Predation of Raoiella indica (Acari: Tenuipalpidae) by Stethorus tridens Gordon (Coleoptera: Coccinellidae)
Monteiro, Vaneska Barbosa  1
; Matheus, Maria Luíza Tavares
1
; Matheus, Maria Luíza Tavares  2
; França-Beltrão, Girleide Vieira
2
; França-Beltrão, Girleide Vieira  3
; Melo, José Wagner da Silva
3
; Melo, José Wagner da Silva  4
; de Lima, Debora Barbosa
4
; de Lima, Debora Barbosa  5
and Gondim-Júnior, Manoel Guedes Côrrea
5
and Gondim-Júnior, Manoel Guedes Côrrea  6
6
1✉ Departamento de Agronomia – Entomologia, Universidade Federal Rural de Pernambuco, Recife, PE, Brasil.
2Departamento de Agronomia – Entomologia, Universidade Federal Rural de Pernambuco, Recife, PE, Brasil.
3Departamento de Agronomia – Entomologia, Universidade Federal Rural de Pernambuco, Recife, PE, Brasil.
4Departamento de Zoologia, Universidade Federal de Pernambuco, Centro de Biociências, Recife, PE, Brasil.
5Departamento de Zoologia, Universidade Federal de Pernambuco, Centro de Biociências, Recife, PE, Brasil.
6Departamento de Agronomia – Entomologia, Universidade Federal Rural de Pernambuco, Recife, PE, Brasil.
2024 - Volume: 64 Issue: 1 pages: 202-212
https://doi.org/10.24349/1hy9-av19Original research
Keywords
Abstract
Introduction
The red palm mite (RPM), Raoiella indica Hirst (Acari: Tenuipalpidae), is a species native to the eastern hemisphere (Beard et al. 2012), that has experienced rapid territorial and host expansion (Gondim Jr. et al. 2012), with a record of plague in 33 countries by the year 2010 and 46 countries by 2022 (Taylor 2022). RPM have been reported in several hosts (Gondim Jr. et al. 2012; Gómez-Moya et al. 2017; Souza et al. 2020; Sousa Neto et al. 2021; Souza et al. 2023), causing damage to plants of agricultural importance in the families Arecaceae and Musaceae (Navia et al. 2015). Several studies have been conducted on the biological control of this pest (Mendes et al. 2018; Filgueiras et al. 2020; Coelho et al. 2020; Lira et al. 2021). This method is ideal for controlling this mite in the medium- and long-term (Gondim Jr. et al. 2012; Navia et al. 2015).
Arthropod biological control agents, including insects and mites, are associated with RPM (Carrillo et al. 2012; Contreras-Bermúdez et al. 2017; Fidelis et al. 2022). Among these biological control agents, predatory insects of the Stethorus genus, such as Stethorus keralicus Kapur, Stethorus parcempunctatus Puttarudriah & ChannaBasavanna and Stethorus tetranychi Kapur (Puttaswamy and Rangaswamy 1976; Gupta 2001; Joshi et al. 2023). These predators consume phytophagous mites, mainly those from the superfamily Tetranychoidea (Hodek and Honěk 1996; Biddinger et al. 2009). Several Stethorus species have been used in biological control programs against tetranychids (Kundoo and Khan 2017). Stethorus punctum LeConte was a successful example of the biological control of mites in the 1970s in the USA, reducing the cost of using acaricides (Asquith and Hull 1973). In Brazil, the beetle Stethorus tridens Gordon preys on tetranychids on plants of the Solanaceae (Britto et al. 2008) and Euphorbiaceae (Costa et al. 2020) families and is being considered a potential biological control agent.
Promising biological control agents can be evaluated through functional and numerical responses, which are key factors that regulate population dynamics in predator-prey systems (Holling 1959). The functional response focuses on changes in individual's consumption rate and food (prey) density (Solomon 1949), while the numerical response focuses on the changes in predator to prey densities (Hassel 1978). Through mathematical and statistical models, it is possible to characterize the type of functional response of the predator (Moffat et al. 2020) and compare its effectiveness with that of other biological control agents (Eini et al. 2023). The functional response involves several parameters, such as handling time (Th) and attack rate (ɑ) (Holling 1959). The handling time corresponds the time spent by the predator to identify, stalk, kill, consume and digest a prey, whereas the attack rate corresponds to proportion of prey captured by each predator per time unit (Holling 1959).
The concept of functional responses is fundamental for studies on biological control, invasive species management, and conservation biology (Dick et al. 2014; Mendes et al. 2018; Coblentz and De Long 2020), making it the first step in assessing whether natural enemies are effective in controlling pests (Lin et al. 2022). In areas recently colonized by RPM, there is an increasing interest in cost-effective control strategies. In South America, RPM attack banana and coconut crops, which are mostly produced by small producers. The predator S. tridens is frequently associated with crops in Brazil, including those in the same region invaded by RPM (Brito et al. 2008; Costa et al. 2020). Therefore, we hypothesized that S. tridens may prey on RPM not only due to the eating habits of the Stethorini tribe (acarophagous) but also due to their association with this pest in India, as reported by Joshi et al. (2023). Thus, studying the potential of generalist native control agents, such as S. tridens, in controlling RPM may indicate prospects for natural and sustainable biological control of pests.
