Oribatid mite fauna of three members of the lichen genus Cladonia in Europe – observations on species richness, endophagous juveniles and their morphology
Pfingstl, Tobias  1
; Vtípilová, Věra
1
; Vtípilová, Věra  2
; Ghlimová, Heda
2
; Ghlimová, Heda  3
; Mourek, Jan
3
; Mourek, Jan  4
; Steinová, Jana
4
; Steinová, Jana  5
; Hessinger, Tobias P. 6
; Schäffer, Sylvia
5
; Hessinger, Tobias P. 6
; Schäffer, Sylvia  7
and Resl, Philipp
7
and Resl, Philipp  8
8
1✉ Department of Biology, University of Graz, Universitaetsplatz 2, 8010 Graz, Austria.
2Department of Botany, Faculty of Science, Charles University, Benátská 2, 12801 Prague, Czech Republic & Institute for Environmental Studies, Faculty of Science, Charles University, Benátská 2, 12801 Prague, Czech Republic.
3Department of Botany, Faculty of Science, Charles University, Benátská 2, 12801 Prague, Czech Republic & National Museum, Mycological Department, Cirkusová 1740, 19300, Prague, Czech Republic
4Department of Biology Education, Faculty of Science, Charles University, Viničná 7, 128 43 Prague, Czech Republic.
5Department of Botany, Faculty of Science, Charles University, Benátská 2, 12801 Prague, Czech Republic.
6Department of Biology, University of Graz, Universitaetsplatz 2, 8010 Graz, Austria.
7Department of Biology, University of Graz, Universitaetsplatz 2, 8010 Graz, Austria.
8Department of Biology, University of Graz, Holteigasse 6, 8010 Graz, Austria.
2025 - Volume: 65 Issue: 4 pages: 1092-1123
https://doi.org/10.24349/nalv-gpxmZooBank LSID: 340B9B69-19C2-4570-A7BF-BC84B5891186
Original research
Keywords
Abstract
Introduction
Lichens are complex symbiotic organisms consisting of fungi and photosynthetic green algae or cyanobacteria as well as other bacteria (Spribille 2018). Lichens exhibit intriguing morphological variation and display a wide array of different (unique) substances (Singh et al. 2025). They thrive on various substrates including trees, rocks and soil and provide an important habitat for a considerable number of taxonomically diverse invertebrates, like Collembola, Psocoptera and Oribatida (e.g. Seyd and Seaward 1984, Prinzing and Wirtz 1997). The latter are mostly known as particle-feeding saprophagous and mycophagous taxa in soil and litter (e.g. Norton 2007), but they are frequently found in various lichens growing on a wide variety of substrates (e.g. Travé 1963, Dalenius 1960, MacLean 1981). Their relationship can range from casual, i.e. mites randomly foraging on the lichen, to highly dependent, i.e. where immature mites develop exclusively within the lichen (Seyd and Seaward 1984). For oribatid mites, lichens may serve as food source (Seyd and Seaward 1984), as shelter (Meier et al. 2002) and in some cases even as breeding ground (Grandjean 1950, 1951; Bellido 1979). While mites apparently benefit in several ways from using lichens as microhabitats, it is hypothesized that lichens, on the other hand, are mainly affected negatively. Some authors (Paulson and Thompson 1911, 1913) regard oribatid mites as the chief pests of lichens, a view that is supported by numerous accounts of mite infestation causing visible deterioration of lichen material (e.g. Laundon 1967, Sowter 1971, Gilbert 1976). However, the grazing activity may still be advantageous for the lichen in some cases, as the fecal pellets of oribatid mites can contain viable ascospores, mycelial fragments and photobiont cells and may thus serve as dispersal vectors of lichens or their symbionts (Meier et al. 2002). Moreover, grazing activity by mites can also induce sturdier growth of lichen branches, resulting in a reduced risk of damage by wind, as shown in the lichen Evernia prunasti (Prinzing and Wirtz 1997).
Although the relationship between oribatid mites and lichens is not yet fully understood, there is a considerable amount of literature showing that such associations are common. For example, Zopf (1907) mentioned that 29 species of larger foliose and fruticose lichens are regularly consumed by oribatid mites, André (1984) calculated that the mean density of oribatid mites in crustose and foliose lichens is approx. 53 individuals/dm2, Materna (2000) reported 47 species of oribatid mites from only four different saxicolous lichens, and Melekhina (2023) found 87 oribatid mite species in 35 species of ground and epiphytic lichens. Seyd and Seaward (1984) even classified mites into three groups according to their association with lichens, i.e. group A represents species that are restricted to lichens as their exclusive biotopes, group B contains species that prefer lichens but also feed occasionally on plants, and group C consists of species that frequently occur in lichens but are also common in other biotopes. Despite this comprehensive basic knowledge, little is known about specific relationships of individual lichens and their associated oribatid mites (Biazrov and Melekhina 1992, Fröberg et al. 2003).
Members of the lichen genus Cladonia were often reported to form close associations with oribatid mites (André 1984, Seyd and Seaward 1984, Bellido 1990, Materna 2000, Melekhina 2023). Cladonia symphycarpa, for example, is suggested to attract certain species of oribatid mites due to its specific secondary metabolites (Reutimann and Scheidegger 1987). Another unspecified Cladonia was reported to harbor high numbers of the oribatid mite Carabodes willmani, whereas its juveniles complete their whole life-cycle burrowing in the lichen thallus (Bellido 1979). This makes Cladonia species excellent models to investigate the intricate relationship of mites and lichens.
Cladonia norvegica is a relatively small, wood-inhabiting species that has, since its original description, regularly been reported to exhibit unusual red spots on its basal squamules and podetia. These spots contain rhodocladonic acid, a substance typically confined to fruiting bodies of red-fruited Cladonia species (clade Erythrocarpae, sensu Stenroos et al. 2018). Interestingly, Cladonia norvegica has ochraceous fruiting bodies and the production of the red pigment is restricted to the red spots (Timdal 1989). There, Timdal (1989) also observed whitish small mites inside holes within the lichen and suggested that the spots are a result of their feeding activity. He assumed these mites were adults, but their reported whitish appearance indicates they were more likely juveniles. Recently, Pino-Bodas et al. (2021) mentioned again that the red spotting of C. norvegica is caused by mite infestation and they further gave a name for the possible culprit, namely the oribatid mite Carabodes marginatus. However, recent molecular investigations (Vtípilová et al. 2025) revealed that red-spotted Cladonia norvegica specimens actually represent a novel species, called Cladonia rubrotincta. Regardless of the taxonomy of the lichen, the cause of the red coloration remains unclear and requires further investigation to determine whether it results from mite activity (adults and/or juveniles) or whether it is a phenotypic feature unrelated to mites.
In order to answer this question, we investigated the oribatid mite fauna of three morphologically similar Cladonia species, namely C. coniocraea, C. norvegica, and C. rubrotincta, collected in different European countries. These three lichens occupy similar habitats. While Cladonia norvegica and C. rubrotincta share the same profile of secondary metabolites and differ in the red pigment and smaller podetia in C. rubrotincta (Vtípilová et al. 2025), the third species, C. coniocraea, differs by its larger thallus size and the presence of different secondary metabolites. Specifically, we aimed (1) to document and compare the oribatid mite fauna present in all studied Cladonia species, (2) to assess if red coloration of C. rubrotincta is correlated with any mite activity, e.g. foraging adults or endophagous juveniles, and in case of a correlation (3) to observe and describe the respective activity, and (4) to describe the morphology of endophagous immature stages of oribatid mites developing in the lichen C. rubrotincta.
Materials and methods
Sample locations
The studied material is comprised of the lichens Cladonia coniocraea, C. norvegica and C. rubrotincta and their associated mites from Austria, Czech Republic, Great Britain and Norway. Samples were mostly collected by the authors of this paper and thus they are not explicitly mentioned here, other collectors are given in the following list.
Austria:
- Soboth, Styria; Cladonia rubrotincta growing on tree stumps in a mixed forest (Fagus sylvatica, Acer pseudoplatanus, Picea abies, Pinus sylvestris, Abies alba); 1168 m a.s.l.; 2 Oct. 2022 and 17 Jun. 2023; leg. M. Liebmann-Reindl; coordinates: 46°40.8560′N 15°2.6190′E and 46°40.9170′N 15°2.4700′E.
- Eibiswald, Styria; Cladonia rubrotincta growing on tree stumps of dead trees and on tree trunks of living trees in a mainly coniferous forest (Pinus sylvestris, Picea abies); ca. 1300 m a.s.l.; 18 May 2022 and 17 Jun. 2023; leg. M. Liebmann-Reindl and P. Resl; coordinates: 46°42.9511′ N 15°4.5732′ E and 46°44.7370′N 15°6.1570′E.
- Hartelsgraben, National Park Gesäuse, Styria; Cladonia rubrotincta growing on deadwood in a mixed forest (Picea abies, Pinus sylvestris, Fagus sylvatica); ca. 1000 m a.s.l.; 24 Aug. 2022; leg. M. Liebmann-Reindl and P. Resl; coordinates 47°35.2916′ N 14°42.3683′ E and 47°35.3500′ N 14°42.3766′ E.
- Eibegg, Styria; Cladonia rubrotincta growing on rotten trees and tree stumps in mixed forest (Fagus sylvatica, Larix decidua, Corylus colurna); ca. 1035m a.s.l.; 25 Sep. 2022; leg. M. Liebmann-Reindl; coordinates: 47°25.2091′ N 15°27.1051′ E.
- Schöckl, Styria; Cladonia rubrotincta and C. coniocraea growing on rotten tree stumps in a mixed forest (Picea abies, Larix decidua, Pinus sylvestris, Abies alba); ca. 1135 m a.s.l.; 16 Jun. 2022, 29 Sep. 2023, 15 Nov. 2024; coordinates: 47°12.2360′ N 15°29.0250′ E and 47°12.2809′ N 15°29.0530′ E.
- Weinebene, Styria; Cladonia rubrotincta and C. coniocraea growing on the stump of Picea abies; ca. 1715 m a.s.l.; 9. Aug. 2023; coordinates: 46°50.5054′N 15°1.2372′E.
- Weinebene, Styria; Cladonia rubrotincta growing on the dead wood; ca. 1715 m a.s.l.; 9. Aug. 2023; coordinates: 46°50.4693′N 15°1.3304′E.
- Fjell, Enebakk, Viken; Cladonia rubrotincta and C. coniocraea growing on lying dead wood; ca. 235 m a.s.l.; 13 Oct. 2022; leg. Einar Timdal and authors; coordinates:
59°47.6520′N 10°58.8073′E. - Fjell, Enebakk, Viken; Cladonia rubrotincta and C. coniocraea growing on the dead wood; ca. 235 m a.s.l.; 13 Oct. 2022; leg. Einar Timdal and authors; coordinates:
59°47.6342′N 10°58.7735′E. - Nordre Vinholtjern, Enebakk, Viken; Cladonia rubrotincta and C. coniocraea growing on base of Betula sp. and on the lying wood; ca. 295 m a.s.l.; 13 Oct. 2022; leg. Einar Timdal and authors; coordinates: 59°47.3018′N 11°0.5039′E.
- Nordre Vinholtjern, Enebakk, Viken; Cladonia rubrotincta and C. coniocraea growing on base of Betula sp.; ca. 295 m a.s.l.; 13 Oct. 2022; leg. Einar Timdal and authors; coordinates: 59°47.2445′N 11°0.5826′E.
- Nordre Vinholtjern, Enebakk, Viken; Cladonia rubrotincta and C. coniocraea growing on the dead wood; ca. 300 m a.s.l.; 13 Oct. 2022; leg. Einar Timdal and authors; coordinates: 59°47.0467′N 11°0.9010′E.
- Lundamo, lake Sokna, Trøndelag; Cladonia rubrotincta and C. coniocraea growing on base of Betula sp.; ca. 255 m a.s.l.; 19 Jun. 2023; coordinates: 63°8.0446′N 10°23.7107′E.
- Trondheim - Østbyen, Løkkstad, Trøndelag; Cladonia rubrotincta and C. coniocraea growing on lying dead wood of Picea abies; ca. 205 m a.s.l.; 20 Jun. 2023; coordinates: 63°21.4704′N 10°33.5338′E.
- Lake Benna, between Mt. Loåsen and brook Loa, Trøndelag; Cladonia norvegica and C. coniocraea growing on the dead wood; ca. 190 m a.s.l.; 19 Jun. 2023; coordinates: 63°11.7375′N 10°15.1328′E.
- Hommelvik, Trøndelag; Cladonia norvegica and C. coniocraea growing on the dead wood; ca. 275 m a.s.l.; 19 Jun. 2023; coordinates: 63°22.2281′N 10°54.4596′E.
- Brøttem, Trøndelag; Cladonia norvegica and C. coniocraea growing on the dead wood; ca. 170 m a.s.l.; 20 Jun. 2023; coordinates: 63°14.9939′N 10°30.9911′E.
- Løkkstad, Trøndelag; Cladonia norvegica and C. coniocraea growing on a base of Picea abies; ca. 200 m a.s.l.; 20 Jun. 2023; coordinates: 63°21.4704′N 10°33.5338′E.
- Stjørdal, Hegra, Trøndelag; Cladonia norvegica and C. coniocraea growing on the dead wood; ca. 185 m a.s.l.; 21 Jun. 2023; coordinates: 63°26.7254′N 11°5.4952′E.
- Novohradské hory Mts.; Cladonia rubrotincta growing on fallen trunk of spruce; ca. 750 m a.s.l.; 29. Oct. 2022; leg. David Svoboda; coordinates: 48°39.9594′N 14°42.0396′E.
- Šumava Mts., Černý Kříž, Volary; Cladonia rubrotincta growing on a dead wood of the coniferous tree in wet spruce forest (with occasional pines and birches); ca. 740 m a.s.l.; 30. Dec. 2021; leg. Zdeněk Palice; coordinates: 48°51.6250′N 13°51.9950′E.
- Šumava Mts., Vydra River Valley; Cladonia rubrotincta growing on the dead wood (spruce) in the spruce forest; ca. 895 m a.s.l.; 24. Sep. 2022; coordinates: 49°4.7358′N 13°30.4118′E.
- Kinlochewe, Scotland; Cladonia rubrotincta growing on a piece of decaying wood covered in mosses in wet mixed forest (Betula, Fraxinus, Pinus); ca. 60 m a.s.l.; 22. Sep. 2023; leg. Lukáš Janošík; coordinates: 57°36.2436′N 5°17.5380′W.
Norway:
Czech Republic:
Great Britain:
Sampling procedure
The lichen was removed with a knife and afterwards transported to the laboratory for the extraction and for observation of mites. The former was achieved by placing the patches of lichen in Berlese-Tullgren funnels for 2-7 days (except for the Scottish samples where mites were extracted manually with a fine brush). Extracted mites (adults) were preserved in pure ethanol and part of them then prepared in permanent slides, using Berlese-mountant, and the rest was prepared in temporary cavity slides using pure lactic acid for identification under the light microscope (Olympus BH-2, Motic DMBA 310, Leitz-Wetzlar SM-LUX).
Observations and drawings
Red colored lichen thallus was cut open with a razorblade or a fine scalpel to check the content. Observations of extracted living juvenile specimens were performed under a stereomicroscope (Leica Wild M8) and photographic documentation was conducted with a Keyence VHX-7000 digital microscope. For graphic depictions, juvenile specimens were embedded in Berlese mountant and drawings were performed with an Olympus BH-2 Microscope equipped with a drawing attachment (camera lucida). Drawings were scanned and afterwards processed and digitized with the free and open-source vector graphics editor Inkscape (https://inkscape.org ![]() ), finally they were further modified with Adobe Photoshop CS.
), finally they were further modified with Adobe Photoshop CS.
Molecular genetic work
For taxonomic identification of juvenile specimens, we used molecular genetic sequence data. We subjected individual juvenile mites, extracted from inside lichen thalli, to direct PCR without prior DNA extraction. PCR amplification, purification of the PCR product and sequencing followed previously published methods (Schäffer et al. 2008). Fragments were purified using the Big Dye XTerminator kit (Thermo Fisher Scientific) and visualized on a 3500XL capillary sequencer (Applied Biosystems). We sequenced the 28S rRNA D3 gene fragment, which is known as a reliable marker for oribatid mite species identification (Lehmitz and Decker 2017), using the standard primers D3A and D3B (Litvaitis et al. 1994). Sequences were aligned manually in MEGA v6. (Tamura et al. 2013) and then verified by comparison with reference sequences from NCBI GenBank. Sequences were also deposited in NCBI GenBank (accession nrs.: PV990091-PV990107).
Statistics based on adult mite fauna
Statistical analyses were performed in the Jamovi software, Version 2.2 (R Core Team 2021; The Jamovi Project 2022) with the package car vers. 3.1-3 (Fox and Weisberg 2020).
The Shapiro-Wilk test for normality was performed for the total number of adult individuals and the number of mite species in individual samples, categorized by lichen species (Cladonia rubrotincta, C. norvegica and C. coniocraea), along with Levene's test for homogeneity of variances. For normally distributed data with equal variances, we used classical (Fisher's) one-way ANOVA. For data deviating significantly from normality, we used Kruskal-Wallis test, a non-parametric variant of one-way ANOVA. For data with unequal variances, we used Welch's modification of one-way ANOVA. We visualized the data with box and violin plots.
General remarks
Mite species lists for different Cladonia species are based on material collected from Austrian and Norwegian locations (1-7, 8-19) and all found species were included in the statistical analyses. For the analysis of the content of the red-colored lichen parts only samples from locations 5, 21-23 were used, and juveniles from locations 3-6, 8, 12-14 and 20 were used for taxonomic identification based on molecular genetic sequence data. Czech samples (20-22) were only used for the investigation of red spots and for molecular genetic sequencing of juveniles and the lichen as well (the latter was part of another study).
Results
The oribatid mite fauna of three different Cladonia members
In total, 61 different oribatid mite species
(Supplementary Table S1 ![]() )
could be identified in all lichen samples. Species with less than four individuals in all samples together were regarded as incidental by-catch and thus we do not address them in the results and discussion sections. The remaining taxa comprised 28 different oribatid mite species from 23 genera and 18 families from nineteen different locations in Austria and Norway. The faunal composition of Cladonia rubrotincta, C. coniocraea and C. norvegica was almost identical in the Norwegian samples, with 12 to 15 different species occurring in each lichen (Table 1). The majority of the species was present in all three lichens in Norway (Table 1), very few, i.e. Suctobelba regia, Suctobelbella arcana, Oribatula tibialis, Chamobates cuspidatus and Tectocepheus velatus sensu lato (based on morphological discrepancies, this species could not be assigned to any known subspecies), were only present in two of the lichens and Eueremaeus cf. valkanovi could only be detected in C. coniocraea. The most abundant species were Carabodes areolatus, C. labyrinthicus, C. marginatus, C. subarcticus, Melanozetes mollicomus and Neobrachychthonius marginatus magnus. In Austria, the faunal composition of C. rubrotincta and C. coniocraea showed specific overlaps but the number of species found in the former lichen was five times higher than in the latter. All the five species present in C. coniocraea were also common in C. rubrotincta. The most abundant species in the Austrian C. rubrotincta samples were Carabodes areolatus, C. labyrinthicus, C. marginatus, C. ornatus, Hermannia gibba, Caleremaeus lignophilus and Liacarus coracinus. A comparison of lichen samples from Norway and Austria revealed a strong overlap in the most abundant species. Carabodes areolatus, C. labyrinthicus, C. marginatus and Melanozetes mollicomus occurred regularly in each lichen in both countries. Carabodes subarcticus, on the other hand, which frequently occurred in all Norwegian lichen samples, was absent in all Austrian samples and the same applies to Hermannia gibba but the other way round.
)
could be identified in all lichen samples. Species with less than four individuals in all samples together were regarded as incidental by-catch and thus we do not address them in the results and discussion sections. The remaining taxa comprised 28 different oribatid mite species from 23 genera and 18 families from nineteen different locations in Austria and Norway. The faunal composition of Cladonia rubrotincta, C. coniocraea and C. norvegica was almost identical in the Norwegian samples, with 12 to 15 different species occurring in each lichen (Table 1). The majority of the species was present in all three lichens in Norway (Table 1), very few, i.e. Suctobelba regia, Suctobelbella arcana, Oribatula tibialis, Chamobates cuspidatus and Tectocepheus velatus sensu lato (based on morphological discrepancies, this species could not be assigned to any known subspecies), were only present in two of the lichens and Eueremaeus cf. valkanovi could only be detected in C. coniocraea. The most abundant species were Carabodes areolatus, C. labyrinthicus, C. marginatus, C. subarcticus, Melanozetes mollicomus and Neobrachychthonius marginatus magnus. In Austria, the faunal composition of C. rubrotincta and C. coniocraea showed specific overlaps but the number of species found in the former lichen was five times higher than in the latter. All the five species present in C. coniocraea were also common in C. rubrotincta. The most abundant species in the Austrian C. rubrotincta samples were Carabodes areolatus, C. labyrinthicus, C. marginatus, C. ornatus, Hermannia gibba, Caleremaeus lignophilus and Liacarus coracinus. A comparison of lichen samples from Norway and Austria revealed a strong overlap in the most abundant species. Carabodes areolatus, C. labyrinthicus, C. marginatus and Melanozetes mollicomus occurred regularly in each lichen in both countries. Carabodes subarcticus, on the other hand, which frequently occurred in all Norwegian lichen samples, was absent in all Austrian samples and the same applies to Hermannia gibba but the other way round.
Download as
Cladonia rubrotincta
Cladonia coniocraea
Norway (12 localities)
N
Norway (12 localities)
N
Carabodes labyrinthicus
37
Carabodes labyrinthicus
31
Carabodes areolatus
71
Carabodes areolatus
92
Carabodes marginatus
52
Carabodes marginatus
40
Carabodes subarticus
5
Carabodes subarticus
31
Melanozetes mollicomus
10
Melanozetes mollicomus
1
Phthiracarus sp.
1
Phthiracarus sp*.
2
Tectocepheus velatus velatus
3
Tectocepheus velatus velatus
4
Zygoribatula exilis
3
Zygoribatula exilis
1
Lagenobates lagenulus
2
Lagenobates lagenulus
12
Neobrachychthonius marginatus magnus
29
Neobrachychthonius marginatus magnus
22
Suctobelba regia
1
Suctobelba regia
4
Tectocepheus velatus sensu lato
3
Chamobates cuspidatus
1
Oribatula tibialis
1
Oribatula tibialis
6
Caleremaeus sp.
2
Eueremaeus cf. valkanovi
1
Suctobelbella arcana
2
Suctobelbella arcana
2
Austria (7 localities)
Austria (2 localities)
Carabodes areolatus
150
Carabodes areolatus
1
Carabodes labyrinthicus
54
Carabodes labyrinthicus
6
Carabodes marginatus
131
Caleremaeus lignophilus*
2
Carabodes ornatus
45
Hermannia gibba
6
Hermannia gibba
90
Ceratoppia bipilis
1
Odontocepheus elongatus
6
Liacarus coracinus
41
Cladonia norvegica
Mycobates carli
11
Norway (7 localities)
N
Mesoplophora pulchra
7
Carabodes labyrinthicus
11
Steganacarus applicatus
4
Carabodes areolatus
52
Caleremaeus lignophilus
46
Carabodes marginatus
21
Melanozetes mollicomus
9
Carabodes subarticus
9
Fuscozetes setosus
15
Melanozetes mollicomus
6
Eupelops torulosus
2
Phthiracarus sp.
1
Mesotritia nuda
4
Tectocepheus velatus velatus
5
Ceratoppia bipilis
5
Zygoribatula exilis
7
Neobrachychthonius marginatus magnus
1
Lagenobates lagenulus
1
Suctobelba regia
1
Neobrachychthonius marginatus magnus
2
Phthiracarus sp.
1
Chamobates cuspidatus
9
Eueremaeus cf. valkanovi
3
Tectocepheus velatus sensu lato
1
A note about Carabodes willmanni in Scotland
Observations on six mites collected manually from C. rubrotincta in Scotland (Locality 23), showed that only a single species of Carabodes was present in this sample, namely C. willmanni, while all the other common Carabodes species, like C. labyrinthicus, C. areolatus and C. marginatus, found in other European countries were absent. Interestingly Carabodes willmanni was absent from the lichen samples from all the other investigated European countries.
Statistical analysis of the oribatid mite fauna of the three investigated Cladonia members
The Shapiro-Wilk normality test showed that species numbers followed a statistically normal distribution, whereas the numbers of individuals deviated significantly from normality (p< 0.004), which may indicate an aggregated distribution (i.e. individual lichen samples contained very different numbers of mite individuals). The test for homogeneity of variances was non-significant except for the number of species of oribatid mites with endophagous juveniles (Carabodes+Mycobates; p = 0.009).
Results of the one-way ANOVA analyses showed that the mean species richness and abundance are similar across the different lichens (p=0.559 and p=0.529 respectively). Cladonia rubrotincta and C. norvegica showed slightly higher average species numbers than Cladonia coniocraea (Figure 1A). Although one sample of C. coniocraea contained almost 100 mite individuals, the majority of samples of all lichen comprised less than 50 mite specimens (Figure 1B).
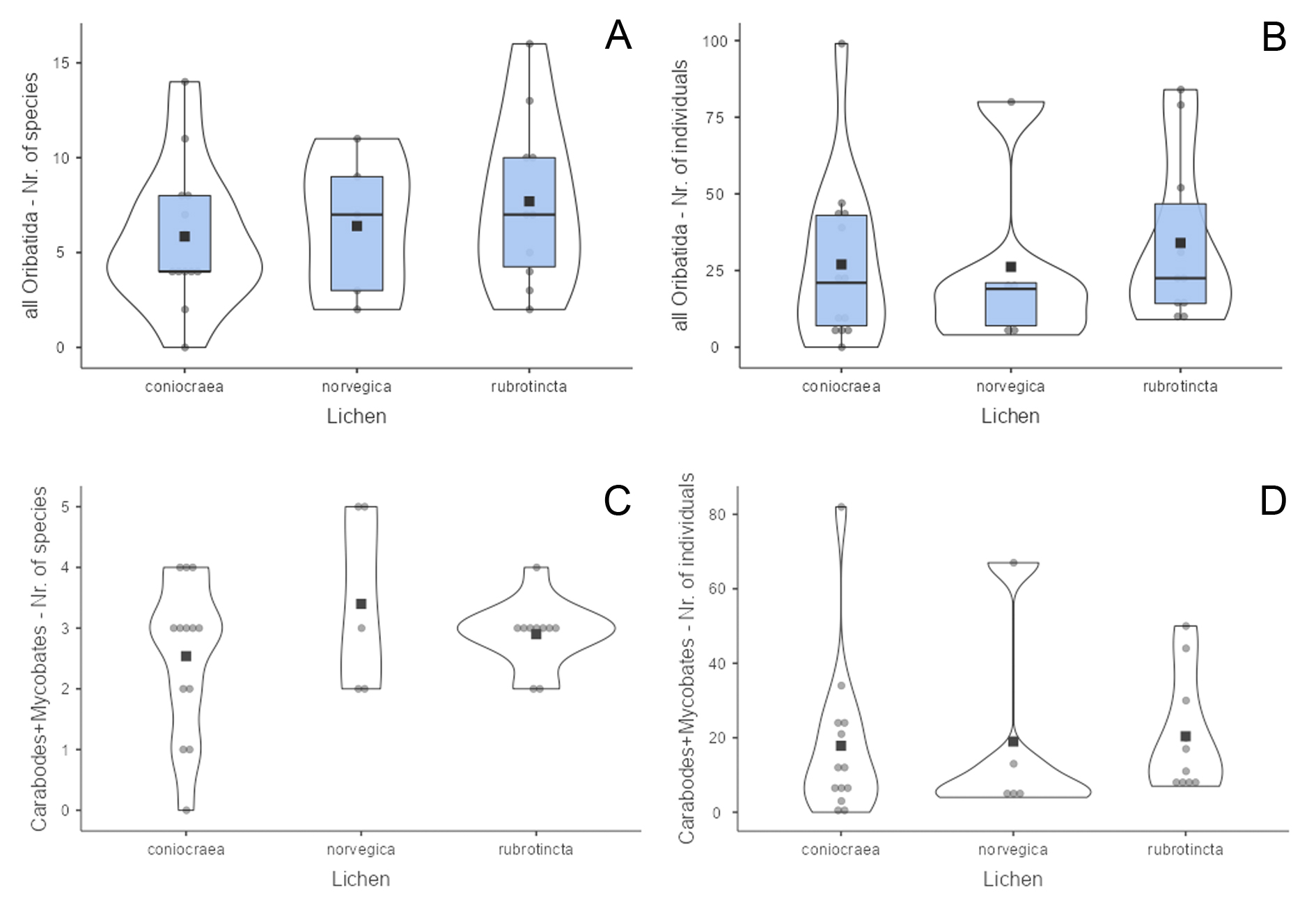


