Quality control of the predatory mite Euseius scutalis (Acari: Phytoseiidae) against Tetranychus turkestani (Acari: Tetranychidae) over 30 generations of rearing on cattail pollen
Gorji, Zahra  1
; Shishehbor, Parviz
1
; Shishehbor, Parviz  2
; Kocheili, Farhan
2
; Kocheili, Farhan  3
and Riahi, Elham
3
and Riahi, Elham  4
4
1Department of Plant Protection, Faculty of Agriculture, Shahid Chamran University of Ahvaz, Ahvaz, Iran.
2Department of Plant Protection, Faculty of Agriculture, Shahid Chamran University of Ahvaz, Ahvaz, Iran.
3Department of Plant Protection, Faculty of Agriculture, Shahid Chamran University of Ahvaz, Ahvaz, Iran.
4✉ Department of Entomology, Faculty of Agriculture, Tarbiat Modares University, P.O. Box 14115-336, Tehran, Iran.
2024 - Volume: 64 Issue: 3 pages: 819-832
https://doi.org/10.24349/gz0z-vqghOriginal research
Keywords
Abstract
Introduction
Tetranychus turkestani Ugarov and Nikolski (Tetranychus atlanticus McGregor), known commonly as the strawberry spider mite, has become an important pest worldwide (Jeppson et al. 1975; Bolland et al. 1998). The species is polyphagous and is known to feed on more than 25 plant species belonging to more than 15 plant families (Jeppson et al. 1975; Kamali et al. 2001). It is found in the United States, Europe, Russia, Japan, China, the Near East, and the Middle East (Jeppson et al. 1975; Helle and Sabelis 1985; Zhang, 2003). The pest is frequent in Khuzestan province, southwest of Iran attacking various plant species from Solanaceae, Cucurbitaceae and Fabaceae (Mossadegh & Kocheili 2003).
Tetranychus turkestani produces extensive webbing and its feeding activity causes white chlorotic spots on the upper side of leaves. Severe infestations can result in the dropping of leaves and a reduction in flowering. In favorable conditions, the mite develops fast thanks to its short developmental period and high reproductive capacity. Heavy infestations lead to a rapid decrease in crop yields and cause serious economic losses (Carey & Bradley 1982; Tomczyk & Kropczynska 1985; Sohrabi & Shishehbor 2008).
Predaceous mites in the Phytoseiidae family play a crucial role in protecting different crops against many phytophagous mites and other pests (Bounfour & McMurtry 1987; McMurtry & Croft 1997). Therefore, they can be used in biocontrol programmes against pest mites and some insects (Gerson et al. 2003; Fouly et al. 2011). The members of genus Euseius are potential predators. They can prey on other food sources like fungal spores (Kostiainen & Hoy 1994; Schausberger 1997), and pollen (Fouly & Hassan 1991; Abou-Awad et al. 1992; Abdel halim et al. 2000). They have a major impact on low spider mite population densities, and could play a preventative role to limit the spread and the growth of the infestations (McMurtry 1992).
The first Euseius scutalis were described by Athias-Henriot (1958) in Algeria as Typhlodromus scutalis. In Middle East countries (Lebanon, Iran, Egypt, Israel, Jordan) and North Africa, E. scutalis is widely reported on a variety of host plants (Bounfour & McMurtury 1987; Momen & Abdel-Khalek 2008). It is one of the most common and widely distributed phytoseiid mites in Iran. Euseius scutalis is classified by McMurtry et al. (2013) as a specialized pollen feeder and generalist predator that is part of the type IV life style. The species is known to prey on spider mites, but also consumes eggs and immature stages of whiteflies, as demonstrated by previous studies (Swirski et al. 1967; Meyerdirk & Coudriet 1986; Nomikou et al. 2001).
Biological data and predation rates are important to evaluate the performance and potential of natural enemies on pest. They are useful in many studies, such as pest management, pesticide resistance, predator-prey relationships, biological control, mass rearing and harvesting (Chi et al. 2020). As the developmental stage and sex of most arthropods have a significant impact on their morphology, physiology, survival, reproduction, and predation rate, it is necessary to accurately describe stage differentiation and include both sexes in the experiments. They enable the achievement of realistic and precise life table analyses and accurately assess the overall population's fitness (Chi et al. 2020). Both sexes and stage differentiation can be included in data analysis, description, interpretation, and practical applications, thanks to the age-stage, two-sex life table. This method has been extensively used in entomological research for the past twenty years. Achieving effective sustainable management of a prey population using phytoseiid mites requires determining their full effects. There are numerous studies that utilize the age-stage, two-sex life table theory to evaluate the effects of diet on phytoseiid mites (Ansari-Shiri et al. 2022; Gravandian et al. 2022; Hadadi et al. 2022; Yazdanpanah et al. 2023a,b).
Among the various types of high-quality plant pollen, there is a commercially available product called NutrimiteTM (Biobest N.V., Westerlo, Belgium). This product is derived from the narrow-leaved cattail (Typha angustifolia L.) and has been recommended as an alternative food source for supporting generalist phytoseiid populations in crop fields and greenhouses (Samaras et al. 2015). Both T. angustifolia and Typha latifolia L. pollen have been shown to enhance phytoseiid performance (Goleva & Zebitz 2013; Vangansbeke et al. 2014). Compared to other plant pollen species, which are primarily insect-pollinated, Typha species have reduced allergenic properties and are more cost-effective and less labor-intensive to collect (Goleva & Zebitz 2013). However, there is a lack of published information on the long-term mass production of different phytoseiid species using this pollen, despite the potential of Typha species for mass rearing generalist phytoseiids.
Diets can affect mites' performance over many generations and not only in one generation (Nguyen et al. 2014). Any nutrient imbalances can cause a decline in the life table parameters of predatory mites over several generations (De Clercq et al. 2005). It is thus vital to investigate E. scutalis biological parameters, and ability to catch and feed on natural prey after being long-term reared on a non-prey food diet. The present work aimed to study with an age-stage, two-sex life table theory, the biology, life table and predation rate of E. scutalis on a diet T. turkestani, after being reared during different periods on cattail pollen. The growth rate and predation activity of four different generations of the species (0, 10th, 20th, 30rd generation) reared on cattail pollen was evaluated after being switched to a diet T. turkestani. It seems that a predatory mite that can feed on both pollen and arthropod prey will likely not be significantly affected by a switch between these two diets. We hypothesize that since E. scutalis is specialized in feeding on pollen, rearing it exclusively on pollen for 30 generations will not affect its performance.
Material and methods
Prey and predator cultures
Tetranychus turkestani was collected from heavily infested leaves of field bindweed plants (Convolvus arvensis L.) grown on the campus of the Faculty of Agriculture, Shahid Chamran University of Ahvaz, Ahvaz, Iran. The spider mites were then reared on cowpea seedlings (Vigna unguiculata (L.) Walp.) in laboratory conditions at a temperature of 25 °C (± 1 °C), 60±10% RH, and 16:8 h (L: D) photoperiod. Wood-frame culture cages (120× 60 ×60 cm) with 210 µm nylon mesh were used to contain the infested plants, with new plants added as needed.
