Is cattail pollen a sustainable long-term food source for the predatory mite, Euseius scutalis (Acari: Phytoseiidae)?
Gorji, Zahra  1
; Shishehbor, Parviz
1
; Shishehbor, Parviz  2
; Kocheili, Farhan
2
; Kocheili, Farhan  3
and Riahi, Elham
3
and Riahi, Elham  4
4
1Department of Plant Protection, Faculty of Agriculture, Shahid Chamran University of Ahvaz, Ahvaz, Iran.
2Department of Plant Protection, Faculty of Agriculture, Shahid Chamran University of Ahvaz, Ahvaz, Iran.
3Department of Plant Protection, Faculty of Agriculture, Shahid Chamran University of Ahvaz, Ahvaz, Iran.
4✉ Department of Entomology, Faculty of Agriculture, Tarbiat Modares University, P.O. Box 14115-336, Tehran, Iran.
2026 - Volume: 66 Issue: 1 pages: 69-80
https://doi.org/10.24349/bpkw-1i07Original research
Keywords
Abstract
Introduction
Phytoseiid mites are widely recognized as important predators of various agricultural pests, including spider mites, thrips, and whiteflies (Barbar et al. 2024). Studies indicate that some species can be economically produced and maintained at high densities, which is essential for effective biological control programs (Vangansbeke et al. 2024). Their significance in integrated pest management (IPM) lies in their ability to utilize alternative food sources, such as pollen, which facilitates their mass rearing and helps populations persist when prey is scarce, thereby enhancing their reliability and long-term effectiveness as biological control agents (Barbar et al. 2024). Integrating phytoseiid mites into IPM strategies improves overall pest management efficacy. This approach allows for a reduced reliance on chemical pesticides, promoting sustainable agricultural practices (Liburd et al. 2019).
The ability of phytoseiid mites to suppress pest populations can be significantly influenced by the type of food available to them during rearing and experimentation, as the nutritional quality and composition of the diet affect their development, reproduction, and predation efficiency (Nomikou et al. 2001; Goleva and Zebitz 2013; Shishehbor et al. 2022; Zergani et al. 2023). Pollen often serves as a crucial alternative food source, enabling these predatory mites to survive and reproduce even when their primary prey is scarce (Goleva and Zebitz 2013). The nutritional composition and value of pollen vary considerably among plant species, which in turn can greatly influence the development, reproduction, and overall performance of phytoseiid mites (Broufas and Koveos 2001; Goleva and Zebitz 2013; Shishehbor et al. 2022; Zergani et al. 2023). Therefore, understanding the nutritional requirements of specific phytoseiid mites and selecting appropriate pollen sources can optimize their effectiveness as biological control agents.
Euseius scutalis (Athias-Henriot, 1958) is a predatory mite belonging to the family Phytoseiidae. It is widely recognized for its role as a biological control agent, preying on various pest species, including spider mites and whiteflies (Swirski et al. 1967; Meyerdirk and Coudriet 1986; Nomikou et al. 2001). This broad diet contributes to its effectiveness in controlling multiple pest species across different crops. The efficacy of this predatory mite can vary depending on the region and crop type, and studies conducted across different geographical locations and crops have provided valuable insights into its adaptability and effectiveness (Bounfour and McMurtry 1987). The availability of alternative food sources, such as pollen, can enhance the establishment and efficacy of E. scutalis (Al-Azzazy and Alhewairini 2024). Pollen can serve as an adequate alternative food source, supporting the population of this predatory mite even in the absence of its primary prey (Nawar 2017; Shishehbor et al. 2022; Zergani et al. 2023; Al-Azzazy and Alhewairini 2024).
The success of mass-rearing programs for predatory mites largely depends on the availability of reliable and nutritionally adequate diets. Many rearing systems rely on a single, readily available food source, which may influence the physiological and reproductive traits of the predators over time. Pollen from cattails (Typha spp.), particularly Typha latifolia L., has been widely used as an alternative or supplemental food for generalist phytoseiid mites because it is rich in nutrients, easy to collect in bulk, and has good storage stability (Gravandian et al. 2022; Gorji et al. 2024). Several studies have demonstrated that cattail pollen supports development, survival, and fecundity of phytoseiid mites such as E. scutalis, Euseius concordis (Chant, 1959), Neoseiulus cucumeris (Oudemans, 1930), Amblyseius swirskii Athias-Henriot, 1962, Amblyseius largoensis (Muma, 1955), and Typhlodromus ornatus (Denmark and Muma, 1973) (Sarmento et al. 2011; Gravandian et al. 2022; Hadadi et al. 2022; Freitas et al. 2023; Gorji et al. 2024).
Despite these benefits, most previous studies have focused on short-term effects, and little is known about the potential long-term consequences of rearing predators on a single pollen type across multiple generations. Multi-generational studies are essential to uncover transgenerational effects and adaptive responses that may not be evident in single-generation experiments. Such studies also allow assessment of whether physiological or genetic constraints limit predator performance over time (Nemati and Riahi 2020; Yazdanpanah et al. 2021). Understanding these dynamics is crucial for improving the stability and efficiency of mass-rearing systems and ensuring the sustained quality of biological control agents. By investigating the long-term effects of cattail pollen on E. scutalis, an effective biological control agent in Iran, this study aims to provide insights into its suitability for prolonged laboratory rearing and its potential implications for integrated pest management programs.
Material and methods
Pollen collection
Cattail pollen was collected in July 2022 from Dorud, Lorestan Province, Western Iran. The pollen was oven-dried at 40 °C for 36 hours and then frozen at -20 °C for long-term storage.
Stock colony of predatory mite
The individuals in the initial E. scutalis colony were collected in February 2021 from marshmallow plants (Althea officinalis L.) infested with Tetranychus turkestani Ugarov and Nikolski, 1937 on the campus of the Faculty of Agriculture, Shahid Chamran University of Ahvaz, Ahvaz, Iran. The E. scutalis colony was reared on a green plastic sheet (6 × 6 × 0.1 cm) placed over a water-saturated sponge in a Petri dish (9 × 1 cm, diameter × height) filled with water. The edges of the green plastic sheet were covered with tissue paper immersed in water to provide necessary moisture and prevent the predators from escaping. Additionally, a few cotton threads were placed on the plastic sheets to provide oviposition sites and shelter for the predators. Before starting the experiments, the Euseius scutalis individuals were reared on date palm (Phoenix dactylifera L.) pollen for one month. The date palm pollen was collected from Magtu Village, Khuzestan Province, and frozen at -20 °C for long-term storage. Predators were maintained under laboratory conditions of 25 ± 1 °C, 60 ± 10% RH, and a photoperiod of 16:8 (L:D) throughout the experiments.
