Two species representing a rare genus Sinaxonopsis (Acari, Hydrachnidiae, Aturidae) from China, with discussion of some taxonomic features
Jia, Lan  1
; Li, Hai-Tao
1
; Li, Hai-Tao  2
and Guo, Jian-Jun
2
and Guo, Jian-Jun  3
3
1Institute of Entomology, Guizhou University; Scientific Observing and Experimental Station of Crop Pests in Guiyang, Ministry of Agricultural and Rural Affairs of the P.R. China, Guiyang 550025, P.R. China.
2Institute of Entomology, Guizhou University; Scientific Observing and Experimental Station of Crop Pests in Guiyang, Ministry of Agricultural and Rural Affairs of the P.R. China, Guiyang 550025, P.R. China.
3✉ Institute of Entomology, Guizhou University; Scientific Observing and Experimental Station of Crop Pests in Guiyang, Ministry of Agricultural and Rural Affairs of the P.R. China, Guiyang 550025, P.R. China.
2024 - Volume: 64 Issue: 2 pages: 554-574
https://doi.org/10.24349/orqe-mqhZooBank LSID: 27F5A3C9-FC58-47CA-AE05-6309DF752E46
Original research
Keywords
Abstract
Introduction
Aturidae Thor, 1900 is a family of water mites that is widely distributed and species-diverse. Members of this family have small, heavily sclerotized, and dorsoventrally flattened bodies (Gerecke 2014; Smit 2020). A rare genus, Sinaxonopsis Yi & Jin, 2012, was established on the following features: (1) dorsal shields formed by a large posterior plate and a pair of anterior platelets; (2) a relatively bigger and plumper body than other aturid mites (Yi and Jin 2012). Until now, only three species, Sinaxonopsis unicucrus Yi & Jin, 2012 (from China), Sinaxonopsis laosensis Pešić & Smit, 2016 (from Laos), Sinaxonopsis siamicus Smit, 2016 (from Thailand), have been recorded worldwide (Yi and Jin 2012; Pešić and Smit 2016; Smit 2016).
Both presence and position of glandularia are important and highly variable features in water mites (Wiles 1997; Jin 1997; Shatrov 2008); this variation is remarkable in Aturidae since, for example, some species of Aturus Kramer, 1875 lack the gland of D4 , and the genus Albia Thon, 1899 lacks of V1 (Wiles 1991; Kim and Chung 1993, 1995; Smit 2017). However, glandularia has received poor attention in Sinaxonopsis, and clarification of glandularia including their quantity, distribution and condition is significant in order to establish the taxonomic status of this group. In addition, as Aturidae species are small in size, distinguishing features are difficult to observe unless specimens are properly mounted, especially in this rare group of Sinaxonopsis, which make species identification much difficult.