When taking into account only the numbers of Carabodes and Mycobates species (which have endophagous juveniles in the lichen), the three Cladonia species also do not differ significantly (p=0.516 and p=0.429). The lichens contain in general three species with an average of 20 specimens (Figures 1C, 1D).
Observation of endophagous behaviour in Cladonia rubrotincta
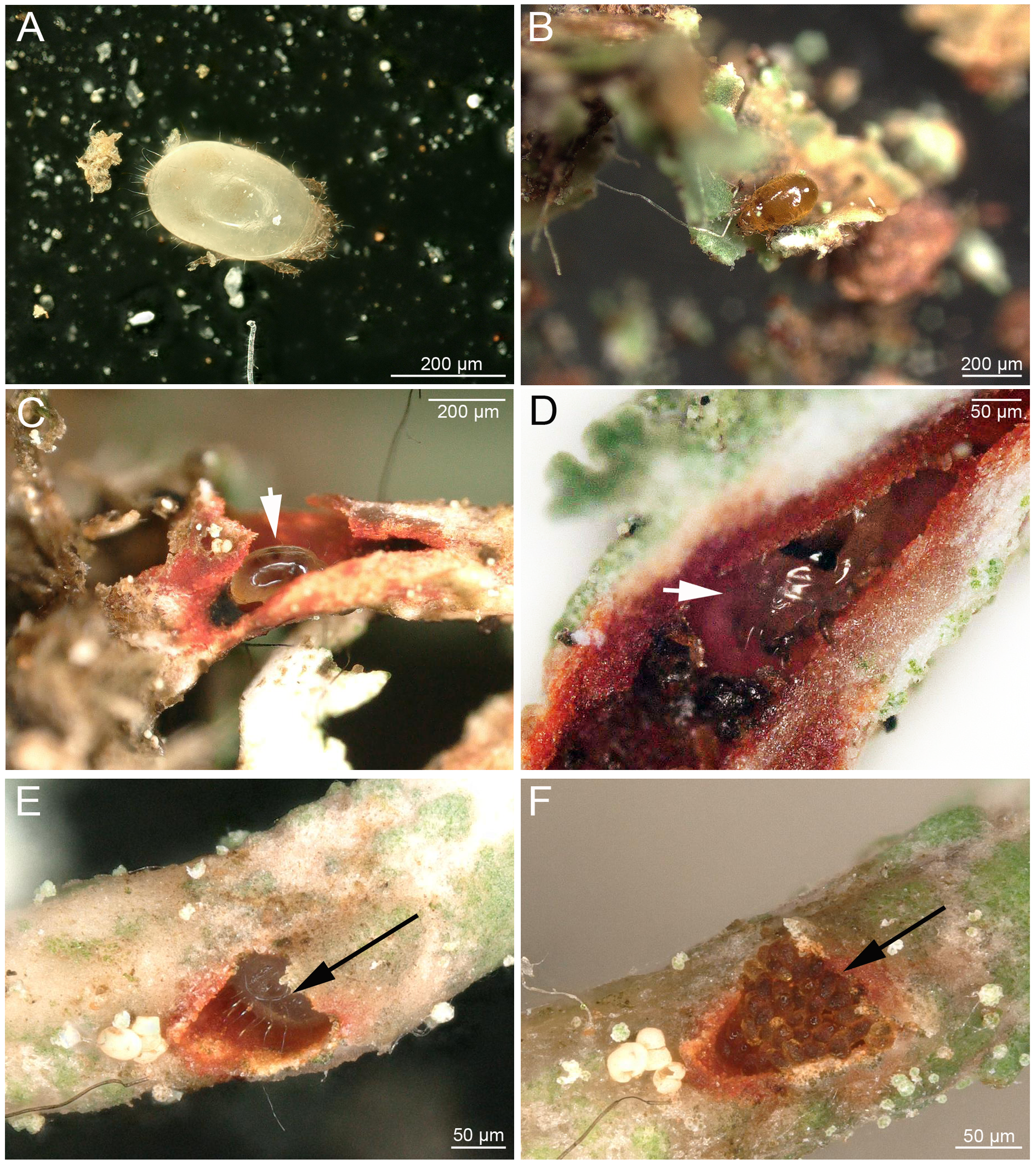


To study the nature of the red spots on the lichen C. rubrotincta, fresh samples were brought to the lab and the spots were cut open under a stereomicroscope. This investigation revealed the common presence of Carabodes juveniles in the red colored lichen thalli. Contrary to adult mites, these juveniles (Figure 2A) were never observed feeding outside on cortex lichen thallus. They were always found tunneling within the lichen, and they occurred in podetia as well as basal squamules. The resulting tunnels were always enclosed by red colored lichen thallus (Figures 3A, B), but it is unclear if this is caused by the feeding and digging activity, or if juveniles just prefer red colored thalli. However, juvenile stages were never found in green lichen thalli. From 208 specifically investigated red spots on C. rubrotincta from four different countries, 24% did not show any sign of endophagous activity, 50% did show an external feeding hole (Figures 3C, D) and either contained fecal pellets or a juvenile specimen (Figures 2C, D; Table 2) or both. About 14% of hollow red spots contained only fecal pellets and 12% were hollow but empty. In two single cases, the hollows harbored juvenile specimens although an external feeding hole or any other artificial entry passage could not be detected. Most of numbers of endophagous mites do not differ significantly between C. rubrotincta from different countries, especially the presence of nymphs and/or fecal pellets is remarkably constant. The hollow but empty spots and the spots without any sign of activity show some variation between the countries, but they represent the minority of cases in all countries. Totally, approximately 75% of all red spots were associated with one or more signs of endophagous activity (hole, pellet, juvenile) (Table 2).



