Two new species the genus Neocunaxiodes (Acariformes: Cunaxidae) from China
Chen, Jian-Xin  1
; Yao, Mao-Yuan
1
; Yao, Mao-Yuan  2
; Wu, You-Fang
2
; Wu, You-Fang  3
; Guo, Jian-Jun
3
; Guo, Jian-Jun  4
; Yi, Tian-Ci
4
; Yi, Tian-Ci  5
and Jin, Dao-Chao
5
and Jin, Dao-Chao  6
6
1College of Agriculture / Key Laboratory of Information Technology for Plant Protection in Characteristic High-Efficiency Agriculture of Central Guizhou, Anshun University, Anshun, 561000, P. R. China.
2College of Agriculture / Key Laboratory of Information Technology for Plant Protection in Characteristic High-Efficiency Agriculture of Central Guizhou, Anshun University, Anshun, 561000, P. R. China.
3College of Agriculture / Key Laboratory of Information Technology for Plant Protection in Characteristic High-Efficiency Agriculture of Central Guizhou, Anshun University, Anshun, 561000, P. R. China.
4Institute of Entomology, Guizhou University; Guizhou Provincial Key Laboratory for Agricultural Pest Management of the Mountainous Region; Scientific Observing and Experimental Station of Crop Pest in Guiyang, Ministry of Agriculture and Rural Affairs of the P. R. China, Guiyang 550025. P. R. China.
5Institute of Entomology, Guizhou University; Guizhou Provincial Key Laboratory for Agricultural Pest Management of the Mountainous Region; Scientific Observing and Experimental Station of Crop Pest in Guiyang, Ministry of Agriculture and Rural Affairs of the P. R. China, Guiyang 550025. P. R. China.
6✉ Institute of Entomology, Guizhou University; Guizhou Provincial Key Laboratory for Agricultural Pest Management of the Mountainous Region; Scientific Observing and Experimental Station of Crop Pest in Guiyang, Ministry of Agriculture and Rural Affairs of the P. R. China, Guiyang 550025. P. R. China.
2025 - Volume: 65 Issue: 3 pages: 900-916
https://doi.org/10.24349/tf8q-wxeuZooBank LSID: 7C723F53-A932-4D72-9866-9C9D0F34E0B0
Original research
Keywords
Abstract
Introduction
Cunaxid mites have a predatory habit and prey on microarthropods and nematodes, and play an important role in agricultural ecosystems (Hernandes et al. 2015, Al-Azzazy & Al-Rehiayani 2022).
The genus Neocunaxoides was erected by Smiley (1975) with Cunaxoides andrei Baker & Hoffmann, 1948 as type species, which belongs to the subfamily Cunaxoidinae (Den Heyer 1978). Neocunaxoides andrei was reported in Russia, China, Poland and Egypt (Kuznetzov & Livshitz 1979, Tseng 1980, Michocka 1987, Hu 1997, Shoala and El Kady 2009). Den Heyer (1980a) reported N. lajumensis, N. rykei and N. zuluensis from South Africa. Tseng (1980) described N. whartoni from Taiwan, China, Skvarla et al. (2014) moved N. whartoni to Pulaeus. Inayatullah & Shahid (1989) described two species, namely, N. dilato and N. kalamiensis from Pakistan. N. biswasi and N. cerasoides were described from India (Gupta & Chattopadhyay 1978, Gupta 1991).
Smiley (1992) synonymized Scutopalus with Neocunaxoides and moved Cunaxoides trepidus to Neocunaxoides. Romeih and Abo-Shnaf (2006) described N. fayoumi from Egypt. N. grandis, N. mahabaeus and N. ornatus were described from Philippine (Corpuz-Raros 1996, Corpuz-Raros & Gruèzo 2007). Lin et al. described N. boltoides, N. fani and N. ovatus from China (Lin et al. 2001, 2003). N. metwallyi was described in Egypt (Fawzy 2007). Corpuz-Raros (2007) described Cunaxoides philippinensis from Philippine and Skvarla et al. (2014) moved Cunaxoides philippinensis Corpuz-Raros, 2007 to genus Neocunaxoides. Castro and Den Heyer (2009) moved Pulaeus trepidus (=Neocunaxoides trepidus) to Scutopalus.
Rocha et al. (2015) described N. promatae from Brazil, Paktinat-Saeij et al. (2016) moved Pulaeus neopectintus (Shiba, 1978) to Neocunaxoides, Alatawi & Kamran (2017) reported N. ornatus from Saudi Arabia, Kalúz & Ermilov (2018) described N. tropicus from Vietnam, and provided a key to adult females of Neocunaxoides species of the world. Later Chen et al. (2020) moved P. neopectintus to Lepidocunaxoides Kalúz & Ermilov, 2019.
Currently, genus Neocunaxoides contains 20 known species in the world, but only have four species reported in China, N. andrei (Baker & Hoffmann, 1948), N. boltoides Lin, 2001, N. fani Lin, 2001 and N. ovatus Lin, Zhang & Ji, 2003 (Tseng 1980, Hu 1997, Lin et al. 2001, Lin et al. 2003, Lin & Zhang 2010). In this work, we add two species new to science N. huangshanensis Chen & Jin sp. nov. and N. pinus Chen & Jin sp. nov. from the fauna of China. In addition, provide a species key of adult females of genus Neocunaxoides from China.
Materials and methods
Samples of moss were collected from Huangshan Scenic Spot, Huangshan city, Anhui Province, Wangjiabang, Ningqiang county, Hanzhong city, Shanxi Province and fallen leaves of Pinus yunnanensis were collected in a forest, Yulong Snow Mountain, Lijiang City, Yunnan Province, P. R. China. Mites were isolated from the samples by using Berlese-Tullgren funnels for eight to twelve hours, preserved in 75% alcohol and then mounted in Hoyer's medium on slides (Walter & Krantz 2009). Line drawings were prepared with the aid of a drawing tube attached to a phase contrast and DIC Nikon Ni E microscope. Photographs were taken using a camera (Nikon DS-Ri 2) attached to the Nikon Ni E microscope and figures were edited with Adobe Photoshop CC 2019. The length of the gnathosoma was measured from the base to the top of the subcapitulum, the length of the idiosoma, from the suture between the gnathosoma and idiosoma to the posterior margin of the idiosoma, the width of the idiosoma at its broadest level and the length of the legs from the ventral insertion of coxae to the tip of the claw. All measurements were taken in micrometers (µm) using the software Nikon NIS Elements AR 4.50 and provided for the holotype and the paratypes. The dorsal and ventral setal nomenclature follows Fisher et al. (2011) and Paktinat-Saeij et al. (2017), legs setal notation follows Den Heyer (1981).
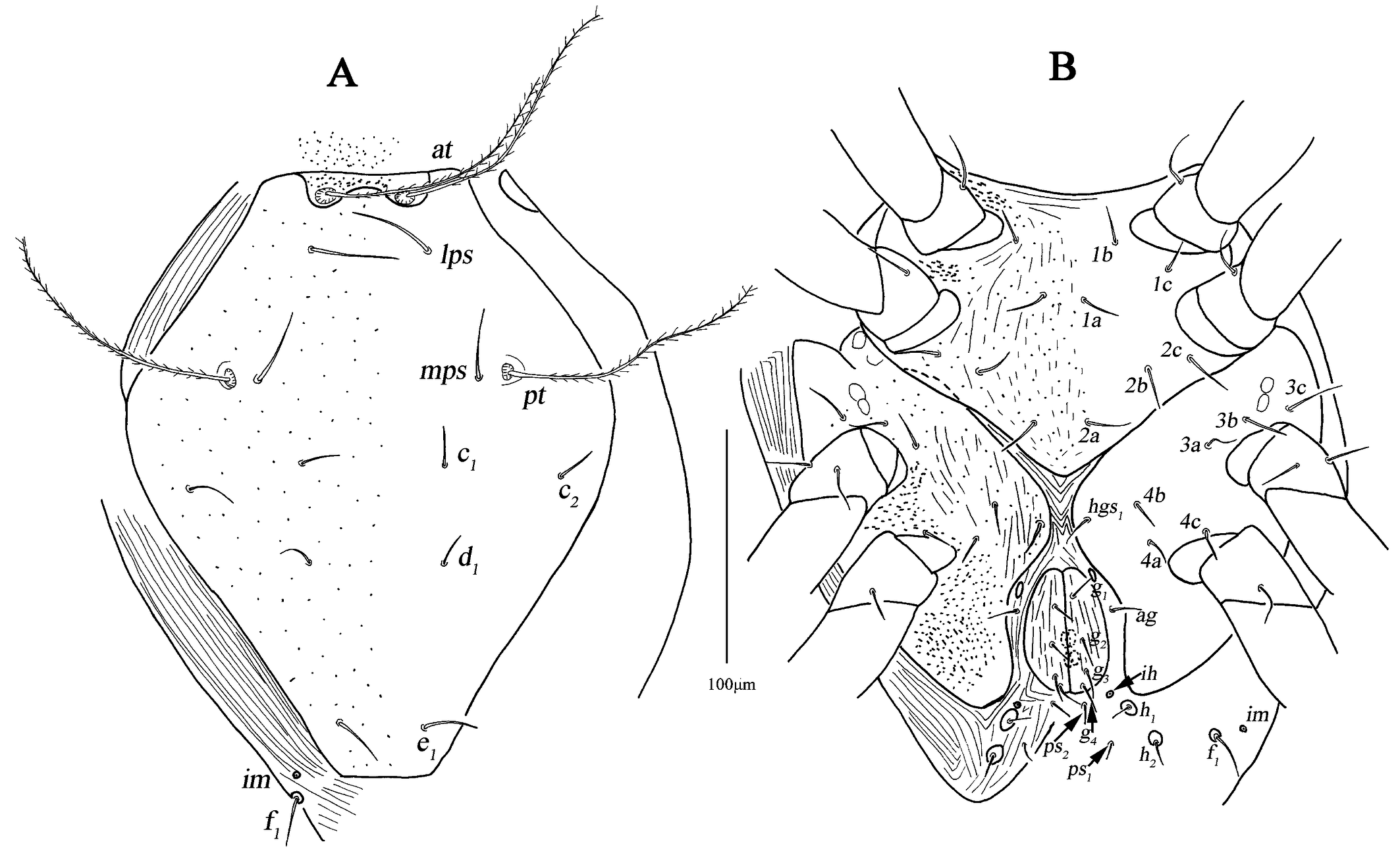
The following abbreviations are used: prodorsum: anterior trichobothria (at), posterior trichobothria (pt), lateral proterosomal (lps), median proterosomal (mps); hysterosoma: internal humerals (c1 ), external humerals (c2 ), internal dorsals (d1 ), internal lumbals (e1 ), internal sacrals (f1 ), internal clunals (h1 ), external clunals (h2 ); venter: propodogastral seta (ppgs), hysterogastral seta (hgs); anal region: pseudanal (ps); genital region: aggenitals (ag), genitals (g); gnathosoma: hypognathals (hg); leg: attenuate (sharply) solenidion (asl), blunt-pointed rod-like solenidion (bsl), famulus (fam), trichobothria (T), simple tactile seta (sts), microseta (mst), dorsoterminal solenidion (dtsl).