A colony of E. scutalis population was obtained in February 2022 from a laboratory-grown culture in the Faculty of Agriculture, Shahid Chamran University of Ahvaz, Ahvaz, Iran. This population had initially been collected from marshmallow plants (Althea officinalis L.) infested with T. turkestani on the campus of the Faculty of Agriculture, Shahid Chamran University of Ahvaz, in February 2021. A stereo-microscope (Olympus SZ51 Stereo Microscope) was used to examine the leaf samples and a thin-tipped brush was used to transfer the predatory mite females to green plastic sheet discs that had been soaked in water in Petri dishes. A mixture of T. turkestani and date palm pollen (Phoenix dactylifera L.) was provided to the plastic discs as a food source for the predators. The date palm pollen was collected from Magtu Village, Khuzestan Province, and stored in a freezer throughout the experiments. Cotton threads were placed on the plastic sheets to provide egg-laying sites and shelter for the predators. To guarantee population continuity, predatory mite's eggs were collected at 1–2-day intervals and new mite colonies were established by transferring them to clean plastic discs. Euseius scutalis individuals were reared on date palm pollen for one month before starting the experiments. All the mite species were reared under laboratory conditions of 25± 1 °C, 60± 10% RH and photoperiod of 16: 8 (L: D) conditions throughout the experiments.
Experimental unit
All experiments were conducted in arenas similar to the rearing arenas used for predator rearing. A green plastic sheet (3 × 3 × 0.1 cm) was positioned over a sponge in a Petri dish (6 × 1 cm, diam × height) containing water. A small amount of sewing threads was added to the green plastic sheet to create shelter and oviposition spots. The rearing arena was provided daily with some cattail pollen, prior to and after E. scutalis adults appeared.
How Euseius scutalis performs before and after long-term rearing on cattail pollen
A random selection of around 150 E. scutalis females was taken from the stock colony and then moved to an experimental unit for oviposition. When the oviposited eggs hatched, defrost cattail pollen (Typha latifolia L.) collected from Dorud County, Lorestan Province, Iran, was offered to mites until they reached adulthood (G1). The diet of female progeny from the first generation was kept unchanged for as long as 30 generations (G30). After 10 (G10), 20 (G20), and 30 (G30) generations, the life table parameters of E. scutalis were estimated when switched to T. turkestani. The trial was set-up with four treatments: individuals from the stock colony (G0), individuals reared on cattail pollen for 10 generations (G10), individuals reared on cattail pollen for 20 generations (G20), and those raised on cattail pollen for 30 generations (G30) were compared. About 100 females were first randomly chosen from the related cultures and put in experimental arenas for 24 hours. The newly laid eggs were transferred individually to the experimental units. After the eggs hatched, T. turkestani was added to the units and the number of consumed prey was replaced daily by new ones. The number of prey provided to each experimental unit was increased by changing the predator stage. Specifically, 20, 25, 30, and 50 immature stages of T. turkestani were offered as food at the larval, protonymphal, deutonymphal, and adult stages, respectively. Each day, experimental arenas were checked and the duration and survival of each life stage were documented. The males and females of each treatment were paired immediately after the adults appeared. When there were more females than males or if a male who was paired with a female died, additional males of the same age from the laboratory colony were paired with them; however, the longevity and number of consumed prey were not documented. There were 51, 41, 43, and 45 replicate experimental arenas used, respectively for the treatments mentioned earlier. The daily data recording was conducted to determine the pre-oviposition period, oviposition days, adult longevity, survival, and fertility. The observations were carried on until all adults had died. The daily sex ratio was estimated by collecting eggs during the oviposition period daily and kept in the same conditions as their female parents until adulthood.
The predation ability of E. scutalis was assessed in G0, G10, G20, and G30 populations in life table experiments as well. Daily recording of the number of dead T. turkestani was done until all predators died. To distinguish between the number of T. turkestani preyed by male and female of E. scutalis, the predation rate of 20 males was examined under the same conditions. The female daily consumption rate was determined by subtracting the average male consumption from the consumption of pairs (Farhadi et al. 2011). All the experimental units were placed in a growth chamber at 25 ± 1 °C, 60 ± 10% RH, and a photoperiod of 16L:8D h.
Statistical analysis
Using TWOSEX-MSChart, the age-stage, two-sex life table method was applied to analyze the life history data of E. scutalis (Chi & Liu 1985; Chi 1988; Chi 2023a). The TWOSEX-MS Chart program was used to estimate age-stage-specific survival rate (sxj ), age-stage-specific fecundity (fxj ), age-specific fecundity (mx ), age-specific survival rate (lx ), and age-specific fecundity (mx ), as well as population growth parameters for each treatment (Chi 2023a). By using this program, the bootstrap method with 100000 re-sampling was used to estimate the means and standard errors of different stage durations, total pre-oviposition period (TPOP), adult pre-oviposition period (APOP), fecundity, oviposition days, and adult longevity, and the life table parameters [intrinsic rate of increase (r), finite rate of increase (λ), gross reproductive rate (GRR), net reproductive rate (R0), and mean generation time (T)] (Chi 2023a). The mean of all parameters was compared between different treatments using the paired bootstrap test.
The computer program CONSUME-MS Chart (Chi 2023b) was used to analyze the daily consumption of all individuals. To compute the age-stage predation rate (cxj ), the daily consumption of each individual was taken into account. The mean number of T. turkestani preyed by an individual E. scutalis during its life span (C0), the number of T. turkestani required for the production of an offspring from a E. scutalis (Qp), the entire consumption capacity of a stable population whose maximum value is one (ψ), and the predation potential of E. scutalis population by combining its growth rate (λ), age-stage predation rate (cxj ), and stable age-stage structure (axj ) (ω) were computed based on Chi and Yang (2003). The bootstrap resampling method was used to estimate the variations and standard errors of predation parameters. Different stages' predation rates were compared using the Tukey test (P >0.05) (SPSS 21.0). Using the TWOSEX-MS Chart program, the comparison of C0, Qp, ψ, and ω was made based on the paired bootstrap test (with 100,000 resampling) (Chi 2023a).
Results
Life table parameters
Euseius scutalis developed on T. turkestani even after being reared for thirty generations on pollen (Table 1). The developmental duration of E. scutalis was significantly affected by increasing the duration of rearing on cattail pollen before switching to T. turkestani. The female protonymphal period was significantly longer in G20 than in G0, G10, and G30; this period was longer in G20 than in G10 in the case of males. The female pre-adult duration in G20 was significantly longer than other treatments. In G10, developmental durations of E. scutalis males were the shortest, followed by those in G30 (Table 1). The female and male adult longevities of E. scutalis were significantly different between various treatments. The longest female adult longevity was obtained in G0 (20.60 days), while there was no significant difference between G10 (14.64 days), G20 (14.97 days), and G30 (14.71 days). The same trend was observed for female total life span (Table 1). The male adult longevity of E. scutalis was not significantly different between G20 and G30, both were significantly shorter than that in G0 and G10. In G0, the male total life span was longest, while there was no significant difference between others (Table 1).


The adult pre-oviposition period (APOP), oviposition days, fecundity and sex ratio were significantly affected by the time of rearing on cattail pollen (Table 2). TPOP (total pre-oviposition period) did not significantly differ between treatments. In older generations (G20 and G30), APOP was shorter than in younger generations (G10). The individuals of G0 had the longest oviposition days (Table 2). There was no significant difference in oviposition days of this predator between G10, G20, and G30. The number of eggs produced by E. scutalis females in G0 and G20 was significantly higher than those laid by the females in G10 and G30 (Table 2). The sex ratio of the predatory mite in G20 (74% female adults/total individuals) was significantly higher than others, while not significantly different to G30. The lowest sex ratio (49%) was observed when E. scutalis fed T. turkestani before rearing on cattail pollen (Table 2).
