Phytoseiidae mites (Acari: Mesostigmata) in pastures from Goiás state, Brazil
Falchi, Vanessa L.  1
; Ribeiro, Lana Letícia C.
1
; Ribeiro, Lana Letícia C.  2
; Demite, Peterson R.
2
; Demite, Peterson R.  3
and Daud, Rodrigo D.
3
and Daud, Rodrigo D.  4
4
1Programa de Pós-Graduação em Biodiversidade Animal, Universidade Federal Goiás (UFG), 74690-900, Goiânia, Goiás, Brazil & TEIA - Laboratório de Taxonomia, Ecologia e Interações de Aracnídeos, Departamento de Ecologia, Instituto de Ciências Biológicas, Universidade Federal de Goiás, UFG, Campus II, 74690-900, Goiânia, Goiás, Brazil.
2Programa de Pós-Graduação em Biodiversidade Animal, Universidade Federal Goiás (UFG), 74690-900, Goiânia, Goiás, Brazil & TEIA - Laboratório de Taxonomia, Ecologia e Interações de Aracnídeos, Departamento de Ecologia, Instituto de Ciências Biológicas, Universidade Federal de Goiás, UFG, Campus II, 74690-900, Goiânia, Goiás, Brazil.
3Instituto Federal de Educação, Ciência e Tecnologia de Roraima, Campus Bonfim, Bonfim, Roraima, Brazil.
4Programa de Pós-Graduação em Biodiversidade Animal, Universidade Federal Goiás (UFG), 74690-900, Goiânia, Goiás, Brazil & TEIA - Laboratório de Taxonomia, Ecologia e Interações de Aracnídeos, Departamento de Ecologia, Instituto de Ciências Biológicas, Universidade Federal de Goiás, UFG, Campus II, 74690-900, Goiânia, Goiás, Brazil.
2025 - Volume: 65 Issue: 4 pages: 1158-1172
https://doi.org/10.24349/dxmj-hesjOriginal research
Keywords
Abstract
Introduction
Phytoseiidae includes more than 2,700 valid species with 257 species recorded in Brazil (Demite et al. 2025). These mites occur primarily on plants (Schmidt 2013), but some species have also been found in soil samples (Moraes et al. 2024). Phytoseiids are natural predators of several phytophagous arthropod species and nematodes. Some species are also used in biological control programs to manage mite and insect pests on crops (McMurtry et al. 2013; Moraes et al. 2024). Because of these attributes, these mites are widely studied in several crops (Döker et al. 2023; Hyte et al. 2023; Möth et al. 2023).
Approximately 40.3% of the territory of the state of Goiás, Brazil, is represented by pastures used for livestock production (LAPIG 2023). Poaceae species are used for fodder and soil cover and are typically adopted by producers due to their high productivity (Shavanov 2021). Despite their ecological and agricultural relevance, studies on mites in Brazilian pastures remain scarce, particularly in the Cerrado biome (Flechtmann 2000, 2004; Flechtmann and Navia 2007; Lofego et al. 2009). Flechtmann (2000) described two new species, Aceria aristidae Flechtmann and Aceria zoostrix Flechtmann (Eriophyidae), on Aristida (Poaceae) in São Paulo. Flechtmann (2004) recorded 10 species of phytophagous mites occurring on Urochloa ruziziensis (R. Germ and C.M. Evrard) Crins (Poaceae) in the state of São Paulo, including description of two new species, namely Catarhinus brachiariae (Diptilomiopidae) Flechtmann and Eotetranychus herbicolus Flechtmann (Tetranychidae). Flechtmann and Navia (2007) reported the first occurrence of Tetranychus (Armenychus) graminae Flechtmann (Tetranychidae) in central Brazil, causing an elephant grass infestation. Furthermore, Lofego et al. (2009) recorded 21 phytoseiid mite species in Brazilian pastures, with the description of a new species, Proprioseiopsis biologicus Lofego, Demite and Moraes.
Understanding the biodiversity of agricultural systems is essential for elucidating the ecological interactions between mites and their host plants. Accordingly, our goal was to provide the first comprehensive survey of Phytoseiidae mites in cultivated pastures across Goiás State, documenting their diversity, abundance, and associations with their host grasses.
Material and methods
Study area
Phytoseiidae mites were sampled during the rainy season in pastures between November and December 2022 in 15 livestock farms from Brazabrantes, Caturaí, Inhumas, Itaberaí, Itauçu and Nova Veneza municipalities, Goiás State, Brazil. Between January and May of 2023, all the same farms were sampled once more, except for the Itauçu 1 (Figure 1, Table 1).
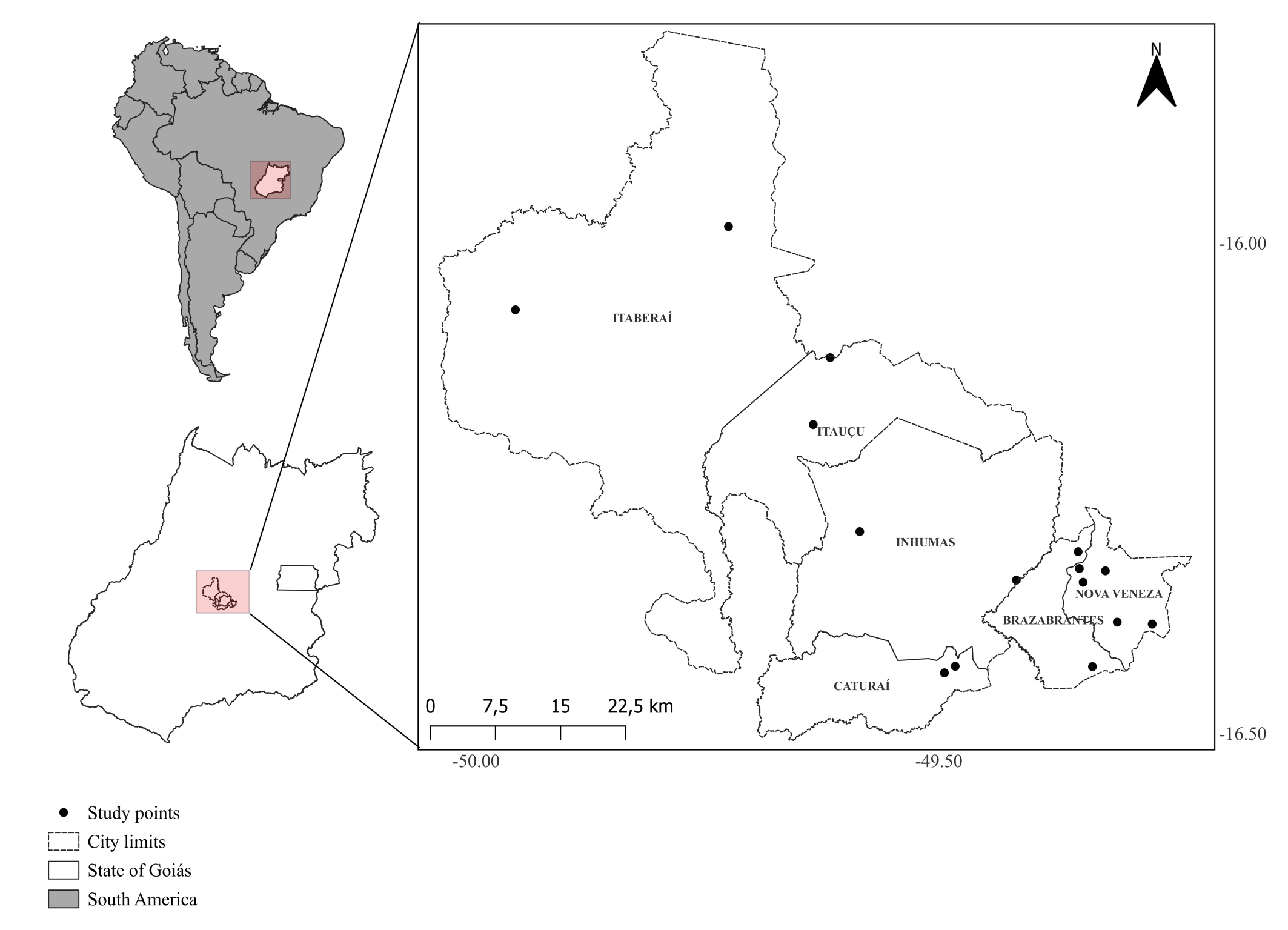


All farms sampled were in the Cerrado domain and grew pastures of the species Cynodon nlemfuensis Vanderyst, Urochloa arrecta (Hack. Ex T. Durand and Schinz) Morrone and Zuloaga, Urochloa brizantha (Hochst. ex A.Rich.) R.D. Webster, Urochloa decumbes (Stapf) R.D.Webster, Urochloa humidicola (Rendle) Morrone and Zuloaga, Urochloa lorentziana (Mez) Morrone and Zuloaga, Urochloa plantaginea (Link) R.D. Webster, and Urochloa ruziziensis (R. Germ. and C.M. Evrard) Crins (Poaceae) for livestock production.