Results
Family Cunaxidae Thor, 1902
Subfamily Cunaxoidinae Den Heyer, 1978
Tribe Pulaeini Den Heyer, 1980b
Genus Neocunaxiodes Smiley, 1975
Generic diagnosis — see Skvarla et al. (2014).
Type species — Cunaxoides andrei Baker & Hoffmann, 1948.
Neocunaxiodes huangshanensis Chen & Jin sp. nov
ZOOBANK: E6C74961-6EC9-4953-8A63-80D52AE3B124 ![]()
(Figs. 1–8)
Diagnosis
Subcapitulum without striae and between hg3 and the base area with two row polygonal pattern; dorsal shield with random punctation; coxae I–II fused medially as a V-shaped single sternal and between coxae II small platelet absent; setal formula of coxal plates I–IV: 3-3-3-3 sts, basifemora I–IV: 3-5-2-0 sts. Female anterior to genital plates with a small platelet; three pairs of pairs of hysterogastral setae (hgs1–3 ); genital plates large and with reticulate pattern, and one pair of small platelets posterolateral situated in genital plates. Male similar to female, differing as follows: anterior to genital plates small platelet absent; one pairs of pairs of hysterogastral setae (hgs1 ); genital plates small and reticulate pattern absent, one pair of small platelets anterolateral situated in genital plates; genu I–IV, tibia I and tarsus I–II with long bsl.
Description
Female (n=4)
Idiosoma 438 (407–430) long, 337 (330–337) wide.
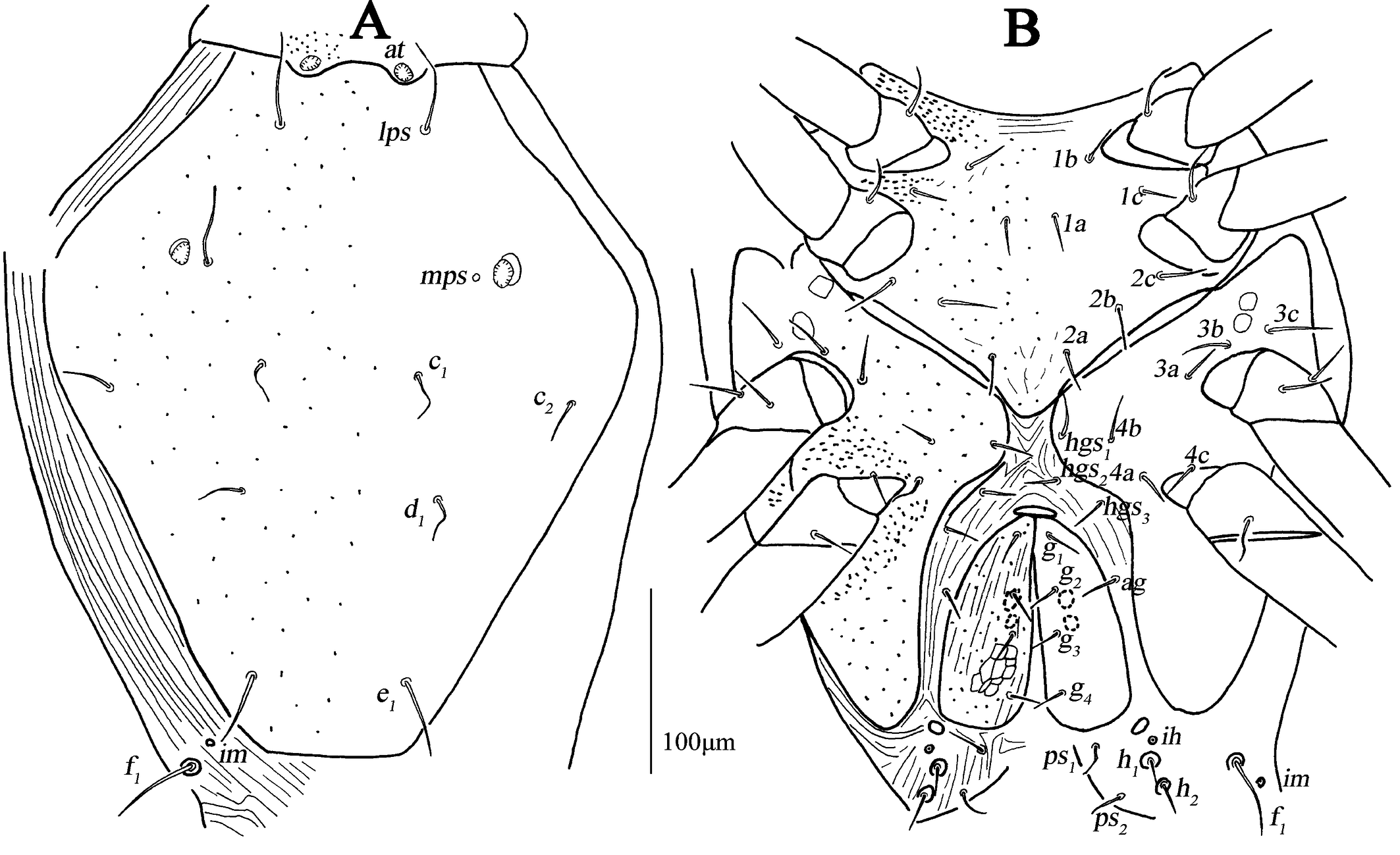


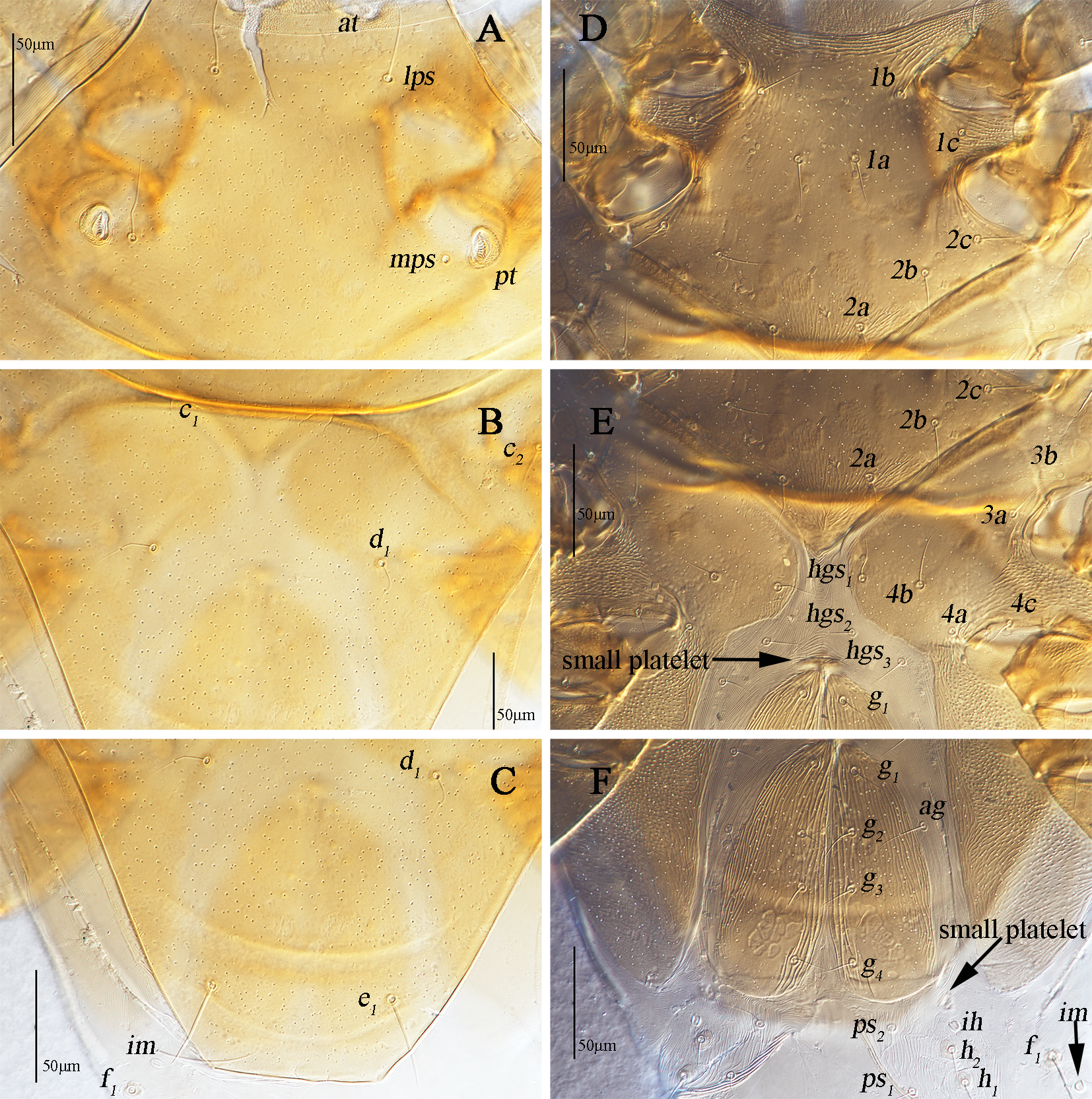


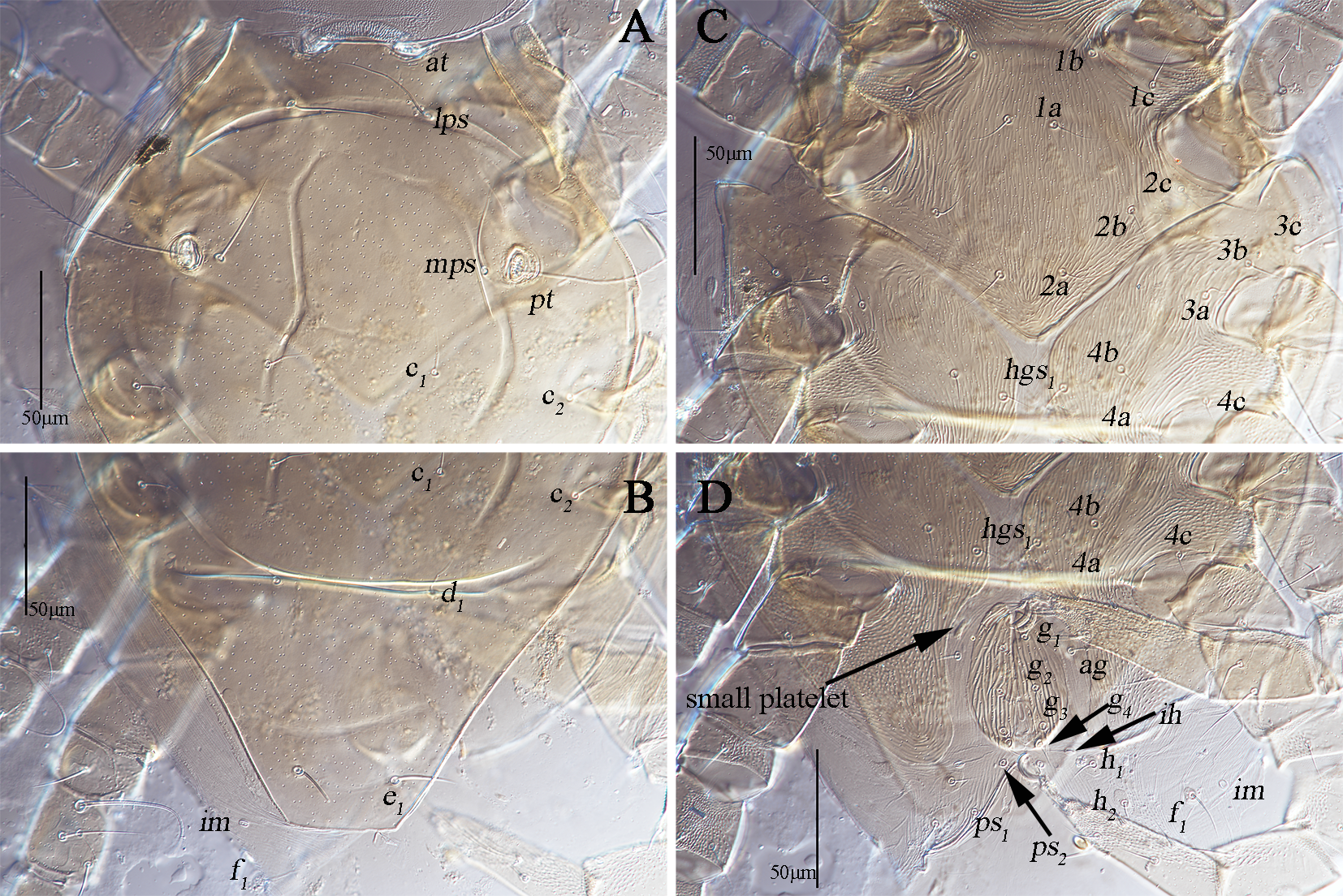
Dorsum (Figs. 1A, 2A–C). Propodosomal shield concave anteromedially, and at off the shield though very close to the edge of the concaves, area anterior to at with some fine papillae. Proterosomal and hysterosomal shields fused, forming a large dorsocentral shield, 370 (370–377) long, 303 (288–303) wide, with random punctation, bearing one pairs of trichobothria (pt), two pairs of tactile setae (lps and mps), four pairs of simple setae (c1 , c2 , d1 , e1 ); f1 situated on small platelets, lateral area outside dorsocentral shield with lengthwise striation and one pair of lyrifissures (im) situated on lateral soft integument anterior to f1 . Setal lengths and distances: at ?