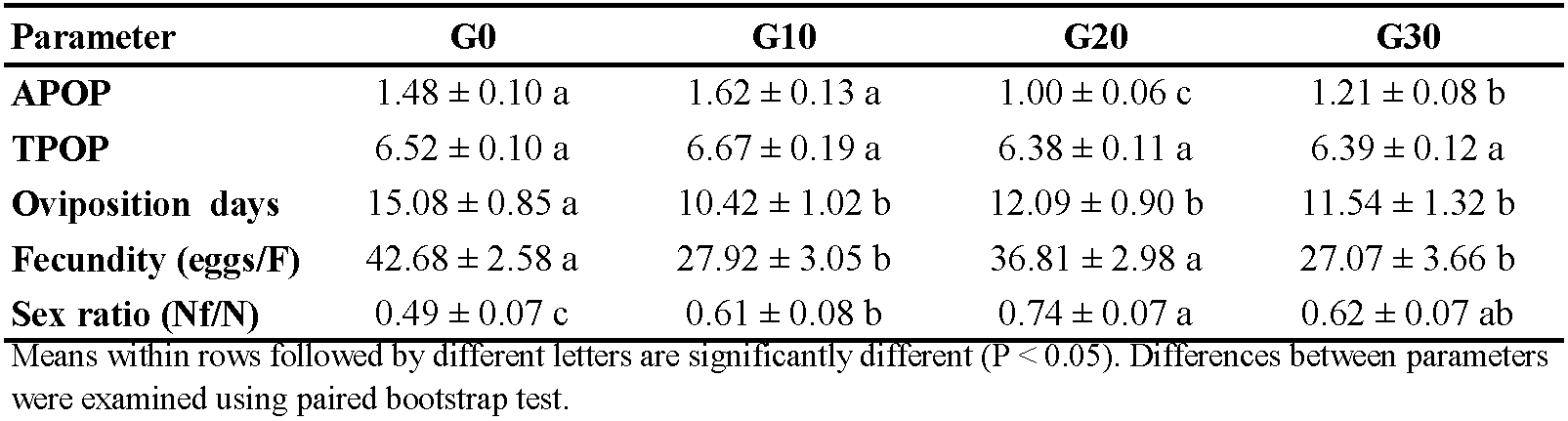


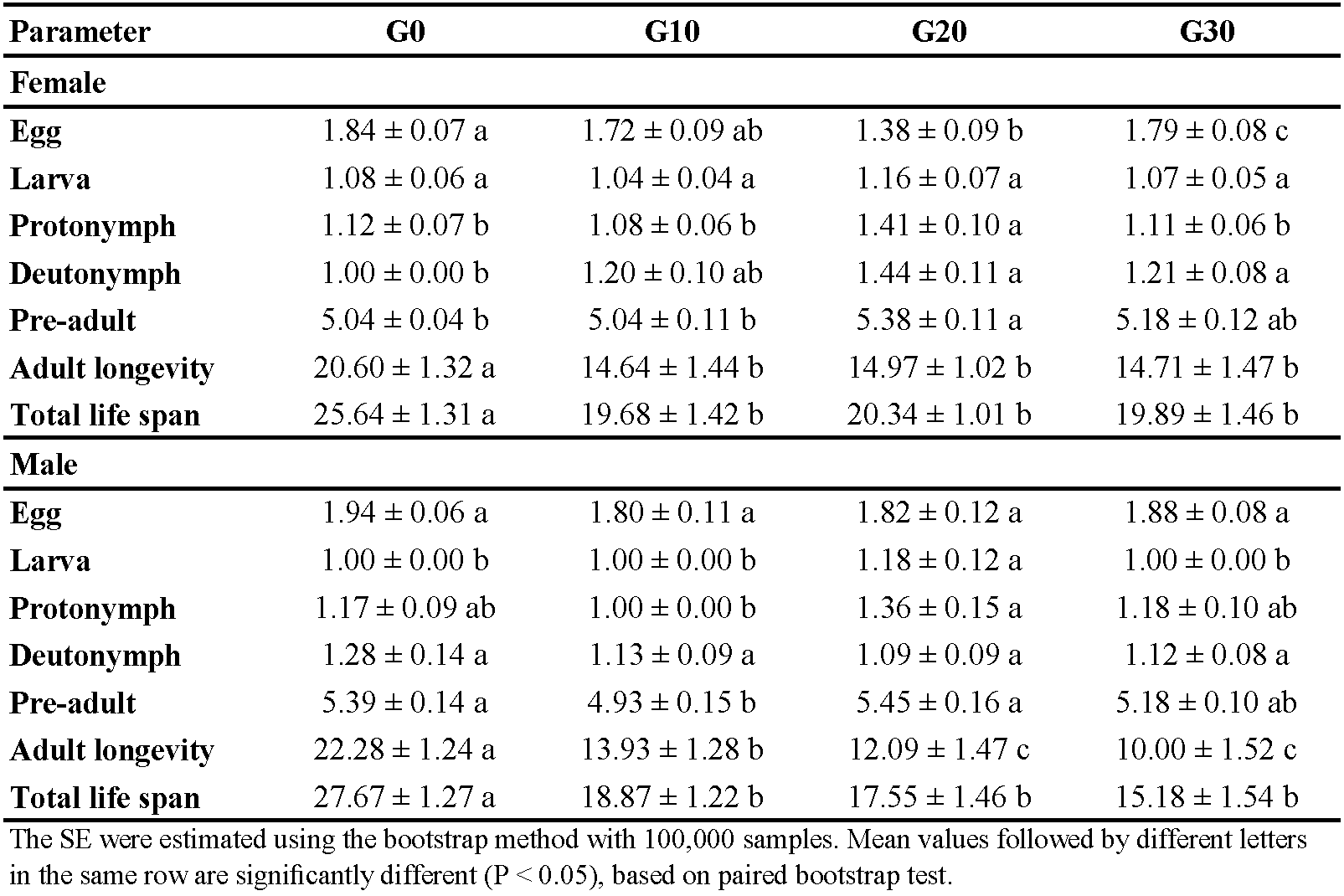
Figure 1 illustrates the age-stage survival rate (sxj ) of E. scutalis before and after different periods of rearing on cattail pollen with obvious overlap between stages as a consequence of differences in development rates among various individuals. Eggs and nymphs experienced generation-based variations in survival rates; rates were higher in G30 and lower in G20. Furthermore, the survival rates of adults in G20 and G30 were higher than G0 and G10 (Figure 1). The female age-specific fecundity (fx ) first raised quickly, peaked, and then decreased. The peak fx of E. scutalis was 3.41 eggs/female in G20, which was higher than that in G10 (3.39 eggs/female), G30 (3.30 eggs/female), and G0 (3.28 eggs/female) (Figure 2). The age-specific survivorship (lx ) for immature stages was 1 in all generations tested except for G0 (0.92). The age specific fecundity (mx ) was highest in the mentioned treatments at the age of 11 (1.91 eggs), 10 (2.11 eggs), 17 (2.92 eggs), and 23 (2.64 eggs) days, respectively (Figure 2).




As shown in Table 3, the population growth parameters of E. scutalis were affected by the time of rearing on cattail pollen when switched to T. turkestani (Table 3). The gross reproductive rate (GRR) in the older generations was significantly higher than that in younger generations. The net reproductive rate (R0) of E. scutalis in G20 was not significantly different from that in G0, but the R0 values in both generations were significantly higher than in G10 and G30 (Table 3). The intrinsic rate of increase (r) and finite rate of increase (λ) of E. scutalis were highest in G20 (r= 0.2793 day -1, λ= 1.3222 day -1) but did not differ significantly from those of in G10 (r= 0.2469 day -1, λ= 1.2801 day -1) (Table 3). The generation time (T) in G30 (13.178 days) was longer than those in G10 (11.48 days) and G20 (11.85 days), while there was no significant difference between G30 and G0 (Table 3).



