A new troglobiont species of Rhagidiidae (Acari: Eupodoidea) from cave sites in the Ural Mountains (Russia)
Khaustov, Alexander A.  1
; Kolesnikov, Vasiliy B.
1
; Kolesnikov, Vasiliy B.  2
; Turbanov, Ilya S.
2
; Turbanov, Ilya S.  3
and Tolstikov, Andrei V.
3
and Tolstikov, Andrei V.  4
4
1✉ Tyumen State University, Tyumen, 6 Volodarskogo Str., 625003, Russia.
2All-Russian Research Institute of Plant Protection, Voronezh, Russia.
3Papanin Institute for Biology of Inland Waters, Russian Academy of Sciences, Borok, Yaroslavl Region, 152742, Russia & Cherepovets State University, Cherepovets, Vologda Region 162600, Russia.
4Tyumen State University, Tyumen, 6 Volodarskogo Str., 625003, Russia.
2024 - Volume: 64 Issue: 3 pages: 787-802
https://doi.org/10.24349/o7sw-gepqZooBank LSID: AEEDBF76-3C11-438C-AC0F-DF3B23B17EF2
Original research
Keywords
Abstract
Introduction
The cosmopolitan family Rhagidiidae Oudemans, 1922 (Acari: Eupodoidea) currently includes 29 genera, one of which is in a fossil form (Zhang et al. 2011; Khaustov 2015). Rhagidiid mites inhabit soil, forest litter, mosses, lichens, etc. Many rhagidiid species are troglobionts, i.e., they live in caves. This group includes all representatives of the genera Traegaardhia Zacharda, 1980; Troglocheles Zacharda, 1980; Elliotia Zacharda, 1980; and Flabellorhagidia Elliott, 1976, as well as some species of the genera Rhagidia Thorell, 1872; Foveacheles Zacharda, 1980; Robustocheles Zacharda, 1980; and Poecilophysis Cambridge, 1876 (Zacharda 1980, 1985, 1987; Zacharda, Elliott 1981; Zacharda, Pugsley 1988; Walter et al. 2009; Zacharda et al. 2011). Some troglobiont species of Rhagidiidae are characterized by troglomorphism that is expressed in an unusually large quantity and size of rhagidial solenidia and unusually long legs (Zacharda 1980). Rhagidiid mites are predators of small arthropods and nematodes (Zacharda 1980).
The family Rhagidiidae on the territory of Russia is insufficiently studied. Zacharda (1983) described and recorded 13 species of Rhagidiidae from various regions of Russia: Foveacheles (Foveacheles) caucasica Zacharda, 1983; F. (F.) cegetensis Zacharda, 1983; F. (F.) brevichelae Zacharda, 1980; and Poecilophysis (Procerocheles) pseudoreflexa Zacharda, 1980 from Central Caucasus; P. (Soprocheles) saxonica (Willmann, 1934) from Central Caucasus and Central Siberia; P. (Dentocheles) pratensis (C.L. Koch, 1835) from Central Caucasus and Kamchatka; Rhagidia (Noerneria) punkva Zacharda, 1980 from Central Siberia; Robustocheles (Robustocheles) mucronata (Willmann, 1936) from Central Caucasus and Central Siberia; Evadorhagidia bezdezensis Zacharda, 1980 from Central Siberia; Coccorhagidia clavifrons (Canestrini, 1886) from Central Caucasus; C. pittardi Strandtmann, 1971, Thoria uniseta (Thor, 1909) and Th. brevisensilla Zacharda, 1980 from Central Caucasus and Central Siberia. Also, Makarova (2002) recorded Robustocheles hilli (Strandtmann, 1971) from the Severnaya Zemlya Archipelago, and Hammenia macrostella Zacharda, 1980 from the Taymyr Peninsula (Makarova 2015). Khaustov (2015) described a new genus and species, Paracrassocheles clavisetosus Khaustov, 2015, from Crimea. Troglobiont species of Rhagidiidae have never been described from Russia.
The genus Foveacheles is one of the largest in the family Rhagidiidae and currently comprises 34 species grouped into 10 subgenera and one species group (Zacharda 1996, 2000). The subgenera Hirschmanniella Zacharda, 1980, Proxistella Zacharda, 1980, Spelaeocheles Zacharda, 1988, Trofocheles Zacharda, 1982, and Usitorhagidia Zacharda, 1980 include troglobiont species (Zacharda 1980, 1982, 1988).
This paper presents the first description of a troglobiont species, Foveacheles uralensis n. sp., from three caves located in the Ural Mountains of Russia.
Material and methods
Mites were collected by hand inside the caves and immediately fixed in 96% alcohol. The collected mites were mounted in Hoyer's medium. The notations for the prodorsal and leg setae follow those of Lindquist and Zacharda (1987) and Baker (1995), while the remaining nomenclature is that applied to eupodoids by Baker (1990). All measurements are given in micrometers (μm) for the holotype; paratypes were not measured because their soft bodies are strongly damaged. In the descriptions of leg setation, the number of solenidia is given in parentheses (μm). Mite morphology was studied using a Carl Zeiss Axio Imager A2 compound microscope with phase contrast and DIC optics, as well as a Bioptic C-400 microscope with DIC optics. Photomicrographs were taken with an AxioCam ICc5 digital camera.
Photographs of the live specimens of Foveacheles uralensis n. sp. were made using a Sony Alpha NEX-5T digital camera equipped with a Sony E 3.5/30 MACRO lens.
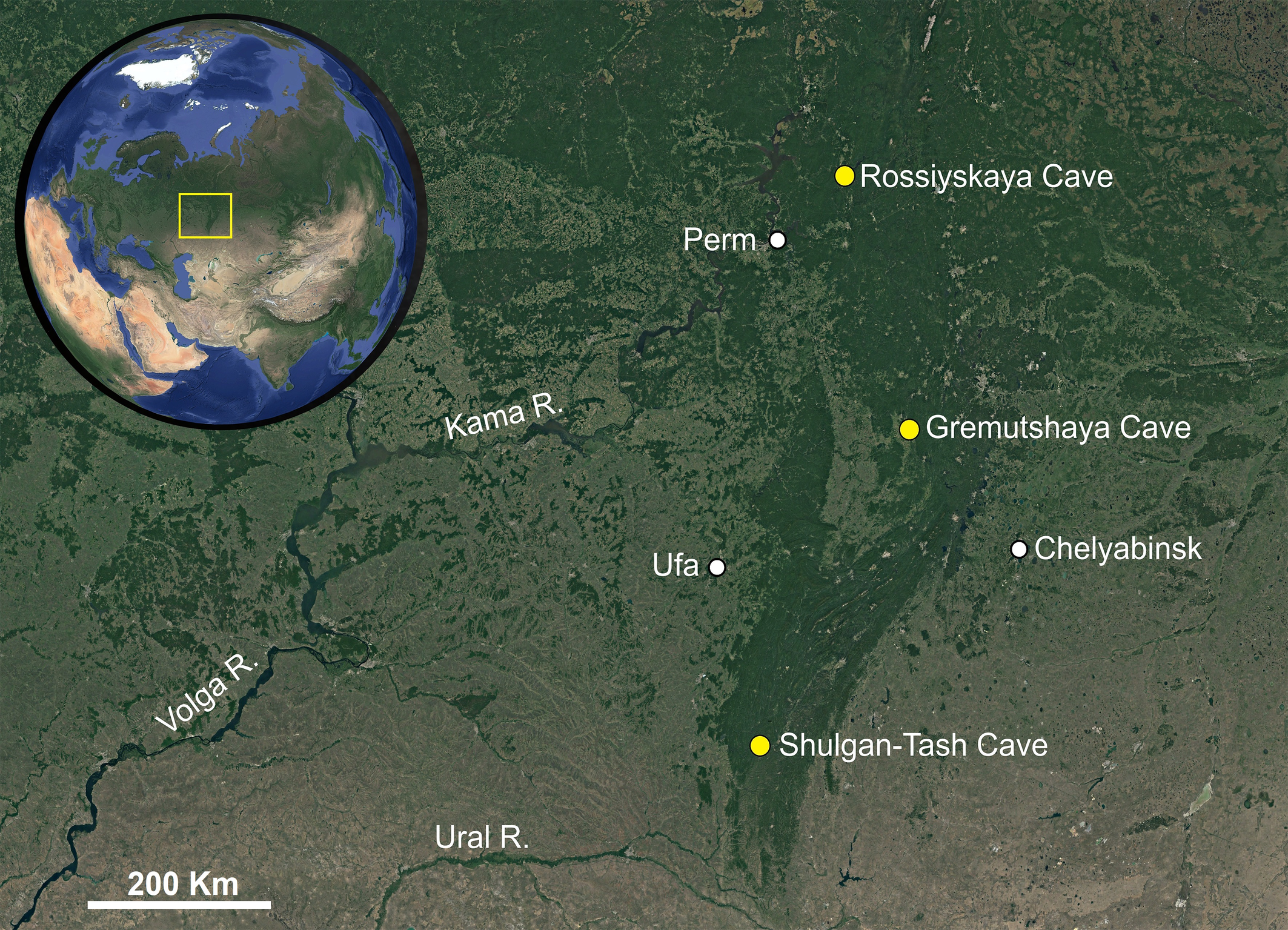
The distribution map (Fig. 1) was created using Google Earth Pro (ver. 7.3.4.8248) and Adobe Photoshop CS6 (ver. 13.0.1.3).


