Update of the national checklist and new records of oribatid mites (Acari: Sarcoptiformes) in conserved relicts within urban areas in Bogota D.C. (Colombia)
Salazar-Fillippo, Andrés A.  1
; Esteban, Tulande-M.
1
; Esteban, Tulande-M.  2
and Miko, Ladislav
2
and Miko, Ladislav  3
3
1✉ Institute for Environmental Studies, Faculty of Science, Charles University, Benátská 2, 128 00, Prague, Czech Republic.
2Jardín Botánico de Bogotá José Celestino Mutis, Subdirección Científica, Av. Calle 63 N° 68-95, 111071, Bogotá, Colombia & Museo Javeriano de Historia Natural, Grupo de taxonomía y sistemática molecular, Laboratorio de Entomología, Cra. 7a #43-82, 110231, Bogotá, Colombia.
3Institute for Environmental Studies, Faculty of Science, Charles University, Benátská 2, 128 00, Prague, Czech Republic & Geobiology Research Group, Department of Biology, University of Antwerp, Universiteitsplein 1C, 2610, Wilrijk, Belgium.
2024 - Volume: 64 Issue: 3 pages: 941-951
https://doi.org/10.24349/kprn-xkaqOriginal research
Keywords
Abstract
Introduction
Reports of oribatid mites in the Neotropical region account for 2404 species (Subías, 2004 - electronic update 2022). A significant proportion of these contributions have taken place during the past decades driven by clear knowledge gaps and a growing interest in soil fauna (Schatz 2004). Unfortunately, these patterns are not consistent throughout the region, and nations like the Republic of Colombia continue to lag behind other South American nations like Argentina, Brazil, or Ecuador (Oliveira et al. 2017; Fredes 2018; Revelo-Tobar 2022). Only 68 species reported in Colombia -compared to \textgreater400 in neighboring nations- encourage urgent attention toward the study of oribatid mites at a national level (Salazar-Fillippo and Miko 2022). These unsatisfying patterns remain constant, as published literature mainly reports oribatid mites at low taxonomic resolution (suborder level), and only one new record and a newly described species were found during the past four years (Niedbala and Ermilov 2022).
Current knowledge of oribatid mites in Colombia is unevenly distributed both in terms of geography and taxonomic groups (Salazar-Fillippo and Miko, 2022). Ptyctimous mites (Niedbała 2004; Niedbala and Ermilov 2022), reports in high-mountain ecosystems (Balogh & Balogh, 1985; Balogh, 1984a, 1984b; Niedbała, 2004), and in conserved areas near major cities (Liu and OConnor 2015; Rueda-Ramirez et al. 2016) have received disproportionate attention leaving other highly diverse and functionally relevant ecosystems aside. Conserved relicts within urban areas represent one of such broadly neglected systems. Their role as biodiversity reservoirs and ecological corridors continues to gain growing attention worldwide (Vergnes et al. 2012; Philpott et al. 2014; Noreika et al. 2015). Whereas the capital, i.e. Bogotá D.C., holds over 25% of the total reports of oribatid mites in Colombia, urban areas remain virtually unknown.
Bogotá is located on the Eastern Andes Mountain range at an altitude of 2600 m.a.s.l. This capital city is densely populated, as only 23% of its nearly 1800 km2 account for urban areas. Over 10 million inhabitants have shaped the metropolitan area of Bogotá into a mosaic of diverse habitats with residential, industrial, commercial, and road infrastructure. However, green areas for recreational use (parks) and conservation of local flora and fauna have remained a priority in certain areas of the city (Alcaldía Mayor de Bogotá, 2021; Corrales and Osorno, 2018). In the present study we are pioneering the study of oribatid mites in two of the most iconic conservation areas within the city of Bogotá D.C. These are the botanical garden José Celestino Mutis and ''Las Mercedes'' forest, respectively representing key reservoirs of soil biodiversity in the Northern border and centre of the city (Orejuela and Celis 2019; Pérez-Suárez 2020). The botanical garden houses 34 living plant collections that emulate different Colombian exotic ecosystems distributed along 19.5 ha. These result in a high diversity mosaic accounting for over a 1000 plant species from 179 families (Orejuela and Celis 2019). ''Las Mercedes'' forest, in contrast, is located in the Northwest of the city within the Thomas van der Hammer natural reserve, and is considered one of the last relicts of fluvio-lacustrine forest, a nearly extinct native ecosystem (Mendoza and Sánchez 2014). This particular ecosystem found in high Andean mountains (2600-2800 m.a.s.l.) owes its name to its former floodplain under the influence of a lake, and its location represents a strategic ecological corridor between hills and wetlands (La Conejera) (Ruiz et al. 2008). In this work, we report 17 new records of oribatid mites (Fig. 1) at a local (Bogotá D.C.) and national level (Colombia) collected in two of the most important conservation areas of the city.