The experimental units were similar to the stock colony rearing units but smaller, consisting of green plastic sheets (3 × 3 × 0.1 cm) placed on water-saturated sponges inside Petri dishes. To provide oviposition sites and shelter, a few cotton fibers were placed on the plastic sheets. The edges of the sheets were covered with moist tissue paper to maintain humidity and prevent mite escape. Water was added daily to prevent drying.
To prevent overlapping of generations, approximately 200 mated females from the stock colony were transferred to new rearing units supplied with cattail pollen. After a 4‑day oviposition period, all adult females were removed, and only the eggs were allowed to develop to adulthood. Once the resulting females became gravid, they were transferred to new rearing units to initiate the next generation. This procedure was repeated for up to 50 generations to establish a long-term colony of E. scutalis maintained on cattail pollen, ensuring that only synchronous cohorts were preserved and that overlapping generations were avoided.
For each selected generation, eggs used in the biological and life table experiments were obtained from this maintained colony. To synchronize the age of eggs, approximately 150 adult E. scutalis females were randomly selected from the colony and transferred to new experimental units for oviposition. Females were allowed to lay eggs for a period of less than 24 hours, after which all adult females were removed. About 100 freshly laid eggs were individually transferred to new experimental units and reared on cattail pollen until adulthood. This process was repeated to establish up to 50 generations (designated G1, G5, G10, G15, G20, G25, G30, G40, and G50) reared exclusively on cattail pollen. For each generation, 100 eggs of the same age were randomly selected and individually transferred to the experimental units. The number of replicates per generation was 65, 69, 61, 56, 59, 59, 55, 53, and 51, respectively. Daily observations were made to record development duration, survival of immature stages, and adult longevity. Fresh pollen was added every two days, replacing the old to prevent fungal contamination. After adult emergence, males and females emerged in the same generation were paired together and monitored daily to record pre-oviposition period, oviposition rate, oviposition duration, fertility, and survival until death. Sex ratio estimation was performed by monitoring eggs laid daily during oviposition in generations G1, G10, G20, G30, G40, and G50 until the offspring reached adulthood. All experiments were conducted under controlled laboratory conditions at 25 ± 1 °C, 60 ± 10% relative humidity, and a photoperiod of 16:8 (light: dark) hours.
Statistical analysis
Raw data of E. scutalis were analyzed using the TWOSEX-MSChart software based on the age-stage, two-sex life table theory (Chi and Liu 1985; Chi 1988, 2023). All parameters, including age-stage-specific survival rate (sxj ), age-stage-specific fecundity (fxj ), age-specific survival rate (lx ), and age-specific fecundity (mx ), as well as population growth parameters for each treatment, were estimated (Chi 2023). Estimations of variances and standard errors for various stage durations, including the total pre-oviposition period (TPOP), adult pre-oviposition period (APOP), fecundity, oviposition days, and adult longevity, along with life table parameters such as the intrinsic rate of increase (r), finite rate of increase (λ), gross reproductive rate (GRR), net reproductive rate (R0), and mean generation time (T) (Chi 2023) were derived using a bootstrap process with 100,000 samples (Huang and Chi 2012). The mean of all parameters was compared across different generations using the paired bootstrap test.