Systematics
Family Rhagidiidae Oudemans, 1922
Genus Foveacheles Zacharda, 1980
Type species: Foveacheles osloensis (Thor, 1934), by original designation
Foveacheles uralensis n. sp.
ZOOBANK: 4862C3D6-E3C8-44FA-A0A3-BC184B30E6B7 ![]()
(Figures 2–11)
Description
Female — Body white in color (Fig. 11). Length of idiosoma 1340, width 560.
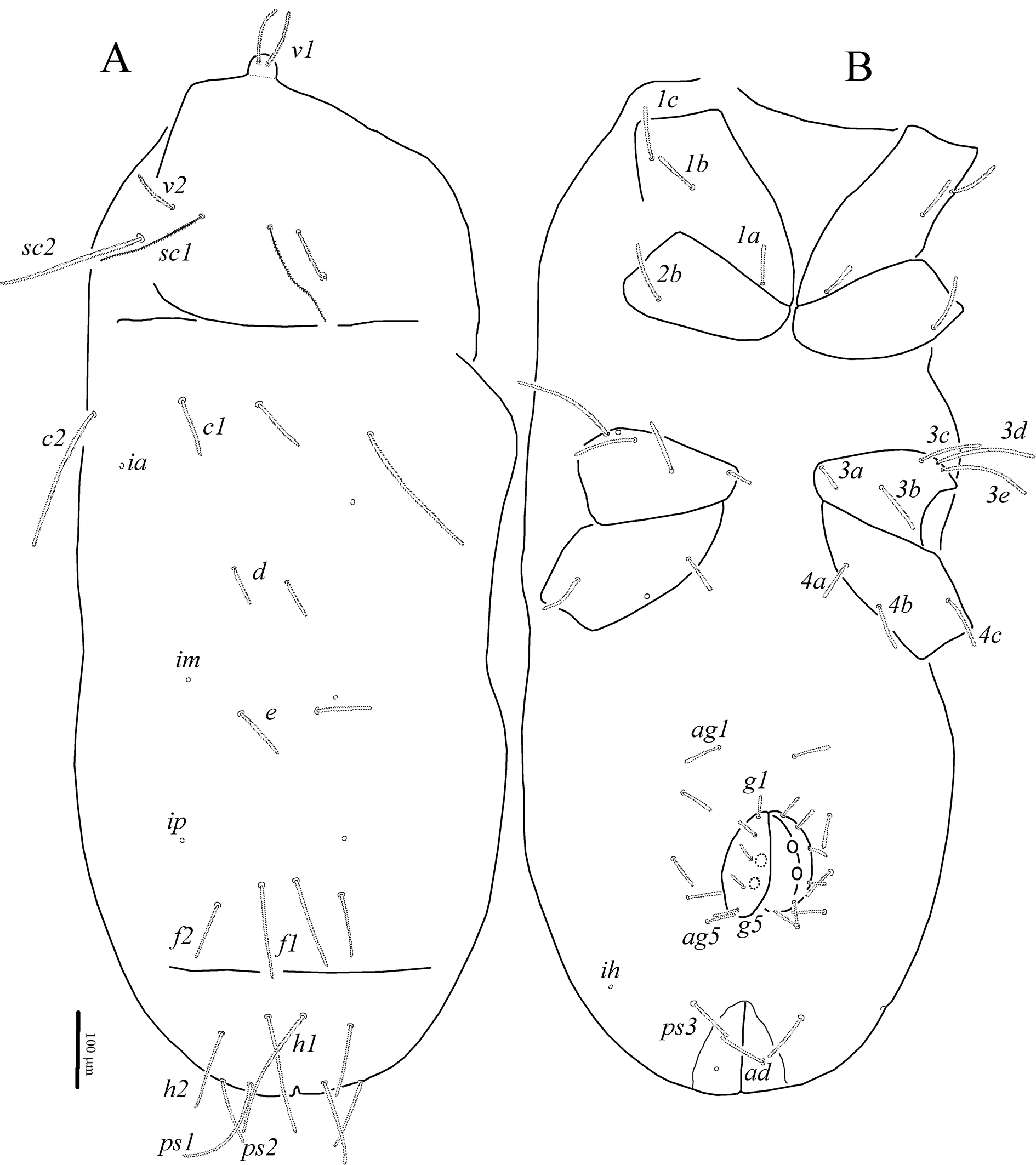




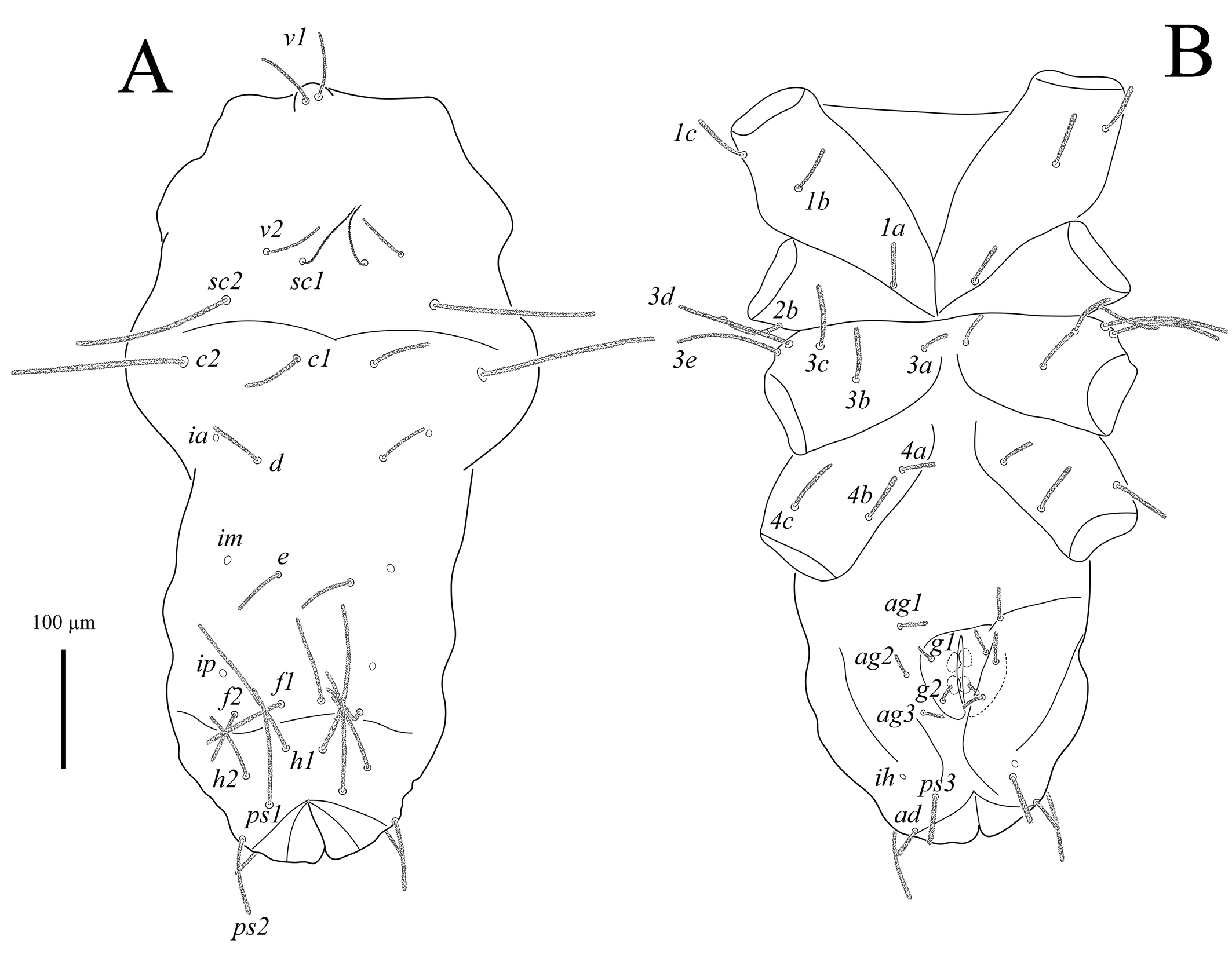
Idiosoma (Figs 2A, B). Cuticle finely striated. All dorsal and ventral setae uniform in shape, densely pilose. Trichobothria sc1 thin, with large barbs; setae v1 situated in bothridia on well-developed anteriorly rounded naso. Transverse furrow visible posteriad setae f1 and f2. Setae f2 present. Lyrifissures ia situated posterolaterally to bases of setae c1; im situated posterolaterad bases of setae d; ip situated anterolaterad bases of setae f2; ih located anterolaterally to setae ps3. Three pairs of pseudanal, one pair of adanal, five pairs of both aggenital and genital setae present. Epimeral formula: 3-1-5-3; in some specimens coxisternal fields I and III with asymmetrical presence of four and six setae, respectively (Fig. 6A). Progenital chamber with two pairs of large, oval genital papillae and well-developed ovipositor with numerous eugenital setae. Lengths of idiosomal setae: v1 102, v2 65, sc1 145, sc2 195, c1 80, c2 187, d1 80, e1 77, f1 123, f2 77, h1 180, h2 107, ps1 148, ps2 95, ps3 65, ad 64, 1a 53, 1b 62, 1c 73, 2b 79, 3a 34, 3b 72, 3c 84, 3d 135, 3e 125, 4a 49, 4b 67, 4c 74, ag1 52, ag2 45, ag3 47, ag4 45, ag5 42, g1 37, g2 36, g3 28, g4 23, g5 35.