Download as
Locality
Geographical coordinates
Nova Veneza 1
16°20’50.03”S, 49°20’43.87”W
Nova Veneza 2
16°19’59.92”S, 49°20’58.99”W
Nova Veneza 3
16°23’21.55”S, 49°16’13.98”W
Nova Veneza 4
16°20’7.40”S, 49°19’17.40”W
Itauçu 1
16°11’18.56”S, 49°38’16.62”W
Itauçu 2
16°7’12.72”S, 49°37’12.58”W
Caturaí 1
16°26’26.66”S, 49°29’40.34”W
Itaberaí 1
15°59’12.91”S, 49°43’50.48”W
Itaberaí 2
16°4’23.77”S, 49°57’33.50”W
Nova Veneza 5
16°23’15.44”S, 49°18’30.00”W
Brazabrantes 1
16°25’59.52”S, 49°20’4.78”W
Caturaí 2
16°26’2.36”S, 49°28’58.51”W
Inhumas 1
16°17’50.28”S, 49°35’13.20”W
Inhumas 2
16°20’44.16”S, 49°25’3.50”W
Brazabrantes 2
16°18’57.89”S, 49°21’4.07”W
Mites collection
We randomly established five sampling plots on each livestock farm, totalizing 480 samples. Each plot was represented by a 1-square-meter area in the pasture where we collected grasses. At each sampling plot, we collected 2 L of grasses in a recipient, including all plant extensions such as stalks, nodes, internodes and leaves, except for rhizomes. Then, the sampled grasses were kept in individual paper bags (one bag per sampling plot) in a Styrofoam box with ice for transport. In the laboratory, the grasses were immersed in a 5 L container of 30% alcohol, which was shaken vigorously to release the Phytoseiidae mites from the plants. After this procedure, the grasses were removed, and the solution was transferred to a 25 µm mesh sieve. The mites that were trapped in the mesh were transferred to a pot containing 70% alcohol.
The samples were observed under stereomicroscope and all mites found were mounted on microscope slides using Hoyer's medium (Moraes et al. 2024). Phytoseiidae mites were counted and identified to the species level under a phase-contrast optical microscope using specialized dichotomous keys for genera and subgenera (Chant and McMurtry 2007) and works on descriptions, redescriptions, and revisions of Phytoseiidae (e.g., Barbosa and Demite 2023; Ferragut and Navia 2024; Lofego et al. 2024).
The list of mites collected in pastures from the state of Goiás, Brazil, includes the following information about the examined material: collection locality, grass species, month of sampling in Roman numerals, year in Arabic numerals, and the number of individuals of each sex (indicated by symbols ♀ and ♂). Additionally, the list provides previous records and observations. We extracted the previous records of Phytoseiidae species from the Phytoseiidae Database (Demite et al. 2025), available at: http://www.lea.esalq.usp.br/phytoseiidae ![]() .
.
Results
We sampled 21 species from 10 genera of Phytoseiidae in pasture crops in Goiás State, Brazil. Among these, seven genera belonged to Amblyseiinae, and three to Typhlodrominae (Table 2).
Download as *Poaceae species codes: Cynodon nlemfuensis (CNL), Urochloa arrecta (UAR), Urochloa brizantha (UBR), Urochloa decumbes (UDE), Urochloa humidicola (UHU), Urochloa plantaginea (UPL) and Urochloa ruziziensis (URU).
Phytoseiidae species
CNL*
UAR
UBR
UDE
UHU
UPL
URU
Amblyseiinae Muma
Amblyseius acalyphus Denmark and Muma
1
Amblyseius atibaiensis Mineiro, Lofego and Sato
27
4
Amblyseius compositus Denmark and Muma
1
Arrenoseius lofegoi Barbosa and Demite
1
Iphiseiodes zuluagai Denmark and Muma
1
Neoseiulus benjamini (Schicha)
1
57
1
2
1
Neoseiulus gracilis (Muma)
1
Neoseiulus paraibensis (Moraes and McMurtry)
1
5
Neoseiulus transversus Denmark and Muma
1
13
1
1
3
1
Proprioseiopsis biologicus Lofego, Demite ans Moraes
11
Proprioseiopsis mexicanus (Garman)
3
2
43
1
4
10
Proprioseiopsis neotropicus (Ehara)
2
Proprioseiopsis ovatus (Garman)
1
2
151
3
18
Proprioseiopsis pentagonalis (Moraes and Mesa)
8
3
Typhlodromalus aripo De Leon
2
Typhlodromalus clavicus Denmark and Muma
1
Typhlodromalus peregrinus Muma
2
1
Typhlodromips mangleae De Leon
5
Typhodrominae Wainstein
Metaseiulus (Metaseiulus) camelliae Chant and Yoshida-Shaul
1
Silvaseius barretoae Yoshida-Shaul and Chant
1
Typhlodromus (Anthoseius) transvaalensis (Nesbitt)
13
1
Among the 21 species recorded, 11 are new records for Goiás State, and two were collected for the first time since their original descriptions, namely, Amblyseius atibaiensis and Proprioseiopsis biologicus.
In total, we collected 416 adult phytoseiid mites that were identified at the species level. The most abundant species was Proprioseiopsis ovatus, followed by Neoseiulus benjamini and Proprioseiopsis mexicanus. Only one individual was sampled for Amblyseius acalyphus, Amblyseius compositus, Arrenoseius lofegoi, Iphiseiodes zuluagai, Neoseiulus gracilis, Metaseiulus (Metaseiulus) camelliae and Silvaseius barretoae. Regarding host plants, U. brizantha hosted the highest richness and abundance of Phytoseiidae mites, followed by U. plantaginea and U. decumbens (Table 2). No phytoseiid mites were recorded on U. lorentziana. The following is a list of Phytoseiidae species sampled in pastures from livestock farms in Goiás State, Brazil.
Phytoseiidae Berlese
Amblyseiinae Muma
Amblyseius Berlese
Amblyseius acalyphus Denmark & Muma
Amblyseius acalyphus Denmark & Muma 1973: 242.
Amblyseius acalyphus — Moraes et al. 1986: 6, 2004: 12; Lofego et al. 2024: 9.
Amblyseius (Amblyseius) acalyphus — Denmark & Muma 1989: 75.
Specimens examined — Itaberaí 1: Urochloa brizantha, IV-2023 (1 ♀).
Previous records — Brazil - Goiás, Mato Grosso, Mato Grosso do Sul, Minas Gerais, Roraima, São Paulo; Guadeloupe.
Observation — this species was previously recorded in the state of Goiás on Bauhinia ungulata L. (Leguminosae), Psidium guajava L. (Myrtaceae), Schinus terebinthifolius Raddi (Anacardiaceae), Brosimum gaudichaudii Trécul (Moraceae) and Protium spruceanum (Bentham) Engler (Burseraceae) (Demite et al. 2017).
Amblyseius atibaiensis Mineiro, Lofego & Sato
Amblyseius atibaiensis Mineiro, Lofego & Sato 2012: 596.
Amblyseius atibaiensis — Lofego et al. 2024: 15.
Specimens examined — Brazabrantes 2: Urochloa brizantha, IV-2023 (6 ♀, 1 ♂). Caturaí 1: Urochloa brizantha, IV-2023 (2 ♀, 1 ♂), V-2023 (2 ♀). Inhumas 2: Urochloa plantaginea, IV-2023 (4 ♀). Itaberaí 1: Urochloa brizantha, IV-2023 (1 ♀, 1 ♂). Nova Veneza 3: Urochloa brizantha, III-2023 (1 ♂). Nova Veneza 4: Urochloa brizantha, III-2023 (2 ♀), IV-2023 (9 ♀), V-2023 (1 ♀).
Previous records — Brazil - São Paulo.
Observation — Amblyseius atibaiensis was first described based on individuals sampled from the soil and litter of coffee crops in São Paulo State, Brazil (Mineiro et al. 2012). Here, A. atibaiensis is recorded for the first time after its original taxonomy description.
Amblyseius compositus Denmark & Muma
Amblyseius compositus Denmark & Muma 1973: 240.