In this paper, a Chinese species new to science, Sinaxonopsis hainanensis sp. nov., is described and illustrated in detail, focusing on the quantity, position, and condition of glandularia. We achieved this through the images of camera, optical microscope, and scanning electron microscope (SEM). Finally, the shape and color of the patches on the large posterior plate of species in Sinaxonopsis were compared between S. unicucrus and S. hainanensis sp. nov., and examined whether they are stable within species, or obviously different between species, and whether they can be used, besides glandularia information, as diagnostic features at the species level in this genus.
Material and methods
The collection, preservation of specimens and slide mounting follow Gu et al. (2020). Canon 5D Mark IV digital camera (Canon, Japan) in conjunction with a Mitutoyo Plan NIR 10x lens (Mitutoyo, Japan) and a Godox MF12 flash as the light source (Godox, China) were used to generate the images of water mites. Leica DM3000 microscope (Leica, Germany) was used to identify and illustrate the specimens. Scanning Electron Microscope (SEM, Nikon, Japan) processing and photography follow Li et al. (2022). All photographs and illustrations were edited with Adobe Photoshop CS6. Measurements were taken using a Nikon Ni-E microscope with a Nikon DS-Ri2 camera (Nikon, Japan) and are given in micrometers (μm).
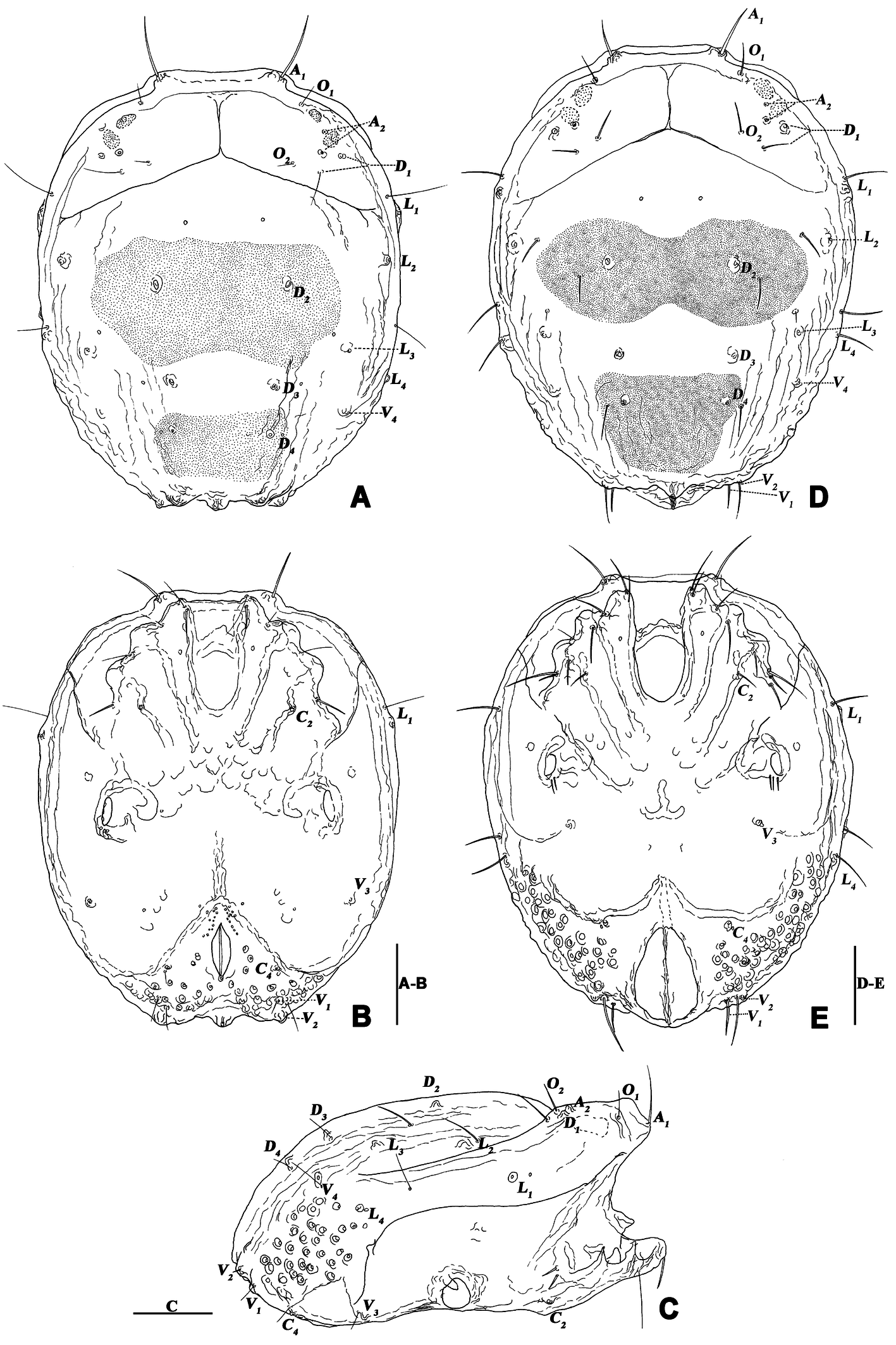
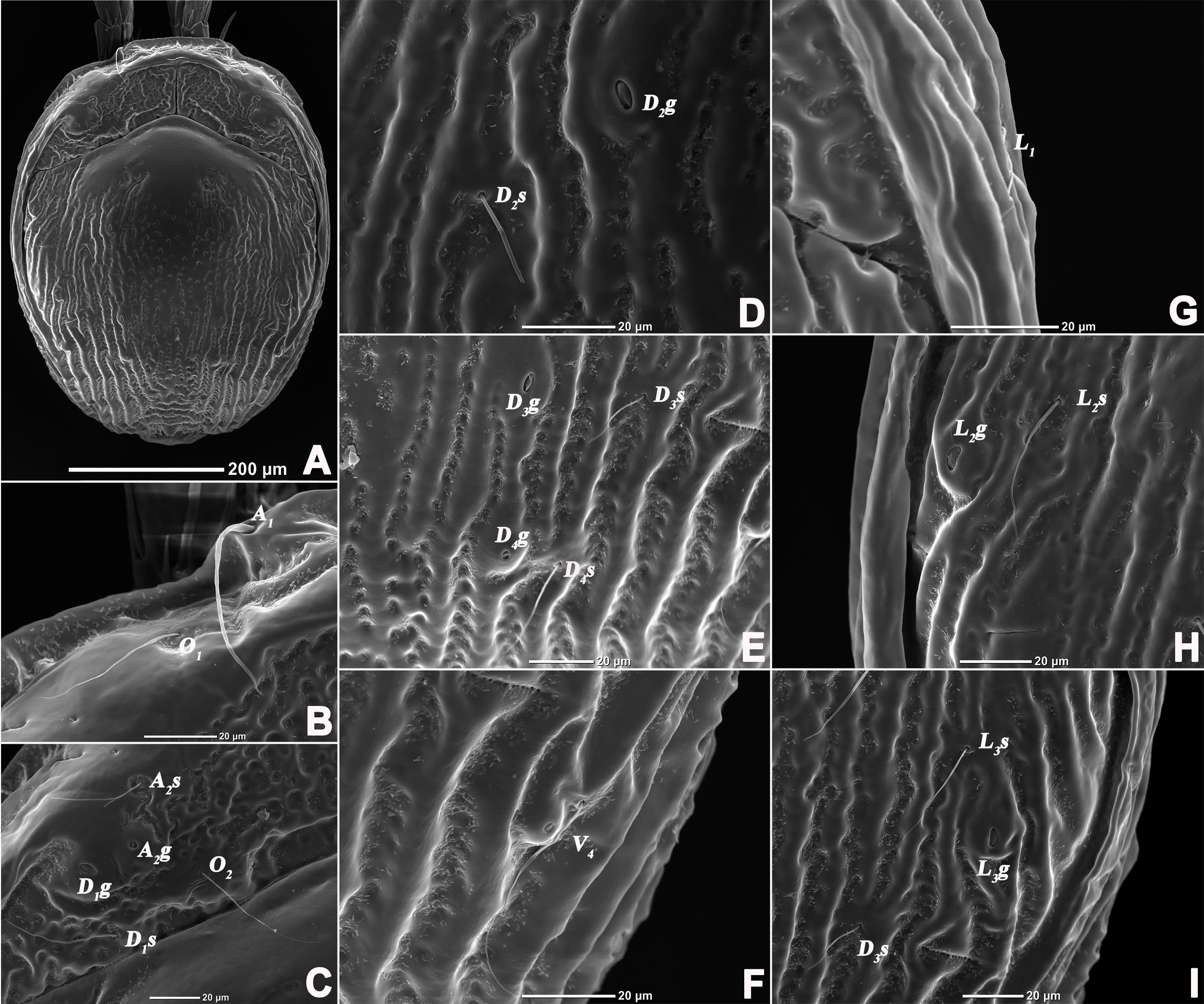
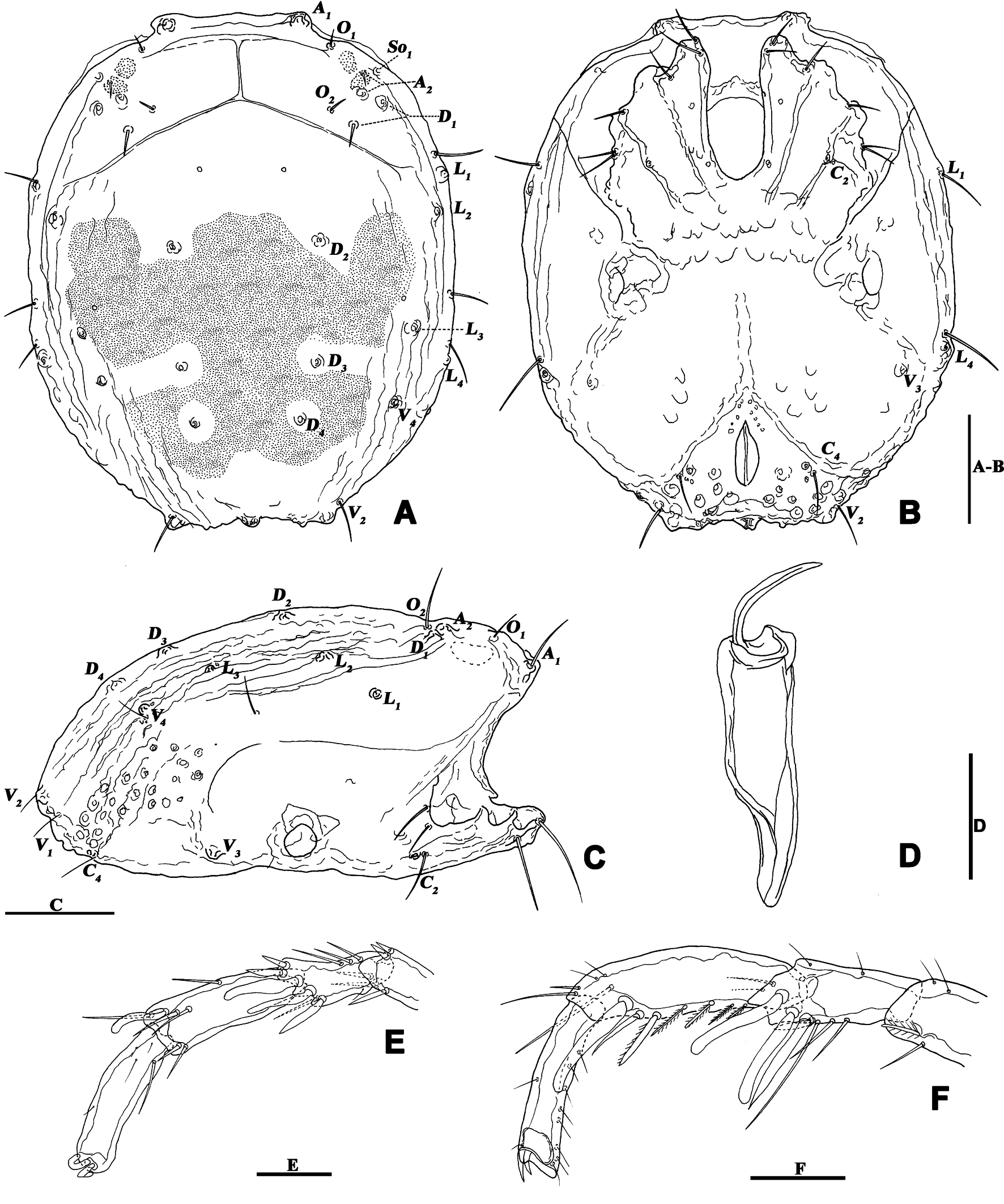
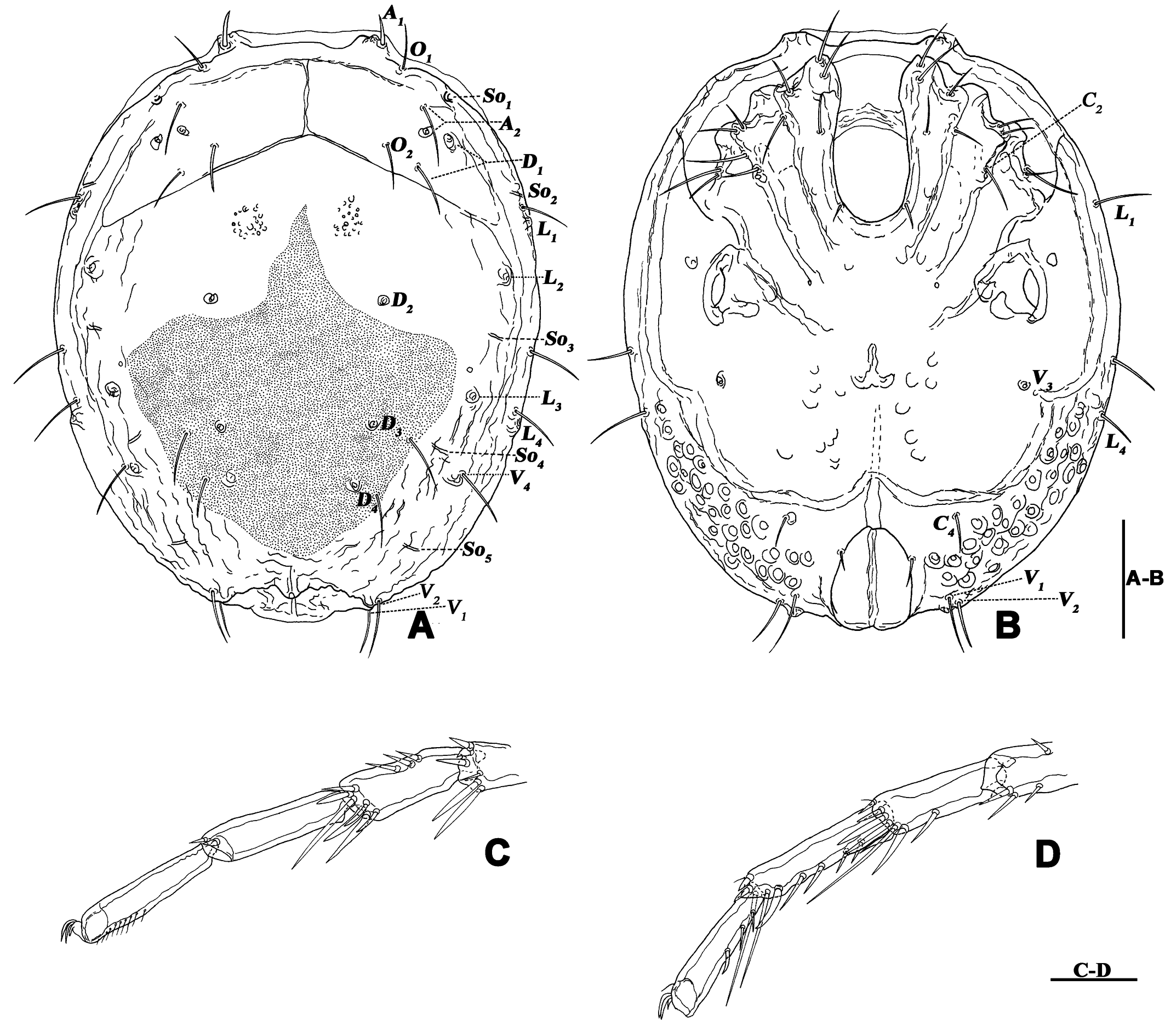
All terminology and abbreviations in this article were taken and updated from Jin (1997): a.s.l. = above sea level, A1 = preantennal glandularia, A2 = postantennal glandularia, A2g = glands of the A2 , A2s = seta of the A2 , C2 and C4 = coxoglandularia 2 and 4, Cx-I–Cx-IV = coxae I–IV, D1 –D4 = dorsoglandularia 1–4, D1g–D4g = glands of the D1 –D4 , D1s–D4s = setae of the D1 –D4 , dL = dorsal length, I-L-1–6, so forth. = first to sixth segment of the first leg, so forth., L = length, L1 –L4 = latero-glandularia 1–4, L1g–L4g = glands of the L1 –L4 , L1s–L4s = setae of the L1 –L4 , O1 = preocularia, O2 = postocularia, P-1–5 = first to fifth segments of the palp (from proximal to distal), V1 –V4 = ventroglandularia 1–4, vL = ventral length, W = width.
The type series were deposited in the Institute of Entomology, Guizhou University, Guiyang, P.R. China (GUGC).
Systematics
Family Aturidae Thor, 1900
Subfamily Axonopsinae K. Viets, 1929
Genus Sinaxonopsis Yi & Jin, 2012
Sinaxonopsis unicucrus Yi & Jin, 2012
(Figures 1–12)
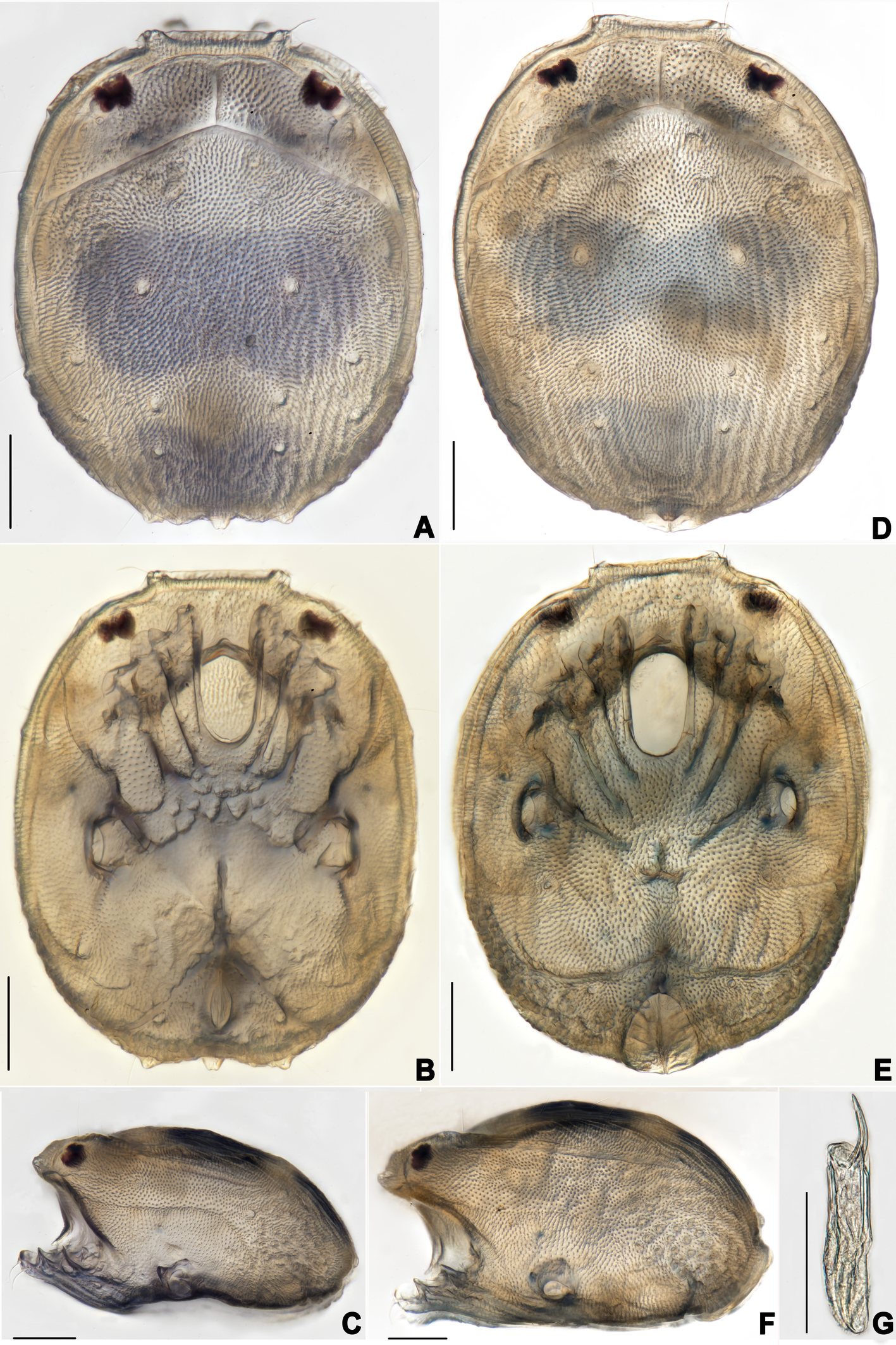

























Habitat — Slow flow of stream, many pebbles covered by mosses at the bottom.