Material and methods



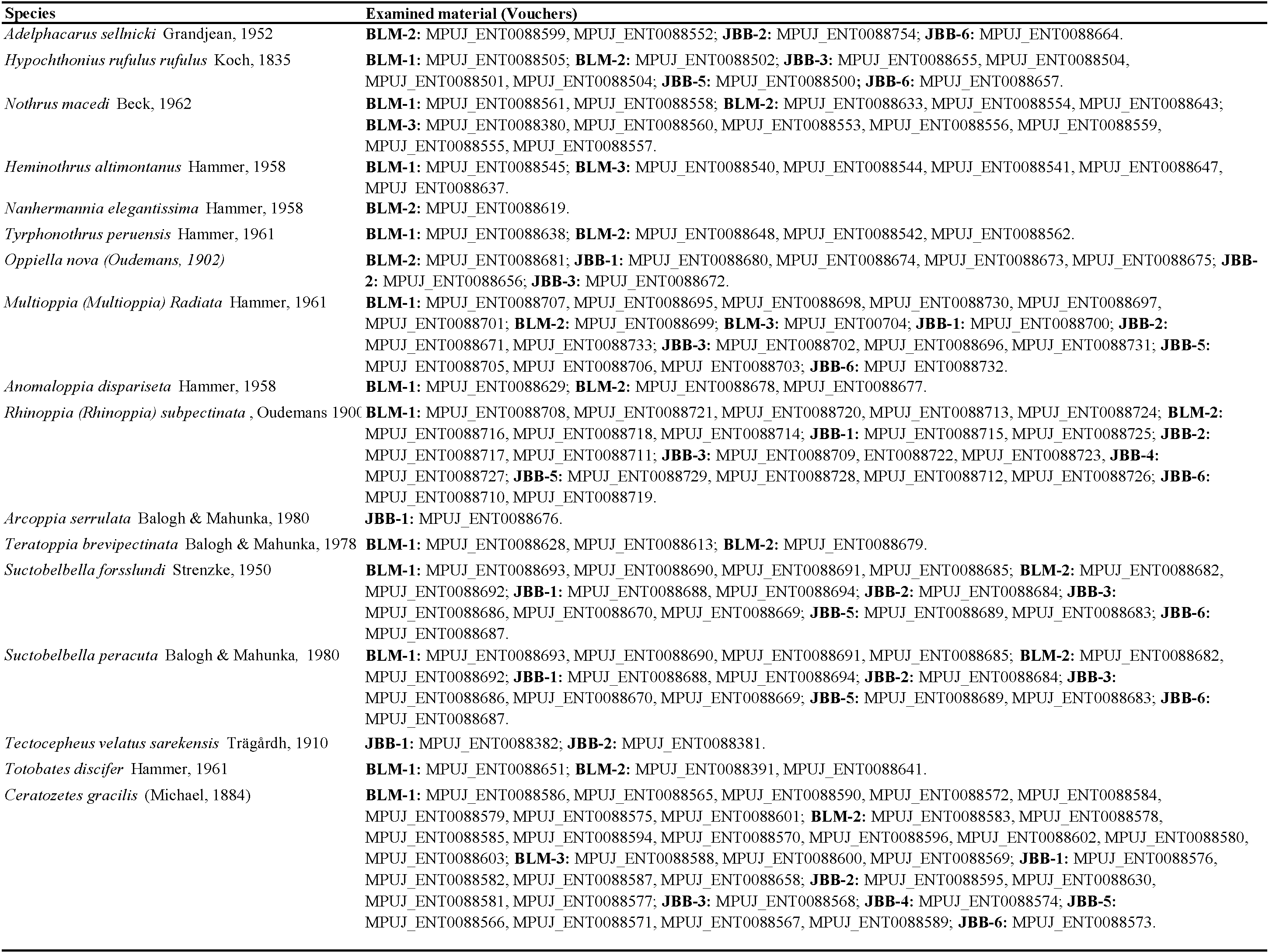
Individuals were collected from the Bogotá Botanical Garden ''José Celestino Mutis'' and the ''Las Mercedes'' forest (Table 1). Five samples were taken from each of the six living collections in the Botanical Garden JCM, accounting for a total of 30. These collections consisted on 1) Cloud forests, 2) Páramo, 3) Cryptogams, 4) Andean forest with Cinchona spp., 5) Arboretum, and 6) high Andean forest with Magnolia spp. ''Las Mercedes'' forest, in contrast, was sampled through three 100 m transects by taking one sample every 10 m, and accounting for a total of 30 samples. Soil cores of 10 cm of diameter and 10 cm of depth were taken in both sites. The extractions of mites from soil samples (785.4 cm3) were performed using Berlese-Tullgren funnels. Specimens were tagged as indicated in the collection vouchers (Table 2) and deposited in the collection of the Museo Javeriano de Historia Natural Lorenzo Uribe J. (referred to as MPUJ-ENT hereafter). The identification of oribatid mites was done using Balogh (1984a, 1984b), Weigmann & Miko (2006), and several keys for specific genera and species. All of the material here examined was collected between April and May 2022 by E. Tulande-M.


We also did a thorough literature revision and included only the documents identifying specimens at least to family level.
New records



CTENACAROIDEA
Adelphacaridae Grandjean, 1954
Genus Adelphacarus Grandjean, 1952
● 1 Adelphacarus sellnicki Grandjean, 1952 (Fig 1. A)