Amblyseius compositus — Moraes et al., 1986: 11, 2004: 21; Lofego et al. 2024: 23.
Amblyseius (Multiseius) compositus — Denmark & Muma 1989: 95.
Specimens examined — Nova Veneza 4: Urochloa brizantha, II-2023 (1 ♀).
Previous records — Brazil - Amazonas, Bahia, Espírito Santo, Goiás, Minas Gerais, Paraná, Pernambuco, Piauí, Rio Grande do Sul, Sergipe, São Paulo, Tocantins.
Observation — this species was previously recorded in Goiás State on P. guajava, S. terebinthifolius, Virola sebifera Fusée-Aublet (Myristicaceae), Cecropia pachystachya Trécul (Urticaceae), Inga edulis von Martius (Leguminosae), Protium spruceanum, Rhamnaceae sp., Rudgea viburnoides, Tocoyena formosa Schumann (Rubiaceae), Trichilia martiana De Candolle (Meliaceae) and an unidentified Myrtaceae species (Demite et al. 2017).
Arrenoseius Wainstein
Arrenoseius lofegoi Barbosa & Demite
Arrenoseius lofegoi Barbosa & Demite 2023: 50.
Arrenoseius lofegoi — Lofego et al. 2024: 87.
Specimens examined — Nova Veneza 3: Urochloa brizantha, IV-2023 (1 ♀).
Previous records — Brazil - Bahia.
Observation — Arrenoseius lofegoi was found in sugarcane (Saccharum officinarum L., Poaceae) crops in the state of Bahia, Brazil (Barbosa and Demite 2023). Arrenoseius lofegoi was previously recorded in Goiás State by Rossetti et al. (2025)
Iphiseiodes De Leon
Iphiseiodes zuluagai Denmark & Muma
Iphiseiodes zuluagai Denmark & Muma 1972: 23.
Iphiseiodes zuluagai — Moraes et al. 1986: 61; 2004: 91; Lofego et al. 2024: 107.
Amblyseius zuluagai — Moraes & Mesa 1988: 79.
Specimens examined — Nova Veneza 4: Urochloa brizantha, II-2023 (1 ♀).
Previous records — Brazil - Acre, Alagoas, Amapá, Amazonas, Bahia, Ceará, Distrito Federal, Goiás, Maranhão, Mato Grosso, Mato Grosso do Sul, Minas Gerais, Pará, Paraná, Paraíba, Pernambuco, Rio de Janeiro, Rio Grande do Norte, Rio Grande do Sul, Rondônia, Roraima, Sergipe, São Paulo, Tocantins; Colombia; Cuba; Dominican Republic; Guadeloupe; Marie Galante; Martinique; Panama; Peru; Puerto Rico; Venezuela.
Observation — previous recorded in Goiás State on Campomanesia pubescens (von Martius ex De Candolle) O. Berg (Myrtaceae), Didymopanax vinosum (Cham and Schltdl) Marchal (Araliaceae), Harconia speciosa Gomes (Apocynaceae), Xylopia aromatica (Lamark) von Martius (Annonaceae), Byrsonima coccolobifolia Kunth (Malpighiaceae), Byrsonima intermedia de Jussieu (Malpighiaceae), Doliocarpus cf. dentatus (Fusée-Aublet) Standley (Dilleniaceae), Ouratea spectabilis von Martius Ex Engler (Ochnaceae), Annona crassiflora von Martius (Annonaceae), Kielmeyera cf. coriacea von Martius and Zucc (Calophyllaceae), Ouratea spectabilis (von Martius) Engler (Ochnaceae), Qualea grandiflora von Martius (Vochysiaceae), two unidentified species of the Myrtaceae (Rezende and Lofego 2011); Zeyheria montana von Martius (Bignoniaceae), Trichilia martiana De Candolle (Meliaceae), Roupala montana Fusée-Aublet (Proteaceae) (Demite et al. 2017), and Miconia albicans (Swartz) Steudel (Melastomataceae) (Moraes et al. 2022).
Neoseiulus Hughes
Neoseiulus benjamini (Schicha)
Amblyseius benjamini Schicha 1981: 203.
Amblyseius (Amblyseius) benjamini — Tseng 1983: 60.
Neoseiulus benjamini — Moraes et al. 1986: 72, 2004: 108; Lofego et al. 2024: 199.
Specimens examined — Brazabrantes 1: Urochloa brizantha, I-2023 (1 ♀); Cynodon nlemfuensis, IV-2023 (1 ♀). Brazabrantes 2: Urochloa brizantha, XI-2022 (1 ♂), I-2023 (1 ♂), III-2023 (1 ♀). Caturaí 1: Urochloa brizantha, XI-2022 (1 ♀), XII-2022 (1 ♂), II-2023 (1 ♀, 1 ♂). Caturaí 2: Urochloa brizantha, XI-2022 (1 ♀). Inhumas 1: Urochloa brizantha, XI-2022 (1 ♂), II-2023 (1 ♂), V-2023 (1 ♀). Inhumas 2: Urochloa plantaginea, III-2023 (1 ♀). Itauçu 2: Urochloa brizantha, XI-2022 (1 ♀). Nova Veneza 1: Urochloa brizantha, XI-2022 (3 ♀), I-2023 (1 ♀), III-2023 (1 ♀), IV-2023 (2 ♀, 1 ♂). Nova Veneza 2: Urochloa brizantha, XII-2022 (1 ♀, 1 ♂), I-2023 (1 ♀, 1 ♂). Nova Veneza 3: Urochloa brizantha, III-2023 (3 ♀), IV-2023 (3 ♀), V (1 ♀). Nova Veneza 4: Urochloa brizantha, XI-2022 (1 ♀), XII-2022 (3 ♀), III-2023 (6 ♀, 3 ♂), IV-2023 (4 ♀, 3 ♂), V-2023 (3 ♀). Nova Veneza 5: Urochloa brizantha, XII-2022 (1 ♀), V-2023 (1 ♀); Urochloa plantaginea, I-2023 (1 ♀). Itaberaí 1: Urochloa humidicola, XI (1 ♀); Urochloa ruziziensis, XI-2022 (1 ♀).
Previous records — Australia; Brazil - Bahia, Distrito Federal, Goiás, Mato Grosso do Sul, Minas Gerais, São Paulo, Tocantins; Chile; Martinique; South Africa.
Observation — previous recorded in Goiás on Trachypogon sp. (Poaceae) and Stryphnodendron adstringens (von Martius) Coville (Fabaceae) (Rezende and Lofego 2011).
Neoseiulus gracilis (Muma)
Cydnodromus gracilis Muma, 1962: 9.
Neoseiulus gracilis — Moraes et al. 1986: 81, 2004: 122; Lofego et al. 2024: 207.
Neoseiulus atrii (Karg) — (synonymy according to Moraes et al. 1997).
Specimens examined — Itauçu 2: Urochloa brizantha, XII-2022 (1 ♀).
Previous records — Brazil - Alagoas, Bahia, Paraíba, Pernambuco, Tocantins; Cuba; Saint Lucia, United States of America; Venezuela.
Observation — this is the first record of this species in Goiás State, Brazil.
Neoseiulus paraibensis (Moraes & McMurtry)
Amblyseius paraibensis Moraes & McMurtry 1983: 135.
Neoseiulus paraibensis — Moraes et al. 1986: 92, 2004: 137; Lofego et al. 2024: 217.
Specimens examined — Caturaí 2: Urochloa arrecta, XI-2022 (1 ♀); Nova Veneza 1: Urochloa brizantha, XI-2022 (1 ♀, 3 ♂). Nova Veneza 3: Urochloa brizantha, XI-2022 (1 ♂).
Previous records — Brazil - Paraná, Paraíba, Rio Grande do Sul, São Paulo; Colômbia; Costa Rica; Cuba; Martinique; Nicaragua; Panama; United States of America.
Observation — here, N. paraibensis is recorded for the first time in Goiás State, Brazil.
Neoseiulus transversus Denmark & Muma
Neoseiulus transversus Denmark & Muma 1973: 267.
Neoseiulus transversus — Moraes et al. 1986: 98, 2004: 148; Lofego et al. 2024: 221.
Specimens examined — Brazabrantes 1: Urochloa brizantha, XI-2022 (1 ♀, 1 ♂). Brazabrantes 2: Urochloa brizantha, XI-2022 (3 ♀), XII-2022 (1 ♂). Caturaí 1: Urochloa brizantha, I-2023 (1 ♀). Itaberaí 1: Urochloa ruziziensis, XII-2022 (1 ♀), Cynodon nlemfuensis, I-2023 (1 ♀). Itaberaí 2: Urochloa decumbens, XI-2022 (1 ♂); Urochloa humidicola, XI-2022 (1 ♀). Nova Veneza 3: Urochloa plantaginea, XI-2022 (1 ♂), I-2023 (1 ♀). Nova Veneza 4: Urochloa brizantha, XII-2022 (1 ♀), I-2023 (1 ♀). Nova Veneza 5: Urochloa brizantha, XI-2022 (1 ♀, 1 ♂), XII-2022 (1 ♀, 1 ♂).