Material examined — 3♂, 9♀, Baotianman National Nature Reserve, Nanyang City, Henan Province, P.R. China (33°46′73″ N, 111°89′98″ E, 555 m a.s.l.), collected by Hai-Tao Li, 11-VI-2021. Slides No. HN-AT-2021061101–2021061112.
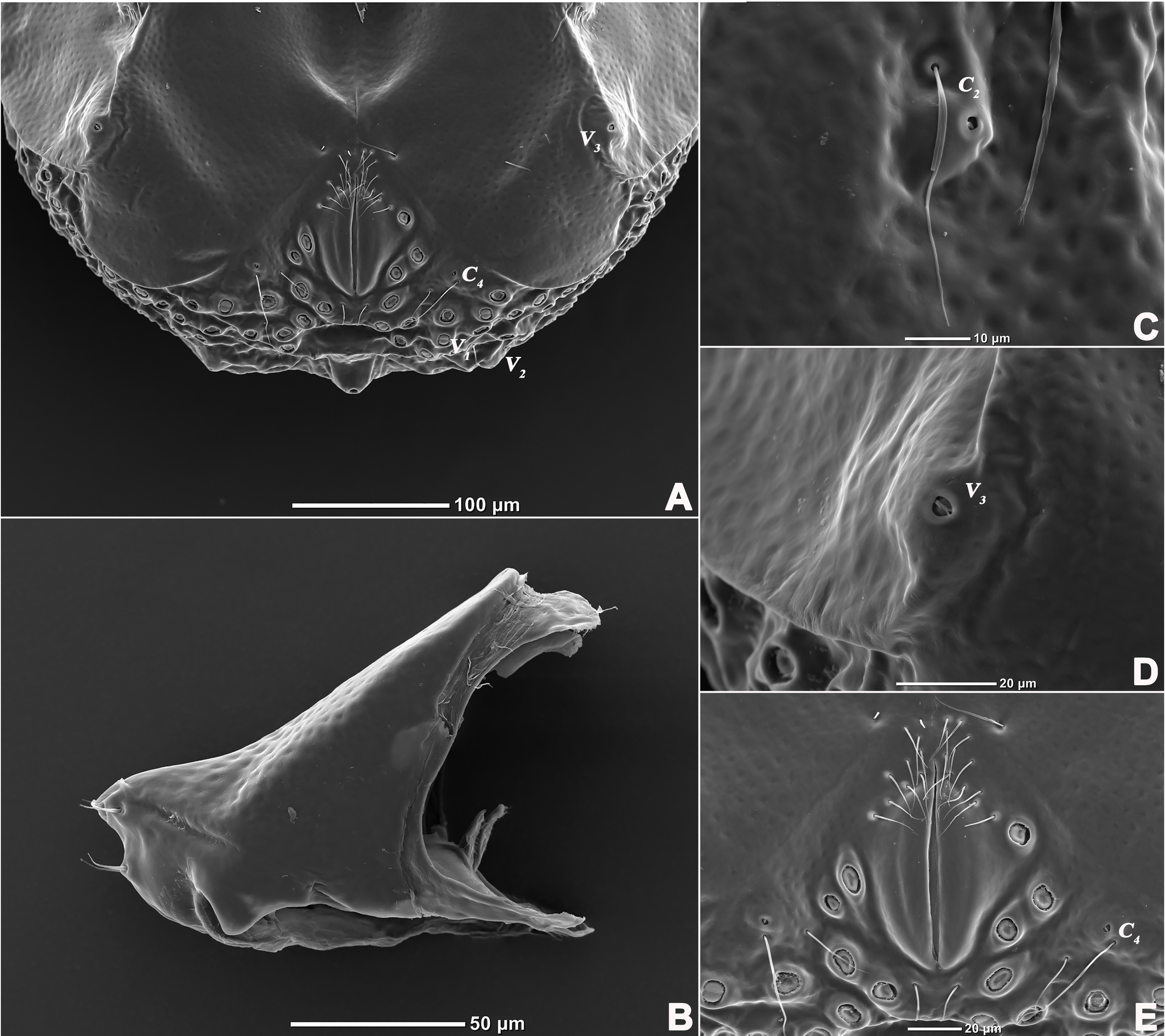
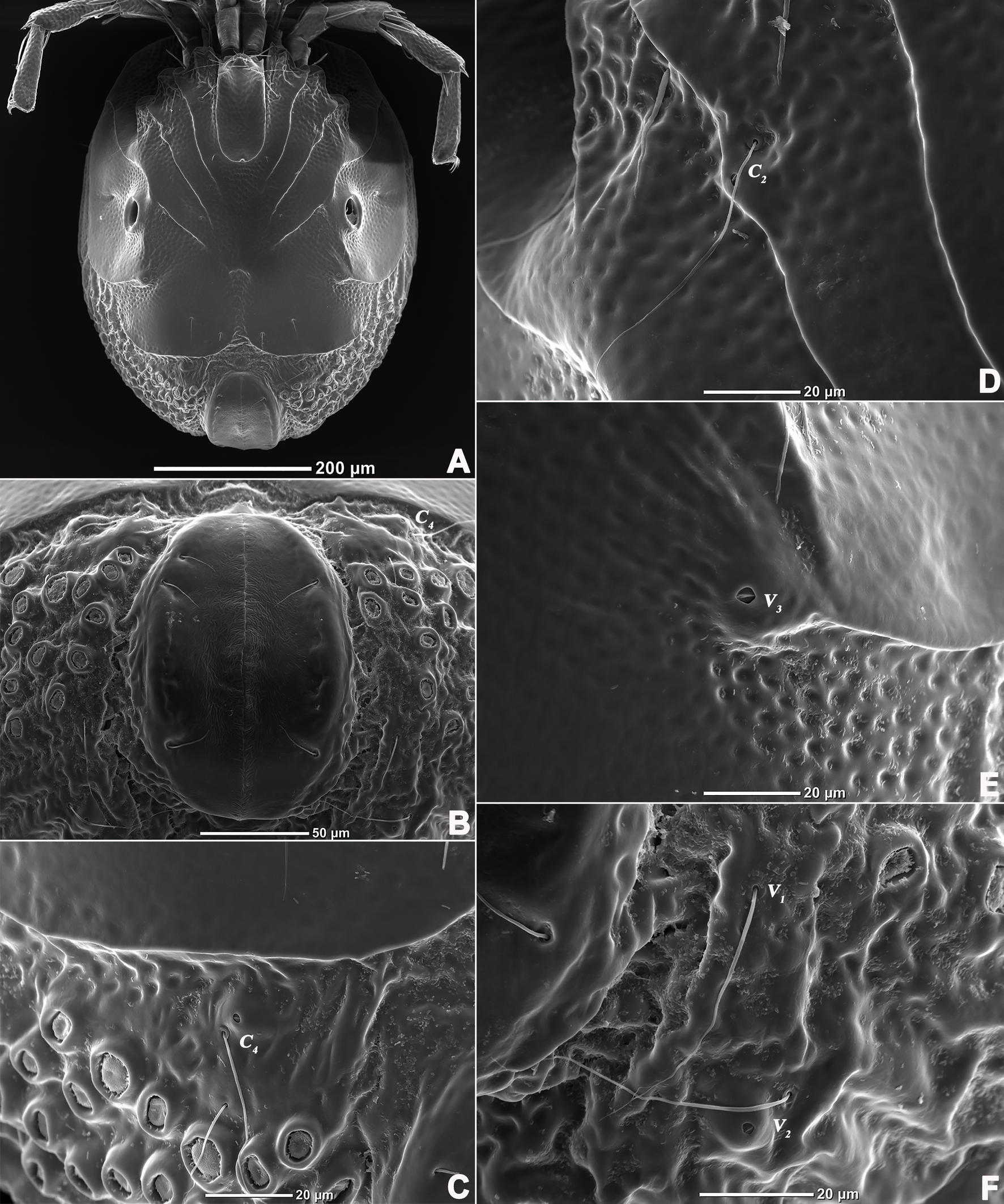
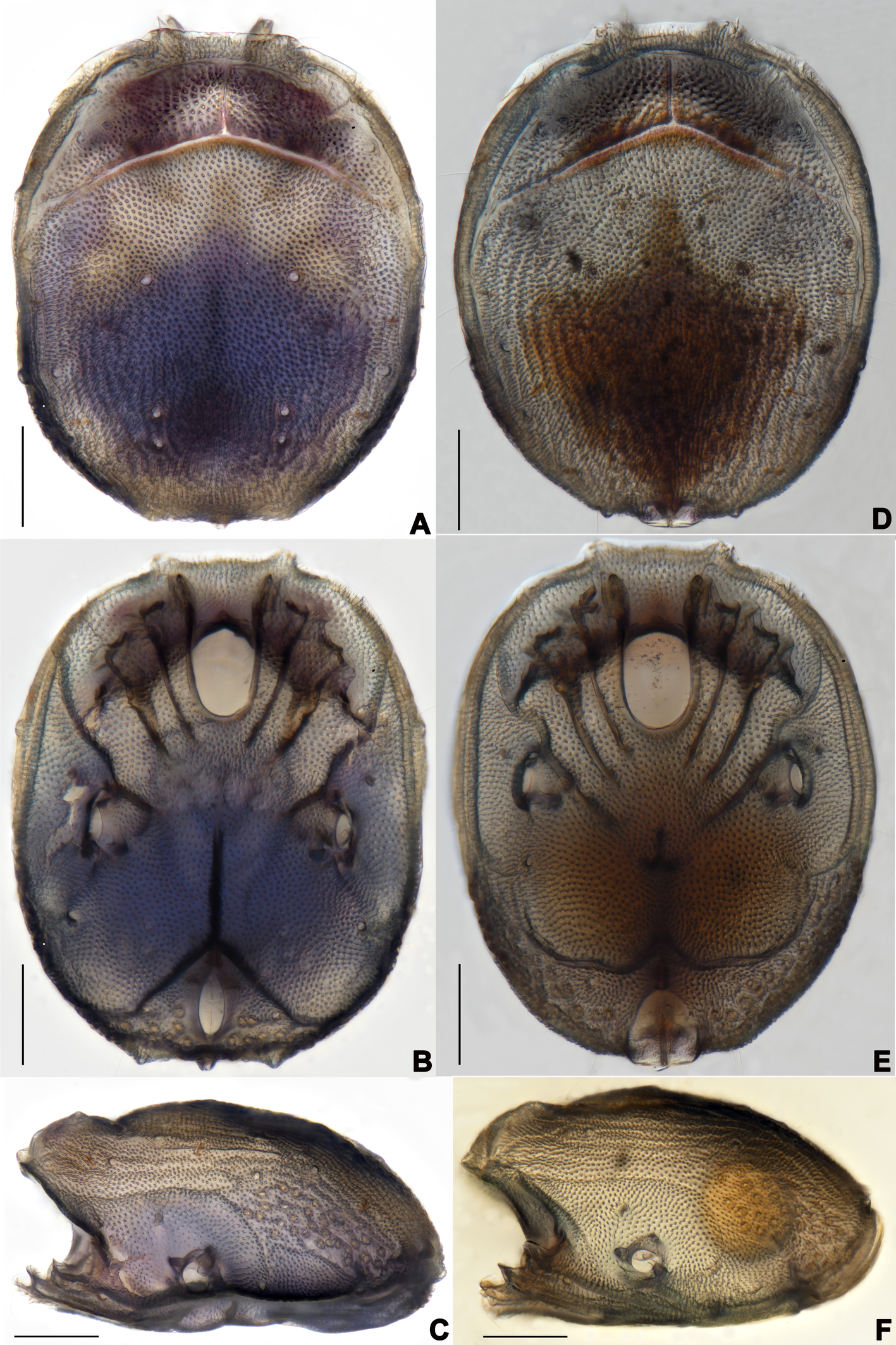
Description — Male (n=3). Idiosoma oval, slightly truncate at anterior end; dorsal and ventral shield present and this last with a conspicuous concavity (Figures 1 and 2). In dorsal view (Figures 1A and 2A): dorsal shield formed by one pair of anterior platelets and a large posterior plate; anterior platelets bearing the lateral eyes, O2 , A2 and D1 ; posterior plate bearing six pairs of glandularia (D2 –D4 , L2 –L3 and V4 ), setae of A2 , D1 –D3 , L2 –L3 all far away from glands, numerous longitudinal stripes, and two eyepatch-like violet spots, of which the anterior one is larger and covering almost 1/3 of the entire dorsal shield, and the posterior one situated at the end of the body; excretory pore protruding, located at the posterior end of the dorsal shield. In ventral view (Figures 1B and 2B): coxal plate single; tips of Cx-I not extending beyond the frontal margin; Cx-IV with distinct suture lines; suture line of Cx-III/IV ending medially to IV-L insertions and not reaching the medial line; C2 located at the posterior margin of Cx-II where suture line of Cx-II/III convex latero-posteriorly (Figure 3C); Cx-IV well extended posteriorly with round posterior margin and laterally with a ridge; IV-L basal segmental fossa without ankles. Genital field (Figure 3A) triangular and slightly depressed, fused to ventral shield; gonopore elongated and somewhat projecting, anteriorly with several genital hairs; with multiple acetabula located on both sides of gonopore to the posterolateral area of idiosoma; C4 located at either side of posterior part of genital pore (Figure 3E).
