Species of Phytoseiidae (Acari: Mesostigmata) predators of thrips and whiteflies: a review
Barbar, Ziad  1
; Skinner, Margaret
1
; Skinner, Margaret  2
; Parker, Bruce L.
2
; Parker, Bruce L.  3
and Kreiter, Serge
3
and Kreiter, Serge  4
4
1✉ Department of Plant Protection, Faculty of Agriculture, Al-Baath University, P.O. Box 77, Al- Sham St., Homs, Syria.
2Entomology Research Laboratory, University of Vermont, 661 Spear Street, Burlington, Vermont 05405-0105, USA.
3Entomology Research Laboratory, University of Vermont, 661 Spear Street, Burlington, Vermont 05405-0105, USA.
4CBGP, Montpellier SupAgro, INRAE, CIRAD, IRD, Univ Montpellier, Montpellier, France.
2024 - Volume: 64 Issue: 3 pages: 745-767
https://doi.org/10.24349/alf8-2ujoOriginal research
Keywords
Abstract
Introduction
Families Aleyrodidae (Hemiptera) and Thripidae (Thysanoptera) constitute two major groups of insects with piercing-sucking mouthparts. They have an economic importance and worldwide distribution (Nakahara 1994; Evans 2007). In the first group, the tobacco whitefly, Bemisia tabaci (Gennadius) and the greenhouse whitefly Trialeurodes vaporariorum (Westwood), are among the most serious pests. They infest more than 800 plant species in the greenhouse and the field. They infest bean, pepper, eggplant, cucumber, tomato and a large number of ornamentals, including species of chrysanthemum, fuchsia, gerbera, poinsettia and rose (Evans 2007; Lee and Zhang 2018; CABI 2023). Their impacts on host plants are significant causing major economic damage. They have also shown a high level of resistance to several insecticides and can act as vectors of plant viruses (Navas-Castillo et al. 2011; Reliy et al. 2011).
Several species in the second group include Frankliniella bispinosa (Morgan), Frankliniella occidentalis (Pergande), Frankliniella schultzei Trybom, Heliothrips haemorrhoidalis (Bouché), Scirtothrips dorsalis Hood, Thrips flavidulus (Bagnall), Thrips palmi Karny, and Thrips tabaci Lindeman. All are among the most important pests of crops and ornamentals throughout the world. They cause direct damage through feeding and sucking out the contents of plant cells (flowers, young foliage, and buds) and indirect damage as vectors of destructive plant viruses in greenhouse and field crops. The most damaging species are F. occidentalis, T. tabaci, T. palmi and S. dorsalis (Nakahara 1994; Jenser and Szenasi 2004; Seal et al. 2006; Cannon et al. 2007; Reitz 2009).
The biological control of whiteflies and thrips is a successful and sustainable approach within IPM (Integrated Pest Management) as it is considered as a permenant and durable method and help to prevent the development of insecticide resistance. IPM provides long-term management by regulating thrips and whitefly populations over time and it is an alternative to insecticides. It reduces the need for repeated pesticide applications that can have negative impacts on non-target organisms, the environment and workers and consumers (Cock et al. 2010; Van Lenteren et al. 2017). With biological control, many parasitoids and predators can be efficient and regulate pest populations. Evans (2007) reported in his catalog more than 50 Aphelinidae (Hymenoptera) parasitoid species of B. tabaci and most of them belong to the genera Encarsia and Eretmocerus (39 and 14 species, respectively). He also reported more than 300 species of insect predators of the families Coccinellidae and Miridae. Aphelinidae, Coccinellidae and Miridae are natural enemies of T. vaporariorum with about 50 total species in these families (Evans 2007). Among these natural enemies, the aphelinid parasitoids Encarsia formosa Gahan, Eretmocerus eremicus (Rose & Zolnerowich), Eretmocerus mundus Mercet and the mirid predators Macrolophus pygmaeus Rambur and Nesidiocoris tenuis (Reuter) are widely used worldwide in the augmentative biological control of whiteflies (Van Lenteren 2012; Gullino et al. 2020).
Regarding predatory insects used as biological control agents of thrips, several species such as Franklinothrips vespiformis (Crawford) (Aeolothripidae), Scolothrips sexmaculatus (Pergande) (Thripidae), the generalist predator Mallada signata (Schneider) (Neuroptera, Chrysopidae), and many species of the genus Orius, such as Orius insidiosus (Say) and Orius laevigatus (Fieber) (Hemiptera, Anthocoridae) are important (Van Lenteren 2012; Gullino et al. 2020).
Many predacious mite species of the family Phytoseiidae (Mesostigmata) can also feed and reproduce on whiteflies and thrips (Gerson et al. 2003; McMurtry et al. 2013). Currently, this family contains about 2,557 valid species (Demite et al. 2024). The well-known species were classified according to their feeding habits and habitats, biological traits, and morphological characteristics in four major types (and several sub-types) of predators (specialized mite predators, selective predators of tetranychid mites, generalist predators, and polliniphagous generalist predators) (McMurtry et al. 2013). Most species of Phytoseiidae are generalist predators (Type III) that feed and reproduce on a wide range of prey including phytophagous mites, thrips, whiteflies, as well as pollen and fungi (McMurtry and Croft 1997; McMurtry et al. 2013). Despite the high number of described species of this family, less than thirty species are practically involved as biological control agents, of which only eleven are used to control whiteflies and/or thrips (Van Lenteren et al. 2017). This could be explained by the little knowledge of their biological features, ability to predation and feeding habits, and lifestyles (McMurtry et al. 2013; Tixier et al. 2020). However, more research has been done since 2000 on additional predacious mites and their potential to control these pests, especially thrips.
This review aims to present data on the phytoseiid species studied under laboratory conditions and in greenhouse crops to manage whiteflies and thrips, to provide information about their efficacy and limitations, and to present future perspectives for their use.
Literature was electronically collected from the Google Scholar internet browser, CABI Abstracts, Science Direct sources, and Semantic Scholar AI research tool, using the search terms ′Phytoseiidae′, ′phytoseiid', or phytoseiid species name in combination with ′whitefly-whiteflies or whitefly species name′, and ′thrips or thrips species name′. We focused on the references published after 2000. According to data from the literature, phytoseiid species were divided into three groups: (i) species predate whiteflies and/or thrips but prefer phytophagous mites, (ii) species used as biological control agents of whiteflies and thrips, and (iii) species need further research for exploration as biological control agents of whiteflies and thrips. They were tabulated according to the pests on which they were tested (whitefly and/or thrips). The names of phytoseiid genera and species in this review follow those used in the taxonomic system summarized in Chant and McMurtry (2007) and in the Phytoseiidae database (Demite et al. 2024), in which other species names and synonyms can be consulted.
Early and current research on Phytoseiidae predators of whiteflies and thrips
From 1960 to about the beginning of 2000, moderate numbers of research (~150) were done and dealt with the biology, ecology and potential use of Phytoseiidae as natural enemies of whiteflies and thrips. The bibliography of Phytoseiidae by Kostiainen and Hoy (1996), showed that Amblyseius swirskii Athias-Henriot, Euseius scutalis (Athias-Henriot) and Phytoseiulus persimilis Athias-Henriot, were the first species tested for their potential as predators of B. tabaci and T. vaporariorum (Teich 1966; Vogel 1969). Amblyseius swirskii and Neoseiulus cucumeris (Oudemans) were also the first two phytoseiid species tested as predators of T. tabaci and F. occidentalis, respectively (Vartapetov and Vasilev 1971; Castagnoli et al. 1990). Later, other phytoseiid predators were examined and involved in the control of further species of whiteflies and thrips (as Heliothrips sylvanus Faure, Scirtothrips aurantii Faure, Scirtothrips citri (Moulton), Scolothrips longicornis Priesner, S. dorsalis, Taeniothrips simplex (Morison), Thrips palmi, and Parabemisia myricae (Kuwana)). Only the three phytoseiid mites, Iphiseius degenerans (Berlese), N. cucumeris and Neoseiulus barkeri Hughes have demonstrated good performances as biological control agents and became commercially available before 2000 to manage thrips in Europe, North America and other areas (Van Lenteren 2012; Knapp et al. 2013; Van Lenteren et al. 2017).
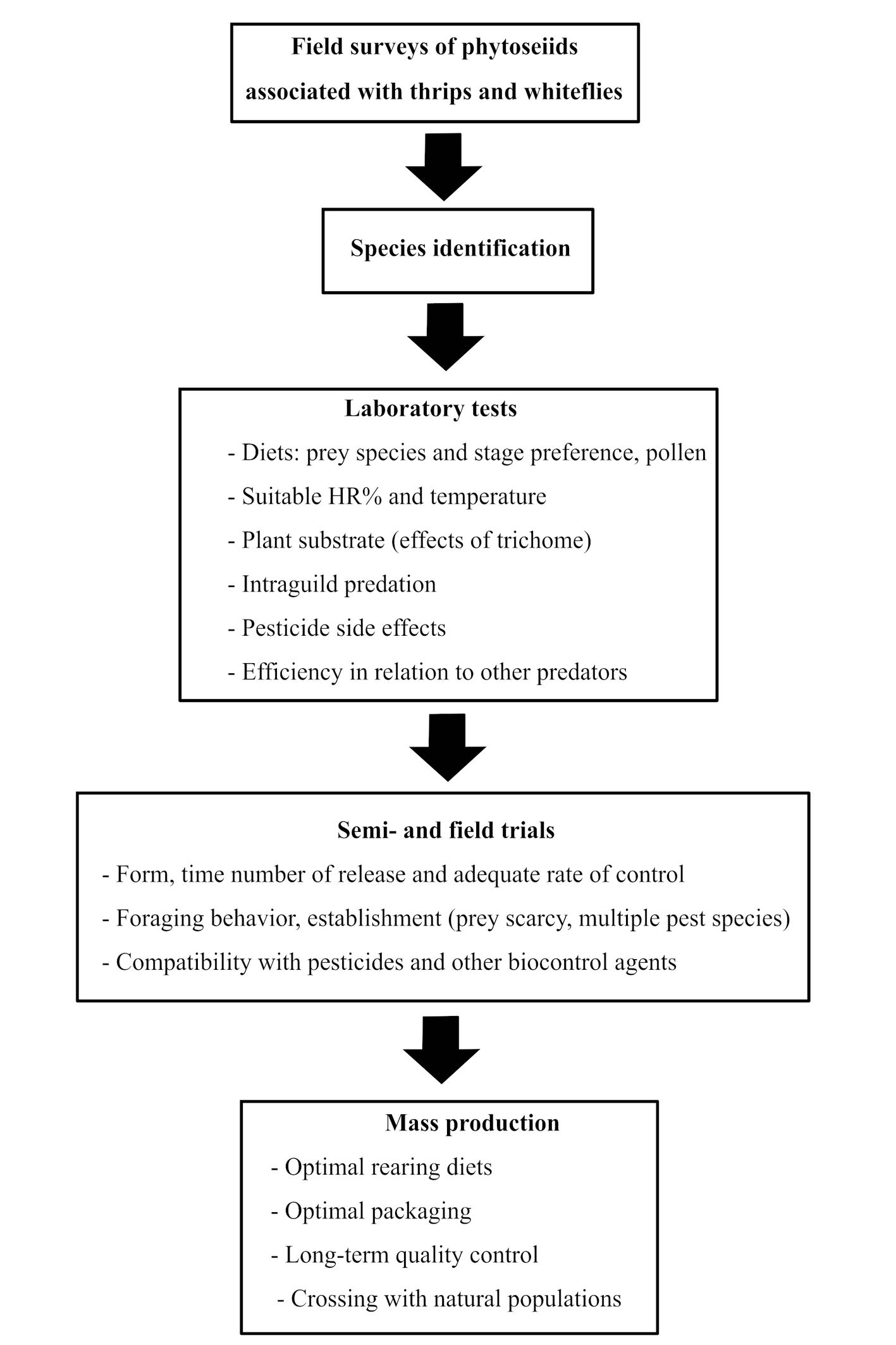


After 2000 and to date, a significant increase in research on the biological control of whiteflies and thrips by Phytoseiidae occurred. The ability of predation and factors affecting the performance of 40 phytoseiid species were examined/re-examined on several whiteflies and thrips taxa under laboratory, semi-field conditions and/or in greenhouses of several crops or ornamental plants like cucumber, sweet pepper, eggplant, bean, tomatoes, roses, chrysanthemum, etc. (Supplementary Table 1, Figure 1). The most targeted pests were F. occidentalis (by 23 tested phytoseiid species) B. tabaci (by 18 species), T. vaporariorum (12 species), T. tabaci (by 11 species), and T. palmi (by 10 species), and S. dorsalis (by six species).
The number of phytoseiid species evaluated against these pests mostly belong to the subfamily Amblyseiinae in particular to three main genera: Euseius (nine species), Amblyseius and Neoseiulus (eight species of each). However, only three species of the subfamily Typhlodrominae [Typhlodromus (Typhlodromus) athiasae Porath and Swirski, Typhlodromus (Typhlodromus) pyri Scheuten and Cydnoseius negevi (Swirski & Amitai)] and one of Phytoseiinae (Phytoseius finitimus Ribaga) have been the subject of studies mentioned above. Most studies involving phytoseiid species were either pollen-feeding generalist predators (type IV) of Euseius and Iphiseius genera, for which pollen constitute an important part of their diet, or generalist predators (type III) living in various habitats [on pubescent leaves (III-a) like Typhlodromalus aripo De Leon and T. pyri, on glabrous leaves (III-b) like A. swirskii and Transeius montdorensis (Schicha), in confined space on dicotyledonous plants (III-c) as Amblyseius herbicolus (Chant), and predators from soil/litter habitats (III-e) like N. barkeri and N. cucumeris] (McMurtry et al. 2013).
Phytoseiid species predate whiteflies and/or thrips but prefer phytophagous mites
This group of mites includes eleven phytoseiid species that are specialized on the Tetranychus genus, in the Tetranychidae or prefer to feed on members of other phytophagous mite families such as Eriophyidae, Tarsonemidae and/or Tenuipalpidae. They are alphabetically presented.
Amblyseius andersoni (Chant) was described was described based on the specimens collected from prune (Rosaceae) from Canada. It is a generalist predator living on glabrous leaves (type III-b) and widely distributed particularly in Europe (Demite et al. 2024). It has been reared on a commercial scale since 1995 and utilized to control several mite species such as the two-spotted spider mite Tetranychus urticae Koch, the apple rust mite Aculus schlechtendali (Nalepa), the European red mite Panonychus ulmi (Koch), the western flower thrips F. occidentalis and the tomato russet mite Aculops lycopersici (Massee) (Knapp et al. 2018). It was commercially available to control mites in Europe, North America, and Asia (Van Lenteren 2012). It was used in conservation and augmentation biological control and its releases were suggested as a preventive measure to slow the increase of T. urticae (McMurtry and Croft 1997; Van Lenteren 2012). However, the results of Sengonca and Dreischer (2001) showed its best performance was on T. tabaci as prey with regard to developmental time, longevity and predation compared with its use on T. urticae control.