The objective of this study was to investigate S. tridens preferences among different developmental stages of RPM and estimating the functional and numerical responses of S. tridens over its most preferred prey-stage of RPM. Additionally, the prey preference of S. tridents was examined between eggs of Tetranychus evansi Baker & Pritchard (its natural prey) and eggs of RPM through two-choice and non-choice experiments.
Material and methods
Obtaining and rearing insects and mites
Stethorus tridens were obtained from individuals collected on the campus of Universidade Federal Rural de Pernambuco (UFRPE) (8°01′S; 34°56′W) in Solanum americanum (Solanaceae) Miller plants infested with T. evansi and on Musa spp. (Musacea) plants infested with Tetranychus abacae Baker & Pritchard. The confirmation of the species was made through the description of the identification key described by Gordon and Chapin (1983). Adult S. tridens were placed in wooden cages (100 × 60 × 80 cm) covered with voile fabric containing a plant of S. americanum infested with T. evansi. Pupae of S. tridens were collected from rearing cages and kept in a Petri dish with a transparent cylindrical plastic lid (9 cm diameter) and a circular opening covered with voile fabric to allow for the passage of air. After emergence, the adults were sexed and used in experiments. Banana seedlings infested with only RPM were kept inside cages similar to those used for the rearing of S. tridens.
Prey stage preference of Stethorus tridens to Raoiella indica
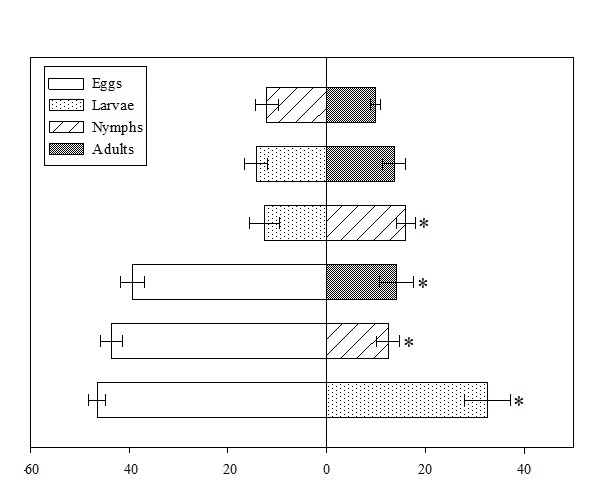
The preferences of S. tridens to RPM developmental stages were evaluated using two-choice tests. The individuals of S. tridens used in this experiment were obtained from S. americanum plants infested with T. evansi. Experimental units were made with acrylic containers (3 cm diameter) containing polyethylene foam (0.5 mm thick), filter paper (2.5 cm diameter), and banana leaf disk (2.5 cm diameter), superimposed in this sequence. The foam was moistened to maintain the leaf turgor. Then, 50 RPM individuals from two developmental stages (egg, larva, nymph, or adult) were randomly placed in all possible combinations, (six combinations in total: egg vs. larva, egg vs. nymph, egg vs. adult, larva vs. nymph, larva vs. adult, and nymph vs. adult). The inactive (egg) and active (larva, nymph, and adult) forms of RPM were transferred to each experimental unit using a brush. After transfer, when the active forms did not move, they were replaced. For the RPM eggs, a blank test was carried out without the use of the predator to assess the physical integrity as a function of handling; however, no correction was necessary, since the mortality rate was fewer than 5%. Subsequently, an adult female S. tridens of unknown age was confined to each experimental unit. Fifteen replicates were performed per combinations, and each female S. tridens represented a replicate. Finally, the experimental units were closed with a lid that had a circular opening closed with voile fabric to allow for the passage of air and kept at 27 °C ± 1.0 °C, 75 ± 10% R.H. and 12 h of photophase. After 24 h, prey consumption was counted. The average consumption among the items offered was compared using the χ2 test (α = 0.05), using the Proc FREQ of SAS (SAS Institute 2008).