Material examined — BLM-2, JBB-2, JBB-6. Total = 3 individuals.
Distribution — Neotropical (Argentina, Brazil, Colombia), Palearctic (Netherlands, Sweden, Spain, United Kingdom).
HYPOCHTHONIOIDEA
Hypochthoniidae Berlese, 1910
Genus Hypochthonius Koch, 1835
● 2 Hypochthonius rufulus rufulus Koch, 1835 (Fig 1. B)
Material examined — BLM-1, BLM-2, JBB-3, JBB-5, JBB-6. Total = 72 individuals.
Distribution — Semicosmopolitan – Holarctic and tropical.
CROTONIOIDEA
Nothridae Berlese, 1896
Genus Nothrus Koch, 1835
● 3 Nothrus macedi Beck, 1962 (Fig 1. C)
Material examined — BLM-1, BLM-2, BLM-3. Total = 19 individuals.
Distribution — Neotropical (Argentina, Brazil, Colombia, Peru) and meridional palearctic (Caucasus and Iran)
Crotoniidae Thorell, 1876
Genus Heminothrus Berlese, 1913
Subgenus Heminothrus (Platynothrus) Berlese, 1913
● 4 Heminothrus altimontanus Hammer, 1958 (Fig 1. D)
Platynothrus altimontanus Hammer, 1958
Material examined — BLM-1, BLM-3. Total = 9 individuals.
Distribution — Neotropical (Bolivia, Chile, Colombia, Peru).
Nanhermanniidae Sellnick, 1928
Genus Nanhermannia Berlese, 1913
● 5 Nanhermannia elegantissima Hammer, 1958 (Fig 1. E)
Material examined — BLM-2. Total = 1 individual.
Distribution — Neotropical (Argentina, Bolivia, Chile, Colombia) and subantarctic (Falkland Islands).
Malaconothridae Berlese, 1916
Genus Tyrphonothrus Knülle, 1957
Subgenus Tyrphonothrus (Cristonothrus) Subías, 2004
● 6 Tyrphonothrus peruensis Hammer, 1961 (Fig 1. F)
Malaconothrus peruensis Hammer, 1961
Material examined — BLM-1, BLM-2. Total = 4 individuals.