(121–130), pt ?(120–126), lps 50 (48–50), mps 40 (30–40), c1 20 (20–21), c2 20 (20–20), d1 25 (24–25), e1 42 (37–42), f1 48 (42–48); at–at 46 (28–51), pt–pt 167 (167–168), lps–lps 77 (74–77), mps–mps 137 (135–137), lps–mps 84 (83–84), at–lps 36 (32–36), pt–mps 14 (13–14), pt–lps 88 (85–88), at–mps 117 (115–117), at–pt 122 (120–122), c1 –c1 82 (81–82), c2 –c2 236 (227–236), d1 –d1 88 (88–97), e1 –e1 80 (77–80), c1–c2 79 (74–79), c1 –d1 66 (66–82), c2 –d1 88 (88–93), d1 –e1 100 (91–99), e1 –f1 59 (33–59).
Venter (Fig. 1B, 2D–F). Coxae I–II fused medially as a V-shaped single sternal plate and with random punctation, coxae III–IV fused and forming paired lateral ventral plates. Areas anterior to coxae I, between coxae I and coxae II, between coxae III and coxae IV, and posterior to coxae IV with strong punctations; coxal plates III–IV with random punctation and a few of reticulate pattern. Posterior edge of coxal plates IV extending and almost reached to anterior edge of genital plates level. Setal formula of coxal plates I–IV: 3(1a–c)-3(2a–c)-3(3a–c)-3(4a–c) sts, length of setae: 1a 19 (13–22), 1b 21 (17–23), 1c 16 (16–23), 2a 22 (17–22), 2b 19 (18–21), 2c 21 (29–31), 3a 21 (21–24), 3b 18 (16–18), 3c 33 (26–29), 4a 9 (7–9), 4b 22 (15–22), 4c 15 (14–15). Three pairs of hysterogastral setae (hgs1 –hgs3 ), lengths of hgs1 –hgs3 : 17 (16–17), 14 (11–14) and 9 (9–15), respectively, hgs1 situated in lateral ventral plates. Areas between lateral ventral plates with longitudinal striation. Small platelet anteriomedially of genital plates present, 23 (18–22) long, 14 (11–12) wide, and one pair of small platelets posterolateral situated in genital plates. Genital plates large, 118 (105–118) long, 42 (40–43) wide, with reticulate pattern, broken longitudinal striation and random punctation, two pairs of visible genital papillae, four pairs of genital setae (g1 –g4 ) arranged longitudinally that 19 (14–18), 18 (14–16), 15 (12–15) and 16 (15–16) in length, respectively; one pair of aggenital setae (ag), long 19 (12–19), almost at g2 level. Anal region with two pairs of pseudanal setae (ps1 –ps2 ) 14 (14–16) and 17 (14–17) in length, respectively, two pairs of platelets setae bearing h1 15 (15–21) and h2 16 (13–16), respectively, and one pair of lyrifissures (ih).


Gnathosoma. Palp (Fig. 3A). Three-segmented, 111 (111–120) long. Palp chaetotaxy: trochanter none; femurogenu with six simple setae; tibiotarsus with five simple setae, one pointed apophysis and one knob-like apophysis; claw well-developed and simple.
Chelicera (Fig. 3B). 184 (170–184) long, with strong punctations; one cheliceral seta 12 (11–12) in length; chela developed.
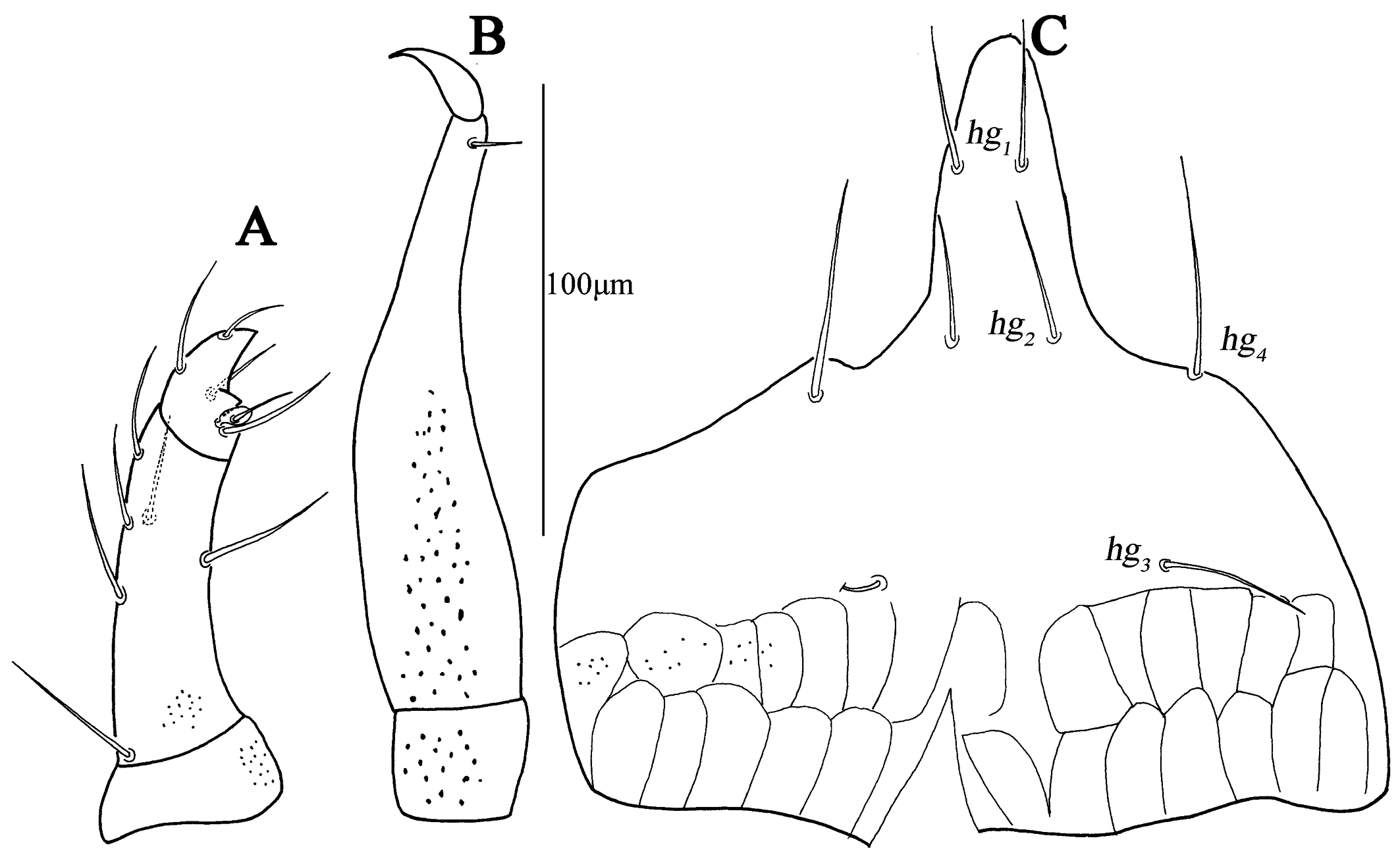
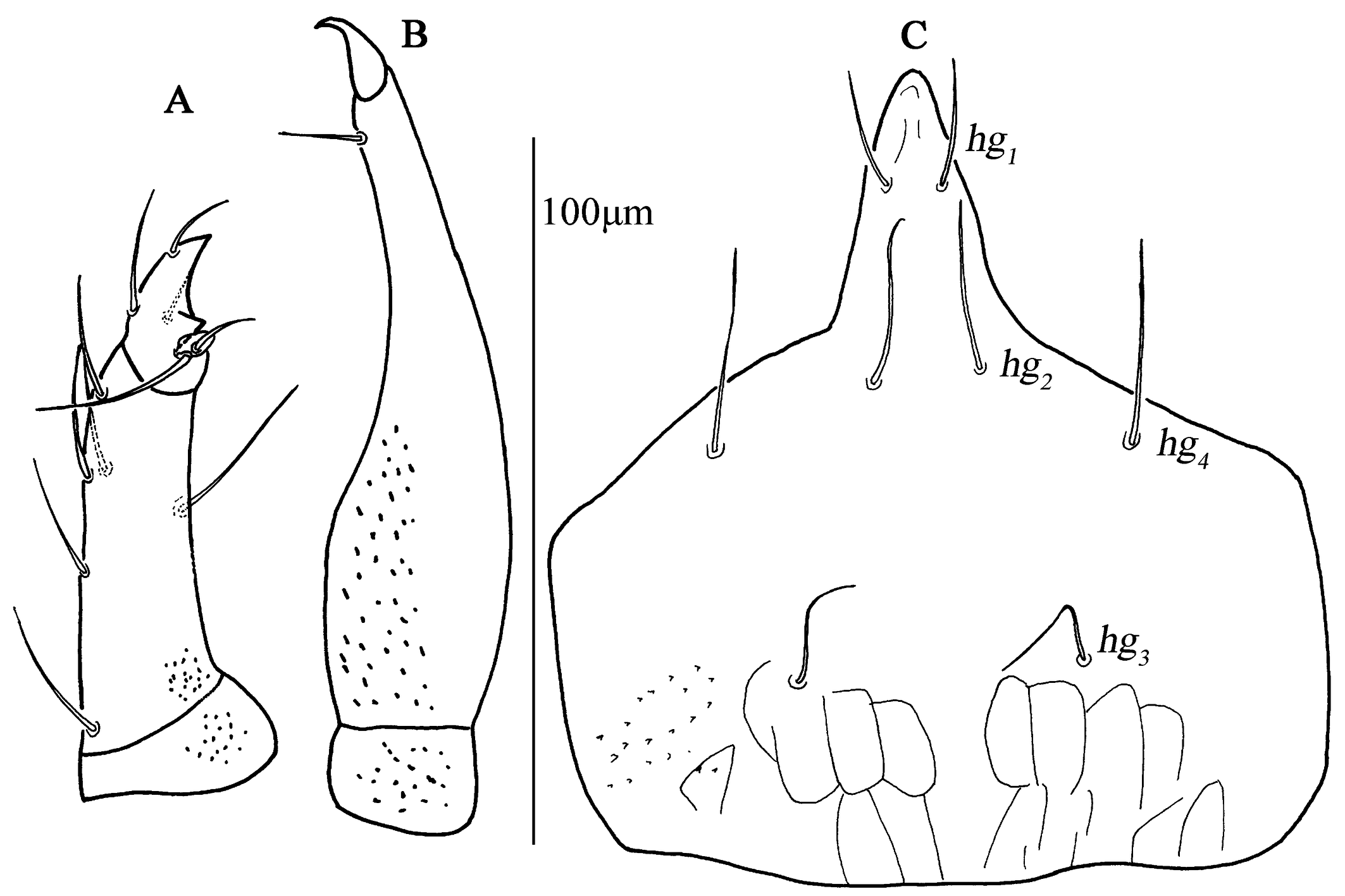
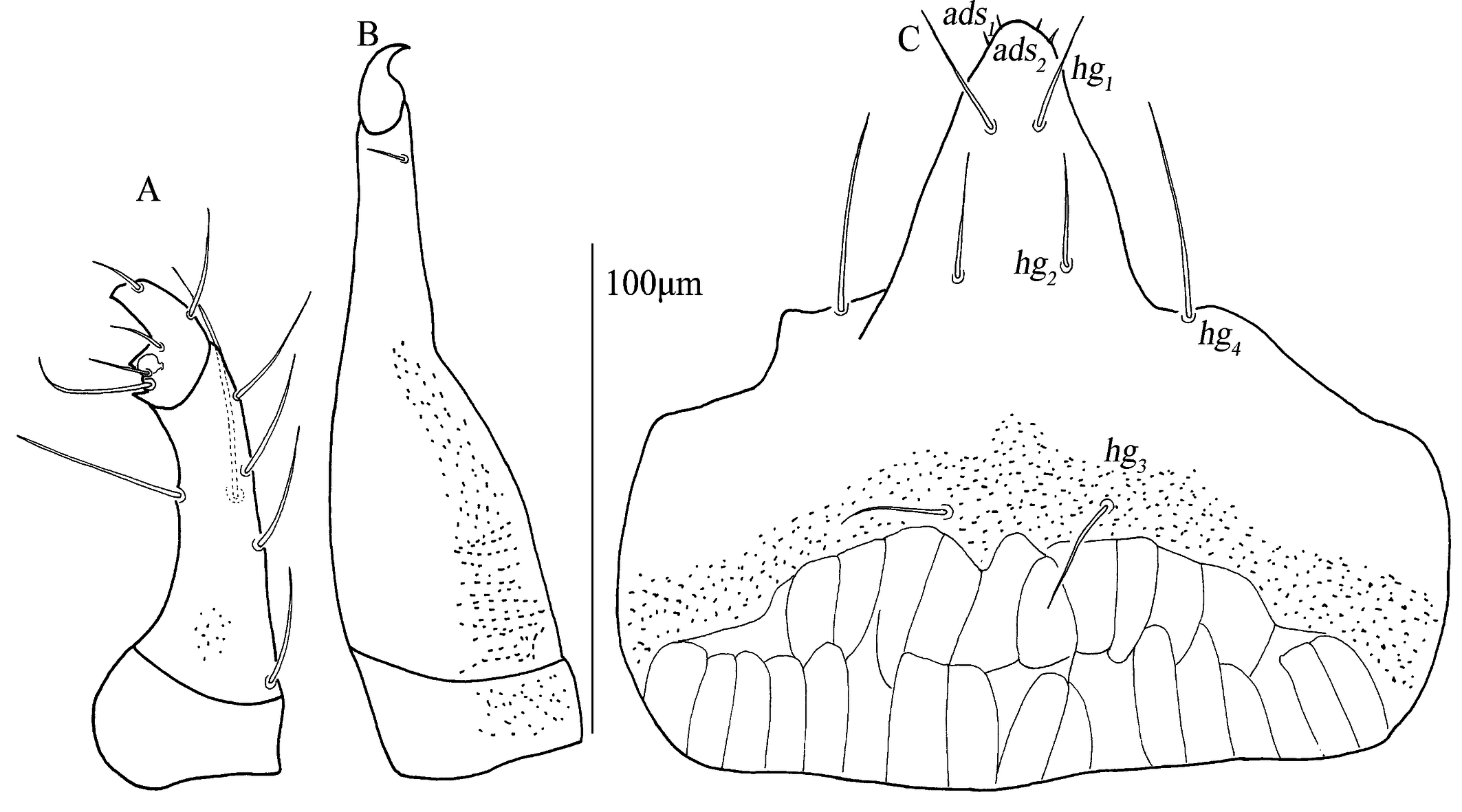
Subcapitulum (Fig. 3C). 182 (170–187) long, 172 (161–169) wide; between hg3 and the base area with two row polygonal pattern; adoral setae absent; four pairs of hypognathal setae (hg1 –hg4 ), lengths of hg1 –hg4 : 27 (25–27), 27 (27–28), 10 (10–22) and 45 (34–45), respectively. Distances of hg setae: hg1 –hg1 13 (13–13), hg2 –hg2 22 (22–23), hg3 –hg3 63 (54–63), hg4 –hg4 82 (82–87), hg1 –hg2 38 (38–38), hg2 –hg3 58 (50–58), hg3 –hg4 46 (46–46).
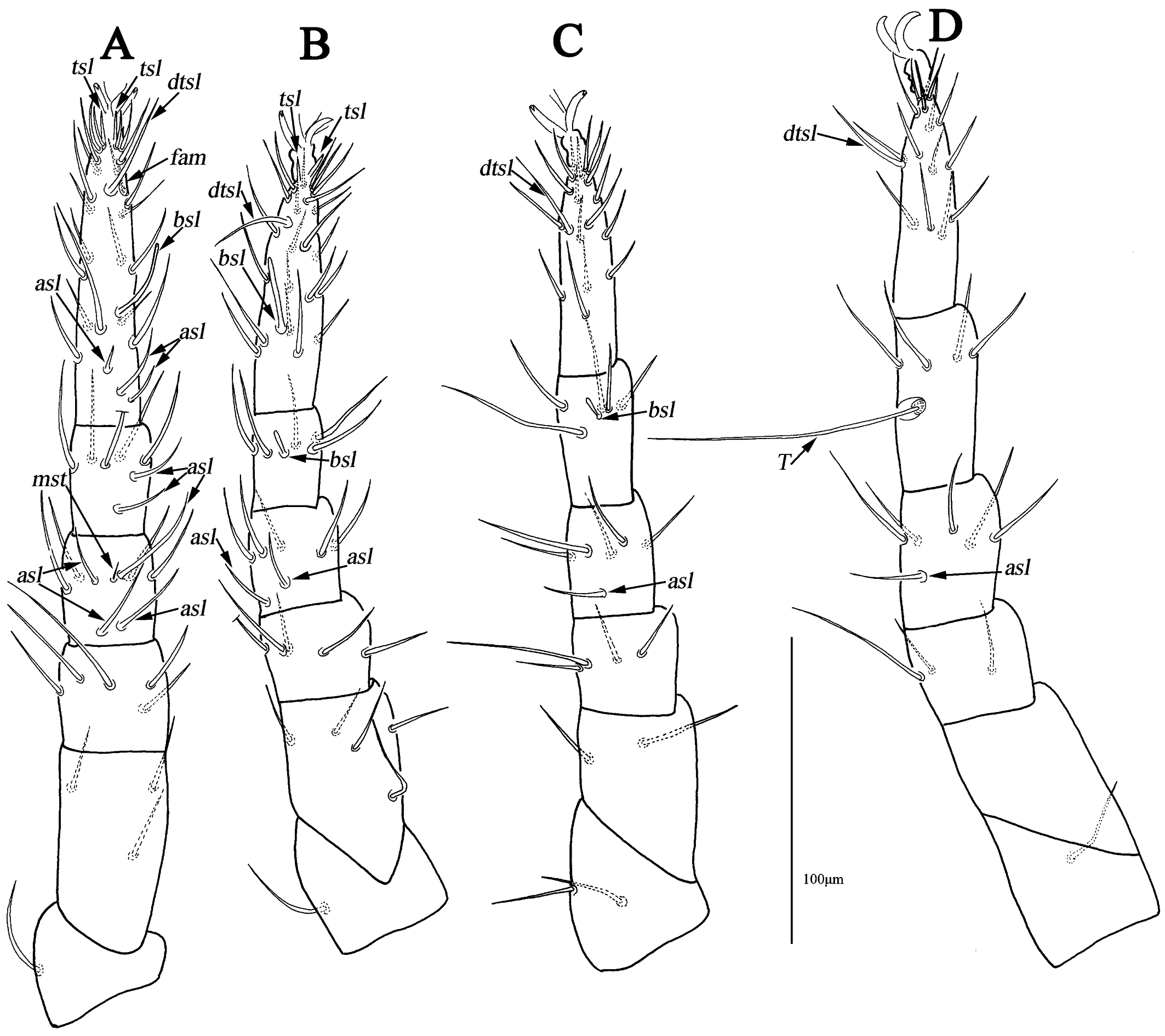


Legs (Figs. 4A–D). Lengths of leg I–IV: 295 (281–295), 256 (241–269), 267 (256–268) and 294 (290–294). Lengths of tarsi I–IV: 100 (95–100), 85 (76–85), 73 (73–80) and 78 (78–80). Seta T smooth on tibia IV 93 (90–93) in length. Legs I–IV chaetotaxy: coxae I–IV 3-3-3-3 sts; trochanters I–IV 1-1-2-1 sts; basifemora I–IV 3-5-2-0 sts; telofemora I–IV 5-5-4-3 sts; genua I–IV 3asl, \{1asl, 1mst\}, 4sts-2asl, 5sts-1asl, 5sts-1asl, 5sts; tibiae I–IV 2asl, 5sts-1bsl, 5sts-1bsl, 5sts-1 smooth T, 4sts; tarsi I–IV 3asl, 1bsl, 1fam, 1dtsl, 2tsl, 21sts-1bsl, 1dtsl, 2tsl, 22sts-1dtsl, 17sts-1dtsl, 15sts.