Download as
Features
Austria
%
Norway
%
Czech Rep.
%
Scotland
%
Total
%
Holes + juveniles or pellets
49
45.4
25
50
26
65
4
40
104
50
Just pellets
15
13.9
6
12
7
17.5
1
10
29
13.9
Hollow but empty
4
3.7
12
24
5
12.5
4
40
25
12
Nothing - just red coloration
40
37
7
14
2
5
1
10
50
24
Total
108
50
40
10
208
Carabodes juveniles were also observed to use their own fecal pellets, like bricklayers, to completely close holes in the lichen (Figures 2E, F). When the burrow was cut open during the investigation, juveniles often tried to escape by swiftly moving deeper into the lichen tissue. When removed from the burrows, they walked rather clumsily outside the lichen. In two cases, adults of Carabodes areolatus were found enclosed in feeding cavities, one of them still carrying parts of the tritonymphal skin.
Juveniles of Mycobates carli (Figure 2B) were also found to be closely associated with Cladonia rubrotincta, although very rarely observed in hollow burrows and podetia inside the lichen thallus. In most cases, they were located in hollow spaces between the lower cortex, anchoring hyphae and the underlying substrate of the lichen. They could be easily revealed by detaching the lichen from the substrate with a forceps. Adults of M. carli were only foraging outside the lichen thallus and were never found in burrows or in the hollow spaces between lichen and underlying substrate.



Our detailed visual investigation of single lichen parts also revealed several mite eggs. These were mostly placed in the red colored hollow cavities (Figure 4A) or embedded in holes or deepenings of the lichen thalli (Figure 4B). Based on their placement within the lichen thallus, we conclude that they belong to Carabodes and not to Mycobates.
As larvae and nymphs tunneling in C. rubrotincta could not be determined to species level due to a lack of prior knowledge about specific juvenile morphology, we performed molecular genetic investigations to infer their taxonomic identity. Nuclear 28S gene fragment D3 analysis of juvenile specimens, extracted manually from the lichen, showed that only four oribatid species use Cladonia rubrotincta for rearing purposes. These species were Carabodes areolatus, C. labyrinthicus, C. marginatus and Mycobates carli (Table 3). In Austrian samples we detected all these four species, whereas in Norwegian samples we found C. marginatus and M. carli, and in Czech samples there was only C. marginatus present.
Download as
Species
ID DNA
Location
Country
D3
Carabodes areolatus
MLR-06
Schöckl
Austria
PV990102
Carabodes labyrinthicus
MLR-07
Schöckl
Austria
PV990103
MLR-08
Schöckl
Austria
PV990104
Carabodes marginatus
MLR-03
Hartelsgraben
Austria
PV990101
MLR-09
Schöckl
Austria
PV990105
vv4-1g
Nordre Vinholtjern
Norway
PV990096
vv3-1c
Fjell
Norway
PV990098
vv13-1c
Novohhradské hory Mts.
Czech Republic
PV990097
Mycobates carli
MLR-01
Eibegg
Austria
PV990099
MLR-02
Eibegg
Austria
PV990100
MLR-10
Schöckl
Austria
PV990106
MLR-12
Schöckl
Austria
PV990107
N28a
Weinebene
Austria
PV990094
N28b
Weinebene
Austria
PV990091
N28Aa
Weinebene
Austria
PV990092
N28Ab
Weinebene
Austria
PV990093
TR7CFb
Løkkstad
Norway
PV990095
Based on this information, we could identify M. carli juveniles unambiguously. Identifying juveniles of each Carabodes species based on morphology, on the other hand, was not feasible and thus the observations mentioned above and the following descriptions should be treated as genus specific rather than species specific.
Description of juvenile morphological features
Family Mycobatidae Grandjean, 1954
Genus Mycobates Hull, 1916
Mycobates carli Schweizer, 1922
Common features of juvenile stages
Apheredermous. Integument soft and smooth, opisthonotum with macrosclerite (pygidial shield). Color light brown, prodorsum and pygidial shield slightly darker sclerotized. Body not wrinkled (wrinkles shown in figures represent a shrinking artefact due to preservation). Humeral organ oh present, consisting of elliptic integumental depression lined with fine pores. Bothridial seta bs clavate with acute tip (almost lanceolate), head spinose. Bothridium cup-like with small lateral triangular lamella. Rostrum relatively short not covering mouthparts completely in dorsal view. Gnathosoma no obvious differences to adult stage (details given below for deutonymph but they apply to all juvenile stages). Opisthonotal setae setiform without excentric porose area at base. Centrodorsal setae and seta h1 located on pygidial shield; pygidial shield covering almost entire opisthonotum in dorsal aspect. Legs monodactylous, setae d absent from tibiae and genua when respective solenidion is present. Developmental formulas (adult excluded): setation gastronotic region (larva to tritonymph) 12-15-15-15, genital (protonymph to tritonymph) 1-3-5, aggenital (protonymph to tritonymph) 0-1-1, adanal (protonymph to tritonymph) 0-3-3, anal (larva to tritonymph) 0-0-0-2.
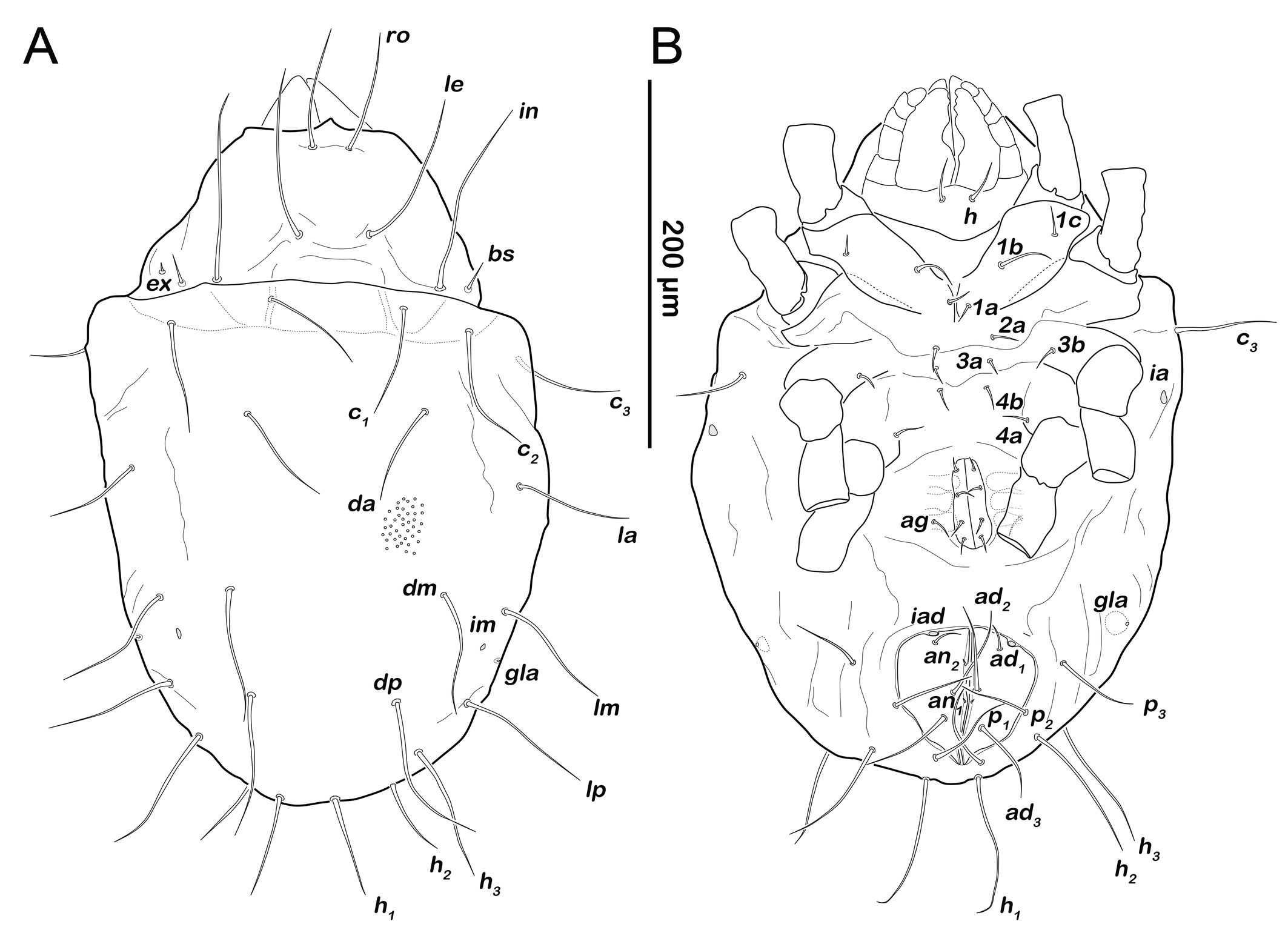
Larva (N=3): length 169–200 µm (mean 190 µm).
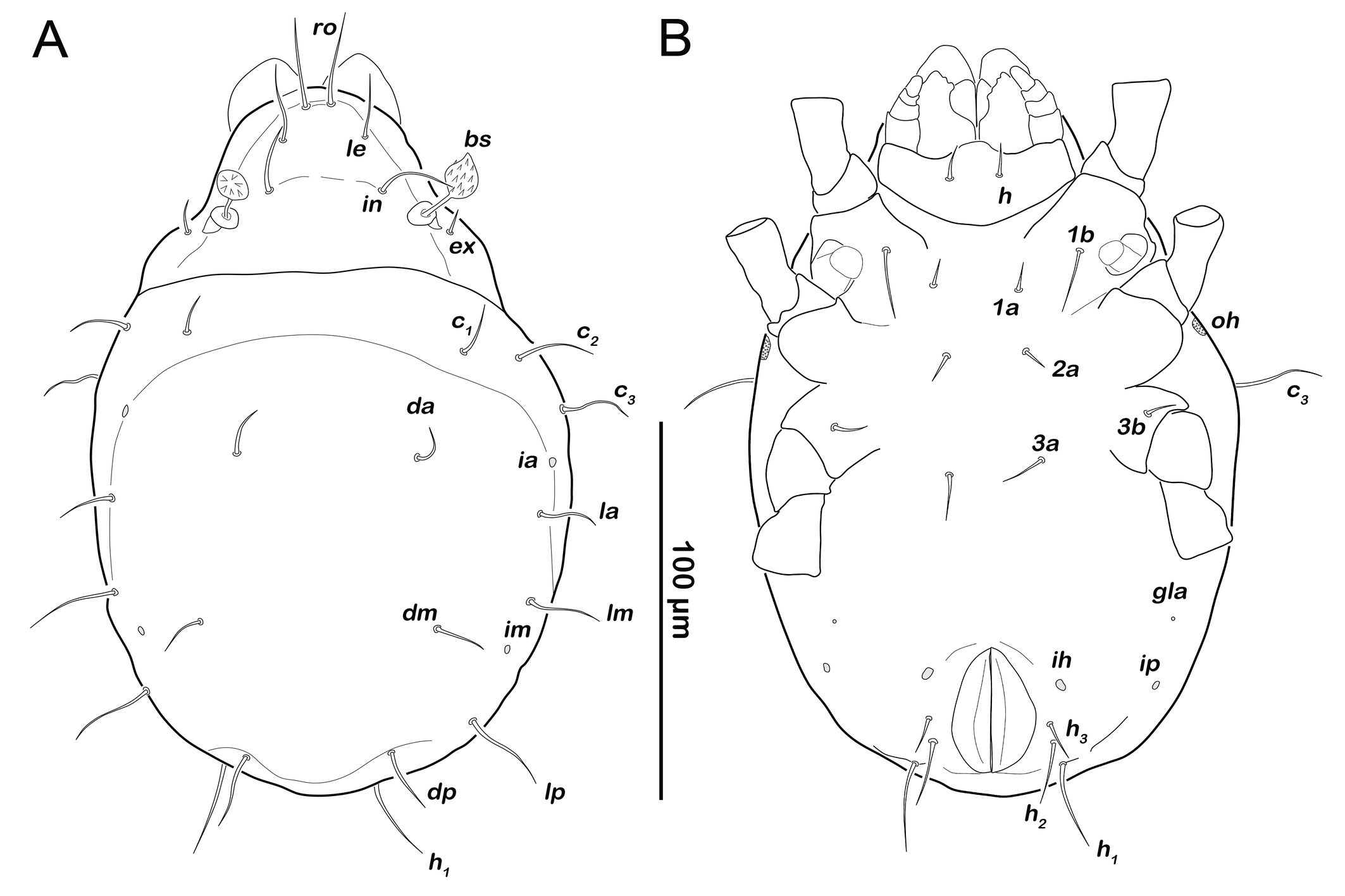