Amblyseius orientalis (Ehara) was described based on the specimens collected from Quercus crispula Blume (Fagaceae) from Japan. It has been recorded from China, Hawaii, India, Japan, Russia and South Korea (Demite et al. 2024) and has been applied in orange and apple orchards for biological control of spider mites (Zhang et al. 2015). This predator was also able to complete its development on B. tabaci eggs. However, developmental duration was longer and fecundity was lower when reared on T. urticae protonymphs, or a mix of both preys. Thus, A. orientalis could be considered as a potential biological control agent of B. tabaci, when it co-occurs with T. urticae (Zhang et al. 2015). It has been mass-produced and Wei et al. (2023) showed that rearing on Carpoglyphus lactis (L.) (Acari: Carpoglyphidae) deutonymphs resulted in lower fecundity than when reared on eggs. This can significantly improve A. orientalis production on a large scale.
Euseius finlandicus (Oudemans) was described based on the specimens collected from Salix caprea L. (Salicaceae) from Finland. It is a pollen feeding generalist predator (type IV) widely distributed in Europe and Asia (Demite et al. 2024). It is used in conservation biological control of mites on deciduous fruits and can be important in control of P. ulmi. It has been commercially available to control mites in Europe since 2000 (Van Lenteren 2012). Euseius finlandicus was more effective in controlling spider mite in cucumber greenhouses and can predate on F. occidentalis and T. vaporariorum. It did not establish better than other predacious species such as N. cucumeris (Messelink et al. 2006; Sarwar et al. 2011).
Neoseiulus californicus (McGregor) was described based on the specimens collected from lemon (Rutaceae) from USA. It has been reported from more than 30 countries particularly in Americas and Europe (Demite et al. 2024). It is a selective predator of tetranychid mites (type II) and particularly associated with species that produce heavy webbing (McMurtry et al. 2013). It is commercially available to control spider mites in Europe, Africa North and South, North and Latin America, and Asia since 1985 (Van Lenteren 2012). First instars of F. occidentalis are aggressively attacked and provide enough nutrients for egg production of N. californicus but its predation rates were low compared with the specialized thrips predator N. cucumeris (Walzer et al. 2004). It can also predate and oviposit on the first instar T. tabaci larvae but juvenile mortalities were very high (only about 16% of newly hatched larvae reached maturity) and seems to be less advantageous than I. degenerans for biological control of this pest (Rahmani et al. 2009, 2016).
Neoseiulus fallacis (Garman) was described based on the specimens collected from apple (Rosaceae) from USA. It has been reported from 18 countries in Americas, Europe, Africa and Australia (Demite et al. 2024). It is a selective predator of tetranychid mites (type II) and was reported to feed on a wide range of mite pests of deciduous fruit orchards and ornamentals (McMurtry et al. 2013). It has been commercially available to control mites in Europe and North America since 1997 (Van Lenteren 2012). Studies showed that females consumed and deposited eggs on T. tabaci nymphs (Abdel-Karim and Abd EL-Wareth 2012) and on first instars of F. occidentalis and T. vaporariorum. Survival and fecundity were low compared to T. urticae (Pratt et al. 1999).
Neoseiulus womersleyi (Schicha) was described based on the specimens collected from strawberry (Rosaceae) from Australia. It has been reported from China, Japan, South Korea and Taiwan (Demite et al. 2024). It is a selective predator of tetranychid mites (type II). It has been commercially available in Asia to control mites since 2005 (Van Lenteren 2012). It was the most efficient predator in controlling F. occidentalis and T. vaporariorum populations on greenhouse cucumber compared with Euseius castaneae (Wang & Xu), Euseius utilis (Liang & Ke) and E. finlandicus (Sarwar et al. 2011).
Phytoseiulus persimilis Athias-Henriot was described based on the specimens collected from Rosa spp. (Rosaceae) from Algeria. It has been reported from more than 40 countries, especially in Europe, northern Africa and Middle East (Demite et al. 2024). It is one of the most comprehensively studied species and commercially used for the control of Tetranychus mites, especially T. urticae (specialized of Tetranychus genus; type I-a). Females of this predator did not attack F. occidentalis larvae and cannot produce eggs. These larvae can be an alternative prey for its immatures, enabling them to develop to adulthood but developmental times prolonged and mortality increased as compared to spider mite prey (Walzer et al. 2004).
Phytoseius finitimus Ribaga was described based on the specimens collected from Buddleja madagascariensis Lamarck (Buddlejaceae) from Italy. It has been reported many times from the Mediterranean region (Demite et al. 2024). This generalist predator lives on plants with pubescent leaves (type III-a) usually found in association with eriophyid and tetranychid mites. It has been commercially available in Europe to control mites since 1990 (Van Lenteren 2012). It has a low impact on whitefly populations (no or very low oviposition on B. tabaci of many tested strains) (Nomikou et al. 2001). Its predation on spider mite eggs and larvae, and on T. vaporariorum crawlers was higher than this on F. occidentalis larvae. The mixed diets of pollen with each of those preys reduced consumption but increased the predator's egg production (Pappas et al. 2013).
Typhlodromalus aripo De Leon was described based on the specimens collected from Solanum stramoniifolium Jacquin (Solanaceae) from Trinidad. It has been reported from many countries in South America and Africa (Demite et al. 2024). It is a generalist predator living on pubescent leaves (type III-a). It can develop and reproduce on B. tabaci crawlers but consumption and fecundity were lower than when feeding on the cassava green mite Mononychellus progresivus Doreste. It is sensitive to extreme low humidity and temperatures compared to other phytoseiids (the optimal conditions for survival were 75% RH and 27 °C) (Mutisya et al. 2014).
Typhlodromus (Typhlodromus) pyri Scheuten was described based on the specimens collected from Pyrus communis L. (Rosaceae) from Germany and widely distributed in Europe and the Middle East (Demite et al. 2024). It is a generalist predator living on pubescent leaves (type III-a).It is known to feed on mites, eggs and larvae of insects, on pollen, fungal spores and hyphae, honeydew and plant saps. It seems to prefer P. ulmi (McMurtry and Croft 1997; Zemek and Prenerova 1997). It has been commercially available in Europe to control mites since 1990 (Van Lenteren 2012). Its ability to control F. occidentalis was studied in cucumber greenhouses but it did not establish better than N. cucumeris (Messelink et al. 2006).
Typhlodromus (Typhlodromus) athiasae Porath and Swirski was described based on the specimens collected from Citrus sp. (Rutaceae) from Israel and has been reported from Azerbaijan, Cyprus, Egypt, France, Greece, Iran, Jordan, Lebanon, Syria, and Turkey (Demite et al. 2024). It has been commercially available in Europe to control mites since 1995 (Van Lenteren 2012). This generalist predator has a significantly lower peak oviposition rate on B. tabaci and pollen than other species as A. swirskii and E. scutalis (Athias-Henriot) (Nomikou et al. 2001).
Phytoseiid species used as biological control agents of whiteflies and thrips
In total, eleven phytoseiid species are biological control agents and commercially available for whitefly and thrips management (Van Lenteren 2012; Van Lenteren et al. 2017).
These species are N. barkeri, N. cucumeris, I. degenerans, E. scutalis, T. montdorensis, A. swirskii, Euseius ovalis (Evans), Amblydromalus limonicus (Garman and McGregor), Euseius amissibilis Meshkov, Gynaeseius liturivorus (Ehara) and Amblyseius tamatavensis Blommers. They are discussed according to their date of commercialization.
Neoseiulus barkeri Hughes based on the specimens collected from germinating barley from England. It has a worldwide distribution (Demite et al. 2024). It is a generalist predator (type III-e) collected mainly from soil or litter habitats and can predate on several phytophagous mite species, thrips and whiteflies (El-Banhawy et al. 2001; Nomikou et al. 2001; McMurtry et al. 2013). It has commercially been used for the biological control of thrips since 1981 (Van Lenteren 2012). Under laboratory conditions, high rates of consumption and fecundity of this predator were observed on the 1st instar T. tabaci (Jafari et al. 2013) and T. flavidulus (Yao et al. 2014) but they were lower on the 1st instar T. palmi than those of N. cucumeris (Komi et al. 2008). In greenhouses with cucumber, the release of N. barkeri at the density of 250 mites/m2 reduced the population of T. tabaci and F. occidentalis by 76% and 41% in six weeks, respectively (Wu et al. 2014). However, it did not establish better than N. cucumeris (Messelink et al. 2006).
Neoseiulus cucumeris (Oudemans) was described based on the specimens collected from Cucumis melo L. (Cucurbitaceae) from France. It is a generalist predator (type III-e) collected mainly from soil or litter habitats and has a wide distribution especially in Europe (Demite et al. 2024). It can predate on many pests including mites, whiteflies and thrips (McMurtry et al. 2013; Li et al. 2017). It has been used commercially for the biological control of mites and thrips since 1985 in Europe, Africa North and South, North and Latin America, Asia, Australia and New Zealand (Van Lenteren 2012). It is now become one of the top four phytoseiid mites used as biological control agents (Knapp et al. 2018). This predator was released in many greenhouse crops and ornamentals. In greenhouses with cucumber, placement of N. cucumeris sachets on plants immediately after planting entirely suppressed populations of F. occidentalis and the damage was reduced (Jacobson et al. 2001). In greenhouse tomatoes, the curatively inundative introductions of this predator at the rate of one sachet (1000 predatory mites per sachet) per plant significantly reduced F. occidentalis populations to low levels (Shipp and Wang 2003). In cyclamen, at the 200 and 350 mites/m2 released rates of N. cucumeris resulted in a rapid decline of F. occidentalis populations (De Courcy Williams 2001). When comparing the efficiency of N. cucumeris with A. limonicus, A. swirskii and E. ovalis, much higher population levels and better control of F. occidentalis in greenhouses cucumber was obtained by these species (Messelink et al. 2006). Moreover, N. cucumeris was found to perform optimally at relatively higher F. occidentalis densities contrarily to A. swirskii which was found to perform better at lower densities and can be released preventively when the crop is flowering and remains present in the crop throughout the entire growing season (Bolckmanns et al. 2005; Dalir et al. 2021). The same results were obtained in greenhouse with pepper in which, both N. cucumeris and A. swirskii were established and reduced numbers of chili thrips S. dorsalis following a single release (30 mites/plant). However, A. swirskii was a more effective predator compared with N. cucumeris (Arthurs et al. 2009). Other biotic and abiotic factors can affect the performance of this predator such as prey species and stages, duration of light, intraguild predation, and application of pesticides, etc. For example, its daily fecundity was slightly higher (2.6 eggs/female/day) on 1st instar of T. flavidulus (Yao et al. 2014) than on 1st instar of T. palmi (2.2 eggs/female/day) (Komi et al. 2008). Furthermore, this predator was not effective for controlling of T. palmi and F. schultzei on blooms of cucumber in the field (Kakkar et al. 2016). Zilahi-Balogh et al. (2007) demonstrated that the use supplemental lighting in greenhouses in northern temperate zones would increase its oviposition and thus improve the biological control of F. occidentalis. Combinations of N. cucumeris with other natural enemies or pesticides would assure sufficiently high control levels of thrips. For example, the introduction of ten N. cucumeris adults per plant (Phaseolus vulgaris L.) and applications of 200 infective juveniles/cm2 of entomopathogenic nematodes (Heterorhabditis bacteriophora Poinar or H. indica Poinar, Karunakar, & David) significantly reduced F. occidentalis populations compared with individual applications of each natural enemy (Ebssa et al. 2006). The same results were obtained with the combination of NeemAzal-U (17% azadirachtin), a formulation developed for root uptake, with N. cucumeris and the soil dwelling mites Hypoaspis aculeifer Canestrini (Acari, Laelapidae) (Thoming and Poehling 2006). Ahmed and Lou (2018) demonstrated that combined use of N. cucumeris and A. swirskii was responsible for the highest positive effect in controlling F. occidentalis on cherry tomato grown compared with the use of each of them alone. However, previous studies (Madadi et al. 2009; Buitenhuis et al. 2010) showed a significant reduction in number of this predator in the presence of minute pirate bugs, Orius albidipennis (Reuter) when used to control T. tabaci. Negative interactions (intraguild predation) between N. cucumeris and A. swirskii also occurred when used together in biological control of F. occidentalis. Side effects of some pesticides used in many agrosystems on N. cucumeris were also tested. For example, the use of Spinosad was not toxic to motile stages of this predator two hours after treatment but female oviposition was reduced by 76% after three days, which indicates that the use of this compound may not be compatible with releases of this predacious mite for controlling of F. occidentalis (Van Dreische et al. 2006). Relatively high mortality of N. cucumeris was observed following exposure to both direct applications and dry residues of Spinosad (Conserve®) and abamectin (Dynamec®) but good survival was recorded for thiacloprid (Calypso®), pymetrozine (Chess®), and dry residues of imazalil (Fungaflor®) (Cuthbertson et al. 2012). In contrast, no effects on this predacious mite were observed when Spinosad was used. The release of this predator in combination with T. montdorensis and Hypoaspis miles (Berlese) (Acari, Laelapidae) together with Spinosad to control F. occidentalis on strawberry was more effective than single species releases (Rahmani et al. 2011).
Iphiseius degenerans (Berlese) was described based on the specimens collected from herb and moss from Italy and has been reported from more than 35 countries particularly in Africa and the Middle East (Demite et al. 2024). It is important to note that the genus Iphiseius does not seem a valid one as its single species is included within the Euseius clade based on a molecular work by Vicente and Tixier (2018). It is a pollen feeding generalist predator (type IV). It was commercialized for thrips control in 1993 (Van Lenteren 2012). This species was reported to feed and oviposit on pollen, T. urticae, larvae of T. tabaci, T. palmi, F. occidentalis or eggs of Ephestia kuehniella (Zeller) (Vantornhout et al. 2005; Cuthbertson et al. 2012). It was unable to develop beyond the protonymph stage when it fed on T. tabaci (Sengonca and Dreischer 2001). Although it is adapted to the Mediterranean region and effectively controls thrips in greenhouses in Northern Europe, its mass production is difficult (Bolckmans et al. 2005). Its establishment was more successful than N. cucumeris on sweet pepper for controlling F. occidentalis but not on cucumber plants (Bolckmans et al. 2005; Messelink et al. 2006). In greenhouses, the releases of I. degenerans in combination with the anthocorid predator, O. insidiosus (a commercial natural enemy of F. occidentalis) did not enhance thrips control on roses (Chow et al. 2008). The application of the insecticide Teflubenzuron (of the benzoyl-phenyl urea group) could be considered harmless on the survival of I. degenerans adults on some plants despite its effectiveness to control F. occidentalis and H. haemorrhoidalis was reduced (Scott Brown et al. 2003). However, the use of spinosad (an insecticide based on chemical compounds found in the bacterial species Saccharopolyspora spinosa Mertz and Yao) may not be compatible with releases of I. degenerans to control F. occidentalis due to its negative effects on its survival and fecundity (Van Dreische et al. 2006; Cuthbertson et al. 2012). Morevere, the insecticides acetamiprid, metaflumizonem, spinetoram and thiamethoxam should be used with caution in greenhouses where I. degenerans was introduced as they highly decreased the fecundity of this predator compared with othe insecticides such as lubendiamide, chlorantraniliprole andmethoxyfenozide (Döker et al. 2015).