Male (n=2)
Idiosoma 273–290 long, 192–238 wide.




Dorsum (Figs. 5A, 6A–B). Resembling the female, dorsocentral shield 246–258 long, 180–202 wide. Setal lengths and distances: at 100–108, pt 104–111, lps 36–52, mps 27–29, c1 11–14, c2 11–15, d1 15–15, e1 26–37, f1 37–42; at–at 32–36, pt–pt 118–118, lps–lps 48–54, mps–mps 88–93, lps–mps 58–59, at–lps 22–26, pt–mps 10–11, pt–lps 61–64, at–mps 80–84, at–pt 86–88, c1 –c1 51–59, c2 –c2 155–158, d1 –d1 54–58, e1 –e1 35–38, c1 –c2 46–46, c1 –d1 42–46, c2 –d1 59–61, d1 –e1 65–68, e1 –f1 36–37.
Venter (Figs. 4B, 6C–D). Resembling the female, platelet absent anterior to genital plates; one pair of small platelets anterolateral situated in genital plates. Setal formula of coxal plates I–IV: 3(1a–c)-3(2a–c)-3(3a–c)-2(4a–b) sts, lengths of setae: 1a 10–12, 1b 14–15, 1c 15–16, 2a 11–14, 2b 11–17, 2c 15–18, 3a 10–10, 3b 12–15, 3c 16–24, 4a 9–10, 4b 11–12, 4c 12–12. One pair of hysterogastral setae (hgs1 ), 11–11 in length. Genital shields relatively small, 45–50 long, 16–16 wide; lengths of setae g1 –g4 : 8–9, 8–9, 8–9, 9–11; one pair of aggenital setae (ag) 11–11 in length; ps1 and ps2 subequal 8–9 in length, h1 15–21 and h2 13–16.


Gnathosoma. Palp (Fig. 7A). Resembling the female, 74–82 long.
Chelicera (Fig. 7B). Resembling the female, 113–118 long; cheliceral seta 8–10 in length.
Subcapitulum (Fig. 7C). Resembling the female, 114–120 long, 98–103 wide; lengths of hg1 –hg4 : 18–20, 21–22, 12–15 and 24–26. Distances of hg setae: hg1 –hg1 7–8, hg2 –hg2 12–16, hg3 –hg3 38–41, hg4 –hg4 45–58, hg1 –hg2 26–28, hg2 –hg3 42–45, hg3 –hg4 33–37.
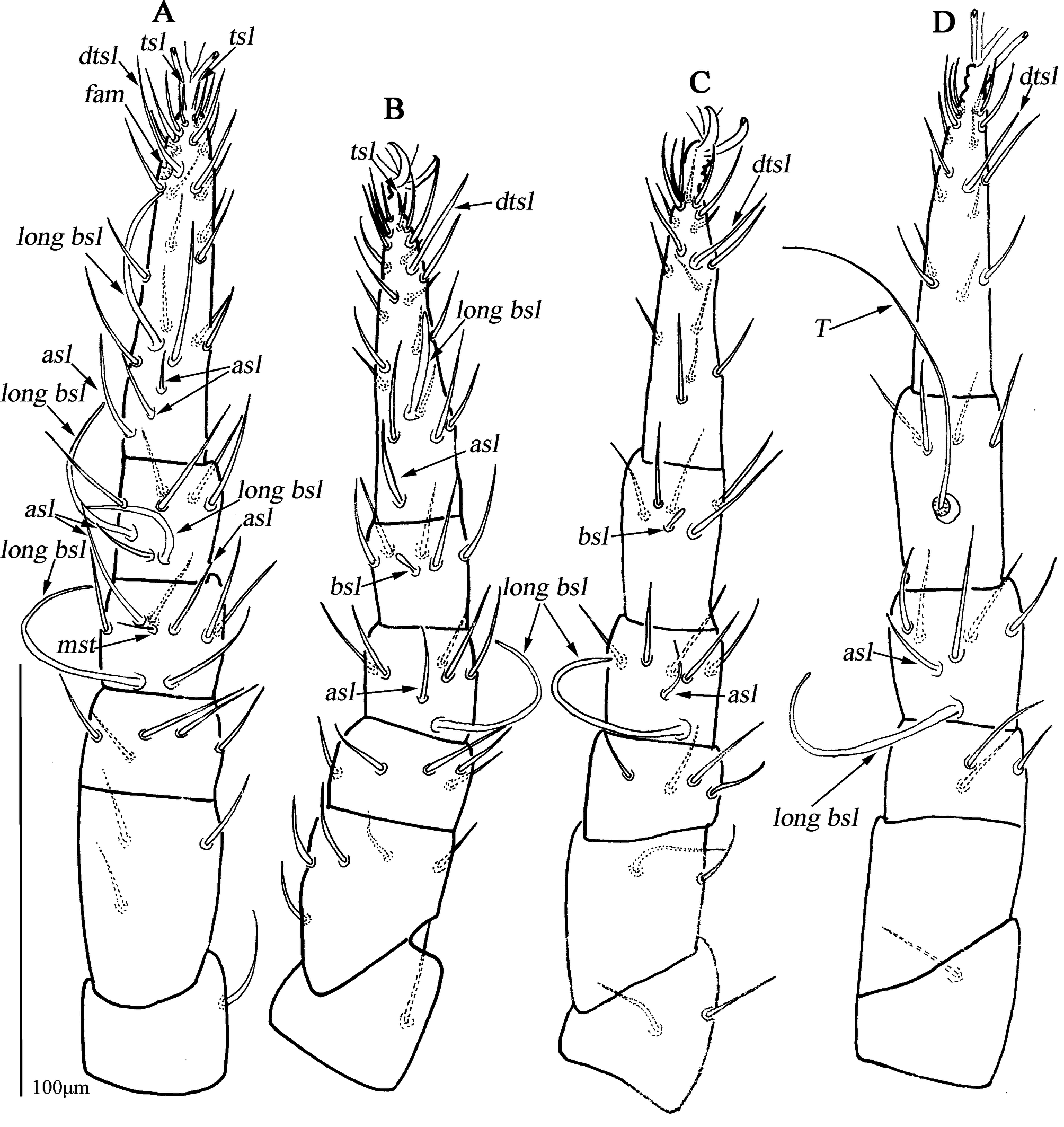


Legs (Fig. 8A–D). Resembling the female, lengths of leg I–IV: 212–224, 180–201, 199–216 and 222–231. Lengths of tarsi I–IV: 80–81, 66–68, 60–67 and 68–73. Seta T 72–74 in length. Legs I–IV chaetotaxy: coxae I–IV 3-3-3-3 sts; trochanters I–IV 1-1-2-1 sts; basifemora I–IV 3-5-2-0 sts; telofemora I–IV 5-5-4-3 sts; genua I–IV 2asl, 1 long bsl, \{1asl, 1mst\}, 4sts-1asl, 1 long bsl, 5sts-1asl, 1 long bsl, 5 sts-1asl, 1 long bsl, 5sts; tibiae I–IV 1asl, 2 long bsl, 5sts-1bsl, 5sts-1bsl, 5sts-1 smooth T, 4sts; tarsi I–IV 3asl, 1 long bsl, 1fam, 1dtsl, 2tsl, 21sts-1asl, 1 long bsl, 1dtsl, 1tsl, 21sts-1dtsl, 17sts-1dtsl, 16sts.
Other developmental stages. Unknown.
Etymology
The new species name is derived from type locality Huangshan Scenic Spot, Huangshan City, Anhui Province, P. R. China.
Remarks
This new species is similar to N. fani Lin, 2001, N. boltoides Lin, 2001, N. zuluensis Den Heyer, 1980, N. lajumensis Den Heyer, 1980, and N. philippinensis (Corpuz-Raros, 2007) (known only from adult female) in having coxae I–II fused medially as a V-shaped single sternal plate (the remaining species coxae I–II not fused medially in this genus)
The new species differs from N. fani by: (1) basifemora I–IV 3-5-2-0 sts (vs. 3-5-2-1 sts in N. fani); (2) tibia III with 1bsl, 5sts (vs. 0bsl, 5sts in N. fani); (3) genital plates with reticulate pattern (vs. without in N. fani); (4) one pair of small platelets posterolateral situated in genital plates present (vs. absent in N. fani); (5) seven hysterogastral setae (vs. six in N. fani). According to the original description, N. fani coxae I–IV 3-3-3-3 sts on the left and 3-3-3-4 sts on the right; We think it's 3-3-3-3 sts, except that the left hgs1 is not on lateral ventral plate and the right hgs1 is on lateral ventral plate.
The new species differs from N. boltoides by: (1) basifemora I–IV 3-5-2-0 sts (vs. 3-4-2-0 sts in N. boltoides); (2) tibia III with 1bsl, 5sts (vs. 0bsl, 5sts in N. boltoides); (3) posterior edge of coxae IV extending beyond edge of genital plates (vs. not extending beyond in N. boltoides); (4) one pair of small platelets posterolateral situated in genital plates present (vs. absent in N. boltoides).
The new species differs from N. zuluensis by: (1) basifemora I–IV 3-5-2-0 sts, (vs. 5-5-2-0 sts in N. zuluensis); (2) telofemora I–IV 5-5-4-3 sts (vs. 3-5-4-3 sts in N. zuluensis); (3) small platelet anteriomedially of genital plates present (vs. absent in N. zuluensis); (4) one pair of small platelets posterolateral situated in genital plates present (vs. absent in N. zuluensis).