Prodorsum (Figure 5A). Rostral seta ro setiform, smooth and large, projecting rostrum. Lamellar seta le setiform, smooth, approx. same size as ro. Interlamellar seta in also smooth and setiform, slightly longer than other prodorsal setae. Exobothridial seta ex setiform, smooth and short.
Gastronotic region (Figure 5A). Twelve pairs of setiform, smooth gastronotic setae (length 16-25 µm), c1-3 , da, dm, dp, la, lm, lp, h1-3 . Cupules well traceable, ia anterior to seta la, im posterior to seta lm, ih anterior to h3 and ip lateral to the latter. Humeral organ oh lateral on a level with leg II. Opisthonotal gland opening gla lateroventrally, between leg III and cupule ip.
Podosoma and venter (Figure 5B). Epimeral setation 2-1-2, most setae short (approx. 10 µm) and spiniform, seta 1b longest (18 µm). Claparède's organ globular, located on posterior edge of epimeron I. Aggenital, genital, adanal and anal setae not developed.
Legs. Setation and solenidia shown in Table 4.
Download as
Instars
Trochanter
Femur
Genu
Tibia
Tarsus
Chaetome
Solenidia
Leg I
larva
-
d, bv´´
(l), s
(l), v´, φ1
(pv), s, (a), (u), (p), (pl), (tc), (ft), ε, ω1
0-2-2-3-16
1-1-1
protonymph
-
-
-
-
ω2
0-2-2-3-16
1-1-2
deutonymph
-
-
-
φ2
-
0-2-2-3-16
1-2-2
tritonymph
v´
(l)
v´
v´´
(it)
1-4-3-4-16
1-2-2
Leg II
larva
-
d, bv´´
(l), s
l´, v´, φ
(pv), s, (a), (u), (p), (tc), (ft), ω1
0-2-2-2-13
1-1-1
protonymph
-
-
-
-
-
0-2-2-2-13
1-1-1
deutonymph
-
-
-
-
ω2
0-2-2-2-13
1-1-2
tritonymph
v´
(l)
v´
v´´
(it)
1-4-3-3-15
1-1-2
Leg III
larva
-
d, ev´
l´, s
v´, φ
(pv), s, (a), (u), (p), (tc), (ft)
0-2-1-1-13
1-1-0
protonymph
-
-
-
-
-
0-2-1-1-13
1-1-0
deutonymph
-
-
-
-
-
0-2-1-1-13
1-1-0
tritonymph
v´
l´
-
v´´
(it)
1-3-1-2-15
1-1-0
Leg IV
protonymph
-
-
-
-
(pv), (u), (p), ft´´
0-0-0-0-7
0-0-0
deutonymph
-
d, ev´
d, l´
l´, d, φ
s, (a), (tc)
0-2-2-2-12
0-1-0
tritonymph
-
-
-
v´´
-
0-2-2-3-12
0-1-0
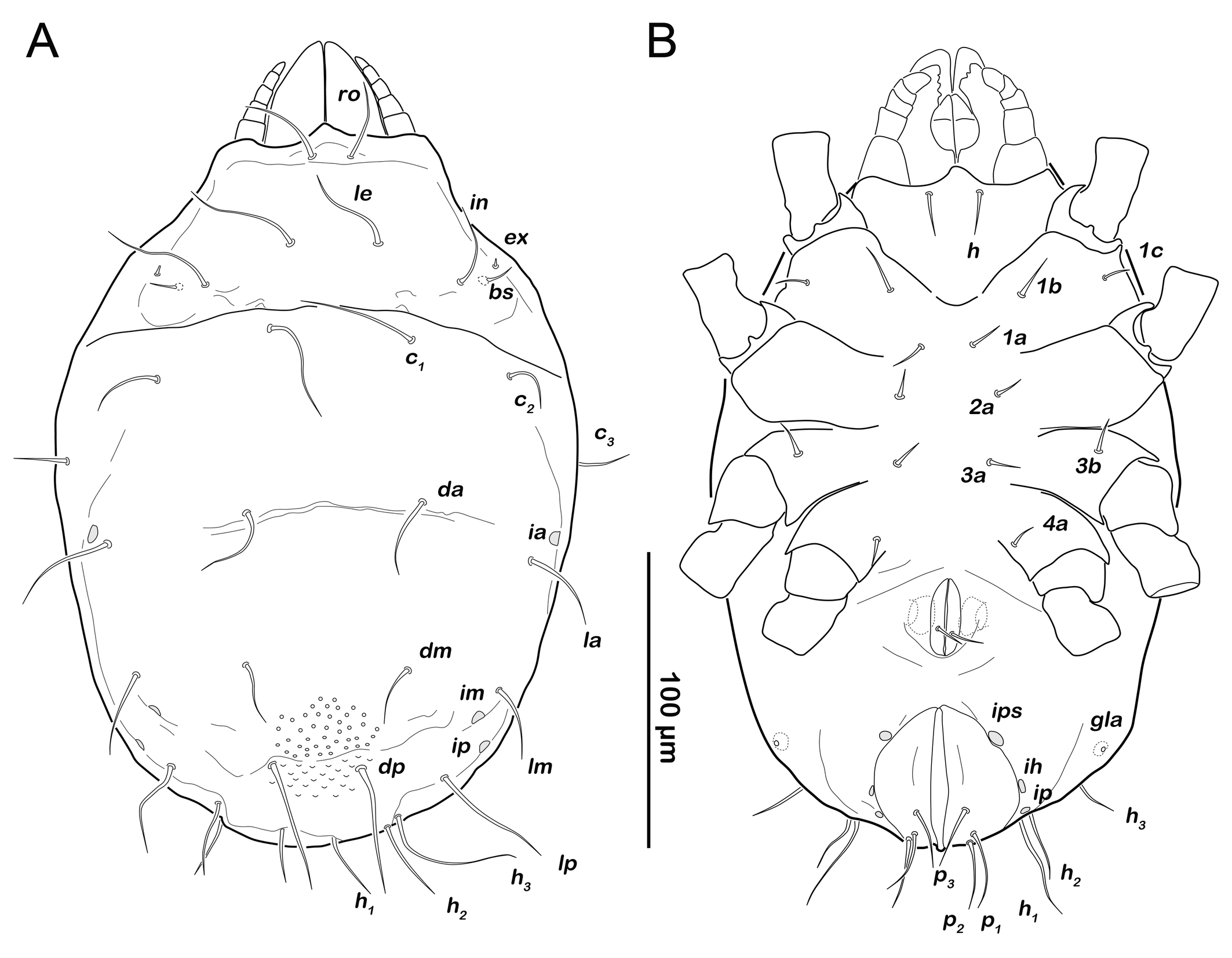
Protonymph (N=7): length 234–286 (mean 262 µm).
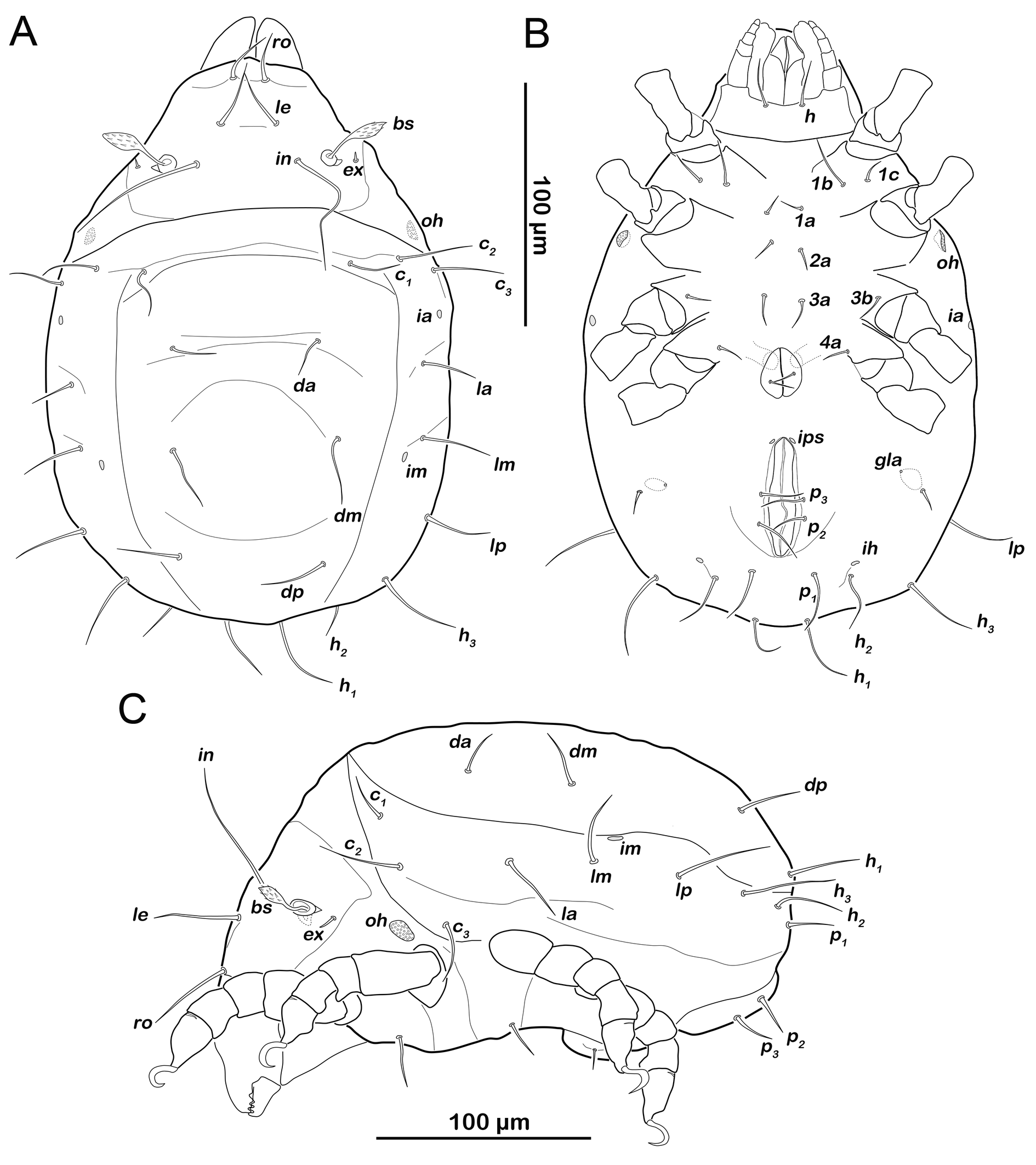


Prodorsum (Figure 6A). Interlamellar seta in (ca. 60 µm) more than twice the length of seta le from this stage.
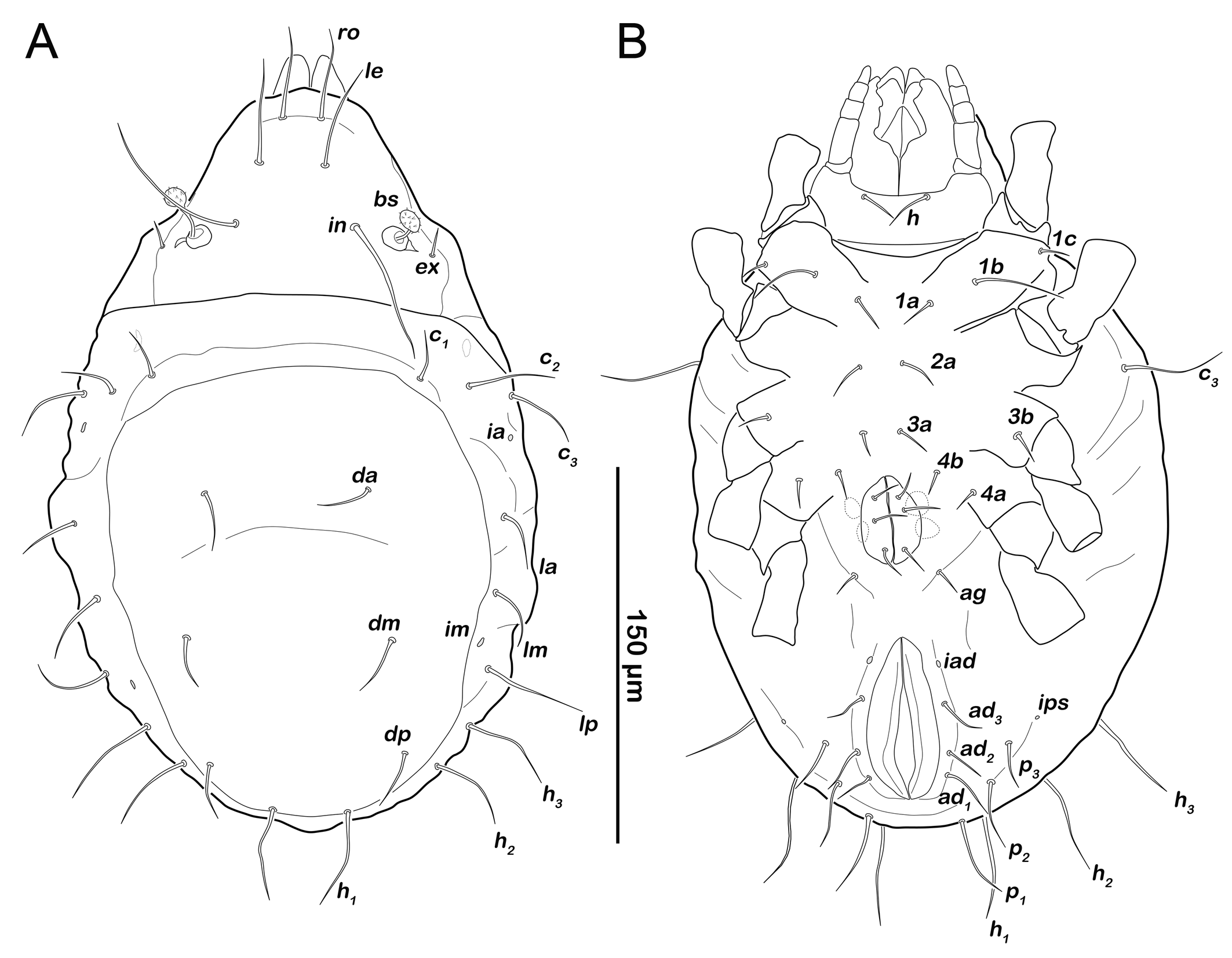
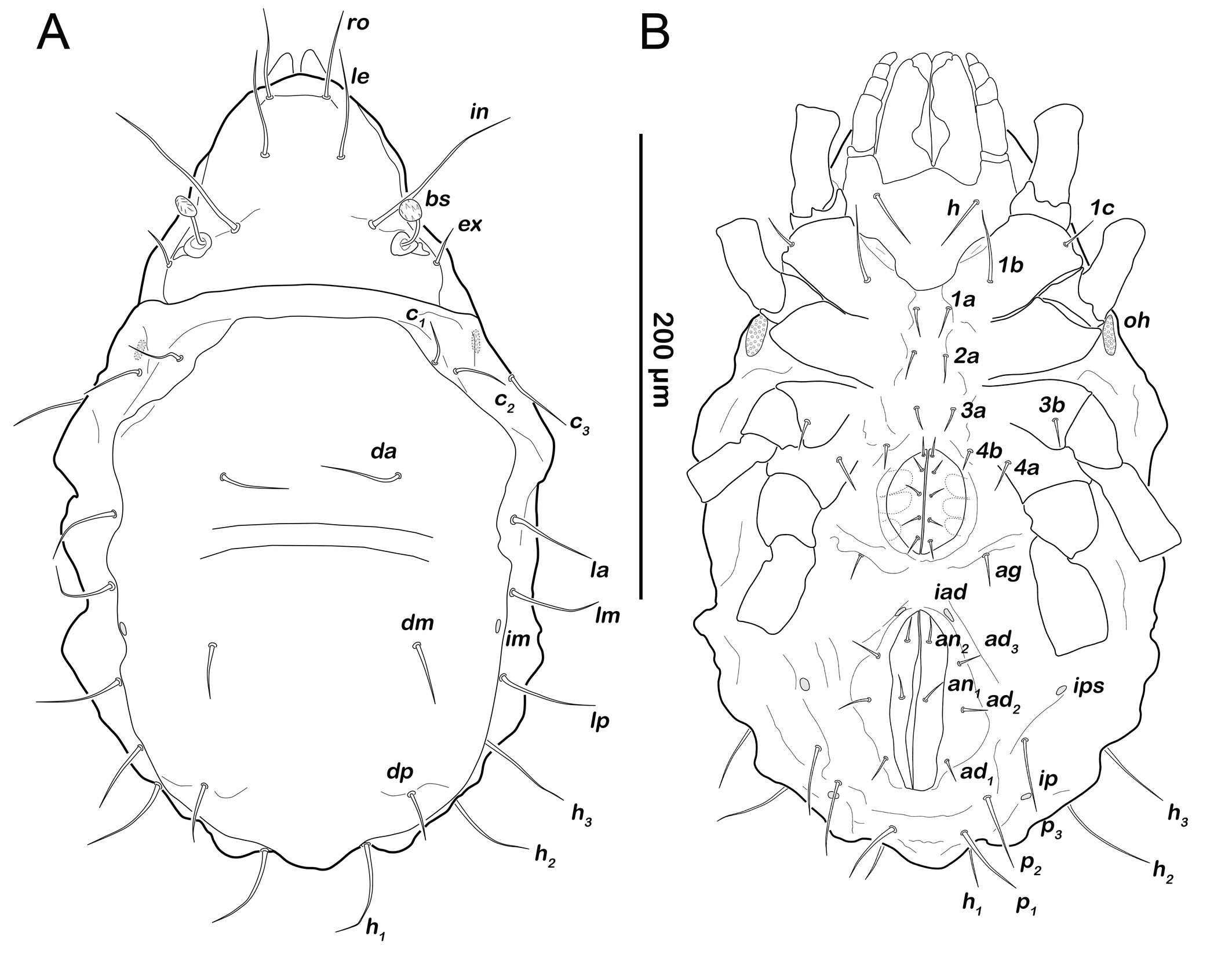
Gastronotic region (Figures 6A, 6C). Fifteen pairs of gastronotic setae (25-36 µm); c1-3 , da, dm, dp, la, lm, lp, h1-3 , p1-3 . Setae p2 and p3 framing anal orifice, seta p1 posterior to anal opening.
Podosoma and venter (Figure 6B). Epimeral setation 3-1-2-1, all setae spiniform (10-20 µm). Seta 1c and 4a added in this stage. One pair of short genital setae. Adanal and anal setae not developed. Cupule ips in paranal position.
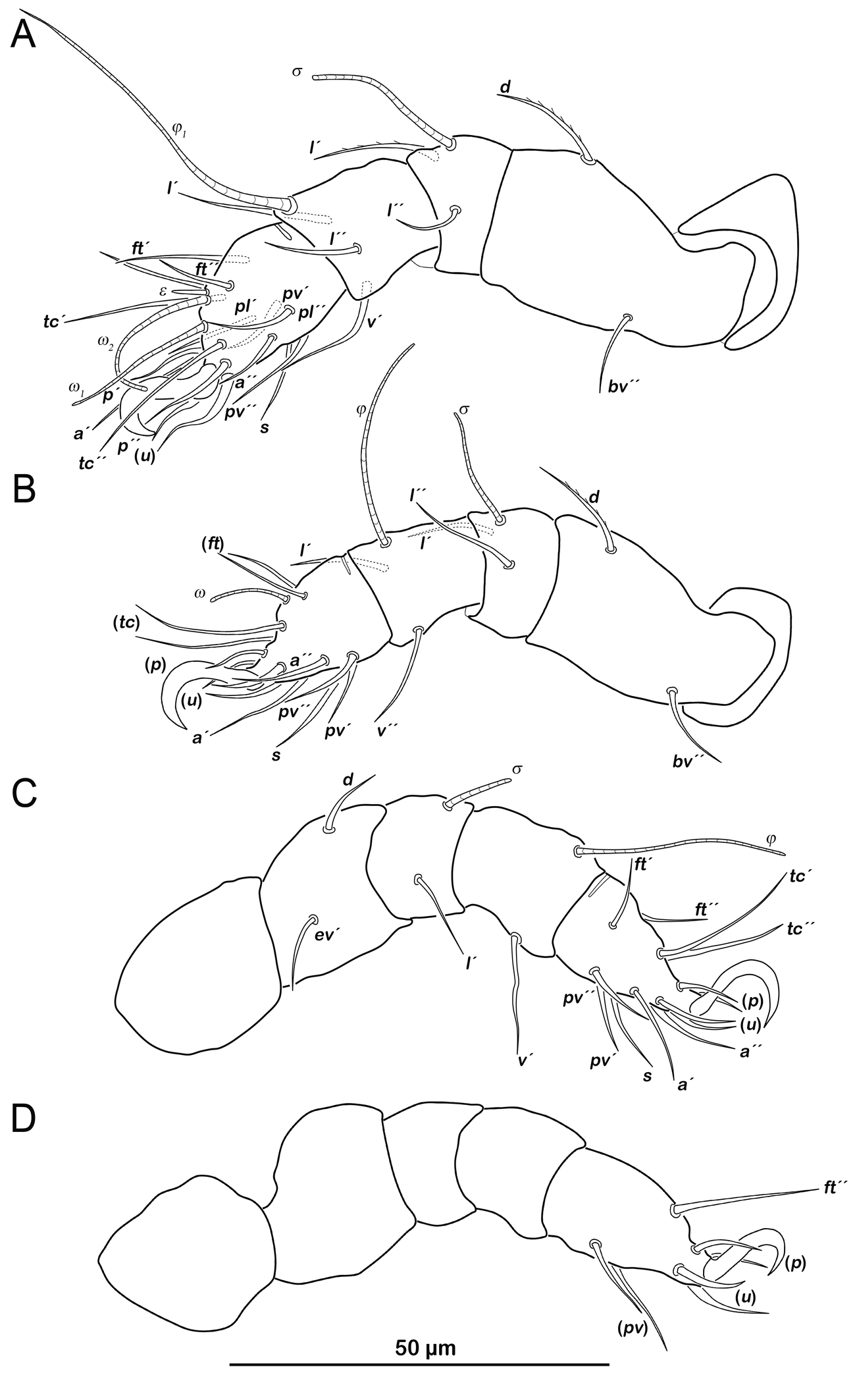


