Complementary description of the female of Typhlodromus (Anthoseius) xini Wu (Acari: Phytoseiidae) with the first description of its male from Sri Lanka
Döker, Ismail  1
; Weliwattage, Akash
1
; Weliwattage, Akash  2
; Jose, Anna
2
; Jose, Anna  3
and Gunasingham, Mikunthan
3
and Gunasingham, Mikunthan  4
4
1✉ Cukurova University, Agricultural Faculty, Department of Plant Protection, Acarology Lab, Adana, Turkey.
2University of Jaffna, Faculty of Agriculture, Department of Agricultural Biology, Ariviyal Nagar, Kilinochchi, Sri Lanka.
3University of Agricultural Sciences, Department of Agricultural Entomology, Bangalore, India.
4University of Jaffna, Faculty of Agriculture, Department of Agricultural Biology, Ariviyal Nagar, Kilinochchi, Sri Lanka.
2024 - Volume: 64 Issue: 3 pages: 833-842
https://doi.org/10.24349/j6jy-pejaZooBank LSID: 556EDE34-8BA4-4B43-8A3B-C903F1EC7E7C
Original research
Keywords
Abstract
Introduction
Species of the family Phytoseiidae (Acari: Mesostigmata) are of great importance for their potential as predators of phytophagous mites and some small soft bodied insects (McMurtry et al. 2013). The subgenus Anthoseius De Leon is one of the largest groups of phytoseiid mites with more than 400 species including synonyms (Ueckermann et al. 2008; Tsolakis and Ragusa 2015; Ferragut and Baumann 2019; Kreiter et al. 2021; Jose et al. 2024).
In Sri Lanka, the family Phytoseiidae has been scarcely studied and only 21 species belonging to 11 genera are known from the country (Demite et al. 2014; Moraes et al. 2004; Khaustov et al. 2021). Among them, Typhlodromus (Anthoseius) bifurcutus Wu, 1983 is the only known species of the subgenus (Moraes et al. 2004).
In this study, we reported Typhlodromus (Anthoseius) xini Wu, 1983 for the first time in Sri Lanka. A complementary description based on its females is provided. In addition, the male of T. (A.) xini is described for the first time.
Material and methods
Leaf samples of Cocos nucifera L. (Arecaceae) infested by spiralling whiteflies were collected from Wennapuwa, Puttalam District, North Western Province, Sri Lanka. Materials were collected and brought to the Department of Agricultural Biology, Faculty of Agriculture, University of Jaffna in sealed polyethylene bags. Individual leaflets were examined under a stereomicroscope (Nikon SMZ18), and predatory mites collected were kept in 70% alcohol. The collected mites were clarified in 60% lactic acid at 50 °C in a hotplate and they were mounted in Hoyer's medium on microscope slides. The permanent slides were examined with an Olympus® CX-41 microscope. Illustrations were prepared by using a U-Da drawing attachment, Camera Lucida. Final corrections were made using a computer program Adobe Photoshop (version CS6). The taxonomic system follows that of Chant and McMurtry (2007). Dorsal setal nomenclature is based on Lindquist and Evans (1965), as adapted by Rowell et al. (1978); ventral setal nomenclature is based on Chant and Yoshida-Shaul (1991); idiosomal dorsal and ventral setal patterns, on Chant and Yoshida-Shaul (1989, 1992); symbols of leg macrosetae, on Athias-Henriot (1957). Nomenclature for idiosomal solenostomes (gland pores) and poroids are based on Athias-Henriot (1971, 1975) and ventral poroids are based on Athias-Henriot (1971). Leg chaetotaxy follows that of Evans (1963). The measurements of each structure are given in micrometers, presented as the mean followed by the respective range in parentheses if more than two specimens examined. The voucher specimens are deposited in the mite collection of the Acarology Laboratory, Cukurova University, Adana, Turkey. As part of our study, we also examined the type specimens of Anthoseius species deposited in National Zoological collections, Prani Vigyan Bhawan, Zoological Survey of India, New Alipore, Kolkata, India.
Results
Typhlodromus (Anthoseius) xini Wu
Typhlodromus xini Wu, 1983: 16. Typhlodromus (Anthoseius) xini Wu, in Wu et al. 2009: 336.
Typhlodromus (Anthoseius) gosabaensis Kar and Karmakar, 2021: 55. New synonymy.
(Figures 1–3)
Material examined
Three females and two males from Cocos nucifera L. (Arecaceae) infested by spiralling whiteflies (Aleurodicus sp., Hemiptera: Aleyrodidae) in Wennapuwa, Puttalam District, North Western Province, Sri Lanka, 7°19′01.97″ N and 79°51′54.47″ E, 18 meters above sea level, 10 January 2024. Two females from the same location, 01 May 2024. All materials collected by Weliwattage A.T.M. Holotype female of Typhlodromus (Anthoseius) gosabaensis has also been examined.