The new species differs from N. lajumensis by: (1) basifemora I–IV 3-5-2-0 sts, (vs. 5-5-4-1 sts in N. lajumensis); (2) telofemora I–IV 5-5-4-3 sts (vs. 3-5-5-3 sts in N. lajumensis); (3) small platelet anteriomedially of genital plates present (vs. absent in N. lajumensis); (4) one pair of small platelets posterolateral situated in genital plates present (vs. absent in N. lajumensis); (5) setae lps longer than mps in length (vs. equal in N. lajumensis).
The new species differs from N. philippinensis by: (1) basifemora I–IV 3-5-2-0 sts, (vs. 4-4-2-0 sts in N. philippinensis); (2) proterosomal and hysterosomal shields fused, forming a large dorsocentral shield and with setae c1 , c2 , d1 , e1 (vs. hysterosomal shield absent and setae c1 , c2 , d1 , e1 on integument sts in N. philippinensis); (3) posterior edge of coxae IV extending beyond edge of genital plates (vs. not extending beyond in N. philippinensis); (4) small platelet anteriomedially of genital plates present (vs. absent in N. philippinensis); (5) one pair of small platelets posterolateral situated in genital plates present (vs. absent in N. philippinensis).
Type series
Holotype, female was collected from moss, Huangshan Scenic Spot (N30°8′19.80″, E118°9′8.35″, elevation 1240 m), Huangshan City, Anhui Province, P. R. China, on 23 May, 2018, by Mao-Yuan Yao, slide No., AH-CU-201805232001. Paratypes, two females and one male the same data as the holotype, slides No.: AH-CU-201805232002–AH-CU-201805232004. Paratypes, one female and one male were collected from moss, Wangjiabang (N32°27′32″, E106°13′13″, elevation 926 m), Ningqiang county, Hanzhong city, Shanxi Province, P. R. China, on 15 July, 2018, by Jian-Xin Chen, slides No.: SX-CU-201807151801–SX-CU-201807151802. All types are deposited in the Institute of Entomology, Guizhou University, Guiyang, P. R. China (GUGC).
Distribution
Anhui Province (Huangshan City), Shanxi Province (Hanzhong City), P. R. China.
Neocunaxiodes pinus Chen & Jin sp. nov.
ZOOBANK: 5090981C-51D8-498D-B659-9E145782206B ![]()
(Figs. 9–12)
Diagnosis
Subcapitulum with papillar striation and between hg3 and the base area with two row polygonal pattern; dorsal shield foveolae and reticulate pattern absent; between coxae II median small platelet absent; anterior to genital plates with a small platelet; setal formula of coxal plates I–IV: 2-2(1)-3-2 sts; combined femora (basifemora + telofemora) I–IV: 8-10-6(4)-4 sts.
Description
Female (n=2)
Idiosoma 390 (402) long, 295 (288) wide.
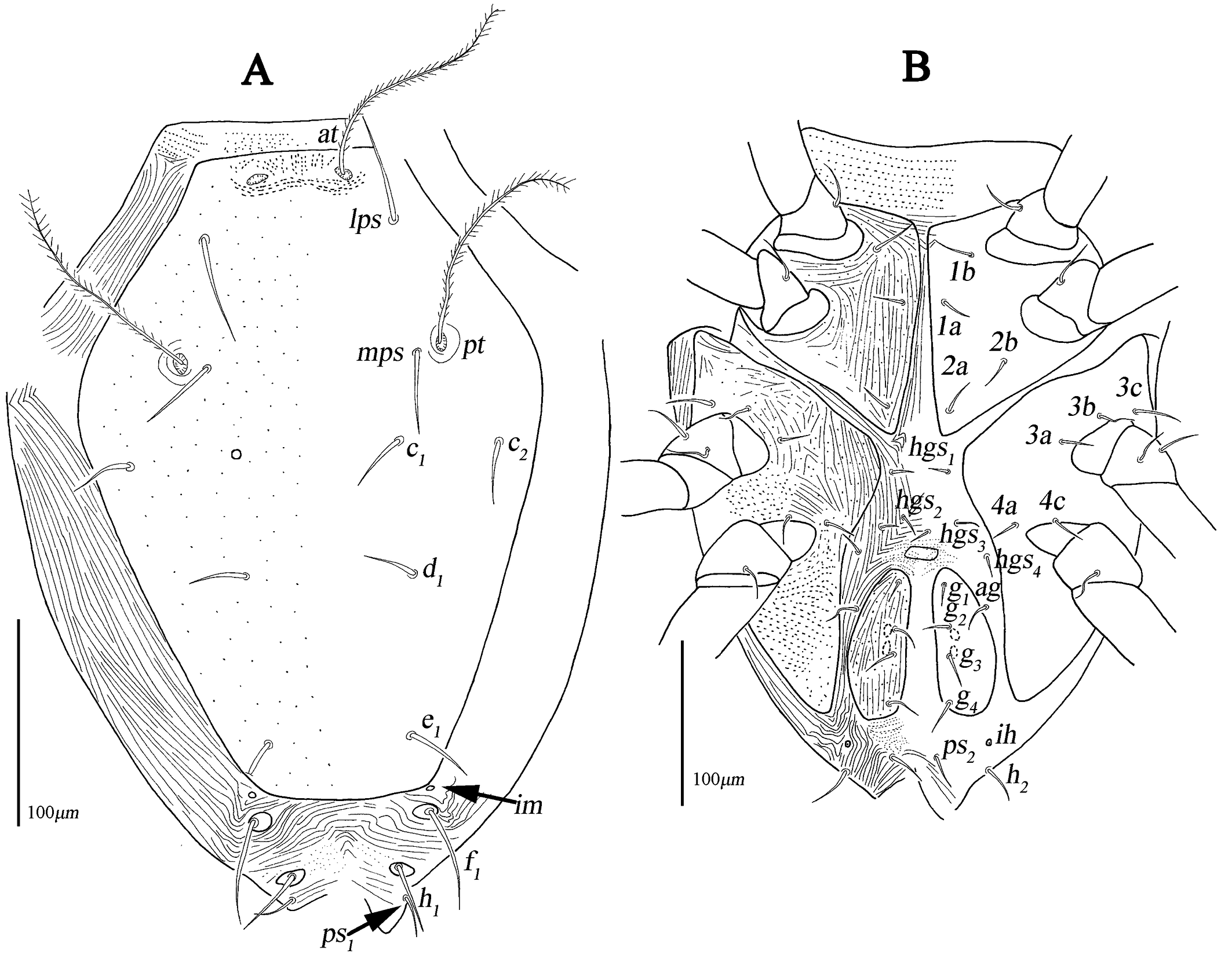


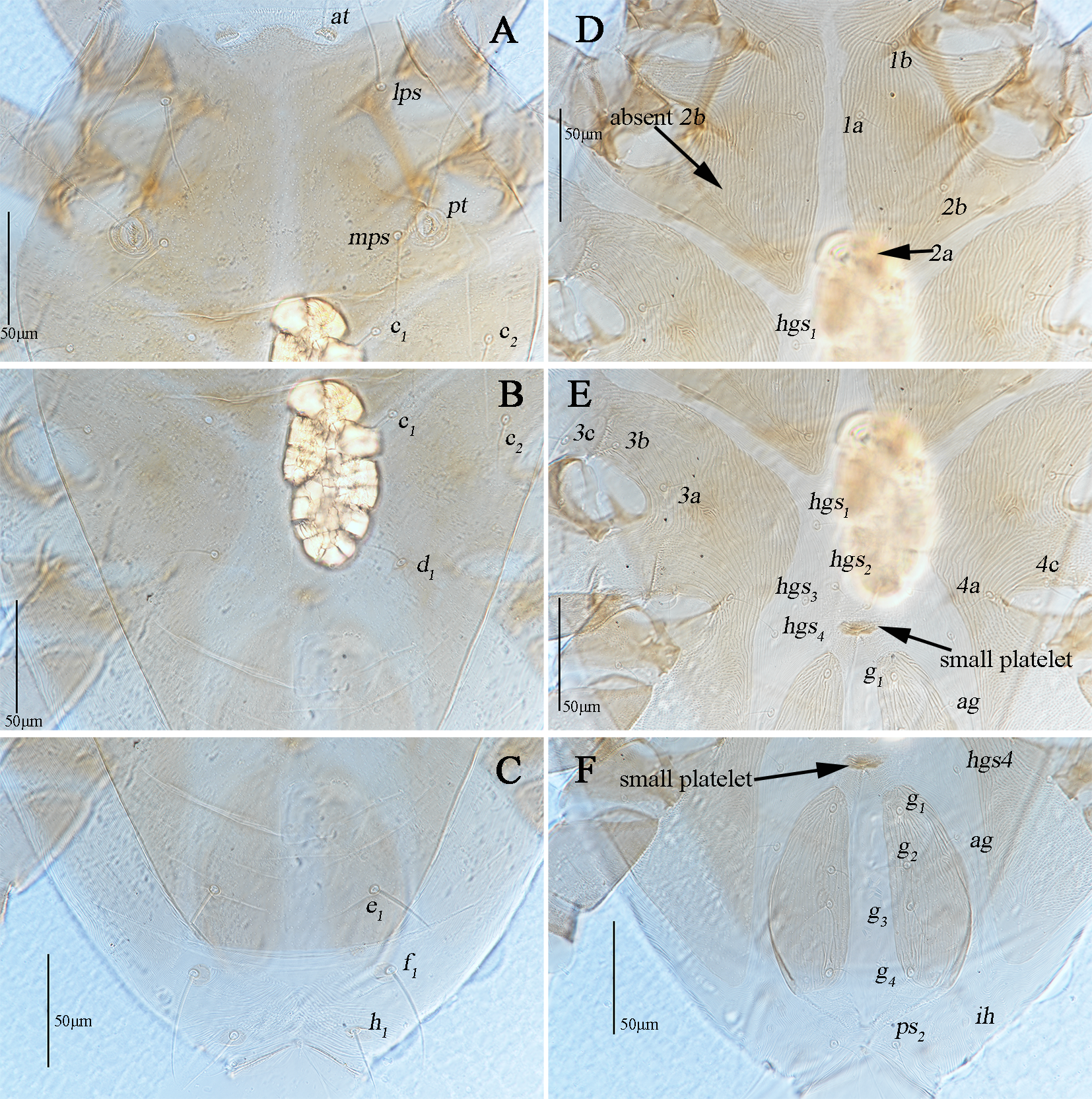


Dorsum (Figs. 9A, 10A–C). Proterosomal and hysterosomal shields fused, forming a large dorsocentral shield, 313 (320) long, 227 (235) wide, with random punctation, bearing two pairs of trichobothria (at and pt), two pairs of tactile setae (lps and mps), four pairs of simple setae (c1 , c2 , d1 , e1 ); f1 and h1 situated on small platelets, lateral area outside dorsocentral shield with lengthwise striation; area between e1 and h1 with transverse striae; one pair of lyrifissures (im) situated on lateral posterior to dorsocentral shield. Setal lengths and distances: at 114 (118), pt 120 (123), lps 52 (47), mps 42 (40), c1 24 (22), c2 26 (24), d1 23 (23), e1 32 (31), f1 50 (52), h1 36 (37), at–at 43 (40), pt–pt 129 (135), lps–lps 96 (101), mps–mps 102 (106), lps–mps 66 (69), at–lps 38 (40), pt–mps 14 (15), pt–lps 61 (58), at–mps 95 (93), at–pt 95 (96), c1 –c1 79 (82), c2 –c2 183 (180), d1 –d1 82 (80), e1 –e1 70 (72), f1 –f1 87 (85), h1 –h1 54 (50), c1 –c2 50 (47), c1 –d1 65 (69), c2 –d1 78 (83), d1 –e1 78 (79), e1 –f1 37 (42), f1 –h1 30 (28).