Legs (Figure 7). Setation and solenidia shown in Table 4.
Deutonymph (N=8): length 292–356 µm (mean 315 µm).


Prodorsum (Figure 8A). No difference to protonymph.
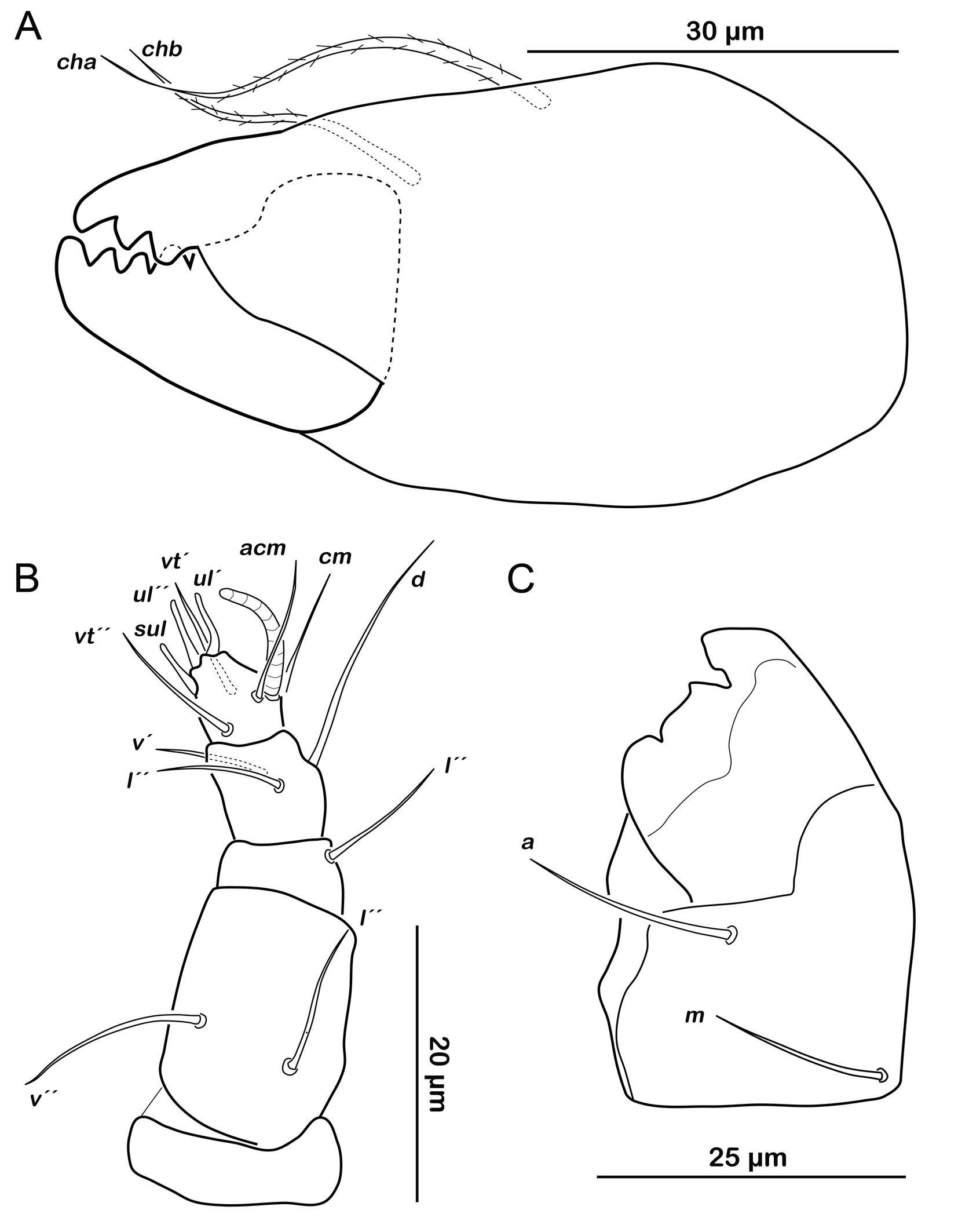
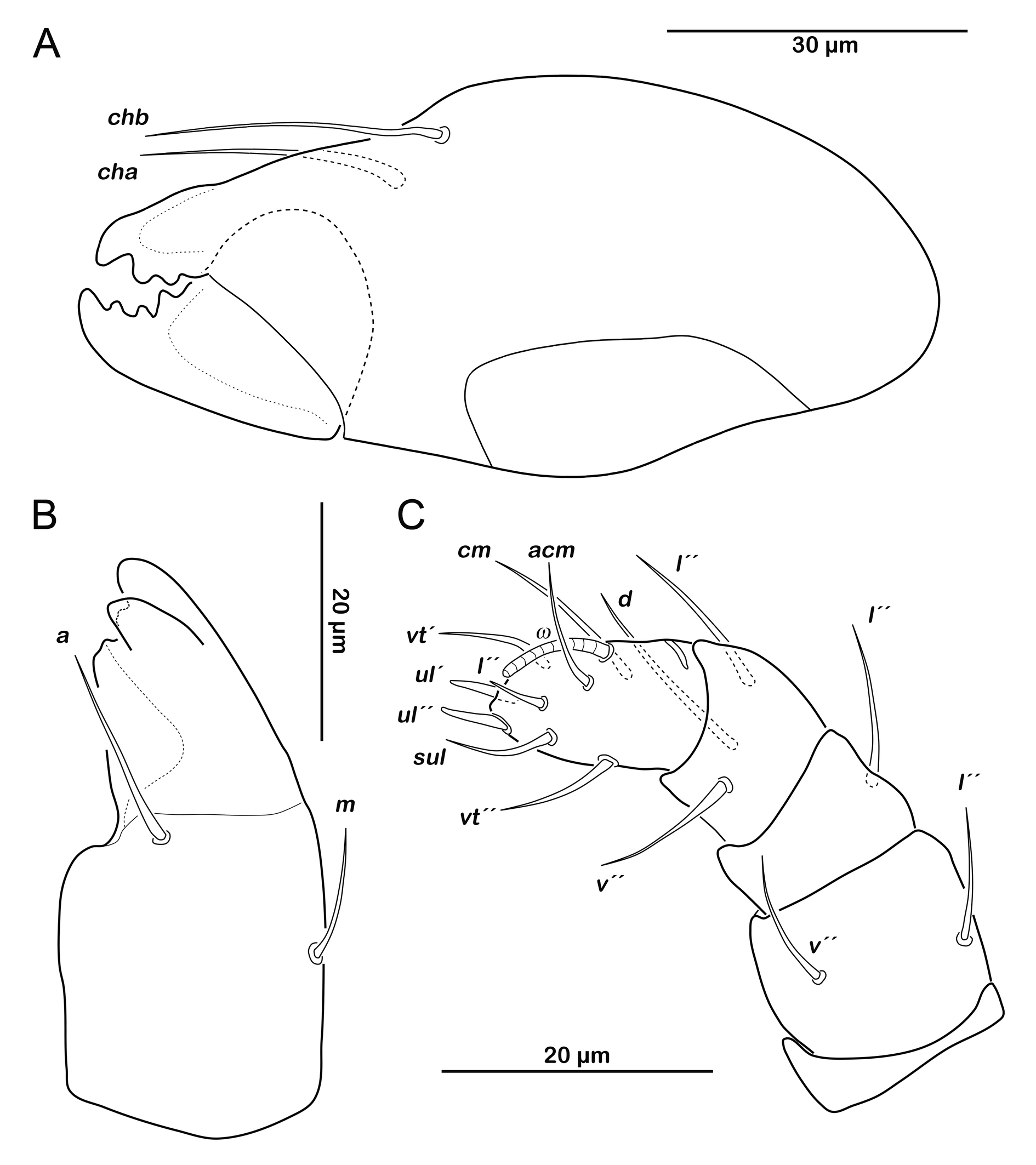
Gnathosoma. Chelicera chelate with strongly sclerotized teeth on fixed and movable digit, setae cha and chb setiform, long (ca. 40 µm) and barbed (Figure 9A). Palp setal formula 0-2-1-3-7 (+ solenidion ω) (Figure 9B). Distal part of rutellum, thin membrane like with longitudinal incisions, setae a and m smooth and setiform (ca. 20 µm) (Figure 9C).


Gastronotic region (Figure 8A). Fifteen pairs of gastronotic setae (22-45 µm), same positions and shapes as in protonymph, except for p1-3 positioned more laterally.
Podosoma and venter (Figure 8B). Epimeral setation 3-1-2-2, seta 4b added in this stage. Three pairs of short genital setae. One pair of aggenital setae. Three pairs of adanal setae (ad1-3 ) flanking anal orifice. Cupule of lyrifissure iad anterior of anal opening.
Legs. Setation and solenidia shown Table 4.
Tritonymph (N=8): length 331–400 µm (mean 364 µm).


Gastronotic region (Figure 10A). Fifteen pairs of gastronotic setae (25-55 µm), no difference to deutonymph.
Podosoma and venter (Figure 10B). Epimeral setation 3-1-2-2, no difference to deutonymph. Five pairs of genital setae. Three pairs of adanal setae (ad1-3 ) and two pairs of short anal setae (an1-2 ) added in this stage.
Legs. Setation and solenidia shown in Table 4.
Family Carabodidae C. L. Koch, 1843
Genus Carabodes C. L. Koch, 1835
Carabodes sp
Common features of juvenile stages
Apheredermous. Integument soft and smooth, opisthonotal surface slightly granular in nymphal stages. Color light yellow, whitish. Body not wrinkled (wrinkles shown in figures represent a shrinking artefact due to preservation). Trichobothrial reduction present, bothridial seta bs very short setiform, bothridium not visible. Gnathosoma with no obvious differences to adult stage, all stages with same features (but in the following, these features are only presented in deutonymph for illustrative purposes). Rostrum relatively short, mouthparts not completely covered in dorsal aspect. Unideficient. Anus subterminal. Legs short, monodactylous with relatively high and robust claws; claw base enveloped by empodial membrane. Seta d absent on tibiae and genua I-III, present on genu and tibia IV from deutonymphal stage. Tarsal setae u short and strongly broadened. Developmental formulas (adult excluded): setation notogaster (larva to tritonymph) 11-15-15-15, (proto- to tritonymph) genital 1-2-4, aggenital variable, adanal (proto- to tritonymph) 0-3-3, anal (deuto- to tritonymph) 0-2.
Egg (N=2) (Figures 4A, 11A): length 157-200 µm (mean 179 µm).
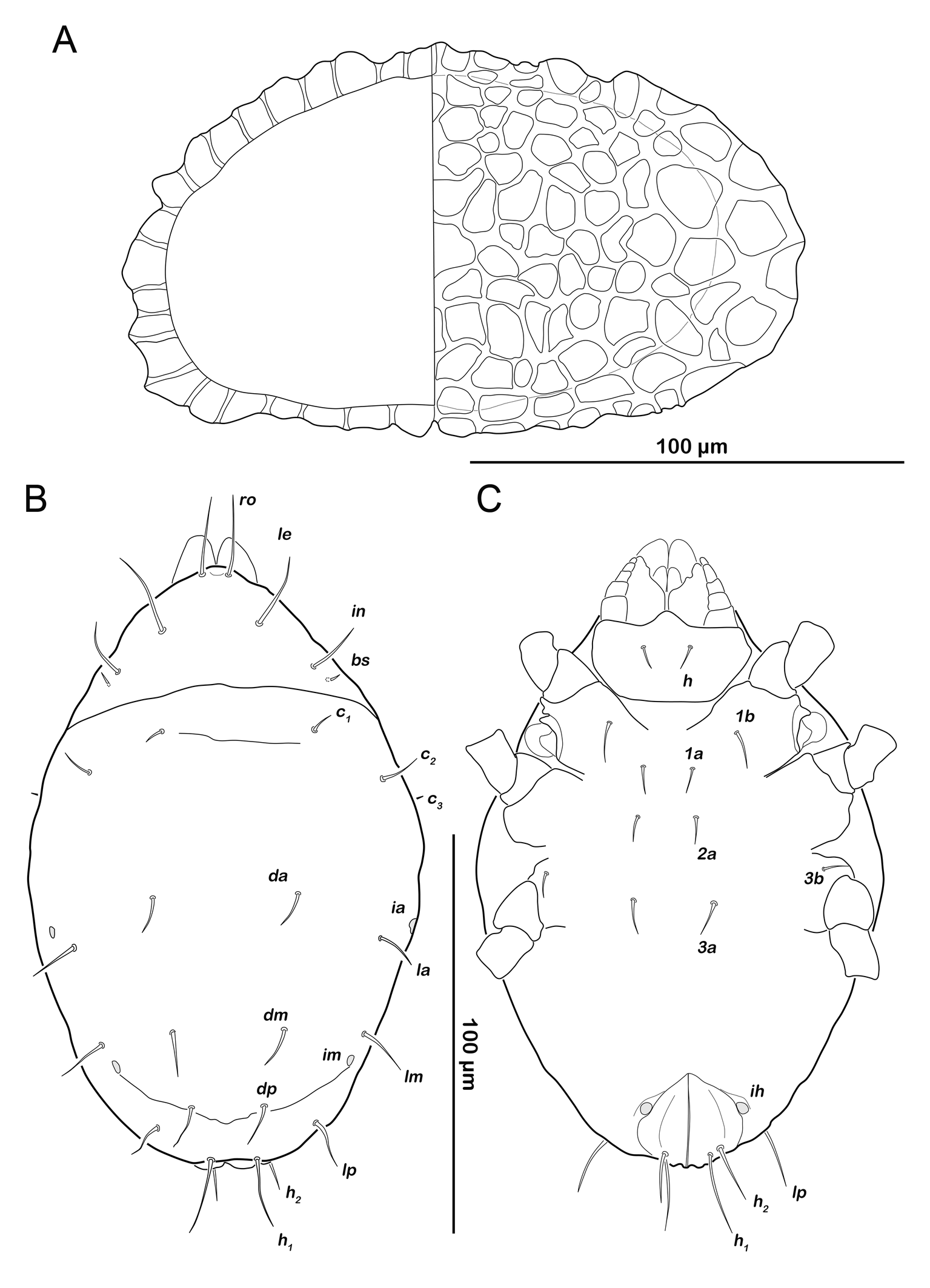


