing r2–r4″, citing the original description of P. posnaniensis and conspecific material collected from Brazil. In the original description of P. posnaniensis, however, these plates are evidently short and asetose, and neither the illustration nor the accompanying text provides any evidence of setae on them. The margins misinterpreted by those authors as anterolateral extensions of the podonotal shield are, in fact, membranous folds, illustrated laterally and medially on the idiosoma in their Figure 2, which do not represent the boundaries of the sclerotized shields. Furthermore, the Brazilian specimens referred to as P. posnaniensis actually possess such anterolateral plates with setae, a feature that does not occur in the true P. posnaniensis. If this condition is genuinely present in the Brazilian material, those specimens most likely represent an undescribed species.
Considering the characters used by Abo-Shnaf et al. (2013) to distinguish P. zaheri from P. posnaniensis, along with the original description of P. denticus and supplementary information kindly provided by Dr. Nasr (SK pers. com.), it appears highly probable that these two species are conspecific. However, because the type material of P. zaheri has not been examined, we treat this synonymy conservatively and regard P. zaheri as a potential junior synonym of P. denticus, pending re-examination of its type material. Moreover, according to Dr. Nasr, extensive sampling of Egyptian soils over the years has yielded only specimens attributable to P. denticus, which further supports this conclusion.
In summary, the available evidence strongly supports that Protogamasellopsis denticus, P. posnaniensis, and Rhodacarella cavernicola represent the same species, with P. denticus having nomenclatural priority. The diagnostic differences used to separate P. rhizoglyphusi from P. posnaniensis are inconsistent and largely based on misinterpretations of earlier descriptions; hence, P. rhizoglyphusi is regarded here as a putative junior synonym of P. denticus. Likewise, P. zaheri, described from Egypt and differing only slightly in leg chaetotaxy, is considered a potential junior synonym of P. denticus, pending re-examination of its type material.
Discussion
In P. denticus, the presternal region typically exhibits four transverse rows of granulated platelets. The sternal shield bears a well-defined, sclerotized anterior margin, which is followed anteriorly by an unsclerotized but lightly granulated area; immediately anterior to this area lies the lowermost (basal) row of presternal platelets, which is usually narrower than the more anterior rows. Above this, three additional rows are present; in most examined specimens, the two lower of these rows are each subdivided into paired left and right elements, whereas the uppermost row generally consists of three discrete platelets.
Minor deviations from this arrangement may occasionally occur, including slight asymmetry between the left and right sides within a single specimen. An example of such asymmetry is illustrated in Figure 9. Given the limited number of specimens available for direct examination and the absence of consistent differences among them, these subdivisions are here regarded as secondary intraspecific variations rather than stable diagnostic characters.
Species descriptions within Protogamasellopsis have generally been based on limited material, often derived from restricted geographic areas. As a result, the extent of morphological variation within species has remained poorly documented, and the stability of several characters traditionally used in diagnoses has not been adequately tested across broader population samples.
Recent studies increasingly suggest that this limitation may have contributed to an underestimation of intraspecific variation and, potentially, to the overlooking of cryptic diversity within the genus. In this context, molecular evidence provides an important independent perspective. For example, a large-scale DNA barcoding study of soil-, nest-, and poultry-associated mites from the Western Palearctic (Young et al. 2019) assigned specimens identified as P. corticalis to multiple BINs, indicating substantial genetic divergence within the nominal species. Notably, the authors did not propose formal taxonomic splitting, but instead emphasized the need for integrative approaches combining morphology, population-level sampling, and molecular data. This independent evidence supports a cautious interpretation of morphological variation in Protogamasellopsis and argues against premature taxonomic conclusions based on limited material.
The material examined in the present study, derived primarily from northern Iran and supplemented by direct or indirect comparisons with type material of several species, is consistent with this broader pattern. Although certain characters—such as the relative lengths of the circumanal setae and the posterior opisthonotal setae—appear to vary among individuals, the available evidence is insufficient to determine whether such differences reflect intraspecific variation or the presence of cryptic species. Accordingly, no definitive taxonomic conclusions are drawn here on this basis alone.