Venter (Figs. 9B, 10D–F). Ventral area between coxae I and the gnathosoma with papillar striation. Coxae I–II divided medially to form sternal plates and with random punctation and striae; coxae III–IV fused to form paired lateral ventral plates and with random punctation, striae and papillar striation. Area between coxae plates I–II with longitudinal striation, between edges of divided sternal plates without a small median platelet. Posterior edge of coxal plates IV extending and almost reached to g4 level. Setal formula of coxal plates I–IV: 2(1a–b)-2(2a–b) (2b absent on the left)-3(3a–c)-2(4a, 4c) sts, length of setae: 1a 15 (14), 1b 23 (24), 2a 14 (15), 2b 13 (12), 3a 17 (18), 3b 18 (18), 3c 24 (26), 4a 11 (12), 4c 12 (12). Four pairs of hysterogastral setae (hgs1 –hgs4 ) subequal 11–12 in length, area between hgs1 and with transverse striation. Anterior to genital plates with a small platelet, long 17 (19), wide 10 (11). Genital plates 93 (88) long, 35 (33) wide, with broken longitudinal striation and random punctation, two pairs of visible genital papillae, four pairs of genital setae (g1 –g4 ) arranged longitudinally that 15 (13), 16 (15), 14 (15) and 18 (19) in length, respectively; one pair of aggenital setae (ag), 13 (12) in length, almost at g2 level. Anal region with two pairs of pseudanal setae (ps1 –ps2 ) 15 (14) and 18 (20) in length, one pair of h2 26 (29) and one pair of lyrifissures (ih).


Gnathosoma. Palp (Fig. 11A). Three-segmented, 107 (111) long. Palp chaetotaxy: trochanter none; femurogenu with six simple setae; tibiotarsus with five simple setae, two pointed apophysis and one knob-like apophysis; claw well-developed and simple.
Chelicera (Fig. 11B). 160 long, with strong punctations; one cheliceral seta 10 (12) in length; chela developed.
Subcapitulum (Fig. 11C). 158 (145) long, 180 (182) wide with papillar striation; between hg3 and the base area with two row polygonal pattern; two pairs of short adoral setae; four pairs of hypognathal setae (hg1 –hg4 ), lengths of hg1 –hg4 : 20 (18), 23 (22), 20 (23) and 40 (43), respectively. Distances of hg setae: hg1 –hg1 12 (9), hg2 –hg2 23 (26), hg3 –hg3 36 (40), hg4 –hg4 77 (73), hg1 –hg2 30 (32), hg2 –hg3 49 (52), hg3 –hg4 41 (44).
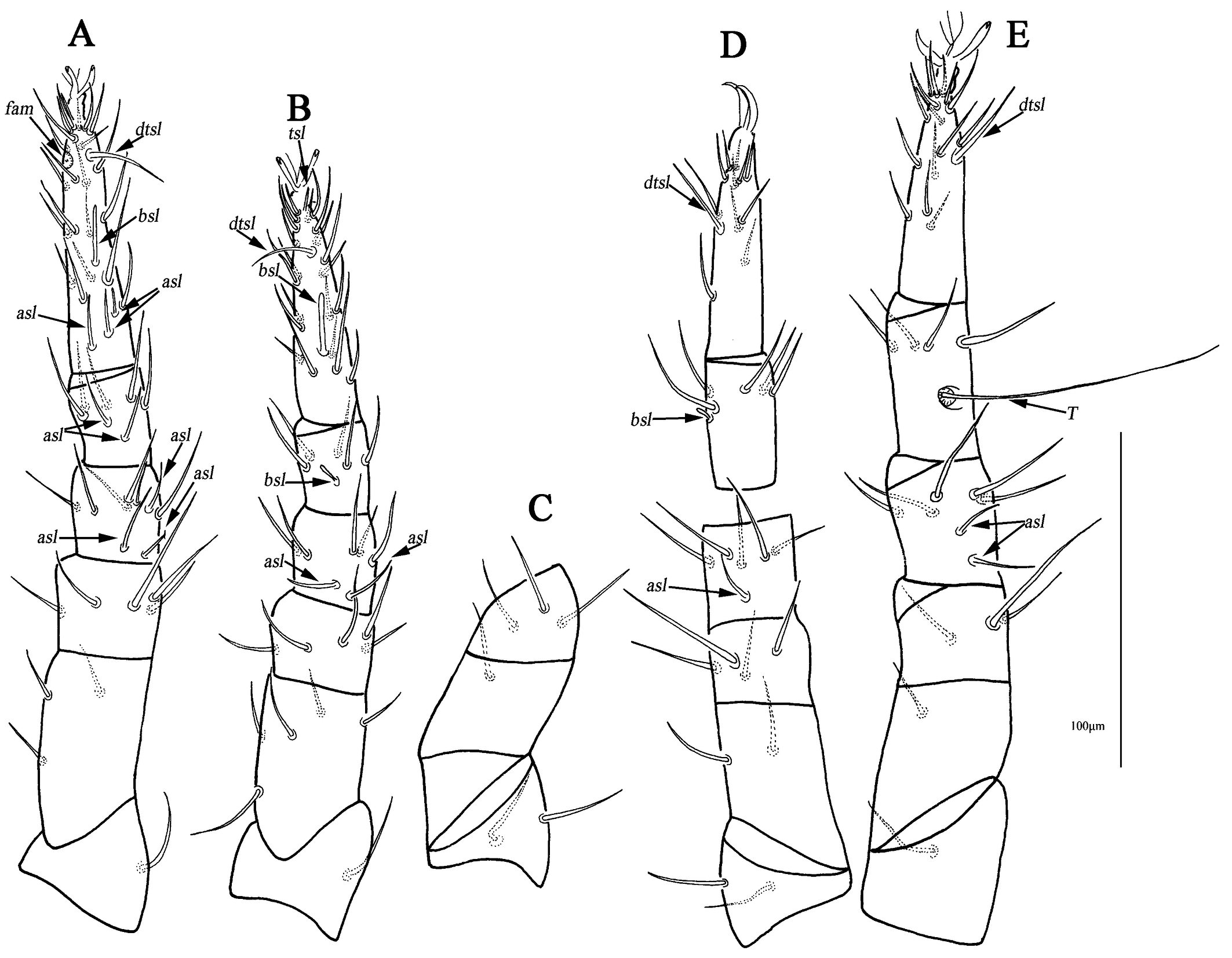


Legs (Fig. 12A–E). Lengths of leg I–IV: 247 (245), 218 (221), 235 (240) and 268 (271). Lengths of tarsi I–IV: 81 (75), 68 (67), 69 (67) and 72 (77). Seta T smooth on tibia IV 90 (93) in length. Legs I–IV chaetotaxy: coxae I–IV 2-2(1) (2b absent on the left)-3-2 sts; trochanters I–IV 1-1-2-1 sts; basifemora I–IV 3-5-2(1) (two setae on the right, one on the left)-1 sts (Figs. 9C–D); telofemora I–IV 5-5-4(3) (four setae on the right, three on the left)-3 sts (Figs. 9C–D); genua I–IV 4asl, 5sts-2asl, 5sts- 1asl, 5sts- 2asl, 5sts; tibiae I–IV 2asl, 5sts-1bsl, 5sts-1bsl, 5sts-1 smooth T, 4sts; tarsi I 2asl, 2bsl, 1fam, 1dtsl, 2tsl, 21sts-1bsl, 1dtsl, 1tsl, 21sts-1dtsl, 11sts-1dtsl, 15sts.
Other developmental stages. Unknown.
Etymology
The new species name derives from the habitat (fallen leaves of Pinus yunnanensis) where the type specimens were collected.
Remarks
The new species is similar to Neocunaxoides kalamiensis Inayatullah & Shahid, 1989 (adult female) in having coxae I–II divided medially to form sternal plates and coxae I–IV 2-2-3-2 sts, but it differs from the latter by the following features: (1) anterior to genital plates with a small platelet (vs. without in N. kalamiensis); (2) subcapitulum between hg3 and the base area with two row polygonal pattern (vs. without in N. kalamiensis); (3) combined femora (basifemora + telofemora) I–IV 8-10-6(4)-4 sts (vs. 9-10-7-4 in N. kalamiensis).
Type series
Holotype, female was collected fallen leaves of Pinus yunnanensis, Yulong Snow Mountain (N26°59′56.44″, E100°11′53.41″, elevation 2670 m), Lijiang City, Yunnan Province, P. R. China, on 27 August, 2018, by Jian-Xin Chen and Juan Li, slide No., YN-CU-201808270401. Paratype, one female the same data as the holotype, slides No.: YN-CU-201808270402. Slides is deposited in the Institute of Entomology, Guizhou University, Guiyang, P. R. China (GUGC).
Distribution
Yunnan Province (Lijiang City), P. R. China
Key to species of Neocunaxides from China (adult females)
1. Coxae I–II fused medially as a single sternal plate
...... 2
— Coxae I–II divided medially to form sternal plates
...... 4
2. Posterior edge of coxal plates IV extending beyond anterior edge of genital plates
...... 3
— Posterior edge of coxal plates IV not extending beyond anterior edge of genital plates
...... N. boltoides Lin, 2001
3. Basifemora IV with 0 sts
...... N. huangshanensis Chen & Jin sp. nov.
— Basifemora IV with 1 sts
...... N. fani Lin, 2001
4. Median platelet between coxae II absent
...... N. pinus Chen & Jin sp. nov.
— Median platelet between coxae II present
...... 5
5. One small platelet anteriomedially of genital plates presen
...... N. ovatus Lin, Zhang & Ji, 2003
— One small platelet anteriomedially of genital plates absent
...... N. andrei (Baker & Hoffmann, 1948)
Acknowledgements
This work was supported by Guizhou Provincial Science and Technology Projects (QKHJC [2024] youth 289 and 410), Guizhou Provincial Science and Technology Projects (QKHJC-ZK[2023]023), National Natural Science Foundation of China (31272357), the Natural Science Research Project of Guizhou Provincial Education Department (Youth Science and Technology Talent Development Programs) (Qianjiaoji [2024] 215), and Key Laboratory of Information Technology for Plant Protection in Characteristic High-Efficiency Agriculture of Central Guizhou (Qianjiaoji [2022] No. 052).