Distribution — Neotropical (Colombia, Peru, Mexico) and India (Tripura).



OPPIOIDEA
Oppidae Sellnick 1937
Genus Oppiella Jacot, 1937
● 7 Oppiella nova (Oudemans, 1902) (Fig 1. G)
Material examined — JBB-1, JBB-2, JBB-3, BLM-2. Total = 101 individuals.
Distribution — Semicosmopolitan (Holarctic: Frequent West Palearctic, India: Uttar Pradesh, New Zeland, and Neotropical: Antilles Is., Argentina, Bolivia, Brazil, Chile, Colombia, Costa Rica, Cuba, Dominican Republic, Ecuador, Mexico, Panama, Peru, Venezuela).
Subfamily Multioppiinae Balogh, 1983
Genus Multioppia Hammer, 1961
Subgenus Multioppia (Multioppia) Hammer, 1961
● 8 Multioppia (Multioppia) radiata Hammer, 1961 (Fig 2. B)
Material examined — JBB-1, JBB-2, JBB-3, JBB-5, JBB-6, BLM-1, BLM-2, BLM-3. Total =151 individuals.
Distribution — Neotropical (Colombia, Peru) and Northeastern India and Iran.
Genus Anomaloppia Subías, 1978
● 9 Anomaloppia dispariseta Hammer, 1958 (Fig 2. C)
Material examined — BLM-1; BLM-2. Total = 20 individuals.
Distribution — Southern: Neotropical (Argentina, Colombia), subantarctic: Falkland Islands, and Iran.
Subfamily : Medioppiinae Subías & Mínguez, 1985
Genus: Rhinoppia Balogh, 1983
Subgenus: Rhinoppia (Rhinoppia) Balogh, 1983
● 10 Rhinoppia (Rhinoppia) subpectinata Oudemans 1900 (Fig 2. D)
Material examined — BLM-1, BLM-2, JBB-1, JBB-2, JBB-3, JBB-4, JBB-5, JBB-6. Total = 502 individuals.
Distribution — Holarctic: except East Palearctic (frequent in West Palearctic), and Senegal. Colombia – New record for the Neotropical region.
Subfamily Arcoppiinae Balogh, 1983
Genus Arcoppia Hammer, 1977
● 11 Arcoppia serrulata Balogh & Mahunka, 1980 (Fig 2. E)
Material examined — JBB-1. Total = 7 individuals.
Distribution — Neotropical (Colombia, Cuba, Mexico) and Asian (Vietnam).
Teratoppiidae Balogh, 1983
Genus Teratoppia Balogh, 1959
Subgenus Teratoppia (Teratoppiella) Balogh, 1983
● 12 Teratoppia brevipectinata Balogh & Mahunka, 1978 (Fig 2. F)
Material examined — BLM-1, BLM-2. Total = 4 individuals.
Distribution — Neotropical (Argentina, Brazil, Chile, Colombia, Peru).
Suctobelbidae Jacot, 1937
Genus Suctobelbella Jacot, 1937
Subgenus Suctobelbella (Flagrosuctobelba) Hammer, 1979
● 13 Suctobelbella forsslundi Strenzke, 1950 (Fig 3. A)
Material examined — BLM-1, BLM-2, JBB-1, JBB-2, JBB-3, JBB-5, JBB-6. Total: 40 individuals.
Distribution — Palearctic and Neotropical (Colombia, Cuba).
● 14 Suctobelbella peracuta Balogh & Mahunka, 1980 (Fig 3. B)
Material examined — BLM-1, BLM-2, JBB-1, JBB-2, JBB-3, JBB-5, JBB-6. Total = 48 individuals.
Distribution — Neotropical (Argentina, Brazil, Colombia, Cuba, Ecuador, Panama) and South Africa.



TECTOCEPHEIODEA
Tectocepheidae Grandjean, 1954
Genus Tectocepheus, Berlese, 1896
● 15 Tectocepheus velatus sarekensis Trägårdh, 1910 (Fig 3. C)
Material examined — JBB-1, JBB-2. Total = 3 individuals.
Distribution — Cosmopolitan.