Euseius scutalis (Athias-Henriot) was described based on the specimens collected from Ceratonia siliqua L. (Fabacae) from Algeria. It has been recorded from many countries in North Africa, Middle-East and Southern Europe (Demite et al. 2024). It is a pollen feeding generalist predator (type IV) (McMurtry et al. 2013) and frequently found in humid climates and seems to be tolerant to hot and dry ones (30 °C was the most favourable temperature for the population increase) (Bounfour and McMurtry 1987; Kasap and Şekerğlu 2004). It is an important predator of tetranychid mites, scale insects and became a commercial biocontrol agent of whiteflies in 2001 (Nomikou et al. 2001; Gerson et al. 2003; Abdelgayed et al. 2020; Zergani et al. 2023). This predator has a high oviposition rate on many plant with pollen (i.e. cattail, alfalfa, broad bean, castor bean and date palm) which could be a suitable alternative food source for their survival and egg reproduction (Nomikou et al. 2001; Nomikou et al. 2003b; Al-Shemmary, 2011; Fouly et al. 2013). Oviposition on B. tabaci immatures was equal to that of A. swirskii and they were able to suppress whitefly populations on cucumber in a greenhouse (Nomikou et al. 2001). However, an unequal distribution of this predator on cucumber plants (more abundant on the lowest leaves) resulted in insufficient controlling of F. occidentalis (abundant on the upper leaves) (Messelink et al. 2006).
Transeius montdorensis (Schicha) was described based on the specimens collected from Datura sp. (Solanaceae) from New Caledonia and was recorded from Australia, Fidji, Tahiti and Vanuatu (Demite et al. 2024). It is a generalist predator found on plants with glabrous leaves (type III-b). This species became commercially available in Europe in 2004 for controlling thrips, whiteflies and tarsonemid mites (Van Lenteren et al. 2017). It is a good predator of F. schultzei, F. occidentalis and T. palmi in strawberry, cucumber, and pepper greenhouses (Steiner et al. 2003; Rahmani et al. 2011; Cuthbertson et al. 2012; Labbé et al. 2019; Téllez et al. 2020). It is an effective control agent of B. tabaci and T. vaporariorum in poinsettia and cucumber in greenhouses and exhibited higher populations than A. swirskii, specifically when climatic conditions were cold. Therefore, its releasing in winter, followed with releases of A. swirskii in spring, may be a good control strategy for greenhouse grown cucumbers (Richter 2017; Téllez et al. 2020).
Amblyseius swirskii Athias-Henriot was described based on the specimens collected from Prunus dulcis (Miller) Webb (Rosaceae) from Israel and found in many countries of the Mediterranean, African subtropical and tropical areas on wild and cultivated plants having glabrous leaves (generalist predator of type III-b), (Zannou and Hanna 2011; McMurtry et al. 2013; Döker et al. 2020; Zahidi et al. 2023). More than 100 papers have been published on biology, behaviour, predation and feeding habits of this species under laboratory, semi-field or field conditions of various crops such as cucumber, sweet pepper, eggplant, bean, tomatoes, and ornamental plants chrysanthemum, roses and poinsettia. Calvo et al. (2015) listed 36 pest species on which A. swirskii has been reported to develop and/or oviposit in laboratory experiments: 18 phytophagous mite species, five thrips species, two species of whiteflies, and the remaining taxa of other insect families. The results of these various research proved the value of A. swirskii that became a successful biological control agent since 2006 and it has been used in augmentative biological control of thrips and whiteflies in more than 50 countries. Furthermore, factors such as feeding on various pests and controlling them simultaneously, its capacity to multiply on pollen in the absence or scarcity of pests and its easy and cheap production on stored product mites are thought to be the main reasons for its worldwide use (Calvo et al. 2015; Knapp et al. 2018). More recently, A. swirskii demonstrated its effectiveness on T. palmi, in commercial open fields of cucumber (Kakkar et al. 2016) and significantly reduced poinsettia thrips (Echinothrips americanus Morgan) densities in greenhouse sweet pepper particularly when cattail pollen was also applied (Ghasemzadeh et al. 2017). However, it was revealed that A. swirskii preferred T. urticae rather than B. tabaci and T. vaporariorum (Heydari et al. 2016; Asadi et al. 2019; Hosseininia et al. 2020). Additional studies showed the impact of several factors on this predacious mite. The acarid mite, C. lactis is the best prey for mass production of this predator (Hosseininia et al. 2020). Furthermore, using living mites of Thyreophagus entomophagus (Laboulbène) as a food source greatly increased its density compared with the use of cattail pollen, a commonly used supplemental food, supporting biological control of thrips and other pests in greenhouse chrysanthemum (Pirayeshfar et al. 2021). The provision of Pinus brutia Tenore pollen also did not result in an effective management of F. occidentalis despite significant increase of its population in greenhouse eggplants and pepper. Thus, thought to be the presence of trichomes on eggplant leaves or the pine pollen was an unsuitable food source for this predator (Kütük 2017, 2018). Combination of A. swirskii with other natural enemies were also examined. Under laboratory conditions, A. swirskii was a stronger competitor compared to A. orientalis or N. californicus even in the presence of B. tabaci eggs (an extra-guild prey) (Guo et al. 2016). On the contrary, it has a higher oviposition rate and a faster development time on an extra-guild prey (B. tabaci eggs) than on intra-guild prey (eggs, larvae and protonymphs of T. negevi) (Momen et al. 2013). In greenhouse conditions, A. swirskii can interfere with the phytoseiid Neoseiulus longispinosus (Evans) and distrupt biological control of T. urticae on papaya plants (Döker et al. 2021). The release of A. swirskii and P. persimilis were highly effective in suppressing B. tabaci, F. occidentalis, and T. urticae populations on cucumber and more effective than many insecticidal and acaricidal sprays in the control greenhouse (Abou-Haidar et al. 2021). The combinations of predatory mirids N. tenuis, M. pygmaeus or predatory anthocorid O. laevigatus with A. swirskii significantly reduced F. occidentalis and B. tabaci populations on sweet pepper (Bouagga et al. 2018). Using the parasitoid E. eremicus and A. swirskii was a feasible strategy for management of B. tabaci on poinsettia and reduced insecticide applications by 25–78% (Vafaie et al. 2021). The application of the entomopathogenic fungus Lecanicillium muscarium R. Zare & W. Gams conidia followed by the release of A. swirskii suppressed T. vaporariorum on roses and was more effective than an application of the control agents separately (Mitina et al. 2021).
Euseius ovalis (Evans) was described based on the specimens collected from rubber plant from Malaysia. It is a pollen feeding generalist predator (type IV) widely distributed in eastern countries of Asia and was also found in Australia, New Zealand, and Mexico. It was very often confused with Euseius ovaloides (Blommers) (Liao et al. 2017). Euseius ovalis typically lives on plants with smooth leaves. (Liao et al. 2017; Demite et al. 2024). They became commercially available in Europe in 2008 and has good potential for the biological control of F. occidentalis and T. vaporariorum in greenhouse cucumbers (Messelink et al. 2006; Van Lenteren et al. 2017). It was also found to be the most effective control agent of T. vaporariorum in greenhouse roses compared to other phytoseiid species such as A. swirskii, A. limonicus, N. californicus and N. cucumeris (Pijnakker et al. 2007). It is also a better predator of the poinsettia thrips, E. americanus pupae on sweet pepper compared to A. swirskii, A. limonicus, and E. amissibilis (Ghasemzadeh et al. 2017).
Amblydromalus limonicus (Garman and McGregor) was described based on the specimens collected from Citrus sp. (Rutaceae) from USA. It has been reported in many countries in Central and South America, Australia, New Zealand, and several European countries including Italy, Portugal, Spain, and Slovenia and Turkey (Tsolakis and Ragusa 2020; Bas et al. 2022; Demite et al. 2024;). It is a generalist predator living on plants with glabrous leaves (type III-b) (McMurtry et al. 2013) and became commercially available in 2012 (Knapp et al. 2013). It is a better predator of F. occidentalis in cucumber greenhouses compared to other species (Messelink et al. 2006) and its biology and predation on many pests (spider mites, T. tabaci, F. occidentalis, B. tabaci and T. vaporariorum) were reviewed by Knapp et al. (2013). Laboratory, semi-field and field trials demonstrated its excellent performance as a biological control agent of thrips and whiteflies in cucumber, gerbera, poinsettia and roses. High predation and reproduction rates were observed on 1st and 2nd instar E. americanus under laboratory conditions (Ghasemzadeh et al. 2017). It has also high potential to control outbreaks of F. occidentalis (Vangansbeke et al. 2014). The addition of Typha angustifolia L. pollen in mixed diets of this pest increases its populations and enhances their biological control capabilities even in the presence of low thrips densities (Samaras et al. 2021). This predator is also a promising candidate for the biological control of the invasive thrips S. dorsalis and may be as effective as A. swirskii in the field (Schoeller et al. 2020). However, its reproductive performance when with fed T. vaporariorum was significantly lower than when fed on F. occidentalis (Vangansbeke et al. 2014). Similar results were obtained in greenhouse cucumber. It was not a suitable candidate for treatment of high populations of T. vaporariorum despite its high level of predation (compared to T. montdorensis and A. swirskii) (Medd and GreatRex 2014). In contrast, releasing A. limonicus on poinsettia proved to be a good addition or alternative to the endoparasitism by the wasp E. formosa to control T. vaporariorum (as it is less susceptible to low temperature during autumn) but slightly less efficient for B. tabaci (Richter 2017).
Euseius amissibilis Meshkov was described based on the specimens collected from Platanus orientalis L. (Platanaceae) from Tajikistan. It was also found in Iran. This species is actually the senior synonym of Euseius gallicus Kreiter and Tixier (Döker et al. 2024) which has been recorded from France (Tixier et al. 2009), Belgium, Germany, Italy, Mauritius, the Netherlands, Slovenia, Tunisia and Turkey (Demite et al. 2024). It is a biocontrol agent of whiteflies and thrips and has been commercially available in Europe since 2013 (Van Houten et al. 2016; Van Lenteren et al. 2017). It has been shown to be a promising control agent of F. occidentalis in cut roses (Pijnakker et al. 2014). Laboratory and semi-field experiments demonstrated that the predator is a suitable biocontrol agent for T. vaporariorum on roses, but not for F. occidentalis (Van Houten et al. 2016), nor for E. americanus on sweet pepper plants even at high densities in the presence of Typha latifolia L. pollen as a supplementary food source (Ghasemzadeh et al. 2017).
Gynaeseius liturivorus (Ehara) was described based on the specimens collected from soybean (Fabaceae) from southern Japan. It has been recorded from Philippines, Sri Lanka, Taiwan and Vietnam (Demites et al. 2024). It is a generalist predator (type-III) (Kreiter et al. 2020). It predates on the eggs and larvae of B. tabaci and T. vaporariorum. It was considered of interest for control of S. dorsalis, F. occidentalis and T. palmi, on which the daily consumption average was 7.4-19.4 1st instar of these preys and laid an average of 1.6-6.8 eggs within 24-48 hours (Mochizuki, 2009). It became a commercially available biocontrol agent of whiteflies and thrips in Asia in 2013 (Van Lenteren et al. 2017).
Amblyseius tamatavensis Blommers was described based on the specimens collected from Citrus hystrix de Candolle (Rutaceae) from Madagascar. This generalist predator (type III) has been reported from over 40 countries in central and South America, Africa, eastern Asia and Australia (Demite et al. 2024). Its females can consume eight B. tabaci eggs daily and has 3.5 times higher oviposition rate than those when fed on T. urticae (Cavalcante et al. 2015a). However, Massaro and Moraes (2019) observed differences in these parameters among different populations of this species and those having larger dorsal and ventrianal shields has the highest rates of daily predation and oviposition. Variations in predation of this pest were also observed according to leaf characteristics of host plants: higher on cotton, potato and bell pepper and lower on tomato and melon (Cavalcante et al. 2017; Barbosa et al. 2019). The acarid mite, Aleuroglyphus ovatus (Troupeau) (Acaridae), seems to be a suitable host for mass production of this predator (Cavalcante et al. 2015a). Amblyseius tamatavensis has been marketed in Brazil as a control agent of B. tabaci (releasing 15 adults per plant, suggests the high potential of this species to control B. tabaci in pepper plants) (Souza and Marucci, 2021). It was also observed to predate on the ficus whitefly Singhiella simplex (Singh) and on F. occidentalis under laboratory conditions (Döker et al. 2018; Jorge et al. 2021).
Phytoseiid species that need further research for exploration as biological control agents of whiteflies and thrips
Eighteen species were studied in this group and many of them showed potential for biological control of thrips and whiteflies.
Amblydromalus lailae (Schicha) was only found on papaw (Caricaceae) in Australia. It is a generalist predator living on glabrous leaves (type III-b) (McMurtry et al. 2013; Demite et al. 2024). It can feed on the broad mite, Polyphagotarsonemus latus (Banks) (Tarsonemidae) and tomato russet mite Aculops lycopersici (Massee) (Eriophyidae) in protected crops. It had an average of seven and two larvae consumption of the first and the second instar of F. schultzei per day, respectively, and thus it is a candidate for the biological control of this pest (Steiner et al. 2003). It is important to note that this species may be the junior synonym of A. limonicus according to Steiner and Goodwin (2005), Minor (2008), and Ma et al. (2018).
Amblyseiella setosa Muma was described based on the specimens collected from orange (Rutaceae) in USA. It was also recorded from Brazil, Peru, and many European and northern African countries (Demite et al. 2024). Very few studies have dealt with the biology of A. setosa. Muma (1971) showed that the tetranychid mite Eutetranychus banksi (McGregor) is an adequate food source of this predator but other species such as Panonychus citri (McGregor) and Eotetranychus sexmaculatus (Riley) were considered only as survival food. Momen et al. (2004) demonstrated the A. setosa had faster development and higher fecundity when fed on the eriophyid mites compared to its feeding on diets of Phoenix dactylifera L. pollen, the scale insect Parlatoria ziziphus (Lucas) (Hemiptera, Diaspididae) and B. tabaci.