Sex ratio
Figure 3 shows how the female predatory mite, E. scutalis, changes the sex ratio of its progeny in response to the female age. The results demonstrate that the predator's age influences the sex ratio in their offspring. In all treatments tested, lifetime sex ratios were consistently female biased. On the first day of oviposition, the sex ratio was male biased in all generations tested, then the percentage of females increased when the age of the predator increased. The three and two last days of oviposition period were male biased in G0 and G30, respectively, while the percentage of males increased in the last day of oviposition in G10 and G20. In other words, during the early and late ages of the oviposition period, the proportion of males produced in this predator increased. Generation does not affect this pattern.
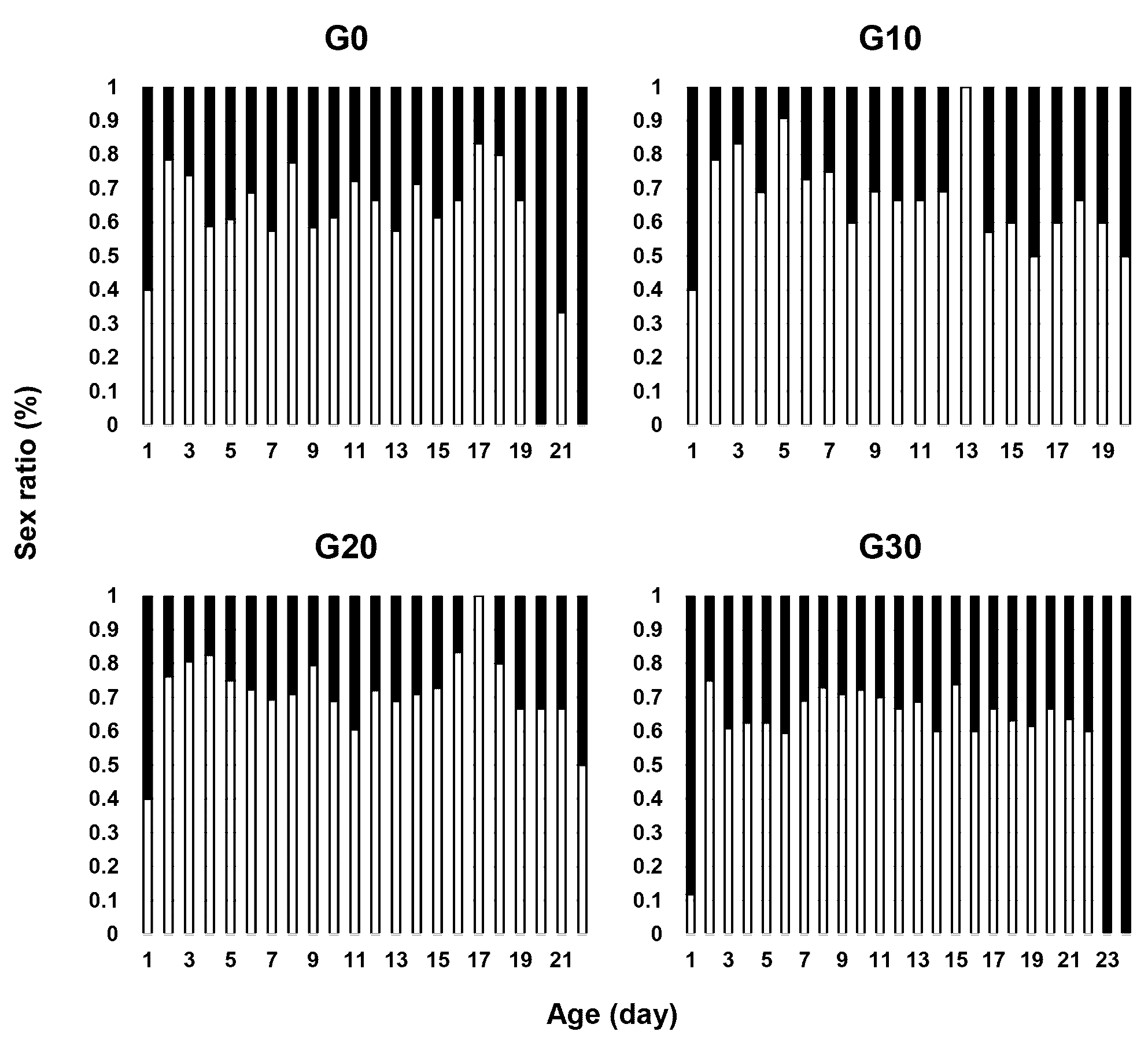


Consumption rate
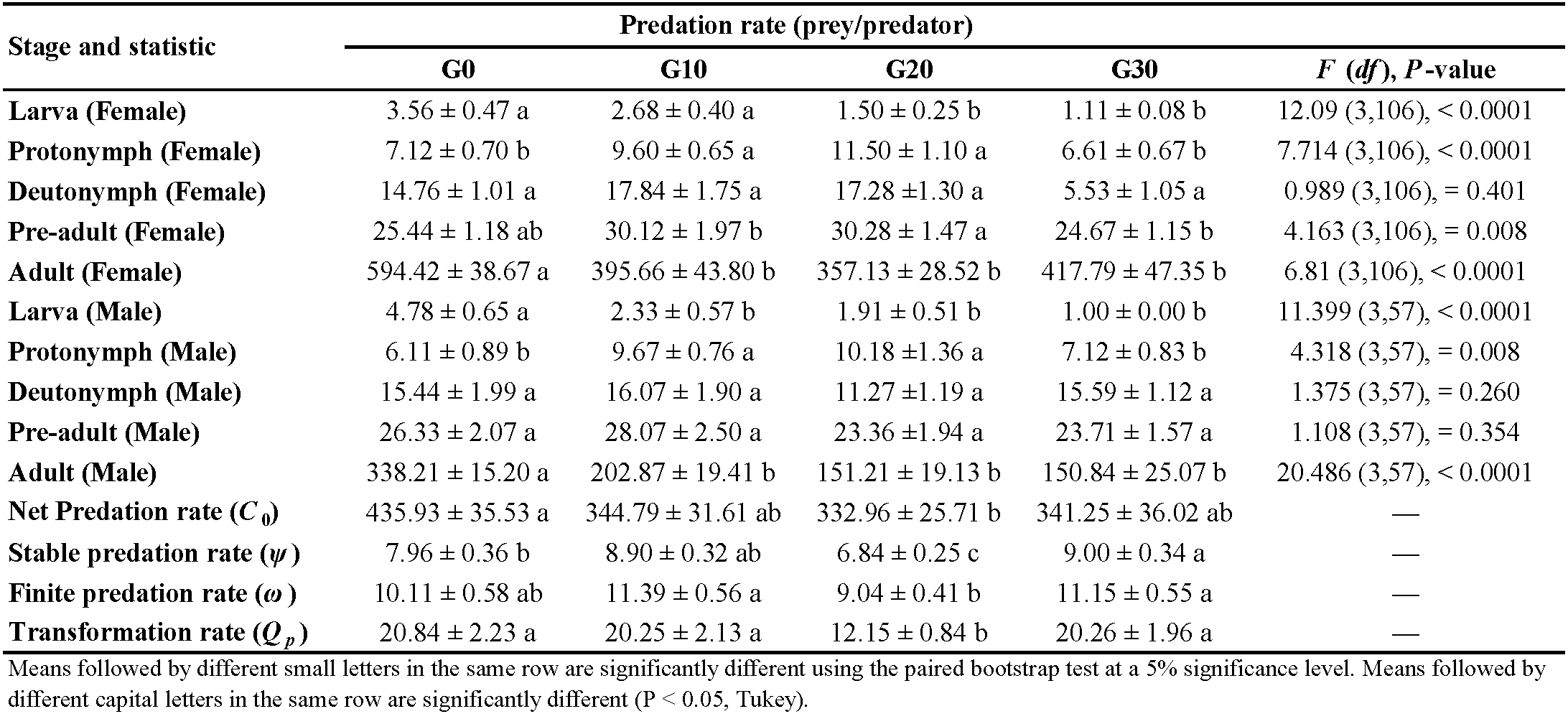
The predation rate of E. scutalis remained constant as the generation increased within the tested generations range (Table 4). The predation rate of the predator increased as stage changed from larva to protonymph, protonymph to deutonymph, and deutonymph to adult. For female and male protonymphs, the predation rate showed no significant difference between G10 (9.60 and 9.67 prey/predator, respectively) and G20 (11.50 and 10.18 prey/predator, respectively), but these values were significantly higher than that in G0 (7.12 and 6.11 prey/predator, respectively) and G30 (6.61 and 7.12 prey/predator, respectively). The total numbers of T. turkestani consumed during the pre-adult stage were 25.44, 30.12, 30.28, and 24.67 per predator for females and 26.33, 28.07, 23.36, and 23.71 per predator for males, in G0, G10, G20, and G30, respectively. The number of prey consumed during female pre-adult duration in G20 was significantly higher than other generations, while the male pre-adult consumption rate was not significantly changed between different generations. The highest female adult (594.42 prey) and male predation rates (338.21 prey) were in G0, and there were no significant differences between G10, G20, and G30 (Table 4).


