Development, reproduction and life table parameters of Tetranychus phaselus Ehara (Acari: Tetranychidae) on two bean host plants
Chen, Chen  1
; Pan, Wei2
; Tong, Xiaoqian3
; Song, Jing4
; Pan, Yang5
and Zhou, Peng
1
; Pan, Wei2
; Tong, Xiaoqian3
; Song, Jing4
; Pan, Yang5
and Zhou, Peng  6
6
1School of Life Sciences, Anqing Normal University, Anqing 246133, China.
2School of Life Sciences, Anqing Normal University, Anqing 246133, China.
3School of Life Sciences, Anhui University, Hefei 230031, China.
4School of Life Sciences, Anqing Normal University, Anqing 246133, China.
5School of Life Sciences, Anqing Normal University, Anqing 246133, China.
6✉ School of Life Sciences, Anqing Normal University, Anqing 246133, China.
2025 - Volume: 65 Issue: 1 pages: 197-203
https://doi.org/10.24349/men5-cyrnOriginal research
Keywords
Abstract
Introduction
Investigations into how host plants affect life history characteristics of pest herbivores help better understand their abundance and distribution, the knowledge of which is important for pest forecast and integrated pest management programs (Santamaría et al. 2018). Host plants vary in chemical traits such as toxins, nutritional quality and quantity, and physical traits such as tissue toughness, which affect development, reproduction, survival and population growth of herbivores (de Lima et al. 2017; Golizadeh et al. 2017; Santamaría et al. 2018; Puspitarini et al. 2021; Segura-Martínez et al. 2023).
Many spider mites in the family Tetranychidae (Acari: Prostigmata) are highly polyphagous pests worldwide. Studies have shown that different host plants affect development, reproduction and demographic parameters of spider mites such as Tetranychus urticae Koch, T. evansi Baker & Pritchard (Santamaría et al. 2018), T. ludeni Zacher (Adango et al. 2006), and T. phaselus Ehara (Ito et al. 2017). Tetranychus phaselus is a polyphagous pest distributed in China, Russia, Japan, and Korea, which is currently considered as a minor pest (Ehara, 1960; Zhao et al. 2013; Gotoh et al. 2015; Jin et al. 2018). It has been recorded to appear on at least 16 host plants including Gossypium spp. (Malvales: Malvaceae), Humulus lupulus L. (Rosales: Cannabaceae), Fatoua villosa (Thunb.) Nakai (Rosales: Moraceae) (Jin et al. 2018; Migeon and Dorkeld 2024), among which the soybeans Glycine max Merr. (Fabales: Fabaceae) and the common beans Phaseolus vulgaris L. (Fabales: Fabaceae) are two most economically important crops cultivated worldwide. Glycine max and P. vulgaris belong to Fabaceae, which are considered favorable host plants of many Tetranychus mites (van den Boom et al. 2003; Ito et al. 2017; Segura-Martínez et al. 2023; Weerawansha et al. 2023; Zhou et al. 2024). Glycine max has more nutritional components such as protein than P. vulgaris (Alcázar-Valle et al. 2020; Li et al. 2024) and may be a more suitable host plant for T. phaselus.
The increasing planting area of host plants may alter species composition and facilitate occurrence of particular pests (Jin et al. 2018). Due to its nutritional and health benefits, there is an increase of demand and cultivation of Fabaceae beans (FAO 2025). For example, the planting area of soybean increased from 6.8 million hectares in 2015 to 10 million hectares in 2022 in mainland China (NBSC 2025), and there is a steady increase of planting area of common beans in temperate countries including China (Wu et al. 2020), which may facilitate the occurrence of T. phaselus. Numerous studies used common bean plants as host species to test the performance of spider mites, for example, Gotoh et al. (2015) tested the fitness of four mites on P. vulgaris in response to different temperatures. However, the performances of many spider mites including T. phaselus on soybean plants are not clear. It is also not clear whether T. phaselus performs better on soybean than on common bean. Here we investigated development, reproduction, survival and life table parameters of T. phaselus on G. max and P. vulgaris and how these traits vary between the two plants. Knowledge generated could contribute to pest forecast and integrated pest management programs for controlling this mite in soybean and common bean growing regions.