Diagnosis
Female — Idiosomal setal pattern 12A:8A/JV–3:ZV (r3 and R1 off shield). Dorsal shield lightly reticulated; with five pairs of solenostomes (gd2, gd4, gd6, gd8 and gd9); dorsal setae serrated; setae Z4, Z5 and S5 spatulate (see description for variations). Peritremes extending between setae j1 and j3. Sternal shield lightly striated or reticulated, with two pairs of setae and with posterior lobe; ventrianal shield pentagonal, reticulated with three pairs of preanal setae and a pair of small preanal solenostomes; seta JV5 serrated and spatulate (bulbs not visible in some specimens). Spermatheca presents a funnel shaped calyx, and distally sclerotized walls; atrium well developed and nodular. Fixed digit of chelicera with two teeth clustered apically and movable digit with one tooth. Most of dorsal and lateral leg setae on most segments serrated. Ventral leg setae simple. Three spatulate macrosetae on leg IV. Trochanter I with six and genu II with seven setae.
Male — Idiosomal setal pattern 12A:8A/11:JV–3,4:ZV–1, 3 (r3 and R1 on shield). Dorsal shield lightly reticulated with six pairs of solenostomes (gd2, gd3, gd4, gd6, gd8 and gd9). Peritremes extending to base of setae j3. Morphology of dorsal setae similar to female (see description for variations). Ventrianal shield fused with peritrematal shield, triangular, reticulated with three pairs of preanal setae, with minute preanal solenostomes. Fixed digit with two teeth and movable digit with one tooth. Spermatophoral process L-shaped with tip of toe bulbous.
Complementary description
Female (n = 5)
(Figures 1–2)
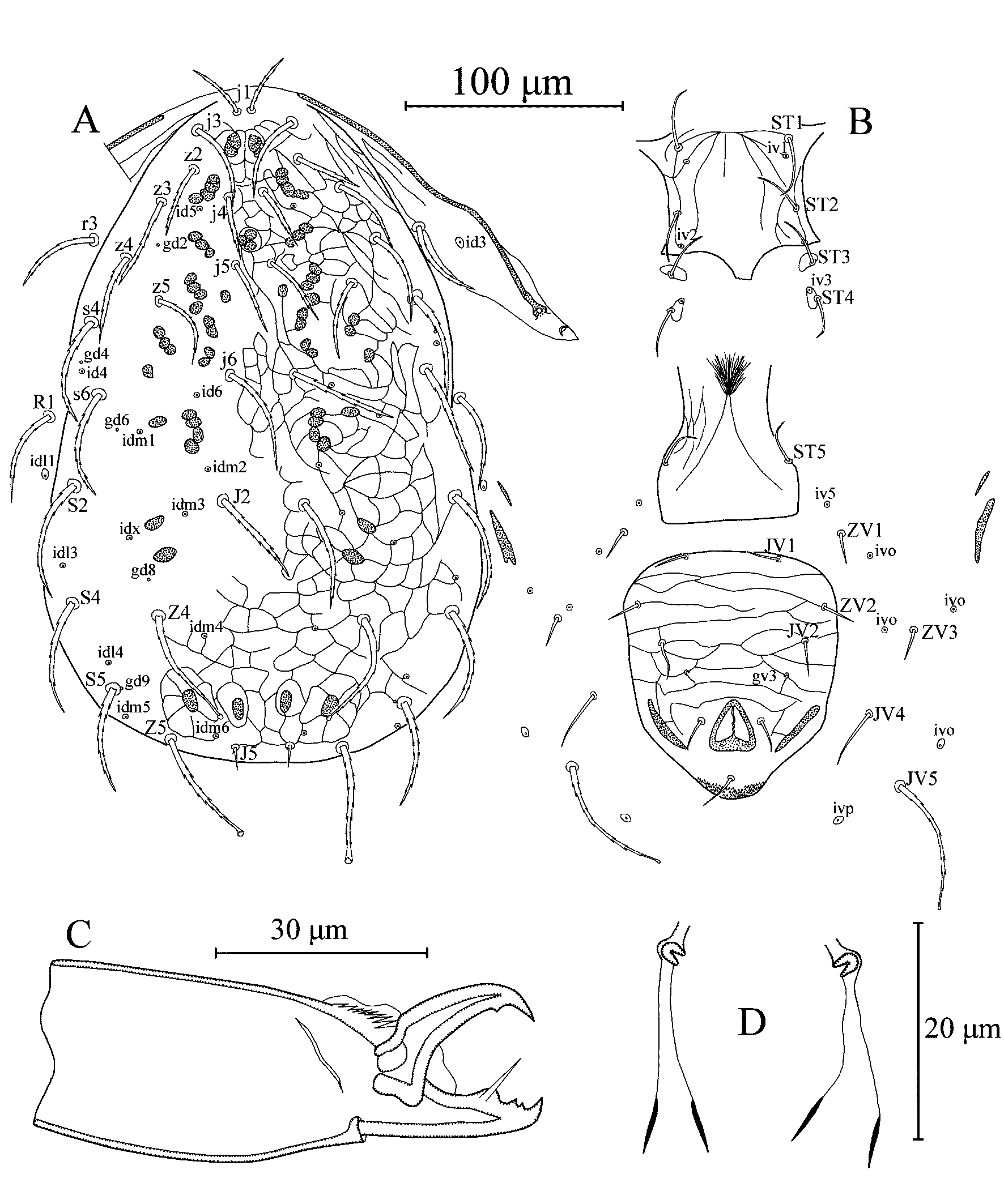


Dorsal idiosoma (Figure 1A) – Dorsal setal pattern 12A:8A (r3 and R1 off shield). Dorsal shield entire, slightly reticulated with five pairs of solenostomes (gd2, gd4, gd6, gd8 and gd9) and 13 pairs of visible poroids (id4, id5, id6, idm1, idm2, idm3, idm4, idm5, idm6, idl1, idl3, idl4 and idx). Length of dorsal shield 308 (298–330), width at level of s4 171 (160–180), width at level of S2 184 (160–195). Dorsal setae serrated; setae Z4 and Z5 spatulate, except one specimen, bulb absent in