Amblyseius eharai Amitai and Swirski was described based on the specimens collected from Dimocarpus longan (Loureiro) (Sapindaceae) from Hong Kong. It was also recorded from China, Georgia, Japan, Malaysia, South Korea, Taiwan and Thailand (Demite et al. 2024). It is a generalist predator living on glabrous leaves (type III-b). Contrarily to A. swirskii, this species is susceptible to low humidity (most of its eggs would not hatch at relative humidities lower than 60%) but they have a relatively low optimal temperature for population growth (Ji et al. 2013; Park et al. 2021). A study conducted by Komi et al. (2008) showed that this predator had higher prey consumption rate on T. palmi than N. cucumeris and was a promising candidate for the biological control of T. palmi in greenhouse vegetable. Side effects of > 50 insecticides, acaricides and fungicides were tested on this predator [also on Amblyseius tsugawai Ehara and Euseius sojaensis (Ehara)] and their effects ranged from harmless to harmful to the survival and fecundity of adult females and immatures (Kishimoto et al. 2018, 2020).
Amblyseius herbicolus (Chant) was described based on the specimens collected from an unidentified Bromeliaceae from USA. It is widely distributed in the Americas and was also recorded from many African, European and Asian countries (Demite et al. 2024). It is a generalist predator living in confined spaces on dicotyledonous plants (type III-c) (McMurtry et al. 2013). Laboratory evaluation of this species as candidate for the control of B. tabaci has showed high levels of egg predation and oviposition (Cavalcante et al. 2015a). High oviposition rates and short developmental durations were also observed when its immatures and females fed on mixed diets of T. vaporariorum eggs and cattail pollen (Typha orientalis C. Presl). This latter seemed suitable as an alternative or additional food source with high nutritional value (Xin and Zhang, 2021). Finally, Oulenziella bakeri (Hughes) (Winterschmidtiidae), A. ovatus and almond pollen could be potential alternative foods for mass rearing programs (Cavalcante et al. 2015a; Hou et al. 2022).
Amblyseius largoensis (Muma) was described based on the specimens collected from Key lime (Rutaceae) from USA. It is widely distributed in Americas, in many African, Middle East, eastern Asian countries and Australia (Demite et al. 2024). It is a type III generalist predator. Under laboratory conditions, predation levels on eggs of B. tabaci were higher than A. limonicus (Cavalcante et al. 2015a) and even than A. swirskii (Fouly et al. 2011). The acarid mite A. ovatus is suitable for its mass production (Cavalcante et al. 2015a). This predator could also be effective for the controlling Aleurodicus cocois (Curtis) (Aleurodidae), a serious pest of cashew, Anacardium occidentale L. especially in low level of pest populations (Alfaia et al. 2018).
Amblyseius tsugawai Ehara was described based on the specimens collected from apple (Rosaceae) in Japan. This generalist predator (type III) has been reported from China and South Korea (Demite et al. 2024). It is better adapted to herbaceous plants (Yang et al. 2019). Although the consumption and oviposition rates of this species on 1st instar of T. palmi were higher than those of N. barkeri, E. sojaensis and Proprioseiopsis nemotoi (Ehara and Amano), these parameters were lower compared to other predators such as G. liturivorus and A. eharai (Komi et al. 2008). Evaluation of this predator against B. tabaci showed its preference for eggs rather than 1er instar but the reproduction was very low suggesting low chance for population establishment (Yang et al. 2019).
Cydnoseius negevi (Swirski and Amitai) was described based on the specimens collected from P. dactylifera from Israel. This generalist predator (type III) has been reported from Egypt, Morocco, Oman, Pakistan, Saudi Arabia, Sudan, and United Arab Emirates (Demite et al. 2024). Date palm pollen was a suitable food source for their mass production and an alternate food source in the field under arid environmental conditions (Alatawi et al. 2018). Similarly, Fouly et al. (2021) showed that eggs of T. urticae and pollen of date palm were the most favourable food for the development and reproduction of this predator compared with eggs of Spodoptera littoralis (Boisduval), Trialeurodes ricini (Misra), and E. kuehniella. A laboratory study by Momen et al. (2009) showed that the total egg reproduction of this predator was the highest on Ricinus communis L. pollen and eggs of B. tabaci compared to other hosts such as the maskell scale Insulaspis pallidula (Green) (Hemiptera: Diaspididae) and the red date scale Phoenicoccus marlatti Cockerell (Hemiptera: Phoenicoccidae) eggs. Female developmental time and longevity were shorter and fecundity was higher when this predator fed on an eriophid A. lycopersici rather than on B. tabaci. Its release reduced B. tabaci populations by 63-84% in open eggplants fields (Elkholy et al. 2022). Releasing this predator in combination with blue sticky traps reduced F. occidentalis about 45% in greenhouse pepper (Sanad and Hassan 2019). In addition to the effects of feeding source, other factor seemed to affect this predator. Momen et al. (2013) showed a high predation rate by A. swirskii on T. negevi eggs (intra-guild prey). It was higher than on an extra-guild prey B. tabaci eggs. Negative effects of the entomopathogenic fungi, Metarhizium anisopliae (Metchnikoff), Beauveria bassiana (Balsamo), Paceliomyces fumosoroseus (Vassiljevsky) were observed on the mobile stages of this predator. Paceliomyces fumosoroseus had the highest rate of reduction in egg production followed by B. bassiana and M. anisoplia (Saad et al. 2021).
Euseius castaneae (Wang and Xu) was described based on the specimens collected from Castanea mollissima Blume (Fagaceae) from China (Demite et al. 2024). Its release to control F. occidentalis and T. vaporariorum in a greenhouse of cucumber showed a reduction in the populations of these pests, but they were unable to develop successfully under greenhouse conditions (Sarwar et al. 2011).
Euseius concordis Chant was described based on the specimens collected from Citrus sp. (Rutacae) from Argentina and has been reported especially from Central and South America, USA, Montenegro, and Portugal (Demite et al. 2024). Several studies showed its ability to feed on pollen and several species of phytophagous mites. This indicates its use as a biological control agent of pests (De Figueiredo et al. 2018; Lopes et al. 2018; Silveira et al. 2020; Dameda et al. 2021). As A. largoensis, this predator could be effective in controlling the whitefly A. cocois, which attacks cashew, when its populations are low (Alfaia et al. 2018).
Euseius nicholsi (Ehara and Lee) was described based on the specimens collected from grass (Poaceae) from Hong Kong. It has been reported from China, Taiwan, and Thailand (Demite et al. 2024). It has been used for reducing T. urticae populations on various crops (Zhang et al. 2018). This species can predate T. flavidulus. The female laid the maximum number of eggs (2.6) at a density of 25 first-instar thrips. The predation and successful attack rates increased with increasing temperatures up to 26 °C and attack rates were reduced afterward (Yao et al. 2014).
Euseius sojaensis (Ehara) was first described based on the specimens collected from mulberry (Moraceae) from Japan. It is a type IV predator and has been reported from Taiwan (Demite et al. 2024) and considered as an effective indigenous natural enemy of some eriophyid mites and spider mites in Japan (Kishimoto 2014; Tsuchida and Masui 2021). This predator affects the population density of S. dorsalis in vineyard and can consume 5.4 larvae/female/day of this pest (Shibao et al. 2004) and its effectiveness was higher on the less hairy grape cultivar ′Pione′ than on the hairy one ′Shine Muscat′ (Tsuchida and Masui 2023). The consumption and oviposition rates of this species on 1st instar of T. palmi were lower than those of A. tsugawai and N. cucumeris (Komi et al. 2008).
Euseius utilis (Liang and Ke) was described based on the specimens collected from Malus domestica Borkhausen (Rosaceae) from China (Demite et al. 2024). As for the predator E. castaneae, releasing of E. utilis to control F. occidentalis and T. vaporariorum species in greenhouse cucumber showed a reduction in the populations of these pests compared to not releasing them in cucumber, but it seems unable to develop successfully under greenhouse conditions (Sarwar et al. 2011).
Neoseiulus bicaudus (Wainstein) was described based on the specimens collected from grass (Poaceae) from Kazakhstan. It has been reported from 30 countries particularly in the Mediterranean and Middle of Asia (Demite et al. 2024). It can feed on spider mite (Li et al. 2015; Zhang et al. 2016) and prefers the 1st instar T. tabaci to adults of the tetranychid mite Tetranychus turkestani Ugarov & Nikolski (Zhang et al. 2017). Releasing this predator in combination with blue sticky traps reduced F. occidentalis population about 57% in greenhouse pepper (Sanad and Hassan 2019).
Neoseiulus tunus (De Leon) was described based on the specimens collected from Psidium guajava L. (Myrtaceae) from Trinidad. It has been reported from Argentina, Brazil, Colombia, Guadeloupe, Jamaica, Marie-Galante, Martinique, and Peru (Demite et al. 2024). High levels of predation on B. tabaci eggs were observed (Cavalcante et al. 2015a). It is promising for management of B. tabaci. The acarid mite A. ovatus seemed was a suitable host for the mass production of N. tunus (Cavalcante et al. 2015a). It was synonymized with N. neotunus (Denmark and Muma) by Kreiter et al. (2018).
Neoseiulus umbraticus (Chant) was described based on the specimens collected from Rubus fruticosus L. (Rosaceae) from England. It has been widely reported from Europe. It was also found in Iran, Morocco, Jamaica and USA (Demite et al. 2024). It was often associated with T. tabaci, P. latus and T. urticae on bean fields (Kazak et al. 2002). It can predate on T. tabaci larvae. However, its predation and oviposition period on this pest were lower thanthose on T. urticae (Sengonca and Dreischer 2001).
Proprioseiopsis lenis (Corpuz & Rimando) was described based on the specimens collected from Citrus reticulata Blanco (Rutaceae) from Philippines. It has been reported from Thailand, Vietnam and Australia (Demite et al. 2024). The results of Nguyen et al. (2019) suggested that P. lenis may have a potential as a natural enemy and could be used in augmentative biological control of spider mites and thrips due to its high oviposition rates on F. occidentalis and T. urticae, and its ability to be reared on the storage mite C. lactis, and its high ovipostion rate on T. latifolia pollen.
Proprioseiopsis mexicanus (Garman) was first described based on the specimens collected from Zinnia sp. (Asteraceae) from USA. This generalist predator (type III) has been reported from Australia, Benin, Brazil, Colombia, Cuba, Ghana, Guadeloupe, Hawaii, Kenya, Martinique, New Zealand, and other countries (Demite et al. 2024). It is a generalist predator and can develop on tarsonemid and spider mites but may require pollen for survival and reproduction (Farfan et al. 2021a, b). This species has the characteristics of successful predator of F. occidentalis and its optimum development time at 35° C was above the temperature optimum of most phytoseiid mites (Emmert et al. 2008). In the same way, this predator could be a helpful biological control agent for T. tabaci and the results indicate its capability to develop and reproduce at a broad range of temperatures, especially above 25° C (Huang et al. 2014).
Proprioseiopsis nemotoi (Ehara and Amano) was described based on the specimens collected from pear (Rosaceae) from Japan. It has been reported from South Korea and classified as a generalist predator from soil/litter habitats (type III-e) (Farfan and Schmidt-Jeffris 2019; Demite et al. 2024). It can predate on T. palmi larvae, but the prey consumption and oviposition rates were lower than those of other species such as N. cucumeris and A. tsugawai (Komi et al. 2008).
Conclusions and future research
A total of 40 phytoseiid species have been shown their ability to predate, develop and reproduce on one or several species of thrips and whiteflies. Among them, 11 species are specialists and prefer feeding on Tetranychidae or other phytophagous mite family members (Eriophyidae, Tarsonemidae or Tenuipalpidae). Eleven species have shown good performance on thrips and whiteflies, and have already been commercially available as biological control agents, most of them on greenhouse crops. They all belong to the subfamily Amblyseiinae and are generalist predators most are found on plants with glabrous leaves, from soil/litter habitats, or are pollen-feeding predators. Amblyseius swirskii was the most studied and used for biological control of at least six thrips and two whiteflies species in greenhouses with bean, cucumber, sweet pepper, eggplant, tomatoes, chrysanthemum, roses and poinsettia. Neoseiulus cucumeris, was mainly used for controlling many thrips species. Amblydromalus limonicus was used for both thrips and whiteflies. The 18 remaining species mostly belong to the subfamily Amblyseiinae and nine of them originated from the oriental biogeographical zone. Amblyseius eharai, P. lenis and P. mexicanus were promising candidates for the biological control of thrips. However, additional studies (i.e. effects of abiotic factors, predation capacity, and ability to mass production) are needed to confirm their performance and potential establishment in the field. Similarly, A. herbicolus, A. largoensis, E. concordis and N. tunus could be promising control agents of whitefly species. However, evaluation of their biological traits, prey species preference and their performances under semi-field and field conditions should essentially be examined and. Few data are available about the performance of the last 11 remaining species as predators of thrips and whiteflies. It is difficult to confirm their effectiveness since extensive future research (evaluation of life history traits, prey preference and alternative food, computability with pesticides, effects of abiotic factors, effects of host plant traits, intraguild predation, development of mass production) is necessary.