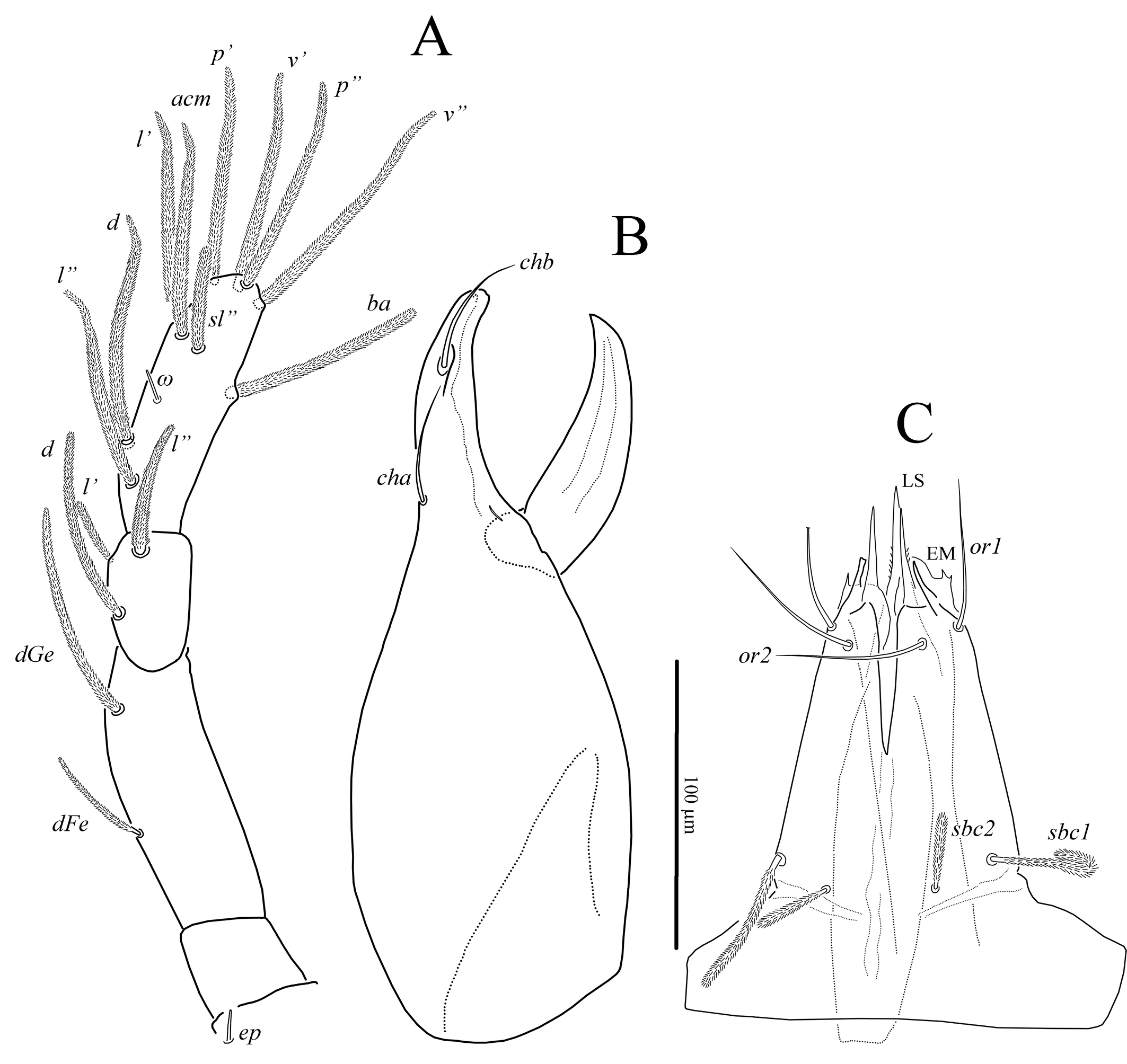
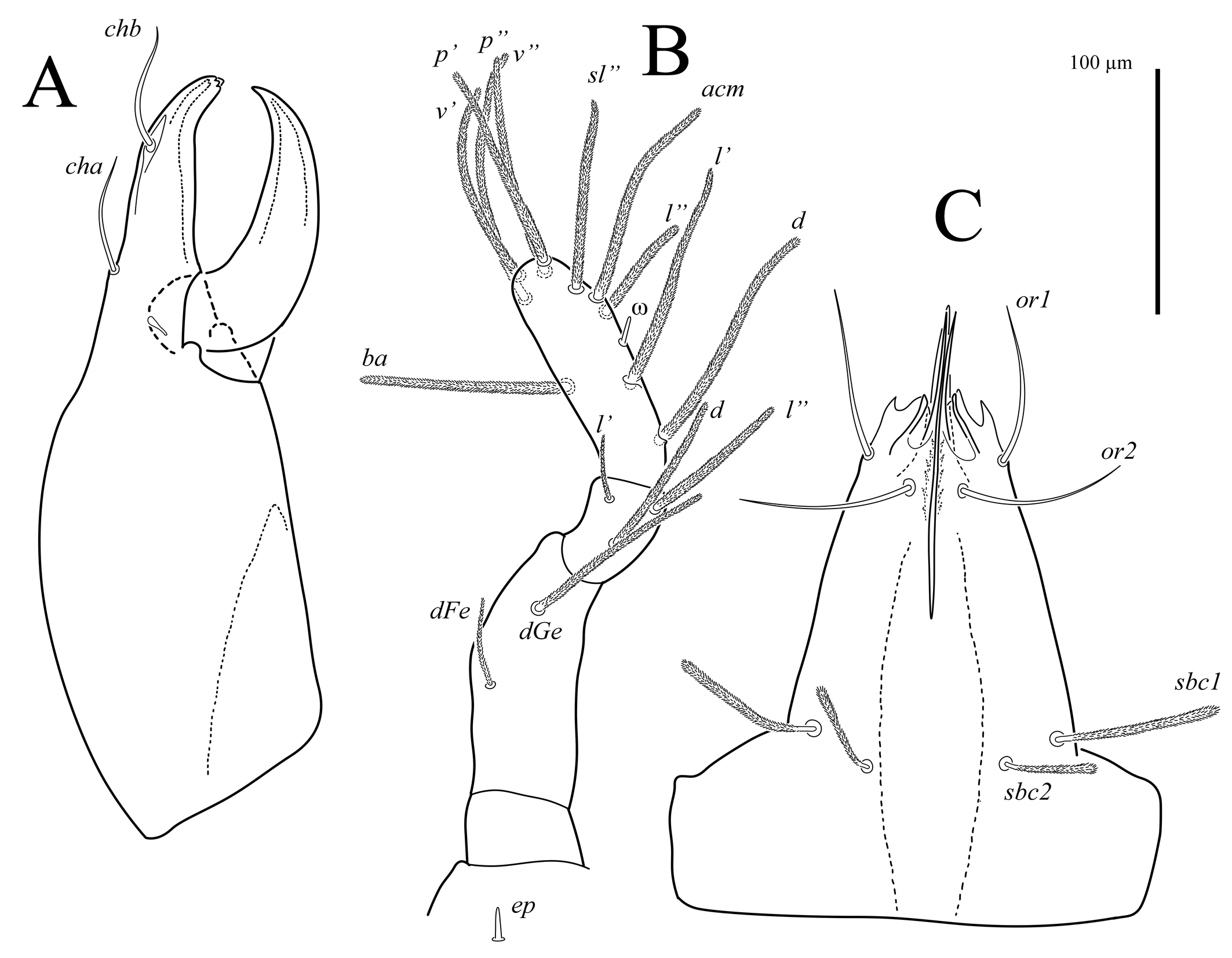
Gnathosoma (Figs 3, 7A). Integument papillate. Subcapitulum (Fig. 3C) roughly triangular, with two pairs of smooth adoral setae (or1-2), located subapically; subcapitular setae sbc1-2 densely pilose, located at level of proximal margin of palp trochanters; labrum (LS) acuminate; external malae (EM) weakly sclerotized, with several small projections. Lengths of subcapitular setae: sbc1 97, sbc2 51, or1 85, or2 89. Chelicera (Fig. 3A) 410 in length; length of movable digit 155. Movable digit with smooth inner margin. With lyrifissure-like structure located near articulation of movable digit with fixed digit. Seta chb situated dorsolaterally in shallow depression (Fig. 7A), seta cha situated dorsally at level of articulation of movable digit with fixed digit; tip of fixed digit with three short cusps. Palps (Fig. 3B) with supracoxal seta ep smooth, baculiform. Length of palp 405. Palp setation: Tr 0, FeGe 2 (dFe, dGe), Ti 3 (d, l′, l″), Ta 10 (1) (d, l′, l″, acm, sl″, p′, p″, v′, v″, ba, ω). All palpal setae densely pilose; solenidion ω small, rod-like.