seta Z4 on right side; setae S5 spatulate only in one specimen. Measurements of dorsal setae as follows: j1 27 (25–28), j3 41 (38–43), j4 31 (30–33), j5 34 (30–36), j6 50 (48–54), J2 54 (50–58), J5 11 (9–13), z2 32 (30–34), z3 42 (39–45), z4 45 (43–46), z5 39 (38–40), Z4 56 (50–60), Z5 57 (55–63), s4 53 (50–55), s6 52 (50–53), S2 57 (53–60), S4 53 (50–55), S5 51 (48–53), r3 40 (38–40) and R1 46 (43–48). Peritremes extending between setae j1 and j3; solenostome gd3 not visible, and poroid id3 visible on peritrematal shield.
Ventral idiosoma (Figure 1B) – Ventral setal pattern 14: JV–3: ZV. Sternal shield lightly striated or reticulated in some specimens; with two pairs of setae (ST1 and ST2), two pairs of poroids (iv1 and iv2) and a posterior lobe; distance between ST1–iv2 49 (48–50), ST2–ST2 51 (48–55). Setae ST3 on separate platelets; metasternal setae ST4 and a pair of poroids (iv3) on metasternal platelets. Genital shield striated some patches of reticulations visible in some specimens, much narrower than ventrianal shield; width at level of setae ST5 60 (58–60); one pair of para-genital poroids iv5 on soft cuticle. Ventrianal shield pentagonal, reticulated with three pairs of pre-anal setae (JV1, JV2 and ZV2); one pair of paraanal (Pa) and a postanal seta (Pst); with a pair of minute preanal solenostomes (gv3) posteromesad setae JV2, distance between gv3 pores 42 (38–47). Length of ventrianal shield 114 (110–117), width at level of setae ZV2 94 (88–100). Setae ZV1, ZV3, JV4 and JV5 and five pairs of poroids (four pairs of ivo and ivp) on integument surrounding ventrianal shield. Setae JV5 serrated with spatulate tip, except two specimen bulbs not visible; much longer and thicker than other ventral setae, 60 (55–63) in length.
Chelicera (Figure 1C) – Fixed digit 26 (25–28) long, with two subapical teeth and pilus dentilis; movable digit 26 (25–26) long with one tooth.
Spermatheca (Figure 1D) – Calyx funnel-shaped, distally well sclerotized walls and connecting atrium with lightly sclerotized (membranous) walls, 21 (19–22) long including atrium, atrium c-shaped and nodular, strongly sclerotized.