Future should be oriented toward surveying indigenous communities of phytoseiids not only in natural ecosystems but also in open-field agroecosystems. In addition, evaluation of the feasibility of their incorporation with exotic species should also be considered in integrated pest management programs. Examining of other species of Amblyseius, Euseius, Neoseiulus, and Proprioseiopsis genera that constitute about 50% of phytoseiids are also essential. More than 140 new phytoseiid species have been described in the last decade. Consideration should also be taken to the sub-genus Typhlodromus (Anthoseius) which contains approximately 400 species but nearly none of them have been examined against thrips and whiteflies. Finally, despite valuable research on phytoseiid biological control agents in some regions of the world such as China, Japan, occidental Europe and Brazil, it seems to be insufficient in other regions. Efforts and financial support should be available for training new taxonomists in collaboration with researchers and experts in systematic biology. The development of new technologies for phytoseiid mass production would increase their widespread use.
acarologia_4721_Supplementary-table-1.pdf
References
- Abdel-Karim H., Abd EL-Wareth, H. M. 2012. Biological aspects of the predatory mite, Amblyseius fallacis Garman (Phytoseiidae) feeding on thrips nymphs under laboratory condition. Egypt. Acad. J. Biol. Sci., 5 (2):197-204. https://doi.org/10.21608/eajbsa.2012.14831
- Abdelgayed A.S., Abd El-Wahed N.M., Ali A.M., Eraky S.A. 2020. Food preference, predation efficiency and life table parameters of Euseius scutalis (Acari: Phytoseiidae) reared on Tenuipalpus punicae (Acari: Tenuipalpidae) and Siphoninus phillyreae (Hemiptera: Aleyrodidae) under constant conditions. SVU-Int. J. Agric. Sci., 2 (2): 428-437. https://doi.org/10.21608/svuijas.2020.45393.1043
- Abou-Haidar A., Tawidian P., Sobh H., Skinner M., Parker B., and Abou-Jawdah Y. 2021. Efficacy of Phytoseiulus persimilis and Amblyseius swirskii for integrated pest management for greenhouse cucumbers under Mediterranean environmental conditions. Can. Entomol., 153(5), 598-615. https://doi.org/10.4039/tce.2021.15
- Ahmed N., Lou M., 2018. Efficacy of two predatory phytoseiid mites in controlling the western flower thrips, Frankliniella occidentalis (Pergande) (Thysanoptera: Thripidae) on cherry tomato grown in a hydroponic system. Egypt. J. Biol. Pest Control, 28 (15). https://doi.org/10.1186/s41938-017-0001-4
- Alatawi F.J., Basahiha J.S., Kamrana M. 2018. Suitability of date palm pollen as an alternative food source for the predatory mite Cydnoseius negevi (Swirski & Amitai) (Acari: Phytoseiidae) at a low relative humidity. Acarologia, 58(2): 357-365. https://doi.org/10.24349/acarologia/20184247
- Alfaia J.P. de, Melo L.L., Monteiro N.V., Lima D.B., Melo J.W.S. 2018. Functional response of the predaceous mites Amblyseius largoensis and Euseius concordis when feeding on eggs of the cashew tree giant whitefly Aleurodicus cocois. Syst. Appl. Acarol., 23(8): 1559-1566. https://doi.org/10.11158/saa.23.8.6
- Ali F.S., Zaher M.A. 2007. Effect of Food and Temperature on The Biology of Typhlodrompis swirskii (Athias-Henriot) (Acari: Phytoseiidae). ACARINES: J. Egypt. Soc. Acarol., 1(1): 17-21. https://doi.org/10.21608/ajesa.2007.4986
- Al-Shemmery K.A. 2011. Plant pollen as an alternative food source for rearing Euseius scutalis (Acari: Phytoseiidae). J. Entomol., 8(4): 365-374. https://doi.org/10.3923/je.2011.365.374
- Arévalo H.A., Fraulo A.B. Liburd O.E. 2009. Management of flower thrips in blueberries in Florida. Fla. Entomol., 92(1):14-17. https://doi.org/10.1653/024.092.0103
- Arthurs S., McKenzie C.L., Chen J., Dogramaci M., Brennan M., Houben K., Osborne L. 2009. Evaluation of Neoseiulus cucumeris and Amblyseius swirskii (Acari: Phytoseiidae) as biological control agents of chilli thrips, Scirtothrips dorsalis (Thysanoptera: Thripidae) on pepper. Biol. Control, 49(1):91-96. https://doi.org/10.1016/j.biocontrol.2009.01.002
- Asadi P., Sedaratian-Jahromi A., Ghane-Jahromi M. Haghani M. 2019. How Spiromesifen affects some biological parameters and switching behavior of predatory mite Amblyseius swirskii (Acari: Phytoseiidae) when feeding on different ratios of mixed preys. Persian J. Acarol., 8(3): 239-251.
- Barbosa M.F.C., Poletti M., Poletti E.C. 2019. Functional response of Amblyseius tamatavensis Blommers (Mesostigmata: Phytoseiidae) to eggs of Bemisia tabaci (Gennadius) (Hemiptera: Aleyrodidae) on five host plants. Biol. Control, 138. https://doi.org/10.1016/j.biocontrol.2019.104030
- Bas H., Döker İ., Ozman-Sullivan S. K. 2022. New records and complementary descriptions of three Phytoseiidae (Acari: Mesostigmata) species from Turkey. Int. J. Acarol., 48(4-5): 393-400. https://doi.org/10.1080/01647954.2022.2082527
- Bolckmans K., van Houten Y. Hoogerbrugge H. 2005. Biological control of whiteflies and western flower thrips in greenhouse sweet peppers with the phytoseiid predator mite Amblyseius swirskii (Athias-Henriot). Second International Symposium on Biological Control Arthropods, 555-565.
- Bouagga S., Urbaneja A., Pérez-Hedo M. 2018. Combined use of predatory mirids with Amblyseius swirskii (Acari: Phytoseiidae) to enhance pest management in sweet pepper. J. Econ. Entomol., 20(10):1-9. https://doi.org/10.1093/jee/toy072
- Bounfour M., McMurtry J.A. 1987. Biology and ecology of Euseius scutalis (Athias-Henriot) (Acarina: Phytoseiidae). Hilgardia, 55(5): 1-23. https://doi.org/10.3733/hilg.v55n05p023
- Buitenhuis R., Shipp L., Scott-Dupree C. 2010. Intraguild vs extra-guild prey: effect on predator fitness and preference of Amblyseius swirskii (Athias-Henriot) and Neoseiulus cucumeris (Oudemans) (Acari: Phytoseiidae). Bull. Entomol. Res., 100(2): 167-173. https://doi.org/10.1017/S0007485309006944
- Buitenhuis R., Shipp L., Scott-Dupree C., Brommit A., Lee W. 2014. Host plant effects on the behavior and performance of Amblyseius swirskii (Acari: Phytoseiidae). Exp. Appl. Acarol., 62(2): 171-180. https://doi.org/10.1007/s10493-013-9735-1
- CABI 2023. CABI Compendium. Wallingford, UK: CAB International. https://www.cabidigitallibrary.org/doi/10.1079/cabicompendium.8927
- Calvo F.J., Knapp M., van Houten Y.M., Hoogerbrugge H., Belda J.E. 2015. Amblyseius swirskii: What made this predatory mite such a successful biocontrol agent? Exp. Appl. Acarol., 65(4): 419-433. https://doi.org/10.1007/s10493-014-9873-0
- Calvo F.J., Bolckmans K., Belda J.E. 2011. Control of Bemisia tabaci and Frankliniella occidentalis in cucumber by Amblyseius swirskii. BioControl, 56(2):185-192. https://doi.org/10.1007/s10526-010-9319-5
- Calvo F.J., Bolckmans K., Belda J.E. 2012. Biological control-based IPM in sweet pepper greenhouses using Amblyseius swirskii (Acari: Phytoseiidae). Biocontrol Sci. Tech., 22(12):1398-1416. https://doi.org/10.1080/09583157.2012.731494
- Cannon R.J.C., Matthews L., Collins D.W. 2007. Review of the pest status and control options for Thrips palmi. Crop Protection, 26(8):1089-1098. doi.org/10.1016/j.cropro.2006.10.023 https://doi.org/10.1016/j.cropro.2006.10.023
- Castagnoli M., del Bene G., Gargani E., Simoni S. 1990. Possibilities of the biological control of Thrips tabaci Lind. and Frankliniella occidentalis (Pergande) (Thys. Thripidae) using Amblyseius cucumeris (Oud.) (Acarina Phytoseiidae)] [in Italian]. Redia 73(1): 53-61.
- Cavalcante A.C.C., Santos V.L.V.D., Rossi L.C., Moraes G.J.D., 2015a. Potential of five Brazilian populations of Phytoseiidae (Acari) for the biological control of Bemisia tabaci (Insecta: Hemiptera). J. Econ. Entomol., 108(1): 29-33. https://doi.org/10.1093/jee/tou003
- Cavalcante A.C.C., Borges L.R., Lourenção A.L., Moraes G.J.D. 2015b. Potential of two populations of Amblyseius swirskii (Acari: Phytoseiidae) for the control of Bemisia tabaci biotype B (Hemiptera: Aleyrodidae) in Brazil. Exp. Appl. Acarol., 67(4): 523-533. https://doi.org/10.1007/s10493-015-9964-6
- Cavalcante A.C.C., Mandro M.E.A., Paes M.R., Moraes G.J.D., 2017. Amblyseius tamatavensis Blommers (Acari: Phytoseiidae) a candidate for biological control of Bemisia tabaci (Gennadius) biotype B (Hemiptera: Aleyrodidae) in Brazil. Inter. J. Acarol., 43(1): 10-15, https://doi.org/10.1080/01647954.2016.1225816
- Chant D.A., McMurtry J.A. 2007. Illustrated keys and diagnoses for the genera and subgenera of the Phytoseiidae of the world (Acari: Mesostigmata). Indira Publishing House, West Bloomfield, Mi, USA, pp. 220.
- Chow A., Chau A., Heinz K.M. 2008. Compatibility of Orius insidiosus (Hemiptera: Anthocoridae) with Amblyseius degenerans (Acari: Phytoseiidae) for control of Frankliniella occidentalis (Thysanoptera: Thripidae) on cut roses. Biol. Control, 44(2): 259-270. https://doi.org/10.1016/j.biocontrol.2007.11.002
- Chow A., Chau A., Heinz K.M. 2010. Compatibility of Amblyseius (Typhlodromips) swirskii (Athias-Henriot) (Acari: Phytoseiidae) and Orius insidiosus (Hemiptera: Anthocoridae) for biological control of Frankliniella occidentalis (Thysanoptera: Thripidae) on roses. Biol. Control, 53(2): 188-196. https://doi.org/10.1016/j.biocontrol.2009.12.008
- Cock M.J.W., van Lenteren JC, Brodeur J., Barratt B.I.P., Bigler F., Bolckmans K., Consoli F.I., Haas F., Mason P.G., Parra J.R.P. 2010. Do new access and benefit sharing procedures under the convention on biological diversity threaten the future of biological control? BioControl, 55(2):199-218. https://doi.org/10.1007/s10526-009-9234-9
- Colomer I., Aguado P., Medina P., Heredia R.M., Fereres A., Beldad J.E., Viňuela E. 2011. Field trial measuring the compatibility of methoxyfenozide and flonicamid with Orius laevigatus Fieber (Hemiptera: Anthocoridae) and Amblyseius swirskii (Athias-Henriot) (Acari: Phytoseiidae) in a commercial pepper greenhouse. Pest. Manag. Sci., 67(10): 1237-1244. https://doi.org/10.1002/ps.2173
- Cuthbertson A.G.S., Mathers J.J., Croft P., Nattriss N., Blackburn L.F., Luo W., Northing P., Murai T., Jacobson R.J., Walters K.F.A. 2012. Prey consumption rates and compatibility with pesticides of four predatory mites from the family Phytoseiidae attacking Thrips palmi Karny (Thysanoptera: Thripidae). Pest. Manag. Sci., 68(9): 1289-1295. https://doi.org/10.1002/ps.3296
- Dalir S., Hajiqanbar H., Fathipoura Y., Khanamanib M. 2021. A comprehensive picture of foraging strategies of Neoseiulus cucumeris and Amblyseius swirskii on western flower thrips. Pest Manag. Sci., 77(12): 5418-5429. https://doi.org/10.1002/ps.6581
- Dameda C., Berté A.L.W., Silva G.L.d., Johann L., Ferla N.J. 2021. Euseius concordis (chant) (Acari: Phytoseiidae) as a potential agent for the control of yerba mate red mite Oligonychus yothersi (McGregor) (Acari: Tetranychidae). Phytoparasitica. 49(3): 377-383. https://doi.org/10.1007/s12600-020-00879-4
- De Courcy Williams M.E. 2001. Biological control of thrips on ornamental crops: interactions between the predatory mite Neoseiulus cucumeris (Acari: Phytoseiidae) and western flower thrips, Frankliniella occidentalis (Thysanoptera: Thripidae), on cyclamen. Biocontrol Sci. Tech., 11(1): 41-55. https://doi.org/10.1080/09583150020029736
- De Figueiredo E.S., Massaro M., Carmo S.D., Moraes G.J.D. 2018. Rearing system for the predatory phytoseiid Euseius concordis (Acari: Phytoseiidae). Exp. Appl. Acarol., 74(1):13-23. https://doi.org/10.1007/s10493-018-0212-8
- Demite P.R., de Moraes G.J., McMurtry J.A., Denmark H.A., Castilho R. C. 2024. Phytoseiidae Database. Available from: http://www.lea.esalq.usp.br/phytoseiidae/. Accessed 13/03/2024
- Doğramaci M., Arthurs S.P., Chen J., McKenzie C., Irrizary F., Osborne L. 2011. Management of chilli thrips Scirtothrips dorsalis (Thysanoptera: Thripidae) on peppers by Amblyseius swirskii (Acari: Phytoseiidae) and Orius insidiosus (Hemiptera: Anthocoridae). Biol. Control, 59(3): 340-347. https://doi.org/10.1016/j.biocontrol.2011.09.008
- Döker İ, Pappas M.L., Samaras K., Triantafyllou A., Kazak C., Broufas G.D. 2015. Compatibility of reduced-risk insecticides with the non-target predatory mite Iphiseius degenerans (Acari: Phytoseiidae). Pest Manag. Sci.,71(9):1267-1273. https://doi.org/10.1002/ps.3921
- Döker I., Hernandez Y. V., Mannion C., Carrilo D. 2018. First report of Amblyseius tamatavensis (Acari: Phytoseiidae) in the United States of America. Inter. J. Acarol., 44 (2-3): 347-360. https://doi.org/10.1080/01647954.2018.1461132
- Döker I., Kazak C., Karut K. 2020. The genus Amblyseius Berlese (Acari: Phytoseiidae) in Turkey with discussion on the identity of Amblyseius meridionalis. Sys. Appl. Acarol., 25(8): 1395-1420. https://doi.org/10.11158/saa.25.8.4
- Döker I., Revynthi A.M., Kazak C., Carrillo D. 2021. Interactions among exotic and native phytoseiids (Acari: Phytoseiidae) affect biocontrol of two-spotted spider mite on papaya. Biological Control, 163, art. no. 104758. https://doi.org/10.1016/j.biocontrol.2021.104758
- Döker I., Khaustov V.A., Joharchi O., Khaustov A.A., Kazakov D.V., Meshkov Y.I. 2024. Integrative taxonomy demonstrates synonyms between Euseius amissibilis Meshkov and Euseius gallicus Kreiter & Tixier (Acari: Phytoseiidae). Sys. Appl. Acarol., 29 (1): 60-77. doi.org/10.11158/saa.29.1.5%20 https://doi.org/10.11158/saa.29.1.5
- Ebssa L., Borgemeister C., Poehling H-M. 2006. Simultaneous application of entomopathogenic nematodes and predatory mites to control western flower thrips Frankliniella occidentalis. Biol. Control, 39(1): 66-74. https://doi.org/10.1016/j.biocontrol.2006.02.005
- El-Banhawy E.M., Hafez S.M., Saber S.A. 2001. Response of Amblyseius cydnodactylon (Phytoseiidae) to increasing prey density of Tetranychus urticae (Tetranychidae) in absence or presence of nymphs of Bemesia tabaci (Homoptera) in Egypt. Inter. J. Acarol., 27(3): 241-244. https://doi.org/10.1080/01647950108684261
- Elkholy S.Z., Abolfadel M.A., Walash E.H. 2022. Biology, life table and efficacy of predatory mite, Cydnoseius negevi (Acari: Phytoseiidae) for controlling some pests on Solanum melongena in Egypt. Acarines, 16(1): 23-28. https://doi.org/10.21608/ajesa.2022.291544
- Emmert C.J., Mizell R.F., Andersen, P.C., Frannk J.H., Jerry L., Stimac J.L. 2008. Effects of contrasting diets and temperatures on reproduction and prey consumption by Proprioseiopsis asetus (Acari: Phytoseiidae). Exp. Appl. Acarol., 44(1): 11-26. https://doi.org/10.1007/s10493-008-9130-5
- Evans G.A. 2007. The whiteflies (Hemiptera: Aleyroididae) of the world and their host plants and natural enemies. USDA/Animal Plant Health Inspection Service (APHIS). 708p.