Material and methods
Mite colony and experimental conditions
Tetranychus phaselus adults were collected on Morus alba L. in Anqing, Anhui province, China, and the colony was established and maintained using 3- to 5-week-old common bean P. vulgaris plants grown in pots. Phaseolus vulgaris and G. max seeds were purchased from local market and were grown in pots. The first expanded leaves cut from 1- to 2-week-old plants were used for experiments. The colony was maintained and the experiments were carried out under the environmental conditions of 24 ± 1 °C, 60 ± 10% RH, and 14:10 h (light:dark) photoperiod in a walk-in climate chamber.
Immature development
Forty adult females were randomly selected from the colony and placed on a P. vulgaris leaf square (3 × 3 cm). Females were allowed to lay eggs for eight hours and then removed from the leaf square. The eggs were individually transferred onto either a P. vulgaris or a G. max leaf circle (1 cm in diameter) using a fine brush. Only data of diploid eggs were collected in this procedure. In spider mites, mated mothers produce haploid (giving rise to sons) and diploid eggs (developing to daughters) whereas virgin mothers produce haploid eggs only. Mated females usually produce female-biased sex ratios (e.g., 3:1). To reduce workload, virgin females were used for producing haploid eggs. Fifty female deutonymphs were randomly selected, placed on a P. vulgaris leaf square (3 × 3 cm) and allowed to develop into 3-d-old adults. Forty adult virgin females were randomly selected and transferred onto a new leaf square and allowed to lay eggs for eight hours. The adults were then removed and each of the eggs was transferred onto either a P. vulgaris or a G. max leaf circle (1 cm in diameter) using a fine brush. The individuals were checked every 24 hours, and the developmental period of eggs, larvae, protonymphs, deutonymphs of males and females were recorded. A total of 60 replicates, i.e., 30 (15 males and 15 females) replicates for each host plant were obtained.
Reproduction, longevity, and offspring survival
Male and female deutonymphs were randomly selected from the colony and each individual was placed on a leaf circle (1.5 cm in diameter) to develop into 1-d-old adults. To obtain mated females, a female and a male were paired on a new leaf circle for 24 hours and then the male was removed. The mating status of a female was checked by examining whether she produced at least a daughter, and only mated females were used for experiment. A new leaf circle was replaced for the female every 24 hours until the female died. The number of eggs laid on each leaf circle was recorded and the eggs were allowed to develop into adults. The number of eggs hatched, the number of offspring that developed into adults, sex ratio, female oviposition period and female longevity were also recorded. Seventeen and 18 replicates were prepared for G. max and P. vulgaris, respectively.
Statistical analysis
All data were analyzed using SAS software (SAS 9.4, SAS Institute Inc., NC, USA). Data distribution was tested using Shapiro-Wilk test (UNIVARIATE Procedure). Data on the male total developmental period (egg to adult) were normally distributed and analyzed using the Student's t-test (TTEST Procedure). Other data were not normally distributed and analyzed using the nonparametric Wilcoxon signed-rank test (NPAR1WAY Procedure). A Gaussian functional model according to Archontoulis and Miguez (2015): \(y=a e^{\left(-0.5\left[\ln \left(x / x_0\right) / b\right]^2\right)}\) was used to fit the daily number of eggs and daughters for each treatment, where x is the age of females (days), a is the peak at time x0 (female age), b is the coefficient controlling the width of the peak. The model allowed us to compare reproduction patterns between treatments. If the 95% confidence limits of a given parameter overlap, then there is no significant difference between treatments.
The life table parameters including the intrinsic rate of increase, the net reproductive rate, mean generation time, and doubling time were calculated. The life table parameters (Jervis et al. 2005) were calculated using the data on daily survival and daughter production of each female. The intrinsic rate of natural increase (r) was calculated by solving the Lotka-Euler equation: \(\sum e^{-r x} l_x m_x=1\), where x is the pivotal age, lx is the proportion of females surviving to age x, and mx is the number of daughters produced per female at age x. The net reproductive rate (\(R_0=\sum l_x m_x\), daughters/female/generation), doubling time [\(D_t=\log _e(2) / r\), days], and generation time [\(T=\log _e\left(R_0\right) / r\), days] were also calculated. The bootstrap method with 50,000 bootstrap samples was used to calculate the pseudo-values of a given parameter and the paired-bootstrap test was employed to compare between two treatments (TTEST Procedure).