Previous records — Argentina; Brazil - Bahia; Goiás; Maranhão; Minas Gerais; Paraíba; Pernambuco, Rio Grande do Sul, São Paulo.
Observation — Neoseiulus transversus has been previously recorded in Goiás State on soybean crops (Glycine max Merril, Fabaceae) (Cavalcante et al. 2017).
Proprioseiopsis Muma
Proprioseiopsis biologicus Lofego, Demite & Moraes
Proprioseiopsis biologicus Lofego, Demite & Moraes, in Lofego et al. (2009): 51.
Proprioseiopsis biologicus — Lofego et al. 2024: 113.
Specimens examined — Brazabrantes 2: Urochloa brizantha, V-2023 (9 ♀). Nova Veneza 1: Urochloa brizantha, XI-2022 (2 ♀).
Previous records — Brazil - São Paulo.
Observation — this species was formally described based on specimens collected from U. decumbens in São Paulo, Brazil (Lofego et al. 2009). This is the first report of P. biologicus since its original description.
Proprioseiopsis mexicanus (Garman)
Amblyseiopsis mexicanus Garman 1958: 75.
Typhlodromus (Amblyseius) mexicanus — Chant 1959: 92.
Amblyseiulus mexicanus — Muma 1961: 278.
Proprioseiopsis mexicanus — Moraes et al. 1986: 118, 2004: 181; Lofego et al. 2024: 117.
Typhlodromus mexicanus — Hirschmann 1962: 5.
Amblyseius mexicanus — Collyer 1964: 634.
Proprioseiopsis (Patinoseius) mexicanus — (synonymy according to Karg 1989).
Proprioseiopsis amotus (Zack) — (synonymy according to Denmark & Evans 2011).
Proprioseiopsis asetus (Chant) — (synonymy according to Denmark & Evans 2011).
Proprioseiopsis clausae (Muma) — (synonymy according to Denmark & Evans 2011).
Proprioseiopsis kogi (Chant and Hansell) — (synonymy according to Denmark & Evans 2011).
Proprioseiopsis putmani (Chant) — (synonymy according to Denmark & Evans 2011).
Proprioseiopsis temperellus (Denmark and Muma) — (synonymy according to Denmark & Evans 2011).
Proprioseiopsis tropicanus (Garman) — (synonymy according to Denmark & Evans 2011).
Proprioseiopsis tulearensis (Blommers) — (synonymy according to Denmark & Evans 2011).
Proprioseiopsis versutus (Zack) — (synonymy according to Denmark & Evans 2011).
Specimens examined — Brazabrantes 1: Urochloa brizantha, II-2023 (2 ♀); Cynodon nlemfuensis, XII-2022 (1 ♀), IV-2023 (1 ♀), V-2023 (1 ♀). Brazabrantes 2: Urochloa brizantha, XI-2022 (2 ♀). Caturaí 1: Urochloa arrecta, XII-2022 (1 ♀). Caturaí 2: Urochloa brizantha, III-2023 (2 ♀), IV-2023 (1 ♀), V-2023 (1 ♀); Urochloa arrecta, XI-2022 (1 ♀). Inhumas 1: Urochloa brizantha, XII-2022 (1 ♀), III-2023 (3 ♀). Inhumas 1: Urochloa brizantha, II-2023 (2 ♀), V-2023 (1 ♀). Inhumas 2: Urochloa brizantha, XII-2022 (3 ♀); Urochloa plantaginea, III-2023 (4 ♀), V-2023 (4 ♀). Itaberaí 1: Urochloa brizantha: II-2023 (1 ♀), V-2023 (4 ♀); Urochloa decumbens, I-2023 (1 ♀). Itaberaí 2: Urochloa humidicola, XII-2022 (1 ♀), I-2023 (1 ♀), III-2023 (1 ♀), IV-2023 (1 ♀). Itauçu 1: Urochloa brizantha, XII-2022 (1 ♀). Itauçu 2: Urochloa brizantha, XI-2022 (1 ♀). Nova Veneza 1: Urochloa brizantha, III-2023 (5 ♀). Nova Veneza 2: Urochloa brizantha, XII-2022 (1 ♀), III-2023 (2 ♀). Nova Veneza 3: Urochloa brizantha, IV-2023 (1 ♀), V-2023 (2 ♀). Nova Veneza 4: Urochloa brizantha, XII-2022 (1 ♀), IV-2023 (2 ♀), V-2023 (2 ♀). Nova Veneza 5: Urochloa brizantha, V-2023 (2 ♀); Urochloa plantaginea, III-2023 (1 ♀, 1 ♂).
Previous records — Australia; Benin; Brazil - Alagoas, Bahia, Distrito Federal, Maranhão, Mato Grosso do Sul, Paraná, Paraíba, Pernambuco, Piauí, Rondônia, São Paulo, Tocantins; Canada; China; Colombia; Costa Rica; Cuba; Galapagos; Ghana; Guadeloupe; Hawaii; Ivory Coast; Jamaica; Japan; Kenya; Madagascar; Martinique; Mexico; New Zealand; Nicaragua; Panama; Peru; Reunion Island; Rodrigues Island (Mauritius); Saudi Arabia; Taiwan; United Arab Emirates; United States of America.
Observation — this is the first record of this species in Goiás State, Brazil.
Proprioseiopsis neotropicus (Ehara)
Amblyseius neotropicus Ehara 1966: 133.
Proprioseiopsis neotropicus — Moraes et al. 1986: 119, 2004: 183; Lofego et al. 2024: 120.
Specimens examined — Nova Veneza 4: Urochloa brizantha, IV-2023 (1 ♀), V-2023 (1 ♀).
Previous records — Argentina; Brazil - Alagoas, Amazonas, Bahia, Mato Grosso, Mato Grosso do Sul, Minas Gerais, Pará, Paraná, Paraíba, Pernambuco, Rio Grande do Sul, Roraima, São Paulo; Colombia; Ecuador; French Guiana; Peru.
Observation — this is the first record of this species in Goiás State, Brazil.
Proprioseiopsis ovatus (Garman)
Amblyseiopsis ovatus Garman 1958: 78.
Typhlodromus (Amblyseius) ovatus — Chant 1959: 90.
Amblyseiulus ovatus — Muma 1961: 278.
Typhlodromus ovatus — Hirschmann 1962: 5.
Amblyseius ovatus — Schuster & Pritchard 1963: 246.
Amblyseius (Amblyseius) ovatus — Tseng 1983: 42.
Proprioseiopsis ovatus — Tuttle & Muma 1973: 11; Moraes et al. 1986: 121, 2004: 151; Lofego et al. 2024: 121.
Proprioseiopsis (Proprioseiopsis) ovatus — Karg 1989: 208.
Proprioseiopsis antonelli Congdon — (synonymy according to Denmark & Evans 2011).
Proprioseiopsis cannaensis (Muma) — (synonymy according to Denmark & Evans 2011).
Proprioseiopsis hundsonianus (Chant and Hansell) — (synonymy according to Denmark & Evans 2011).
Proprioseiopsis parapeltatus Wu and Chou — (synonymy according to Tseng 1983).
Proprioseiopsis peltatus (Van der Merwe) — (synonymy according to Tseng 1983).
