Khaustovella insolita n. gen., n. sp. (Acari, Oribatida, Scheloribatidae), an arboreal mite from Vietnam
Ermilov, Sergey G.  1
1
1✉ University of Tyumen, X-BIO Institute, Tyumen, Russia.
2026 - Volume: 66 Issue: 1 pages: 81-91
https://doi.org/10.24349/9z0x-83r2ZooBank LSID: 1F3CE372-CE79-4931-B5D4-26BF1C1A00DE
Original research
Keywords
Abstract
Introduction
Cat Tien National Park is located in southern Vietnam. It has an area of 720 km2 and protects one of the largest areas of lowland tropical forests left in Vietnam. The oribatid mite (Acari, Oribatida) fauna of the park's soil and litter has been relatively well investigated (e.g., Ermilov et al. 2012, 2025a; Ermilov and Anichkin 2013). In recent years, the ecology of the park's arboreal oribatid mites has been actively studied (Salavatulin et al. 2022; Ermilov and Salavatulin 2025).
During taxonomic identifications of oribatid mites of the family Scheloribatidae associated with trees in the Cat Tien National Park, one new species representing a new genus, was found. The main goal of the present paper is to describe Khaustovella n. gen., with Khaustovella insolita n. sp. as type species, based on the females.
Scheloribatidae is a large family, comprising about 390 species belonging to 49 supraspecies taxa (personal data). Representatives of the family are widespread, occurring in all continents and biogeographical regions and inhabiting diverse natural and anthropogenic habitats. Some Vietnamese scheloribatids are arboreal (Ermilov and Anichkin 2014; Ermilov 2023; Ermilov and Salavatulin 2023), however, they cannot be confused with representatives of Khaustovella insolita n. sp., which has a unique morphology (see Generic diagnosis below).
Material and methods
Sampling
Tree branches were collected via climbing different trees (Dipterocarpus turbinatus, Lauraceae sp., Mangifera sp., Polyalthia sp.) using spikes and other special equipment at the height of 15–25 m above ground in the Cat Tien National Park, southern Vietnam. Mites were subsequently extracted by high-pressure flushing and by heptane flotation in laboratory conditions. Detailed descriptions of arboreal acarofauna collections and extraction techniques are presented in Salavatulin (2019).
Observation and documentation