References
- Alatawi, F.J. & Kamran, M. 2017. Predatory prostigmatid mite (Acari: Trombidiformes) fauna of the date palm agro-ecosystem in Saudi Arabia. Systematic and Applied Acarology, 22(9): 1444-1475. https://doi.org/10.11158/saa.22.9.11
- Al-Azzazy, M. M. & Al-Rehiayani, S. M. 2022. The soil mite Cunaxa capreolus (Acari: Cunaxidae) as a predator of the root-knot nematode, Meloidogyne incognita and the citrus Nematode, Tylenchulus semipenetrans: Implications for biological control. Acarologia, 62(1): 174-185. https://doi.org/10.24349/lo4p-42kf
- Baker, E.W. & Hoffmann, A. 1948. Acaros de la familia Cunaxidae. Anales de la Escuela Nacional de Ciencias Biologicas Mexico, 5(3-4): 229-273.
- Castro, T.M.M.G. & Den Heyer, J. 2009. A revision of the genus Pulaeus Den Heyer, with description of a new genus and four new Brazilian species (Acari: Prostigmata: Cunaxidae). Zootaxa, 2141: 20-36. https://doi.org/10.11646/zootaxa.2141.1.2
- Corpuz-Raros, L.A. 1996. Philippine predatory mites of the family Cunaxidae (Acari). 6. Genus Neocunaxoides Smiley with a new species record from Central Kalimantan, Borneo, Indonesia. Asia Life Sciences, 5(2): 125-140
- Corpuz-Raros, L.A. 2007. Additional species of Bonziinae and Cunaxoidinae and description of the male Coleoscirus horidula (Tseng) (Coleoscirinae) from the Philippine (Cunaxidae, Acari). Asia Life Sciences, 16(2): 153-173.
- Corpuz-Raros, L.A. & Gruèzo, W.S. 2007. Two new species of predatory mites of the family Cunaxidae (Acari) from Java, Indonesia. Asia Life Sciences, 16(2): 175-181
- Chen, J.X., Guo, J.J., Yi, T.C. & Jin, D.C. 2020. A new species of genus Lepidocunaxides with an updated diagnosis of the genus. Systematic and Applied Acarology, 25(2): 178-192. https://doi.org/10.11158/saa.25.2.2
- Den Heyer, J. 1978. A new cunaxid subfamily and neotype designation of Cunaxoides croceus (Koch, 1838) (Prostigmata: Acari). Acarologia, 20(3): 338-350.
- Den Heyer, J. 1980a. Three new Afrotropical species of Neocunaxoides Smiley (Actinedida: Acarida). Phytophylactica, 12(3): 129-146.
- Den Heyer, J. 1980b. A classification system for the family Cunaxidae (Actinedida: Acarida). Publications of the University of the North, series A, 23: 1-12.
- Den Heyer, J. 1981. Systematics of the family Cunaxidae Thor, 1902 (Actinedida: Acarida). Publication of the University of the North, series A, 24: 1-19.
- Fawzy, M.M.H. 2007. Two new species of cunaxid mites on cotton and castor plants from Egypt (Actinedida: Cunaxidae). Egyptian Journal of Agricultural Research, 85(6): 2065-? https://doi.org/10.21608/ejar.2007.211340
- Fisher, J.R., Skvarla, M.J., Bauchan, G.R., Ochoa, R. & Dowling, A.P.G. 2011. Trachymolgus purpureus sp. n., an armored snout mite (Acari, Bdellidae) from the Ozark highlands: morphology, development, and key to Trachymolgus Berlese. ZooKey, 125: 1-34. https://doi.org/10.3897/zookeys.125.1875
- Gupta, S.K. 1991. Studies on the predatory prostigmatid mites of Northeast with descriptions of new species and new records from India. Records of the Zoological Survey of India, 88(1-4): 207-239 https://doi.org/10.26515/rzsi/v88/i2/1991/161345
- Gupta, S.K. & Chattopadhyay, S. 1978. Studies on Acari associated with bird nests in Bengal, India. International Journal of Acarology, 2: 77-86
- Hernandes, F.A., Castro, T.M.M.G. & Venancio, R. 2015. Prostigmata (Acari: Trombidiformes) as biological control agents. pp. 151-184, Chapter 6 in Carrillo, D., Moraes, G.J. & Peña, J.E. (Eds.), Prospects for Biological Control of Plant Feeding Mites and Other Harmful Organisms. Springer, PP. 151-184. https://doi.org/10.1007/978-3-319-15042-0_6
- Hu, S.J. 1997. Cunaxid mites recorded in China. Journal of Ninbo Teachers College, 15(1): 56-59. [in Chinese]
- Inayatullah, I. & Shahid, M. 1989. Two new predatory mites of genus Neocunaxoides Smiley (Acarina: Cunaxidae) from Pakistan. Pakistan Journal of Zoology, 21(3): 221-228.
- Kalúz, S. & Ermilov, S.G. 2018. A new species of the genus Neocunaxoides (Acari: Prostigmata: Cunaxidae) from Vietnam. Biologia, 73: 1223-1228. https://doi.org/10.2478/s11756-018-0140-4
- Kalúz, S. & Ermilov, S.G. 2019. A new genus of Pulaeini (Acari: Prostigmata: Cunaxidae) from South-East Asia. Zootaxa, 4619(2): 382-390. https://doi.org/10.11646/zootaxa.4619.2.12
- Kuznetzov, N.N. & Livshitz, I.Z. 1979. Predatory mites of the Nikita Botanical Gardens (Acariformes: Bdellidae, Cunaxidae Camarobiidae). Trudy Gosudarstvennogo Nikitskogo Botanicheskogo Sada, 79: 51-105.
- Lin, J.Z., Zhang, Y.Z. & Ji, J. 2001. Three new species of Cunaxidae from Fujian, China (Acari: Cunaxidae). Systematic and Applied Acarology, 6: 145-153. https://doi.org/10.11158/saa.6.1.19
- Lin, J.Z., Zhang, Y.Z. & Ji, J. 2003. A new species of Neocunaxoides from Fujian, China (Acari: Cunaxidae). Systematic and Applied Acarology, 8: 101-106. https://doi.org/10.11158/saa.8.1.13
- Lin, J.Z. & Zhang, Z.Q. 2010. Bdelloidea of China: a review of progress on systematics and biology, with a checklist of species. Zoosymposia, 4, 42-50. In: Zhang, Z.Q., Hong, X.Y., Fan, Q.H., (Eds.) Xin, J.L. Centenary: Progress in Chinese Acarology. Zoosymposia, 4: 1-345 https://doi.org/10.11646/zoosymposia.4.1.3
- Michocka, S. 1987. Mites (Acari) of the Bdellidae and Cunaxidae families in Poland. Monografie Fauny Polski, 14: 1-127
- Paktinat-Saeij, S., Bagheri, M., Castro, T.M.M.G., Saboori, A., Gharekhani, G. & Ghobari, H. 2017. New species and records of Cunaxinae mites (Acari: Trombidiformes: Cunaxidae) from Iran. Systematic and Applied Acarology, 22(9): 1277-1294. https://doi.org/10.11158/saa.22.9.1
- Paktinat-Saeij, S., Castro, T.M.D., Bagheri, M., Skvarla, M.J. & Moraes, G.J. 2016. Two new species and eight new combinations of Pulaeini Berelese (Acari: Cunaxidae) from Iran, with key to species of Lupaeus and Pulaeus in the world. Systematic and Applied Acarology, 21(6): 778-790. https://doi.org/10.11158/saa.21.6.5
- Romeih, A.H.M. and Abo-Shnaf, R.I.A. 2006. A NEW SPECIES OF THE GENUS NEOCUNAXOIDES (Acari: Cunaxidae) IN EGYPT. Journal of Plant Protection and Pathology, 31(12): 7973-7979. https://doi.org/10.21608/jppp.2006.235363
- Rocha, M.S., Rodrigues, E.N.L. & Ferla, N.J. 2015. New species and records of cunaxid mites (Acari: Cunaxidae) from soil in Southern Brazil. Zootaxa, 3981(1): 056-070. https://doi.org/10.11646/zootaxa.3981.1.2
- Shiba, M. 1978. Taxonomic investigation on free-living Prostigmara from the Malay Peninsula. Nature and Life in South East Asia, 7: 83-229.
- Shoala, S.M.E. and El Kady, G.A. 2009. The Cunaxid Mite Neocunaxoides andrei (Baker & Hoffmann) as a Biological Control Agent of the Root-Knot Nematode Meloidogyne javanica Chitwood. Acarines: Journal of the Egyptian Society of Acarology, 3(1): 55-58. https://doi.org/10.21608/ajesa.2009.4966
- Skvarla, M.J., Fisher, J.R. & Dowling, A.P.G. 2014. A review of Cunaxidae (Acariformes, Trombidiformes): Histories and diagnoses of subfamilies and genera, keys to world species, and some new locality records. ZooKeys, 418: 1-103. https://doi.org/10.3897/zookeys.418.7629
- Smiley, R.L. 1975. A generic revision of the mites of the family Cunaxidae (Acarina). Annals of the Entomological Society of America, 68(2): 227-244. https://doi.org/10.1093/aesa/68.2.227
- Smiley, R.L. 1992. The Predatory Mite Family Cunaxidae (Acari) of the World with a New Classification. West Bloomington, Michigan, Indira Publishing House, 356 pp.
- Thor, S. 1902. Zur Systematik der Acarinenfamilien Bdellidae Koch, 1842, Grube 1859, Eupodidae Koch, 1842 und Cunaxidae Sig Thor, 1902. Verhandlungen der kaiserlich-koniglichen zoologish-botanischen Gesellschaft in Wien, pp. 159-165.
- Tseng, Y.H. 1980. Taxonomical study of the mite family Cunaxidae from Taiwan (Acarina: Tromoidiformes). Quarterly Journal of the Taiwan Museum, 33(3-4): 253-277. [in Chinese]
- Walter, D.E. & Krantz, G.W. 2009. Collecting, rearing, and preparing specimens. In: Krantz, G.W. & Walter, D.E. (Eds.), A Manual of Acarology, 3rd edition. Lubbock, Tex., Texas Tech University Press, pp. 83-96.



2025-04-17
Date accepted:
2025-05-29
Date published:
2025-09-04
Edited by:
Akashi Hernandes, Fabio

This work is licensed under a Creative Commons Attribution 4.0 International License
2025 Chen, Jian-Xin; Yao, Mao-Yuan; Wu, You-Fang; Guo, Jian-Jun; Yi, Tian-Ci and Jin, Dao-Chao
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