Specimens examined — Brazabrantes 1: Urochloa brizantha, XI-2022 (1 ♀); Cynodon nlemfuensis, IV-2023 (1 ♀). Brazabrantes 2: Urochloa brizantha, II-2023 (1 ♀), III-2023 (3 ♀, 1 M), IV-2023 (3 ♀), V-2023 (4 ♀). Caturaí 1: Urochloa brizantha, XII-2022 (5 ♀), I-2023 (1 ♀), III-2023 (8 ♀, 2 ♂), IV-2023 (7 ♀, 1 ♂), V-2023 (7 ♀, 4 ♂). Caturaí 2: Urochloa arrecta, XI-2022 (1 ♀). Inhumas 1: Urochloa brizantha, XI-2022 (1 ♀), XII-2022 (1 ♀), IV-2023 (1 ♀), V-2023 (1 ♀). Inhumas 2: Urochloa plantaginea, III-2023 (4 ♀), V-2023 (7 ♀). Itaberaí 1: Urochloa brizantha, XII-2022 (2 ♀), IV-2023 (1 ♀), V-2023 (1 ♀). Itaberaí 2: Urochloa humidicola, I-2023 (1 ♀), IV-2023 (1 ♀), V-2023 (1 ♀). Itauçu 1: Urochloa brizantha, XII-2022 (1 ♀). Nova Veneza 1: Urochloa brizantha, XII-2022 (1 ♀), II-2023 (10 ♀), IV-2023 (2 ♀), V-2023 (1 ♀). Nova Veneza 2: Urochloa brizantha, XII-2022 (2 ♀), IV-2023 (19 ♀), V-2023 (18 ♀, 3 ♂). Nova Veneza 3: Urochloa brizantha, XI-2022 (1 ♀), III-2023 (2 ♀), IV-2023 (17 ♀). Nova Veneza 4: Urochloa brizantha, XI-2022 (1 ♀), III-2023 (12 ♀), IV-2023 (1 ♀, 1 ♂), V-2023 (2 ♀); Urochloa arrecta, XI-2022 (1 ♀). Nova Veneza 5: Urochloa plantaginea, I-2023 (1 ♀), III-2023 (1 ♀), IV-2023 (4 ♀), V-2023 (1 ♀).
Previous records — Argentina; Australia; Brazil - Alagoas, Bahia, Distrito Federal, Goiás, Mato Grosso, Mato Grosso do Sul, Minas Gerais, Pará, Paraná, Rio Grande do Sul, São Paulo; Canada; China; Colombia; Costa Rica; Cuba; DR Congo; Ecuador; Egypt; El Salvador; Fiji; French Guiana; Ghana; Grande Comore Island (Comoros); Guadeloupe; Guyana; Hawaii; Honduras; India; Ivory Coast; Japan; Lesotho; Madagascar; Malaysia; Malawi; Marie Galante; Martinique; Mayotte Island; Mozambique; New Caledonia; Papua New Guinea; Paraguay; Peru; Philippines; Puerto Rico; Reunion Island; Saudi Arabia; Sierra Leone; South Africa; Spain; Sri Lanka; Taiwan; Thailand; Turkey; United States of America; Venezuela; Zimbabwe.
Observation — Proprioseiopsis ovatus has been previously recorded on Psidium guineense Swartz (Myrtaceae) and Guapira noxia (Netto) Lundell (Nyctaginaceae) in the state of Goiás (Demite et al. 2017).
Proprioseiopsis pentagonalis (Moraes & Mesa)
Amblyseius pentagonalis Moraes & Mesa, in Moraes et al. (1991): 127.
Proprioseiopsis pentagonalis — Moraes et al. 2004: 186; Lofego et al. 2024: 124.
Specimens examined — Inhumas 2: Urochloa brizantha, V-2023 (5 ♀); Urochloa plantaginea (Poaceae), V-2023 (3 ♀). Nova Veneza 3: Urochloa brizantha, IV-2023 (3 ♀).
Previous records — Brazil - Amazonas, Bahia, Maranhão, Piauí, São Paulo; Colombia; Guadeloupe.
Observation — this is the first record of this species in Goiás State, Brazil.
Typhlodromalus Muma
Typhlodromalus aripo De Leon
Amblyseius aripo De Leon 1967: 21.
Typhlodromalus aripo — Moraes & McMurtry 1983: 132; Moraes et al. 1986: 128, 2004: 195. Lofego et al. 2024: 169.
Specimens examined — Nova Veneza 1: Urochloa brizantha, II-2023 (1 ♀). Nova Veneza 4: Urochloa brizantha, II-2023 (1 ♂).
Previous records — Argentina; Benin; Brazil - Alagoas, Amazonas, Bahia, Ceará, Distrito Federal, Espírito Santo, Goiás, Maranhão, Mato Grosso, Mato Grosso do Sul, Minas Gerais, Pará, Paraná, Paraíba, Pernambuco, Piauí, Rio Grande do Norte, Rio Grande do Sul, Santa Catarina, Sergipe, São Paulo, Tocantins; Cameroon; Colombia; Congo; Costa Rica; El Salvador; Guadeloupe; Guyana; Jamaica; Kenya; Malawi; Mexico; Mozambique; Paraguay; Peru; Trinidad; Uganda.
Observation — Typhlodromalus aripo has previously been recorded in Goiás State on Virola sebifera Fusée-Aublet (Myristicaceae), Vernonanthura sp. (Compositae), Luehea divaricata von Martius (Malvaceae), Cecropia pachystachya Trécul (Urticaceae), Zeyheria montana von Martius (Bignoniaceae), Magonia pubescens de Saint-Hilaire (Sapindaceae), Schefflera macrocarpa (von Chamisso and von Schlechtendal) Frodin (Araliaceae), Tibouchina sp. (Melastomataceae) and unidentified species of Rubiaceae and Malpighiaceae (Demite et al. 2017).
Typhlodromalus clavicus Denmark & Muma
Typhlodromalus clavicus Denmark & Muma 1973: 257.
Typhlodromalus simus — Moraes et al. 1986: 134, 2004: 204; Lofego et al. 2024: 171.
Specimens examined — Brazabrantes 1: Cynodon nlemfuensis, V-2023 (1 ♀).
Previous records — Brazil - São Paulo, Tocantins; El Salvador; Honduras.
Observation — this is the first record of this species in Goiás State, Brazil.
Typhlodromalus peregrinus Muma
Typhlodromus peregrinusMum a 1955: 270.
Typhlodromalus peregrinus — Moraes et al. 1986: 132, 2004: 202; Lofego et al. 2024: 176.
Typhlodromus (Typhlodromopsis) peregrinus — De Leon 1959: 114.
Typhlodromus (Amblyseius) peregrinus — Chant 1959: 97.
Amblyseius (Typhlodromalus) peregrinus — Muma 1961: 288.
Amblyseius peregrinus — Muma and Denmark 1962: 60.
Amblyseius (Amblyseius) peregrinus — Van der Merwe 1968: 113.
Typhlodromalus evansi (Chant) — (synonymy according to Muma 1964).
Typhlodromalus primulae (Chant) — (synonymy according to Muma 1964).
Typhlodromalus robiniae (Chant) — (synonymy according to Muma 1964).
Typhlodromalus sextus (Garman, 1958) — (synonymy according to Denmark & Evans 2011).
Examined material — Nova Veneza 3: Urochloa plantaginea, XI-2022 (1 ♀). Nova Veneza 4: Urochloa brizantha, II-2023 (2 ♀);
Previous records — Argentina; Brazil - Acre, Alagoas, Amazonas, Bahia, Ceará, Goiás, Mato Grosso, Paraíba, Pernambuco, Rio de Janeiro, Rio Grande do Norte, Rio Grande do Sul, Sergipe, São Paulo, Tocantins; Colombia; Costa Rica; Cuba; Dominican Republic; Ecuador; Guadeloupe; Guatemala; Guyana; Hawaii; Honduras; Martinique; Mexico; Nicaragua; Peru; Puerto Rico; Suriname; United States of America; Venezuela.
Observation — Typhlodromalus peregrinus has previously been recorded on Protium spruceanum (Bentham) Engler (Burseraceae) in Goiás State (Demite et al. 2017).
Typhlodromips De Leon
Typhlodromips mangleae De Leon
Typhlodromips mangleae De Leon 1967: 28.
Typhlodromips mangleaem — Moraes et al., 1986: 143, 2004: 217; Lofego et al 2024: 251.
Amblyseius mangleae — Moraes & Mesa 1988: 75.
Specimens examined — Brazabrantes 1: Urochloa brizantha, (1 ♀). Nova Veneza 2: Urochloa brizantha, V-2023 (1 ♀). Nova Veneza 4: Urochloa brizantha, II-2023 (1 ♀), III-2023 (1 ♀), V-2023 (1 ♀).
Previous records — Brazil - Alagoas, Bahia, Ceará, Distrito Federal, Maranhão, Mato Grosso do Sul, Minas Gerais, Pará, Paraná, Pernambuco, Rio Grande do Sul, São Paulo, Tocantins; Colombia; Peru; Puerto Rico; Trindad.
Observation — this is the first record of this species in Goiás State, Brazil.
Typhlodrominae Wainstein
Metaseiulus Muma
Metaseiulus (Metaseiulus) camelliae (Chant & Yoshida-Shaul)
Typhlodromus camelliae Chant & Yoshida-Shaul 1983: 1053.
Typhlodromina camelliae — Moraes et al. 1986: 236.
Metaseiulus (Metaseiulus) camelliae — Moraes et al. 2004: 278.