The age-specific consumption rates (kx ), age-specific net consumption rates (qx ), and the cumulative consumption rate (Cx ) of E. scutalis are shown in Figure 4. The value of kx and qx remained the same until the first death occurred, as indicated by Figure 4. The first peaks of these parameters of E. scutalis fed on T. turkestani occurred at the age of 9, 7, 8, and 8 days in G0, G10, G20, and G30, respectively.
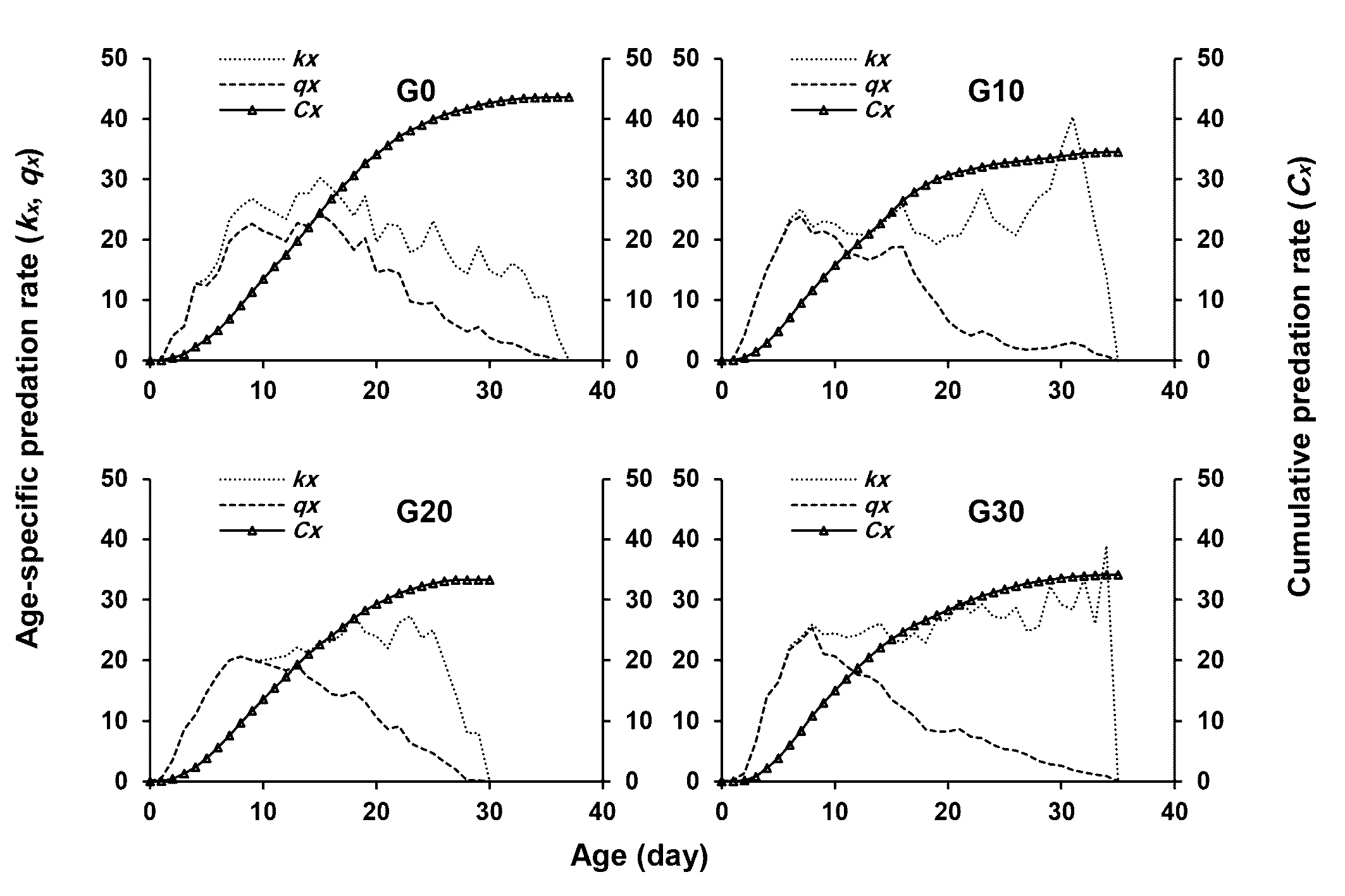


The net predation rate (C0) changed significantly among generations tested (Table 4). Each E. scutalis consumed an average of 435.93 T. turkestani over its lifetime when exposed to it before long-term rearing on cattail pollen, which did not differ significantly from the number of prey fed after ten (344.79) and thirty generations (341.25) on this pollen. The finite predation rate (ω) in G20 was lower than that in G30 and G10, while there was no significant difference between other three generations. The transformation rate (Qp ), the average number of prey needed to produce one predator egg, in G20 was 12.15 prey, which was significantly lower than that in G0 (20.84 prey), G10 (20.25 prey), and G30 (20.26 prey) (Table 4). Stable predation rate (ψ) was lowest in G20, and highest in G30, which the letter was not significantly different from that in G10.
Discussion
One of the most significant concerns in the biological control industry today is the quality of the natural enemies produced (Chambers 1977; Morrison & King 1977; Dicke et al. 1989; van Lentern et al. 1993; Kim et al. 2001). Natural enemies may lose wild-insect traits during production due to genetic variability loss (Paspati et al. 2019). Therefore, monitoring the production, the process, and the product over successive laboratory generations is a major step in mass-rearing of natural enemies for biological control (Parra 2008). The overall quality of a laboratory population is assessed based on its effectiveness against the targeted prey after release (Huettel 1976). Pollen is a common food source for rearing and sustaining predatory mite populations.