Color yellow to light orange. External layer (exochorion) thick (5-18µm), thickest at the poles, consisting of a remarkable net-like structure.
Larva (N=1): length 160 µm.
Prodorsum (Figure 11B). Rostral seta ro setiform, smooth and long, projecting rostrum. Lamellar seta le setiform, smooth, approx. same size as ro. Interlamellar seta in also smooth and setiform, slightly shorter than ro and in. Exobothridial seta ex minute, difficult to observe in this stage. Bothridial seta bs very short, setiform.
Gastronotic region (Figure 11B). Eleven pairs of setiform, smooth gastronotic setae (length 6-18 µm), c1-3 , da, dm, dp, la, lm, lp, h1-2 , h3 absent. Cupules well traceable, ia anterior to seta la, im posterior to seta lm, ih flanking anterior part of anal orifice. Opisthonotal gland opening gla hardly discernible in this stage.
Podosoma and venter (Figure 11C). Epimeral setation 2-1-2, most setae short (approx. 10 µm) and spiniform. Claparède's organ globular, located on lateral posterior edge of epimeron I, partly covered by protective seta, developed as thin triangular lamella. Aggenital, genital, adanal and anal setae not developed.
Legs. Setation and solenidia shown in Table 5.
Download as
Instars
Trochanter
Femur
Genu
Tibia
Tarsus
Chaetome
Solenidia
Leg I
larva
-
d, bv´´
(l), s
(l), v´, φ1
(pv), s, (a), (u), (p), (tc), (ft), ε, ω1
0-2-2-3-14
1-1-1
protonymph
-
-
-
-
ω2
0-2-2-3-14
1-1-2
deutonymph
-
-
-
φ2
-
0-2-2-3-14
1-2-2
tritonymph
v´
l´
-
v´´
(it)
1-3-2-4-16
1-2-2
Leg II
larva
-
d, bv´´
(l), s
l´, v´, φ
(pv), s, (a), (u), (p), (tc), (ft), ω1
0-2-2-2-13
1-1-1
protonymph
-
-
-
-
-
0-2-2-2-13
1-1-1
deutonymph
-
-
-
l´´
ω2
0-2-2-3-13
1-1-2
tritonymph
v´
-
v´
-
(it)
1-2-3-3-15
1-1-2
Leg III
larva
-
d, ev´
l´, s
v´, φ
(pv), s, (a), (u), (p), (tc), (ft)
0-2-1-1-13
1-1-0
protonymph
-
-
-
-
-
0-2-1-1-13
1-1-0
deutonymph
v´
-
-
-
-
1-2-1-1-13
1-1-0
tritonymph
-
-
-
-
(it)
1-2-1-1-15
1-1-0
Leg IV
protonymph
-
-
-
-
(pv), (u), (p), ft´´
0-0-0-0-7
0-0-0
deutonymph
-
d, ev´
d
v´, φ
s, (a), (tc)
0-2-1-1-12
0-1-0
tritonymph
v´
-
-
v´´
-
1-2-1-2-12
0-1-0
Protonymph (N=1): length 268 µm.


Prodorsum (Figure 12A). Interlamellar seta in same size as ro and le in this stage (approx. 31 µm), exobothridial seta ex visible as minute seta from this stage.
Gastronotic region (Figures 12A). Fifteen pairs of relatively long gastronotic setae (25-44 µm); c1-3 , da, dm, dp, la, lm, lp, h1-3 , p1-3 . Setae p1-3 framing anal orifice.
Podosoma and venter (Figure 12B). Epimeral setation 3-1-2-1, all setae spiniform (12-14 µm). Seta 1c and 4a added in this stage. One pair of short genital setae. Adanal and anal setae not developed. Cupule ips in paranal position, ih and ip laterally to anal opening.
Legs. Setation and solenidia shown in Table 5.
Deutonymph (N=7): length 246–369 µm (mean 287 µm).
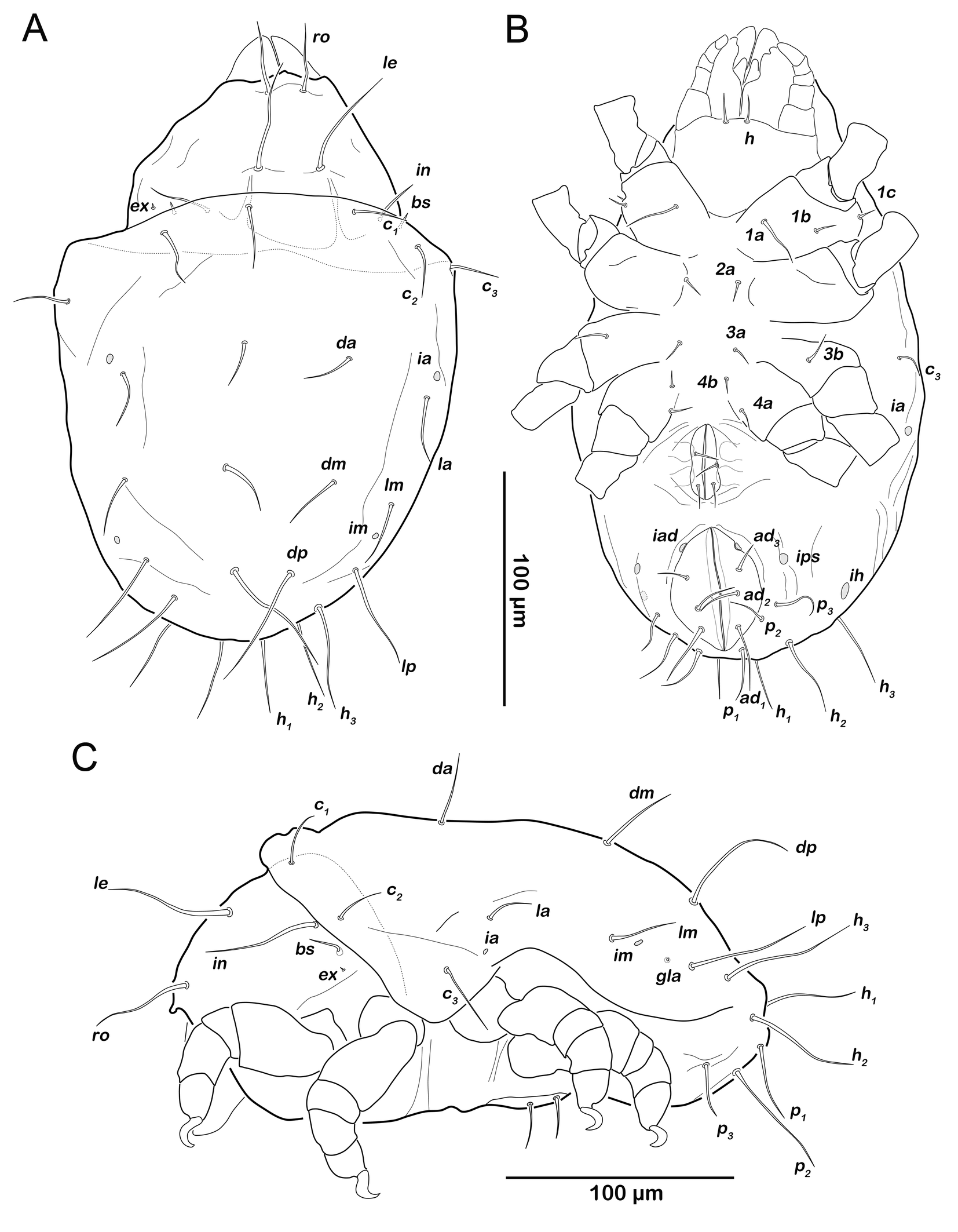


Prodorsum (Figure 13A, C). No difference to protonymph.
Gnathosoma. Chelicera chelate with strongly sclerotized teeth on fixed and movable digit, setae cha and chb setiform, long (ca. 30 µm) (Figure 14A). Distal part of rutellum, thin membrane like with longitudinal incisions, setae a and m smooth and setiform (ca. 20 µm) (Figure 14B). Palp setal formula 0-2-1-3-8 (+ solenidion ω) (Figure 14C).


Gastronotic region (Figure 13A). Fifteen pairs of gastronotic setae (20-60 µm), same positions and shapes as in protonymph, except for p1-3 positioned more laterally.
Podosoma and venter (Figure 13B). Epimeral setation 3-1-2-2, seta 4b added in this stage. Two pairs of short genital setae. One pair of aggenital setae absent. Three pairs of adanal setae (ad1-3 ) flanking anal orifice. Cupule of lyrifissure iad on anterior edge of anal opening.
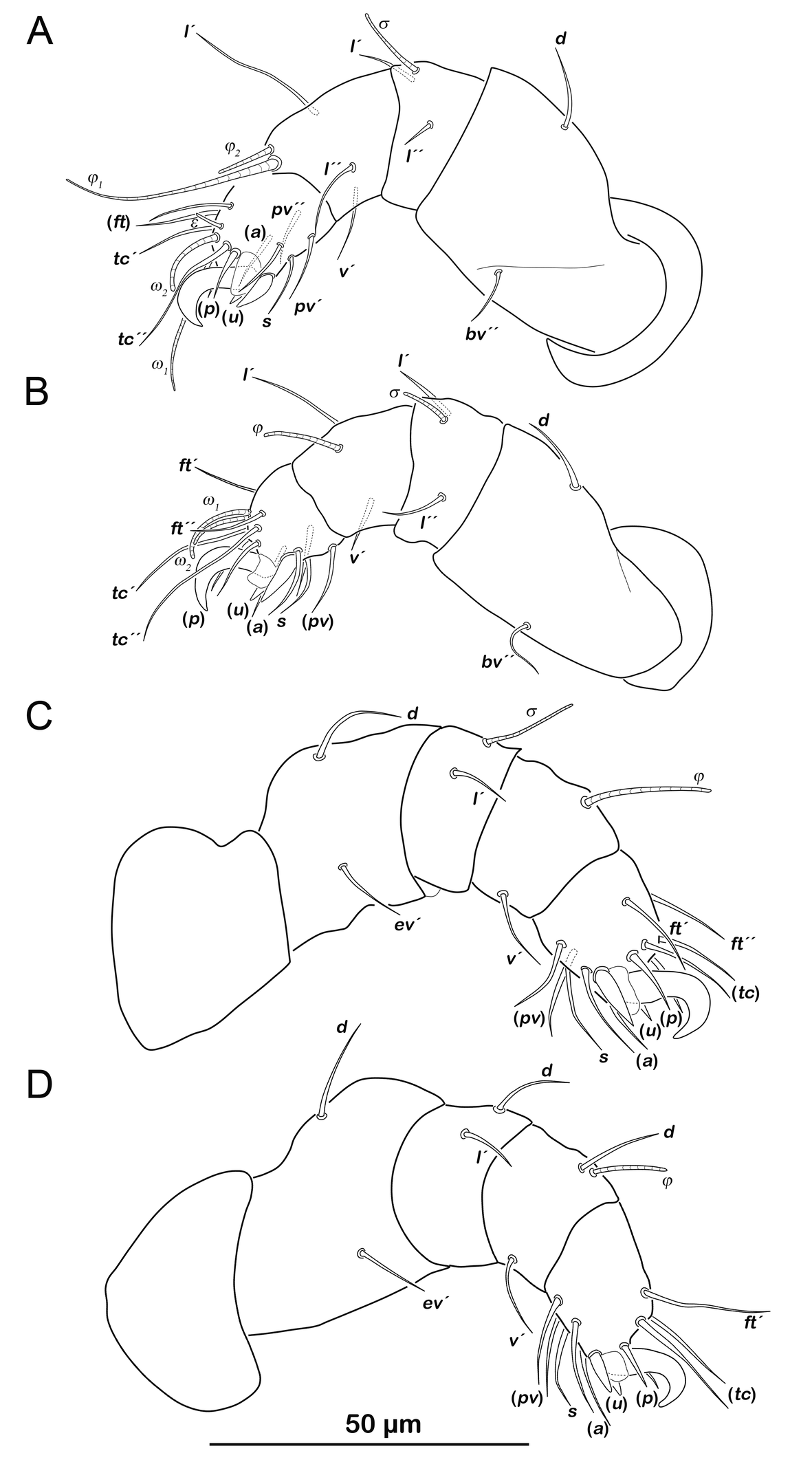


Legs (Figure 15). Setation and solenidia see Table 5.
Tritonymph (N=2): length 388–419 µm (mean 404 µm).