Gnathosoma with pointed dorsal apodemed, ventral apodeme blunt and almost equal in length, with two pairs of setae at the tip of the rostrum (Figure 3B). Cheliceral two-segmented, chelicera claw curved knife in shape (Figure 1G).
Palp five-segmented (Figure 4); P-1 with one dorsal seta on the inner side; P-2 with two dorsal setae on the outer side, three dorsal setae on the inner side; P-3 short, with three dorsal setae on the outer side; P-4 longest, basally and distally concave on venter, medially convex with a pair of long setae.
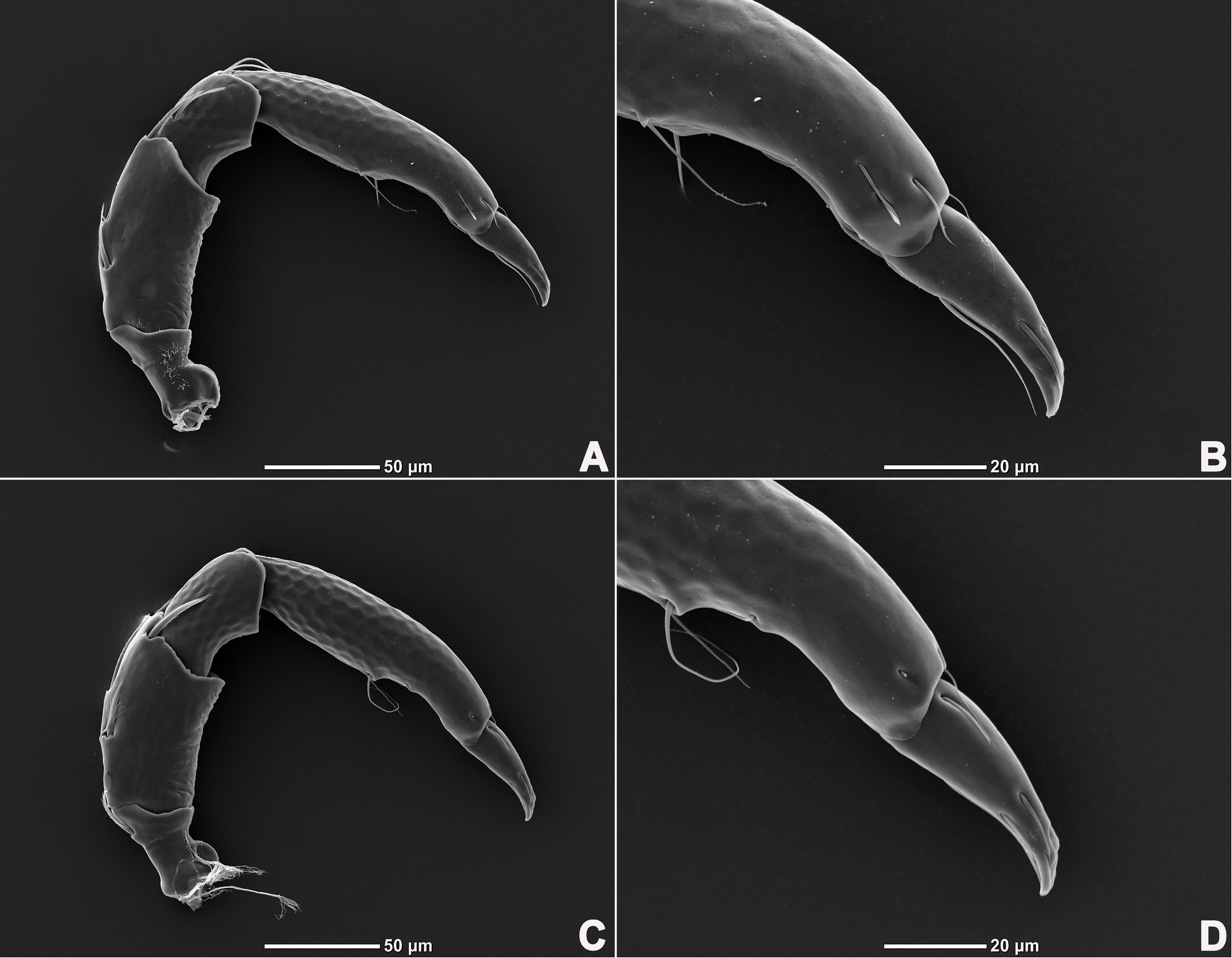