Our results indicated that the development, reproduction and population growth rate of E. scutalis on T. turkestani were not impacted by the different rearing periods on cattail pollen, ranging from 10 to 30 generations. The duration of rearing on this pollen did not have any significant effects on female and male pre-adult period, female adult longevity, female and male total lifespan, and oviposition days. Furthermore, despite fluctuations in G20, no changes were observed between G10 and G30 in terms of fecundity, sex ratio, intrinsic rate of increase, and finite rate of increase. It can be concluded that the predator's performance remained constant during 30 generations of rearing on cattail pollen. Similar to our findings, duration of rearing (ranging from 10 to 30 generations) on almond pollen did not significantly impact the finite rate of increase (λ) and intrinsic rate of increase (r) of N. cucumeris after switching to T. urticae (Yazdanpanah et al. 2023b). Another study demonstrated that although a longer period of rearing on cattail pollen (20 generations) before switching to T. urticae resulted in shorter adult longevity, fewer oviposition days, reduced fecundity, GRR, and R0 of A. swirskii compared to a shorter period (10 generations) of feeding on this pollen, the intrinsic and finite rates of increase remained unchanged (Hadadi et al. 2022). In contrast to our results, Gravandian et al. (2022) found that individuals of N. cucumeris, which was reared on cattail pollen for a longer period before being switched to Tetranychus urticae, exhibited shorter adult longevity and oviposition days, along with a higher population growth rate compared to those fed on the pollen for a shorter duration before the switch to T. urticae.
As mentioned previously, evaluating the effectiveness of the rearing process and the quality of the of reared organisms is necessary to assess their quality over time (Huettel 1976; Parra 2008). To ensure the success of a biological control program, it is crucial to monitor the performance of a predator that has been reared on a non-prey diet for a long period when it switches to a prey diet. Therefore, in this study we compared the life table parameters of E. scutalis before (G0) and after long-term rearing (G30) on cattail pollen when exposed to T. turkestani. Our results revealed that although the female and male adult longevity, female and male total life span, APOP, oviposition days, and fecundity of individuals of G30 was shorter or lower than those of G0, pre-adult duration, TPOP, R0, r, λ, and T remained consistent. Given the significance of intrinsic rate of increase (r) as the most important parameter summarizing all demographic and biological parameters of a population (Birch 1948), it can be concluded that long-term rearing on cattail pollen did not negatively affect the performance and success rate of this beneficial biocontrol agent. Both populations (G0 and G30) were fed exclusively on date palm pollen for at least one month before experiments and this might explain why the G0 population was not significantly different from the G30 population. In other words, the biological parameters and population growth of the population that was fed date palm pollen and then transferred to prey did not differ much from those fed cattail pollen after initially being fed date palm and then transferred to prey. From this, we can conclude that there is not a significant difference in nutritional value between date pollen and cattail pollen for this predator. Similarly, stable performance of other phytoseiid mite, A. swirskii, before and after twenty generations of rearing on saffron pollen (Pourbahram et al. 2022), and ten generations of rearing on bird-of paradise pollen (Yazdanpanah et al. 2023a) has been previously reported. In another study, a higher population growth rate and net reproductive rate of A. swirskii were estimated after forty-five generations of rearing on almond pollen when switched to T. urticae compared to the initial population (Ansari-Shiri et al. 2022).
After long-term rearing on alternative food, predators should still be capable of locating, seizing, and killing target prey (Grenier & De Clercq 2003). Our results indicated that E. scutalis can still catch and kill its natural prey (T. turkestani) even after thirty generations of rearing on cattail pollen. Similar results were observed for N. cucumeris against T. urticae after twenty generations (Gravandian et al. 2022) and thirty generations (Yazdanpanah et al. 2023b). Amblyseius swirskii also remained capable of preying on T. urticae after being reared on almond and maize pollen for six generations (Nemati & Riahi 2020), bird-of-paradise pollen for ten generations (Yazdanpanah et al. 2023a), saffron pollen for twenty generations (Pourbahram et al. 2022), and almond pollen for forty-five generations (Ansari-Shiri et al. 2022).
The potential predation ability of natural enemies can be evaluated using the finite predation rate (ω) (Chi et al. 2011), which remained constant even as the length of the rearing period on cattail pollen increased (from ten to thirty generations) in the current study. This parameter serves as a standard measure by establishing a connection between the finite rate, stable age-stage distribution, and age-stage specific predation rate (Chi et al. 2011). The predation rate of both immature and adult stages, as well as the net predation rate of E. scutalis when exposed to T. turkestani, remained stable regardless of the rearing period on pollen. In addition, individuals from G30, preyed on the same number of T. turkestani as individuals from G10, with no significant difference in stable predation rate, finite predation rate, and transformation rate between these populations. Similar to our results, the duration of rearing on pollen before switching to natural prey did not notably affect the stable predation rate and finite predation rate of A. swirskii in other studies (Hadadi et al. 2022; Pourbahram et al. 2022). In contrast, Gravandian et al. (2022) discovered that a longer period of rearing on cattail pollen led to a more stable and finite predation rates of N. cucumeris on T. urticae compared to a shorter period. Yazdanpanah et al. (2023b) reported opposite results. By comparing the finite predation rate of E. scutalis between the G30 and the initial population (G0), we found that the predation potential of this predatory mite against T. turkestani remained unaffected after 30 generations of rearing on cattail pollen, aligning with trends observed in previous studies (Ansari-Shiri et al. 2022; Hadadi et al. 2022; Pourbahram et al. 2022).
Phenotypic plasticity refers to the ability of animals to adjust their developmental, physiological, or behavioral characteristics in response to changes in their environment (Stephen et al. 2011). Various environmental factors can induce plastic responses in organisms (Westneat et al. 2019). Research on insect nutrition has demonstrated that the composition of the diet can greatly influence plasticity (Koyama et al. 2013). Insects that fail to adapt to new food sources in a timely manner may enter diapause or even die (Gu et al. 2021). Based on our findings, E. scutalis appears to possess the ability to maintain population stability due to its high plasticity in response to environmental changes. In other words, this predatory mite shows no difficulty in adapting to different diets, whether it is pollen or prey. This is consistent with the study of McMurtry et al. (2013) that mentioned this predator's ability to be a specialized pollen feeder and a generalist predator. This characteristic allows E. scutalis to easily adjust to various diets within a short period of time, without experiencing a significant decline in fitness. In contrast, A. swirskii took some time to adjust to a new food source after switching its diet (Hadadi et al. 2022). This finding confirms that A. swirskii is a Type III predator, rather than a specialist pollen feeder (Type IV). This suggests that although A. swirskii can consume pollen, it is not as highly specialized as Type IV predators. Consequently, its ability to adapt to diet changes may differ from that of E. scutalis. There is a need to explore field applications of mass-reared predatory mites and the nutritional changes that occur when diets are altered in further researches.
In conclusion, our results showed that E. scutalis, considered a promising biological control agent against certain mites and insect pests, maintained successful predation on T. turkestani even after a long-term rearing on cattail pollen. Moreover, the biological and predation parameters of this predator did not suffer any adverse effects due to the long-term feeding history on cattail pollen. The growth and consumption rates of the predators remained consistent when they switched from cattail pollen to prey, comparable to the population that was not reared on cattail pollen at all. Consequently, E. scutalis specimens that have been reared for an extended period on cattail pollen may represent excellent candidates for establishing an effective predator on plants infested with T. turkestani. However, further research is necessary to assess the effectiveness of these mass-reared predators in greenhouses or fields.
Acknowledgements
Financial support (Grant no. SCU.AP1401.400) provided by the research deputy of Shahid Chamran University of Ahvaz, Ahvaz, Iran is gratefully acknowledged.
References
- Abdel Halim S.M., Hanna M.A., Abdella M.M., Ramadan M.F. 2000. Evaluation of food type, availability of food and competition as factors affecting mass rearing of the predaceous mite Euseius scutalis (A.-H.) (Acari: Phytoseiidae). Egyptian J. Biol. Pest Control., 10(1): 33-38.