Results
Development time and adult longevity showed variability among generations (Table 1). The pre-adult duration for both female and male mites was shortest in generation 30 (G30). The female developmental time ranged from 4.09 days in G30 to 5.63 days G40, while it varied from 4.17 days in G30 to 5.55 days in G50 for males. As the number of generations of E. scutalis reared on cattail pollen increased, the lifespan of female mites showed some fluctuations (Table 1). The adult longevity of E. scutalis females varied slightly among generations, with the highest value recorded in G1 (Table 1). However, these differences were not consistent or biologically meaningful, suggesting that prolonged rearing on cattail pollen did not substantially affect adult lifespan. The mean adult longevity of males followed a similar trend, showing only minor, statistically insignificant fluctuations across generations. In both femaes and males, total lifespan did not differ significantly among generations, reflecting overall developmental stability.
Download as The SEs were estimated using the bootstrap method with 100,000 samples. Mean values followed by different letters in the same row are significantly different (P < 0.05, paired bootstrap test).
Parameter
G1
G5
G10
G15
G20
G25
G30
G40
G50
Female
Egg
1.83 ± 0.07 abc
1.98 ± 0.04 a
1.77 ± 0.08 bc
1.49 ± 0.08 a
1.54 ± 0.08 d
1.68 ± 0.08 cd
1.00 ± 0.00 e
1.86 ± 0.06 abc
1.9 ± 0.06 ab
Larva
1.03 ± 0.03 a
1.04 ± 0.03 a
1.03 ± 0.03 a
1.00 ± 0.00 a
1.00 ± 0.00 a
1.00 ± 0.00 a
1.00 ± 0.00 a
1.00 ± 0.00 a
1.10 ± 0.06 a
Protonymph
1.1 ± 0.06 cdef
1.02 ± 0.02 ef
1.4 ± 0.09 a
1. 3 ± 0.08 ab
1. 1 ± 0.05 de
1.12 ± 0.06 bcde
1.00 ± 0.00 f
1.2 ± 0.07 abcd
1.3 ± 0.09 abc
Deutonymph
1.21 ± 0.08 bcde
1.10 ± 0.04 cde
1.4 ± 0.1 ab
1.05 ± 0.04 e
1.44 ± 0.09 a
1.32 ± 0.12 abcd
1.09 ± 0.05 de
1.51 ± 0.16 ab
1.3 ± 0.1 abc
Pre- adult
5.17 ± 0.1 b
5.15 ± 0.07 b
5.6 ± 0.1 a
4.84 ± 0.07 c
5.08 ± 0.07 b
5.12 ± 0.14 bc
4.09 ± 0.05 d
5.63 ± 0.15 a
5.6 ± 0.1 a
Adult longevity
17.97 ± 1.39 a
13.62 ± 0.75 be
14.77 ± 1.62 abce
13.76 ± 0.94 bce
10.95 ± 1.13 cde
10.91 ± 1.31 e
11.94 ± 1.37 bcde
14.23 ± 1.28 bce
10.93 ± 0.77 de
Total life span
23.14 ± 1.41 a
18.77 ± 0.76 a
20.37 ± 1.63 ab
18.59 ± 0.96 a
16.03 ± 1.11 a
16.03 ± 1.3 a
16.03 ± 1.36 a
19.86 ± 1.28 ab
16.53 ± 0.78 b
Male
Egg
1.84 ± 0.07 b
1.95 ± 0.09 ab
1.79 ± 0.08 b
1.5 ± 0.12 c
1.50 ± 0.11 c
1.8 ± 0.09 b
1.00 ± 0.00 d
2.00 ± 0.00 a
1.85 ± 0.08 ab
Larva
1.00 ± 0.00 a
1.10 ± 0.07 a
1.17 ± 0.1 a
1.00 ± 0.00 a
1.00 ± 0.00 a
1.00 ± 0.00 a
1.00 ± 0.00 a
1.06 ± 0.03 a
1.05 ± 0.05 a
Protonymph
1.06 ± 0.04 bc
1.10 ± 0.07 bc
1.17 ± 0.07 b
1.28 ± 0.11 ab
1.05 ± 0.05 bc
1.15 ± 0.08 bc
1.00 ± 0.00 c
1.12 ± 0.08 bc
1.55 ± 0.17 a
Deutonymph
1.29 ± 0.09 a
1.00 ± 0.00 c
1.29 ± 0.09 a
1.06 ± 0.06 abc
1.2 ± 0.09 ab
1.25 ± 0.12 ab
1.09 ± 0.05 ab
1.29 ± 0.14 ab
1.1 ± 0.07 bc
Pre- adult
5.19 ± 0.1 ab
5.15 ± 0.11 bc
5.42 ± 0.12 ab
4.83 ± 0.12 cd
4.75 ± 0.1 d
5.2 ± 0.14 ab
4.17 ± 0.09 e
5.41 ± 0.15 ab
5.55 ± 0.17 a
Adult longevity
10.06 ± 1.15 abc
12.8 ± 0.83 a
9.12 ± 1.11 c
10.78 ± 1.06 abc
9.6 ± 1.05 bc
10.15 ± 1.34 abc
12.44 ± 1.05 ab
10.94 ± 1.9 abc
11.15 ± 0.84 abc
Total life span
15.26 ± 1.12 ab
17.95 ± 0.83 a
14.54 ± 1.09 b
15.61 ± 1.05 ab
14.35 ± 1.06 b
15.35 ± 1.33 ab
16.61 ± 1.07 ab
16.35 ± 1.86 ab
16.7 ± 0.89 ab