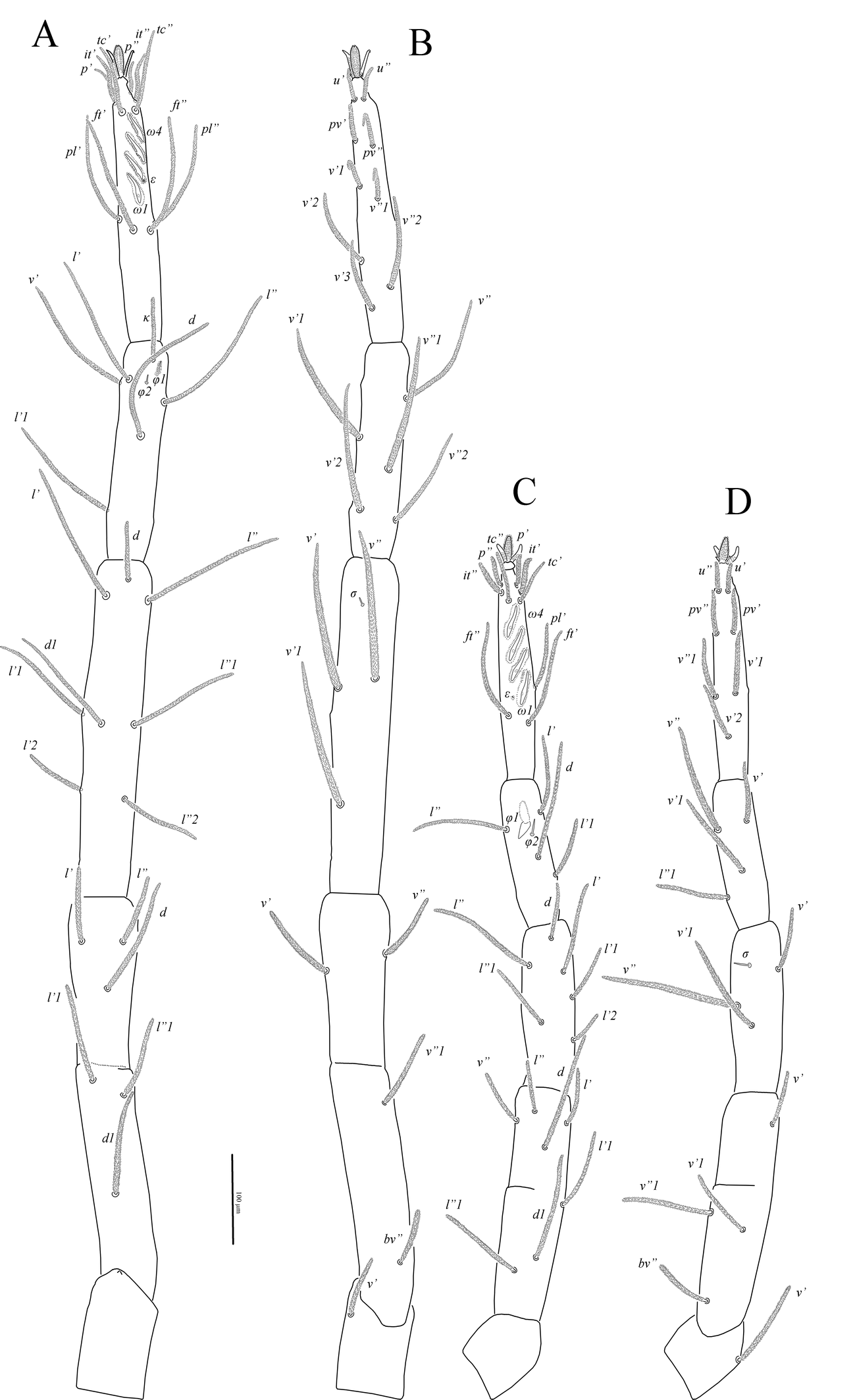


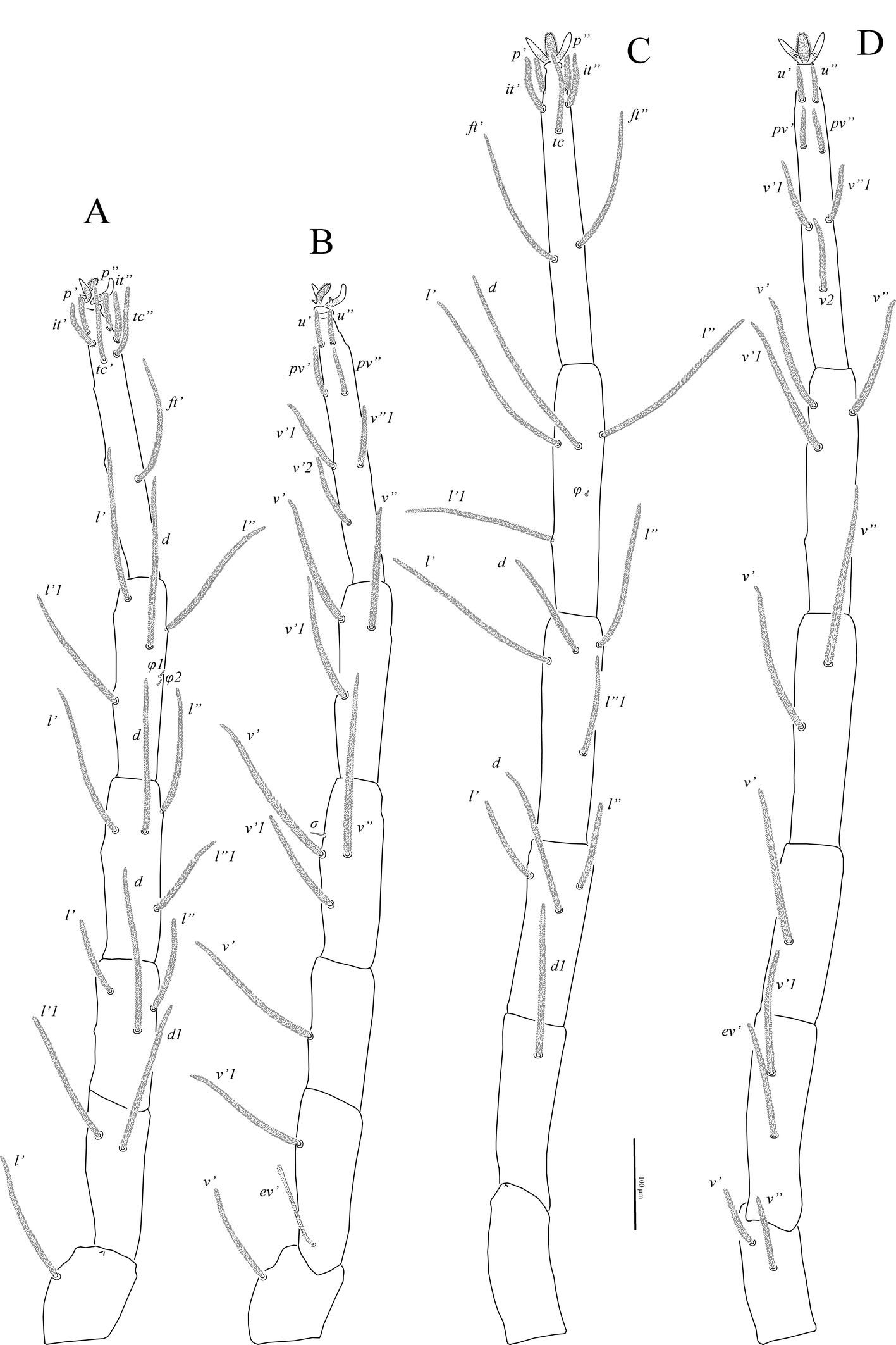


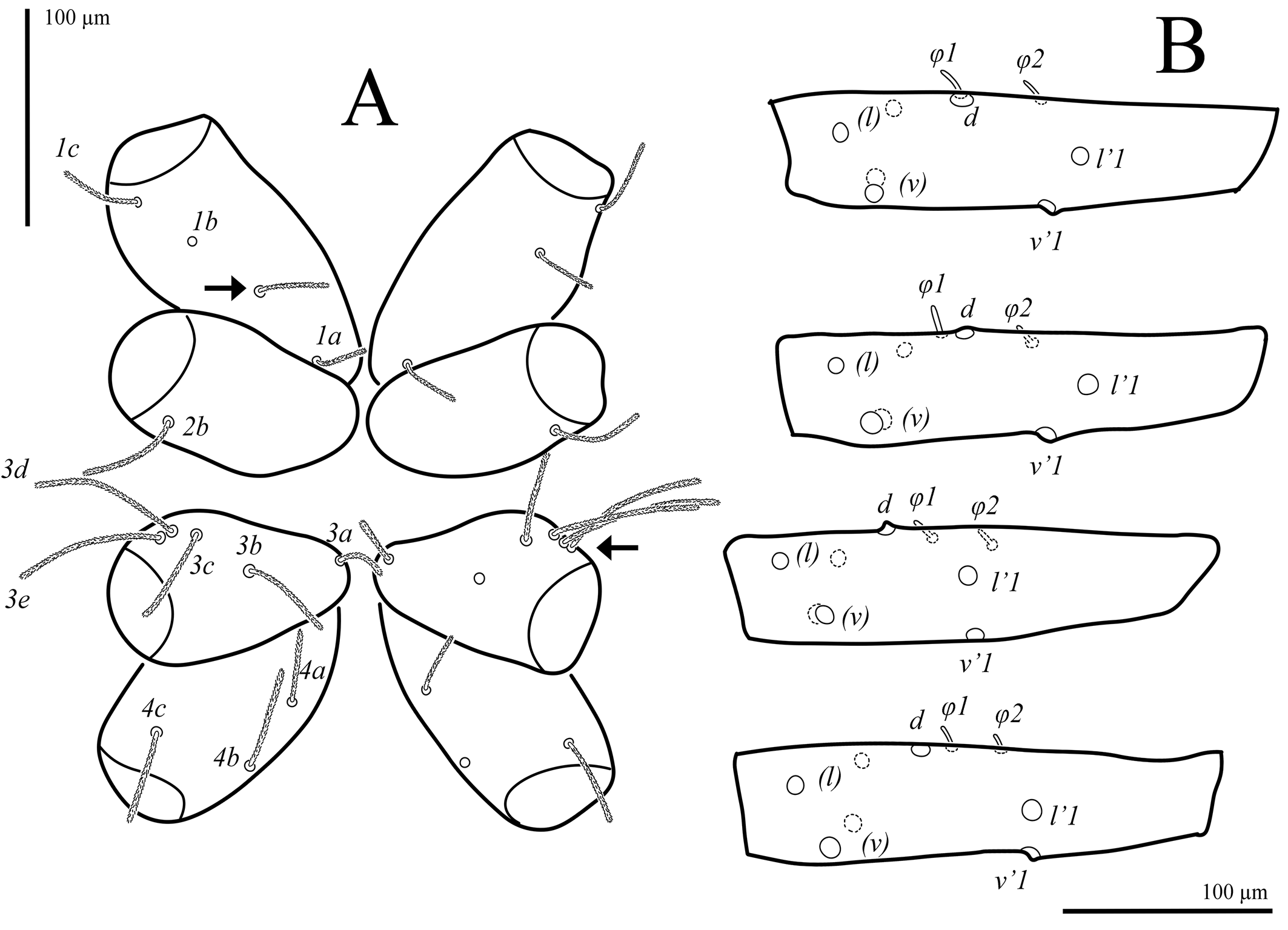


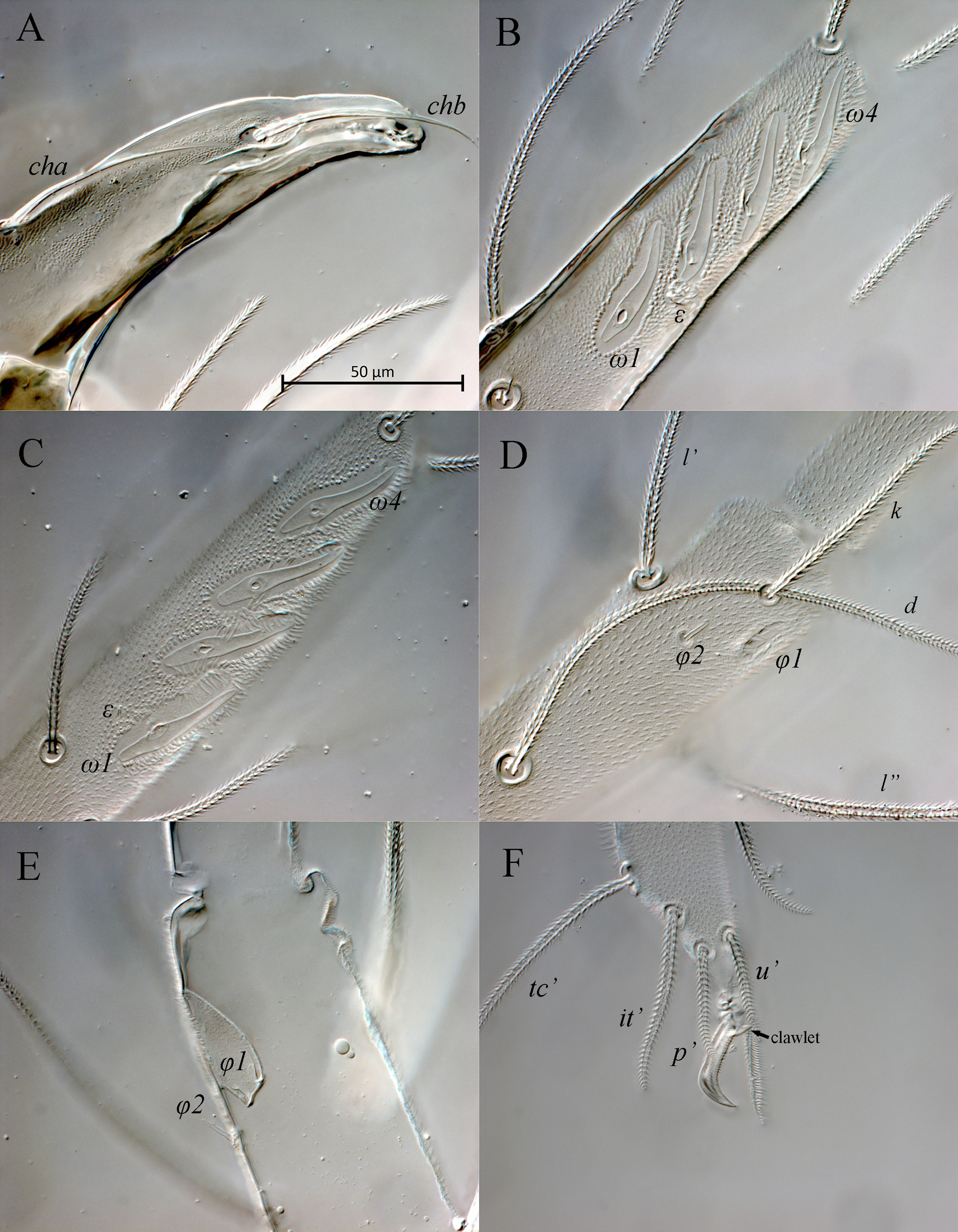


Legs (Figs 4, 5, 7B–F). Lengths of legs: leg I 1470, leg II 1050, leg III 1160, leg IV 1415. All leg setae densely pilose. Tarsal claws with large basal clawlets (Fig. 7F). Distal part of claws on tarsi II-IV weakly sclerotized, distinctly flattened and rounded in dorso-ventral aspect. Leg I (Figs 4A, B, 7B): Supracoxal setae el of the same shape and length as palpal supracoxal setae ep. Femur completely divided into basi- and telofemur. Leg setation: Tr 1 (v′), Fe 5+5 (d, l′, l″, v′, v″, d1, l′1, l″1, v″1, bv″), Ge 11(1σ) (d, l′, l″, v′, v″, d1, l′1, l″1, v′1, l′2, l″2, σ), Ti 11(2φ) (k, d, l′, l″, v′, v″, l′1, v′1, v″1, v′2, v″2, φ1, φ2), Ta 19(4ω, ε) (p′, p″, it′, it″, tc′, tc″, ft′, ft″, pl′, pl″, u′, u″, pv′, pv″, v′1, v″1, v′2, v″2, v′3, ε, ω1-ω4). Rhagidial organ I with four rhagidial solenidia arranged obliquely in separate pits; famulus ε (stellate seta) located near the base of rhagidial solenidion ω2 (Fig. 7B). Rhagidial solenidion φ1 situated in shallow depression just posterolaterad seta k; small erect solenidion φ2 located dorsodistally, posteromesad rhagidial solenidion φ1 at level of seta l′ (Fig. 7D). Small erect solenidion σ on genu located anteroventrally, almost at level of setae (l). Leg II (Figs 4C, D, 7C, E): Femur incompletely divided into basi- and telofemur. Leg setation: Tr 1 (v′), Fe 6+5 (d, l′, l″, v′, v″, d1, l′1, l″1, v′1, v″1, bv″), Ge 