- Abou-Awad B.A., Reda A.S., Elsawi S.A. 1992. Effects of artificial and natural diets on the development and reproduction of two phytoseiid mites Amblyseius gossipi and Amblyseius swirskii (Acari: Phytoseiidae). Insect Sci. Appl., 13: 441-445. https://doi.org/10.1017/S1742758400013746
- Ansari-Shiri H., Fathipour Y., Hajiqanbar H., Riahi E., Riddick E.W. 2022. Quality control of the predatory mite Amblyseius swirskii during long-term rearing on almond Prunus amygdalis pollen. Arthropod-Plant Inte., 16: 645-655. https://doi.org/10.1007/s11829-022-09929-6
- Athias-Henriot C. 1958. Contribution à la connaissance du genre Typhlodromus Scheuten. (Acariens Parasitiformes, Phytoseiidae). Description de deux espèces nouvelles d'Algérie et clé des espèces du groupe finlandicus. Rev. Patol. Veg. Entom. Agric. France, 37(2): 181-186.
- Birch L.C. 1948. The intrinsic rate of natural increase of an insect population. J. Anim. Ecol., 17: 15-26. https://doi.org/10.2307/1605
- Bolland H.R., Gutierrez J., Flechtmann C.H.W. 1998. World catalogue of the spider mite family (Acari: Tetranychidae), with reference to taxonomy, synonymy, host plants and distribution. Leiden, The Netherlands:.Brill academic Publisher. 392 pp.
- Bounfour M., McMurtry J.A. 1987. Biology and ecology of Euseius scutalis (Athias-Henriot) (Acarina: Phytoseiidae). Hilgardia, 55: 1-23. https://doi.org/10.3733/hilg.v55n05p023
- Carey J.R., Bradley J.W. 1982. Developmental rate, vital schedules, sex ratios, and life tables of Tetranychus urticae, T. turkestani and T. pacificus (Acarina: Tetranychidae) on cotton. Acarologia, 23(4): 333-345.
- Chambers D. L. 1977. Quality control in mass rearing. Annu. Rev. Entomol, 22(1): 289-308. https://doi.org/10.1146/annurev.en.22.010177.001445
- Chi H. 1988. Life-table analysis incorporating both sexes and variable development rates among individuals. Environ. Entomol., 17: 26-34. https://doi.org/10.1093/ee/17.1.26
- Chi H. 2023a. CONSUME-MSChart: computer program for consumption rate analysis based on the age stage, two-sex life table.2019.02.01 ed. http://140.120.197.173/Ecology/prod02.htm. Accessed 19 Feb 2023
- Chi H. 2023b. TWOSEX-MSChart: a computer program for the agestage, two-sex life table analysis. 2019.01.09 Edition. http://140.120.197.173/Ecology/prod02.htm. Accessed 19 Feb 2023
- Chi H., Liu H. 1985. Two new methods for the study of insect population ecology. Bull. Inst. Zool. Acad. Sin., 24: 225-240.
- Chi H., Yang T. 2003. Two-sex life table and predation rate of Propylaea japonica Thunberg (Coleoptera: Coccinellidae) fed on Myzus persicae (Sulzer). Environ. Entomol., 32: 327-333. https://doi.org/10.1603/0046-225X-32.2.327
- Chi H., Huang Y.B., Allahyari H., Yu J.Z., Mou D.F., Yang T.C., Farhadi R., Gholizadeh M. 2011. Finite predation rate: a novel parameter for the quantitative measurement of predation potential of predator at population level. Nat. Prec. https://doi.org/10.1038/npre.2011.6651.1
- Chi H., You M., Atlihan R., Smith C.L., Kavousi A., Özgökçe M.S., et al. 2020. Age-stage, two-sex life table: an introduction to theory, data analysis, and application. Entomol. Gen., 40: 103-124. https://doi.org/10.1127/entomologia/2020/0936
- De Clercq P., Arijs Y., van Meir T., van Stappen G., Sorgeloos P., Dewettinck K., Rey M., Grenier S., Febvay G. 2005. Nutritional value of brine shrimp cysts as a factitious food for Orius laevigatus (Heteroptera: Anthocoridae). Biocontrol Sci. Technol., 15: 467-479. https://doi.org/10.1080/09583150500086706
- Dicke M., de Jong M., Alers M.P.T., Stelder F.C.T., Wunderink R., Post J., 1989. Quality control of mass‐reared arthropods: Nutritional effects on performance of predatory mites 1. J. Appl. Entomol, 108(1-5): 462-475. https://doi.org/10.1111/j.1439-0418.1989.tb00480.x
- Fouly A.H., Al-Daghairi M.A., Abdel Baky N.F. 2011. Biological aspects and life tables of Typhlodromips swirskii (Acari: Phytoseiidae) fed Bemisia tabaci (Homoptera: Aleyrodidae). J. Entomol., 8(1): 52-62. https://doi.org/10.3923/je.2011.52.62
- Fouly A.H., Hassan M.F. 1991. Effect of crowding and food level on the predaceous phytoseiid mite Amblyseius gossipi (El-Badry) fed on white fly Bemisia tabaci (Gennadius). Bull. Zool. Soc. Egypt, 40: 141-146.
- Gerson U., Smiley R.L., Ochoa R. 2003. Mites (Acari) for Pest Control. UK: Blackwell Science, Oxord, UK. 539 pp. https://doi.org/10.1002/9780470750995
- Goleva I., Zebitz C.P.W. 2013. Suitability of different pollen as alternative food for the predatory mite Amblyseius swirskii (Acari, Phytoseiidae). Exp. Appl. Acarol., 61: 259-283. https://doi.org/10.1007/s10493-013-9700-z
- Gravandian M., Fathipour Y., Hajiqanbar H., Riahi E., Riddick E.W. 2022. Long-term effects of cattail Typha latifolia pollen on development, reproduction, and predation capacity of Neoseiulus cucumeris, a predator of Tetranychus urticae. BioControl, 67: 149-160. https://doi.org/10.1007/s10526-021-10116-4
- Grenier S., De Clercq P. 2003. Comparison of artificially vs. naturally reared natural enemies and their potential for use in biological control. In: van Lenteren J. (Ed). Quality control and production of biological control agents. Theory and testing procedures. Wallingford, UK: CABI Publishing. p. 115-131. https://doi.org/10.1079/9780851996882.0115
- Gu H.J., Dai C.C., Yang Y.Z., Lu Y.H. 2021. Effects of different preys on development and fecundity of Hippodamia variegata (Goeze). J. of Plant Prot., 48(05): 1187-1188.
- Hadadi A., Fathipour Y., Hajiqanbar H., Riahi E. 2022. Long-term effects of cattail pollen on development, population growth potential, and predation capacity of Amblyseius swirskii (Acari: Phytoseiidae). Biocontrol Sci. Technol., 32: 1403-1416. https://doi.org/10.1080/09583157.2022.2134555
- Helle W., Sabelis M.W. 1985. Spider mites: their biology, natural enemies and control. Volume 1A. Amsterdam, The Netherlands: Elsevier Science Publisher. 405 pp.
- Huettel, M.D. 1976. Monitoring the quality of laboratory-reared insects: A biological and behavioral perspective. Environ. Entomol., 5: 807-14. https://doi.org/10.1093/ee/5.5.807
- Jeppson L.R., Keifer H.H., Baker E.W. 1975. Mites injurious to economic plants. Berkeley, USA: University of California Press. 614 pp. https://doi.org/10.1525/9780520335431
- Kamali K., Ostovan H., Atamehr A. 2001. A catalogue of mites and ticks (Acari) of Iran. Tehran, Iran: Islamic Azad University Scientific Publication Centre. pp. 192.