For measurement and illustration, specimens were mounted in lactic acid on temporary cavity slides. Body length was measured in lateral view, from the tip of the rostrum to the posterior edge of the notogaster. Body width refers to the maximum width of the notogaster in dorsal view. Curved structures, e.g. setae, were measured from the side to avoid foreshortening. All body measurements are presented in micrometers (µm). Formulas for leg setation are given in parentheses according to the sequence trochanter-femur-genu-tibia-tarsus (famulus of tarsus I included). Formulas for leg solenidia are given in square brackets, according to the sequence genu-tibia-tarsus. Paired structures are described in the singular, unless otherwise noted. Drawings were made with a camera lucida using a Leica DM 2500 light microscope. Images were obtained with an AxioCam ICc3 camera using a Carl Zeiss transmission light microscope Axio Lab.A1.
Terminology
Morphological terminology used in this paper follows that of F. Grandjean: see Travé and Vachon (1975) for references, Norton (1977) for leg setal nomenclature, and Norton and Behan-Pelletier (2009) for overview.
Abbreviations
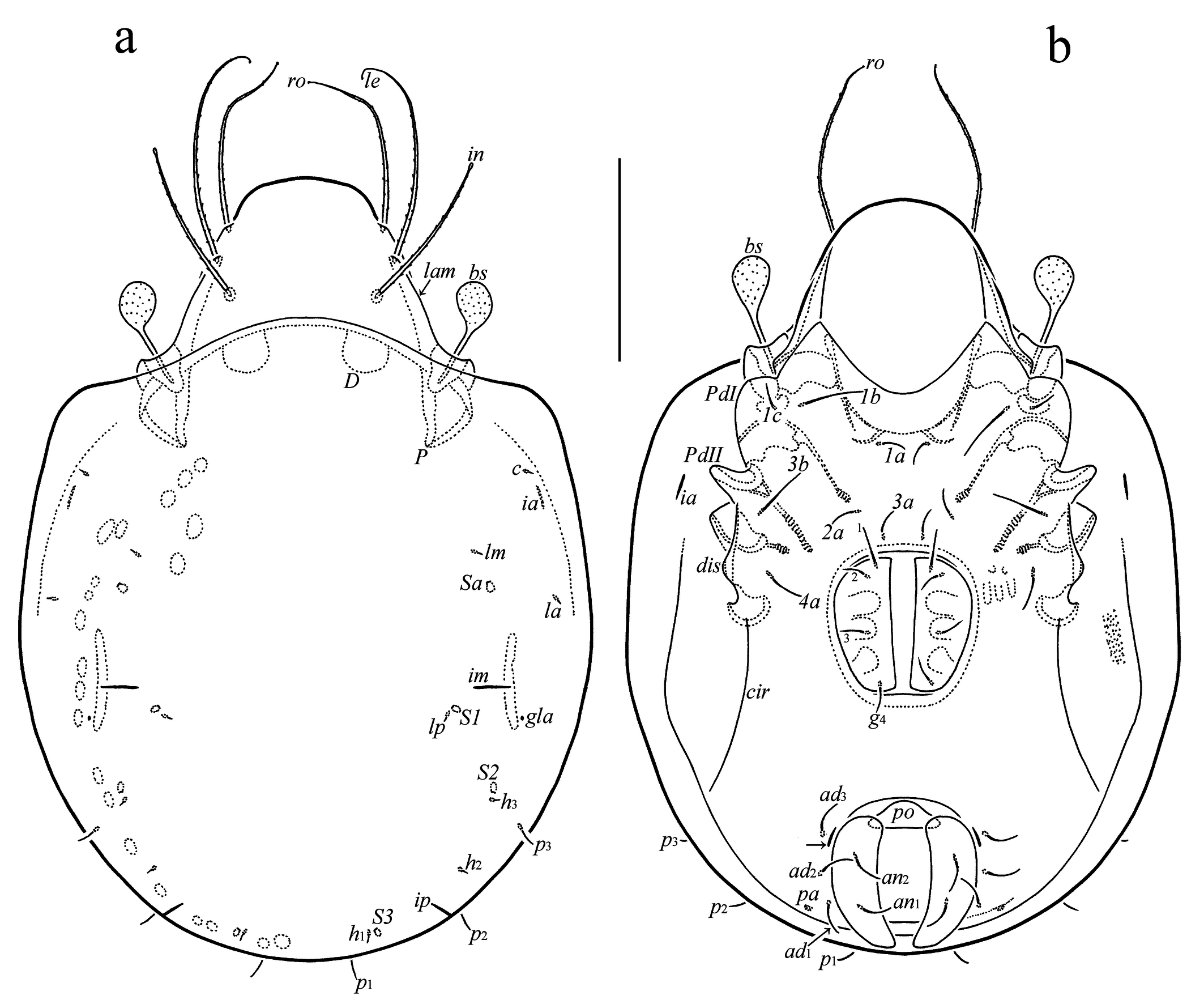
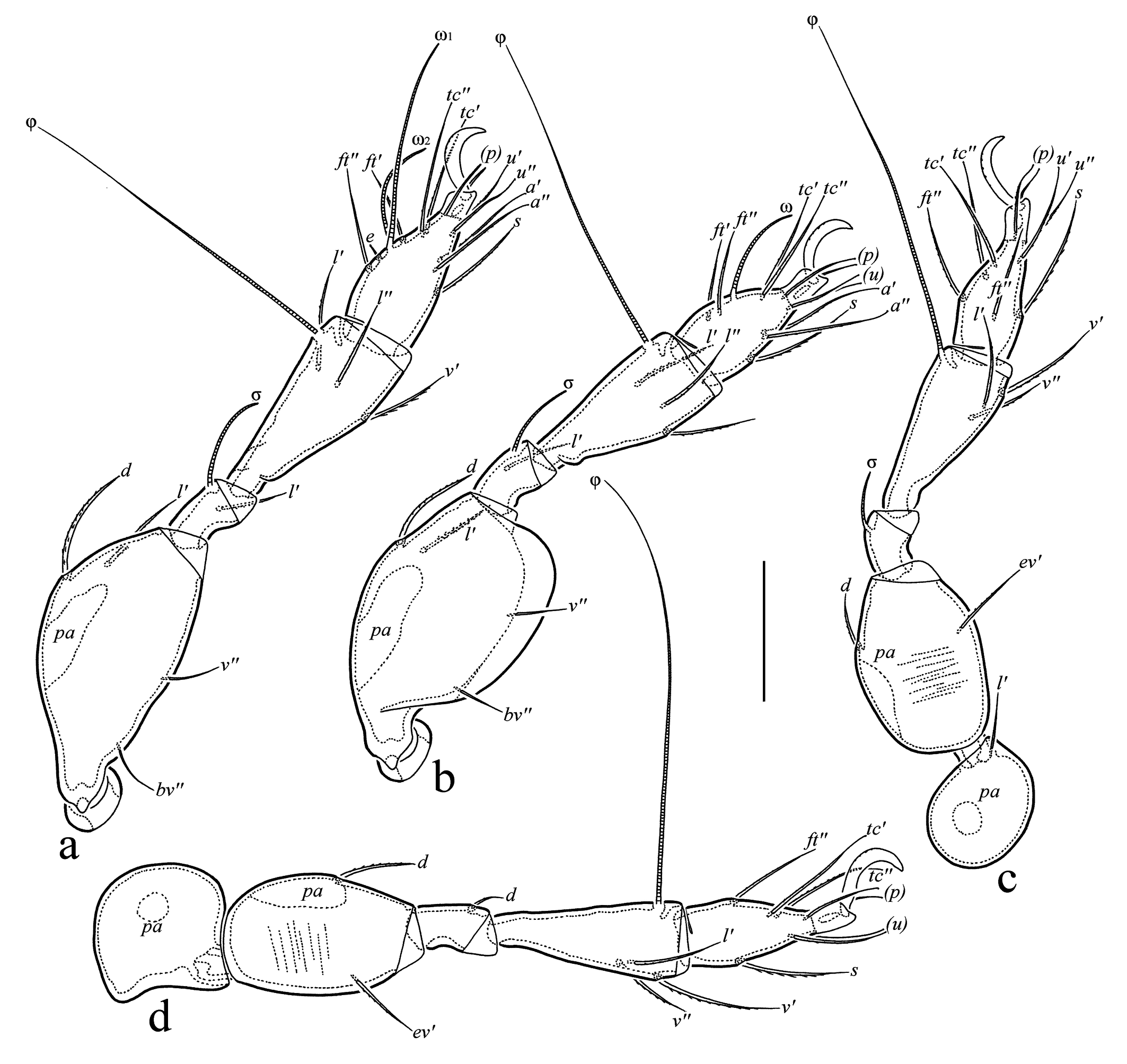
Prodorsum: lam = lamella; plam = prolamella; slam = sublamella; kf = keel-shaped ridge; ro, le, in, bs = rostral, lamellar, interlamellar, and bothridial setae, respectively; bo = bothridium; D = dorsophragma; P = pleurophragma. Notogaster: c, la, lm, lp, h1–h3, p1–p3 = setae; Sa, S1, S2, S3 = saccules; ia, im, ip, ih, ips = lyrifissures; gla = opisthonotal gland opening. Gnathosoma: a, m, h = subcapitular setae; or = adoral seta; d, l, cm, acm, ul, su, lt, vt, sup, inf = palp setae; ω = palp solenidion; cha, chb = cheliceral setae; Tg = Trägårdh's organ. Epimeral and lateral podosomal regions: 1a, 1b, 1c, 2a, 3a, 3b, 4a = epimeral setae; PdI, PdII = pedotecta I and II, respectively; dis = discidium; cir = circumpedal carina. Anogenital region: g, ag, an, ad = genital, aggenital, anal, and adanal setae, respectively; iad = adanal lyrifissure; pa = porose area; po = preanal organ. Legs: pa = porose area; ω, σ, φ = solenidia; e = famulus; d, l, v, ev, bv, ft, tc, p, u, a, s = setae.