9(1σ) (d, l′, l″, v′, v″, l′1, l″1, v′1, l′2, σ), Ti 8(2φ) (d, l′, l″, v′, v″, l′1, l″1, v′1, φ1, φ2), Ta 16(4ω, ε) (p′, p″, it′, it″, tc′, tc″, ft′, ft″, pl′, u′, u″, pv′, pv″, v′1, v″1, v′2, ε, ω1-ω4). Rhagidial organ II with four rhagidial solenidia arranged obliquely in separate pits; spiniform famulus ε located laterad basal part of rhagidial solenidion ω1 (Fig. 7C). Dorsodistal rhagidial solenidion φ1 large, lanceolate, situated in deep depression with large dorsal opening (Fig. 7E); small erect solenidion φ2 located dorsodistally, posteriad rhagidial solenidion φ1. Small erect solenidion σ on genu located anteroventrally, almost at level of seta v′. Leg III (Figs 5A, B, 6B, 7C): Leg setation: Tr 2 (l′, v′), Fe 4+4 (d, l′, l″, v′, d1, l′1, v′1, ev′), Ge 7 (1σ) (d, l′, l″, v′, v″, l″1, v′1, σ), Ti 7 (2φ) (d, l′, l″, v′, v″, l′1, v′1, φ1, φ2), Ta 14 (p′, p″, it′, it″, tc′, tc″, ft′, u′, u″, pv′, pv″, v′1, v″1, v′2); femur completely divided into basi- and telofemur; erect solenidia φ1 and φ2 very small, located dorsomedially, distance between bases of solenidia variable (Fig. 6B); one female paratype with only one solenidion on tibia III. Small erect solenidion σ on genu located ventrodistally, slightly anteriad seta v′. Leg IV (Fig. 5C, D): Leg setation: Tr 2 (v′, v″), Fe 3+4 (d, l′, l″, v′, d1, v′1, ev′), Ge 6 (d, l′, l″, v′, v″, l″1), Ti 7 (1φ) (d, l′, l″, v′, v″, l′1, v′1, φ), Ta 14 (p′, p″, it′, it″, tc, ft′, ft″, u′, u″, pv′, pv″, v′1, v″1, v′2); femur completely divided into basi- and telofemur. Minute erect solenidion φ situated middorsally.