Results and discussion
Our results show that T. phaselus performed differently on G. max and P. vulgaris in terms of the development time, reproduction and life-table parameters. Host plants have had a strong influence on the biological parameters of several species of tetranychids (de Lima et al. 2017; Golizadeh et al. 2017; Santamaría et al. 2018; Puspitarini et al. 2021; Segura-Martínez et al. 2023). The total developmental period of T. phaselus from egg to adult was significantly shorter on G. max than on P. vulgaris for both males and females. In spider mites, the male has a shorter immature stage than the female. Additionally, males typically stop growing after the protonymphal stage and are smaller than females upon emergence as adults (Li and Zhang 2018). These differences may result in divergent nutritional demands across developmental stages between males and females. Our results show that females on G. max had significantly shorter developmental period at deutonymph stage, whereas males had significantly shorter developmental period at the larva and protonymph stage (Table 1). Similarly, different growth rates on different host plant between males and females was also found in the red spider mite T. merganser Boudreaux (Segura-Martínez et al. 2023). Our results show that T. phaselus had significantly higher egg-to-adult survival on G. max than on P. vulgaris (Table 2). Specifically, egg hatchability was significantly higher on G. max than on P. vulgaris but larva-to-adult survival was similar between the two host plants indicating that egg stage is more affected by host plants (Table 2). It is widely reported that mothers of spider mites dynamically adjust their resource provisioning to offspring (e.g., egg size) in response to environmental conditions such as host plant quality variations (Dahirel et al. 2019), population density fluctuations (Weerawansha et al. 2022), and changes in mating status (Zhou et al. 2018). The differences in egg hatchability between the two bean plants may be attributed to variations in host plant quality (e.g., leaf nutrient composition) and maternal effects. Faster developmental rate may allow a shorter life cycle and faster population growth rate, and together with higher offspring survival, these features may lead to larger population size on G. max.