- Farfan M.A., Schmidt-Jeffris R.A. 2019. Biodiversity of Phytoseiidae (Acari: Mesostigmata) of annual specialty crop systems: the current state of knowledge worldwide and the need for study in North America. In: Skvarla, M., Ochoa, R., Verle Rodrigues, J., Hutcheson, H. (eds) Contemporary Acarology. Springer, Cham. https://doi.org/10.1007/978-3-030-17265-7_6
- Farfan M.A., Coffey J., Schmidt-Jeffris R. 2021a. Evaluation of Tarsonemus bilobatus and Podosphaera xanthii as suitable resources for Proprioseiopsis mexicanus in cucurbit systems in the Southeast USA. Exp. Appl. Acarol., 85(1): 31-40. https://doi.org/10.1007/s10493-021-00658-8
- Farfan M.A., Coffey J., Schmidt-Jeffris R. 2021b. Suitability of food resources for Proprioseiopsis mexicanus, a potentially important natural enemy in eastern USA agroecosystems. Exp. Appl. Acarol., 84(1): 121-134. https://doi.org/10.1007/s10493-021-00622-6
- Fouly A.H., Awadalla S.S., Ata T.E., Marouf E.A., 2021. Influence of alternative food sources on different biological aspects of Cydnoseius negevi (Acari: Phytoseiidae). J. Plant Prot. Path., Mansoura Univ., 12 (4): 295-301. https://doi.org/10.21608/jppp.2021.171265
- Fouly A.H., Al-Deghairi M.A., Abdel Baky N.F. 2011. Biological aspects and life tables of Typhlodromips swirskii (Acari: Phytoseiidae) fed Bemisia tabaci (Hemiptera: Aleyroididae). J. Entomol., 8(1):52-62. https://doi.org/10.3923/je.2011.52.62
- Fouly A.H., Nassar O.A., Osman M.A. 2013. Biology and life tables of Euseius scutalis (A.-H.) reared on different kinds of food. J. Entomol., 10(4):199-206. https://doi.org/10.3923/je.2013.199.206
- Gerson U., Smiley R.L., Ochoa R. 2003. Mites (Acari) for pest control. Blackwell Science, Oxford, UK, pp. 539. https://doi.org/10.1002/9780470750995
- Ghasemzadeh S., Leman A., Messelink G.J. 2017. Biological control of Echinothrips americanus by phytoseiid predatory mites and the effect of pollen as supplemental food. Exp. App. Acarol., 73(2): 209-221. https://doi.org/10.1007/s10493-017-0191-1
- Gullino M.L., Albajes R., Nicot P.C. 2020. Integrated pest and disease management in greenhouse crops, Second Edition, Springer Nature Switzerland AG, 691 pp. https://doi.org/10.1007/978-3-030-22304-5_14
- Guo Y., Lv., J., Jiang, X., Wang B., Gao Y., Wang E., Xu X. 2016. Intraguild predation between Amblyseius swirskii and two native Chinese predatory mite species and their development on intraguild prey. Sci. Rep., 6:1-7. https://doi.org/10.1038/srep22992
- Heydari S., Allahyari H., Golpayegani A.Z, Farhadi R. 2016. The effect of the simultaneous presence of two prey species on the predation rate of Amblyseius swirskii (Acari: Phytoseiidae). Proceeding of 22nd Iranian plant protection congress 27-30 August 2016, College of Agriculture and Natural Resources, University of Tehran, Karaj, IRAN, 605.
- Hosseininia A., Khanjani M., Asadi M., Soltani J. 2020. Life history of the Predatory Mite Amblyseius swirskii (Athias- Henriot) (Acari: Phytoseiidae) on Tetranychus urticae Koch (Acari: Tetranychidae), Carpoglyphus lactis Linnaeus (Acari: Carpoglyphidae) and Trialeurodes vaporariorum (Westwood) (Hemiptera: Aleyrodidae). J. Ornam. Plants, 10(3): 155-166.
- Hou F., Ni Z.-H., Zou M.-T., Zhu R., Yi T.-C., Guo J-J., Jin D-C. 2022. The effects of alternative foods on life history and cannibalism of Amblyseius herbicolus (Acari: Phytoseiidae). Insects, 13(11): 1036. https://doi.org/10.3390/insects13111036
- Huang J.H., Freed S., Wang L.S., Qin W.J., Chen H.F., Qin H.G. 2014. Effect of temperature on development and reproduction of Proprioseiopsis asetus (Acari: Phytoseiidae) fed on asparagus thrips, Thrips tabaci. Exp. Appl. Acarol., 64(2): 235-244. https://doi.org/10.1007/s10493-014-9819-6
- Jacobson R.J., Croft P., Fenlon J. 2001. Suppressing establishment of Frankliniella occidentalis Pergande (Thysanoptera: Thripidae) in cucumber crops by prophylactic release of Amblyseius cucumeris Oudemans (Acarina: Phytoseiidae) Biocontrol Sci. Tech., 11(1): 27-34. https://doi.org/10.1080/09583150020029718
- Jafari S., Abassi N., Bahirae F. 2013. Demographic parameters of Neoseiulus barkeri (Acari: Phytoseiidae) fed on Thrips tabaci (Thysanoptera: Thripidae). Persian J. Acarol., 2 (2): 287-296. https://doi.org/10.22073/pja.v2i2.10031
- Jenser G., Szenasi A. 2004. Review of the biology and vector capability of Thrips tabaci Lindeman (Thysanoptera: Thripidae). Acta Phytopathol. Entomol. Hung., 39(1-3):137-155. https://doi.org/10.1556/APhyt.39.2004.1-3.14
- Ji J., Lin T., Zhang Y., Sun S., Saito Y., Lin J., Chen X. 2013. Effects of starvation and humidity on the development and survival of Amblyseius swirskii, Agistemus exsertus and Amblyseius eharai. Sys. Appl. Acarol., 18(4):321-328. https://doi.org/10.11158/saa.18.4.2
- Jorge S.J., Demite P.R., Moraes G.J.D. 2021. First report of Amblyseius tamatavensis Blommers, 1974 (Acari: Phytoseiidae) in Peru, with predation observation and a key for the Amblyseius species reported so far from that country. Entomol. Commun., 3: ec03037. https://doi.org/10.37486/2675-1305.ec03037
- Kakkar G., Kumar V., Seal D.R., Liburd O.E., Stansly P.A. 2016. Predation by Neoseiulus cucumeris and Amblyseius swirskii on Thrips palmi and Frankliniella schultzei on cucumber. Biol. Control, 92: 85-91. https://doi.org/10.1016/j.biocontrol.2015.10.004
- Kasap I. and Şekeroğlu E. 2004. Life history of Euseius scutalis feeding on citrus red mite Panonychus citri at various temperatures. BioControl, 49(6): 645-654. https://doi.org/10.1023/B:BICO.0000046733.53887.2b
- Kazak C., Yildiz S., Şekeroğlu E. 2002. Biological characteristics and life tables of Amblyseius umbraticus Chant (Acari, Phytoseiidae) at three constant temperatures. J. Pest Sci., 75(5): 118-121. https://doi.org/10.1046/j.1472-8206.2002.02034.x
- Kishimoto H. 2014. Development and oviposition of six native phytoseiid species (Acari: Phytoseiidae) reared on pink citrus rust mite, Aculops pelekassi (Keifer) (Acari: Eriophyidae). J. Acarol. Soc. Jpn., 23(2): 71-77. https://doi.org/10.2300/acari.23.71
- Kishimoto H., Yaginuma K., Toyama M. 2018. Effects of pesticides on four native generalist phytoseiid species (Acari: Phytoseiidae). Jpn. J. Appl. Entomol. Zool., 62(1): 29-39. https://doi.org/10.1303/jjaez.2018.29
- Kishimoto H., Yaginuma K., Furihata S, Ito T., Yaegashi H., Toyama M., 2020. Effects of fungicides on four native generalist phytoseiid species (Acari: Phytoseiidae). Jpn. J. Appl. Entomol. Zool., 64(4): 175-182. https://doi.org/10.1303/jjaez.2020.175
- Knapp M., van Houten Y., Hoogerbrugge H., Bolckmans K. 2013. Amblydromalus limonicus (Acari: Phytoseiidae) as a biocontrol agent: literature review and new findings. Acarologia, 53(2): 191-202. https://doi.org/10.1051/acarologia/20132088
- Knapp M., van Houten Y, van Baal E., Groot T., 2018. Use of predatory mites in commercial biocontrol: current status and future prospects. Acarologia, 58 (Suppl): 72-82. https://doi.org/10.24349/acarologia/20184275
- Komi K., Arakawa R., Amano H., 2008. Predatory potential against Thrips palmi Karny of some native phytoseiid mites (Acari: Phytoseiidae) occurring on greenhouse vegetable crops in Kochi prefecture. Japan. J. Acarol. Soc. Jpn., 17(1): 29-35. https://doi.org/10.2300/acari.17.29
- Kostiainen T.S., Hoy M.A. 1996. The Phytoseiidae as biological control agents of pest mites and insects. A bibliography. Monograph 17, University of Florida, Agricultural Experiment Station, 355 pp.
- Kozlova E., Moor V., Krasavina L. 2020. Application of phytoseiid mites for whitefly control on roses in the North-West of Russia, BIO Web of Conferences 18, IV All-Russian Plant Protection Congress, 1-5. https://doi.org/10.1051/bioconf/20201800016
- Kreiter S., Bopp M-C., Douin M., Nguyen D.T., Wyckhuys K. 2020. Phytoseiidae of Vietnam (Acari: Mesostigmata) with description of a new species. Acarologia 60(1): 75-110. https://doi.org/10.24349/acarologia/20204362
- Kreiter S., Zriki G., Ryckewaert P., Pancarte C., Douin M., Tixier M.-S. 2018. Phytoseiid mites of Martinique, with redescription of four species and new records (Acari: Mesostigmata). Acarologia, 58(2): 366-407. https://doi.org/10.24349/acarologia/20184248
- Kütük H., 2017. Performance of the predator Amblyseius swirskii (Acari: Phytoseiidae) on greenhouse eggplants in the absence and presence of pine Pinus brutia (Pinales: Pinaceae) pollen. Entomol. Res., 47(40): 263-269. https://doi.org/10.1111/1748-5967.12222
- Kütük H., 2018. Performance of the predator Amblyseius swirskii (Athias-Henriot) (Acari: Phytoseiidae) on plastic greenhouse pepper sprayed vs unsprayed pine pollen. Derim, 35(2):135-140. https://doi.org/10.16882/derim.2018.396951
- Kütük H., Yigit A. 2011. Pre-establishment of Amblyseius swirskii (Athias-Henriot) (Acari: Phytoseiidae) using Pinus brutia (Ten.) (Pinales: Pinaceae) pollen for thrips (Thysanoptera: Thripidae) control in green¬house peppers. Int. J. Acarol., 37(sup 1), 95-101. https://doi.org/10.1080/01647954.2010.540081
- Labbé R.M., Gagnier D., Shipp L. 2019. Comparison of Transeius montdorensis (Acari: Phytoseiidae) to other phytoseiid mites for the short-season suppression of western flower thrips, Frankliniella occidentalis (Thysanoptera: Thripidae). Environ. Entomol., 20 (20): 1-8. https://doi.org/10.1093/ee/nvz017
- Lee M.H., Zhang Z. 2018. Assessing the augmentation of Amblydromalus limonicus with the supplementation of pollen, thread, and substrates to combat greenhouse whitefly populations. Sci. Rep., 8: 1-14. https://doi.org/10.1038/s41598-018-30018-3
- Li Y-T., Jiang J-Y-Q., Huang Y-Q., Wang Z-H., Zhang J-P. 2015. Effects of temperature on development and reproduction of Neoseiulus bicaudus (Phytoseiidae) feeding on Tetranychus turkestani (Tetranychidae). Sys. Appl. Acarol., 20(5): 478-490. https://doi.org/10.11158/saa.20.5.4
- Li M., Yang N., Wan F., Liu L., Chen Y., Li J., Fu J. 2017. Functional response of Neoseiulus cucumeris (Oudemans) (Acari: Phytoseiidae) to Bemisia tabaci (Gennadius) on tomato leaves. Biocontrol Sci. Tech., 27(5): 677-685. https://doi.org/10.1080/09583157.2017.1328484
- Liao J-R., Ho C-C., Ko C-C. 2017. Species of the genus Euseius Wainstein (Acari: Phytoseiidae: Amblyseiinae) from Taiwan. Zootaxa, 4226(2): 205-228. https://doi.org/10.11646/zootaxa.4226.2.3
- Lopes P.C., Kanno R.H., Sourassou N.F., Moraes G.J.D. 2018. Effect of temperature and diet on the morphology of Euseius concordis (Acari: Phytoseiidae). Sys. Appl. Acarol., 23(7): 1322-1332. https://doi.org/10.11158/saa.23.7.9
- Ma M., Fan Q-H., Zhang Z-Q.2018. Morphological ontogeny of Amblydromalus limonicus (Acari: Phytoseiidae). Syst. Appl. Acarol., 23(9): 1741-1765. https://doi.org/10.11158/saa.23.9.3
- Madadi H., Annie Enkegaard A., Brødsgaard H.F., Kharrazi-Pakdel A., Ashouri A., Mohaghegh-Neishabouri J. 2009. Interactions between Orius albidipennis (Heteroptera: Anthocoridae) and Neoseiulus cucumeris (Acari: Phytoseiidae): Effects of host plants under microcosm condition. Biol. Control 50(2): 137-142. https://doi.org/10.1016/j.biocontrol.2009.03.015
- Manjunatha M., Hanchinal S.G., Kulkarni S.V. 2001. Interaction between Amblyseius ovalis and Polyphagotarsonemus latus and efficacy of A. ovalis on chilli mite and thrips. Karnataka J. Agri. Sci., 14(2): 506-509.