Functional and numerical response of Stethorus tridens
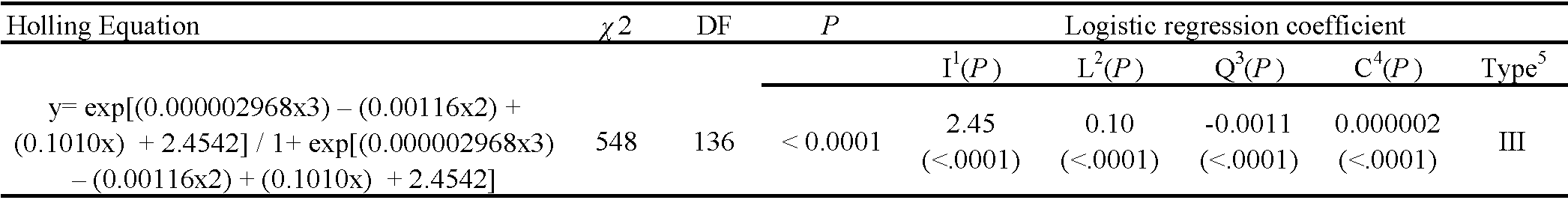
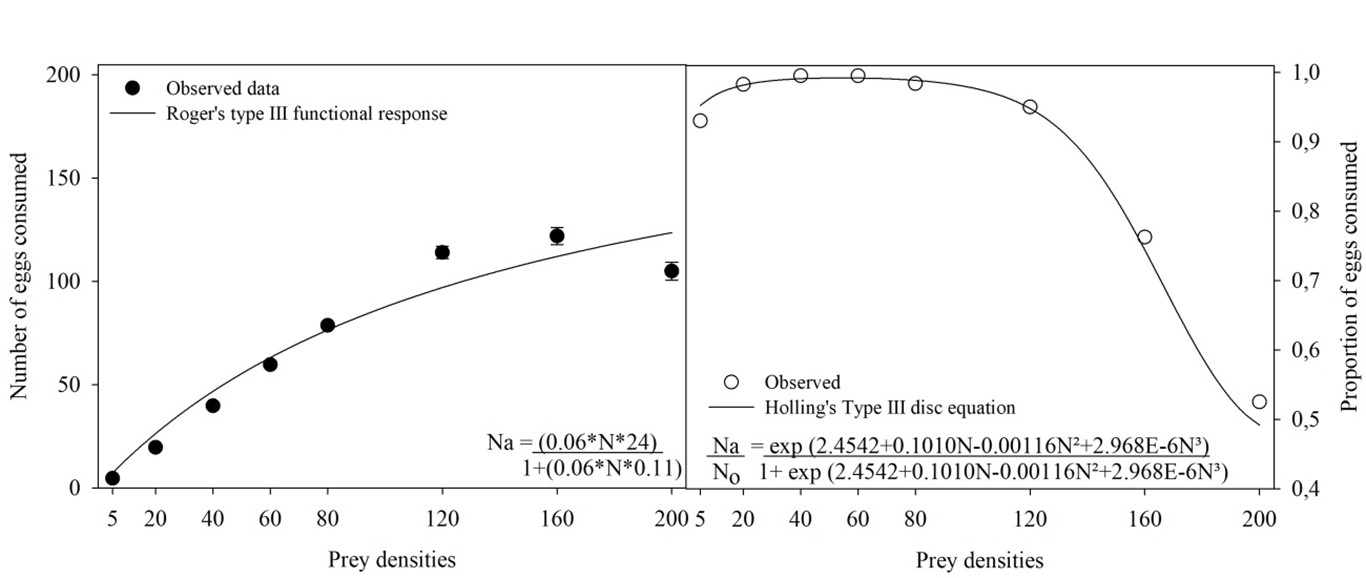
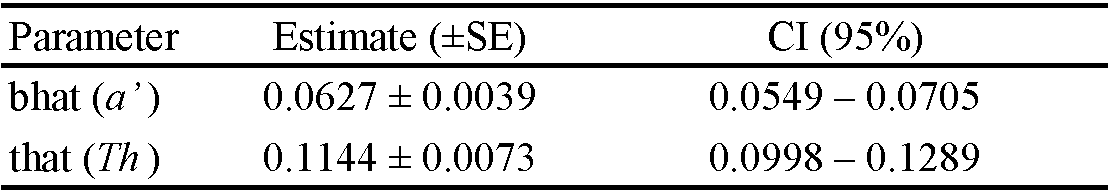
The RPM eggs were transferred from the rearing unit to the experimental unit and randomly distributed at densities of 5, 10, 20, 40, 60, 120, 160, or 200 eggs/experimental unit. Adult females of S. tridens, of unknown age were removed from rearing and kept under food deprivation for 4 h before being confined to the experimental units. After 24 h of confinement, the number of eggs consumed and deposited was counted. To determine the type of functional response, a logistic regression of the proportion of prey consumed as a function of the density of prey offered was performed using the protocol described by Juliano (1993) (using Proc. CATMOD of SAS, SAS Institute 2008). Based on the obtained equation, the significance of the regression coefficient and sign of the linear coefficient were observed to determine the type of functional response. When not significant, the linear coefficient indicated a type I response (linear increase in prey consumption as a function of density); when negative and significant, the response was type II (rapidly increasing prey consumption as a function of density, passing through reduction until stabilization, plateau); and when positive and significant, the response was type III (accelerated increase in prey consumption with prey density, presenting a sigmoidal response) (Holling 1959). Because the experiments were conducted with prey depletion, the attack rate (a) and handling time (Th ) were estimated using the equation \(Ne = N_0 \{1 - exp [ (d + bN_0) (T_h N_e – T) / (1 + cN_0) ] \}\) (Rogers 1972), using nonlinear regression with the least squares method (using ProcNLIN of SAS, SAS Institute 2008), where Ne is the number of prey consumed during time T, N0 is the initial density of the prey, a is the coefficient of the attack rate (ratio of prey captured by the predator to search time), and Th is the handling time (the time required for the predator to identify, stalk, kill, consume, and digest the prey); and b, c, and d are constants of the function that relate a and N0 in the type III functional response: \(a = (d + bN_0) / (1 + cN_0)\).