Download as
Treatment
Egg
Larva
Protonymph
Deutonymph
Total (egg to adult)
Female
G. max
4.73 ± 0.25
2.20 ± 0.20
2.33 ± 0.19
3.07 ± 0.15
12.33 ± 0.36
P. vulgaris
4.67 ± 0.13
2.67 ± 0.39
3.13 ± 0.39
5.47 ± 0.58
15.93 ± 1.14
c² (df)
0.1184 (1)
0.4642 (1)
1.7632 (1)
12.9261 (1)
9.7195 (1)
P
0.7308
0.4956
0.1842
0.0003
0.0018
Male
G. max
5.20 ± 0.14
1.47 ± 0.13
2.47 ± 0.26
3.80 ± 0.45
12.93 ± 0.46
P. vulgaris
5.40 ± 0.13
3.73 ± 0.21
3.53 ± 0.29
4.27 ± 0.33
16.93 ± 0.45
c² (df)
0.9218 (1)
21.7107 (1)
5.8975 (1)
1.8145 (1)
t = 6.18 (df=28)
P
0.337
< 0.0001
0.0152
0.178
< 0.0001
Download as
Host plant
G. max
P. vulgaris
c² (df)
P
No. eggs
64.59 ± 8.83
24.89 ± 3.58
9.23 (1)
0.0024
Oviposition period
14.59 ± 1.59
13.72 ± 1.49
0.11 (1)
0.7408
Daily fecundity (No. eggs/female/day)
3.47 ± 0.36
1.58 ± 0.22
11.56 (1)
0.0007
Egg hatchability (%)
98.59 ± 0.41
93.11 ± 1.83
4.26 (1)
0.039
Larva-to-adult survival
93.49 ± 1.60
88.37 ± 2.93
1.72 (1)
0.1896
Egg-to-adult survival
92.19 ± 1.69
82.16 ± 3.10
6.56 (1)
0.0104
Daughters (%)
79.07 ± 2.82
70.05 ± 4.72
2.21 (1)
0.1373
Longevity
17.29 ± 1.39
16.44 ± 1.45
0.08 (1)
0.7785
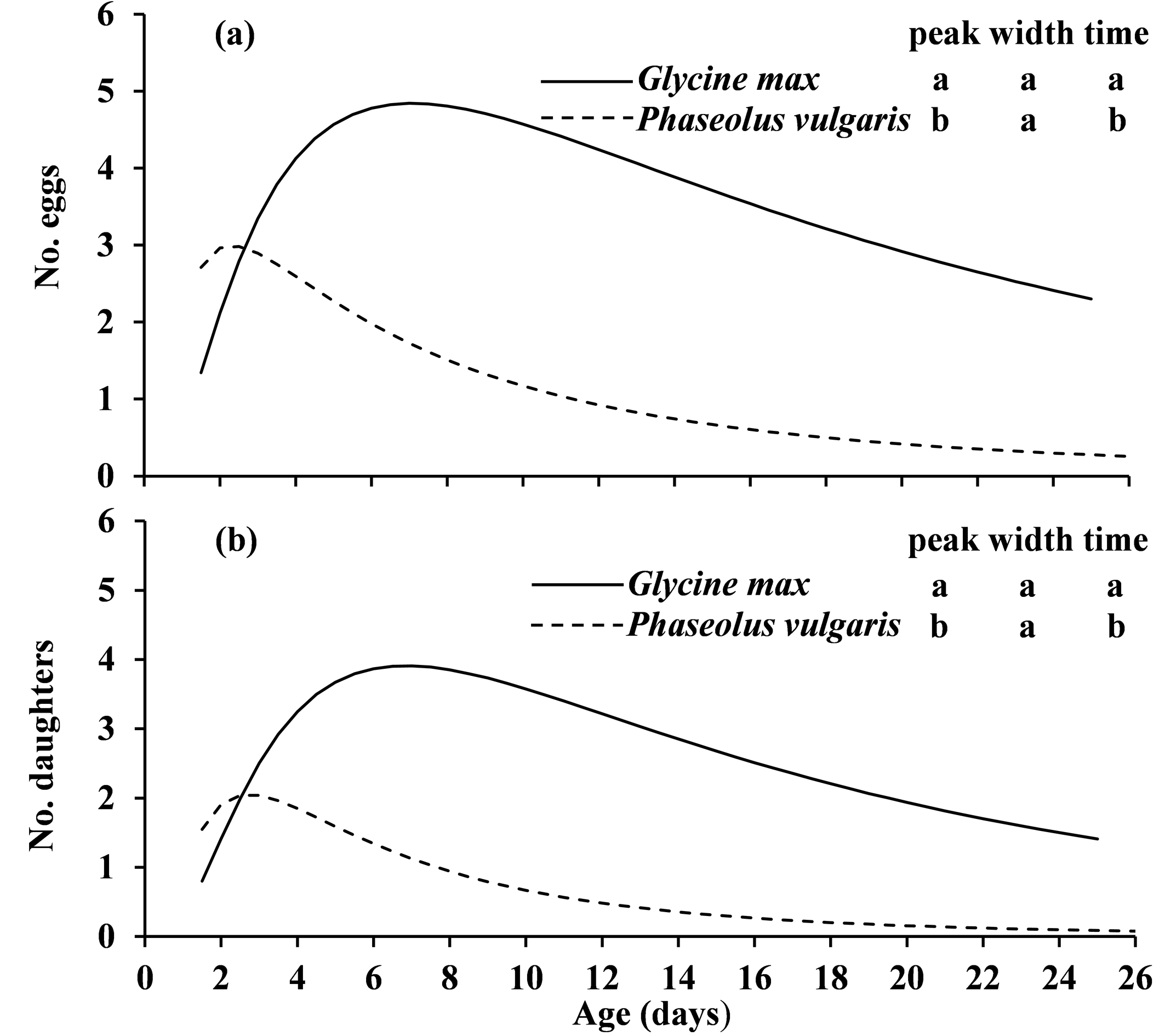


Tetranychus phaselus females had similar oviposition period and longevity on the two host plants but produced significantly more daily and lifetime number of eggs on G. max than on P. vulgaris (Table 2). The higher reproductive output is also illustrated in Figure 1 where the peaks of daily number of eggs and daughters produced were significantly higher and delayed when fed on G. max than those fed on P. vulgaris (Figure 1). Ito et al. (2017) reported that T. phaselus produced about 11 eggs on the favorable host plant P. vulgaris within the first five days, which is similar with present study (about 13 eggs within five days). Our results suggest that G. max is a more favorable host plant for T. phaselus, probably because it is higher in some nutritional components such as protein. Similarly, higher nitrogen content leads to faster development and higher fecundity in Panonychus ulmi (Koch) (Breukel and Post 1959), T. ludeni (Adango et al. 2006), and T. urticae (Wermelinger et al. 1991).