Significant differences in pre-oviposition period (APOP), total pre-oviposition period (TPOP), oviposition days, fecundity, and sex ratio were observed among generations (Table 2). The APOP ranged from 1.15 days in G5 (shortest) to 3.07 days in G10 (longest) (Table 2). However, no clear increasing or decreasing trend was evident over the generations. The total pre-oviposition period (TPOP) fluctuated among generations, with the highest value recorded in G10 and the lowest in G30. Although some generations differed significantly from others, the variations did not follow a consistent pattern with the duration of rearing on cattail pollen. The duration of oviposition showed some fluctuations across generations, with a slight decreasing trend observed, although these differences were not statistically significant (Table 2). The highest number of eggs was observed in G5, which did not differ significantly from generations G1, G15, and G30. The overall pattern did not show a consistent trend across generations. The sex ratio varied slightly, being highest in G5 and lowest in C1, but the differences were not statistically significant among most generations (Table 2).
Download as Means within rows followed by different letters are significantly different (P < 0.05). Differences between parameters were examined using paired bootstrap test.
Parameter
G1
G5
G10
G15
G20
G25
G30
G40
G50
APOP (day)
2.04 ± 0.26 a
1.15 ± 0.05 d
3.07 ± 0.38 a
1.69 ± 0.18 ab
1.65 ± 0.16 ab
1.59 ± 0.12 b
1.49 ± 0.09 c
2.00 ± 0.21 a
1.82 ± 0.15 ab
TPOP (day)
7.21 ± 0.32 bc
6.29 ± 0.07 d
8.59 ± 0.44 a
6.53 ± 0.22 cd
6.65 ± 0.16 c
6.62 ± 0.17 cd
5.57 ± 0.08 e
7.64 ± 0.25 b
7.43 ± 0.2 b
Oviposition days (day)
12.14 ± 1.06 a
9.33 ± 0.75 b
7.78 ± 1.06 bc
8.67 ± 0.73 b
7.91 ± 1.04 bc
7.76 ± 1.01 bc
7.60 ± 1.03 bc
8.36 ± 0.80 b
5.89 ± 0.57 c
Fecundity (eggs/female)
18.31 ± 1.93 ab
21.33 ± 1.94 a
11.07 ± 1.91 cd
17.51 ± 1.64 ab
12. 9 ± 2.04 bcd
13.38 ± 2.08 bcd
16.66 ± 2.20 abc
15.51 ± 1.63 bc
10.63 ± 1.31 d
Sex ratio (Nf/N)
0.45 ± 0.06 c
0.70 ± 0.06 a
0.49 ± 0.06 bc
0.66 ± 0.06 ab
0.66 ± 0.06 ab
0.58 ± 0.06 abc
0.64 ± 0.06 ab
0.66 ± 0.06 ab
0.59 ± 0.07 abc
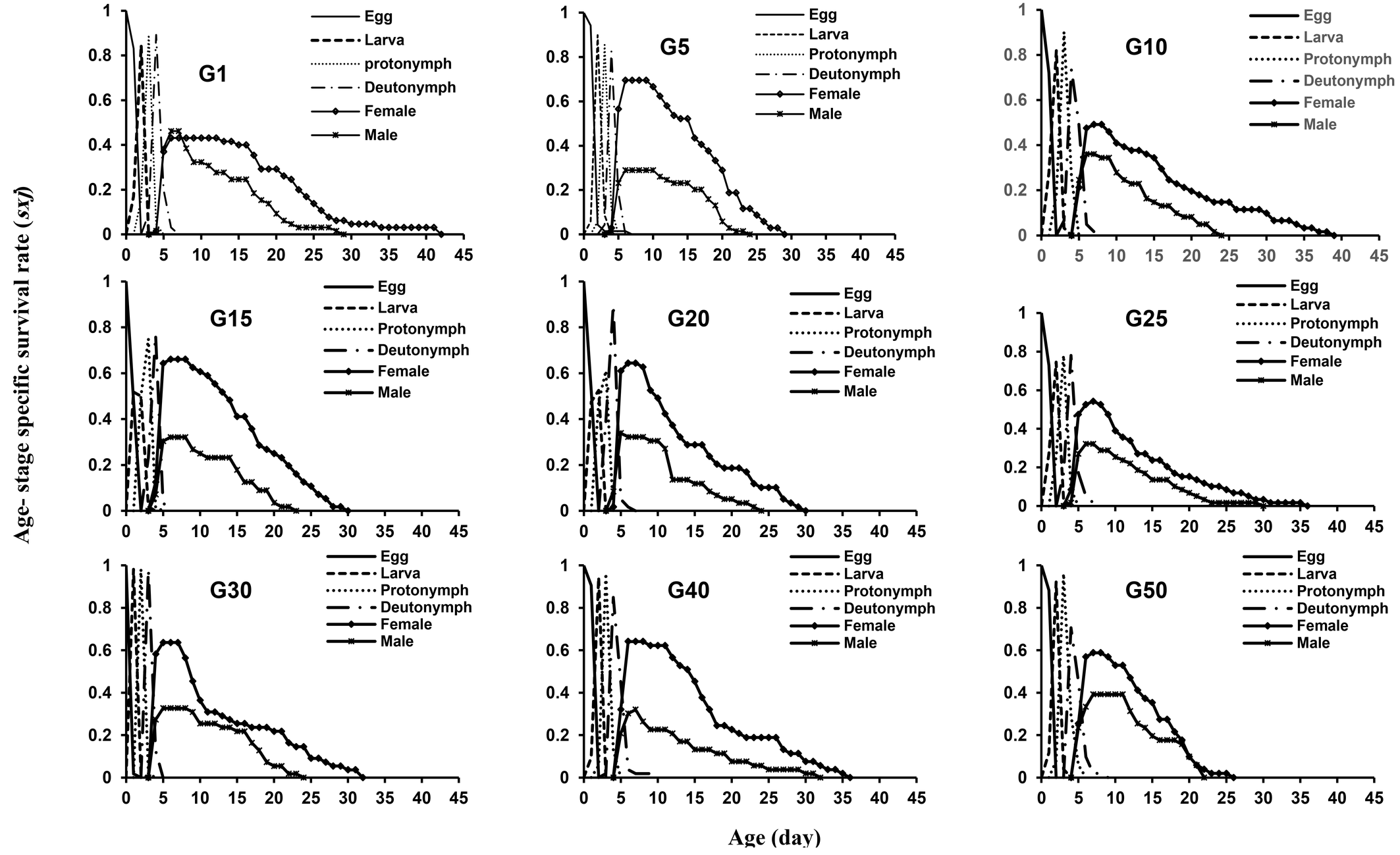
Figure 1 indicates the age-stage specific survival rate (sxj ) of E. scutalis during 50 generations reared on cattail pollen, displaying the probability that an individual predatory mite would survive to age x and stage j. There is an obvious overlap between stages, which contributes to the differences in developmental rates among individuals, as seen in these curves. The general pattern of survival is similar across generations, but slight differences can still be observed. In particular, individuals from the generations G5, G15, G20, G30, and G40 maintained slightly higher survival probabilities in the early adult stage compared with other generations. Although the overall shapes of the curves remain largely consistent.


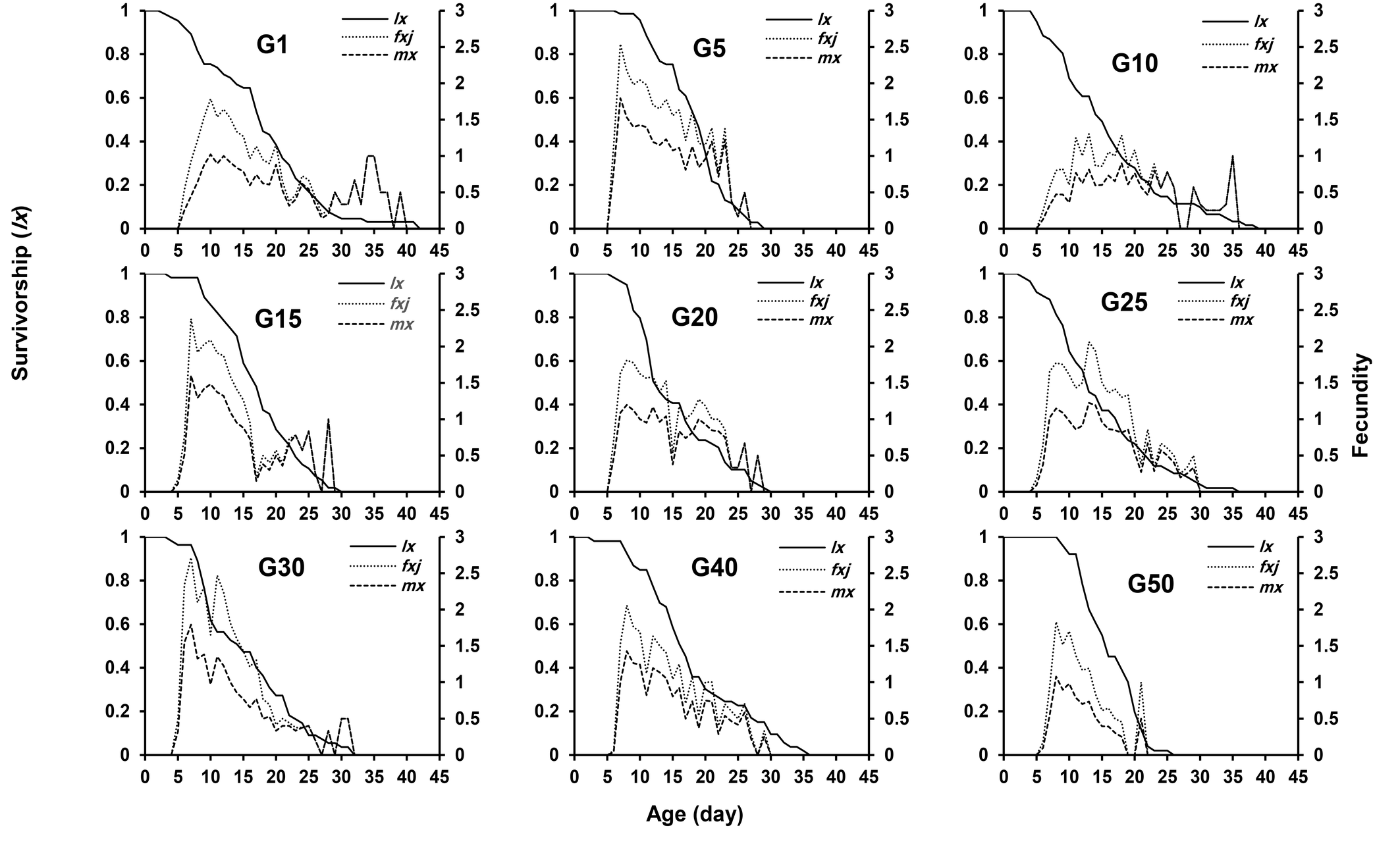
The age-specific survival (lx ) of E. scutalis on the first day of adult emergence in G1, G5, G10, G15, G20, G25, G30, G40, and G50 was 1.00, 0.92, 1.00, 0.98, 0.98, 0.92, 0.96, 0.98, and 1.00, respectively (Figure 2). The female age-specific fecundity (fx ) of this mite was highest on the 10th day (1.79 eggs/female/day), with additional peaks on the 7th day (2.54 eggs/female/day), 13th day (1.30 eggs/female/day), 7th day (2.38 eggs/female/day), 7th day (1.81 eggs/female/day), 13th day (2.06 eggs/female/day), 7th day (2.71 eggs/female/day), 8th day (2.06 eggs/female/day), and 8th day (1.83 eggs/female/day) in the aforementioned generations, respectively (Figure 2).