Another character that may prove informative in future taxonomic studies of Protogamasellopsis is the structure of the spermatheca. Unfortunately, in the material examined here, the components of this structure could not be observed with sufficient clarity, likely due to the condition of the specimens and the degree of clearing during slide preparation. As a result, no detailed interpretation of spermathecal morphology is attempted in the present study. Nevertheless, careful documentation of the spermatheca at early stages of slide preparation, before excessive clearing occurs, is strongly recommended. Detailed comparative data on this structure, especially when combined with population-level sampling and molecular evidence, may provide additional characters of taxonomic value for resolving species limits within this group.
Resolving these questions will require comprehensive revisionary work based on specimens collected from multiple geographic regions, with particular emphasis on the re-examination of type material and the inclusion of molecular data where possible. Until such data become available, observed morphological differences within Protogamasellopsis are best interpreted conservatively, within a framework that explicitly acknowledges the current limitations of sampling and comparative material.
Taken together, the evidence presented here demonstrates that several characters historically regarded as stable or diagnostic in Protogamasellopsis exhibit broader intraspecific variability than previously recognized. Reliable species delimitation within the genus will therefore require a cautious, integrative approach that emphasizes combinations of characters rather than isolated traits, supported by renewed examination of problematic type series and, where feasible, molecular data. Such approaches will be critical for refining the generic concept and establishing a more robust and stable taxonomic framework for Protogamasellopsis.
Acknowledgments
We are grateful to Dr. Hans Klompen (USA) for providing access to the type material of Rhodacarella cavernicola, which greatly facilitated our comparative analyses. We also thank Dr. Abdelrady K. Nasr (Egypt) for his valuable assistance in examining specimens of Protogamasellopsis denticus and for generously sharing photographs and additional information. Our sincere thanks are extended to Dr. Dariusz Gwiazdowicz (Poland) for examining the type material of P. posnaniensis and kindly providing detailed notes and documentation. We also thank Dr. Bruce Halliday (Australia) for sharing relevant literature. The project was supported by the Department of Plant Protection, College of Agricultural Sciences, University of Guilan, Rasht, Iran, which is greatly appreciated.
References
- Abo-Shnaf R., Castilho R.C., Moraes G.J. 2013. Two new species of Rhodacaridae (Acari: Mesostigmata) from Egypt and a key to the species of the family from the Mediterranean region. Zootaxa, 3718 (1): 28-38. https://doi.org/10.11646/zootaxa.3718.1.2
- Amin M.R., Khanjani M. 2022. Post embryonic stages of a new species of the genus Protogamasellopsis Evans & Purvis (Rhodacaridae: Acari) from Iran. Int. J. Acarol., 48(1): 67-75. https://doi.org/10.1080/01647954.2021.2022758
- Amin M.R., Khanjani, M. 2024. Development, reproduction and survival Protogamasellopsis rhizoglyphusi and Gaeolaelaps aculeifer (Mesostigmata: Rhodacaridae, Laelapidae) feeding on two astigmatine mite prey and notes on the behavior of P. rhizoglyphusi. Syst. Appl. Acarol., 29(1): 109-124. https://doi.org/10.11158/saa.29.1.8
- Anonymous. 2025. Rhodacaridae Database. Available from: http://www.lea.esalq.usp.br/rhodacaridae/busca/arvore (accessed 28/8/2025).
- Athias-Henriot C. 1975. Nouvelles notes sur les Amblyseiini II. Le relevé organotaxique de la face dorsale adulte (Gamasides Protoadenique, Phytoseiidae). Acarologia, 17: 20-29.