Taxonomy
Family Scheloribatidae
Genus Khaustovella n. gen.
ZOOBANK: D8974F4E-29C0-4DED-A776-B0927883AF57 ![]()
Type species: Khaustovella insolita n. sp.
Generic diagnosis (adult)
With character states of Scheloribatidae (Weigmann 2006; Norton and Behan-Pelletier 2009). Body size. Small, length less than 300. Integument. Body without heavy sculpturing and ornamentation. Prodorsum. Rostrum rounded. Lamella simple, without cusp, positioned submarginally. Translamella and tutorium absent. Prolamella absent or present. Sublamella and keel-shaped ridge present. Sublamellar porose area absent. Rostral, lamellar, and interlamellar setae long, setiform. Bothridial seta clavate, with well-developed head. Bothridium cup-shaped, with lateral scale. Exobothridial seta absent. Dorsosejugal porose area absent. Notogaster. Anterior margin of notogaster convex medially. Pteromorph broadly rounded, immovable, slightly curved ventrad. Octotaxic system as saccules. Ten pairs of short, simple setae. Gnathosoma. Subcapitulum diarthric. Chelicera chelate-dentate. Epimeral and lateral podosomal regions. Epimeres III and IV with reduced number of setae. Pedotecta I and II represented by small lamina. Humeral porose areas and custodium absent. Discidium and circumpedal carina present. Anogenital region. Genital aperture comparatively large, distance between genital and anal apertures equal to length of genital aperture or slightly shorter. Four pairs of genital, two pairs of anal, and three pairs of adanal setae. Aggenital setae absent. Adanal lyrifissure located close and parallel to anal plate. One pair of adanal porose areas located posterolateral to anal aperture. Marginal porose area absent. Legs. All tarsi with one claw. Porose area present on femora I–IV and on trochanters III, IV. Leg chaetome strikingly reduced.
Etymology
The generic name is dedicated to Dr. Alexander A. Khaustov, a famous acarologist from the University of Tyumen, Tyumen, Russia, for his extensive and invaluable contributions to our knowledge of mites.
Remarks
Khaustovella n. gen. differs from the other supraspecies taxa of Oripodoidea by two apomorphies: the presence of one pair of distinct porose areas in the adanal region (posterolateral to anal aperture), and the striking reduction of the leg chaetome. Also, see the Discussion section below.
Khaustovella insolita n. sp.
ZOOBANK: 9A2F981E-2B2F-4023-B032-E92BE357E413 ![]()
(Figures 1–4)
Type material
Holotype (female) and 21 paratypes (females) — Southern Vietnam, Dong Nai Province, Dong Nai Biosphere Reserve, Cat Tien National Park, 11°25′ N, 107°25′ E, about 130 m a.s.l., branches from the Dipterocarpus turbinatus tree at the height of 15–25 m, 8.XII.2021 (V.M. Salavatulin).
The holotype is deposited in the collection of the Senckenberg Museum of Natural History, Görlitz, Germany. Six paratypes are deposited in the collection of the University of Tyumen, Museum of Zoology, Tyumen, Russia. Fifteen paratypes are in the personal collection of S.G. Ermilov. All specimens are preserved in 70% solution of ethanol with a drop of glycerol.
Additional (non-type) material — Two adults (females): same as the type material, but branches from the Mangifera sp. tree (8.XII.2021). Two adults (females): same as the type material, but branches from the Polyalthia sp. tree (12.XII.2021). One adult (female): same as the type material, but branches from the Lauraceae sp. tree (12.XII.2021).
All specimens (in 70% solution of ethanol with a drop of glycerol) are in the personal collection of S.G. Ermilov.
Diagnosis
Presumably parthenogenetic. Body length 184–225. Rostrum broadly rounded. Prolamella absent or present. Keel-shaped ridge broad. Rostral and lamellar setae long, setiform, curved in distal half, slightly swollen apically, barbed. Interlamellar seta long, setiform, erect, distinctly swollen apically, barbed. Bothridial seta medium-sized, clavate, roughened. All notogastral setae short, acicular. Palptarsus with one seta vt. Epimeral setal formula 3-1-2-1. Epimeral and anogenital setae simple, thin. Distance between genital and anal apertures equal to length of genital aperture or shorter. Adanal setae inserted close and lateral to anal plate. One pair of adanal porose areas small. Leg chaetome strongly reduced, e.g., setal formula of trochanters 0-0-1-0, genua I and II with one seta, setae it and pv absent on all tarsi, tibia I and tarsus II with one solenidion.
Description
Measurements – Body length 221 (holotype), 184–225 (paratypes). Body width 154 (holotype), 142–176.


Integument (Figures 3c, d, 4a) – Body color light brown. Surface microfoveolate (visible under high magnification in dissected specimens). Lateral side of body with dense microgranulate cerotegument. Paraxial side of leg femora III and IV partially with transverse ridges ridges.