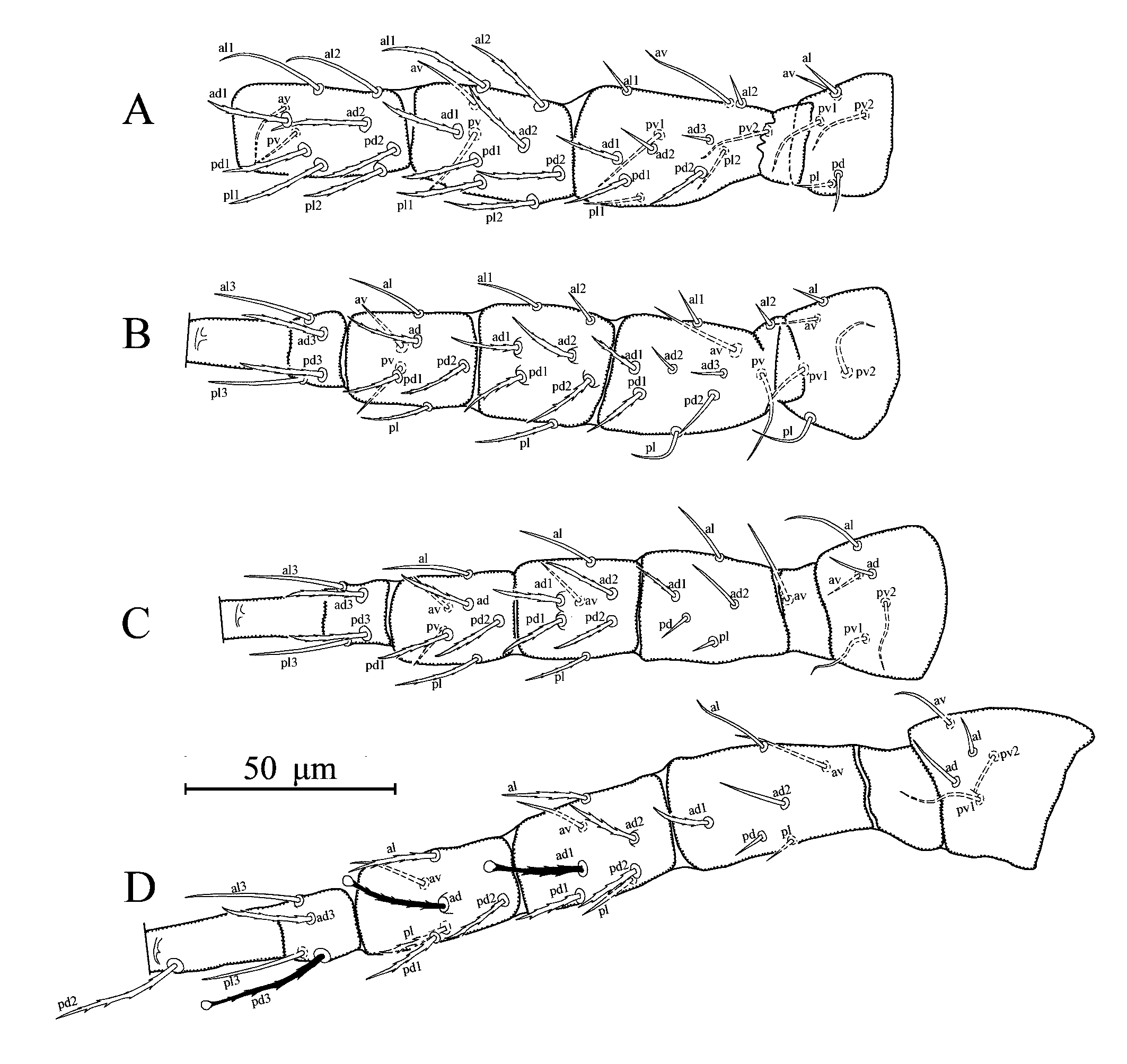


Legs (Figures 2A–D) – Length of legs (excluding pretarsus): I, 273 (260–288); II, 225 (220–230); III, 234 (225–245); IV, 286 (275–300). Chaetotactic formulae as follows: leg I: coxa 0 0/1 0/1 0, trochanter 1 0/1 1/2 1, femur 2 3/1 2/2 2, genu 2 2/1 2/1 2, tibia 2 2/1 2/1 2; leg II: coxa 0 0/1 0/1 0, trochanter 1 0/1 0/2 1, femur 2 3/1 2/1 1, genu 2 2/0 2/0 1, tibia 1 1/1 2/1 1; leg III: coxa 0 1/0 0/1 0, trochanter 1 1/1 0/2 0, femur 1 2/1 1/0 1, genu 1 2/1 2/0 1, tibia 1 1/1 2/1 1; leg IV: coxa 0 0/1 0/ 0 0, trochanter 1 1/1 0/2 0, femur 1 2/1 1/0 1, genu 1 2/1 2/0 1, tibia 1 1/1 2/0 1. Serrated setae as follows, leg I: femur ad1, pd1 and pd2, genu al1, al2, ad1, ad2, pd1, pd2, pl1, and pl2, tibia ad1, ad2, pd1, pd2, pl1, and pl2; legII: femur ad1and pd1, genu ad1, ad2, pd1, pd2 and pl, tibia ad, pd1, pd2 and pl, basitarsus ad3 and pd3; leg III: femur ad1, genu ad1, ad2, pd1, pd2 and pl, tibia ad, pd1, pd2 and pl, basitarsus ad3 and pd3; leg IV: femur ad1, genu al, ad1, ad2, pd1 and pd2, tibia al, ad, pd1, pd2 and pl, basitarsus ad3 and pd3. Leg IV with three spatulate macroseta; SgeIV (ad1) 25 (23–26), StiIV (ad) 24 (23–25) and StIV (pd3) 32 (30–33) long; other legs without macroseta.