Metaseiulus camelliae — Bellini et al. 2005 479.
Specimens examined — Itauçu 2: Urochloa brizantha, XII-2022 (1 ♀).
Previous records — Argentina; Brazil - Minas Gerais, Rio Grande do Sul, Santa Catarina, São Paulo; Chile; Uruguay.
Observation — this is the first record of M. (M.) camelliae in Goiás State.
Silvaseius Chant & McMurtry
Silvaseius barretoae Yoshida-Shaul & Chant
Cydnodromella barretoae Yoshida-Shaul & Chant 1991: 93.
Silvaseius barretoae — Chant & McMurtry 1994: 241; Moraes et al. 2004: 304.
Specimens examined — Itaberaí 1: Urochloa brizantha, IV-2023 (1 ♀).
Previous records — Argentina; Brazil - Mato Grosso, São Paulo; Costa Rica.
Observation — this is the first record of this species in Goiás State, Brazil.
Typhlodromus Scheuten
Typhlodromus (Anthoseius) transvaalensis (Nesbitt)
Kampimodromus transvaalensis Nesbitt 1951: 55.
Typhlodromus (Typhlodromus) transvaalensis — Chant 1959: 60.
Neoseiulus transvaalensis — Muma 1961: 295.
Typhlodromus (Neoseiulus) transvaalensis — Pritchard & Baker 1962: 222.
Mumaseius transvaalensis — Abbasova 1970: 1410.
Clavidromus transvaalensis — Muma & Denmark 1971: 238; Moraes et al. 1986: 182.
Anthoseius (Anthoseius) transvaalensis — Wainstein & Vartapetov 1973: 104.
Anthoseius transvaalensis — Beglyarov 1981: 21.
Typhlodromus (Anthoseius) transvaalensis — Chant & McMurtry 1994: 252; Moraes et al. 2004: 355.
Anthoseius (Clavidromus) transvaalensis — Kolodochka 2006: 145.
Typhlodromus (Anthoseius) jackmickleyi De Leon — (synonymy according to Muma & Denmark 1968).
Typhlodromus (Anthoseius) pectinatus Athias-Henriot — (synonymy according to Muma & Denmark 1968).
Specimens examined — Caturaí 1: Urochloa brizantha, XII-2022 (2 ♀). Inhumas: Urochloa brizantha, XII-2022 (1 ♀). Nova Veneza 1: Urochloa brizantha, III-2023 (1 ♀). Nova Veneza 2: Urochloa brizantha, I-2023 (1 ♀). Nova Veneza 3: Urochloa brizantha, XII-2022 (1 ♀), III-2023 (6 ♀). Itaberaí 1: Urochloa brizantha, IV (1 ♀). Itaberaí 2: Urochloa humidicola, XII-2022 (1 ♀).
Previous records — Algeria; Anjouan Island; Argentina; Australia; Queensland; Azerbaijan; Brazil - Amazonas, Bahia, Mato Grosso, Pará, Paraná, Pernambuco, Rio Grande do Sul, São Paulo, Tocantins; Cameroon; Cape Verde; China; Colombia; Costa Rica; Cuba; Egypt; Georgia; Guinea; Hawaii; India; Indonesia; Iran; Israel; Italy; Japan; Jordan; Kenya; Mexico; New Caledonia; Panama; Peru; Philippines; Reunion Island; Singapore; South Africa; Spain; Taiwan; United States of America.
Observation — Typhlodromus (Anthoseius) transvaalensis was previously recorded in Goiás State by Rossetti et al. (2025).
Discussion
The grass species used for pastures in Goiás State act as reservoirs for predatory mites. In the present study, 21 Phytoseiidae species were recorded in pastures used for livestock. These results were similar to the findings of Lofego et al. (2009), which in turn, reported 21 phytoseiid mites on grasses of the species U. decumbens, U. brizantha, U. ruziziensis, Melinis minutiflora, Urochloa lorentziana, and Pennisetum purpureum in São Paulo State. Previous studies on native plants in the Cerrado recorded 34 species of Phytoseiidae (Demite et al. 2017), while another survey reported 26 species of mites from the same family (Rezende and Lofego 2011). In the Pantanal biome, Mendonça et al. (2019) reported 35 Phytoseiidae species associated with native trees. Additionally, collections carried out in the Cerrado, Pantanal, and Amazon Forest recorded nine, 16, and 11 species, respectively (Conceição et al. 2021), while another sampling in the same biomes recorded five, five, and 17 phytoseiid species, respectively (Demite et al. 2021). Rossetti et al. (2025) recorded 19 phytoseiid species in remnants of Cerrado vegetation in the state of Goiás. Thus, pasture areas, although simplified ecosystems, can exhibit considerable diversity when compared to natural vegetation remnants.
Among Phytoseiidae species sampled, only I. zuluagai, N. benjamini, P. mexicanus and P. biologicus have been previously reported in Brazilian grasslands (Lofego et al. 2009). For the remaining species, this study provides the first record in Brazilian pasture crops. Proprioseiopsis ovata was the most abundant species in the evaluated pastures. Species from this genus are widely used in natural biological control. For example, P. mexicanus, the third most abundant phytoseiid species sampled here, is a generalist predator with the potential to control arthropod pests (Yuan et al., 2025). Proprioseiopsis mexicanus has also been recorded at high abundances during the wet season (Mailloux et al. 2010). Neoseiulus benjamini was the second most abundant species in pastures from livestock farms in Goiás State. This species was previously reported on monocotyledonous plants from the families Poaceae and Cyperaceae (Demite et al. 2017; Ferragut and Navia 2022).
Our results showed that Poaceae crops support a rich assemblage of Phytoseiidae species. Therefore, assessing the biodiversity in pastures is essential for crop management, as these grass species are widely used for soil cover in Brazilian agroecosystems (Flechtmann 2004; Aguilar-Fenollosa et al. 2011; Blanco-Pérez et al. 2020). These results highlights the importance of avoiding farm management practices that affect predator populations (Farfan et al. 2021). However, more studies are necessary to increase our knowledge of the biodiversity associated with pasture crops and the interactions between mites and grasses.
Acknowledgements
Financial support was provided by CAPES/FAPEG within the ''Apoio aos Programas de Pós-Graduação Emergentes e em Consolidação em Áreas Prioritárias nos Estados» Program [Proc. nº 202010267000426]. V.L. Falchi was supported by a scholarship from the ''Coordenação de Aperfeiçoamento de Pessoal de Nível Superior» (CAPES) and Fundação de Amparo à Pesquisa do Estado de Goiás (FAPEG). L.L.C. Ribeiro receives a scholarship from CAPES/FAPEG. To the researcher Antônio Carlos Lofego (Universidade Estadual Paulista – UNESP, São José do Rio Preto, São Paulo, Brazil) for providing the microscope used for the identification of Phytoseiidae. We also thank Maria Victória Cordeiro Gama (Universidade Federal de Goiás) for reviewing the manuscript.
References
- Abbasova E.D. 1970. Little known species and new subspecies of the genus Mumaseius De Leon (Acarina: Phytoseiidae). Zoologicheskii Zhurnal, 49: 1410-1414.
- Aguilar-Fenollosa E., Ibáñez-Gual M.V., Pascual-Ruiz S., Hurtado M., Jacas J.A. 2011. Effect of ground-cover management on spider mites and their phytoseiid natural enemies in clementine mandarin orchards (I): Bottom-up regulation mechanisms. Biol. Control., 59: 158-170. https://doi.org/10.1016/j.biocontrol.2011.06.013
- Barbosa M.F.C., Demite P.R. 2023. A new species of Arrenoseius Wainstein (Mesostigmata: Phytoseiidae) from Brazil, with a world key to the genus. Int. J. Acarol., 49: 49-53. https://doi.org/10.1080/01647954.2023.2178505
- Beglyarov G.A. 1981 Keys to the determination of phytoseiid mites of the USSR. Information Bulletin International Organization for Biological Control of Noxious Animals and Plants, East Palaearctic Section, Leningrad Russia. 97 pp.
- Bellini M.R., Moraes G.J. de, Feres R.J.F. 2005. Ácaros (Acari) de dois sistemas de cultivo da seringueira no noroeste do estado de São Paulo. Neotrop. Entomol., 34: 475-484. https://doi.org/10.1590/S1519-566X2005000300017
- Blanco-Pérez R., Sáenz-Romo M.G., Vicente-Díez I., Ibáñez-Pascual S., Martínez-Villar E., Marco-Mancebón V.S., Pérez-Moreno I., Campos-Herrera R. 2020. Impact of vineyard ground cover management on the occurrence and activity of entomopathogenic nematodes and associated soil organisms. Agricul. Ecosyst. Environ., 301: 1-11. https://doi.org/10.1016/j.agee.2020.107028
- Cavalcante A.C.C., Demite P.R., Carneiro M.E.S., Nunes L.S., Pereira A.I.A. 2017. Mites (Acari) associated with soybean culture (Glycine max), in the southeast region of Goiás state. Magistra, 29: 266-272.