Life table parameters
The gross reproductive rate (GRR) of E. scutalis showed some variation across generations (Table 3). There was no significant difference between G1 and G5, then decreased slightly in G10, and remained stable from G15 to G40. However, a noticeable decline in GRR was observed in G50. The net reproductive rate (R₀) increased markedly up to G5 and after a decline in G10, remained statistically similar until G40, with a decrease observed in G50. The intrinsic rate of increase (r) and finite rate of increase (λ) followed a similar trend across generations. Both parameters increased from G1 to G5 and then statistically declined in G10. These parameters remained statistically unchanged up to G50.
Download as The SE were estimated using the bootstrap technique with 100,000 samples. Mean values followed by different letters in the same row are significantly different (P < 0.05), based on paired bootstrap test increase. GRR: gross reproductive rate, R0: net reproductive rate; r: intrinsic rate of increase λ: finite rate of increase, and T mean generation time.
Parameter
G1
G5
G10
G15
G20
G25
G30
G40
G50
GRR (eggs/individual)
19.86 ± 4.04 ab
22.03 ± 2.30 a
15.53 ± 2.34 b
18.54 ± 1.95 ab
17.94 ± 2.64 ab
17.23 ± 2.68 ab
16.76 ± 2.38 ab
16.76 ± 2.38 ab
8.18 ± 1.41 c
R0 (eggs/individual)
8.17 ± 1.40 bc
14.84 ± 1.78 a
5.44 ± 1.16 c
11.57 ± 1.54 ab
8.52 ± 1.55 bc
7.71 ± 1.46 bc
10.24 ± 1.47 b
10.24 ± 1.47 b
6.25 ± 1.05 c
r (day-1)
0.16 ± 0.01 cd
0.24 ± 0.01 a
0.12 ± 0.02 d
0.23 ± 0.01 ab
0.19 ± 0.02 bc
0.18 ± 0.02 c
0.20 ± 0.01 bc
0.20 ± 0.01 bc
0.17 ± 0.01 cd
λ (day-1)
1.17 ± 0.01 cd
1.27 ± 0.01 a
1.12 ± 0.02 d
1.25 ± 0.02 ab
1.21 ± 0.02 bc
1.20 ± 0.02 c
1.21 ± 0.01 bc
1.21 ± 0.01 bc
1.18 ± 0.02 cd
T (day)
13.01 ± 0.39 a
11.13 ± 0.23 bc
13.91 ± 0.73 a
10.79 ± 0.21 c
11.17 ± 0.30 bc
11.34 ± 0.34 bc
11.82 ± 0.34 b
11.82 ± 0.34 b
11.03 ± 0.28 bc
Offspring sex ratio
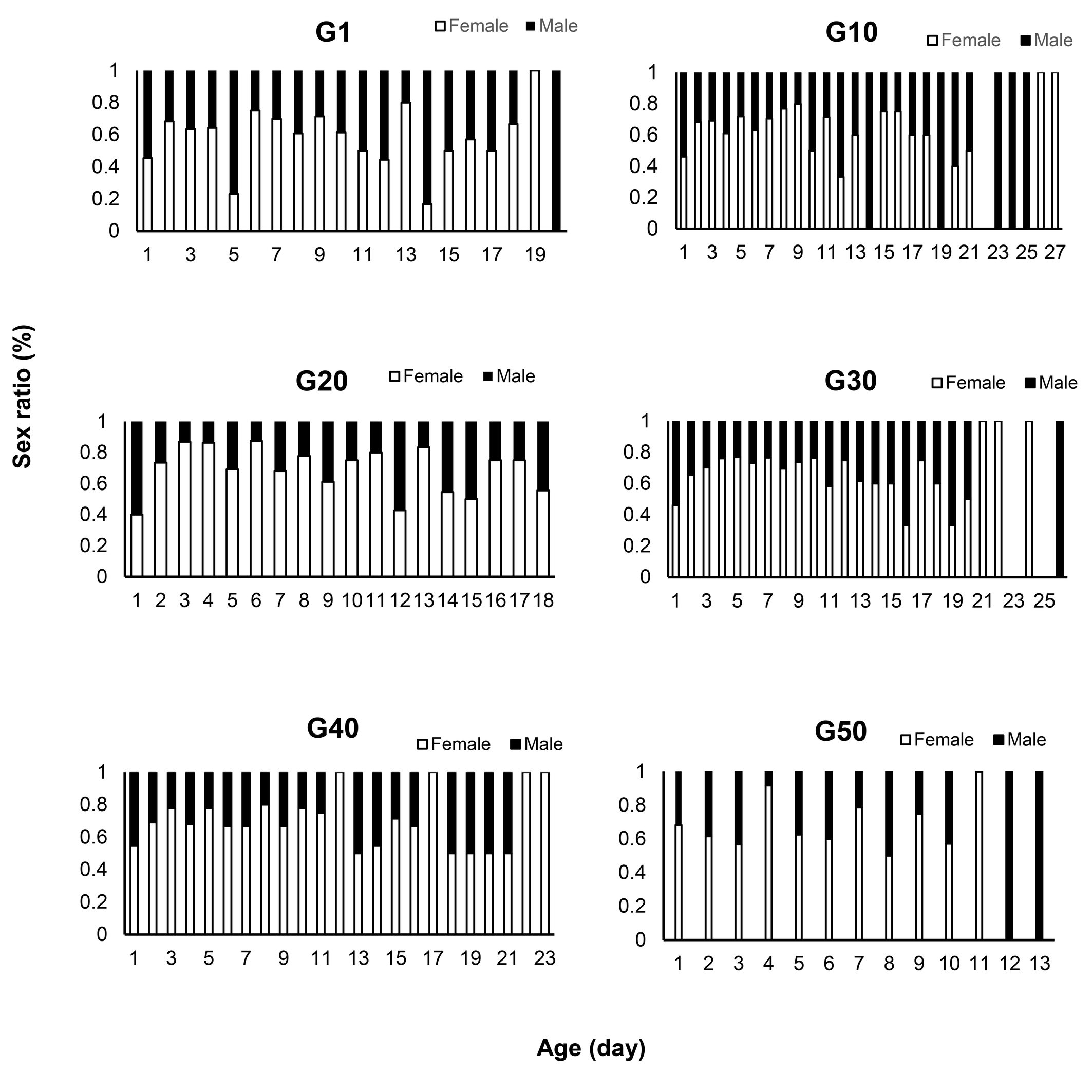
Figure 3 illustrates how the age of the female predator influences the sex ratio of their progeny. The sex ratio varied among generations and over the course of oviposition. In all generations, the sex ratio of the offspring was biased towards females during most of the egg-laying period. However, in the first, 30th and 50th generations, the sex ratio skewed towards males during the first and last days of oviposition. In the 40th generation, the sex ratio remained biased towards females throughout the entire egg-laying period.