Description of Male (n = 2)
(Figure 3)
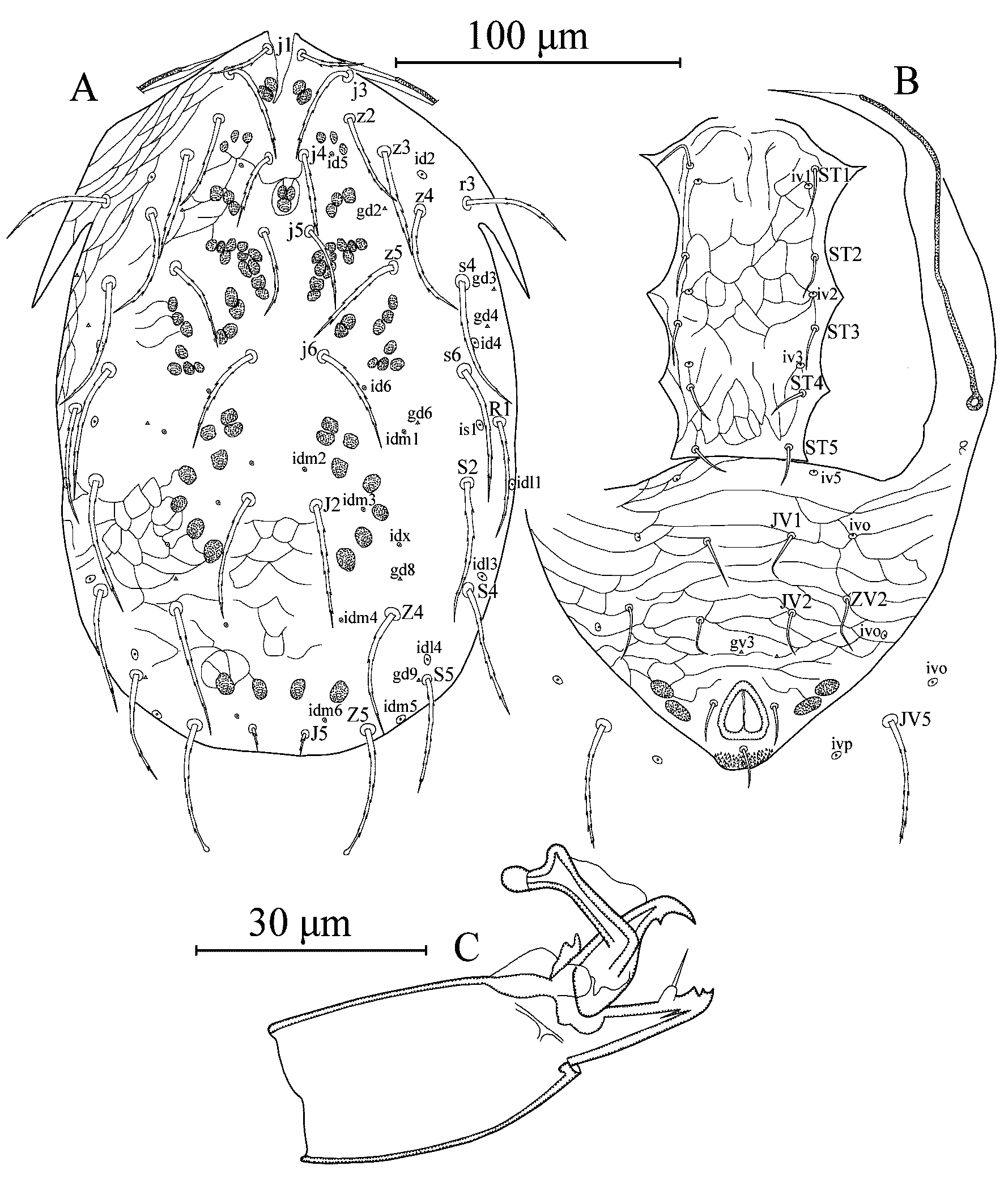


Dorsal idiosoma (Figure 3A) – Dorsal setal pattern 12A:8A (r3 and R1 on shield). Dorsal shield entire; oval; lightly reticulated, reticulations in anterior part of the illustrated specimen not visible, with six pairs of solenostomes (gd2, gd3, gd4, gd6, gd8 and gd9) and 15 pairs of visible poroids (id2, id4, id5, id6, idm1, idm2, idm3, idm4, idm5, idm6, is1, idl1, idl3, ild4 and idx). Length of dorsal shield 245–250, width at level of s4 150–155, width at level of S2 153–165. Dorsal setae serrate, only setae Z5 spatulate in the illustrated specimen, setae Z4, Z5 and R1 and seta S5 (right side) spatulate in another specimen. Measurements of dorsal setae as follows: j1 23–24, j3 37–38, j4 26–28, j5 28–30, j6 40–42, J2 44, J5 10–11, z2 31–34, z3 35–36, z4 40–41, z5 30–32 (right side 38 in the illustrated specimen), Z4 45–47, Z5 45–47, s4 44–48, s6 44–48, S2 47–51, S4 43–46, S5 38–41, r3 37–38 and R1 40–42. Peritremes extending level of setae j3.