- Chant D.A., McMurtry J.A. 1994 A review of the subfamilies Phytoseiinae and Typhlodrominae (Acari: Phytoseiidae). Int. J. Acarol., 20: 223-310. https://doi.org/10.1080/01647959408684022
- Chant D.A., McMurtry J.A. 2007. Illustrated keys and diagnoses for the genera and subgenera of the Phytoseiidae of the world (Acari: Mesostigmata). Indira Publishing House. 219 pp.
- Chant D.A. 1955. Notes on mites of the genus Typhlodromus Scheuten, 1857 (Acarina: Laelaptidae), with description of the males of some species and the female of a new species. Can. Ent., 887: 496-503. https://doi.org/10.4039/Ent87496-11
- Chant D.A. 1959. Phytoseiid mites (Acarina: Phytoseiidae). Part I. Bionomics of seven species in southeastern England. Part II. A taxonomic review of the family Phytoseiidae, with descriptions of thirty-eight new species. Can. Entomol., 91: 45-166. https://doi.org/10.4039/entm9112fv
- Chant D.A., Yoshida-Shaul E. 1983. A world review of five similar species groups in the genus Typhlodromus Scheuten: Part II. The conspicuus and cornus groups (Acarina: Phytoseiidae). Can. J. Zool., 61: 1041-1057. https://doi.org/10.1139/z83-138
- Collyer E. 1964. The occurrence of some mites of the family Phytoseiidae in New Zealand, and descriptions of seven new species. Acarologia, 6: 632-646.
- Conceição E.M, Demite P.R., Rezende J.M., Carnniello M.A., Lofego A.C. 2021. Phytoseiidae (Acari: Parasitiformes: Mesostigmata) inhabiting native plants from three biomes in Mato Grosso State, Brazil, with description of a new species. Syst. Appl. Acarol., 26: 2268-2286. https://doi.org/10.11158/saa.26.12.6
- De Leon D. 1959. Seven new Typholodromus from Mexico with collection notes on three other species (Acarina: Phytoseiidae). Fla. Entomol., 42: 113-121. https://doi.org/10.2307/3492606
- De Leon D. 1967. Some mites of the Caribbean Area. Part I. Acarina on plants in Trinidad, West Indies. Allen Press Inc., Lawrence, Kansas, USA. 66 pp.
- Demite P.R., Dias M.A., Cavalcante A.C.C., Ramos M.V.V., Lofego A.C. 2017. Phytoseiid mites (Acari: Mesostigmata: Phytoseiidae) associated with Cerrado biome plants in Brazil, with description of a new species. Syst. Appl. Acarol., 22: 2141-2177. https://doi.org/10.11158/saa.22.12.9
- Demite P.R., Moraes G.J. de, McMurtry J.A., Denmark H.A., Castilho R.C. 2025. Phytoseiidae Database. Available from: \textless www.lea.esalq.usp.br/phytoseiidae\textgreater. Accessed 09 January 2025.
- Denmark H.A., Evans G.A. 2011. Phytoseiidae of North America and Hawaii (Acari: Mesostigmata). Indira Publishing House, West Bloomfield, USA. 451 pp.
- Denmark H.A., Muma M.H. 1989. A revision of the genus Amblyseius Berlese, 1914 (Acari: Phytoseiidae). Occasional Papers of the Florida State Collection of Arthropods. 149 pp.
- Denmark H.A., Muma M.H. 1973. Phytoseiidae mites of Brazil (Acarina: Phytoseiidae). Rev. Bras. Biol., 33: 235-276.
- Denmark H.A., Muma M.H. 1972. Some Phytoseiidae of Colombia (Acarina: Phytoseiidae). Fla. Entomol., 55: 19-29. https://doi.org/10.2307/3493637
- Döker I., Revynthi A.M., Kazak C., Carrillo D. 2023. Interactions among exotic and native phytoseiids (Acari: Phytoseiidae) affect biocontrol of two-spotted spider mite on papaya. Biol. Control., 163: 1-8. https://doi.org/10.1016/j.biocontrol.2021.104758
- Ehara S. 1966. Some mites associated with plants in the state of São Paul, Brazil, with a list of plant mites of South America. Jpn. J. Zool., 15: 129-150.
- Farfan M.A., Coffey J., Schmidt-Jeffris R.A. 2021. Suitability of food resources for Proprioseiopsis mexicanus, a potentially important natural enemy in eastern USA agroecosystems. Exp. Appl. Acarol., 84: 121-134. https://doi.org/10.1007/s10493-021-00622-6
- Ferragut F., Navia D. 2024. Phytoseius Ribaga (Mesostigmata: Phytoseiidae) in the Brazilian Espinhaço Range: seven new species, renaming of the plumifer species group, and a critical review of its taxonomic characters. Zootaxa, 5493: 201-245. https://doi.org/10.11646/zootaxa.5493.3.1
- Ferragut F., Navia, D. 2022. The genus Neoseiulus Hughes (Mesostigmata: Phytoseiidae) in the Espinhaço Range, a great reservoir of biodiversity in Brazil. Zootaxa, 5120: 523-542. https://doi.org/10.11646/zootaxa.5120.4.4
- Flechtmann C.H.W. 2004. Two new plant feeding mites from Urochloa ruziziensis in citrus groves in São Paulo, Brazil and new distribution records of other plant mites in Brazil. Zootaxa, 708: 1-11. https://doi.org/10.11646/zootaxa.708.1.1
- Flechtmann C.H.W. 2000. Two new species of Aceria (Acari: Eriophyidae) on grass from Brazil. Int. J. Acarol., 26: 335-338. https://doi.org/10.1080/01647950008684207
- Flechtmann C.H.W., Navia D. 2007. Tetranychus (Armenychus) graminae Flechtmann, 1971 (Acari: Tetranychidae) infesting elephant grass, Pennisetum purpureum Schum. (Poaceae), in Central Brazil. Int. J. Acarol., 33: 323-32. https://doi.org/10.1080/01647950708683693
- Garman P. 1958. New species belonging to the genera Amblyseius and Amblyseiopsis with keys to Amblyseius, Amblyseiopsis, and Phytoseiulus. Ann. Entomol. Soc. Am., 51: 69-79. https://doi.org/10.1093/aesa/51.1.69
- Hirschmann W. 1962. Gangystematik der Parasitiformes. Acarologie Schriftenreihe fur Vergleichende Milbenkunde, Hirschmann-Verlag, Furth/Bay, 5. 80 pp.
- Hyte K., Rhodes E.M., Mallinger R., Liburd O.E. 2023. Effect of sweet alyssum on efficacy and sustainability of Neoseiulus californicus (Acari: Phytoseiidae) for biological control of spider mites in multiple cultivars of organic strawberries. Crop. Prot., 174: 1-7. https://doi.org/10.1016/j.cropro.2023.106436
- Kolodochka L.A. 2006. Phytoseiid mites of the Palaerarctic Region (Parasitiformes, Phytoseiidae): faunistic, taxonomy, ecomorphology, evolution. Vestnik Zoologia, suppl. 21. 250 pp.
- LAPIG, Laboratório de Processamento de Imagens e Geoprocessamento. Atlas das pastagens. Available from: https://atlasdaspastagens.ufg.br/. Accessed 07 January 2025.