Discussion
Euseius scutalis was able to develop and reproduce on cattail pollen for 50 generations, supporting most previous studies that indicated this pollen could sustain the development, survival, and reproduction of other phytoseiid mites, including A. swirskii and N. cucumeris (for up to 25 generations each) (Hadadi et al. 2022; Gravandian et al. 2022). The outcomes of this investigation indicate that long-term rearing of E. scutalis on cattail pollen significantly modifies its duration of development, reproduction, and life table characteristics. These results provide insights into the potential of E. scutalis to adjust its biological characteristics when maintained on a non-prey diet over several generations. Our findings indicate that while cattail pollen supports survival and reproduction, some generational trends suggest possible limitations in performance.
Pollen-feeding nutritional stress may have little apparent impact in the first generation but can influence offspring quality. Previous studies on phytoseiid mites have suggested that prolonged exposure to non-prey diets can reduce reproductive traits and offspring performance in subsequent generations (Nguyen et al. 2014; Nemati and Riahi 2020). Maternal malnutrition has also been associated with decreased egg viability, delayed development, or shortened longevity in these later generations. In our study, some variations were observed in the duration of immature stages among generations, but these differences did not follow a clear or consistent trend. This pattern suggests that prolonged feeding on cattail pollen did not systematically accelerate development. By contrast to our study, adaptation to non-prey food resulted in an initial increase in developmental efficiency, followed by a decline due to nutritional deficiencies or genetic bottlenecks (Yazdanpanah et al. 2021; Hadadi et al. 2022).
Reproductive traits, including the pre-oviposition period (APOP and TPOP), oviposition days, fecundity, and sex ratio, could vary between generations. Notably, despite slight variations, no significant differences were observed in APOP and TPOP between G1 and G50. This phenomenon may be due to a primary adaptation response, and the presence of such fluctuations could represent an initial maladaptive phase. The increased fecundity and longer oviposition days observed in earlier generations, followed by a decline in generations G40–G50, could reflect the influence of initial generational exposure to cattail pollen on reproductive performance. However, extended rearing on this single diet, as well as potential genetic factors such as inbreeding, may have contributed to the observed decline in reproductive output. However, the effects of diet and genetic background cannot be conclusively separated in this study.
Reproductive performance of E. scutalis, measured as lifetime fecundity, ranged from 11.07 to 21.33 eggs per female across generations. These values are comparable to previous findings on pollen diets, such as 18.31 eggs on cattail pollen (Bazazzadeh et al. 2025), 17.20 on hopbush pollen, 12.64 on castor bean pollen, and 17.39 on date palm pollen (Zergani et al. 2023). However, higher fecundity is often reported on prey-based diets, including 31.14 eggs on T. turkestani (Zergani et al. 2023), 29.68 on Tetranychus urticae Koch, 1836 (Osman et al. 2013), 37.9 on Pectinophora gossypiella (Saunders, 1844), and 14.2 on T. urticae (Salwa et al. 2017). These variations reflect differences in food quality, prey availability, and strain-specific performance, and they indicate that pollen diets generally support moderate reproduction compared with high-quality prey.
Intrinsic rate of increase is a crucial parameter in biological control. A one-generation test can show whether mites are able to survive and reproduce on certain diet, but multi-generation assessments demonstrate whether the population would increase or persist over the long term. If every subsequent generation has decreased survival and growth rate, pollens as a long-term food source would not be feasible. Population growth parameters of E. scutalis were almost constant from the initial generation to the 20th generation, then decreased and remained constant until the 50th generation. The r and λ parameters first increased slightly and then plateaued after a decline. This pattern aligns with observations in other phytoseiid mites, where optimal reproduction occured during early generations but declined in later generations due to nutrient limitations or genetic drift (Nemati and Riahi, 2020; Yazdanpanah et al., 2022).
The intrinsic rate of increase of E. scutalis in our study ranged from 0.16 to 0.24 across different generations. These values are within the range previously reported for this species on various diets. For instance, Bazazzadeh et al. (2025) found an r value of 0.16 on cattail pollen, while Bonfour and McMurtry (1987) reported an r of 0.188 for a Moroccan strain fed on red ice plant pollen. Other studies have documented r values of 0.1675 on hopbush pollen, 0.1692 on castor bean pollen, and 0.1996 on date palm pollen (Zergani et al. 2023). Higher rates (0.234 day⁻¹) were observed in a Turkish strain feeding on Panonychus ulmi (Koch, 1836) (Kasap and Şkeroğlu 2004) and in laboratory studies by Fouly et al. (2013), who reported r values of 0.232, 0.191, and 0.175 when E. scutalis was provided with date palm pollen, T. urticae, and Bemisia tabaci (Gennadius, 1889), respectively. Even greater variation has been noted on alternative diets such as Ricinus communis pollen (0.3379), Aceria ficus (Cotte, 1920) (0.2180), and Rhyncaphytoptus ficifoliae (Nalepa, 1892) (0.2158) (Momen and Abdel-Khalek 2008, 2014). These contrasts likely reflect differences among E. scutalis strains, host or diet quality, and experimental conditions (temperature, humidity, photoperiod).
Differences in sex ratio among phytoseiid species are mainly related to species-specific traits, particularly in laboratory populations (Amano and Chant 1977). Factors such as mating behavior, diet, and environmental conditions can influence the sex ratio during oviposition (Amano and Chant 1977). In our study, females produced more males at the beginning and again toward the end of their reproductive period. Early in oviposition, higher male production may ensure enough males for mating, while later, reduced sperm reserves likely result in more unfertilized eggs, shifting the sex ratio toward males. Similar trends have been observed in Typhlodromus caudiglans (Schuster, 1959) (Putman 1962), T. occidentalis (Nesbitt, 1951) (Croft and McMurtry 1972), Amblyseius andersoni (Chant, 1957) (Amano and Chant 1978), E. scutalis (Gorji et al. 2024; Bazazzadeh et al. 2025), and Phytoseiulus persimilis Athias-henriot, 1957 (Schulten et al. 1978), although environmental and species-specific factors may influence the pattern.
It is important to note that the observed changes in population parameters over multiple generations may not be solely due to the single-pollen diet. Laboratory colonies maintained for many generations can experience genetic bottlenecks or inbreeding, which may negatively affect reproductive performance and developmental traits (Mackauer 1976; Carbonnelle et al. 2007; Tien et al. 2011). Incorporating newly field-collected individuals into laboratory colonies has been shown to mitigate these detrimental effects by increasing genetic diversity and maintaining colony vigor (Henter and Fenster 2003; Nunney 2003). Therefore, the decline in reproductive parameters observed in later generations could result from a combination of dietary limitations and long-term laboratory rearing effects, rather than the diet alone.