Prodorsum (Figure 16A). Prodorsal setae ro, le and in very long (100-110µm).
Gastronotic region (Figure 16A). Fifteen pairs of long gastronotic setae (59-78 µm), no difference to deutonymph.
Podosoma and venter (Figure 16B). Epimeral setation 3-1-2-2, no difference to deutonymph. Four pairs of genital setae. Three pairs of adanal setae (ad1-3 ), and two pairs of minute anal setae an1-2 .
Legs. Setation and solenidia shown in Table 5.
Discussion
Cladonia as habitat for oribatid mites
The oribatid mite fauna of the three investigated Cladonia species exhibits a high degree of similarity, regardless of their geographic origin. Statistics confirmed this similarity between lichens in terms of mite species richness and numbers of individuals. All three investigated lichens contain an average of five to seven mite species. This corresponds well with the observation of Materna (2000) who found an average of 6.6 oribatid mite species at a single stand of saxicolous lichens. The majority of most abundant species was the same, regardless of country and lichen species, suggesting that these often-detected species are frequent inhabitants or visitors of the lichen environment. Mites that are only found rarely and in low numbers, such as Eueremaeus cf. valkanovi or Eupelops torulosus, may represent species that inhabit adjacent habitats and occasionally wander into the lichen by chance; others are probably species that mostly occur in or on the underlying wood on which the lichen grows. Caleremaeus lignophilus, for example, is basically restricted to decaying wood as substrate (Lienhard and Krisper 2021), or Mesotritia nuda is often reported from old tree stumps (Weigmann 2006). However, species numbers and abundances should be viewed with caution because samples were taken at different times of the year and without standardizing the amount of substrate (i.e. some samples included a thicker layer of dead wood than others). Despite this methodical variation, the occurrence of Carabodes areolatus, C. marginatus, C. labyrinthicus, Melanozetes mollicomus and Neobrachychthonius marginatus magnus was so consistent in almost all samples that we conclude that these species are closely associated with all three investigated Cladonia types in continental Europe. Carabodes subarcticus and Hermannia gibba should also be viewed as common inhabitants of the here studied lichens, but their occurrence is geographically limited. Hermannia gibba does not occur in the whole Scandinavian area, where it is replaced by H. scabra and H. reticulata (e.g. Bäumler 1970) (although not found in our samples), explaining its absence in the Norwegian samples. Carabodes subarcticus, on the other hand, is a boreal species with a preference for more northern coniferous forests (e.g. Weigmann et al. 2015); therefore, it was absent in the more southern Austrian samples.
In Scottish Cladonia rubrotincta samples only Carabodes willmanni but none of the otherwise common Carabodes species could be detected. Carabodes willmanni is well known for its close association with Cladonia lichens, as its juveniles have previously been observed to be endophagous within them (Bellido 1978, 1979). Consequently, it seems reasonable to assume that Carabodes willmanni is responsible for the red coloration of Cladonia rubrotincta in Scotland. It remains unclear why other Carabodes species, such as C. marginatus and C. labyrinthicus, which should also occur in this geographic area (Luxton 1996, Pino-Bodas et al. 2021), were absent from our samples.
Apart from these biogeographic peculiarities, Carabodes species were found in each Cladonia sample and they appeared in high abundances, which strongly indicates that these mites commonly inhabit lichen microhabitats. Carabodes areolatus and C. marginatus were classified in group C by Seyd and Seaward (1984), which means they are species that frequently occur in lichens but are equally common in other biotopes. While C. areolatus was only rarely reported from lichens, C. marginatus was suggested to predominantly inhabit lichens (Kunst 1968) and was listed as characteristic for soil-dwelling lichens (Melekhina 2023). Moreover, Pino-Bodas et al. (2021) mentioned Carabodes marginatus as a possible cause for the red spotting of Cladonia norvegica. The consistent occurrence of C. marginatus in the three Cladonia species further corroborates its preference for these lichens as habitat. Carabodes labyrinthicus was classified in group B by Seyd and Seaward (1984) and there are numerous accounts of this species being an abundant inhabitant of diverse foliose and fruticose lichens (Kunst 1968, André 1984, Biazrov and Melekhina 1992, Materna 2000). It was one of the dominant species across 17 different species of epiphytic lichens (Melekhina 2023), and thus, it clearly shows a strong ecological association with lichens. Carabodes subarcticus was regularly found in high numbers in Cladonia arbuscula in Russia (Ermilov 2011, as Cladonia sylvatica sensu auct. brit.) and was suggested to be a characteristic faunal element of ground lichens (Melekhina 2023). The regular occurrence in C. coniocraea, C. norvegica and C. rubrotincta in Norway further confirms this species as a typical inhabitant of lichens in the more northern Eurasian regions. Carabodes willmanni juveniles were documented to be endophagous in Cladonia (Bellido 1978, 1979), and thus this species was classified in group B by Seyd and Seaward (1984). However, in the present study, Carabodes willmanni was only recovered in the Scottish samples of Cladonia rubrotincta. There is undoubtedly a close association of C. willmanni with Cladonia lichen, but factors such as distribution, competition with other Carabodes species, or preference for specific Cladonia hosts may geographically or ecologically limit this association. Further investigations are needed to clarify this.
In general, members of Carabodes strongly dominate the mite fauna in the lichen habitat (Wehner et al. 2018) and seem to prefer lichen as a food resource and breeding ground. All Carabodes species found herein, except for C. subarcticus, were observed to feed on the lichen and also have endophagous juveniles inside the lichens. The latter clearly classifies them as members of group B in the sense of Seyd and Seaward (1984), because this group is characterized by having lichen-restricted juveniles. Carabodes subarcticus juveniles have so far only been reared in the lab, where they were observed to stay on the lichen and molt in holes made in the lichen thalli (Ermilov 2011). Based on this rather vague observation, the existence of endophagy in this species remains unclear, but morphological features are indicative of such behaviour (discussed in the following sections).
Mycobates carli was only found in C. rubrotincta in Austria and was absent from all other samples. However, molecular genetic identification revealed the presence of at least one juvenile in one location in Norway which indicates the presence of M. carli and suggests that this species also uses C. rubrotincta for the development of its juveniles, just like it does in Austria, albeit in much lower abundances. This species shows a strong affinity to lichens in general. Kunst (1968) categorized M. carli as a species predominantly living in lichens, and Materna (2000) observed this species to be a frequent inhabitant of lichen covers. The closely related species Mycobates parmeliae was classified as a lichenophage with its juvenile stages being restricted to the lichen (Travé 1963, Seyd and Seaward 1984), and M. carli was suggested to be the ecological vicariant of M. parmeliae in the high central European Alps (Materna 2000). This hypothesis is supported by the fact that M. carli was found in the Austrian Cladonia rubrotincta, which prefers decaying wood of tree stumps and trunks often shared with other lichens and mosses at higher elevations in Central Europe as habitat (Vtípilová et al. 2025).
All species common to the lichen C. rubrotincta, i.e. Carabodes areolatus, C. labyrinthicus, C. marginatus and Mycobates carli, have been observed in this study to feed on red and green lichen material and to deposit fecal pellets in the respective colors. Detailed feeding experiments have been conducted by our group to investigate whether mite feeding activity causes the production of rhodocladonic acid, and whether the acid acts as repellent or attractant, but this will be presented and discussed in a separate publication.
Apart from that, our results demonstrate that Carabodes areolatus, C. labyrinthicus, C. marginatus and Mycobates carli show strong ecological affinity to Cladonia rubrotincta, as they use it for foraging and sheltered development. As mentioned before and shown in this study, these species are also common in many other lichens, e.g. other members of Cladonia, but also Evernia, Cetraria, Hypogymnia etc. (e.g. André 1984, Materna 2000, Melekhina 2023) and we assume that these other lichens are also used as a food source and kindergarten, although there are yet no reports for the latter.
Endophagous juveniles
Molecular genetic investigation of 19 juveniles, contained in burrows of Cladonia rubrotincta from Austria, the Czech Republic, and Norway, revealed that Carabodes areolatus, C. labyrinthicus, C. marginatus and Mycobates carli possess endophagous tunneling juveniles, which develop within the lichen thallus. Based on morphology, it is impossible to reliably distinguish juveniles of Carabodes and therefore behavioural observations cannot be assigned to individual species with absolute certainty. However, the basic principles of endophagous behaviour in lichen thallus are most likely the same for each species and thus probably apply to all Carabodes species.
Present observations clearly indicate that all species complete their whole ontogeny as endophages and leave the burrows after their final molt to adults. Endophagous juveniles in lichens have been reported for Dometorina plantivaga (Grandjean 1951), Carabodes willmanni (Bellido 1978, 1979, 1990) and Mycobates sarekensis (Seniczak et al. 2015). Unfortunately, the exact lichen species in which they develop was not specified. Grandjean (1951) stated that D. plantivaga occurred in a ′lichen gris' (a grey lichen) that grew on maple (Acer campestris). Bellido (1978, 1979, 1990) reported C. willmanni from Cladonia in France, but did not further state which species of this genus (we found it in Cladonia rubrotincta in Scotland), and Seniczak et al. (2015) did not mention in which specific lichen or lichens juveniles of M. sarekensis were found. However, Carabodes willmanni was observed to lay single eggs into decomposed parts of the lichen (Bellido 1978, 1979); the eggs showed no obvious ornamentation and were covered by a slimy layer which thickened at one of the poles to form a kind of cuff (Bellido 1978). The eggs we found in Cladonia rubrotincta all exhibited a distinct, thick and ornamented exochorion. Thick and heavily ornamented exochorions were also reported from several other oribatid mite taxa, e.g. Scutovertex, Rhysotritia and Microtritia (Märkel and Meyer 1959, Krisper et al. 2008), and are supposed to primarily protect against desiccations (Witaliński 1993). As the lichen itself already provides some protection against desiccation (Bellido 1990) it is unlikely that the exochorion of eggs found in C. rubrotincta serves the exact same function. Other examples further demonstrate that eggs deposited in lichens do not necessarily need additional protection against desiccation. The arboricol Camisia segnis deposits eggs with a smooth, translucent and extremely thin surface into lichens, where it is completely enveloped by the lichen thallus and the prelarval stage is already formed during deposition (Grandjean 1950). Ermilov (2011) reported Carabodes subarcticus, another characteristic inhabitant of epiphytic lichens (Melekhina 2023), to also deposit smooth semitransparent eggs during laboratory experiments, but the detailed circumstances of deposition remain elusive. In the present case, the unusual thick and ornamented exochorion of the Carabodes eggs may still serve as some kind of protection but against what exactly is yet unclear.
Concerning the morphology and behaviour of the subsequent juvenile stages, there is strong conformity among all known endophagous species. Juveniles have basically a very soft integument with reduced sclerotization (Michael 1882, Bellido 1990), are rather inactive (Bellido 1979, Ermilov 2011) and show morphological adaptations to the burrowing lifestyle (Bellido 1978, 1979; Seniczak et al. 2015). The latter includes a stockier body, curved inward short legs and reduced bothridial setae (Bellido 1978, Seniczak et al. 2015). These characters are more pronounced in Carabodes than in Mycobates juveniles. The subterminal position of the anus in Carabodes juveniles may be included to these adaptive characters because placing fecal pellets terminally is surely an advantage in narrow holes or tunnels. The chelicerae also seem to be larger and oriented in a more frontal position, as they are never covered by the rostrum in dorsal view. Grandjean (1951) observed an interesting behaviour in this respect: juvenile stages of D. plantivaga repaired holes in their burrows with their own fecal pellets and exactly the same behaviour was shown by Carabodes juveniles in this study. Closing holes to the outside may keep the tunnels moist and may prevent water, debris or predators from entering. A part of the consumed lichen tissue could pass the digestive tract of mites unaltered and serve as starting point for regrowing lichen tissue to close the holes in addition to fecal pellets alone. This could provide additional protection and it could explain why we found two juveniles in hollow burrows without an external feeding hole or any trace of it. However, this will have to be tested in dedicated experiments.
It also remains to be tested specifically if the endophagous behaviour of juvenile mites inhabiting C. rubrotincta causes rhodocladonic acid production. At this point, we can confirm that endophagous juveniles were consistently found in red, rhodocladonic acid containing, lichen thalli. This indicates a strong correlation between the substance and the presence of juvenile mites. We also regularly observed feeding activity of adult mites and a considerable number of red spots on C. rubrotincta did not contain juveniles. Such red spots could be the result of adult mite feeding activity. Rhodocladonic acid production is not immediate and, in contrast to mite feeding activity, cannot be observed directly. We therefore plan specific experiments to investigate the role of rhodocladonic acid in this interaction.
The present observations and data confirm the endophagous nature of the genera Carabodes and Mycobates. We therefore suggest that juvenile endophagy is a common phenomenon among these taxa and that several different lichen species may be used. In some cases, there is also indication that certain fungi may be used for endophagous development, as for example tree polypore as suggested for Carabodes erectus and Carabodes polyporetes (Reeves 1991, 1992).
Juvenile morphology
Download as
Character
M. acuspidatus
M. corticeus
M. carli
M. parmeliae
M. royi
M. sarekensis
length larva
300
-
190 (169-200)
-
250
257
length protonymph
367 (332-397)
240
262 (234-286)
-
335 (297-384)
297
length deutonymph
427 (413-454)
329 (322-336)
315 (292-356)
-
453 (393-480)
377
length tritonymph
580 (567-599)
443 (384-480)
364 (331-400)
-
542 (441-566)
449
prodorsal porose area
present
present
absent
absent
absent
absent
setae ro, le, in
barbed
barbed
smooth
smooth
barbed
barbed
bothridial seta
barbed
barbed
barbed
smooth
barbed
barbed
humeral organ in larva
not evident
-
evident
-
evident
evident
transverse depression pygidial shield
evident
conspicuous
faint
conspicuous
conspicuous
conspicuous
Presently, there are 35 species of Mycobates known worldwide (Subías 2022), but data on juveniles is only available for six of them, Mycobates acuspidatus, M. carli, M. corticeus, M. parmeliae, M. royi and M. sarekensis (Michael 1884, Palacios-Vargas and Vázquez 1988, Behan-Pelletier et al. 2001, Seniczak et al. 2015). Among these species, there is a general consistency in the juvenile habitus and morphology; all show a stocky appearance, the same developmental setal formulas, short barbed, clavate bothridial setae, a porose humeral organ and a large pygidial shield bearing the centrodorsal setae and seta h1 . Differences between species (Table 6) are mostly subtle and unsuitable for reliable species distinction, or should at least be interpreted with caution. For example, rostral, lamellar and interlamellar setae are barbed in M. royi (Palacios-Vargas and Vázquez 1988), M. acuspidatus, M. corticeus (Behan-Pelletier et al. 2001) and M. sarekensis (Seniczak et al. 2015), whereas in M. carli and M. parmeliae (Michael 1884) they are smooth. However, the latter description was made very early, and faint barbs may have been overlooked due to the limited microscopic means at that time. Mean body lengths also vary between species, e.g. juveniles of M. carli are apparently the smallest, but size ranges do often overlap (see Table 6). Mycobates acuspidatus and M. corticeus show a large prodorsal porose area posterior to seta in between the bothridia (Behan-Pelletier et al. 2001) and this character lacks in all other species (Michael 1884, Palacios-Vargas and Vázquez 1988, Seniczak et al. 2015), nevertheless porosity can be very fine and therefore be overlooked. Despite these possible uncertainties, it is very important to give detailed information on all juvenile stages, because distinctive features may still be found, and it provides a comprehensive insight into juvenile morphology of the genus in general, which further can be helpful in larger systematic considerations.