- Braga A.F. & Castilho, R.D.C., 2024. Article ZOOTAXA. Zootaxa, 5497(1): 070-082. https://doi.org/10.11646/zootaxa.5497.1.3
- Castilho R.C., Moraes G.J., Halliday B. 2012. Catalogue of the mite family Rhodacaridae Oudemans, with notes on the classification of the Rhodacaroidea (Acari: Mesostigmata). Zootaxa, 69: 1-69. https://doi.org/10.11646/zootaxa.3471.1.1
- Castilho R.C., Moraes G.J., Silva E.S., Silva L.O. 2009. Predation potential and biology of Protogamasellopsis posnaniensis Wisniewski & Hirschmann (Acari: Rhodacaridae). Biol. Control, 48: 164-167. https://doi.org/10.1016/j.biocontrol.2008.10.004
- Castilho R.C., Venancio R., Narita, J.P.Z. 2015. Mesostigmata as biological control agents, with emphasis on Rhodacaroidea and Parasitoidea. In: Carrillo D., Moraes G.J., Peña, J.E. (eds.), Prospects for biological control of plant feeding mites and other harmful organisms. Cham: Springer International Publishing, p.1-31. https://doi.org/10.1007/978-3-319-15042-0_1
- Evans G.O. 1963a. Observations on the chaetotaxy of the legs in the free-living Gamasina (Acari: Mesostigmata). Bull. Brit. Mus. (Nat. Hist.) Zool., 10: 277-303. https://doi.org/10.1080/00222936308651393
- Evans G.O. 1963b. Some observations on the chaetotaxy of the pedipalps in the Mesostigmata (Acari). Ann. & Mag. Nat. Hist. Ser. 13 (6): 513-527. https://doi.org/10.1080/00222936308651393
- Evans G.O., Purvis, G. 1987. A new ascid mite from St Helena with observations on the Protogamasellus complex (Acari: Mesostigmata). J. Nat. Hist., 21: 855-861. https://doi.org/10.1080/00222938700770511
- Halliday R.B., Walter D.E., Lindquist, E.E. 1998. Revision of the Australian Ascidae (Acarina: Mesostigmata). Invertebr. Taxon., 12: 1-54. https://doi.org/10.1071/IT96029
- Johnston D.E., Moraza M.L. 1991. The idiosomal adenotaxy and poroidotaxy of Zerconidae (Mesostigmata: Zerconina). In: Dusbábek F., Bukva, V. (eds.), Modern Acarology, Vol. 2. Academia, Prague, p. 349-356.
- Karg W. 1994a. Raubmilben der Ascidae, Ameroseiidae, Rhodacaridae und Macrochelidae auf dem Galapagos-Archipel (Acarina, Parasitiformes). Mitteilungen aus dem Museum fur Naturkunde in Berlin. Zoologisches Museum und Institut fur Spezielle Zoologie (Berlin), 70: 113-131. https://doi.org/10.1002/mmnz.19940700107
- Karg W. 1994b. Raubmilben der Cohors Gamasina Leach (Acarina, Parasitiformes) vom Galapagos-Archipel. Mitt. Zool. Mus. Berl. 70: 179-216. https://doi.org/10.1002/mmnz.4840700202
- Karg W. 2000. Zur Systematik der Raubmilbenfamilien Hypoaspididae v. Vitzthum, 1941 und Rhodacaridae Oudemans, 1902 (Acarina, Parasitiformes) mit neuen Arten aus Sud- und Mittelamerika. Mitt. Zool. Mus. Berl., 76: 243-262. https://doi.org/10.1002/mmnz.4850760207
- Kazemi S., Rajaei, A. 2013. An annotated checklist of Iranian Mesostigmata (Acari), excluding the family Phytoseiidae. Persian J. Acarol., 2(1): 63-158. https://doi.org/10.22073/pja.v2i1.9950
- Lindquist E.E. 1994. Some observations on the chaetotaxy of the caudal body region of gamasine mites (Acari: Mesostigmata), with a modified notation for some ventrolateral body setae. Acarologia, 35: 323-326.