Prodorsum (Figures 1a, 2a, 4b–d) – Rostrum broadly rounded. Lamella as long as half of prodorsum. Translamella absent. Prolamella absent or present, complete (reaching insertion of rostral seta), or developed partially. Sublamella long. Keel-shaped ridge broad. Rostral (49–56) and lamellar (56–64) setae setiform, curved in distal half, slightly swollen apically, barbed. Interlamellar seta (49–52) setiform, erect, distinctly swollen apically, barbed. Bothridial seta (26–30) clavate, roughened, with short stalk and longer head. Head of bothridial seta slightly elongate (visible in lateral view), broadly rounded distally. Dorsophragmata comparatively large, distance between them equal to 1–1.5 of dorsophragma width.
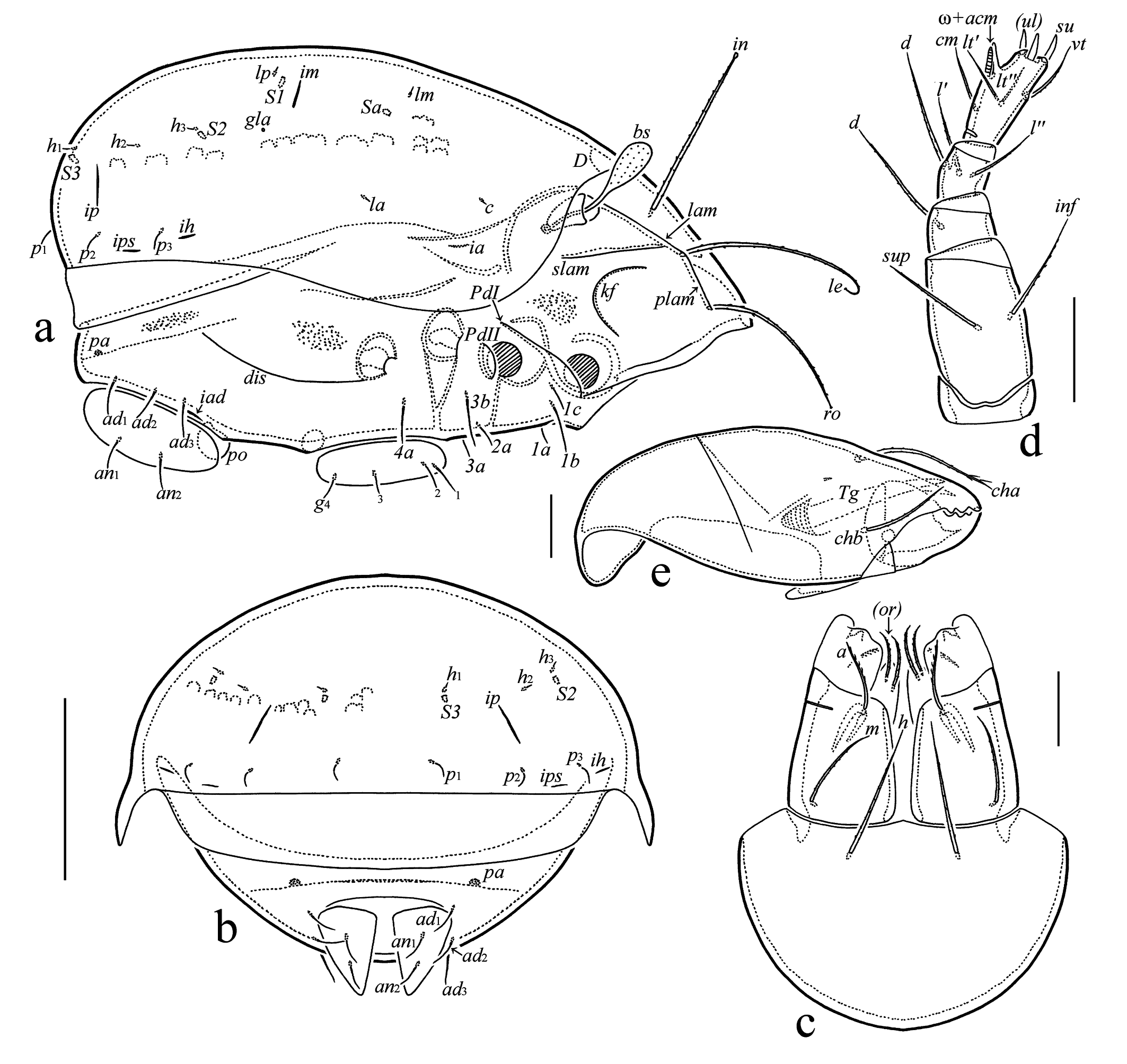


Notogaster (Figures 1a, 2a, b, 4e) – Pteromorph medium-sized, broadly rounded laterally, its anterior margin slightly oblique. Setae (p1–p3 7; c, la, lm, lp, h1–h3 4) acicular. Saccules (Sa, S1, S2, S3) drop-like, Sa located posterior to lm, S1 close to lp, S2 close to h3, S3 close to h1. Opisthonotal gland opening and all lyrifissures distinct.
Gnathosoma (Figures 2c–e) – Subcapitulum size 61–67 × 41–49. Subcapitular setae h (19) and m (15) setiform, thin, roughened, seta a (11) setiform, with several distinct barbs unilaterally. Two pairs of adoral setae (7) setiform, roughened. Palp length 41–49. Palp setal formula 0-2-1-3-8(+ω). One seta vt present on palptarsus. Postpalpal seta (4) spiniform. Chelicera length 71–75. Cheliceral seta cha (22) setiform, slightly barbed, with some cilia in distal part, chb (15) setiform, slightly barbed.