- Lofego A.C., Barbosa M.F. de C., Demite P.R., Moraes G.J. de. 2024. Phytoseiidae (Acari: Mesostigmata) of the subfamily Amblyseiinae from Brazil. Zootaxa, 5439: 1-306. https://doi.org/10.11646/zootaxa.5439.1.1
- Lofego A.C., Demite P.R., Kishimoto R.G., Moraes G.J. de. 2009. Phytoseiid mites on grasses in Brazil (Acari: Phytoseiidae). Zootaxa, 2240: 41-59. https://doi.org/10.11646/zootaxa.2240.1.3
- Mailloux J., Le Bellec F., Kreiter S., Tixier, M.-S. 2010. Influence of ground cover management on diversity and density of phytoseiid mites (Acari: Phytoseiidae) in Guadeloupean citrus orchards. Exp. Appl. Acarol., 52: 275-290. https://doi.org/10.1007/s10493-010-9367-7
- McMurtry J.A., Moraes G.J. de, Sourassou N.F. 2013. Revision of the lifestyles of phytoseiid mites (Acari: Phytoseiidae) and implications for biological control strategies. Syst. Appl. Acarol., 18: 297-320. https://doi.org/10.11158/saa.18.4.1
- Mendonça A.L, Lofego A.C., Pott A., Daud R.D., Demite P.R. 2019. Phytoseiidae (Parasitiformes: Mesostigmata) from the Pantanal, Mato Grosso do Sul State, Brazil. Syst. Appl. Acarol., 24: 587-612. https://doi.org/10.11158/saa.24.4.6
- Mineiro J.L.C., Lofego A.C., Sato M.E. 2012. Phytoseiid mites (Acari: Phytoseiidae) found in soil and litter from Atibaia, State of São Paulo, Brazil, with description of two new species. Int. J. Acarol., 38: 595-604. https://doi.org/10.1080/01647954.2012.713001
- Moraes G.J. de, McMurtry J.A., Denmark H.A. 1986. A catalog of the mite family Phytoseiidae. References to taxonomy, synonymy, distribution and habitat. Embrapa, Brasília, Brazil. 353 pp.
- Moraes G.J. de, McMurtry J.A., Denmark H.A., Campos C.B. 2004. Revised catalog of the mite family Phytoseiidae. Zootaxa, 434: 1-494. https://doi.org/10.11646/zootaxa.434.1.1
- Moraes G.J. de, Melo E.L., Gondim Jr. M.G.C. 1997. Description of a new species of phytoseiid mite from northeastern Brazil and redescriptions of Neoseiulus gracilis (Acari, Phytoseiidae). Fla. Entomol., 80: 319-324. https://doi.org/10.2307/3495765
- Moraes G.J. de, Castilho R.C. de, Flechtmann C.H.W. 2024. Manual de Acarologia - Acarologia Básica e Ácaros de Plantas Cultivadas no Brasil. Fealq. 486 pp. https://doi.org/10.37856/9786589722588.fealq.2024.485
- Moraes G.J. de, McMurtry J.A. 1983. Phytoseiid mites (Acarina) of Northeastern Brazil with descriptions of four new species. Int. J. Acarol., 9: 131-148. https://doi.org/10.1080/01647958308683326
- Moraes G.J. de, Mesa N.C., Braun A. 1991. Some phytoseiid mites of Latin America (Acari: Phytoseiidae). Int. J. Acarol., 17: 117-139. https://doi.org/10.1080/01647959108683892
- Moraes G.J. de, Mesa N.C. 1988. Mites of the family Phytoseiidae (Acari) in Colombia, with descriptions of three new species. Int. J. Acarol., 14: 71-88. https://doi.org/10.1080/01647958808683790
- Moraes V. de S., Nunes S.N., Demite P.R., Daud R.D. 2022. Vegetation structure define mite assemblage on plants: a case study in Cerrado biome. Entomol. Commun., 4: 1-4. https://doi.org/10.37486/2675-1305.ec04029
- Möth S., Richart-Cervera S., Herrera R.A., Hoffmann C., Kolb S., Popescu D., Reiff J.M., Rusch A., Tolle P., Walzer A., Winter S. 2023. Local management and landscape composition affect predatory mites in European wine-growing regions. Agricul. Ecosyst. Environ., 344: 1-15. https://doi.org/10.1016/j.agee.2022.108292
- Muma, M.H. 1964. Annotated list and keys to Phytoseiidae (Acarina: Mesostigmata) associated with Florida citrus. University of Florida Agricultural Experiment Station Bulletin, 685, 1-42.
- Muma M.H., Denmark H.A. 1962. Intraspecific variation in Phytoseiidae (Acarina: Mesostigmata). Fla. Entomol., 45: 57-65. https://doi.org/10.2307/3492217
- Muma M.H., Denmark H.A. 1968. Some generic descriptions and name change in the family Phytoseiidae (Acarina: Mesostigmata). Fla. Entomol., 51: 229-240. https://doi.org/10.2307/3493424
- Muma M.H. 1962. New Phytoseiidae (Acarina: Mesostigmata) from Florida. Fla. Entomol, 45: 1-10. https://doi.org/10.2307/3492897
- Muma M.H. 1955. Phytoseiidae (Acarina) associated with citrus in Florida. Ann. Entomol. Soc. Am., 48: 262-272. https://doi.org/10.1093/aesa/48.4.262
- Muma M.H. 1961. Subfamilies, genera, and species of Phytoseiidae (Acarina: Mesostigmata). Bull. Fla. State Mus., 5: 267-302. https://doi.org/10.58782/flmnh.tqpo4380
- Nesbitt H.H.J. 1951. A taxonomic study of the Phytoseiidae (Family Laelaptidae) predaceous upon Tetranychidae of economic importance. Zoologische Verhandelingen, 12 pp.
- Pritchard A.E., Baker E.W. 1962. Mites of the family Phytoseiidae from Central Africa, with remarks on genera of the world. Hilgardia, 33: 205-309. https://doi.org/10.3733/hilg.v33n07p205
- Rezende J.M., Lofego A.C. 2011. Phytoseiidae (Acari: Mesostigmata) on plants of the central region of the Brazilian Cerrado. Acarologia, 51: 449-463. https://doi.org/10.1051/acarologia/20112027
- Rossetti O.C., Barroso G., Demite P.R., de Lima E.L., Daud R.D. 2025. Phytoseiidae mite (Parasitiformes: Phytoseiidae) assemblages from different Cerrado vegetation types. Acarologia, 65: 505-518. https://doi.org/10.24349/3rdh-xfoj
- Schicha E. 1981. A new species of Amblyseius (Acari: Phytoseiidae) from Australia compared with ten closely related species from Asia, America and Africa. Int. J. Acarol., 7: 203-216. https://doi.org/10.1080/01647958108683262
- Schmidt R. A. 2013. Leaf structures affect predatory mites (Acari: Phytoseiidae) and biological control: a review. Exp. Appl. Acarol., 62: 1-17. https://doi.org/10.1007/s10493-013-9730-6
- Schuster R.O., Pritchard A.E. 1963. Phytoseiid mites of California. Hilgardia: 34, 191-285. https://doi.org/10.3733/hilg.v34n07p191
- Shavanov M.V. 2021. The role of food crops within the Poaceae and Fabaceae families as nutritional plants. IOP Conf. Ser. Earth and Environ. Sci: 624, 1-8. https://doi.org/10.1088/1755-1315/624/1/012111
- Tseng Y.H. 1983. Further study on phytoseiid mites from Taiwan (Acarina: Mesostigmata). Chin. J. Entomol: 3, 33-74.
- Tuttle D.M., Muma M.H. 1973. Phytoseiidae (Acarina: Mesostigmata) inhabiting agricultural and other plants in Arizona. Agricultural Experiment Station Technical Bulletin 208. University of Arizona, Tucson, Arizona. 55 pp.
- Ueckermann E.A., Loots G.C. 1988. The African species of the subgenera Anthoseius De Leon and Amblyseius Berlese (Acari: Phytoseiidae). Entomology Memoir, Department of Agriculture and Water Supply, Republic of South Africa, 73. 168 pp.
- Van der Merwe G.G. 1968. A taxonomic study of the family Phytoseiidae (Acari) in South Africa with contributions to the biology of two species. Entomol. Mem.: 18, 1-198.
- Wainstein B.A., Vartapetov S.G. 1973. Predatory mites of the family Phytoseiidae (Parasitiformes) of Adzharskaya ASSR. Akademiya Nauk Armyanskoy SSR, Biologicheskiy Zhurnal Armenii: 26, 102-105.
- Yoshida-Shaul E., Chant D.A. 1991. Adult ventral setal patterns in the family Phytoseiidae (Acari: Gamasina). Int. J. Acarol.: 17, 187-199. https://doi.org/10.1080/01647959108683906
- Yuan X., Xie Y., Zhang B., Xu X., Liu Z., Wang E., Wei G.S. 2025. Assessment of predatory mite Proprioseiopsis mexicanus as a biocontrol agent against multiple sap-suckling pests. Syst. Appl. Acarol.: 30, 275-285. https://doi.org/10.11158/saa.30.2.7



2025-10-07
Date accepted:
2025-11-03
Date published:
2025-11-14
Edited by:
Kreiter, Serge

This work is licensed under a Creative Commons Attribution 4.0 International License
2025 Falchi, Vanessa L.; Ribeiro, Lana Letícia C.; Demite, Peterson R. and Daud, Rodrigo D.
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