Download as
Treatment
rm (daughters/female/day)
Ro (daughters/female/generation)
T (days)
Dt (days)
G. max
0.2010 ± 0.0001
48.41 ± 0.44
19.30 ± 0.04
3.45 ± 0.00
P. vulgaris
0.1369 ± 0.0010
15.32 ± 0.18
19.93 ± 0.09
5.07 ± 0.04
P
< 0.0001
0.0007
0.0013
< 0.0001
Life table parameters indicate the population growth rates of species in the present generation and subsequent generations and thus a reliable tool to assess the performance of phytophagous arthropods on different host plants. We show that females fed on G. max had significantly higher intrinsic rate of increase (rm ) and net reproductive rate (Ro) and shorter generation time (T) and doubling time (Dt) (Table 3). The values of life table parameters (rm , Ro, T, and Dt) obtained from the two host plants in our study fell within the ranges documented for Tetranychus species in the literature (Kasap 2004; Adango et al. 2006; Sedaratian et al. 2011; Golizadeh et al. 2017; Zhou et al. 2021; de Lima et al. 2022; Ristyadi et al. 2022; Segura-Martínez et al. 2023). The higher rm on soybean may due to higher reproductive rate (Figure 1) and shorter generation time (Golizadeh et al. 2017; Osman et al. 2019), which is suggested to be the most appropriate index to evaluate the performance of pests on host plants (Golizadeh et al. 2017). These results further suggest that G. max is more susceptive to T. phaselus attack than P. vulgaris. Life table parameters can reflect the occurrence of pests on host plants in the field (Adango et al. 2006; de Lima et al. 2017), and thus provide essential information for forecasting and developing pest management strategies (Sedaratian et al. 2011; Segura-Martínez et al. 2023).
In summary, we show that T. phaselus had shorter developmental period, higher egg-to-adult survival and produced more daily and lifetime number of eggs on G. max than on P. vulgaris. Furthermore, females fed on G. max had significantly higher intrinsic rate of increase (rm ) and net reproductive rate (Ro) and shorter generation time (T) and doubling time (Dt). These results suggest that G. max is more susceptive to T. phaselus attack. Considering that G. max and P. vulgaris are favorable host plants for T. phaselus, the increasing planting area of these beans particularly soybeans may facilitate the occurrence of T. phaselus.
Acknowledgments
We thank Professor Tian-Ci Yi for identification of this spider mite to species. We also thank two anonymous reviewers and the editor, Dr. Dejan Marčić, for their constructive comments and suggestions, which have significantly improved the paper. The work reported here was supported by the Key Scientific Research Programs of Universities in Anhui Province (2022AH051052; 2024AH051125; 2024AH051080; 2024AH040170), and the Provincial Graduate Education Quality Engineering Project of Anhui Province (2023zybj021; 2024yjshhsfkc040).