Male — Differs from female in having elongate sperm sac typical for rhagidiids, as illustrated for F. halltalensis Zacharda, (2000, Fig. 4A).




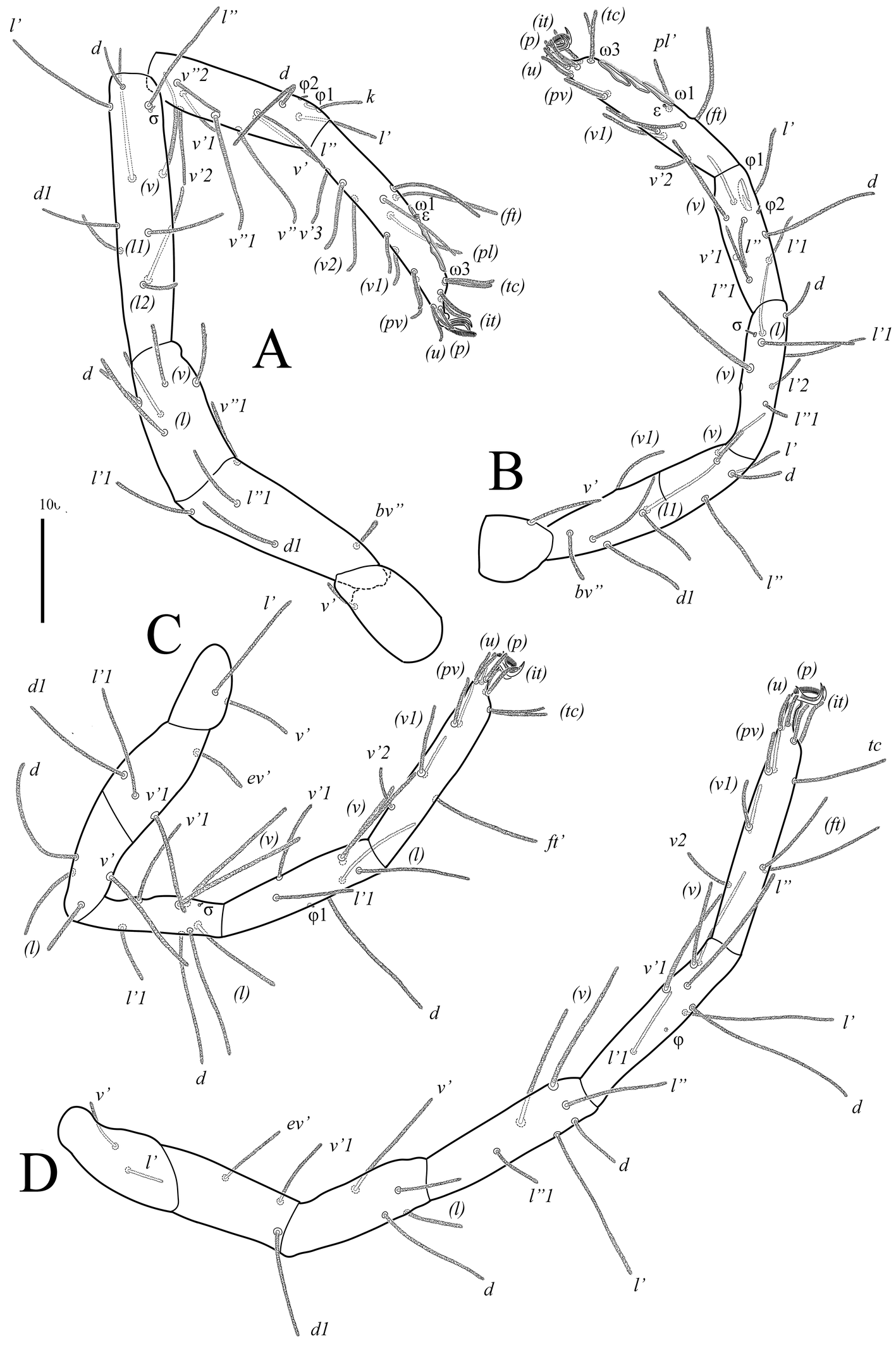


Deutonymph — (Figs 8–10). In general similar to adults with the following differences: three pairs of aggenital and two pairs of genital setae; eugenital setae absent; rhagidial organs I and II with three oblique solenidia each; famulus ε on tarsus I located near basal part of solenidion ω1; basifemur I with five setae (v′1 absent); genu I with 10 setae (v′1 absent); tibia I with 10 setae (l′1 absent); genu II with eight setae (v′1 absent); tibia III with one solenidion.
Other stages unknown.



Type material
Holotype female, slide N° T-Rhag-1, Russia, Chelyabinsk Oblast, Gremutshaya Cave, са. 56°17′ N 59°18′ E, 03 January 2024, collected by E.V. Lyubavina and L. Pshennikova. Paratypes: one female and one male, same data; one deutonymph and three females, Russia, the Republic of Bashkortostan, Shulgan-Tash Cave, ca. 53°03′ N 57°04′ E, 19 February 2018, collected by O.Ya. Chervyatsova; two males and one female, Russia, Perm Krai, Rossiyskaya Cave, ca. 58°49′ N 57°37′ E, 12 December 2020, collected by P.V. Somchenko.
Type deposition
The holotype and one paratype deutonymph, along with four female and two male paratypes are deposited in the mite collection of the Zoological Institute of RAS, St. Petersburg, Russia; one female and one male paratype are deposited in the mite collection of the Tyumen State University Museum of Zoology, Tyumen, Russia.
Etymology
The name of the new species refers to its geographical distribution, i.e., the caves of the Ural Mountains.
Differential diagnosis
The new species is placed in the genus Foveacheles as it possesses: filiform trichobothria; chelicerae that have, in lateral aspect, a distinct dorsal saddle-shaped depression, the fixed digit with a soft, sharp dorsal ridge at the base of which seta chb is inserted dorsolaterally in a shallow depression (Fig. 3B, 7A), and dorsal cha; rhagidial organs I and II with four rhagidial solenidia and, respectively, a stellate and spiniform famulus, and the epimeral formula 3-1-5-3 (Zacharda 2000).
Foveacheles comprises 10 subgenera and one species group (Zacharda 1996, 2000).
According to the key to the subgenera and their species (Zacharda 1996), the new species does not match any subgenus. In the presence of four rhagidial solenidia in each of the rhagidial organs (I and II) and erect solenidia φ2 on tibiae I and II, the new species is similar to the species from the subgenera Proxistella Zacharda, 1980, Usitorhagidia Zacharda, 1980, and Foveacheles Zacharda, 1980. It differs from the representatives of these subgenera in having the epimeral formula 3-1-5-3 (vs. 3-1-6-3 in the aforementioned subgenera). The new species differs from all subgenera in having solenidion φ2 on tibiae I and II located dorsodistally, just posteriad solenidia φ1 (vs. solenidia φ2 on tibiae I and II located dorsomedially in the other subgenera). The new species differs from the representatives of the subgenera Foveacheles, Proxistella, and Hirschmannetta in having solenidion φ1 on tibia II located in a deep depression with large dorsal opening (vs. solenidion φ1 on tibia II located in a deep depression with a small terminal pore in the aforementioned subgenera). The new species is most similar to the gigantea species group (Zacharda 2000) and the monotypic subgenus Spelaeocheles Zacharda, 1988 in having a lyrifissure-like structure on the fixed digit of the chelicera. Zacharda (2000) remarked that the chelicerae and rhagidial organs of another monotypic subgenus, Hirschmannetta Zacharda, 1980, are strikingly similar to those of the gigantea species group, but the presence of the cheliceral lyrifissure-like structure is yet to be confirmed. The new species differs from all species with a lyrifissure-like structure on the chelicerae in having solenidia φ2 on tibiae I and II located dorsodistally, just posteriad solenidia φ1 (vs. solenidia φ2 on tibiae I and II located dorsomedially in the other species).