- Massaro M. and Moraes G. J. D. 2019. Predation and oviposition potential of Brazilian populations of the predatory mite Amblyseius tamatavensis (Acari: Phytoseiidae) on eggs of Bemisia tabaci (Insecta: Hemiptera). Acarologia, 59(1): 120-128. https://doi.org/10.24349/acarologia/20194314
- McMurtry J.A., Moraes G.J.D., Sourassou N.F. 2013. Revision of the lifestyles of phytoseiid mites (Acari: Phytoseiidae) and implications for biological control strategies. Sys. Appl. Acarol., 18(4): 297-320. https://doi.org/10.11158/saa.18.4.1
- McMurtry J.A., Croft B.A. 1997. Life styles of phytoseiid mites and their role in biological control. Ann. Rev. Entomol., 42: 291-321. https://doi.org/10.1146/annurev.ento.42.1.291
- Medd N.C. and GreatRex R.M. 2014. An evaluation of three predatory mite species for the control of greenhouse whitefly (Trialeurodes vaporariorum). Pest Manag. Sci., 70(10): 1492-1496. https://doi.org/10.1002/ps.5433
- Messelink G.J., van Maanen R., van Steenpaal S.E.F., Janssen A. 2008. Biological control of thrips and whiteflies by a shared predator: two pests are better than one. Biol. Control, 44(3): 372-379. https://doi.org/10.1016/j.biocontrol.2007.10.017
- Messelink G.J., van Maanen R., van Holstein-Saj R, Sabelis M.W., Janssen A. 2010. Pest species diversity enhances control of spider mites and whiteflies by a generalist phytoseiid predator. Biocontrol, 55(3): 387-398. https://doi.org/10.1007/s10526-009-9258-1
- Messelink G.J., van Steenpaal S.E.F., Ramakers P.M.J. 2006. Evaluation of phytoseiid predators for control of western flower thrips on greenhouse cucumber. BioControl, 51(6): 753-768. https://doi.org/10.1007/s10526-006-9013-9
- Messelink G.J., van Steenpaal S., van Wensveen W. 2005 Typhlodromips swirskii (Athias-Henriot) (Acari: Phytoseiidae): a new predator for thrips control in greenhouse cucumber. IOBC/WPRS Bulletin, 28(1): 183-186.
- Minor M. 2008. Phytoseiidae of New Zealand ( http://keys.lucidcentral.org/keys/v3/phytoseiidae/key/Phytoseiidae/Media/Html/about.htm) Accessed 30 October 2023.
- Mitina G.V., Krasavina L.P., Trapeznikova O.V. 2021. Computability of the fungus Lecanicillium muscarium and the predatory mite Amblyseius swirskii for their combined application against the greenhouse whitefly Trialeurodes vaporariorum. Plant Protect. News, 104(3): 163-170. https://doi.org/10.31993/2308-6459-2021-104-3-15026
- Mochizuki M. 2009. Development, Reproduction and Prey Consumption of the Thrips Predator Gynaeseius liturivorus (Ehara) (Acari: Phytoseiidae). J. Acarol. Soc. Jpn., 18(2): 73-84. https://doi.org/10.2300/acari.18.73
- Mohamed N.A., Imam I. 2019. The effect of different preys on behavior of the predatory mite Amblyseius swirskii, Egypt. J. Desert Res., 69(3): Special Issue, 47-57. https://doi.org/10.21608/ejdr.2020.18475.1063
- Momen F.M., Rasmy A.H., Zaher M.A., Nawar M.S., Abou-Elella G.M. 2004. Dietary effect on the development, reproduction and sex ratio of the predatory mite Amblyseius denmarki Zaher & El-Borolosy (Acari: Phytoseiidae). Int. J. Trop. Insect Sci., 24 (2):192-195. https://doi.org/10.1079/IJT200414
- Momen F. M., Abdel-Khalek A., El-Sawi, S. 2009. Life tables of the predatory mite Typhlodromus negevi feeding on prey insect species and pollen diet (Acari: Phytoseiidae). Acta Phytopathol. Entomol. Hung., 44(2): 353-366. https://doi.org/10.1556/APhyt.44.2009.2.12
- Momen F.M., Hussein H., Reda A.S. 2013. Intra-guild vs extra-guild prey: effect on development, predation and preference of Typhlodromus negevi Swirski and Amitai and Typhlodromips swirskii (Athias-Henriot) (Acari: Phytoseiidae), Acta Phytopathol. Entomol. Hung., 48 (1): 95-106. https://doi.org/10.1556/APhyt.48.2013.1.9
- Muma M.H. 1971. Food habits of Phytoseiidae (Acarina: Mesostigmata) including common species in Florida citrus. Fla. Entomol., 54 (1): 21-34. https://doi.org/10.2307/3493786
- Mutisya D., Kariuki C.W., El-banhawy E.M., Khamala C. 2014. Typhlqdromalus aripo de leon (Acari: Phytoseiidae) development and reproduction on major cassava pests at different temperatures and humidities: an indication of enhanced mite resilience. Acarologia, 54(4): 395-407. https://doi.org/10.1051/acarologia/20142145
- Nakahara S. 1994. The genus Thrips Linnaeus (Thysanoptera: Thripidae) of the New World, United States Department of Agriculture, Agr. Res. Ser. Tech. Bull., 1822, 183pp.
- Navas-Castillo J., Fiallo-Olivé E., Sánchez-Campos S. 2011. Emerging virus diseases transmitted by whiteflies. Ann. Rev. Phytopathol., 49: 219-248. https://doi.org/10.1146/annurev-phyto-072910-095235
- Nguyen D.T., Jonckheere W., Nguyen V.H., Van Leeuwen T., De Clercq P. 2019. Life tables and feeding habits of Proprioseiopsis lenis (Acari: Phytoseiidae) and implications for its biological control potential in Southeast Asia. Syst. Appl. Acarol., 24(5): 857-865. https://doi.org/10.11158/saa.24.5.9
- Nomikou M., Janssen A., Shraag R., Sabelis M.M. 2001. Phytoseiid predators as potential biological con¬trol agents for Bemisia tabaci. Exp. Appl. Acarol., 25(4): 271-291. https://doi.org/10.1023/A:1017976725685
- Nomikou M., Janssen A., Schraag R., Sabelis M.W. 2002. Phytoseiid predators suppress populations of Bemisia tabaci on cucumber plants with alternative food. Exp. Appl. Acarol., 27(1-2): 57-68. https://doi.org/10.1023/A:1021559421344
- Nomikou M., Janssen A., Sabelis M.W. 2003a. Phytoseiid predator of whitefly feeds on plant tissue. Exp. Appl. Acarol., 31(1-2): 27-36. https://doi.org/10.1023/B:APPA.0000005150.33813.04
- Nomikou M., Janssen A., Schraag R., Sabelis, M.W. 2003b. Phytoseiid predators of whiteflies feed and reproduce on non-prey food sources. Exp. Appl. Acarol., 31(1-2): 15-26. https://doi.org/10.1023/B:APPA.0000005142.31959.e8
- Nomikou M., Janssen A., Schraag R., Sabelis M.W. 2004. Vulnerability of Bemisia tabaci immatures to phytoseiid predators: consequences for oviposition and influence of alternative food. Entomol. Exp. Appl., 110 (Special issue): 95-102. https://doi.org/10.1111/j.0013-8703.2004.00114.x
- Pappas M.L., Xanthis C., Samaras K., Koveos D.S., Broufas G.D. 2013. Potential of the predatory mite Phytoseius finitimus (Acari: Phytoseiidae) to feed and reproduce on greenhouse pests. Exp. Appl. Acarol., 61(4): 387-401. https://doi.org/10.1007/s10493-013-9711-9
- Park Y-G., Lee J-H., Lim U.T. 2021. Functional response of Amblyseius eharai (Acari: Phytoseiidae) on Tetranychus urticae (Acari: Tetranychidae). PLoS ONE 16(12): e0260861. https://doi.org/10.1371/journal.pone.0260861
- Pijnakker J., Victoria N.G., Ramakers P.M. 2007. Predatory mites for biocontrol of the greenhouse whitefly, Trialeurodes vaporariorum in cut roses. Proc. IVth International Symposium on Rose Research and Cultivation Ed. H.B. Pemberton Acta Hort. 751. https://doi.org/10.17660/ActaHortic.2007.751.33
- Pijnakker J., de Souza A., Wäckers F. 2014. Euseius gallicus, a bodyguard for roses, Integrated Control in Protected Crops, Temperate Climate. IOBC-WPRS Bulletin, 102:191-195.
- Pirayeshfar F., Safavi S.A., Moayeri H.R.S., Messelink, G.J. 2021. Provision of astigmatid mites as supplementary food increases the density of the predatory mite Amblyseius swirskii in greenhouse crops, but does not support the omnivorous pest, western flower thrips. BioControl, 66(4): 511-522. https://doi.org/10.1007/s10526-021-10092-9
- Pratt P.D., Croft B.A. 1999. Expanded distribution of the bamboo spider mite Schizotetranychus longus (Acari; Tetranychidae), and predation by Neoseiulus fallacis (Acari: Phytoseiidae). Acarologia, 40(2): 191-197.
- Rahman T., Spafford H., Broughton S. 2011. Single versus multiple releases of predatory mites combined with spinosad for the management of western flower thrips in strawberry. Crop Protection, 30(4): 468-475. https://doi.org/10.1016/j.cropro.2010.11.027
- Rahmani H., Fathipur Y., Kamali K. 2009. Life history and population growth parameters of Neoseiulus californicus (Acari: Phytoseiidae) fed on Thrips tabaci (Thysanoptera: Thripidae) in laboratory condi¬tions. Syst. Appl. Acarol., 14(2): 91-100. https://doi.org/10.11158/saa.14.2.2
- Rahmani H., Hoseini M., Saboori A., Walzer A. 2016. Prey preference of the predatory mite Neoseiulus californicus (Mesostigmata: Phytoseiidae) when offered two major pest species, the two spotted spider mite and the onion thrips. Int. J. Acarol., 42(6): 319-323. https://doi.org/10.1080/01647954.2016.1191540
- Reitz S.R. 2009. Biology and ecology of the western flower thrips (Thysanoptera: Thripidae): the making of a pest. Fla. Entomol., 92(1): 7-13. https://doi.org/10.1653/024.092.0102
- Riley D.G., Joseph S.V., Srinivasan R., Diffie S. 2011. Thrips vectors of tospoviruses. J. Integr. Pest Manag., 2(1): 1-10. https://doi.org/10.1603/IPM10020
- Richter E. 2017. Efficacy of two predatory mite species to control whiteflies infesting poinsettia plants compared to the standard parasitoid Encarsia formosa. Proc. III International Symposium on Organic Greenhouse Horticulture Eds.: G.B. Öztekin and Y. Tüzel, 413-417. https://doi.org/10.17660/ActaHortic.2017.1164.53
- Saad N.M., Yousef A.A., Fouly A.H. 2021. Side Effect of Indigenous Entomopathogenic Fungi on the Predatory Mite, Cydnoseius negevi (Swirski and Amitai) (Acari: Phytoseiidae). J. Plant Prot. Pathol., 12 (3): 215-223. https://doi.org/10.21608/jppp.2021.66659.1020
- Samaras K., Pappas M.L., Pekas A., Wäckers F.L., Broufas G.D. 2021. Benefits of a balanced diet? Mixing prey with pollen is advantageous for the phytoseiid predator Amblydromalus limonicus. Biol. Control, 155, 104531. https://doi.org/10.1016/j.biocontrol.2021.104531
- Sanad A.S., Hassan G.M. 2019. Controlling the western flower thrips, Frankliniella occidentalis (Pergande) (Thysanoptera: Thripidae) by releasing the predatory phytoseiid mites and pesticides on pepper in a greenhouse. Egypt. J. Biol. Pest Control, 29:1-8. https://doi.org/10.1186/s41938-019-0186-9
- Sarwar M.H., Xuenong X., Endong W., Kongming W. 2011. The potential of four mite species (Acari: Phytoseiidae) as predators of sucking pests on protected cucumber (Cucumis sativus L.) crop. Afr. J. Agric. Res., 6: 73-78. https://doi.org/10.5897/AJAR10.670
- Schoeller E.N., McKenzie C.L., Osborne L.S. 2020. Comparison of the phytoseiid mites Amblyseius swirskii and Amblydromalus limonicus for biological control of chilli thrips, Scirtothrips dorsalis (Thysanoptera: Thripidae). Exp. Appl. Acarol., 82(3): 309-318. https://doi.org/10.1007/s10493-020-00556-5
- Scott Brown A.S., Simmonds M. S.J., Blaney W.M. 2003. Influence of a short exposure to teflubenzuron residues on the predation of thrips by Iphiseius degenerans (Acari: Phytoseiidae) and Orius laevigatus (Hemiptera: Anthocoridae). Pest Manag. Sci., 59:1255-1259. https://doi.org/10.1002/ps.749
- Seal D.R., Ciomperlik M., Richards M.L., Klassen W. 2006. Comparative effectiveness of chemical insecticides against the chilli thrips Scirtothrips dorsalis Hood (Thysanoptera: Thripidae), on pepper and their compatibility with natural enemies. Crop Protection, 25(9): 949-955. https://doi.org/10.1016/j.cropro.2005.12.008
- Seiedy M. 2015. Compatibility of Amblyseius swirskii (Acari: Phytoseiidae) and Beauveria bassiana for biological control of Trialeurodes vaporariorum (Hemiptera: Aleyrodidae). Syst. Appl. Acarol., 20(7):731-738. https://doi.org/10.11158/saa.20.7.2
- Seiedy M., Soleymani S., Hakimitabar M. 2016. Development and reproduction of the predatory mite Amblyseius swirskii Athias-Henriot (Acari: Phytoseiidae) on Tetranychus urticae Koch (Acari: Tetranychidae) and Bemisia tabaci Gennadius (Heteroptera: Aleyrodidae). Int. J. Acarol., 43(2): 160-164. https://doi.org/10.1080/01647954.2016.1248486
- Sengonca C., Drescher K. 2001. Laboratory studies on the suitability of Thrips tabaci Lindeman (Thysanoptera, Thripidae) as prey for the development, longevity, reproduction and predation of four predatory mite species of the genus Amblyseius (Acari, Phytoseiidae). J. Plant Dis. Protect., 108(1): 66-76.