In conclusion, our findings suggest that cattail pollen can serve as a suitable diet for short-to-midium term rearing of E. scutalis, supporting development, survival, and reproduction across multiple generations. While early generations exhibited improved developmental and reproductive performance, later generations experienced a decline in these parameters, suggesting potential physiological constraints. Our results show that this pollen can support growth well up to generation 30, but for reproduction it can only have beneficial effects on the efficiency of this mite up to generation 15. Future studies should explore genetic and physiological mechanisms underlying these adaptations and investigate potential diet supplementation strategies to enhance population performance over multiple generations.
Acknowledgment
Financial support (Grant no. SCU.AP1403.400) provided by the research deputy of Shahid Chamran University of Ahvaz, Ahvaz, Iran is gratefully acknowledged.
References
- Al-Azzazy M.M., Alhewairini S.S. 2024. The potential of two phytoseiid mites as predators of the grape erineum mite, Colomerus vitis. Plants, 13: 1953. https://doi.org/10.3390/plants13141953
- Amano H., Chant D.A. 1978. Some factors affecting reproduction and sex ratios in two species of predacius mites, Phytoseiulus persimilis Athias-henriot and Amblyseius andersoni (Chant) (Acarina: Phytoseiidae). Canadian J. Zool., 56: 1593- 1607. https://doi.org/10.1139/z78-221
- Barbar Z., Skinner M., Parker B.L., Kreiter S. 2024. Species of Phytoseiidae (Acari: Mesostigmata) predators of thrips and whiteflies: a review. Acarologia, 64(3): 745-767. https://doi.org/10.24349/alf8-2ujo
- Bazazzadeh F., Shishehbor P., Gorji Z., Gravandian M., Esfandiari M., Riahi E. 2025. Effect of different constant temperatures on the biological characteristics and life table parameters of Euseius scutalis (Acari: Phytoseiidae) fed on cattail pollen. Acarologia, 65(1): 242-254. https://doi.org/10.24349/thvk-w602
- Bounfour M., McMurtry J.A. 1987. Biology and ecology of Euseius scutalis (Athias-Henriot) (Acarina: Phytoseiidae). Hilgardia, 55 (5): 1- 23. https://doi.org/10.3733/hilg.v55n05p023
- Broufas G.D., Koveos D.S. 2001. Development, survival and reproduction of Euseius finlandicus (Acari: Phytoseiidae) at different constant temperatures. Exp. Appl. Acarol., 25: 441- 460. https://doi.org/10.1023/A:1011801703707
- Carbonnelle S., Hance T., Migeon A., Baret P., Cros‐Arteil S., Navajas M. 2007. Microsatellite markers reveal spatial genetic structure of Tetranychus urticae (Acari: Tetranychidae). Exp. Appl. Acarol., 42: 129-138. https://doi.org/10.1007/s10493-007-9068-z
- Chi H. 1988. Life-table analysis incorporating both sexes and variable development rates among individuals. Environ. Entomol., 17: 26-34. https://doi.org/10.1093/ee/17.1.26
- Chi H. 2023. CONSUME-MSChart: computer program for consumption rate analysis based on the age stage, two-sex life table.2019.02.01 ed. http:// 140. 120. 197. 173/ Ecology/ prod02. Htm. Accessed 19 Feb 2023
- Chi H., Liu H. 1985. Two new methods for the study of insect population ecology. Bull. Inst. Zool. Acad. Sin., 24: 225-240.
- Croft B.A., McMurtry J.A. 1972. Comparative studies on four strains of Typhlodromus occidentalis Nesbitt (Acarina: Phytoseiidae). IV. L.ife history studies. Acarologia, 13: 460-470.
- Fouly A., Nassar O.A, Osman M. A. 2013. Biology and life tables of Euseius scutalis (A.-H.) reared on different kinds of food. J. Entomol., 10(4): 199-206. https://doi.org/10.3923/je.2013.199.206
- Freitas G.S.D., Jumbo L.O.V., Rêgo A.S., Galvão A.S., Do Amaral E.A., Teodoro A.V. 2023. Pollen as sole food source for two generalist predatory mite species (Acari: Phytoseiidae). Syst. Appl. Acarol., 28(10), 1569-1578. https://doi.org/10.11158/saa.28.10.1
- Goleva I., Zebitz C.P.W. 2013. Suitability of different pollen as alternative food for the predatory mite Amblyseius swirskii. Exp. Appl. Acarol., 61: 259-283. https://doi.org/10.1007/s10493-013-9700-z
- Gorji Z., Shishehbor P., Kocheili F., Riahi E. 2024. Quality control of the predatory mite Euseius scutalis (Acari: Phytoseiidae) against Tetranychus turkestani (Acari: Tetranychidae) over 30 generations of rearing on cattail pollen. Acarologia, 64(3): 1-14. https://doi.org/10.24349/gz0z-vqgh
- Gravandian M., Fathipour Y., Hajiqanbar H., Riahi E., Riddick E.W. 2022. Long-term effects of cattail Typha latifolia pollen on development, reproduction, and predation capacity of Neoseiulus cucumeris, a predator of Tetranychus urticae. BioControl, 67: 149-160. https://doi.org/10.1007/s10526-021-10116-4
- Hadadi A., Fathipour Y., Hajiqanbar H., Riahi E. 2022. Long-term effects of cattail pollen on development, population growth potential, and predation capacity of Amblyseius swirskii (Acari: Phytoseiidae). Biocontrol Sci. Technol., 32: 1403-1416. https://doi.org/10.1080/09583157.2022.2134555
- Henter H.J., Fenster C.B. 2003. Potential for and limits to genetic rescue via immigration in an aphid parasitoid. J. Evolution Biol., 16: 124-132.