References
- Adango E., Onzo A., Hanna R., Atachi P., James B. 2006. Comparative demography of the spider mite,Tetranychus ludeni, on two host plants in West Africa. J. Insect Sci., 6: 1-9. https://doi.org/10.1673/031.006.4901
- Alcázar-Valle M., Lugo-Cervantes E., Mojica L., Morales-Hernández N., Reyes-Ramírez H., Enríquez-Vara J.N., García-Morales S. 2020. Bioactive compounds, antioxidant activity, and antinutritional content of legumes: a comparison between four Phaseolus species. Molecules, 25: 3528. https://doi.org/10.3390/molecules25153528
- Archontoulis S.V., Miguez F.E. 2015. Nonlinear regression models and applications in agricultural research. Agron. J., 107: 786-798. https://doi.org/10.2134/agronj2012.0506
- Breukel L.M., Post A. 1959. The influence of the manurial treatment of orchards on the population density of Metatetranychus ulmi (Koch) (Acari, Tetranychidae). Entomol. Exp. Appl., 2: 38-47. https://doi.org/10.1111/j.1570-7458.1959.tb02095.x
- Dahirel M., Masier S., Renault D., Bonte D. 2019. The distinct phenotypic signatures of dispersal and stress in an arthropod model: from physiology to life history. J. Exp. Biol., 222: jeb203596. https://doi.org/10.1242/jeb.203596
- de Lima R.P., Bezerra M.M., de Moraes G.J., Furtado I. 2017. Life table of the red spider mite Tetranychus bastosi (Acari: Tetranychidae) on different host plants. Acarologia, 57: 601-605. https://doi.org/10.24349/acarologia/20174177
- de Lima R.P., Guzzo É.C., Breda M.O. 2022. Biological performance of Tetranychus bastosi (Acari: Tetranychidae) on different hosts. Acarologia, 62: 1210-1216. https://doi.org/10.24349/k2gx-swz0
- Ehara S. 1960. On some Japanese tetranychid mites of economic importance. Jpn. J. Appl. Entomol. Zool., 4: 234-241. https://doi.org/10.1303/jjaez.4.234
- FAO. Food and Agriculture Organization of the United Nations: Crops and livestock products [Internet] [assessed on 04 March 2025]. Available from: https://www.fao.org/faostat/en/#data/QCL/visualize
- Golizadeh A., Ghavidel S., Razmjou J., Fathi S.A.A., Hassanpour M. 2017. Comparative life table analysis of Tetranychus urticae Koch (Acari: Tetranychidae) on ten rose cultivars. Acarologia, 57: 607-616. https://doi.org/10.1111/j.1744-7917.2010.01379.x
- Gotoh T., Moriya D., Nachman G. 2015. Development and reproduction of five Tetranychus species (Acari: Tetranychidae): Do they all have the potential to become major pests? Exp. Appl. Acarol., 66: 453-79. https://doi.org/10.1007/s10493-015-9919-y
- Ito K., Fukuda T., Arakawa R. 2017. Host plant and field density of Tetranychus phaselus Ehara (Acari: Tetranychidae). J. Acarol. Soc. Jpn., 26: 13-24. https://doi.org/10.2300/acari.26.13
- Jervis M.A., Copland M.J.W. & Harvey J.A. 2005. The life-cycle. In: Jervis M.A. (Ed). Insects as natural enemies: a practical perspective. Dordrecht: Springer. p. 73-165. https://doi.org/10.1007/1-4020-2625-0_2
- Jin P.-Y., Tian L., Chen L., Hong X.-Y. 2018. Spider mites of agricultural importance in China, with focus on species composition during the last decade (2008-2017). Syst. Appl. Acarol., 23: 2087-2098. https://doi.org/10.11158/saa.23.11.1
- Kasap İ. 2004. Effect of apple cultivar and of temperature on the biology and life table parameters of the twospotted spider mite Tetranychus urticae. Phytoparasitica, 32: 73-82. https://doi.org/10.1007/BF02980863
- Li G.-Y., Zhang Z.-Q. 2018. Does size matter? Fecundity and longevity of spider mites (Tetranychus urticae) in relation to mating and food availability. Syst. Appl. Acarol., 23: 1796-1808. https://doi.org/10.11158/saa.23.9.6
- Li S., Chen J., Hao X., Ji X., Zhu Y., Chen X., Yao Y. 2024. A systematic review of black soybean (Glycine max (L.) Merr.): nutritional composition, bioactive compounds, health benefits, and processing to application. Food Front., 5: 1188-1211. https://doi.org/10.1002/fft2.376
- Migeon A., Dorkeld F. 2011. Spider Mites Web: a comprehensive database for the Tetranychidae. Trends in Acarology: 557-560. [Available from: https://www1.montpellier.inrae.fr/CBGP/spmweb/ Acessed 24 Sept 2024]. https://doi.org/10.1007/978-90-481-9837-5_96