Epimeral and lateral podosomal regions (Figures 1b, 2a, 4f) – Epimeral setal formula 3-1-2-1. Setae (1b, 3b 15; 4a 11; 1a, 1c, 2a, 3a 7–9) simple, thin. Discidium subtriangular, with rounded tip. Circumpedal carina comparatively short, directed to acetabulum IV.


Anogenital region (Figures 1b, 2a, b, 4g–i) – Genital aperture slightly smaller than anal aperture (measured in lateral view). Typical anogenital setal formula 4-0-2-3, but asymmetry in number of genital (four versus three) and anal (two versus one) setae registered in some specimens. Genital (g1 11; g2–g4 7), anal (9–11), and adanal (11) setae simple, thin. All adanal setae inserted close and lateral to anal plate. Adanal lyrifissure located close and parallel to anterolateral margin of anal plate. One pair of adanal porose areas circular (4) or oval (7 × 4), located on weakly visible transverse carina.
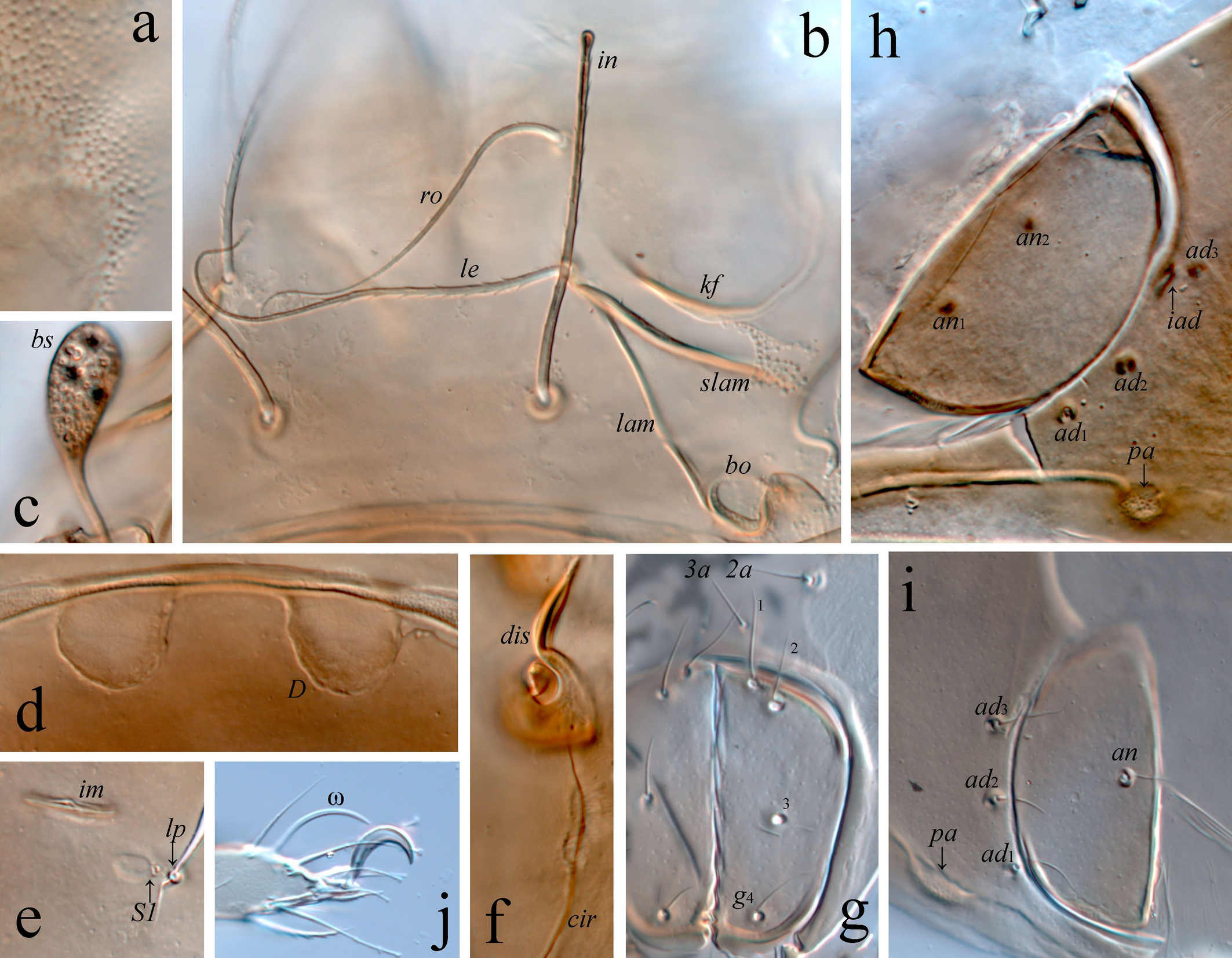


Legs (Figures 3a–d, 4j) – Claw of each tarsus strong, slightly barbed on dorsal side. Dorsoparaxial porose area on femora I–IV and on trochanters III, IV well visible. Tibia I and II with weak tubercle proximoventrally. Formulas of leg setation and solenidia: I (0-4-1-3-12) [1-1-2], II (0-4-1-3(or 4)-11) [1-1-1], III (1-2-0-3-9) [1-1-0], IV (0-2-1-3-8) [0-1-0]. Homology of setae and solenidia indicated in Table 1. Trochanters I, II, and IV without setae, trochanter III with one seta v′ (l′ absent). Femora I, II without seta l″, femur III without seta l′. Genua I and II with seta l′ (l″ and v′ absent), genua III and IV without seta l′. Tibia I without seta v″ and second solenidion. Tarsus I without setae it′, it″, pv′, pv″, pl′, and pl″, tarsus II without setae it′, it″, pv′, pv″, and second solenidion, tarsus III without setae it′, it″, a′, a″, pv′, and pv″, tarsus IV without setae a′, a″, pv′, and pv″. Seta s of tarsus I setiform, barbed (not eupathidial). Many setae of tarsi I–IV slightly swollen apically.