Ventral idiosoma (Figure 3B) – Ventral setal pattern 11: JV–3,4: ZV–1,3 (seta JV2 absent in left side in one specimen). Sternogenital shield reticulated; with five pairs of setae (ST1, ST2, ST3, ST4 and ST5), three pairs of poroids (iv1, iv2 and iv3) and a straight posterior margin; distance between ST1–ST5 95–96, ST3–ST3 50–51. Ventrianal shield fused with peritrematal shields, triangular, reticulated; with three pairs of pre-anal setae (JV1, JV2, and ZV2), seta JV2 in left side absent in one specimen; one pair of paraanal (Pa) and a postanal seta (Pst); with a pair of minute preanal solenostomes (gv3); distance between gv3–gv3 19 in the illustrated specimen, 36 in another specimen; and with three pairs of poroids (iv5 and two pairs of ivo). Length of ventrianal shield 106–108, width at anterior margins 143–150. Setae JV5, serrate without knob, 45–47 in length. A pair of ivo and ivp on integument surrounding ventrianal shield.
Chelicera (Figure 3C) – Fixed digit 20 long with two subapical teeth and pilus dentilis; movable digit 18 long with one tooth. Spermatophoral process L-shaped, with toe strongly developed, bulbous and with hyaline bulb.
Legs – Length of legs (excluding pretarsus): I, 225–233; II, 190–195; III, 185–203; IV, 250–255. Leg chaetotaxy same as female. Serrated setae as follows, leg I: femur ad1 and pd1, genu al1, al2, ad1, ad2, pd1, pd2, pl1, and pl2, tibia ad1, ad2, pd1, pd2, pl1, and pl2; legII: femur ad1and pd1, genu ad1, ad2, pd1, pd2 and pl, tibia ad, pd1, pd2 and pl, basitarsus ad3 and pd3; leg III: femur ad1, genu ad1, ad2, pd1, pd2 and pl, tibia al, ad, pd1, pd2 and pl, basitarsus ad3 and pd3; leg IV: femur ad1, genu al, ad1, ad2, pd1 and pd2, tibia al, ad, pd1, pd2 and pl, basitarsus ad3 and pd3. Leg IV with three spatulate macrosetae; SgeIV (ad1) 21–22, StiIV (ad) 22 and StIV (pd1) 25–28; other legs without macroseta.
Remarks
Typhlodromus (Anthoseius) xini was described by Wu (1983) based on the holotype female collected from banana in Hainan Island, Guangdong, China. This is the first report of T. (A.) xini in Sri Lanka. The unknown males of the species are also described for the first time. Morphological characters and measurements of the female specimens examined in this study fit well with those provided in its original description. However, setae Z4, Z5, S5 (in some specimens), and JV5 (in some specimens) are spatulate in the examined females but forked in the holotype (Wu 1983; Wu et al. 2009). It should be noted here that these setal bulbs consist of hyaline membranes that are lightly sclerotized and might not be visible under the optic material used at that time. However, the well-sclerotized apical parts of these setae at the connection of hyaline membranes exactly seem to be forked. Furthermore, preanal pores are present in the current material but absent in the holotype. It should also be noted here that these preanal pores are smaller than those of other mites in the subgenus Anthoseius, and observable only by using an oil lens (1.25 NA). Therefore, we consider these differences as variations or flaws of the original description due to the quality of the microscope used at that time.
The specimens of T. (A.) xini examined in this study resemble T. (A.) gosabaensis Kar & Karmakar, 2021, a species recently described from West Bengal (India), in many aspects such as the nature of the dorsal and ventral idiosoma (including the shape and the length of the setae). However, based on the original description, T. (A.) gosabaensis show important morphological differences with T. (A.) xini: (1) the absence of gd4 and the presence of gd5 (vs opposite situation stands for T. (A.) xini; (2) chelicera dentition (including arrangement and positions of teeth); (3) only dorsal and lateral setae are serrated on some leg segments but serration never observed for ventral setae in T. (A.) xini (vs all leg setae serrated); (4) Chaetotactic formula of genu II 2 2/0 2/0 1 in T. (A.) xini (vs 1 2/0 2/1 1); (5) measurements of seta z4 45 (43–46) in T. (A.) xini (vs 33 (32–34)). We could successfully reach and examine the holotype, which was deposited in the National Zoological Collections, Prani Vigyan Bhawan, Kolkata, India. Our observation on T. (A.) gosabaensis holotype revealed that the gd5 solenostomes are absent and gd4 are present. Moreover, we also considered other differences mentioned above and concluded that these are different from the original description of T. (A.) gosabaensis but identical to the complementary description of T. (A.) xini provided here. Therefore, T. (A.) gosabaensis is proposed as a new junior synonymy of T. (A.) xini.
Taxonomic relationship of Typhlodromus (Anthoseius) xini with other species in the subgenus Anthoseius
Typhlodromus (Anthoseius) xini shows affinity to several species in the subgenus Anthoseius such as T. (A.) diumbokus Schicha & Corpuz-Raros, 1992, T. (A.) hartlandrowei Evans, 1958, T. (A.) neohartlandrowei Zannou, Moraes & Oliveira, in Ueckermann et al. (2008), and T. (A.) transvaalensis (Nesbitt, 1951). All of these species are similar to each other based on the nature of their dorsal and ventral aspects, chelicera dentition, and spermetheca morphology. Most importantly, ventral seta JV3 is absent in all of these species.