The new species is also similar to the representatives of the genus Traegaardhia Zacharda, 1980 in general appearance (discussed further below) and ecology (all species of Traegaardhia are troglobionts). Among Traegaardhia-species, the new species is most similar to T. distosolenidia Zacharda, 2010 in having solenidia φ2 on tibiae I and II located dorsodistally. The new species differs from T. distosolenidia in having four obliquely aligned solenidia in the rhagidial organ II (vs. three solenidia arranged in tandem in T. distosolenidia).
Discussion
In the key to the genera of Rhagidiidae (Zacharda 1980), the genera Foveacheles and Traegaardhia were separated at the couplet concerning two character states: 1) the number of aggenital setae (6-7 pairs in Traegaardhia and 5 pairs in Foveacheles), and 2) the shape of the insertion pit of solenidion φ1 on tibia II (longitudinally opened in Traegaardhia and in a deep insertion pit with terminal pore in Foveacheles). However, Zacharda et al. (2010), in the revision of the genus Traegaardhia, redescribed the type species Traegaardhia dalmatina (Willmann, 1939) with solenidion φ1 on tibia II in a deep depression with a small terminal pore. Solenidion φ1 on tibia II in the other species described was either of the latter form or recessed in a deep pit with an elongated, broadly open surface pore. The above authors also described several new species with five pairs of aggenital setae. Zacharda et al. (2010) noted that the genera Foveacheles and Traegaardhia are very similar; in fact, he even moved a couple of troglobiont species from Foveacheles to Traegaardhia. According to the latest diagnosis of the genus Traegaardhia (Zacharda et al. 2010), the main differences between Traegaardhia and Foveacheles are as follows: ''Traegaardhia species have the cheliceral digits elongated and attenuated, the distal cheliceral seta is inserted laterodorsally in a weakly expressed depression (as seen in lateral aspect); in Foveacheles species the distal cheliceral seta is also inserted laterodorsally but in the distinct, well-developed, distally bounded and proximally open depression, and the cheliceral digits are usually much shorter and thick″. The length and width of the cheliceral digits is a subjective character which is difficult to apply at generic level. However, the location of the cheliceral seta chb in the new species is, undoubtedly, as in the genus Foveacheles. In view of the variability, and sometimes overlapping, of character states within each genus, the generic boundary between the two genera warrants re-examination.
Not much is known about the ecology of Foveacheles uralensis n. sp. Most often, F. uralensis specimens were found in the aphotic parts of caves, on the surfaces of microponds. Springtails (Hexapoda: Collembola) are noted among its food items (see Fig. 11).
In accordance with recent reviews of the cave biota of Russia (Turbanov et al. 2016a, b, c; Golovatch et al. 2018) the core of the cave fauna of the Urals consists of a variety of troglophile and trogloxene elements. At the same time, the fauna of the caves of the Urals is significantly depleted in specialized stygobionts and troglobionts in comparison with those of Crimea, the Caucasus, and the Far East of Russia. Thus, among stygobionts, the only species known is the amphipod Uralocrangonyx chlebnikovi Borutzky, 1928 (Amphipoda: Crangonyctidae) from caves in the Perm Region (Borutzky 1928; Marin, Palatov 2022); and among the known troglobiont springtails (Collembola), Philotella olgae Kniss and Thibaud, 1995 (Poduromorpha: Neanuridae), Ceratophysella kapoviensis (Babenko, 1994), Schaefferia baschkirica Kniss, 1985 (Poduromorpha: Hypogastruridae), Plutomurus baschkiricus (Skorikow, 1900) (Entomobryomorpha: Oncopoduridae), and Sericeotoma knissi Potapov, 1991 (Entomobryomorpha: Isotomidae) from caves in the Republic of Bashkortostan (Skorikow 1900; Kniss 1985; Potapov 1991; Babenko et al. 1994; Kniss and Thibaud 1995). Also, the presence of the troglobiont Foveacheles sp. – described in this paper as a new species – has been repeatedly indicated in the Shulgan-Tash Cave (Bashkortostan) (Abdullin et al. 2012; Kapralov 2015; Turbanov et al. 2016b; Golovatch et al. 2018; Lyakhnitsky, Chervyatsova, 2019). Among other Rhagidiidae, Rhagidia breviseta Zacharda, 1995 and Rhagidia sp. were cited as troglophiles for caves in the Ural Mountains of Bashkortostan (Kniss 1984; Kapralov 2015). However, in regards to the latter statement, the record of R. breviseta is doubtful because it was originally described from caves in the Nearctic region, and troglobiont species of Rhagidiidae are usually endemic for the region.
The reason for rather impoverished troglobiont fauna of the Urals lies in the global climatic changes associated with the glacial and interglacial periods. The above notion stems from the fact that the most likely sources of terrestrial troglobiont fauna consisted of forest litter inhabitants, as well as soil invertebrates, including those sheltering under rocks and in rock crevices. During periods of cold weather and ice sheet formation, a number of species found refuge in karst cavities where the temperature regime is more stable (a kind of refugium). Having penetrated caves and acquired specific adaptations, organisms became specialized (troglobionts, stygobionts) and began to occupy their ecological niches (Jeannel 1959, 1965; Vandel 1964; Birstein, Ljovuschkin 1967).
Recent studies in the Ural Mountain caves based on calcite dating (Dublyansky et al. 2018, 2021) have shown that in the Late Pleistocene (ca. 0.078–0.010 Ma) the growth of speleothems in caves ceased, which is associated with the spread of permafrost in northern Eurasia at this time. In our opinion, the beginning of this Late Pleistocene glaciation has provoked the ancestral terrestrial form of Foveacheles uralensis n. sp. –inhabiting the Urals in the interglacial period– to go into caves in search of stable suitable habitat conditions. This also explains the occurrence of the same species in caves over such a vast area, from the Middle to the Southern Urals – ca. 670 km from north to south (see Fig. 1). The temporally insignificant allopatric isolation of a number of cave populations seems insufficient for species divergence. However, this problem can be more reliably addressed by molecular genetic analysis.
Acknowledgements
The authors are very grateful to Elena Lyubavina and Lyudmila Pshennikova for the materials from the Gremutshaya Cave. We also thank Elena Lyubavina and Olga Ya. Chervyatsova for providing materials and photographs of the Foveacheles uralensis n. sp. from the Shulgan-Tash Cave. The authors also express gratitude to Polina V. Somchenko for providing materials from the Rossiyskaya Cave and to Violetta Karipova for helping with the logistics.
The research of Ilya S. Turbanov was conducted within the framework of the State Assignment No. 124032500016-4.
References
- Abdullin Sh.R., Kapralov L.I., Kuzmina L.I., Ryss A.Yu., Snitko V.P., Chervyatsova O.Ya., Chertoprud E.S. 2012. The biota of Shulgan-Tash (Kapovaya) Cave: A popular science essay. Shulgan-Tash Reserve, Irgizly. 25 pp [in Russian].
- Babenko A.B., Potapov M.B., Stebaeva S.K., Chernova N.M. 1994. Keys to Collembola of the fauna of Russia and adjacent countries: Family Hypogastruridae. Moscow: Nauka. Pp. 336 [in Russian].
- Baker A.S. 1990. A survey of external morphology of mites of the superfamily Eupodoidea Banks, 1894 (Acari: Acariformes). J. Nat. Hist., 24: 1227-1261. https://doi.org/10.1080/00222939000770741
- Baker A.S. 1995. A redescription of Halotydeus destructor (Tucker) (Prostigmata: Penthaleidae) with a survey of ontogenetic setal development in the superfamily Eupodoidea. Int. J. Acarol., 21: 2 61-282. https://doi.org/10.1080/01647959508684069
- Borutzky E.W. 1928. Materialien über die Fauna der unterirdischen Gewässer: Crangonyx chlebnikovi sp. n. (Amphipoda) aus den Höhlen der mittleren Urals. Zool. Anz., 77 (9-10): 253-259.
- Birstein J.A., Ljovuschkin S.I. 1967. Some results and problem in studying of the subterranean fauna of the USSR. Zool. Zh., 46 (10): 1509-1535 [in Russian, with English summary].
- Dublyansky Y., Moseley G.E., Lyakhnitsky Y., Cheng H., Lawrence Edwards R., Scholz D., Koltai G., Spötl C. 2018. Late Palaeolithic cave art and permafrost in the Southern Ural. Scient. Rep., 8 (1): 12080. https://doi.org/10.1038/s41598-018-30049-w
- Dublyansky Y., Shirokov V., Moseley G.E., Kosintsev P.A., Lawrence Edwards R., Spötl C. 2021. 230Th dating of flowstone from Ignatievskaya Cave, Russia: Age constraints of rock art and paleoclimate inferences. Geoarchaeology, 36: 532-545. https://doi.org/10.1002/gea.21851
- Golovatch S.I., Palatov D.M., Turbanov I.S., Kniss V.A., Gazaryan S., Snit′ko V.P., Decu,V., Juberthie Ch., Nazareanu G. 2018. Subterranean biota of the European part of Russia: A review. Invert. Zool., 15 (2): 153-213. https://doi.org/10.15298/invertzool.15.2.01
- Jeannel R. 1959. Situation geographique et peuplement des caverns. Ann. Spéléol., 14: 333-338.