- Kim J.H., Broadbent A.B., Lee S.G. 2001. Quality control of the mass-reared predatory mite, Amblyseius cucumeris (Acarina: Phytoseiidae). J. Asia Pac. Entomol, 4(2): 175-179. https://doi.org/10.1016/S1226-8615(08)60120-X
- Kostiainen T., Hoy M.A. 1994. Egg-harvesting allows large scale rearing of Amblyseius finlandicus (Acari: Phytoseiidae) in the laboratory. Exp. Appl. Acarol., 18: 155-165. https://doi.org/10.1007/BF02353683
- Koyama T., Mendes C.C., Mirth C.K. 2013. Mechanisms regulating nutrition-dependent developmental plasticity through organ-specific effects in insects. Front. Physiol., 4: 263. https://doi.org/10.3389/fphys.2013.00263
- McMurtry J.A. Dynamics and potential impact of ′generalist′ phytoseiids in agroecosystems and possibilities for establishment of exotic species. Exp. Appl. Acarol., 14: 371-382. https://doi.org/10.1007/BF01200574
- McMurtry J.A., Croft B.A. 1997. Life-styles of Phytoseiid mites and their roles in biological control. Annu. Rev. Entomol., 42: 291-321. https://doi.org/10.1146/annurev.ento.42.1.291
- McMurtry J.A., De Moraes G.J., Sourassou N.F. 2013. Revision of the lifestyles of phytoseiid mites (Acari: Phytoseiidae) and implications for biological control strategies. Syst. Appl. Acarol., 18: 297-320. https://doi.org/10.11158/saa.18.4.1
- Meyerdirk D.E., Coudriet D.L. 1986. Evaluation of two biotypes of Euseius scutalis (Acarina: Phytoseiidae) as predators of Bemisia tabaci (Homoptera: Aleyrodidae). J. Econ. Entomol., 79(3): 659-663. https://doi.org/10.1093/jee/79.3.659
- Momen F.M., Abdel-Khalek A. 2008. Influence of diet on biology and life-table parameters of the predacious mite Euseius scutalis (A.-H.) (Acari: Phytoseiidae). Acta Phytopathol. Entomol. Hungarica, 41(6): 418430. https://doi.org/10.1080/03235400600813508
- Morrison R.K., King E.G. 1977. Mass production of natural enemies. In: Ridgway R.L., Vinson S.B. (Eds). Biological control by augmentation of natural enemies. New York: Plenum: 183-217. https://doi.org/10.1007/978-1-4684-2871-1_6
- Mossadegh M.S., Kocheili F. 2003. A semi descriptive checklist of identified species of arthropods (agricultural, medical) and other pests from Khuzestan, Iran. Shahid Chamran U., Ahvaz Press, Ahvaz, Iran.
- Nemati A., Riahi E. 2020. Does feeding on pollen grains affect the performance of Amblyseius swirskii (Acari: Phytoseiidae) during subsequent generations? Bull. Entomol. Res., 110: 449-456. https://doi.org/10.1017/S0007485319000804
- Nguyen D.T., Vangansbeke D., De Clercq P. 2014. Artificial and factitious foods support the development and reproduction of the predatory mite Amblyseius swirskii. Exp. Appl. Acarol., 62: 181-194. https://doi.org/10.1007/s10493-013-9749-8
- Nomikou M., Janssen, A., Schraag R., Sabelis M.W. 2001. Phytoseiid predators as potential biological control agents for Bemicia tabaci. Exp. Appl. Acarol., 25: 271 - 291. https://doi.org/10.1023/A:1017976725685
- Parra J.R.P. 2008. Mass rearing of natural enemies. In: Capinera J.L. (Eds). Encyclopedia of entomology. Dordrecht: Springer. https://doi.org/10.1007/978-1-4020-6359-6_1741
- Paspati A., Ferguson K.B., Verhulst E.C., Urbaneja A., González‐Cabrera J., Pannebakker B.A. 2019. Effect of mass rearing on the genetic diversity of the predatory mite Amblyseius swirskii. Entomol Exp Appli, 167(7): 670-681. https://doi.org/10.1111/eea.12811
- Pourbahram P., Hajiqanbar H., Yazdanpanah S., Fathipour Y. 2022. Generation-dependent demography and predation capacity of Amblyseius swirskii (Acari: Phytoseiidae) fed on saffron pollen. Int. J. Acarol., 48: 669-678. https://doi.org/10.1080/01647954.2023.2171481
- Samaras K., Pappas M.L., Fytas E., Broufas G.D. 2015. Pollen suitability for the development and reproduction of Amblydromalus limonicus (Acari: Phytoseiidae). BioControl, 60: 773-782. https://doi.org/10.1007/s10526-015-9680-5
- Schausberger P. 1997. Inter- and intraspecific predation on immatures by adult females in Euseius finlandicus, Typhlodromus pyri and Kampimodromus aberrans (Acari: Phytoseiidae). Exp. Appl. Acarol., 21: 131-150.
- Sohrabi F., Shishehbor P. 2008. Effect of host plant and temperature on growth and reproduction of the strawberry spider mite Tetranychus turkestani Ugarov and Nikolski (Acari: Tetranychidae). Syst. Appl. Acarol., 13: 26-32. https://doi.org/10.11158/saa.13.1.2
- Stephen J.S., Gregory A.S., Nathan L. 2011. Polyphenism in insects. Curr. Biol., 21(18): 738-749. https://doi.org/10.1016/j.cub.2011.06.006
- Swirski E., Amitai S., Dorzia N. 1967. Laboratory studies on the feeding, development and reproduction of the predaceous mites Amblyseius rubini Swirski and Amitai and Amblyseius swirskii Athias-Henriot (Acarina: Phytoseiidae) on various kinds of food substances. Israel J. Agric. Res., 17: 101-112.
- Tomczyk A., Kropczynska D. 1985. Effects of host plants. In: Helle W., Sabelis M.W. (Eds). Spider mites: their biology, natural enemies and control. Volume 1A. Amsterdam, The Netherlands: Elsevier Science Publisher: 317-330.
- Vangansbeke D., Nguyen D.T., Audenaert J., Verhoeven R., Deforce K., Gobin B., Tirry L., De Clercq P. 2014. Diet dependent cannibalism in the omnivorous phytoseiid mite Amblydromalus limonicus. Biol. Control., 74: 30-35. https://doi.org/10.1016/j.biocontrol.2014.03.015
- van Lenteren J.C., Bigler F., Waddington C. 1993. Quality control guidelines for natural enemies. Proc. 7th workshop global IOBC working group quality control of mass reared arthropods: 222-230.
- Westneat D.F., Potts L.J., Sasser K.L., Shaffer J.D. 2019. Causes and consequences of phenotypic plasticity in complex environments. Trends Ecol. Evol., 34(6): 555-568. https://doi.org/10.1016/j.tree.2019.02.010
- Yazdanpanah Sh., Pourbahram P., Fathipour Y. 2023a. Bird-of-paradise, thorn apple and maize pollens are promising alternative diets for the long-term rearing of predatory mite Amblyseius swirskii (Acari: Phytoseiidae). Syst. Appl. Acarol., 28(10): 1678-1690. https://doi.org/10.11158/saa.28.10.8
- Yazdanpanah Sh., Fathipour Y., Riahi E., Zalucki M.P. 2023b. Effects of diet switching from almond pollen to natural prey on predation capacity of Neoseiulus cucumeris. J. Appl. Entomol., 147: 72-84. https://doi.org/10.1111/jen.13081
- Zhang Z.Q. 2003. Mites of greenhouses: identification, biology and control. London, UK: CABI Publishing,. pp. 244. https://doi.org/10.1079/9780851995908.0000



2023-11-21
Date accepted:
2024-06-13
Date published:
2024-06-20
Edited by:
Kreiter, Serge

This work is licensed under a Creative Commons Attribution 4.0 International License
2024 Gorji, Zahra; Shishehbor, Parviz; Kocheili, Farhan and Riahi, Elham
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