Typhlodromus (A.) xini can be separated from T. (A.) diumbokus by having much longer and serrated dorsal seta S5 which is minute and smooth in the latter species. It is also different from T. (A.) hartlandrowei by having much shorter setae z4, S2 and S4, and by having longer setae z2 and S5 (Kreiter et al. 2020). From, T. (A.) neohartlandrowei by having longer seta S5 and shorter setae z4, Z4, s6, S2, S4, and R1 (Ueckermann et al. 2008). Moreover, T. (A.) xini is also different from T. (A.) transvaalensis by having seven setae on genu II as oppose to eight in the latter species. In addition, T. (A.) xini has much longer dorsal setae compared to T. (A.) transvaalensis. Shape of the ventrianal shield is also one of the basic differences between these two species (Ueckermann et al. 2008; Liao et al. 2020).
It is interesting to report that T. (A.) xini is also similar to T. (A.) foraminosus (Schuster, 1966) described from Galapagos Islands located in Neotropical realm, based on the dorsal setal measurements. However, it can be separated from the latter species by having a pentagonal ventrianal shield, which is narrower and slightly elongated in T. (A.) foraminosus. In addition, measurements of macrosetae SgeIV and StIV are 25 (23–26) and 32 (30–33) in T. (A.) xini as oppose to 38 and 50 in T. (A.) foraminosus, respectively.
Finally, T. (A.) xini is also different from all of the aforementioned species, by having many serrated dorsal and lateral setae on most of the leg segments. Considering all of the morphological similarities between these species, it is apparent that all of the aforementioned species share a common ancestor. Therefore, additional molecular analysis, in combination with the use of morphological characters, would help to confirm the taxonomic relationship and evolutionary history of these species.
Acknowledgements
We are grateful to Dr. Jhih-Rong Liao (Systematic Zoology Laboratory, Department of Biological Sciences, Tokyo Metropolitan University, Tokyo, Japan) for translation of the original description of T. (A.) xini from Chinese. Dr. Shelley Acharya for her help in examination of the holotype female of T. (A.) gosabaensis deposited in National Zoological collections, Prani Vigyan Bhawan, New Alipore, Kolkata, India. Study of Ismail Döker was supported by Cukurova University Scientific Projects Foundation Units, grant number, FAY-2022-14495. We also extend our gratitude to Prof. K. Pakeerathan, Head, Department of Agricultural Biology, Faculty of Agriculture, University of Jaffna, for the continuous support given throughout the research period.
References
- Athias-Henriot C. 1957. Phytoseiidae et Aceosejidae (Acarina, Gamasina) d'Algérie. I. Genres Blattisocius Keegan, Iphiseius Berlese, Amblyseius Berlese, Phytoseius Ribaga, Phytoseiulus Evans. Bull. Soc. Hist. Natur. Afr. Nord, 48: 319-352.
- Athias-Henriot C. 1971. La divergence néotaxique des Gamasides (Arachnides). Bull. Scientif. Bourgogne, 28: 93-106.
- Athias-Henriot C. 1975. Nouvelles notes sur les Amblyseiini. II. Le relevé organotaxique de la face dorsale adulte (Gamasides, protoadéniques, Phytoseiidae). Acarologia, 17: 20-29.
- Chant D.A., McMurtry J.A. 2007. Illustrated keys and diognoses for the genera and subgenera of the Phytoseiidae of the world (Acari: Mesostigmata). West Bloomfield, Indira Publishing House, 219 pp.
- Chant D.A., Yoshida-Shaul E. 1989. Adult dorsal setal patterns in the family Phytoseiidae (Acari: Gamasina). Int. J. Acarol., 15: 219-223. https://doi.org/10.1080/01647958908683852
- Chant D.A., Yoshida-Shaul E. 1991. Adult ventral setal patterns in the family Phytoseiidae (Acari: Gamasina). Int. J. Acarol., 17: 187-199. https://doi.org/10.1080/01647959108683906
- Chant D.A., Yoshida-Shaul E. 1992. Adult idiosomal setal patterns in the family Phytoseiidae (Acari: Gamasina). Int. J. Acarol., 18: 177-193. https://doi.org/10.1080/01647959208683949
- Demite P.R., McMurtry J.A., Moraes G.J. de 2014. Phytoseiidae Database: a website for taxonomic and distributional information on phytoseiid mites (Acari). Zootaxa, 3795: 571-577. https://doi.org/10.11646/zootaxa.3795.5.6
- Evans G.O. 1958. Some mesostigamatid mites from a nest of social spiders in Uganda. Ann. Mag. Nat. Hist., Ser. 13, 1: 580-590. https://doi.org/10.1080/00222935808650985