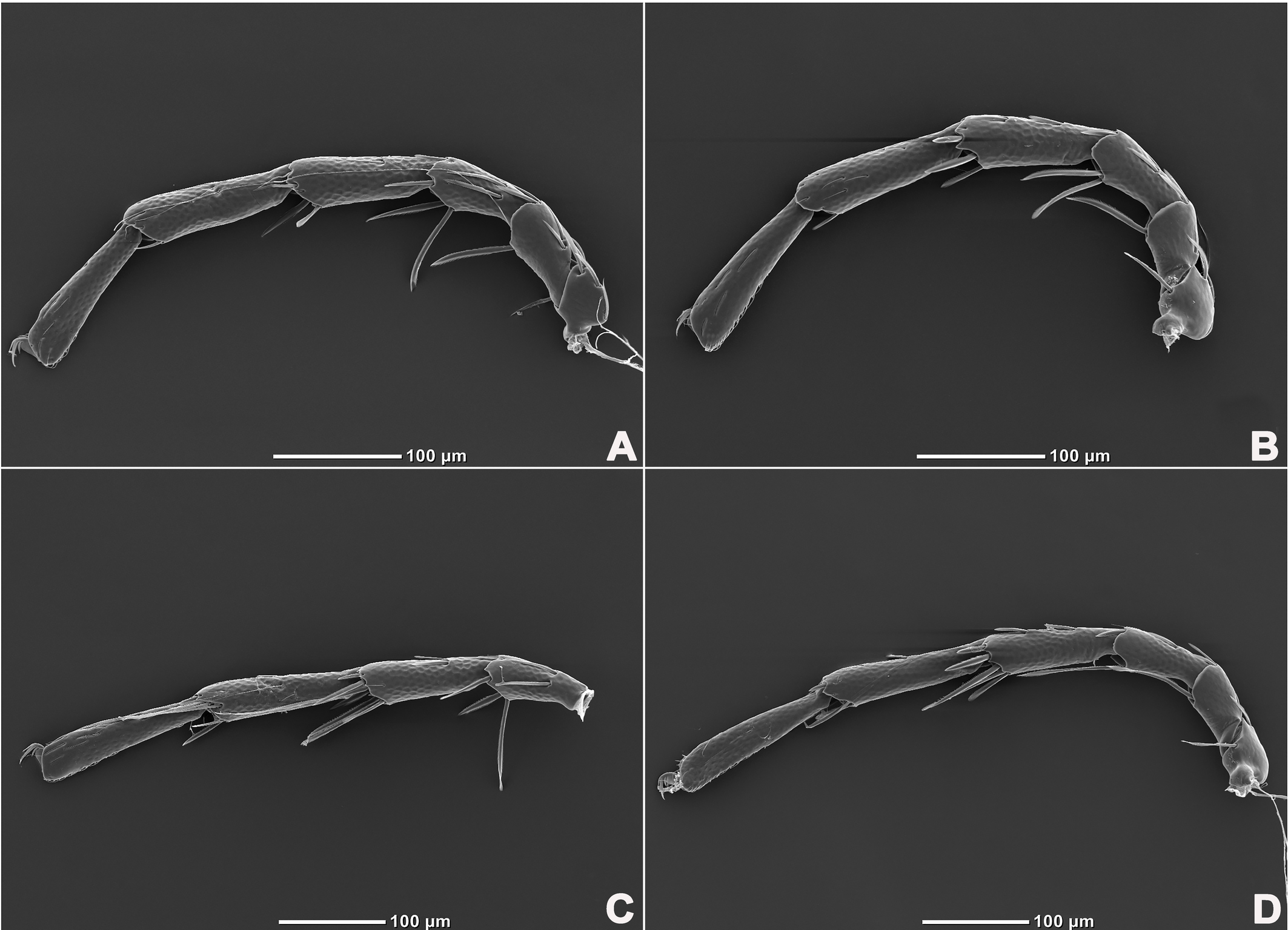
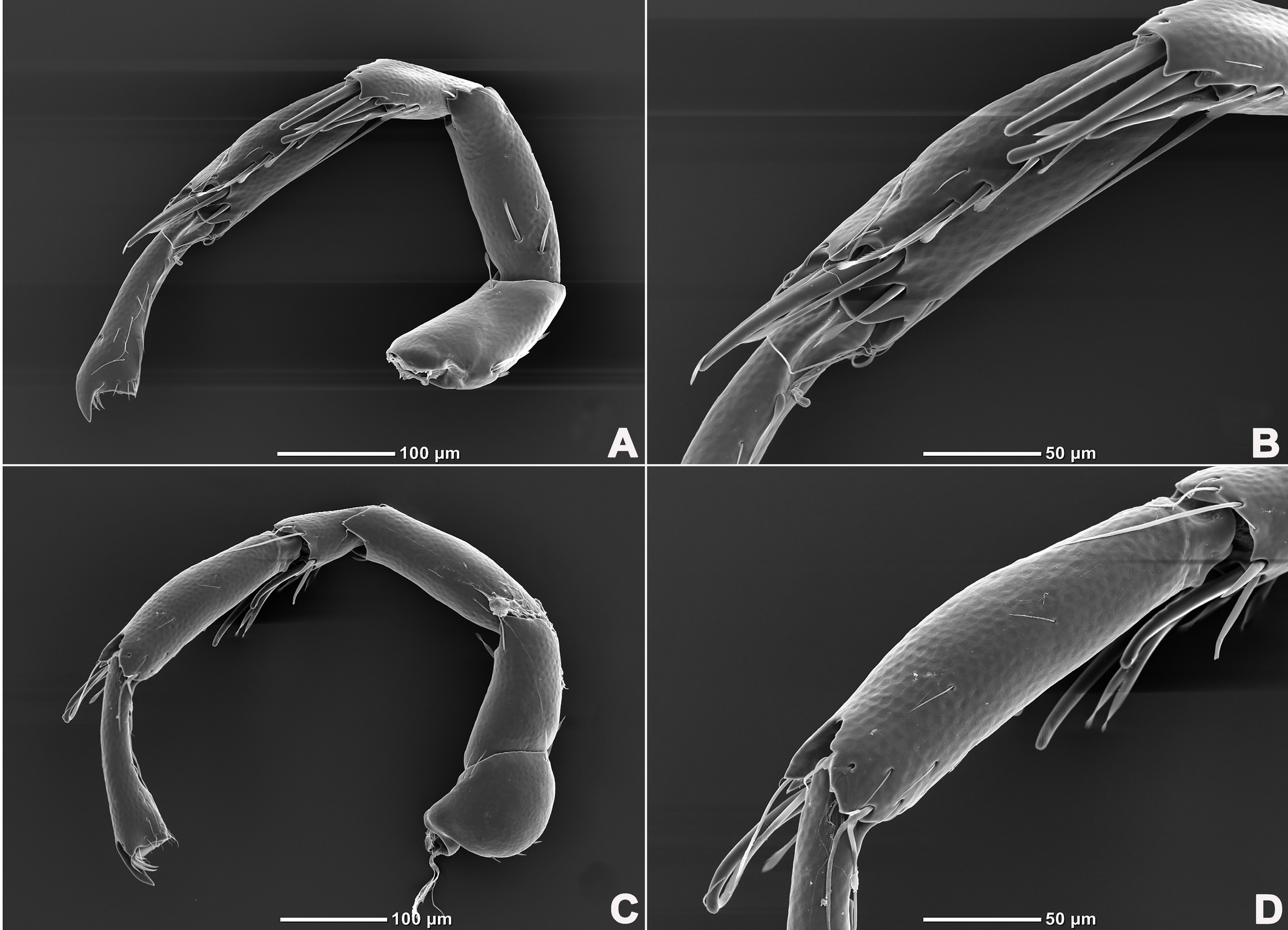
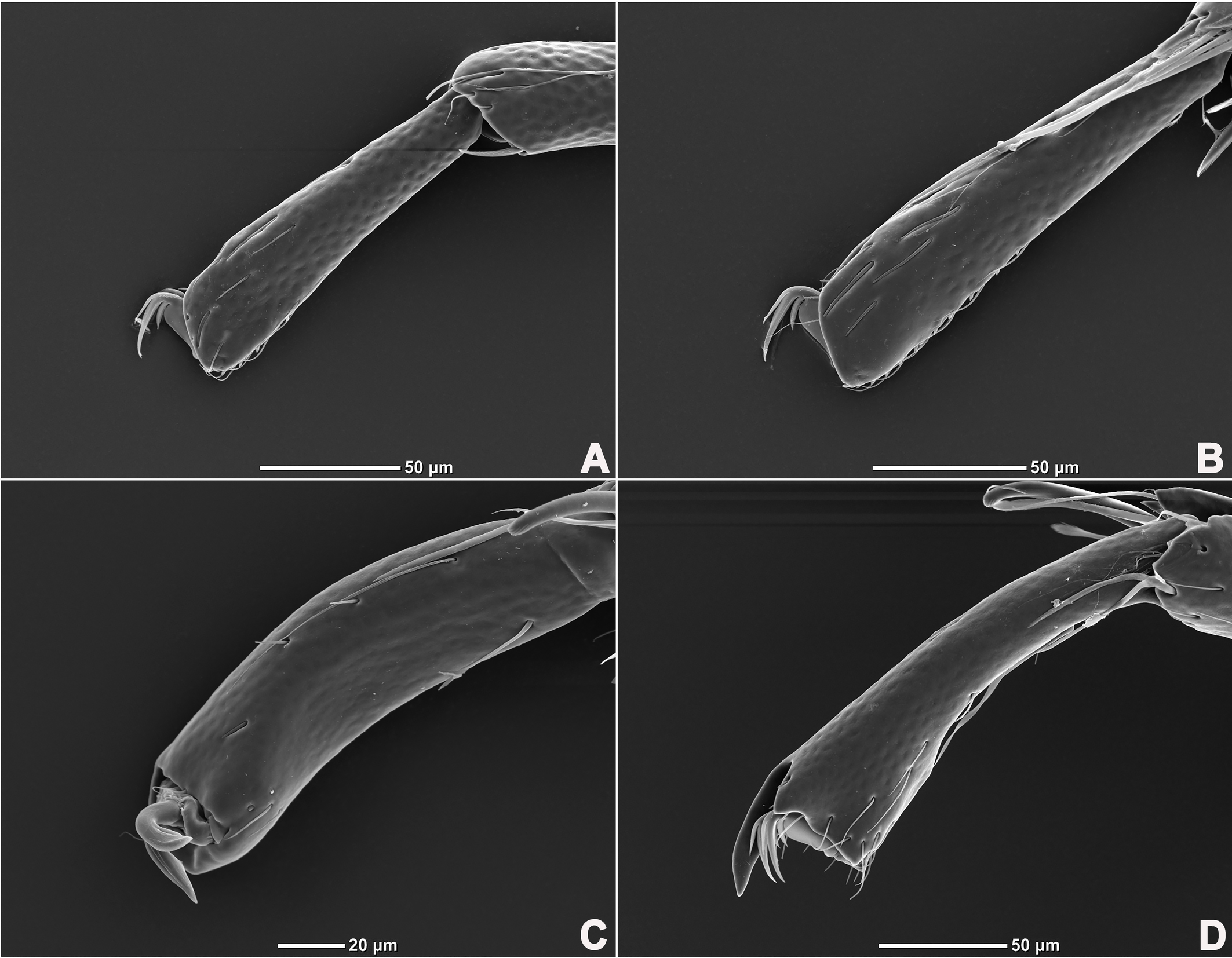
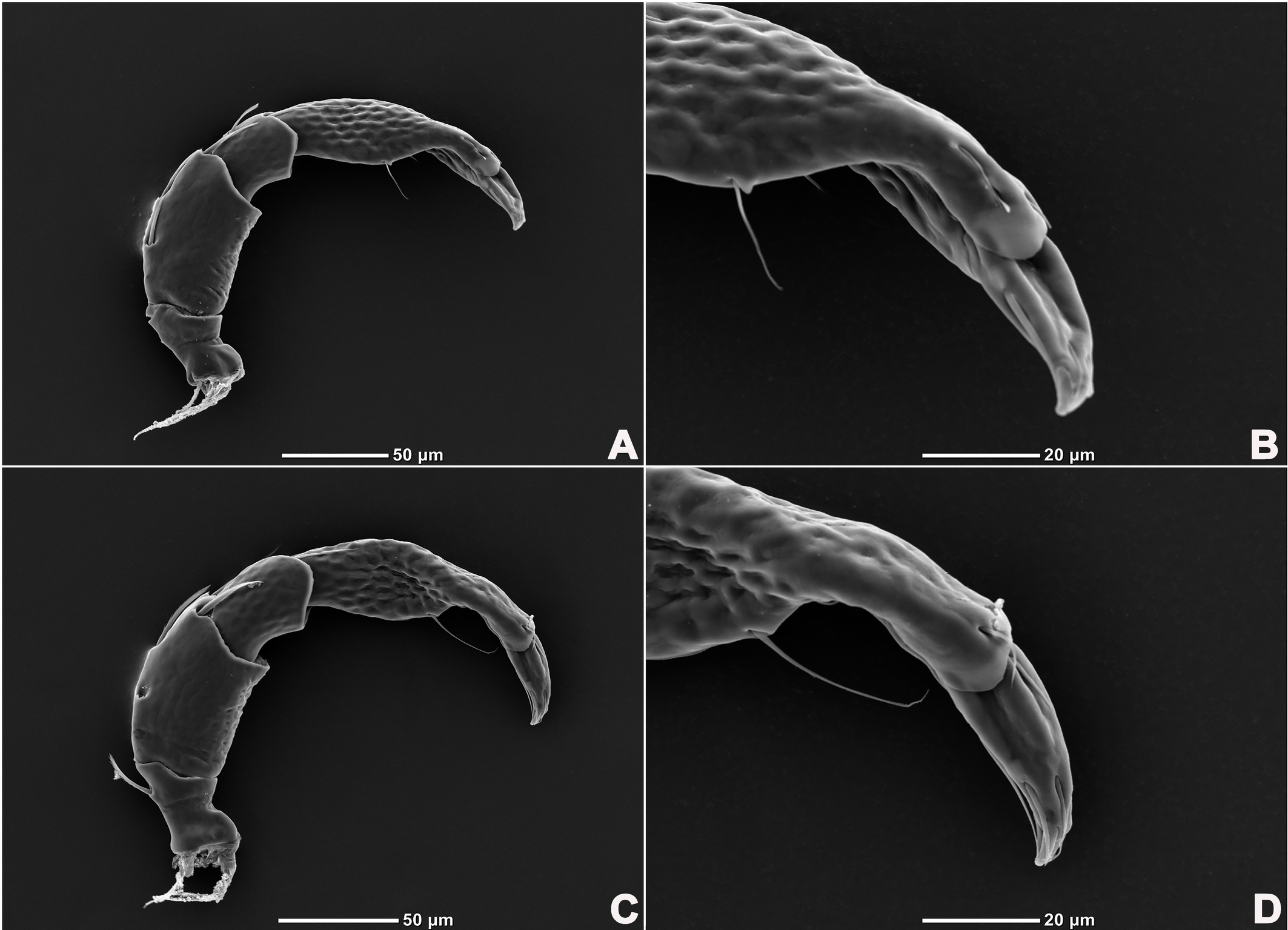
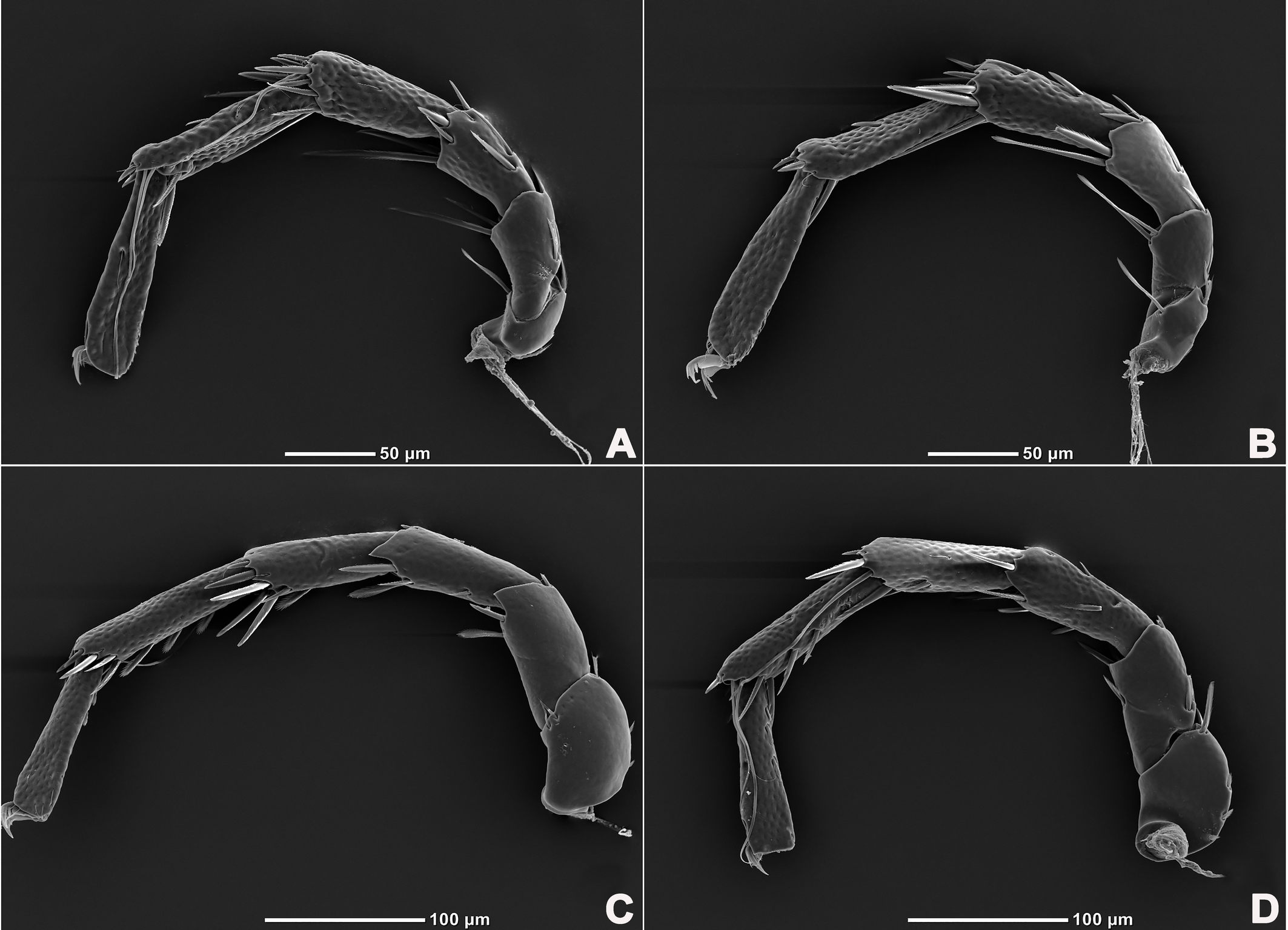
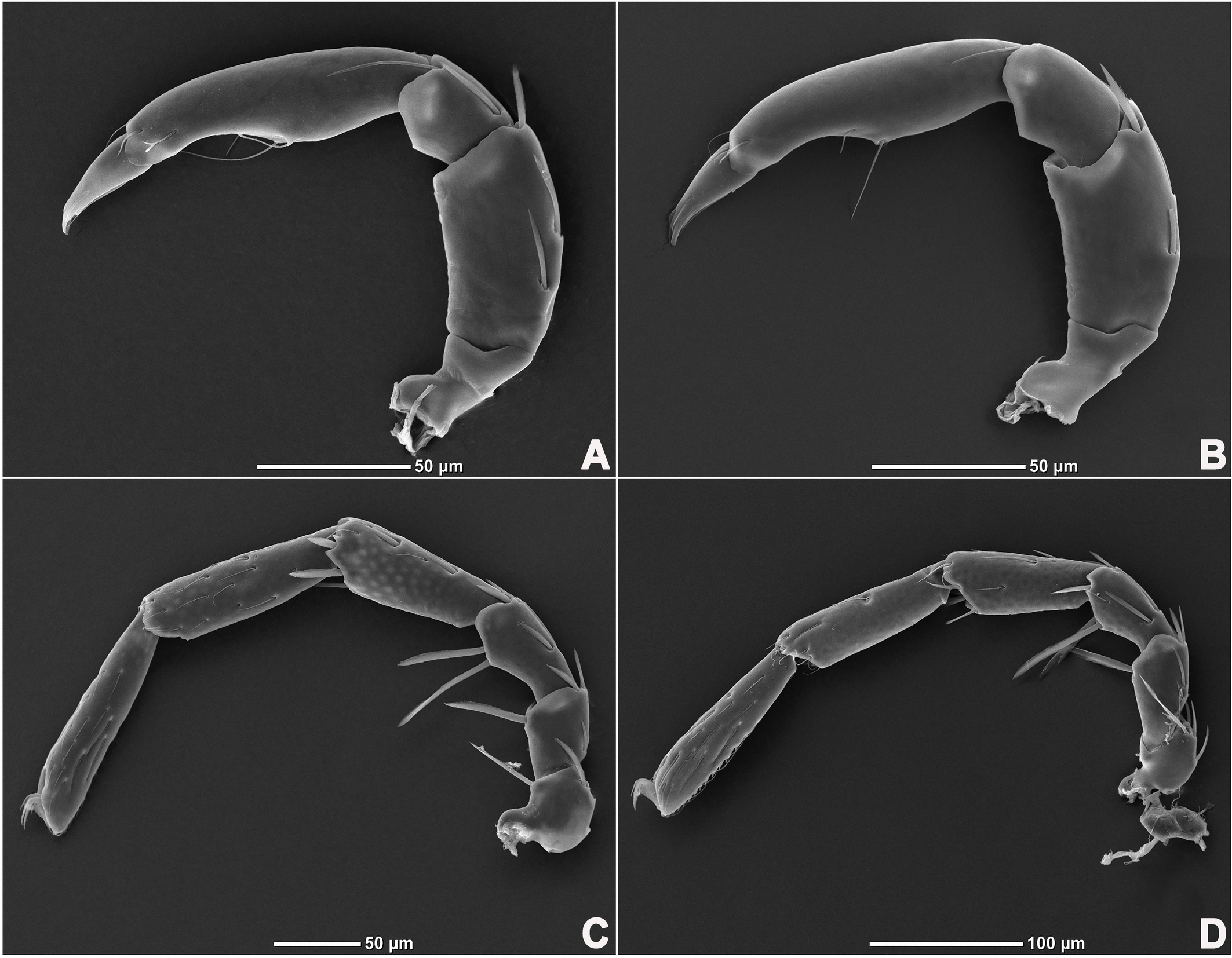
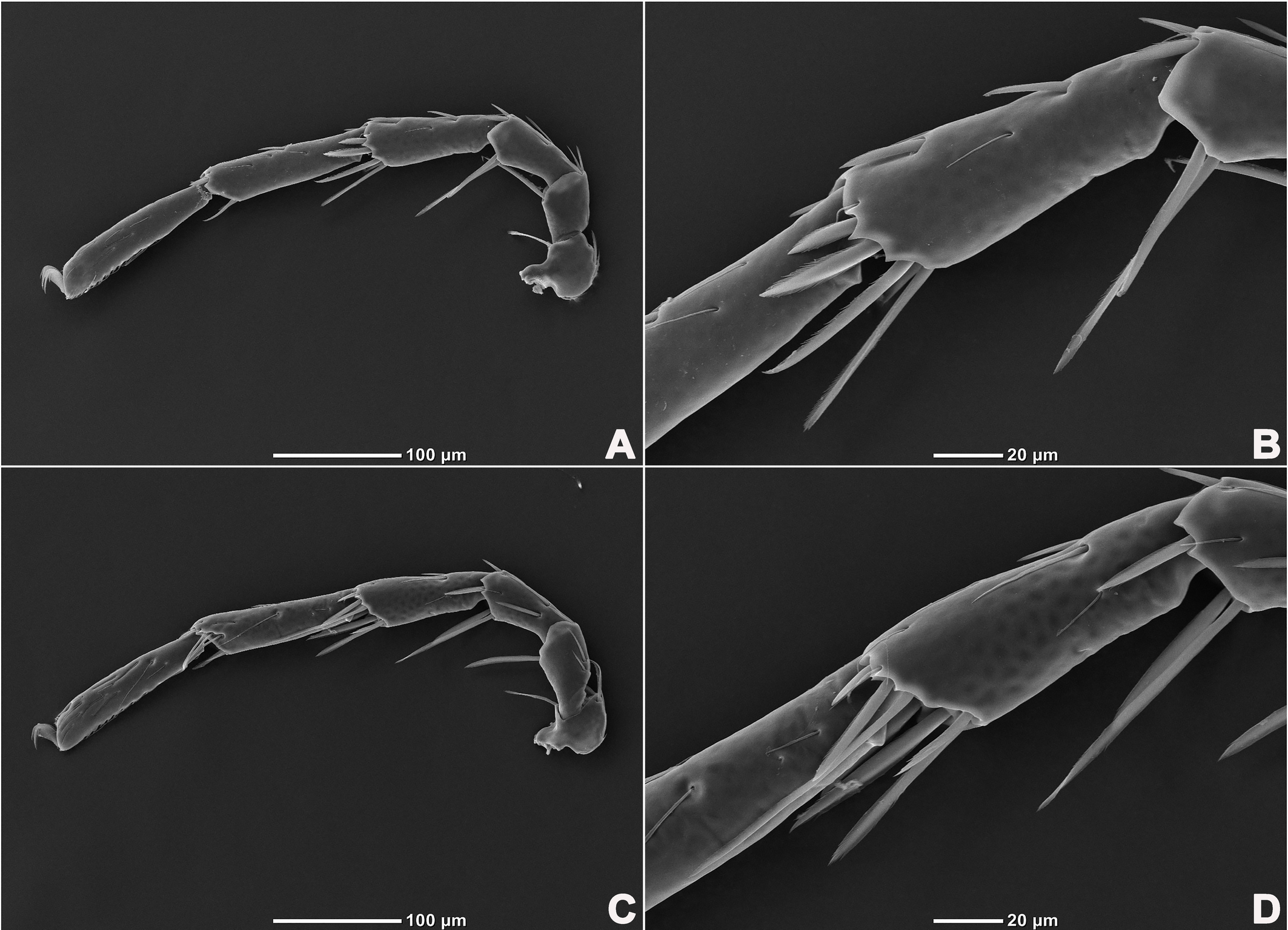
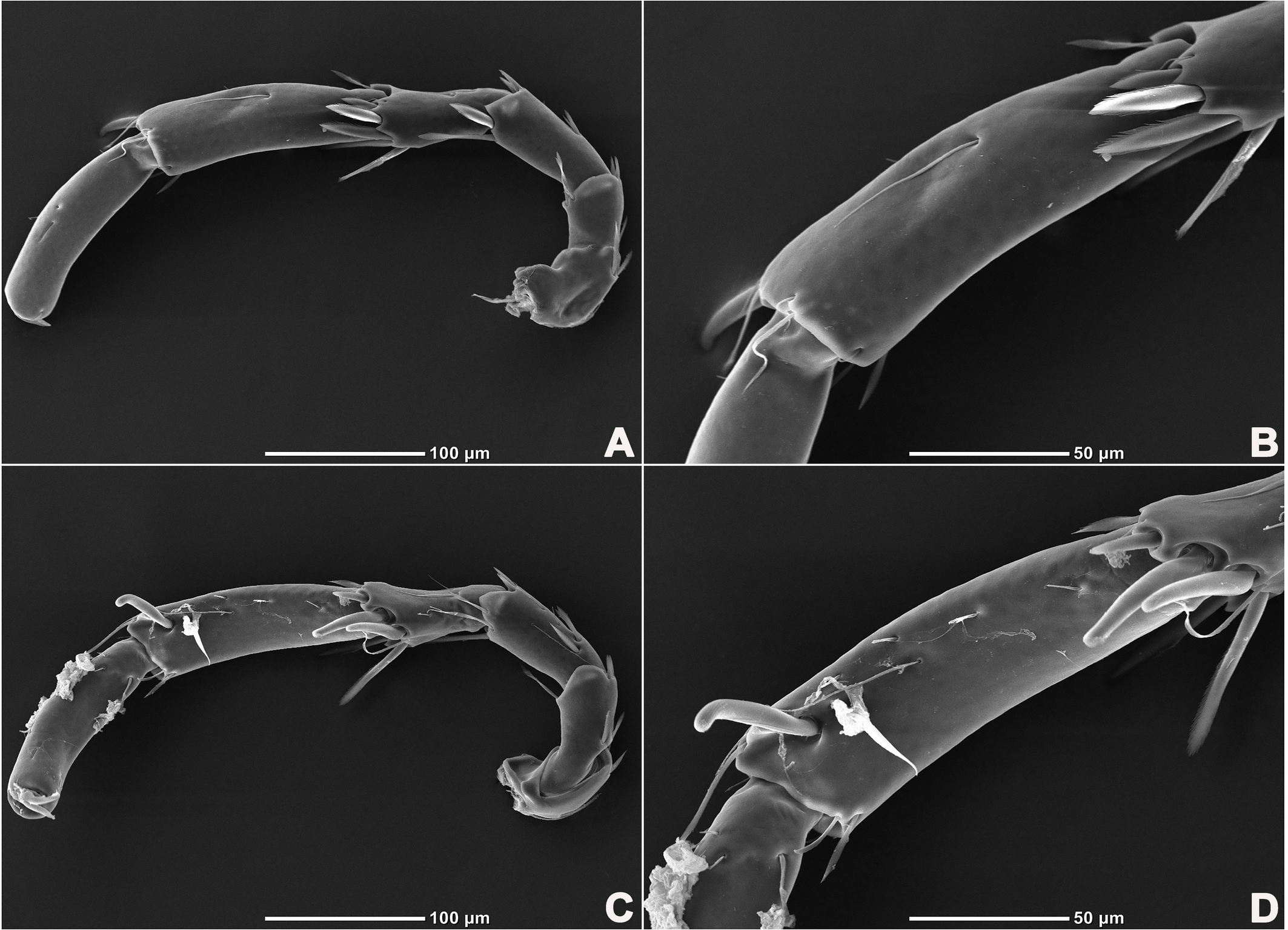
Legs six-segmented; ventral margin of I-L-6 and II-L-6 with dense small setae; claws with one dorsal and one ventral clawlet (Figures 5, 8A and 8B); III-L and IV-L exhibiting significant sexual dimorphism; III-L-4 with four large setae at the end of the outer side, two of which are blunt and cylindrical; III-L-5 ending in one large cylindrical seta curved towards the ventral surface on the outer side; III-L-6 ventrally curved, with a pair of heteromorphic claws (Figures 6 and 8C). IV-L-4 with a developed irregular protrusion and four large setae at the end of the outer side, the two last of which are blunt and cylindrical; IV-L-5 with one cylindrical seta at the end; lateral margin with a row of setae neatly arranged; IV-L-6 terminally enlarged, with a particularly well-developed dorsal process; features of the claws the same as I-L and II-L (Figures 7 and 8A).