- Shibao M., Ehara S., Hosomi A., Tanaka H. 2004 Seasonal fluctuation in population density of phytoseiid mites and the yellow tea thrips, Scirtothrips dorsalis Hood (Thysanoptera: Thripidae) on grape, and predation of the thrips by Euseius sojaensis (Ehara) (Acari: Phytoseiidae). Appl. Entomol. Zool., 39 (4): 727-730. https://doi.org/10.1303/aez.2004.727
- Shipp J.L., Wang K.S. 2003. Evaluation of Amblyseius cucumeris (Acari: Phytoseiidae) and Orius insidiosus (Hemiptera: Anthocoridae) for control of Frankliniella occidentalis (Thysanoptera: Thripidae) on greenhouse tomatoes. Biol. Control, 28(3): 271-281. https://doi.org/10.1016/S1049-9644(03)00091-4
- Silveira E.C.d., Reis P.R., Siqueira M.F., Toledo M.A., Liska G.R., Cirillo M.A. 2020. Functional response of Euseius concordis feeding on Oligonychus ilicis (Acari: Phytoseiidae, Tetranychidae), Exp. Appl. Acarol., 80(1): 2015-226. https://doi.org/10.1007/s10493-019-00454-5
- Soleymani S., Hakimitabar M., Seiedy M. 2016a. Food preference of Amblyseius swirskii (Acari: Phytoseiidae) on different stages of Tetranychus urticae (Acari: Tetranychidae) and Bemisia tabaci (Hemiptera: Aleyrodidae). Persian J. Acarol., 5 (1): 63-70. https://doi.org/10.22073/pja.v5i1.14712
- Soleymani S., Hakimitabar M., Seiedy M. 2016b. Prey preference of predatory mite Amblyseius swirskii (Acari: Phytoseiidae) on Tetranychus urticae (Acari: Tetranychidae) and Bemisia tabaci (Hemiptera: Aleyrodidae). Biocontrol Sci. Techn., 26 (4): 562-569. https://doi.org/10.1080/09583157.2015.1133808
- Souza B., Marucci R.C. 2021. Biological control in ornamental plants: from basic to applied knowledge. Orn. Hortic., 27 (2): 255-267. https://doi.org/10.1590/2447-536x.v27i2.2365
- Steiner M.Y., Goodwin S. 2005. Challenges for the implementation of integrated pest management of cucumber pests in protected crops - an Australian perspective. Acta Hortic., 731: 309-315. https://doi.org/10.17660/ActaHortic.2007.731.42
- Steiner M.Y., Goodwin S., Wellham T.M., Barchia I.M., Spohr, L.J. 2003. Biological studies of the Australian predatory mite Typhlodromalus lailae (Schicha) (Acari: Phytosiidae). Aus. J. Entomol., 42(2): 131-137. https://doi.org/10.1046/j.1440-6055.2003.00344.x
- Teich, Y. 1966. Mites of the family Phytoseiidae as predators of the tobacco whitefly, Bemisia tabaci Gennadius. Israel J. Agric. Res., 16(3): 141-142.
- Téllez M.M., Cabello T., Gámez M., Burguillo F.J., Rodríguez E. 2020. Comparative study of two predatory mites Amblyseius swirskii Athias-Henriot and Transeius montdorensis (Schicha) by predator-prey models for improving biological control of greenhouse cucumber. Ecol. Model., 431, 109197. https://doi.org/10.1016/j.ecolmodel.2020.109197
- Thoming G., Poehling H.-M. 2006. Integrating soil-applied Azadirachtin with Amblyseius cucumeris (Acari: Phytoseiidae) and Hypoaspis aculeifer (Acari: Laelapidae) for the management of Frankliniella occidentalis (Thysanoptera: Thripidae). Environ. Entomol., 35(3): 746-756. https://doi.org/10.1603/0046-225X-35.3.746
- Tixier M.-S., K.reiter S., Okassa M., Cheval B. 2009. A new species of the genus Euseius Wainstein (Acari: Phytoseiidae) from France. J. Nat. Hist., 44 (3-4): 241-254. https://doi.org/10.1080/00222930903383529
- Tixier M.-S., Douin M., Rocio O., Gonzalez L., Pount B., Kreiter S. 2020. Distribution and biological features of Typhlodromus (Anthoseius) recki (Acari: Phytoseiidae) on Tetranychus urticae, T. evansi (Acari: Tetranychidae) and Aculops lycopersici (Acari: Eriophyidae). Acarologia, 60(4): 684-697. https://doi.org/10.24349/acarologia/20204396
- Tsolakis H., Ragusa E. 2020. Specie aliene di recente introduzione in Italia: stato dell′arte. Atti Accademia Nazionale Italiana di Entomologia, 68:167-175.
- Tsuchida Y., Masui S. 2021. Biological control of the Japanese pear rust mite, Eriophyes chibaensis (Acari: Eriophyidae) and the Kanzawa spider mite, Tetranychus kanzawai (Acari: Tetranychidae) with Euseius sojaensis (Acari: Phytoseiidae). Exp. Appl. Acarol., 84(4):673-686. https://doi.org/10.1007/s10493-021-00644-0
- Tsuchida Y., Masui S. 2023. Efficacy of biocontrol of the yellow tea thrips and the Kanzawa spider mite with the generalist phytoseiid mite Euseius sojaensis differs between grape cultivars with different leaf morphological traits. BioControl, 68(4): 425-434. https://doi.org/10.1007/s10526-023-10201-w
- Vafaie E.K., Pemberton H.B., G.M., Kerns D.L., Eubanks M.D., Heinz K.M. 2021. Using multiple natural enemies to manage sweet potato whiteflies (Hemiptera: Aleyrodidae) in commercial poinsettia (Malpighiales: Euphorbiaceae) Production. J. Integr. Pest Manag., 12(1): 1-13. https://doi.org/10.1093/jipm/pmab010
- Van Driesche R.G., Lyon S.M., Nunn C.R. 2006. Compatibility of spinosad with predacious mites (Acari: Phytoseiidae) used to control western flower thrips (Thysanoptera: Thripidae) in greenhouse crops. Fla Entomol., 89(3): 396-401. https://doi.org/10.1653/0015-4040(2006)89%5B396:COSWPM%5D2.0.CO;2
- Van Houten Y.M., Hoogerbrugge H., Oude Lenferink K., Knapp M., Bolckmans K.J. 2016. Evaluation of Euseius gallicus as a biological control agent of western flower thrips and greenhouse whitefly in rose. J. Acarol. Soc. Jpn., 25(S1): 147-159. https://doi.org/10.2300/acari.25.Suppl_147
- Van Lenteren J.C. 2012. The state of commercial augmentative biological control: plenty of natural enemies, but a frustrating lack of uptake. BioControl, 57(1): 1-20. https://doi.org/10.1007/s10526-011-9395-1
- Van Lenteren J.C., Bolckmans K., Köhl J., Ravensberg W.J., Urbaneja A. 2017. Biological control using invertebrates and microorganisms: plenty of new opportunities. BioControl, 63(1): 39-59. https://doi.org/10.1007/s10526-017-9801-4
- Vangansbeke D., Nguyen D.T., Audenaert J., Verhoeven R., Deforce K., Gobin B., Tirry L., Clercq P.D. 2014. Diet-dependent cannibalism in the omnivorous phytoseiid mite Amblydromalus limonicus. Biol. Control, 74: 30-35. https://doi.org/10.1016/j.biocontrol.2014.03.015
- Vantornhout I., Minnaert H.L., Tirry L., Clercq P.D. 2005. Influence of diet on life table parameters of Iphiseius degenerans (Acari: Phytoseiidae). Exp. Appl. Acarol., 35(3): 183-195. https://doi.org/10.1007/s10493-004-3940-x
- Vartapetov S.G., Vasilev P.A. 1971. [Amblyseius swirskii Athias-Henriot (Parasitiformes, Phytoseiidae) predator of the tobacco thrips (Thrips tabaci Lind.)] [in Russian]. Biol. Zh. Arm. 24(11): 106-107.
- Vicente Dos Santos V., Tixier M.-S. 2018. Integrative taxonomy approach for analysing evolutionary history of the tribe Euseiini Chant & McMurtry (Acari: Phytoseiidae). Syst. Biodiversity, 16(3): 302-319. https://doi.org/10.1080/14772000.2017.1401562
- Vogel W. 1969. [The use of Phytoseiulus riegeli Dosse and Encarsia formosa Gahan for the control of mites (Tetranychus urticae Koch) and white flies (Trialeyrodes vaporariorum Westw.) in glasshouses] [in French]. Organ. Eur. Mediterr. Prot. Plant. Publ. Ser. A (52): 45-48
- Walzer A., Paulus H.F., Schausberger P. 2004. Ontogenetic shifts in intraguild predation on thrips by phytoseiid mites: the relevance of body size and diet specialization. Bull. Entomol. Res., 94 (6): 577-84. https://doi.org/10.1079/BER2004329
- Wei J., Liu Y., Sheng F., Wang E., Zhang B., Xu X. 2023. Predatory mite Amblyseius orientalis prefers egg stage and low density of Carpoglyphus lactis prey. Exp. Appl. Acarol., 90 (3-4): 267-276. https://doi.org/10.1007/s10493-023-00805-3
- Wimmer D., Hoffmann D., Schausberger P. 2008. Prey suitability of western flower thrips, Frankliniella occidentalis, and onion thrips, Thrips tabaci, for the predatory mite Amblyseius swirskii. Biocontrol Sci. Techn., 18 (6): 533-542. https://doi.org/10.1080/09583150802029784
- Wu S., Gao Y., Xu X., Wang E., Wang Y., Lei Z. 2014. Evaluation of Stratiolaelaos scimitus and Neoseiulus barkeri for biological control of thrips on greenhouse cucumbers. Biocontrol Sci. Techn., 24(10): 1110-1121. https://doi.org/10.1080/09583157.2014.924478
- Xiao Y., Avery P., Chen J., McKenzie C., Osborne L. 2012a. Ornamental pepper as banker plants for establishment of Amblyseius swirskii (Acari: Phytoseiidae) for biological control of multiple pests in greenhouse vegetable production. Biol. Control, 63(3): 279-286. https://doi.org/10.1016/j.biocontrol.2012.09.007
- Xin T. and Zhang Z. 2021. Suitability of pollen as an alternative food source for different developmental stages of Amblyseius herbicolus (Chant) (Acari: Phytoseiidae) to facilitate predation on whitefly eggs. Acarologia, 61(4): 790-801. https://doi.org/10.24349/bIV1-2heN
- Xu X., Enkegaard A. 2010. Prey preference of the predatory mite, Amblyseius swirskii between first-instar western flower thrips Frankliniella occidentalis and nymphs of the two-spotted spider mite Tetranychus urticae. J. Insect Sci., 10(149): 1-11. https://doi.org/10.1673/031.010.14109
- Yang J., Lv J., Liu J., Xu X., Wang E. 2019. Prey preference, reproductive performance, and life table of Amblyseius tsugawai (Acari: Phytoseiidae) feeding on Tetranychus urticae and Bemisia tabaci. Syst. Appl. Acarol., 24: 404-413. https://doi.org/10.11158/saa.24.3.6
- Yao H., Zheng W., Tariq K., Zhang H. 2014. Functional and numerical responses of three species of Predatory phytoseiid mites (Acari: Phytoseiidae) to Thrips flavidulus (Thysanoptera: Thripidae). Neotrop. Entomol., 43(5): 437-445. https://doi.org/10.1007/s13744-014-0229-6
- Zahidi A., Akchour A., Kreiter S., Tixier M-S., Msanda F., El Mousadik A. 2023. Phytoseiid mites (Acari: Mesostigmata) from Central West Morocco: new records and key to females of all recorded Moroccan species. Acarologia 63(3): 691-724. https://doi.org/10.24349/v5of-5oe1
- Zannou I.D., Hanna R. 2011. Clarifying the identity of Amblyseius swirskii and Amblyseius rykei (Acari: Phytoseiidae): are they two distinct species or two populations of one species? Exp. Appl. Acarol., 53(4): 339-347. https://doi.org/10.1007/s10493-010-9412-6
- Zemek R. Prenerova E. 1997. Powdery mildew (Ascomycotina: Erysiphales) as an alternative food for the predatory mite Typhlodromus pyri Scheuten (Acari: Phytoseiidae). Exp. Appl. Acarol., 21(6-7): 405-414. https://doi.org/10.1023/A:1018427812075
- Zergani A., Parvis S., Naser Nakkai F., Riahi E. 2023. Life history traits and population parameters of the predatory mite Euseius scutalis (Acari: Phytoseiidae) fed on Tetranychus turkestani (Acari: Tetranychidae) and pollen from three different plants. Acarologia, 63(3): 945-954. https://doi.org/10.24349/mrqf-arrz
- Zhang Y., Jiang J., Zhang Y., Qiu Y., Zhang J. 2017. Functional response and prey preference of Neoseiulus bicaudus (Mesostigmata: Phytoseiidae) to three important pests in Xinjiang, China. Environ. Entomol., 46 (3): 538 - 543. https://doi.org/10.1093/ee/nvx073
- Zhang X., Lv J., Hu Y., Wang B., Chen X., Xu X., Wang E. 2015. Prey preference and life table of Amblyseius orientalis on Bemisia tabaci and Tetranychus cinnabarinus. PLoS ONE 10(10): e0138820. https://doi.org/10.1371/journal.pone.0138820
- Zhang Y-N., Guo D-D., Jiang J-Y-Q., Zhang Y-J., Zhang J-P. 2016. Effects of host plant species on the development and reproduction of Neoseiulus bicaudus (Phytoseiidae) feeding on Tetranychus turkestani (Tetranychidae). Syst. Appl. Acarol., 21(5):647-656. https://doi.org/10.11158/saa.21.5.6
- Zhang X-N., Guo J-J., Zou X. Jin D-C. 2018. Pathogenic differences of the entomopathogenic fungus Isaria cateniannulata to the spider mite Tetranychus urticae (Trombidiformes: Tetranychidae) and its predator Euseius nicholsi (Mesostigmata: Phytoseiidae). Exp. Appl. Acarol., 75(1): 69-84. https://doi.org/10.1007/s10493-018-0247-x
- Zilahi-Balogh G.M.G., Shipp J.L., Cloutier C., Brodeur J. 2007. Predation by Neoseiulus cucumeris on western flower thrips, and its oviposition on greenhouse cucumber under winter vs summer conditions in a temperate climate. Biol. Control, 40(2): 160-167. https://doi.org/10.1016/j.biocontrol.2006.10.011



2024-04-03
Date accepted:
2024-05-23
Date published:
2024-05-29
Edited by:
Tsolakis, Haralabos

This work is licensed under a Creative Commons Attribution 4.0 International License
2024 Barbar, Ziad; Skinner, Margaret; Parker, Bruce L. and Kreiter, Serge
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