ORIPODOIDEA
Haplozetidae Grandjean, 1936
Genus Totobates Hammer, 1961
● 16 Totobates discifer Hammer, 1961 (Fig 3. D)
Material examined — BLM-1, BLM-2. Total = 7 individuals.
Distribution — Neotropical (Argentina, Chile, Ecuador, Peru).
CERATOZETOIDEA
Ceratozetidae Jacot, 1925
Genus Ceratozetes Berlese, 1908
● 17 Ceratozetes gracilis (Michael, 1884) (Fig 3. E)
Material examined — BLM-1, BLM-2, BLM-3, JBB-1, JBB-2, JBB-3. JBB-4, JBB-5, JBB-6. Total = 1018 individual.
Distribution — Cosmopolitan: Holarctic, African (Island Santa Helena and South Africa), Asian, New Zealand, Neotropical (Chile, Colombia), and Antarctic (Island South Shetland).
Literature review
● 18 Phthiracarus alienus Niedbała, 2022
Material examined — Sierra Nevada de Santa Marta, above the village Minca, ca. 700 m a.s.l., litter from deciduous forest, 21.02.2020 (leg. W. Niedbała) (Niedbala and Ermilov 2022). Total = 1 individual.
Distribution — Colombia.
● 19 Atropacarus (Hoplophorella) vitrinus (Berlese, 1913)
Material examined — Sierra Nevada de Santa Marta, above the village Minca, ca. 700 m a.s.l., litter from deciduous forest, 21.02.2020 (leg. W. Niedbała) (Niedbala and Ermilov 2022). Total = 7 individuals.
Distribution — Semicosmopolitan - Neotropical: Antilles Is., Argentina, Belize, Brazil, Colombia, Costa Rica, Cuba, Ecuador, Guatemala, Jamaica, Mexico, Panama, Peru, Venezuela.
Discussion
We reported a total of 17 species of oribatid mites in Bogota D.C. that represent new records for the Republic of Colombia, and 2 additional records in the revised literature. This leads to a total of 87 oribatid mite species reported in the country, representing an update of the last published checklist i.e. 68 species (Salazar-Fillippo & Miko, 2022). The genera Totobates, Tectocepheus, Nothrus, Nanhermannia, and Heminotrus (formerly Platynothrus) are also reported for the first time in the country. Whereas this manuscript only includes new reports instead of the total number of species sampled in both sites (JBB and BLM), it is notable that 14 out of the 17 species were found in the Las Mercedes forests, and 7 were exclusive to this site. This contradicts our expectations that a botanical garden including soils and plants from several ecosystems, and receiving visitors from all over the world would show an extremely high species diversity (Aoki 1992; Skubała et al. 2001; Niedbała 2018). Such findings also highlight the importance of conserved natural systems, particularly in a highly unexplored neotropical country.
We stress the importance of conserved relicts within urban areas as reservoirs and ecological corridors of biodiversity (Vergnes et al. 2012; Philpott et al. 2014). Las Mercedes forests is a relatively well-conserved area with over 70 species of plants and undergoing restoration processes in some of its sites (Pérez-Suárez 2020). Moreover, we expect that the number of oribatid mite species in the city of Bogota D.C will increase dramatically when sampling in the southwestern zones, as the eastern hills of the city and the Sumapaz paramo are key ecological corridors within the city.
In terms of the species remarks, we would like to highlight the report of Rhinoppia (Rhinoppia) subpectinata Oudemans 1900, a Holarctic species that was found both in the botanical garden JCM and Las Mercedes forests. Whereas the report in the botanical garden may have been the result of accidental introduction by foreign tourists (anthropogenic passive transport) (Weiss 1915; Zaitsev et al. 2020), their presence in highly conserved alluvial forests (Las Mercedes) give an indication of a broader geographical distribution.