Female (n=9). Characteristics almost similar to male, except for the following: from the lateral view, the ventral margin of idiosoma straight, without concavity (Figure 1F); genital field well developed and situated at the ventral end of the body (Figures 1E, 2E and 10A); gonopore large and terminally located (Figure 10B); V3 more anteriorly located, and lateral to the genital field (Figures 10A and 10E), and C4 located anteriorly on both sides of the gonopore (Figure 10C). III-L-4 terminally with one long swimming seta and several additional setae; III-L-5 with two long swimming setae and two short pointed setae (Figures 12A and 12B); IV-L-4 terminally with one long swimming seta and six large additional setae; IV-L-5 terminally with two long swimming setae and five large additional setae, and with a row of setae inserted on lateral margins (Figures 12C and 12D).
Measurements — Male (n=3). Idiosoma L/W 521 (521–566)/442 (442–497); anterior platelets L/W 221 (221–222)/78 (78–89), posterior plate L/W 407 (407–444)/411 (411–446); gnathosomal bay L 136 (136–152); gonopore L/W 71 (71–72)/21 (21–24), ratio 3.36 (3.0–3.36). gnathosoma vL 127 (127–139), dL 145 (145–146); chelicera total L 141 (141–168), claw L 50 (50–52), basal segment L 107 (107–123), L basal segment/claw ratio 2.14 (2.14–2.45). Palp: dL/H, dL/H ratio: P-1, 42 (42–43)/27 (27–29), 1.44 (1.44–1.59); P-2, 83 (83–84)/29 (29–30), 2.88 (2.80–2.88); P-3, 45 (42–45)/25 (25–26), 1.79 (1.61–1.79); P-4, 120 (120–124)/23 (21–23), 5.32 (5.32–5.83); P-5, 46 (46–47)/17 (17–18), 2.61 (2.61–2.79); Legs: dL of I-L-1–6: 57 (57–58), 59 (55–59), 78 (78–83), 116 (116–119), 143 (143–144), 149 (149–156); dL of II-L-1–6: 56 (56–59), 60 (60–62), 88 (88–91), 131 (131–133), 164 (161–164), 170 (166–170); dL of III-L-1–6: 78 (78–80), 61 (56–61), 94 (94–106), 102 (99–102), 195 (195–198), 182 (177–182); dL of IV-L-1–6: 98 (98–123), 134 (122–134), 192 (192–207), 103 (103–113), 230 (225–230), 198 (198–206).
Female (n=9). Idiosoma L/W 572–654/463–525; anterior platelets L/W 217–238/76–84, posterior plate L/W 455–507/430–489; gnathosomal bay L 159–171, gnathosoma vL 122–146, dL 146–158. Palp: dL of P-1–5, 47–51, 82–94, 41–45, 114–125, 44–45. Legs: dL of I-L-1–6: 47–56, 50–56, 63–70, 94–105, 113–122, 118–134; dL of II-L-1–6: 51–53, 52–57, 69–73,108–111, 131–144, 138–146; dL of III-L-1–6: 56–63, 54–55, 72–74, 107–116, 138–141, 143–145; dL of IV-L-1–6: 94–109, 76–85, 109–119, 112–124, 148–164, 136–155.
Remarks — The specimens from Henan Province were identified as S. unicucrus Yi & Jin, 2012 due to the following characteristics: (1) dorsal shield formed by one pair of anterior platelets and a large posterior plate; (2) III-L-4 with two blunt and cylindrical setae at the end; (3) III-L-5 terminally with one large cylindrical seta curving toward the ventral surface; (4) III-L-6 with a pair of heteromorphic claws; (5) IV-L-4 with a developed irregular protrusion and four large setae at the end; (6) IV-L-5 with well-developed protrude collar and three heteromorphic setae at the end, the nearest to the back is cylindrical and much thicker than the other two; (7) IV-L-6 with a particularly well-developed dorsal process (Yi and Jin 2012).
The species was firstly discovered in Anhui Province, which is the neighbor of Henan Province. Meanwhile, the species was redescribed in this paper, due to the lack of some important characteristic information, e.g. the glandularia, the patches of the large posterior dorsal plate, etc.
Distribution — China (Anhui, Henan).
Sinaxonopsis hainanensis sp. nov.
ZOOBANK: 4AB02675-9B86-42FF-87EE-203ABED00AC4 ![]()
(Figures 13–21)




















Habitat — Stream with numerous rocks and aquatic plants at the bottom (Figure 13).
Material examined — Holotype: male, Diaoluoshan Nature Reserve, Benhao Town, Lingshui Li Autonomous County, Hainan Province, P.R. China (18°67′08″ N, 109°89′43″ E, 270 m a.s.l.), collected by Hai-Tao Li, Yu-Lin Zheng, and Yu-Hao Zhang, 13-IV-2023. Slides No. HN-AT-2023041301. Paratypes: 3♂, the same data as the holotype, Slide No. HN-AT-2023041302–2023041304; 2♂, 1♀, Qinglv waterfall, Yaga Scenic Area, Bawangling National Nature Reserve, Qicha Town, Changjiang Li Autonomous County, Hainan Province, P.R. China (19°08′48″ N, 109°12′39″ E, 463 m a.s.l.), collected by Hai-Tao Li, Yu-Lin Zheng, and Yu-Hao Zhang, 23-IV-2023. Slides No. HN-AT-2023042301–2023042303.
Diagnosis — Dorsal shield formed by one pair of anterior platelets and a large posterior plate; anterior platelets bearing the lateral eyes, O2 , A2 and D1 ; posterior plate bearing six pairs of glandularia, D2 –D4 , L2 –L3 and V4 . Male. Idiosoma blue to purple in color, with an irregular bluish-purple pattern, genital field triangular; genital pore L/W 2.86–3.07; IV-L-5 ventrally with a row of plumose setae; IV-L-6 without distinct processes. Female. Idiosoma brownish to yellow, with an irregular brownish-yellow pattern on the posterior dorsal plate; genital field well developed and placed at the ventral end of the body.
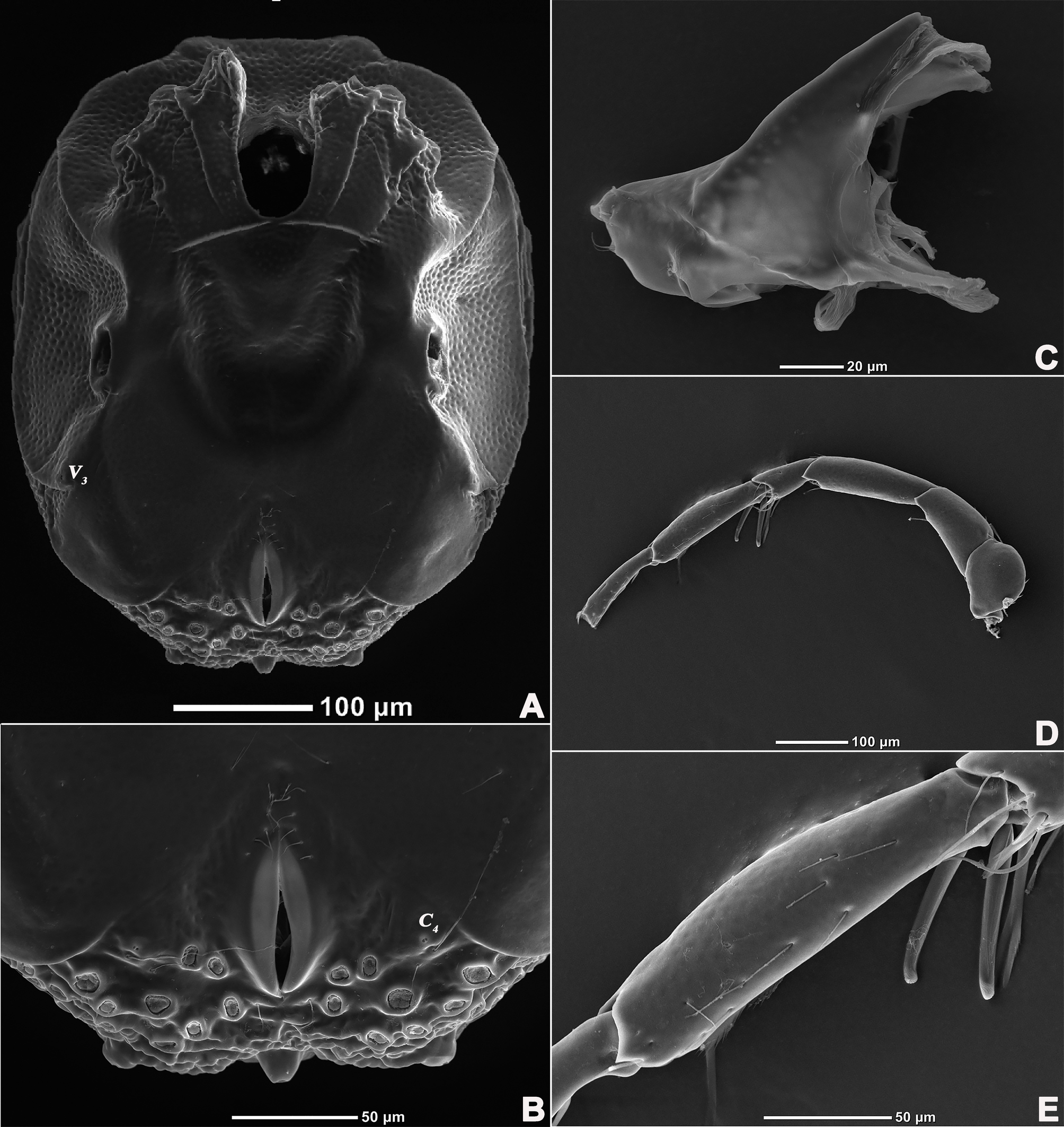
Description — Male (n=6). Idiosoma oval, blue-purple color, slightly truncate at anterior end; dorsal and ventral shields present (Figures 14 and 15). In dorsal view (Figures 14A, 15A and 16A): dorsal shield formed by one pair of anterior platelets and a large posterior plate; anterior platelets bearing the lateral eyes, O2 , A2 and D1 ; posterior plate bearing six pairs of glandularia, D2 –D4 , L2 –L3 and V4 (Figure 16A), setae of A2 , D1 –D3 , L2 –L3 all far away from glands, numerous longitudinal stripes, and with an irregular bluish-purple pattern, almost 2/3 of the entire large posterior plate (Figure 15A); excretory pore protruding, located at the posterior end of dorsal shield. In ventral view (Figures 14B, 15B and 16B): coxal plate single; tips of Cx-I not extending beyond frontal margin; Cx-IV with distinct suture lines, suture line of Cx-III/IV ending medially to IV-L insertions and not reaching the medial line; C2 located at the posterior margin of Cx-II where suture line of Cx-II/III slightly curved; Cx-IV well extended posteriorly with round posterior margin and laterally with a ridge; basal segmental fossa of IV-L without ankles (Figure 18A). Genital field triangular and slightly depressed, fused to ventral shield; gonopore elongated and somewhat projecting, anteriorly with several genital hairs; multiple acetabula located on both sides of gonopore in the posterolateral area of idiosoma (Figure 18B); C4 located at either side of posterior part of genital pore.
Gnathosoma with pointed dorsal apodeme, ventral apodeme blunt and almost equal in length; two pairs of setae at the peak of the rostrum (Figure 18C). Chelicera two-segmented; cheliceral claw curved knife shaped (Figure 1D).
Palp five-segmented (Figures 19A and 19B); P-1 dorsally with one seta socket on the outer side, without seta; P-2 with three dorsal setae on the outer, two setae on the inner sides; P-3 short, with two dorsal setae on the outer side; P-4, basally and distally concave on venter, medially convex with a pair of long setae.