Prey preference of Stethorus tridens by RPM or Tetranychus evansi eggs
The individuals of S. tridens used in this experiment were obtained from Musa spp. plants infested with T. abacae. Initially, a confinement test (non-choice test) was conducted to evaluate the consumption of S. tridens on RPM or T. evansi eggs. For this purpose, the same experimental unit used in previous experiments was used. In each unit, 120 eggs were offered. A female subject deprived of food for 4 h was placed in the experimental unit. Twenty females were subjected to this test for each treatment (RPM or T. evansi eggs). Each female represents a repetition. After 24 h, consumption was counted. The experiment was maintained at 27 °C ± 1.0 °C, 75 ± 10% RH. and 12 h of photophase. The results were subjected to normality (Kolmogorov test) and homogeneity (Bartlett test) tests using Proc TTEST of SAS (SAS Institute 2008). Two-choice tests were conducted using the same experimental units and abiotic conditions as those used in the confinement test (non-choice test). Under the leaf fragments of the experimental unit, 60 eggs of RPM and 60 eggs of T. evansi were randomly placed. A female S. tridens subject to 4 h of food deprivation was placed in each experimental unit, and 20 females were tested. After 24 h, the eggs consumed within the experimental unit of each prey species were counted. Mean consumption among prey was compared using the χ2 test (α = 0.05) using the Proc FREQ of SAS (SAS Institute 2008).
Results
Prey stage preference of Stethorus tridens to Raoiella indica
Stethorus tridens showed preference for eggs over others prey-stages of RPM (Figure 1) (χ2 > 37; df = 1; P < 0.0001) and preference for nymphs over larva (χ2 = 6; df = 1; P = 0.0138). In the other combinations there was no difference (χ2 = 0.2; df = 1; P = 0.6248 to larva vs adult; χ2 = 3; df = 1; P = 0.0613 to nymph vs adult). Thus, egg was the stage of development chosen to perform the functional and numerical response of S. tridens.


Functional and numerical response of Stethorus tridens
Stethorus tridens females showed a type III functional response since the linear coefficient was positive and significant (Table 1). The consumption rate increased rapidly at intermediate densities of 20, 40, 60, and 80 eggs, with average consumption of 20, 40, 60, and 79 eggs, respectively (Figure 2). The attack rate (a) and handling time (Th ) were 2.65 and 0.20, respectively (Table 2). Despite the high predation observed, this was not enough to stimulate predator oviposition.






Prey preference of Stethorus tridens by RPM or Tetranychus evansi eggs
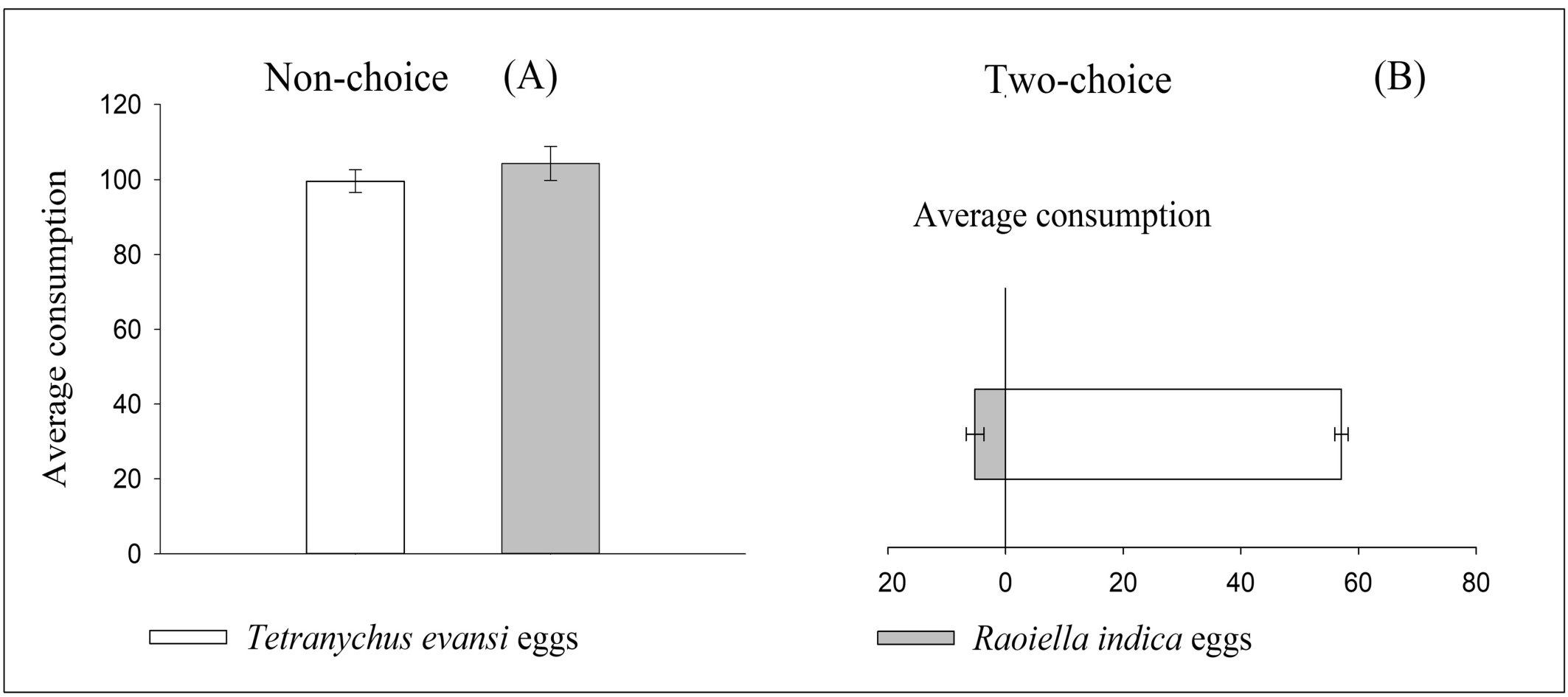
In the confinement test (non-choice test), S. tridens had an average consumption of approximately 100 and 105 eggs in 24 h of T. evansi and RPM, respectively, showing no significant (t33.2 = –1.28; P = 0.2111). When submitted to the two-choice experiment, S. tridens preferred T. evansi (57 eggs) over RPM (5 eggs) (χ2 = 865; df = 1; P < 0.0001) (Figure 3).