Download as Note: Tr, Fe, Ge, Ti, Ta = trochanter, femur, genu, tibia, and tarsus, respectively. Roman letters refer to normal setae, Greek letters refer to solenidia. Prime (ʹ) and double-prime (ʺ) indicate anterior and posterior setae of a pseudosymmetrical pair. Parentheses refer to both members of the pair collectively. *** = Seta v’’ of tibia II present or absent.
Leg
Tr
Fe
Ge
Ti
Ta
I
—
d, l’, bv’‘, v’’
l’,* *σ
(l),* v’*, φ
(ft),* (tc), (p), (u), (a), s, e*, ω1, ω2
II
—
d,* l’, bv’‘, v’’*
l’, σ
(l),* v’, v’’**, φ
(ft),* (tc), (p), (u), (a), s*, ω
III
v’
d, ev’
σ
l’,* (v)*, φ
(ft), (tc),* (p), (u), s*
IV
—
d,* ev’*
d
l’,* (v)*, φ
ft’’,* (tc), (p), (u), s*
Remarks
Khaustovella insolita n. sp. is most similar to Scheloribates (Scheloribates) fuscosensillus Corpuz-Raros, 1980 from the Philippines and Vietnam, S. (S.) tobagoensis Ermilov, 2018 from the Antillas, and S. (Perscheloribates) nodosus (Corpuz-Raros, 1980) from the Oriental region (all species are presented in the personal collection of the author) in having long, curved in distal half and slightly swollen apically rostral and lamella setae, long and distinctly swollen interlamellar seta, and medium-sized bothridial seta with large and broadly rounded head. Moreover, K. insolita n. sp., S. (S.) fuscosensillus, and S. (S.) tobagoensis with comparatively similar location of the adanal setae (close to anal aperture) and lack of the aggenital setae. However, the new species differs from S. (S.) fuscosensillus, S. (S.) tobagoensis, and S. (P.) nodosus by the presence (versus absence) of one pair of the adanal porose areas, striking reduction of the leg chaetome, and comparatively large genital aperture (distance between genital and anal is shortened), the epimeral formula (3-1-2-1 versus 3-1-3-3 in S. (S.) fuscosensillus and S. (P.) nodosus, 3-1-2-3 in S. (S.) tobagoensis), and the absence (versus presence) of the exobothridial seta, sublamellar porose area, and marginal porose area.
Etymology
The specific epithet insolita comes from the Latin word for ''unusual» and alludes to the unusual strong reduction (for Oripodoidea) of the leg setae.
Discussion
1. Parthenogenesis — A total of 27 adults of K. insolita n. sp. were collected from four tree species. The specimens varied greatly (for small species) in size (length 184–225), but all were the females, with a clearly visible ovipositor. Parthenogenesis or absence of males (in the presence of a large number of specimens) are indicated for some species of Oripodoidea (e.g., Protoribates spp., Rostrozetes spp.; see Maraun et al. 2022; Bomfim et al. 2024). Thus, the new species is probably parthenogenetic.
2. Morphology — Khaustovella n. gen. is characterized by two apomorphies. Firstly, one pair of adanal porose areas are present, located posterolateral to anal aperture. Porose areas on the ventral plate are known in Brachypylina. For example, they are developed in the anogenital region in males of Hellenamerus ionicus Mahunka, 1974 (Ameroidea) and some species of Machadobelba Balogh, 1958 (Oppioidea) (see Bernini and Arcidiacono 1985; Ermilov et al. 2025b). However, the porose areas are located anterolateral to anal aperture in these species. Also, possibly porose organs are present on the anal plates in males of Heterozetes aquaticus (Banks, 1895), and on the ventral plate in males of H. minnesotensis (Ewing, 1913) (Ceratozetoidea) (see Behan-Pelletier and Eamer 2003).
Secondly, the striking reduction of the leg setation and solenidia is present, e.g.: setal formula of trochanters, I to IV, 0-0-1-0; genua I and II with one seta; setal formula of tarsi, I to IV, 12-11-9-8, including simultaneous absence of setae it and pv on all tarsi; tarsus II and tibia I with one solenidion each. Usually, scheloribatid species have the following character states: setal formula of trochanters 1-1-2-1; genua I and II with two or three setae; setal formula of tarsi 18 to 20-15-15-12, including presence of setae it and pv; tarsus II and tibia I with two solenidion each. Some species of Scheloribatidae (for example, from the genera Ecuadoribates Ermilov and Kalúz, 2012 and Phylloribatula Balogh and Mahunka, 1978; see Ermilov and Friedrich 2016; Ermilov and Salavatulin 2023) and some species of the other families of Oripodoidea (see Behan-Pelletier et al. 2002; Ermilov et al. 2017, 2023; Ermilov and Salavatulin 2025; including partial leg setal comparisons in Behan-Pelletier et al. 2002 and Ermilov et al. 2017) can also have loss of setae and solenidia on the selective segments, however, setal formula of trochanters 0-0-1-0, the presence of only one seta (l′) on the genua I, II, and simultaneous absence of the setae it and pv on tarsi are unknown in Scheloribatidae and the related families.
Thus, the presence of the adanal porose areas in the posterolateral position to anal aperture as well as the striking reduction of the leg chaetome is unique to Khaustovella n. gen. among Oripodoidea.