- Evans G.O. 1963. Observations on the chaetotaxy of the legs in the free-living Gamasina (Acari: Mesostigmata). Bull. Br. Mus. Nat. Hist., Zool., 10: 275-303. https://doi.org/10.5962/bhl.part.20528
- Ferragut F., Baumann J. 2019. New phytoseiid mites (Mesosstigmata: Phytoseiidae) of Mauritius, with the description of two new species. Syst. Appl. Acarol., 24: 825-856. https://doi.org/10.11158/saa.24.5.8
- Jose A., Döker I., Gowda C.C., Hiremath R. 2024. Complementary description of three species of the subgenus Anthoseius De Leon (Acari: Phytoseiidae) from India. Syst. Appl. Acarol., 29: 305-323. https://doi.org/10.11158/saa.29.2.10
- Kar A., Karmakar K. 2021. Description of three new species of phytoseiid mites (Acari: Mesostigmata) from Sundarban, West Bengal, India. Int. J. Acarol., 47: 51-60. https://doi.org/10.1080/01647954.2020.1870555
- Khaustov V., Döker I., Joharchi O., Ermilov S.G. 2021. A new species of Proprioseiulus Muma (Acari: Phytoseiidae) from Sri Lanka with a key to world species. Syst. Appl. Acarol., 26: 464-473. https://doi.org/10.11158/saa.26.2.10
- Kreiter S., Payet R.-M., Mouigni H., Douin M., Tixier M.-S., Azali H.A. 2021. New records of phytoseiid mites (Acari: Mesostigmata) of Grande Comore Island (Comoros Archipelago). Acarologia, 61: 241-273. https://doi.org/10.24349/acarologia/20214429
- Kreiter S., Payet R.-M., Douin M., Fontaine O., Fillâtre J., Le Bellec F. 2020. Phytoseiidae of La Réunion Island (Acari: Mesostigmata): three new species and two males described, new synonymies, and new records. Acarologia, 60: 111-195. https://doi.org/10.24349/acarologia/20204361
- Liao L.-R., Ho C.-C., Lee H.-C., Ko C.-C. 2020. Phytoseiidae of Taiwan (Acari: Mesostigmata). Taipei, Taiwan, National Taiwan University Press, 538 pp.
- Lindquist E.E., Evans G.O. 1965. Taxonomic concepts in the Ascidae, with a modified setal nomenclature for the idiosoma of the Gamasina (Acarina: Mesostigmata). Mem. Ent. Soc. Can., 47: 1-64. https://doi.org/10.4039/entm9747fv
- McMurtry J.A., Moraes G.J. de., Sourassou N.F. 2013. Revision of the lifestyles of phytoseiid mites (Acari: Phytoseiidae) and implications for biological control strategies. Syst. Appl. Acarol., 18: 297-320. https://doi.org/10.11158/saa.18.4.1
- Moraes G.J. de, Lopes P.C., Fernando C.P. 2004. Phytoseiid mite (Acari: Phytoseiidae) of coconut growing areas in Sri Lanka, with descriptions of three new species. J. Acarol. Soc. Jpn., 13: 141-160. https://doi.org/10.2300/acari.13.141
- Nesbitt H.H.J. 1951. A taxonomic study of the Phytoseiidae (Family Laelaptidae) predaceous upon Tetranychidae of economic importance. Zool. Verh., 12, 64 pp.+ 32 plates.
- Rowell H.L., Chant D.A., Hansell R.I.C. 1978. The determination of setal homologies and setal patterns on the dorsal shield in the family Phytoseiidae (Acarina: Mesostigmata). Can. Entomol., 110: 859-876. https://doi.org/10.4039/Ent110859-8
- Schicha E., Corpuz-Raros L.A. 1992. Phytoseiidae of the Philippines. Bloomfield, Indira Publishing House, 190 pp.
- Schuster R.O. 1966. Phytoseiidae of the Galapos Islands (Acarina: Mesostigmata). Pac. Insects, 8: 319-339.
- Tsolakis H., Ragusa S. 2015. Considerations on systematics of the Phytoseiidae (Acari: Mesostigmata), with definition of a new species group and description of a new species. Zootaxa, 3926: 229-243. https://doi.org/10.11646/zootaxa.3926.2.4
- Ueckermann E.A., Zannou I.D., Moraes G.J. de, Oliveira A.R. de, Hanna R., Yaninek, J.S. 2008. Phytoseiid mites of the tribe Typhlodromini (Acari: Phytoseiidae) from sub-Saharan Africa. Zootaxa, 1901: 1-122. https://doi.org/10.11646/zootaxa.1901.1.1
- Wu W.N. 1983. Two new species of Typhlodromus Scheuten (Acarina: Phytoseiidae) from China. Zool Res. Zool. Res. (China), 4: 15-18 [in Chinese].
- Wu W.N., Ou J.F., Huang J.L. 2009. Fauna Sinica, Invertebrata vol. 47. Arachnida Acari: Phytoseiidae. Beijing, Science Press, 511 pp. [In Chinese with English abstract].



2024-05-13
Date accepted:
2024-06-15
Date published:
2024-06-21
Edited by:
Kreiter, Serge

This work is licensed under a Creative Commons Attribution 4.0 International License
2024 Döker, Ismail; Weliwattage, Akash; Jose, Anna and Gunasingham, Mikunthan
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