Discussion
Stethorus tridens preyed on all development stages of RPM, with a marked preference for eggs over others stages of the prey. This characteristic has been observed in several species of the genus Stethorus (Ullah 2000; Ali et al. 2016; Costa et al. 2017). For example, Costa et al. (2017) found that S. tridens has a feeding preference for the eggs and larvae of the mite Tetranychus bastosi Tuttle, Baker & Sales. Taghizadeh et al. (2018) showed that the consumption rate of the predator Stethorus gilvifrons Mulsant was higher in the eggs of Tetranychus urticae Koch, followed by protonymphs and females. Ragkou et al. (2004) discovered that there was a higher consumption of T. urticae eggs compared to the predation of adult mites by Stethorus punctillum Weise. Predation on prey with higher nutritional value increases the fitness of the predator for greater survival and fecundity (Eubanks and Denno 2000). The egg, besides being a motionless phase, is easy to handle (Costa et al. 2017) and ingest compared to nymphs and adults, which have a sclerotized cuticle (Ganjisaffar and Perring 2015). In addition, RPM eggs are smaller compared to the other stages of development, making the egg preferable for the predator. Egg weight is also lower than that of other developmental stages; therefore, predators need higher egg consumption to obtain the same amount of nutrients (Ganjisaffar and Perring 2015). The preference for eggs shown by S. tridens is desirable for pest management, since the predator will kill eggs before they hatch and begin to feed (Filgueiras et al. 2020).
The functional response of S. tridens to RPM eggs followed the type III model. According to Holling (1965), predators exhibiting type III response are the only ones with potential to regulate the density of their prey. Later, it was reinforced by Fernández-Arhex and Corley (2003) and also by Pervez and Omkar (2005). At low prey densities such predators have lower efficiency allow the prey population to recover from population bottlenecks, avoiding local extinctions, whereas at high densities, the speed of consumption increase helping to avert outbreak (Bruzzone et al. 2022). Although it is not common among coccinelids, the type III functional response was also detected for others coccinelids, such as such as S. gilvifrons feeding on Tetranychus turkestani Ugarov & Nikolski (Sohrabi and Shishehbor 2007), Eriopis connexa Germar and Nephus includens Kirsch when preying upon aphids (Sarmento et al. 2007; Bayoumy 2011) and Nephus arcuatus Kapur when preying upon mealybug (Zarghami et al. 2014). It was also detected for S. tridens feeding on a different prey (eggs from T. bastosi) (Costa et al. 2017).
In the present study, we observed that RPM eggs were not able to support the S. tridens reproduction, even in the highest density. Consequently, the numerical response could not be calculated. A similar observation was made when S. tridens was fed T. bastosi eggs (Costa et al. 2017). Some prey consumed in the field by coccinellids may not be nutritionally suitable for its development and reproduction (Hagen 1987; Hodek and Honěk 1996). When the prey has poor quality, the developmental time, oviposition, fecundity and fertility may be affected. Preys consumed by coccinellids are often classified as essential or alternative (Hodek 1973; Hodek and Honěk, 1996; Evans et al. 1999). Prey that supports pre-imaginal development with low mortality and enables an elevated reproduction is considered essential, while that which serves only as an energy source and is able to prolong survival is considered as alternative (Hodek 1973; Hodek and Honěk, 1996; Evans et al. 1999). Thus, the results presented here suggest that RPM eggs may be considered as alternative prey to S. tridens.
Although tetranychid eggs were slightly larger than tenuipalpids, the average consumption in the feedlots was virtually the same. However, when tetranychid eggs were offered with tenuipalpid eggs, S. tridens preferred tetranychid eggs. In this case, the difference in egg size may have influenced the food preference of S. tridens, because when the predator is faced with two types of prey, it selects one that is more likely to maximize its net energy gain and fitness (Stephens and Krebs 1986). Thus, larger prey is more likely to be selected by predators because of their higher energy content (Soares et al. 2004). Another factor that may have influenced the preference of S. tridens is the adaptability or previous experience of feeding this predator tetranychids, and some studies have shown that subsequent feeding behavior can be influenced by previous feeding experience (Zarghami et al. 2014), including acarophagous coccinellids (Houck, 1986). This is evident from the association of S. tridens with mites of the Tetranychidae family in the field (Britto et al. 2008; Costa et al. 2017, 2020). This shows that in the field, coccinellids may not be efficient in controlling RPM if another pest (Tetranychidae) is present. However, a predator's preference may change according to prey availability and adaptation to the new prey (Murdoch 1969; Jaworski et al. 2013). This preference can influence the population dynamics of low-preferred species within the same niche (Holt and Lawton 1994).