Additionally, Khaustovella n. gen. is characterized by the absence of the sublamellar porose area and exobothridial seta, the presence of one pair of setae on the epimere IV and comparatively large genital opening with shortened distance between genital and anal apertures (equal to length of genital aperture or slightly shorter). Usually, scheloribatid species are characterized by the presence of the sublamellar porose area and exobothridial seta, the presence of three (or two) pairs of setae on the epimere IV, and comparatively small genital opening with large distance between genital and anal apertures. However, for example, among Scheloribatidae, some representatives of Monoschelobates Balogh and Mahunka, 1969 without sublamellar porose area (see Ermilov and OConnor 2021), and some representatives of Scheloribates Berlese, 1908 without exobothridial seta (see Ermilov and Martens 2014; Ermilov et al. 2021; Ermilov and Salavatulin 2023). Also, some species of Oribatulidae, Oripodidae, and Symbioribatidae with one pair of setae on the epimere IV (see Balogh and Mahunka 1978; Ermilov et al. 2023, 2025), and species of Campbellobates Wallwork, 1964 have comparatively large genital opening and shortened distance between genital and anal apertures (see Hammer 1967; Corpuz-Raros 2010).
Thus, the additional character states are not apomorphies, but their combination is unusual to Khaustovella n. gen. among Oripodoidea.
Acknowledgements
I thank Dr. V.M. Behan-Pelletier (Agriculture and Agri-Food Canada, Ottawa, Canada) for consultations, Vladimir M. Salavatulin (University of Tyumen, Tyumen, Russia) for oribatid mite sampling, Dr. Julia Baumann (University of Graz, Graz, Austria), two anonymous reviewers for their valuable comments, and the staff of the Cat Tien National Park for support during the field work. The work was performed within the framework of the Joint Russian-Vietnamese Biological Expedition, financially supported by the Russian Academy of Sciences. Collecting of materials was conducted under an Agreement on the scientific cooperation between Cat Tien National Park and the Joint Russian-Vietnamese Tropical Research and Technological Center.
References
- Balogh J. 1958. Oribatides nouvelles de l′Afrique tropicale. Rev. Zool. Bot. Afr. 58: 1-34.
- Balogh J., Mahunka S. 1969. The scientific results of the Hungarian soil zoological expeditions to South America. 10. Acari: Oribatids, collected by the second expedition. I. Acta Zool. Acad. Sci. Hung., 15: 1-21.
- Balogh J., Mahunka S. 1978. New data to the knowledge of the oribatid fauna of the Neogea (Acari). III. Acta Zool. Acad. Sci. Hung., 24: 269-299.
- Banks N. 1895. Some Acarians from a sphagnum swamp. J. N.Y. Ent. Soc., 3: 128-130.
- Behan-Pelletier V.M., Eamer B. 2003. Zetomimidae (Acari: Oribatida) of North America. In: Smith I.M. (Ed.). An Acarological Tribute to David R. Cook (from Yankee Springs to Wheeny Creek). Michigan: Indira Publishing House. pp. 21-56.
- Behan-Pelletier V.M., Clayton M., Humble L. 2002. Parapirnodus (Acari: Oribatida: Scheloribatidae) of canopy habitats in Western Canada. Acarologia, 42: 75-88.
- Bernini F., Arcidiacono R. 1985. Notulae oribatologicae XXXV. Some new records for the oribatid Italian fauna (Acarida, Oribatida) and the description of the male of Hellenamerus ionicus (Mahunka). Animalia, 12: 129-146.
- Bomfim G.S., Delabie J.H.C., Oliveira A.R. 2024. A new species of Rostrozetes (Acari: Oribatida: Haplozetidae) from Brazil. Zootaxa, 5556: 205-217. https://doi.org/10.11646/zootaxa.5556.1.15
- Corpuz-Raros L. 1980. Philippine Oribatei (Acarina) V. Scheloribates Berlese and related genera (Oribatulidae). Kalikasan, 9: 169-245.
- Corpuz-Raros L.A. 2010. Some mites of the family Oripodidae (Acari: Oribatida) from the Philippines. Philip. Ent., 24: 1-17. https://doi.org/10.59852/TPE-A577V24I1
- Ermilov S.G. 2018. New species of oribatid mites of the superfamily Oripodoidea Jacot 1925 (Acari, Oribatida) from Trinidad and Tobago. Zool. Zh., 97: 136-145 (In Russian) https://doi.org/10.7868/S0044513418020022 English version: 2018. Ent. Rev., 98: 245-254. https://doi.org/10.1134/S0013873818020161
- Ermilov S.G. 2023. New species of Tuberemaeus (Acari, Oribatida, Scheloribatidae) from trees in Southern Vietnam. Syst. Appl. Acarol., 28: 1344-1355. https://doi.org/10.11158/saa.28.8.6
- Ermilov S.G., Anichkin A.E. 2013. Collection of oribatid mites (Acari: Oribatida) from Dong Nai Biosphere Reserve of Southern Vietnam, with description of three new species. Ann. Zool., 63: 177-193. https://doi.org/10.3161/000345413X669513
- Ermilov S.G., Anichkin A.E. 2014. A new species of Scheloribates (Scheloribates) from Vietnam, with notes on taxonomic status of some taxa in Scheloribatidae (Acari, Oribatida). Int. J. Acarol., 40: 109-116. https://doi.org/10.1080/01647954.2014.885564
- Ermilov S.G., Friedrich S. 2016. New species of the genera Scheloribates and Perscheloribates (Acari, Oribatida, Scheloribatidae) from Amazonian Peru. Syst. Appl. Acarol., 21: 703-712. https://doi.org/10.11158/saa.21.6.1