- Lindquist E.E., Evans G.O. 1965. Taxonomic concepts in the Ascidae, with a modified setal nomenclature for the idiosoma of the Gamasina (Acarina: Mesostigmata). Mem. Entomol. Soc. Can., 47: 1-64. https://doi.org/10.4039/entm9747fv
- Lindquist E.E., Krantz G.W., Walter D.E. 2009. Order Mesostigmata. In: Krantz G.W., Walter D.E. (eds.), A Manual of Acarology, Third Edition, Texas Tech University Press, Lubbock, Texas, p. 124-232.
- Manson D.C.M. 1972. A new mite of the genus Protogamasellus (Acarina: Ascidae). Acarologia, 13: 437-445.
- Momen F., Fahim S., Barghout, M. 2020. Mass production of predatory mites and their efficacy for controlling pests. Cottage Industry of Biocontrol Agents and Their Applications: Practical Aspects to Deal Biologically with Pests and Stresses Facing Strategic Crops, pp.157-200. https://doi.org/10.1007/978-3-030-33161-0_5
- Moraes G.J. de, Britto E.P., Mineiro J.L.C., Halliday B. 2016. Catalogue of the mite families Ascidae Voigts & Oudemans, Blattisociidae Garman and Melicharidae Hirschmann (Acari: Mesostigmata). Zootaxa, 4112 (1): 1-299. https://doi.org/10.11646/zootaxa.4112.1.1
- Moraza M.L. 2004. Rhodacarella, a new genus of Rhodacaridae mites from North America (Acari: Mesostigmata: Rhodacaridae). Zootaxa, 470: 1-10. https://doi.org/10.11646/zootaxa.470.1.1
- Rossini L.A.C.J., Prado T.J., Ferreira R.J., Soares P.L.M., de Moraes G.J., Castilho R.C. 2022. Suitability of the soybean cyst nematode as prey to Protogamasellopsis zaheri (Acari: Mesostigmata: Rhodacaridae). Biol. Control, 170: 104905. https://doi.org/10.1016/j.biocontrol.2022.104905
- Rueda-Ramirez D., Palevsky E., Ruess L. 2022. Soil Nematodes as a means of conservation of soil predatory mites for Biocontrol. Agronomy, 13(1): 32. https://doi.org/10.3390/agronomy13010032
- Shaw M. 1999. Mites and ticks from wedge-tailed shearwater (Puffinus pacificus) burrows on Masthead Island. Queensl. Nat. 37: 43-47.
- Wiśniewski J., Hirschmann W. 1991. Protogamasellopsis posnaniensis nov. spec. (Acarina: Mesostigmata) aus Palmenhaus in Pollen. Bulletin of the Polish Academy of Sciences. Biol. Sci. 39: 189-194.
- Young M.R., Moraza M.L., Ueckermann E., Heylen D., Baardsen L.F., Lima-Barbero J.F., Gal S., Gavish-Regev E., Gottlieb Y., Roy L., Recht E., El Adouzi M., Palevsky E. 2019. Linking morphological and molecular taxonomy for the identification of poultry house, soil, and nest dwelling mites in the Western Palearctic. Sci. Rep. 9: 5784. https://doi.org/10.1038/s41598-019-41958-9
- Zaher M.A. 1986. Survey and Ecological Studies on Phytophagous, Predaceous and Soil Mites in Egypt. II-A: Predaceous & No phytophagous Mites in Egypt (Nile Valley and Delta). Text PL. 480 Programme U.S.A., Project No. EG-ARS-30, Grant No. FG-EG-139, 567 pp.



2026-01-23
Date accepted:
2026-02-04
Date published:
2026-02-06
Edited by:
Faraji, Farid

This work is licensed under a Creative Commons Attribution 4.0 International License
2026 Mohammadi, Leila; Hajizadeh, Jalil and Kazemi, Shahrooz
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