The average consumption of eggs by S. tridens was much higher in the confinement than in the experiment with freedom of choice. This observation is not only true for S. tridens; other predators, such as Typhlodromus (Anthoseius) bagdasarjani Wainstein & Arutunjan, 1967 (Bazgir et al. 2020) and Orius minutus Linnaeus (Song et al. 2018) show the same behavior when fed mites. The number of preys consumed by a predator depends on its physiological, nutritional, and morphological requirements (Soares et al. 2004). Prey, in turn, differs in terms of energy content and costs associated with capture and consumption (Eubanks and Denno 2000), and the predator tends to select the most suitable prey. Therefore, it is possible that in the two-choice experiment, S. tridens dedicated a significant part of its time to searching for the preferred prey, which reduced the number of eggs consumed.
Smaller predators, particularly those belonging to the Phytoseiidae family, have been demonstrated as effective agents in managing the RPM. Among them, Amblyseius largoensis Muma, Neoseiulus barkeri Hughes, and Iphiseiodes zuluagai Denmark & Muma showed a type II functional response when fed RPM (Carrillo and Peña 2012; Mendes et al. 2018; Filgueiras et al. 2020; Coelho et al. 2020). This quality is particularly valuable in the context of biological control programs aimed at maintaining RPM populations at low levels. Amblyseius largoensis has been found to be very promising for the control of RPM in the field and demonstrated selectivity for some acaricides used to control RPM (Assis et al. 2013). This implies that A. largoensis could be a viable asset for sustainable pest management strategies. Conversely, at high RPM densities, S. tridens, with its type III functional response, could also make a significant contribution to biological control. In this context, an approach that combines phytoseiids and Stethorini seems promising for effective RPM control in the field. A relevant example is the successful pest control strategy implemented in New Zealand, where the combined action of Stethorus bifidus Kapur and Phytoseiulus persimilis Athias-Henriot resulted in a rapid reduction in the populations of Tetranychus lintearius Dufour ultimately leading to an equilibrium condition for the pest over several years (Hill et al. 2000). However, further research is warranted to determine the potential impact of S. tridens on phytoseiids. Despite the strong preference of S. tridens for preying on Tetranychidae, these predators can to consume RPM, which may help in managing this pest in the future. Moreover, an understanding of S. tridens food preferences suggests that the predator may primarily target native phytophagous mites, such as T. abacae on bananas (Vasconcelos et al. 2004) and Oligonychus pratensis Banks on coconut palms (Calvet et al. 2022). However, in the absence of these preferred options, it would readily switch to attacking RPM. This adaptive behavior highlights the versatility of S. tridens in effectively managing various pest populations. These possibilities open up promising avenues for future research, emphasizing the need to explore predator-prey interactions and their implications for sustainable pest control strategies.
Acknowledgments
The authors are grateful to Fundação de Amparo a Ciência e Tecnologia de Pernambuco (FACEPE) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (Proc. No. 306092/2021-2) for the financial support of this study.
References
- Ali A., Ahmad S., Maula F., Ali Khan I., Yasmin B. 2016. Effect of temperature on food consumption of the black ladybird beetle, Stethorus punctum Leconte (Coleoptera: Coccinillidae) reared on the two spotted spider mite, Tetranychus urticae under different constant temperatures. J. Entomol. Zool. Stud., 4(1): 628-632.
- Asquith D., Hull L.A. 1973. Stethorus punctum and pest-population responses to pesticide treatments on apple trees. J. Econ. Entomol., 66(5): 1197-1204. https://doi.org/10.1093/jee/66.5.1197
- Assis C.P., Morais E.G., Gondim M.G. 2013. Toxicity of acaricides to Raoiella indica and their selectivity for its predator, Amblyseius largoensis (Acari: Tenuipalpidae: Phytoseiidae). Exp. Appl. Acarol., 60: 357-365. https://doi.org/10.1007/s10493-012-9647-5
- Bayoumy M.H. 2011. Foraging behavior of the coccinellid Nephus includens (Coleoptera: Coccinellidae) in response to Aphis gossypii (Hemiptera: Aphididae) with particular emphasis on larval parasitism. Environ. Entomol., 40(4): 835-843. https://doi.org/10.1603/EN10298
- Bazgir F., Shakarami J., Jafari S. 2020. Prey-stage preferences, functional and numerical responses, and mutual interference of Typhlodromus bagdasarjani (Acari: Phytoseiidae) on Eotetranychus frosti (Tetranychidae). Int. J. Acarol., 46(3): 185-191. https://doi.org/10.1080/01647954.2020.1734657
- Beard J.J., Ochoa R., Bauchan G.R., Welbourn W.C., Pooley C., Dowling, A.P.G. 2012. External mouthpart morphology in the Tenuipalpidae (Tetranychoidea): Raoiella a case study. Exp. Appl. Acarol., 57, 227-255. https://doi.org/10.1007/s10493-012-9540-2
- Biddinger D.J., Weber D.C., Hull L.A. 2009. Coccinellidae as predators of mites: Stethorini in biological control. Biol. Control, 51(2): 268-283. https://doi.org/10.1016/j.biocontrol.2009.05.014