- NBSC. National Bureau of Statistics of China: National data [Internet] [assessed on 04 Mar 2025]. Available from: https://data.stats.gov.cn/easyquery.htm?cn=C01
- Osman M.A., Al Dhafar Z.M., Alqahtani A.M. 2019. Biological responses of the two-spotted spider mite, Tetranychus urticae to different host plant. Arch. Phytopathol. Plant Prot., 52: 1229-1238. https://doi.org/10.1080/03235408.2019.1703299
- Puspitarini R.D., Fernando I., Rachmawati R., Hadi M.S., Rizali A. 2021. Host plant variability affects the development and reproduction of Tetranychus urticae. Int. J. Acarol., 47: 381-386. https://doi.org/10.1080/01647954.2021.1915377
- Ristyadi D., He X.Z., Wang Q. 2022. Predator- and killed prey-induced fears bear significant cost to an invasive spider mite: implications in pest management. Pest Manag. Sci., 78: 5456-5462. https://doi.org/10.1002/ps.7168
- Santamaría M.E., Auger P., Martínez M., Migeon A., Castanera P., Díaz I., Navajas M., Ortego F. 2018. Host plant use by two distinct lineages of the tomato red spider mite, Tetranychus evansi, differing in their distribution range. J. Pest Sci., 91: 169-179. https://doi.org/10.1007/s10340-017-0852-1
- Sedaratian A., Fathipour Y., Moharramipour S. 2011. Comparative life table analysis of Tetranychus urticae (Acari: Tetranychidae) on 14 soybean genotypes. Insect Sci., 18: 541-553. https://doi.org/10.1111/j.1744-7917.2010.01379.x
- Segura-Martínez M.T.d.J., Ordaz-Silva S., Hernández-Juárez A., Heinz-Castro R.T.Q., Mora-Ravelo S.G., Chacón-Hernández J.C. 2023. Life table parameters of Tetranychus merganser Boudreaux (Acari: Tetranychidae) on five host plants. Insects, 14: 473. https://doi.org/10.3390/insects14050473
- van den Boom C.E.M., van Beek T., Dicke M. 2003. Differences among plant species in acceptance by the spider mite Tetranychus urticae Koch. J. Appl. Entomol., 127: 177-183. https://doi.org/10.1046/j.1439-0418.2003.00726.x
- Weerawansha N., Wang Q., He X.Z. 2022. Comparing the effects of social environments and life history traits on sex allocation in a haplodiploid spider mite. Syst. Appl. Acarol., 27: 2123 - 2130. https://doi.org/10.11158/saa.27.10.20
- Weerawansha N., Wang Q., He X.Z. 2023. Reproductive plasticity in response to the changing cluster size during the breeding period: a case study in a spider mite. Exp. Appl. Acarol., 91: 237-250. https://doi.org/10.1007/s10493-023-00834-y
- Wermelinger B., Oertli J.J., Baumgärtner J. 1991. Environmental factors affecting the life-tables of Tetranychus urticae (Acari: Tetranychidae) III. Host-plant nutrition. Exp. Appl. Acarol., 12: 259-274. https://doi.org/10.1007/BF01193472
- Wu J., Wang L., Fu J., Chen J., Wei S., Zhang S., Zhang J., Tang Y., Chen M., Zhu J. et al. 2020. Resequencing of 683 common bean genotypes identifies yield component trait associations across a north-south cline. Nat. Genet., 52: 118-125. https://doi.org/10.1038/s41588-019-0546-0
- Zhao D.-X., Chen D.-S., Ge C., Gotoh T., Hong X.-Y. 2013. Multiple infections with Cardinium and two strains of wolbachia in the spider mite Tetranychus phaselus Ehara: revealing new forces driving the spread of wolbachia. PLoS One, 8: e54964. https://doi.org/10.1371/journal.pone.0054964
- Zhou P., He X.Z., Chen C., Wang Q. 2021. Resource relocations in relation to dispersal in Tetranychus ludeni Zacher. Syst. Appl. Acarol., 26: 2018-2026. https://doi.org/10.11158/saa.26.11.3
- Zhou P., He X.Z., Wang Q. 2018. Sons from virgin mothers produce more daughters in a haplodiploid mite. Syst. Appl. Acarol., 23: 1869-1878. https://doi.org/10.11158/saa.23.9.13
- Zhou P., He X.Z., Chen C., Wang Q. 2024. Age and density of mated females affect dispersal strategies in spider mite Tetranychus ludeni Zacher. Insects, 15, 387. https://doi.org/10.3390/insects15060387



2024-09-26
Date accepted:
2025-03-19
Date published:
2025-03-24
Edited by:
Marčić, Dejan

This work is licensed under a Creative Commons Attribution 4.0 International License
2025 Chen, Chen; Pan, Wei; Tong, Xiaoqian; Song, Jing; Pan, Yang and Zhou, Peng
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