Download as
Character
Carabodes sp.
C. erectus
C. labyrinthicus
C. marginatus
C. polyporetes
C. subarcticus
C. willmanni
length larva
160
230 (215-255)
-
-
207 (180-230)
(184-196)
180 (160-197)
length protonymph
268
262 (230-310)
300 (285-320)
-
283 (260-330)
(213-232)
235 (200-257)
length deutonymph
287 (246-369)
331 (310-380)
404 (343-487)
-
346 (305-380)
(249-298)
275 (245-306)
length tritonymph
404 (388-419)
470 (400-520)
482 (446-526)
-
469 (435-505)
(332-415)
370 (320-413)
prodorsal microsclerite
absent
absent
absent
absent
absent
absent
present
bothridial seta
setiform
setiform
setiform
clavate
setiform
setiform
setiform
aggenital setation (Pn-Dn-Tn)
0-0-1
0-1-2
0-0/1-1
-
0-1-2
0-1-1
0-0-1
gastronotic median cornicle k
absent
present
absent
absent
present
absent
absent
Altogether, 134 Carabodes species are known worldwide (Subías 2022), but information on juveniles is only given for seven of these species: Carabodes coriaceus, C. erectus, C. labyrinthicus, C. marginatus, C. polyporetes, C. subarcticus and C. willmanni (Norton and Ermilov 2014). Comparing the juvenile morphologies of these few species provides a similar picture as shown in Mycobates, there is large conformity in habitus, lacking sclerotization, reduction of bothridial organs, setal developmental formulas and shortened legs (Michael 1880, 1884; Grandjean 1954; André 1975; Bellido 1978; Reeves 1991, 1992; Ermilov 2011). But there are also distinct differences between certain species or at least groups of species, which require some discussion (Table 7). Carabodes willmanni shows a distinct circular median microsclerite on its prodorsum (Bellido 1978), a feature that is clearly absent in all other species. Carabodes polyporetes and C. erectus possess a medial sclerotized cornicle k on its posterior gastronotic region (Reeves 1991, 1992), but none of the other species exhibits this character. Reeves (1991, 1992) stated that this cornicle is similar to the ones present in Damaeidae and Ameridae, where it serves to attach the exuviae from the preceding stage. Reeves (1991) observed this cornicle in other Carabodes juveniles, he reared in the lab, but he never saw them carrying an exuvial scalp and thus assumes a different, yet unknown, function for this unusual structure. The clavate bothridial seta of C. marginatus nymphs noted and illustrated by Michael (1884) is also unique among Carabodes juveniles because all other species show strongly reduced bothridial organs as a supposed adaptation to the endophagous lifestyle (e.g. Bellido 1978). Although we detected C. marginatus nymphs in Cladonia rubrotinca with molecular genetic means, we found no individual with a clavate bothridial seta among our morphologically investigated specimens. Grandjean (1954) provided the developmental aggenital setation for C. labyrinthicus with one pair of aggenital seta appearing in the tritonymphal stage, C. willmanni (Bellido 1978) and the herein investigated juveniles conform to this pattern, but C. polyporetes, C. erectus and C. subarcticus clearly deviate with having the aggenital setae already in the deutonymphal stage (Reeves 1991, 1992; Ermilov 2011). Moreover, André (1975) provided additional information on the juvenile stages of C. labyrinthicus and noted that the aggenital setae in his specimens were also present in the deutonymph, which is in contrast to the formula given by Grandjean (1954). Again, our molecular genetic investigations identified C. labyrinthicus in our C. rubrotincta samples, but the studied specimens exhibited the respective setae only in the last nymphal stage, as reported by Grandjean (1954). These differences may either be variations of certain geographic populations or they are the result of misidentifications. The endophagous lifestyle of Carabodes juveniles and their syntopic occurrence in the exact same patch of lichen clearly complicates identification, and that is why we refrained from assigning the herein described juveniles to distinct species. The studied specimens lacked distinct differences as given above, still we observed large variances in size, as for example most of the deutonymphs had a length of 240-289 µm and two showed values from 338-370 µm, which may be an indication that there were different species present in the morphologically studied material. However, most current DNA extraction methods for mite juveniles are destructive and do not allow to study morphology of the respective specimens before and after DNA extraction, preventing a combined analysis of morphological and molecular data. Carabodes areolatus was the most abundant species in all samples, and we found freshly molt adults in the lichen; moreover, as earlier descriptions of C. marginatus (Michael 1884) and C. labyrinthicus (André 1975) deviate from the herein given descriptions, it is assumed that the herein given juvenile stages represent C. areolatus specimens. Future studies clearly need to overcome these problems by using molecular genetic methods allowing to morphologically investigate the sequenced individual in detail either before or after DNA extraction, and by performing specific rearing experiments.
Acknowledgements
First of all, we would like to thank Einar Timdal for his help with collecting samples in Norway and his support during the project. We also thank Maria Liebmann-Reindl for generating preliminary data in her Master-thesis and Lukáš Jánošík, Zdeněk Palice and David Svoboda for providing material. Finally, we thank the Nationalpark Gesäuse GmbH for allowing us to take Cladonia samples.
Funding
This study was supported by projects N. 96p3 and 98p2 of the AKTION Austria – Czech Republic Initiative and by the Ministry of Culture of the Czech Republic (DKRVO 2024–2028/3.II.b, National Museum, 00023272).
acarologia_4837_Supplementary_Table_S1.pdf
References
- André H.M. 1975. Observations sur les Acariens corticoles de Belgique. Fondation Universitaire Luxembourgeoise, Série «Notes de Recherche», 4: 5-31.
- André H.M. 1984. Notes on the ecology of corticolous epiphyte dwellers. 3. Oribatida. Acarologia, 25: 385-395.
- Bäumler W. 1970. Zur Morphologie, Biologie und Ökologie von Hermannia gibba (Acarina, Oribatei) unter Berücksichtigung einiger Begleitarten. Z. Angew. Entomol., 66(3): 257-277.
- Behan-Pelletier V.M., Eamer B., Clayton M. 2001. Mycobatidae (Acari:Oribatida) of Pacific Northwest canopy habitats. Can. Entomol., 133: 755-775. https://doi.org/10.4039/Ent133755-6
- Bellido A. 1978. Developpement postembryonnaire de Carabodes willmanni Bernini, 1975 (Acari, Oribatei). Acarologia, 20: 419-432.
- Bellido A. 1979. Écologie de Carabodes willmanni Bernini, 1975 (Acari, Oribatei) dans les formations pionnières de la lande armoricaine. Rev. Ecol. Biol. Sol., 16(2): 195-218.
- Bellido A. 1990. Caractéristiques biodémographiques d'un acarien oribate (Carabodes willmanni) des pelouses xérophiles. Can. J. Zool., 68: 2221-2229. https://doi.org/10.1139/z90-309
- Biazrov L.G., Melekhina E.N. 1992. Oribatid mites in lichen consortia of Northern Scandinavia (a case study of the Varangerfjord). Bull. Mosc. Obshch. Isp. Prir., Biol. Ser., 97: 73-79. [In Russian]
- Dalenius J. 1960. Studies on the Oribatei (Acari) on the Tornetrásk Territory in Swedish Lapland. 1. A list of the habitats, and the composition of their Oribatid fauna. Oikos, 11: 80-124. https://doi.org/10.2307/3564885
- Ermilov S. 2011. The Biology of the Development of the Oribatid Mite Carabodes subarcticus (Acari, Carabodidae). Entomol. Rev., 91: 515-523. https://doi.org/10.1134/S0013873811040154
- Fox J., Weisberg S. 2020. car: Companion to Applied Regression (version 3.1-3). [R package]. https://cran.r-project.org/package=car.
- Fröberg L, Solhøy T., Baur A., Baur B. 2003. Lichen specificity of Oribatid mites (Acari; Oribatida) on limestone walls in the Great Alvar of Öland, Sweden. Ent. Tidskr., 124: 177-182.
- Gilbert O.L. 1976. A lichen-arthropod community. Lichenologist, 8: 98. https://doi.org/10.1017/S0024282976000133
- Grandjean F. 1950. Observations éthologiques sur Camisia segnis (Herm.) et Platynothrus peltifer (Koch) (Acariens). Bull. Mus., 2e série, 22(2): 224-231.
- Grandjean F. 1951. Sur deux espèces du genre Dometorina n. g. et les moeurs de D. plantivaga (Berl.) (Acariens, Oribates). Bull. Soc. Zool. France, 75: 224-242.
- Grandjean F. 1954. Essai de classification des Oribates (Acariens). Bull. Soc. Zool. France, 78: 421-446.
- Krisper G., Pfingstl T., Ebermann E. 2008. SEM-Investigations on the exochorion of scutoverticid eggs. Soil Org., 80: 217-221.
- Kunst M. 1968. Mites of the superorder Oribatida (in Czech). Habilitation thesis, Charles University, Prague, 1-1548.
- Laundon J.R. 1967. A study of the lichen flora of London. The Lichenologist, 3: 277-327. https://doi.org/10.1017/S0024282967000362
- Lehmitz R., Decker P. 2017. The nuclear 28S gene fragment D3 as species marker in oribatid mites (Acari, Oribatida) from German peatlands. Exp. Appl. Acarol., 71: 259-276. https://doi.org/10.1007/s10493-017-0126-x
- Lienhard A., Krisper G. 2021. Hidden biodiversity in microarthropods (Acari, Oribatida, Eremaeoidea, Caleremaeus). Sci. Rep., 11: 23123. https://doi.org/10.1038/s41598-021-02602-7
- Litvaitis M.K., Nunn G., Thomas W.K., Kocher T.D. 1994. A molecular approach for the identification of meiofaunal turbellarians (Platyhelminthes, Turbellaria). Mar. Biol., 120: 437-442. DOI: 10.1007/BF00680218 https://doi.org/10.1007/BF00680218
- Luxton M. 1996. Oribatid mites of the British Isles: a check-list and notes on biogeography (Acari: Oribatida). J. Nat. Hist., 30(6): 803-822. https://doi.org/10.1080/00222939600770441
- MacLean S.F.J. 1981. Introduction invertebrates. In: International Biological Programme 25. Tundra ecosystems: A Comparative Analysis. Cambridge University Press: Cambridge, UK, 1981; pp. 509-516.
- Märkel M., Meyer I. 1959. Zur Systematik der deutschen Euphthiracarini. Zool. Anz., 163: 327-342
- Materna J. 2000. Oribatid communities (Acari: Oribatida) inhabiting saxicolous mosses and lichens in the Krkonoše Mts. (Czech Republic). Pedobiologia, 44: 40-62. https://doi.org/10.1078/S0031-4056(04)70027-X
- Meier F.A., Scherrer S., Honegger R. 2002. Faecal pellets of lichenivorous mites contain viable cells of the lichen-forming ascomycete Xanthoria parietina and its green algal photobiont, Trebouxia arboricola. Biol. J. Linn. Soc., 76: 259-268. https://doi.org/10.1111/j.1095-8312.2002.tb02087.x
- Melekhina E.N. 2023. Lichen-Associated Oribatid Mites in the Taiga Zone of Northeast European Russia: Taxonomical Composition and Geographical Distribution of Species. Diversity, 15: 599. https://doi.org/10.3390/d15050599
- Michael A.D. 1880. Further contribution to the knowledge of British Oribatidae. (Part I). Royal Microsc. Soc., 3: 32-43. https://doi.org/10.1111/j.1365-2818.1880.tb05649.x
- Michael A. D. 1882. Further notes on British Oribatidae. J. R. Micr. Soc. (ser. 2) 2: 1-19.
- Michael AD. 1884. British Oribatidae. Volume I. London: Ray Society.
- Norton R.A. 2007. Holistic acarology and ultimate causes: Examples from the oribatid mites. In: Morales-Malacara J.B., Behan-Pelletier V., Ueckermann E., Pérez T.M., Estrada-Venegas E.G., Badil M. (Eds.). Acarology XI: Proceedings of the International Congress; Instituto de Biología and Facultad de Ciencias, Universidad Nacional Autónoma de México, Sociedad Latinoamericana de Acarología, México. p. 3-20.
- Norton R.A., Ermilov S.G. 2014. Catalogue and historical overview of juvenile instars of oribatid mites (Acari: Oribatida). Zootaxa, 3833: 1-132. https://doi.org/10.11646/zootaxa.3833.1.1
- Palacios-Vargas JG, Vázquez IM. 1988. A new Mexican arboreal Mycobates (Oribatei: Mycobatidae). Acarologia, 29: 87-93.
- Paulson R. & Thompson P.G. 1911. Reports on the lichens of Epping Forest. Essex Naturalist, 16: 136-145.
- Paulson R., Thompson P.G. 1913. Reports on the lichens of Epping Forest. Essex Naturalist, 17: 90-105.
- Pino-Bodas R., Sanderson N., Cannon P., Aptroot A., Coppins B., Orange A., Simkin J. 2021. Lecanorales: Cladoniaceae, including the genera Cladonia, Pilophorus and Pycnothelia. Revisions of British and Irish Lichens, 19: 1-45.
- Prinzing A., Wirtz H.-P. 1997. The epiphytic lichen, Evernia prunastri L., as a habitat for arthropods: shelter from dessication, food-limitation and indirect mutualism. In: Stork N.E., Adis J & Didham R.K. (eds.). Canopy Arthropods. Chapman & Hall, London, pp. 478-494.
- R CORE TEAM 2021. R: A Language and environment for statistical computing. Version 4.1 [Computer software]. https://cran.r-project.org.
- Reeves R.M. 1991. Carabodes niger Banks, C. polyporetes n.sp., and unverified records of C. areolatus Berlese (Acari: Oribatida: Carabodidae) in North America. Can. J. Zool., 69: 2925-2934. https://doi.org/10.1139/z91-413
- Reeves R.M. 1992. Carabodes of the eastern United States and adjacent Canada (Acari: Oribatida: Carabodidae). Can. J. Zool., 70: 2042-2058. https://doi.org/10.1139/z92-276
- Reutimann P., Scheidegger C. 1987. Importance of lichen secondary products in food choice of two oribatid mites (Acari) in an alpine meadow system. J. Chem. Ecol., 13: 363-369. https://doi.org/10.1007/BF01025896
- Schäffer S., Krisper G., Pfingstl T., Sturmbauer C. 2008. Description of Scutovertex pileatus sp. nov. (Acari, Oribatida, Scutoverticidae) and molecular phylogenetic investigation of congeneric species in Austria. Zool. Anz., 247: 249-258. https://doi.org/10.1016/j.jcz.2008.02.001
- Seniczak S., Seniczak A., Coulson S.J. 2015. Morphology, distribution and biology of Mycobates sarekensis (Acari: Oribatida: Punctoribatidae). Int. J. Acarol., 41: 663-675. https://doi.org/10.1080/01647954.2015.1102968
- Seyd E.L., Seaward M.R.D. 1984. The association of oribatid mites with lichens. Zool. J. Linn. Soc., 80: 369-420. https://doi.org/10.1111/j.1096-3642.1984.tb02552.x
- Singh G., Dal Grande F., Martin F.M., Medema M.H. 2025. Breaking into nature's secret medicine cabinet: Lichens - a biochemical goldmine ready for discovery. New Phytol., 246(2): 437-449. https://doi.org/10.1111/nph.70003
- Sowter F.A. 1971. Mites (Acari) and lichens. The Lichenologist, 5: 176. https://doi.org/10.1017/S0024282971000185
- Spribille T. 2018. Relative symbiont input and the lichen symbiotic outcome. Curr. Opin. Plant. Biol., 44: 57-63. https://doi.org/10.1016/j.pbi.2018.02.007
- Stenroos S., Pino‐Bodas R., Hyvönen J., Lumbsch H.T., Ahti T. 2018. Phylogeny of the family Cladoniaceae (Lecanoromycetes, Ascomycota) based on sequences of multiple loci. Cladistics, 35: 351-384. https://doi.org/10.1111/cla.12363
- Subías L.S. 2022. Listado sistemático, sinonímico y biogeográfico de los ácaros oribátidos (Acariformes, Oribatida) del mundo (excepto fósiles). Monografías electrónicas, 12: 1-538.
- Tamura K, Stecher G, Peterson D, Filipski A, Kumar S. 2013. MEGA6: molecular evolutionary genetics analysis version 6.0. Mol. Biol. Evol., 30: 2725-2729. DOI 10.1093/molbev/mst197. https://doi.org/10.1093/molbev/mst197
- THE JAMOVI PROJECT 2022. jamovi (Version 2.3) [Computer Software]. https://www.jamovi.org.
- Timdal E. 1989. The production of rhodocladonic acid in Cladonia bacilliformis and C. norvegica triggered by the presence of a lichenicolous mite. Graph. Scr., 2: 125-127.
- Travé J. 1963. Ecologie et biologie des Oribates (Acariens) saxicoles et arboricoles. Vie et Milieu, 14: 267.
- Vtípilová V., Timdal E., Stodůlková E., Semerád J., Resl P., Steinová J. 2025. Cladonia rubrotincta, a new species distinguished from C. norvegica. The Lichenologist: Published online 2025:1-13. https://doi.org/10.1017/S002428292510128X
- Wehner K., Heethoff M., Brückner A. 2018. Sex ratios of oribatid mite assemblages differ among microhabitats. Soil Org., 90: 13-21.
- Weigmann G. 2006. Hornmilben (Oribatida). Die Tierwelt Deutschlands. Goecke & Evers, Keltern: 520 pp.
- Weigmann G., Horak F., Franke K., Christian A. 2015. Verbreitung und Ökologie der Hornmilben (Oribatida) in Deutschland. Peckiana, 10: 1-171.
- Witaliński, W. 1993. Egg shells in mites: vitelline envelope and chorion in Acaridida (Acari). Exp. Appl. Acarol., 17: 321-344 https://doi.org/10.1007/BF00058596
- Zopf W. 1907. Biologische und morphologische Beobachtungen an Flechten. Ber. Dtsch. Bot. Ges., 25: 233-238. https://doi.org/10.1111/j.1438-8677.1907.tb06614.x



2025-07-03
Date accepted:
2025-10-20
Date published:
2025-11-05
Edited by:
Baumann, Julia

This work is licensed under a Creative Commons Attribution 4.0 International License
2025 Pfingstl, Tobias; Vtípilová, Věra; Ghlimová, Heda; Mourek, Jan; Steinová, Jana; Hessinger, Tobias P. ; Schäffer, Sylvia and Resl, Philipp
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