- Britto E.P., Gondim Jr. M.G.C., Torres J.B., Fiaboe K.K.M., Moraes G.J., Knapp, M. 2008. Predation and reproductive output of the ladybird beetle Stethorus tridens preying on tomato red spider mite Tetranychus evansi. BioControl, 54: 363-368. https://doi.org/10.1007/s10526-008-9178-5
- Bruzzone O.A., Aguirre M.B., Hill J.G., Virla E.G., Logarzo G. 2022. Revisiting the influence of learning in predator functional response, how it can lead to shapes different from type III. Ecol. Evol., 12(2): e8593. https://doi.org/10.1002/ece3.8593
- Calvet E.C., Lima D.B., Melo J.W., Gondim Jr. M.G.C. 2022. The expansion of invasive mite Raoiella indica can be improved by coexistence with Oligonychus pratensis. Ann. Appl. Biol., 181(3): 288-297. https://doi.org/10.1111/aab.12773
- Carrillo D., Amalin D., Hosein F., Roda A., Duncan R.E., Peña J.E. 2012. Host plant range of Raoiella indica (Acari: Tenuipalpidae) in areas of invasion of the New World. Exp. Appl. Acarol., 57: 271-289. https://doi.org/10.1007/s10493-011-9487-8
- Carrillo D., Peña J.E. 2012. Prey-stage preferences and functional and numerical responses of Amblyseius largoensis (Acari: Phytoseiidae) to Raoiella indica (Acari: Tenuipalpidae). Exp. Appl. Acarol., 57: 361-372. https://doi.org/10.1007/s10493-011-9488-7
- Coblentz K.E., De Long J.P. 2020. Predator‐dependent functional responses alter the coexistence and indirect effects among prey that share a predator. Oikos, 129(9): 1404-1414. https://doi.org/10.1111/oik.07309
- Coelho C.R., Galvão A.S., Santos M.C., Farias A.P., Teodoro A.V. 2020. Effectiveness of three species of predatory mites (Acari: Phytoseiidae) for controlling Raoiella indica (Acari: Tenuipalpidae). Rev. Prot. Veg., 35: 1-9.
- Contreras-Bermúdez Y., Palomares-Pérez M., Galhou A., Suaste-Dzul A.P., Sarmiento-Cordero M.A., Sánchez-González J.A., Arredondo-Bernal Y.H.C. 2017. Chrysopids (Neuroptera: Chrysopidae) associated with Raoiella indica (Acari: Tenuipalpidae) in Colima, Mexico. J. Entomol. Sci., 52(4): 460-462. https://doi.org/10.18474/JES17-62.1
- Costa J.F., Matos C.H.C., Oliveira C.R.F., Silva T.G., Neto I.F.L. 2017. Functional and numerical responses of Stethorus tridens Gordon (Coleoptera: Coccinellidae) preying on Tetranychus bastosi Tuttle, Baker & Sales (Acari: Tetranychidae) on physic nut (Jatropha curcas). Biol. Control, 111: 1-5. https://doi.org/10.1016/j.biocontrol.2017.04.015
- Costa J.F., Matos C.H.C., Oliveira C.R.F., Santos G.A. 2020. Biology and life table of Stethorus tridens fed Tetranychus bastosi on physic nut. Bull. Insectology, 73(1): 111-116. https://doi.org/10.1016/j.biocontrol.2017.04.015
- Dick J.T.A., Alexander M.E., Jeschke J.M., Ricciardi A., MacIsaac H.J., Robinson T.B., Kumschick S., Weyl O.L.F., Dunn A.M., Hatcher M.J., Paterson R.A., Farnsworth K.D., Richardson D.M. 2014. Advancing impact prediction and hypothesis testing in invasion ecology using a comparative functional response approach. Biol Invasions, 16: 735-753. https://doi.org/10.1007/s10530-013-0550-8
- Eini N., Jafari S., Fathipour Y., Prager S.M. 2023. Experienced generation-dependent functional and numerical responses of Neoseiulus californicus (Acari: Phytoseiidae) long-term reared on thorn apple pollen. Acarol., 63(2): 539-552. https://doi.org/10.24349/isgo-9oic
- Eubanks M.D., Denno R.F. 2000. Health food versus fast food: the effects of prey quality and mobility on prey selection by a generalist predator and indirect interactions among prey species. Ecol. Entomol., 25(2): 140-146. https://doi.org/10.1046/j.1365-2311.2000.00243.x
- Evans E.W., Stevenson A.T., Richards, D.R. 1999. Essential versus alternative foods of insect predators: benefits of a mixed diet. Oecologia, 121: 107-112. https://doi.org/10.1007/s004420050911
- Fernández-Arhex V., Corley J.C. 2003. The functional response of parasitoids and its implications for biological control. Biocontrol Sci. Technol., 13(4): 403-413. https://doi.org/10.1080/0958315031000104523
- Fidelis E.G., Farias E.D.S., Junior R.J.D.S. 2022. Contribution of natural enemies and weather to the population buildup of Raoiella indica on coconut. Syst. Appl. Acarol., 27(8): 1535-1546. https://doi.org/10.11158/saa.27.8.6
- Filgueiras R.M.C., Mendes J.A., Sousa Neto E.P., Monteiro N.V., Melo J.W.S. 2020. Neoseiulus barkeri Hughes (Acari: Phytoseiidae) as a potential control agent for Raoiella indica Hirst (Acari: Tenuipalpidae). Syst. Appl. Acarol. 25(4): 593-606. https://doi.org/10.11158/saa.25.4.1
- Ganjisaffar F., Perring T.M. 2015. Prey stage preference and functional response of the predatory mite Galendromus flumenis to Oligonychus pratensis. Biol. Control, 82: 40-45. https://doi.org/10.1016/j.biocontrol.2014.12.004
- Gómez-Moya C.A., Lima T.P., Morais E.G., Gondim Jr. M.G., Moraes G.D. 2017. Hosts of Raoiella indica Hirst (Acari: Tenuipalpidae) native to the Brazilian Amazon. J. Agric. Sci., 9(4): 86-94. https://doi.org/10.5539/jas.v9n4p86
- Gondim Jr. M.G.C., Castro T.M., Marsaro Jr. A.L., Navia D., Melo J.W., Demite P.R., Moraes G.J. 2012. Can the red palm mite threaten the Amazon vegetation? Syst. Biodivers., 10(4): 527-535. https://doi.org/10.1080/14772000.2012.752415
- Gordon R.D., Chapin E.A. 1983. A revision of the new world species of Stethorus Weise (Coleoptera: Coccinellidae). Trans. Am. Entomol. Soc., 109(3): 229-276.