- Ermilov S.G., Kalúz S. 2012. A new subgenus and three new species of oribatid mites of the family Scheloribatidae (Acari: Oribatida) from Ecuador. Ann. Zool. 62: 773-787. https://doi.org/10.3161/000345412X659795
- Ermilov S.G., Martens J. 2014. A new species of Perscheloribates (Acari, Oribatida, Scheloribatidae) from Nepal with a key to all species of the genus from the Oriental region. Acarina, 22: 14-19. https://doi.org/10.3897/zookeys.431.8120
- Ermilov S.G., OConnor B. 2021. New species of Monoschelobates Balogh et Mahunka 1969 and Multoribates Hammer 1961 (Acari, Oribatida, Scheloribatidae) phoretic on passalid beetles from the Neotropical region. Zool. Zh., 100: 618-626. https://doi.org/10.31857/S0044513421060040
- Ermilov S.G., Salavatulin V.M. 2023. Taxonomic contribution to the knowledge of the oribatid mite family Scheloribatidae (Acari, Oribatida), with description of three new species from Vietnam. Zootaxa, 5336: 233-246. https://doi.org/10.11646/zootaxa.5336.2.5
- Ermilov S.G., Salavatulin V.M. 2025. New faunistic data on arboreal oribatid mites (Acari: Oribatida) of Vietnam. Acarina, 33: 3-7. https://doi.org/10.21684/0132-8077-2025-33-1-3-7
- Ermilov S.G., Niedbała W., Anichkin A.E. 2012. Oribatid mites of Dong Nai Biosphere Reserve (=Cat Tien National Park) of Southern Vietnam, with description of a new species of Pergalumna (Acari, Oribatida, Galumnidae). Acarina, 20: 20-28. https://doi.org/10.31610/zsr/2011.20.2.200
- Ermilov S.G., Salavatulin V.M., Khaustov V.A. 2025a. Additions to the oribatid mite fauna of Cat Tien National Park, Vietnam, with description of a new species of Dolicheremaeus (Acari, Oribatida, Otocepheidae). Pers. J. Acarol., 14: 413-424. https://doi.org/10.22073/pja.v14i3.87212
- Ermilov S.G., Salavatulin V.M., Kolesnikov V.B. 2023. Contribution to knowledge of the oribatid mite genus Symbioribates (Acari, Oribatida, Symbioribatidae), with descriptions of two new arboreal species from Vietnam. Zootaxa, 5325: 556-570. https://doi.org/10.11646/zootaxa.5325.4.6
- Ermilov S.G., Hugo-Coetzee E.A., Theron P.D., Behan-Pelletier V.M. 2017. Contribution to the knowledge of the oribatid mite family Nesozetidae (Acari, Oribatida). Zootaxa, 4358: 311-327. https://doi.org/10.11646/zootaxa.4358.2.5
- Ermilov S.G., Subías L.S., Shtanchaeva U.Ya., Friedrich S. 2021. New sacculonotic Oripodoidea (Acari: Oribatida) from Peru. Zootaxa, 5048: 422-434. https://doi.org/10.11646/zootaxa.5048.3.7
- Ermilov S.G., Subías L.S., Shtanchaeva U.Ya., Kalúz S. 2025b. A new sexually dimorphic species of Machadobelba (Acari, Oribatida, Machadobelbidae) from Malaysia. Pers. J. Acarol., 14: 335-343. https://doi.org/10.22073/pja.v14i2.86956
- Ewing H.E. 1913. New Acarina. Part I. Bull. Am. Mus. Nat. Hist., 32: 93-121.
- Hammer M. 1967. Investigations on the oribatid fauna of New Zealand. Part II. Det Kong. Dansk. Vidensk. Selsk. Biol. Skr., 15: 1-60.
- Mahunka S. 1974. Neue und interessante Milben aus dem Genfer Museum. XII. Beitrag zur Kenntnis der Oribatiden-Fauna Griechenlands (Acari). Rev. suisse Zool., 81: 569-590. https://doi.org/10.5962/bhl.part.76024
- Maraun M., Bischof P.S.P., Klemp F.L., Pollack J., Raab L., Schmerbach J., Schaefer I., Scheu S., Caruso T. 2022. "Jack-of-all-trades» is parthenogenetic. Ecol. Evol., 12: e9036. https://doi.org/10.1002/ece3.9036
- Norton R.A. 1977. A review of F. Grandjean's system of leg chaetotaxy in the Oribatei (Acari) and its application to the family Damaeidae. In: Dindal D.L. (Ed.). Biology of Oribatid Mites. Syracuse: SUNY College of Environmental Science and Forestry. pp. 33-61.
- Norton R.A., Behan-Pelletier V.M. 2009. Suborder Oribatida. Chapter 15. In: Krantz G.W., Walter, D.E. (Eds). A Manual of Acarology. Lubbock : Texas Tech University Press. pp. 430-564.
- Salavatulin V.M. 2019. Microhabitat distribution of arboreal oribatid mites (Oribatida), associated with the Siberian pine (Pinus sibirica) of Western Siberia. Exp. Appl. Acarol., 78: 469-483. https://doi.org/10.1007/s10493-019-00401-4
- Salavatulin V.M., Ermilov S.G., Kudrin A.A., Nguyen T.D. 2022. Initial data on arboreal oribatid mites (Acari, Oribatida) from Vietnam. Acarina, 30: 103-108. https://doi.org/10.21684/0132-8077-2022-30-2-103-108
- Travé J., Vachon M. 1975. François Grandjean. 1882-1975 (Notice biographique et bibliographique). Acarologia, 17: 1-19.
- Wallwork J.A. 1964. Insects of Campbell island. Appendix. Campbellobates acanthus n. gen., n. sp. (Acari: Cryptostigmata). Pacific Ins. Monogr., 7: 601-606.
- Weigmann G. 2006. Hornmilben (Oribatida). Die Tierwelt Deutschlands. Teil 76. Keltern: Goecke and Evers. 520 pp.



2025-11-03
Date accepted:
2026-01-16
Date published:
2026-01-05
Edited by:
Baumann, Julia

This work is licensed under a Creative Commons Attribution 4.0 International License
2026 Ermilov, Sergey G.
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