Reporting 17 new records of oribatid mites within urban areas in a ten million-inhabitant city is a mere reflection of a highly unexplored soil group in a megadiverse nation in terms of its aboveground taxa (Arbeláez-Cortés 2013). This study reflects our intentions to lead the study of this group in the Republic of Colombia, both to determine their status (e.g. richness and abundance) across habitats and to encourage their application for bioindication in the near future. We are convinced that this is the first step to better exploit outstanding bioindication attributes of Oribatida to support the assessment of conservation and post-disturbance restoration processes in a nation that still possesses pristine and yet highly vulnerable ecosystems (Williams et al. 2024). We thus further encourage and offer our support to local researchers to continue exploring the clearly underrepresented study of oribatid mites in the Republic of Colombia. Likewise, we acknowledge our willingness to engage in new collaborations to further reach our goal to further report and describe local species.
Acknowledgments
This research has been funded by the Alcaldía Mayor de Bogota D.C, investment project 7679: Research for the conservation of ecosystems and flora of Bogota D.C district region.
We acknowledge the help of Prof. Giovanny Fagua from Pontificia Universidad Javeriana for his continuous support essential for this project. The authors would also like to thank Dr. Pfingstl, and two anonymous reviewers for their valuable comments and suggestions.
References
- Alcaldía Mayor de Bogotá. 2021. Decreto distrital No. 555 del 29 de Diciembre del 2021. Revisión general del Plan de Ordenamiento Territorial de Bogotá D.C.
- Aoki J. 1992. Oribatid Mites Inhabiting Orchid Plants in Greenhouse. Journal of the Acarological Society of Japan, 1(1):7-13. https://doi.org/10.2300/acari.1.7
- Arbeláez-Cortés E. 2013. Knowledge of Colombian biodiversity: published and indexed. Biodiversity and Conservation, 22(12):2875-2906. https://doi.org/10.1007/s10531-013-0560-y
- Balogh J, Balogh P. 1985. Studies on the Anderemaeidae J . Balogh, 1972 (Acari, Oribatei). Opuscula Zoologica Budapest, 19-20:41-48.
- Balogh P. 1984a. Oribatids from Colombia I. Acta Zoologica Hungarica, 30(1-2):29-51.
- Balogh P. 1984b. Oribatids from Colombia II. Acta Zoologica Hungarica, 30(3-4):315-326.
- Corrales E, Osorno V. 2018. Análisis de trade-off en el borde de una reserva forestal en el gradiente urbano-rural de Bogotá. Territorios, 38:137-165. https://doi.org/10.12804/revistas.urosario.edu.co/territorios/a.5909
- Fredes, N.A. 2018. Catalogue of oribatid mites (Acari: Oribatida) from Argentina. Zootaxa, 4406 (1), 1-190. https://doi.org/10.11646/zootaxa.4406.1.1
- Liu D, OConnor BM. 2015. Ptyctimous mites (Acari, oribatida) from colombia, with description of a new species and some remarks on the validity of Atropacarus (hoplophorella) andrei (Balogh, 1958). Systematic and Applied Acarology, 20(1):61-70. https://doi.org/10.11158/saa.20.1.7
- Mendoza L, Sánchez F. 2014. Mamíferos de la Hacienda las mercedes, un área rural al norte de Bogotá, Colombia. Boletin Cientifico del Centro de Museos, 18(2):157-171.
- Niedbała W. 2004. Ptyctimous mites (Acari, Oribatida) of the neotropical region. Annales Zoologici, 54(1):1-288.
- Niedbała W. 2018. Further data on the occurrence of ptyctimous mites (Acari, Oribatida) in European Palm houses. International Journal of Acarology, 44(8):382-385. https://doi.org/10.1080/01647954.2018.1529054
- Niedbala W, Ermilov SG. 2022. New species and records of ptyctimous mites (Acarina, Oribatida) from the Neotropical region. Systematic and Applied Acarology, https://doi.org/10.11158/saa.27.8.8
- Noreika N, Pajunen T, Kotze DJ. 2015. Urban mires as hotspots of epigaeic arthropod diversity. Biodiversity and Conservation, 24(12):2991-3007. https://doi.org/10.1007/s10531-015-0990-9
- Oliveira, A.R., Argolo, P.S., De Moraes, G.J., Norton, R.A. & Schatz, H. 2017. A checklist of the oribatid mite species (Acari: Oribatida) of Brazil. Zootaxa, 4245 (1), 1-89. https://doi.org/10.11646/zootaxa.4245.1.1
- Orejuela A, Celis M. 2019. Guía de géneros de angiospermas y gimnospermas del Jardín Botánico de Bogotá (Serie Colecciones Vivas N°. 2). Bogotá: Jardín Botánico de Bogotá José Celestino Mutis.