- Huang Y.B., Chi H. 2012. Age-stage, two-sex life tables of Bactrocera cucurbitae (Coquillett) (Diptera: Tephritidae) with a discussion on the problem of applying female age specific life tables to insect populations. Insect Sci., 19(2): 263-273. https://doi.org/10.1111/j.1744-7917.2011.01424.x
- Kasap İ., Şekeroğlu E. 2004. Life history of Euseius scutalis feeding on citrus red mite Panonychus citri at various temperatures. BioControl, 49: 645-654. https://doi.org/10.1023/B:BICO.0000046733.53887.2b
- Liburd O.E., Lopez L., Carrillo D., Revynthi A.M., Olaniyi O., Akyazi, R. 2019. Integrated pest management of mites. Burleigh Dodds Ser. Agric. Sci, 69, 841-886. https://doi.org/10.19103/AS.2019.0047.26
- Mackauer M. 1976. Genetic aspects of insect rearing. Entomophaga, 21: 45-51. https://doi.org/10.1146/annurev.en.21.010176.002101
- Meyerdirk D.E., Coudriet D.L. 1986. Evaluation of two biotypes of Euseius scutalis (Acarina: Phytoseiidae) as predators of Bemisia tabaci (Homoptera: Aleyrodidae). J. Econ. Entomol., 79 (3): 659-663. https://doi.org/10.1093/jee/79.3.659
- Momen F., Abdel-Khalek A. 2008. Influence of diet on biology and life-table parameters of the predacious mite Euseius scutalis (A.H.) (Acari: Phytoseiidae). Arch. Phytopathol. Plant Prot., 41(6), 418-430. https://doi.org/10.1080/03235400600813508
- Momen F., Abdel-Khalek A. 2014. Influence of diet on biology and life-table parameters of the predacious mite Euseius scutalis (A.H.) (Acari: Phytoseiidae). Arch. Phytopathol. Plant Prot.,41(6): 599-610. https://doi.org/10.1080/03235400600813508
- Nawar M.A. 2017. Biology and thermal requirments of Euseius scutalis (Athias-Henriot) fed on three pest prey types and pollen. Acarines, 11: 21-27. https://doi.org/10.21608/ajesa.2017.164164
- Nemati A., Riahi E. 2020. Does feeding on pollen grains affect the performance of Amblyseius swirskii (Acari: Phytoseiidae) during subsequent generations? Bull Entomol. Research, 110: 449-456. https://doi.org/10.1017/S0007485319000804 https://doi.org/10.1017/S0007485319000804
- Nguyen D.T., Vangansbeke D., De Clercq P. 2014. Artificial and factitious foods support the development and reproduction of the predatory mite Amblyseius swirskii. Exp. Appl. Acarol., 62(2): 181-194. https://doi.org/10.1007/s10493-013-9749-8
- Nomikou M., Janssen A., Schraag R., Sabelis M.W. 2001. Phytoseiid predators as potential biological control agents for Bemicia tabaci. Exp. Appl. Acarol., 25: 271-291. https://doi.org/10.1023/A:1017976725685
- Nunney L. 2003. The effects of population bottlenecks on inbreeding depression in the fruit fly Drosophila melanogaster. Genetics, 165: 1621-1628.
- Osman M.A., Afia Y.E., El‑Sawi S.A. 2013. Biology and life tables of Euseius scutalis (Acari: Phytoseiidae) reared on different diets. J. Entomol., 10(4): 199-206. https://doi.org/10.3923/je.2013.199.206
- Putman W.L. 1962. Life-history and behavior of the predacious mite Typhlodromus (T.) caudiglans (Acarina: Phytoseiidae) in Ontario, with notes on the prey of related species. Can. Entomol., 94: 163-177. https://doi.org/10.4039/Ent94163-2
- Salwa M. E., El- Shanawy Sholla Rania. M. Kandil Mervet A.A. 2017. Biological studies on the phytoseiid mite Euseius scutalis (Athias‑Henriot), reared on Pectinophora gossypiella (Saunders) eggs and Tetranychus urticae Koch in relation to prey biochemistry. Egypt. J. Biol. Pest Control, 27(2): 173-178.
- Sarmento R.A., Venzon M., Pallini A., Oliveira C.L., Souza O.F., Molina-Rugama, A.J. 2011. Predation, development and reproduction of Euseius concordis (Acari: Phytoseiidae) fed with cattail pollen (Typha angustifolia). Exp. Applied Acarol., 55: 63-72.
- Schulten G.G.M., Vanarendonk R.C.M., Russell V.M., Roorda F.A. 1978. Copulation, egg production and sex ratio in Phytoseulus persimilis and Amblyseius bibens (Acari: Phytoseiidae). Entomol. Exp. Appl. Acarol., 24: 145-153. https://doi.org/10.1111/j.1570-7458.1978.tb02764.x
- Shishehbor P., Rahmani Piyani A., Riahi E. 2022. Effect of different pollen diets in comparison to a natural prey, Tetranychus turkestani (Acari: Tetranychidae), on development, survival, and reproduction of Euseius scutalis (Acari: Phytoseiidae). Syst. Appl. Acarol., 23 (10): 211- 2122. https://doi.org/10.11158/saa.27.10.19
- Swirski E., Amitai S., Dorzia N. 1967. Laboratory studies on the feeding, development and oviposition of the predacious mite Amblyseius rubini Swirski and Amitai and Amblyseius swirski Athias-Henriot (Acari: Phytoseiidae) on various kinds of food substances. Isr. J. Agric. Res., 17: 101-119.
- Tien N. S. H., Massourakis D. T., Sabelis M. W. 2011. Avoidance of mating among relatives in the two-spotted spider mite Tetranychus urticae. Exp. Appl. Acarol., 54: 27-38. https://doi.org/10.1007/s10493-011-9431-y
- Vangansbeke D., Duarte M.V.A., Merckx J., Benavente A., Stevens I., Debie H., Viaene N., Guilbaud M., Pijnakker J., Steel H., Wäckers F. 2024. The potential application of Czenspinskia transversostriata in biological control. Exp. Appl. Acarol., 93(3): 549-562. doi: 10.1007/s10493-024-00945-0. https://doi.org/10.1007/s10493-024-00945-0
- Yazdanpanah S., Fathipour Y., Riahi E., Zalucki M.P. 2022. Pollen alone or a mixture of pollen types? Assessing their suitability for mass rearing of Neoseiulus cucumeris (Acari: Phytoseiidae) over 20 generations. J. Insect Sci., 22 (4): 6. https://doi.org/10.1093/jisesa/ieac043
- Yazdanpanah S., Fathipour Y., Riahi E., Zalucki M.P. 2021. Mass production of Neoseiulus cucumeris (Acari: Phytoseiidae): an assessment of 50 generations reared on almond pollen. J. Econ. Entomol., 114(6): 2021, 2255-2263. https://doi.org/10.1093/jee/toab163
- Zergani A., Shishehbor P., Naser Nakkai F., Riahi E. 2023. Life history traits and population parameters of the predatory mite Euseius scutalis (Acari: Phytoseiidae) fed on Tetranychus turkestani (Acari: Tetranychidae) and pollen from three different plants. Acarologia, 63(3): 945-954. https://doi.org/10.24349/mrqf-arrz



2025-06-29
Date accepted:
2025-12-21
Date published:
2026-02-05
Edited by:
Tsolakis, Haralabos

This work is licensed under a Creative Commons Attribution 4.0 International License
2026 Gorji, Zahra; Shishehbor, Parviz; Kocheili, Farhan and Riahi, Elham
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