- Jeannel R. 1965. Le genèse du peuplement des milieux souterrains. Rev. ècol. boil. sol, 2: 1-22.
- Kapralov S.A. 2015. Arthropods of caves in the European part of Russia and the Southern Urals. In: Turbanov I.S., Marin I.N., Gongalsky K.B. (Eds.), Materials of the All-Russia Youth Conference «Biospeleology of the Caucasus and other regions of Russia», Moscow, December 3-5, 2015. Severtzov Institute of Ecology and Evolution of RAS, Papanin Institute of Biology of Inland Waters of RAS & Speleological Сlub «Zelenye Kamneyedy», P. 21-23 [in Russian].
- Khaustov A.A. 2015. A new genus and species of the family Rhagidiidae (Acari: Eupodoidea) from Crimea. Int. J. Acarol., 41(7): 617-623. https://doi.org/10.1080/01647954.2015.1085899
- Kniss V.A. 1984. On the fauna of karst caves of southeastern Bashkiria. Fauna and ecology of springtails (Collembola). Moscow: Nauka. P. 123-128 [in Russian].
- Kniss V.A. 1985. Springtails of the genus Schaefferia (Collembola, Hypogastruridae) from caves of south-west Bashkiria. Zool. Zh., 64 (6): 934-937 [in Russian, with English summary].
- Kniss V.A., Thibaud J.-M. 1995. Une nouvelle espèce de Philotella des grottes d'Oural du Sud (Collembola, Neanuridae, Pseudachorutidae). Rev. Fr. Entomol., 17 (2): 49-51.
- Lindquist E.E., Zacharda M. 1987. A new genus and species of Rhagidiidae (Acari: Prostigmata) from Chihuahuan Desert litter in New Mexico. Can. J. Zool., 65: 2149-2158. https://doi.org/10.1139/z87-328
- Lyakhnitsky Y.S., Chervyatsova O.Ya. 2019. Kapova Cave (Shulgan-Tash). Atlas of Russian caves. Moscow: Russian Geographical Society, Russian Union of Speleologists. P. 421-429 [in Russian].
- Makarova O.L. 2002. Acarocenoses (Acariformes, Parasitiformes) in polar deserts: 1. Mite assemblages of the Severnaya Zemlya Archipelado: structure of fauna and abundance. Entomol. Rev., 82 (7): 839-856.
- Makarova O.L. 2015. 18.3. Acari. In: The Greenland Entomofauna. An Identification Manual of Insects, Spiders and their Allies (Böcher J. et al., eds). Leiden: Koninklijke Brill NV. P. 705-856.
- Marin I.N., Palatov D.M. 2022. Uralocrangonyx gen.n. (Amphipoda: Crangonyctidae) from the Southern Ural, Russia. Arthr. Sel., 31 (2): 183-195. https://doi.org/10.15298/arthsel.31.2.07
- Potapov M.B. 1991. Species of the genus Isotoma subgenus Parisotoma Bagnall, 1940 and Sericeotoma subgen. nov. (Collembola, Isotomidae) of USSR fauna. Acta Zool. Cracov., 34 (1): 267-301.
- Skorikow A.S. 1900. On the systematics of the genus Tomocerus (Collembola) with description of a new species from Eastern Russia. Tr. Obshch. Ispyt. Prir. Khark. Univer., 35: 73-82 [in Russian].
- Turbanov I.S., Palatov D.M., Golovatch S.I. 2016a. The present state of the art of biospeleology in Russia and the countries of the former Soviet Union: A review of the cave (endogean) invertebrate fauna. 1. Introduction - Crustacea. Zool. Zh., 95 (10): 1136-1159 [in Russian, with English summary]. https://doi.org/10.1134/S0013873816070162
- Turbanov I.S., Palatov D.M., Golovatch S.I. 2016b. The present state of the art of biospeleology in Russia and the countries of the former Soviet Union: A review of the cave (endogean) invertebrate fauna. 2. Arachnida - Acknowledgments. Zool. Zh., 95 (11): 1283-1304 [in Russian, with English summary]. https://doi.org/10.7868/S0044513416110064
- Turbanov I.S., Palatov D.M., Golovatch S.I. 2016c. The present state of the art of biospeleology in Russia and the countries of the former Soviet Union: A review of the cave (endogean) invertebrate fauna. 3. Bibliography and summary. Zool. Zh., 95 (12): 1399-1426 [in Russian, with English summary]. https://doi.org/10.7868/S0044513416120187
- Vandel A. 1964. Biospéologie. La Biologie des Animaux Cavernicoles. Paris: Gauthier-Villars. pp. 679.
- Walter D.E., Lindquist E.E., Smith I.M., Cook D.R., Krantz G.W. 2009. Order Trombidiformes In: Krantz G.W., Walter D.E. (Eds.) A Manual of Acarology. Third Edition. Texas: Texas Tech University Press, Lubbock. p. 223-420.
- Zacharda M. 1980. Soil mites of the family Rhagidiidae (Actinedida: Eupodoidea). Morphology, systematics, ecology. Acta Univ. Carol. Biol., 1978: 489-785.
- Zacharda M. 1982. The cavernicolous fauna of Hawaiian lava tubes. 13. A new subgenus and two new species of Rhagidiidae (Acari: Eupodoidea). Pacif. Ins., 24(3-4): 275-280.
- Zacharda M. 1983. The Rhagidiidae (Acarina: Prostigmata) from the central Caucasus, Siberia, and some other parts of the USSR. Věst. Čs. Společ. Zool., 47: 304-318.
- Zacharda M. 1985. New Rhagidiidae (Acarina: Prostigmata) from caves of the U.S.A. Věst. Čs. Společ. Zool., 49: 67-80.
- Zacharda M. 1987. New taxa of Rhagidiidae (Acari: Prostigmata) from Pyrenean caves. Can. J. Zool., 65(8): 2051-2056. https://doi.org/10.1139/z87-313
- Zacharda M. 1988. Foveacheles (Spelaeocheles) troglodyta sp. n., subgen. n. (Acari: Prostigmata: Rhagidiidae) from Stratenska Cave, the Western Carpathians, Slovakia. Věst. Čs. Společ. Zool., 52: 73-78.
- Zacharda M. 1996. New taxa of Rhagidiidae (Acari: Prostigmata) from North America. Part IV. Genus Foveacheles Zacharda, with a key to world species. Can. J. Zool., 74(2): 343-356. https://doi.org/10.1139/z96-042
- Zacharda M. 2000. New species of the rhagidiid genus Foveacheles (Acari: Prostigmata: Eupodoidea) with a lyrifissure-like structure on the chelicerae. J. Nat. Hist., 34: 247-265. https://doi.org/10.1080/002229300299624
- Zacharda M., Elliott W.R. 1981. New species of the family Rhagidiidae (Acarina: Actinedida: Eupodoidea) from California Caves. Acta Univ. Carol. - Biol., 30(9): 463-475.
- Zacharda M., Fong D., Hobbs III H.H., Piva E., Slay M.L., Taylor S.G. 2010. A review of the genus Traegaardhia (Acari, Prostigmata, Rhagidiidae) with descriptions of new species and a key to species. Zootaxa, 2471: 1-64. https://doi.org/10.11646/zootaxa.2474.1.1
- Zacharda M., Grafitti J., Piva E. 2011. New taxa of Rhagidia and Foveacheles (Acari: Prostigmata: Rhagidiidae) from Italian and French caves, with keys to adults of subgenera Deharvengiella and Mediostella. J. Nat. Hist., 45(11-12): 667-683. https://doi.org/10.1080/00222933.2010.535915
- Zacharda M., Pugsley C.W. 1988. Robustocheles occulta sp.n., a new troglobitic mite (Acari: Prostigmata: Rhagidiidae) from North American caves. Can. J. Zool., 66(3): 646-650. https://doi.org/10.1139/z88-096
- Zhang Z.-Q., Fan Q.-H., Pesic V., Smit H., Bochkov A.V., Khaustov A.A., Baker A., Wohltmann A., Wen T.-H., Amrine J.W., Beron P., Lin J.-Z., Gabrys G., Husband R. 2011. Order Trombidiformes Reuter, 1909. In: Zhang, Z.-Q. (Ed.), Animal biodiversity: an outline of higher-level classification and survey of taxonomic richness. Zootaxa, 3148: 129-138. https://doi.org/10.11646/zootaxa.3148.1.24



2024-04-30
Date accepted:
2024-05-31
Date published:
2024-06-03
Edited by:
Faraji, Farid

This work is licensed under a Creative Commons Attribution 4.0 International License
2024 Khaustov, Alexander A.; Kolesnikov, Vasiliy B.; Turbanov, Ilya S. and Tolstikov, Andrei V.
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