- Pérez-Suárez V. 2020. Evaluation of the progress of recovery of aflooded and non-floodable plain forest relict by characterizing the composition and vegetal structure. Revista Luna Azul, 50(50):156-173. https://doi.org/10.17151/luaz.2020.50.8
- Philpott SM, Cotton J, Bichier P, Friedrich RL, Moorhead LC, Uno S, Valdez M. 2014. Local and landscape drivers of arthropod abundance, richness, and trophic composition in urban habitats. Urban Ecosystems, 17(2):513-532. https://doi.org/10.1007/s11252-013-0333-0
- Revelo-Tobar H. 2022. Checklist of Oribatid mites (Acari: Oribatida) of Ecuador.Zootaxa, 5210(1): 001-096. https://doi.org/10.11646/zootaxa.5210.1.1
- Rueda-Ramirez D, De Moraes GJ, Varela A. 2016. Cambios en los ensambles de Mesostigmata y Oribatida edáficos en diferentes usos de suelo en Colombia. In: II Congreso Latinoamericano de Acarología, p. 36-40.
- Ruiz TF, Otero GJ, Ramírez AD, Trespalacios GO. 2008. Biodiversidad y conectividad ecológica en la localidad de Suba. Bogotá D.C., Colombia.
- Salazar-Fillippo AA, Miko L. 2022. Checklist of the oribatid mites (Acari: Oribatida) of the Republic of Colombia. Zootaxa, 5087(1):35-58. https://doi.org/10.11646/zootaxa.5087.1.2
- Schatz H. 2004. Diversity and global distribution of oribatid mites (Acari, Oribatida) - evaluation. Phytophaga, 14:485-500.
- Skubała P, Niemi R, Mleczko I. 2001. Oribatid mite fauna of greenhouses in Upper Silesia, Poland. Fragmenta Faunistica, 44:365-370. https://doi.org/10.3161/00159301FF2001.44.2.365
- Subías LS. 2004 (electronic update 2022). Listado sistemático, sinonímico y biogeográfico de los ácaros oribátidos (Acariformes: Oribatida) del mundo (excepto fósiles). Graellsia, 60:3-305. https://doi.org/10.3989/graellsia.2004.v60.iExtra.218
- Vergnes A, Viol IL, Clergeau P. 2012. Green corridors in urban landscapes affect the arthropod communities of domestic gardens. Biological Conservation, 145(1):171-178. https://doi.org/10.1016/j.biocon.2011.11.002
- Weigmann, G & Miko, L. (2006): Hornmilben (Oribatida). In: Dahl, Die Tierwelt Deutschlands, vol. 76. - Goecke & Evers, Keltern: 520 pp.
- Weiss HB. 1915. Insect importations into New Jersey during the spring of 1915. The Canadian Entomologist, 47(10):326-328. https://doi.org/10.4039/Ent47326-10
- Williams BA, López‐Cubillos S, Ochoa‐Quintero JM, Crouzeilles R, Villa‐Piñeros M, Isaacs Cubides PJ, Schmoeller M, Marin W, Tedesco A, Bastos D, et al. 2024. Bringing the forest back: Restoration priorities in Colombia. Diversity and Distributions, 30(4):e13821. https://doi.org/10.1111/ddi.13821
- Zaitsev AS, Ryabinin NA, Tarasov AI, Shakhab SV. 2020. Potential anthropogenic influence on oribatid mite communities in ancient to modern settlements of the Russian Far East. International Journal of Acarology, 46(5):322-326. https://doi.org/10.1080/01647954.2020.1801838



2024-05-30
Date accepted:
2024-08-07
Date published:
2024-08-30
Edited by:
Pfingstl, Tobias

This work is licensed under a Creative Commons Attribution 4.0 International License
2024 Salazar-Fillippo, Andrés A.; Esteban, Tulande-M. and Miko, Ladislav
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