Legs six-segmented; ventral margin and dorsal margin of I-L-6 and II-L-6 with a dense patch of small setae (Figures 20B and 20D), claws with one dorsal and one ventral clawlet (Figures 19C, 19D and 20); III-L and IV-L exhibiting significant sexual dimorphism; III-L-4 with three large setae on the end of the inner side, two of which are cylindrical; III-L-5 terminally with one ventrally curved large seta on the outer side; III-L-6 without distinct specific setae, with a pair of heteromorphic claws (Figures 16E and 21); IV-L-4 with a well-developed projection bearing three large setae on the inner side (Figure 18E), the two last of which are blunt and slightly curved; IV-L-5 ending with one angular projection, ventrally with a row of plumose setae and one large cylindrical seta at the end; IV-L-6 without distinct processes, with a claw same as that of I-L and II-L (Figures 16F and 18D).
Female (n=1). Characteristics almost similar to male, except for the following: idiosoma brownish to yellow; from the lateral view, the ventral margin of idiosoma slightly projecting (Figures 14C and 15D); genital field well developed at the ventral end of the body; gonopore large and terminally located (Figure 15E); V3 positioned further anterior to the genital field; C4 located anterolateral to the tip of the gonopore (Figure 17B). III-L without heteromorphic setae; claws same as in I-L and II-L (Figure 17C); IV-L-4 ending without prominence, with one long swimming hair; IV-L-5 with one angular prominence at the end, two long swimming setae and one ventrally curved large seta, and one row of tiny setae on venter (Figure 17D).
Measurements — Male (n=6). Idiosoma L/W 508 (478–534)/424 (408–442); anterior platelets L/W 198 (193–214)/73 (70–73), posterior plate L/W 405 (384–425)/388 (384–419); gnathosomal bay L 142 (137–144); gonopore L/W 71 (68–74)/25 (22–25), ratio 2.86 (2.86–3.22). gnathosoma vL 129 (125–129), dL 142 (1431–144); chelicera total L 171 (148–171), claw L 52 (42–52), basal segment L 114 (106–130), L basal segment/claw ratio 2.19 (2.19–3.09). Palp: dL/H, dL/H ratio: P-1, 44 (43–45)/23 (23–24), 1.91 (1.76–1.91); P-2, 79 (76–79)/28 (26–28), 2.73 (2.73–2.99); P-3, 37 (37–46)/23 (23–31), 1.61 (1.30–1.61); P-4, 106 (106–109), 5.91 (5.91–6.02); P-5, 42 (41–42)/11 (11–16), 3.63 (2.70–3.63); Legs: dL of I-L-1–6: 47 (44–47), 43 (43–53), 67 (67–72), 111 (105–114), 116 (116–140), 128 (128–144); dL of II-L-1–6: 54 (47–55), 62 (49–62), 79 (72–82), 113 (108–125), 144 (135–145), 139 (139–153); dL of III-L-1–6: 72 (72–82), 53 (53–60), 82 (78–89), 99 (83–99), 151 (151–171), 147 (142–149); dL of IV-L-1–6: 108 (107–118), 115 (113–118), 185 (179–190), 89 (89–108), 178 (175–191), 145 (134–159).
Female (n=1). Idiosoma L/W 513/422; anterior platelets L/W 204/58, posterior plate L/W 443/394; gnathosomal bay L 151. gnathosoma vL 134, dL 148. Palp: dL of P-1–5, 45, 81, 43, 105, 44; Legs: dL of I-L-1–6: 48, 43, 61, 90, 111, 118; dL of II-L-1–6: 59, 52, 67, 103, 131, 137; dL of III-L-1–6: 55, 56, 69, 109, 124, 129; dL of IV-L-1–6: 93, 73, 97, 103, 137, 121.
Remarks — Due to the morphology of the dorsum (formed by one pair of anterior platelets and a large posterior plate), genital field (with multiple acetabula) and palp (P-4 with a pair of long setae) of the specimens from Hainan, China, the new species, S. hainanensis sp. nov., resembles S. laosensis Pešić & Smit, 2016 from Laos. However, the new species differs from S. laosensis in the following aspects: (1) S. hainanensis sp. nov. (L/W 508 (478–534)/424 (408–442)) smaller than S. laosensis (L/W 581/443); (2) S. hainanensis sp. nov. body bluish-purple, but colourless in S. laosensis; (3) IV-L-5 venter with a row of plumose setae in S. hainanensis sp. nov., but only one large pennate seta and two slender setae in S. laosensis.
Etymology — The name ''hainanensis'' refers to the type location, where the specimens were collected.
Distribution — China (Hainan).
Discussion
Glandularia arrangement on the mites body surface is a peculiar feature of each species (Lundblad 1930; Jin and Li 1997; Wiles 1997) and it is considered to indicate a possible way of evolution in this phylogenetic lineage (Shatrov et al. 2019). However, some species of Aturidae lack the glandularia V1 , the gland of D4 , therefore, this study revealed that S. unicucrus and S. hainanensis sp. nov. had the glandularia V1 and the gland D4g. Besides, this study confirms that this genus has 16 pairs of glandularia, i.e. A1 , A2 , C2 , C4 , D1 –D4 , L1 –L4 , V1 –V4 , and two pairs of ocularia (O1 , O2 ), which is consistent with the common patterns in water mites.
It is important to acknowledge that previous studies on Aturidae may have overlooked certain features due to the small size of the specimens and outdated experimental equipment and techniques. Therefore, future studies should focus on glandulars, particularly D4 and V1 , to clarify their presence, absence, and position. This will provide a basis for further exploration in phylogenetic research of Aturidae.
Patterns (presence or absence, shape, color and size) of large posterior dorsal plate is an important characteristic in species identification of Aturidae, and has been widely used, e.g. the genus of Axonopsis, Hexaxonopsis, Kongsbergia, Vicinaxonopsis, etc. (Asadi et al. 2010; Pešić et al. 2010; Radwell and Smith 2012; Pešić and Gerecke 2014; Pešić and Smit 2016; Smit 2016). Among the two species illustrated in this paper, eyepatch-like violet spots of the lager posterior plate in S. unicucrus and irregular bluish-purple pattern in S. hainanensis sp. nov. are embedding into the chitin exoskeletons and maintain intraspecific stability and interspecies differences. Therefore, the patterns (presence or absence, shape, color and size) of the large posterior plate could be used as discriminative features for species identification of Sinaxonopsis. At the same time, we should pay more attention to illustrate it in detail, to provide enough materials for future taxonomic studies of Aturidae. The present results have not only provided potentially effective characters for species identification in Sinaxonopsis, but also materials for phylogenetic analysis based on glandularia.
Acknowledgements
We are indebted to Yu-Lin Zheng (College of Pharmacy, Dali University) and Yu-Hao Zhang (Institute of Entomology, Guizhou University) for providing the specimens. This research was supported by Guizhou Provincial Science and Technology Projects (Qiankehe Pingtai Rencai-GCC [2022] 029-1) and the National Nature Science Foundation of China (32260125).
References
- Asadi M., Pešić V., Etemadi I. 2010. A revised survey of water mites (Acari: Hydrachnidia) from Iran: new synonyms and descriptions of three new species. Zootaxa, 2628(1): 43-55. https://doi.org/10.11646/zootaxa.2628.1.3
- Gerecke R. 2014. Studies on European species of the water mite family Aturidae Thor (Acari: Hy-drachnidia). Zootaxa, 3841(1): 1-46. https://doi.org/10.11646/zootaxa.3841.1.1
- Gu X.-Y., Jia L., Jin D.-C., Guo J.-J. 2020. Four new species of Torrenticola (Acari, Hydrachnidia, Torrenticolidae) from Northeastern China. Zootaxa, 4779(2): 245-259. https://doi.org/10.11646/zootaxa.4779.2.6
- Jin D.-C. 1997. Hydrachnellae-morphology systematics a primary study of Chinese fauna. Guiyang: Guizhou Science and Technology Publishing House, pp. 356. (in Chinese)
- Jin D. -C., Li L. -S. 1997. On glandularia morphology of water mites (Acari, Actinedida, Hydrachnellae) and evolutionary theory of the mite soma. Acta Entomologica Sinica, 43(3): 231-246.
- Kim I.-H., Chung, K. -S. 1993. Water Mites of the Genus Aturus (Acarina: Aturidae) in Korea. The Korean Journal of Zoology, 36: 329-341.
- Kim I.-H., Chung, K. -S. 1995. Water mites of the genus Aturus (Acarina: Aturidae) from the eastern side of Korea, including five new species. The Korean Journal of Zoology, 38: 269-285.
- Li H.-T., Jin D.-C., Guo J.-J. 2022. Acucapito hainanensis sp. nov., the first record of the family Acucapitidae Wiles, 1996 (Acari, Hydrachnidia) from China. Acarologia, 62(1): 250-261. https://doi.org/10.24349/h7mp-06fx
- Lundblad O. 1930. Über die Anatomie von Arrhenurus mediorotundatus und die Hautdrüsen der Arrhenurus-Arten. Zeitschrift für Morphologie und Ökologie der Tiere, 17(1/2): 302-338. https://doi.org/10.1007/BF00406261
- Pešić V., Gerecke R. 2014. Water mites from caves of the Ha Giang province, northern Vietnam (Acari: Hydrachnidia). Zootaxa, 3774(4): 367-380. https://doi.org/10.11646/zootaxa.3774.4.5
- Pešić V., Smit H. 2016. New records of water mites from Southeast Asia (Acari: Hydrachnidia) with the description of two new genera and 12 new species. Acarologia, 56(3): 393-433. https://doi.org/10.1051/acarologia/20162251
- Pešić V., Smit H., Gerecke R., Di S. -A. 2010. The water mites (Acari: Hydrachnidia) of the Balkan peninsula, a revised survey with new records and descriptions of five new taxa. Zootaxa, 2586(1): 1-100. https://doi.org/10.11646/zootaxa.2586.1.1
- Radwell A. -J., Smith I. -M. 2012. North American members of the reticulata-like species group of the water mite genus Kongsbergia (Acari: Hydrachnidiae: Aturidae). Zootaxa, 3540(1): 1-49. https://doi.org/10.11646/zootaxa.3540.1.1
- Shatrov A. -B. 2008. Organization of unusual idiosomal glands in a water mite, Teutonia cometes (Teutoniidae). Experimental and Applied Acarology, 44: 249-263. https://doi.org/10.1007/s10493-008-9148-8
- Shatrov A. -B., Soldatenko E. -V., Stolbov V. -A., Smirnov P. -A., Petukhova, O. -A. 2019. Ultrastruc-ture and functional morphology of dermal glands in the freshwater mite Limnochares aquatica (L., 1758) (Acariformes, Limnocharidae). Arthropod structure & development, 49: 85-102. https://doi.org/10.1016/j.asd.2018.11.010
- Smit H. 2016. The water mite family Aturidae Thor, 1900 from Southeast Asia (Acari: Hydrachnidia) with the description of one new genus and 14 new species. Acarologia, 56(3): 341-365. https://doi.org/10.1051/acarologia/20162248
- Smit H. 2017. New species of aturid water mites from southern Africa (Acari: Hydrachnidia: Aturidae). Zootaxa, 4273(2): 287-295. https://doi.org/10.11646/zootaxa.4273.2.9
- Smit H. 2020. Water mites of the world, with keys to the families, subfamilies, genera and subgenera (Acari: Hydrachnidia). Leiden, Monografieën van de Nederlandse Entomologische Vereniging, pp. 774.
- Wiles P.-R. 1991. Watermites (Hydrachnidia: Aturidae) of the genus Albia Thon from Asia and Australasia with descriptions of eleven new species. Insect Systematics & Evolution, 22(4): 465-487. https://doi.org/10.1163/187631291X00264
- Wiles P.-R. 1997. The homology of glands and glandularia in the water mites (Acari: Hydrachnidia). Journal of Natural History, 31(8): 1237-1251. https://doi.org/10.1080/00222939700770671
- Yi T.-C., Jin D.-C. 2012. Description of Sinaxonopsis unicucrus sp. nov. et gen. nov. (Acari: Hydrachnidia: Aturidae) from Anhui Province, China. International Journal of Acarology, 38(5): 402-409. https://doi.org/10.1080/01647954.2012.657802



2024-02-03
Date accepted:
2024-04-05
Date published:
2024-04-23
Edited by:
Auger, Philippe

This work is licensed under a Creative Commons Attribution 4.0 International License
2024 Jia, Lan; Li, Hai-Tao and Guo, Jian-Jun
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