- Gupta Y.N. 2001. A conspectus of natural enemies of phytophagous mites and mites as potential biological control agents of agricutural pests in India. In: Halliday R.B., Walter D.E., Proctor H.C., Norton R.A. Colloff M.J. (Eds). Proceedings of the Xth International Congress of Acarology, CSIRO Publishing, Australia, p. 484-497.
- Hagen K.S.1987. Nutritional ecology of terrestrial insect predators. In: F. Slansky Junior and J.G. Rodriquez, eds. Nutritional ecology of insects, mites, spiders, and related invertebrates. New York: John Wiley & Sons. pp. 533-577.
- Hassell M.P. 1978. The dynamics of arthropod predator-prey systems. Princeton, NJ, Princeton University Press, 248 pp.
- Hill R.L., Gourlay A.H., Fowler S.V. 2000. The biological control program against gorse in New Zealand. In Proceedings of the X international Symposium on Biological Control of Weeds. Montana State University Bozeman, Montana, USA. 917: 909-917.
- Hodek I. 1973. Biology of Coccinellidae. Prague: Academia the Hague. https://doi.org/10.1007/978-94-010-2712-0
- Hodek I., Honěk A. 1996. Ecology of Coccinellidae. Kluwer Academic Publishers. Dordrecht., 464. https://doi.org/10.1007/978-94-017-1349-8
- Holling C.S. 1959. Some characteristics of simple types of predation and parasitism. Can. Entomol. 91(7): 385-398. https://doi.org/10.4039/Ent91385-7
- Holling C.S. 1965. The functional response of predators to prey density and its role in mimicry and population regulation. Mem. Ent. Soc. Can., 97(S45): 5-60. https://doi.org/10.4039/entm9745fv
- Holt R.D., Lawton J.H. 1994. The ecological consequences of shared natural enemies. Annu. Rev. Ecol. Evol. Syst., 25(1): 495-520. https://doi.org/10.1146/annurev.es.25.110194.002431
- Houck M.A. 1986. Prey preference in Stethorus punctum (Coleoptera: Coccinellidae). Environ. Entomol., 15(4): 967-970. https://doi.org/10.1093/ee/15.4.967
- Jaworski C.C., Bompard A., Genies L., Amiens-Desneux E., Desneux N. 2013. Preference and prey switching in a generalist predator attacking local and invasive alien pests. PLoS One, 8(12): e82231. https://doi.org/10.1371/journal.pone.0082231
- Joshi M., Muralidharan C.M., Sharma K.M., Patel P.S., Varadharasu P.R. 2023. Seasonal incidence of Raoiella indica Hirst (Acari: Tenuipalpidae) on different varieties of date palm in Kachchh region of Western India. Persian J. Acarol., 12(1): 91-100. https://doi.org/10.22073/pja.v12i1.76729
- Juliano S.A. 1993. Nonlinear Curve Fitting: predation and funtional response curves. In: Scheiner S.M., Gurevitch J. (Eds). Design and analysis of ecological experiments. Chapman and Hall, New York, p. 159-182.
- Kundoo A.A., Khan A.A. 2017. Coccinellids as biological control agents of soft bodied insects: A review. J. Entomol. Zool. Stud., 5(5): 1362-1373.
- Lin T., Chen Y., Chen Y., Lin S., Hu J., Zhao J., Yang G., Yang F. Wei, H. 2022. Temperature-dependent functional response of the arboreal rove beetle, Oligota flavicornis (Coleoptera: Staphylinidae), a voracious predator of Tetranychus urticae (Acarina: Tetranychidae). J. Econ. Entomol., 116(1): 90-97. https://doi.org/10.1093/jee/toac170
- Lira V.A., Jumbo L.O.V., Freitas G.S., Rêgo A.S., Galvão A.S., Teodoro A.V. 2021. Efficacy of Amblyseius largoensis (Muma) as a biocontrol agent of the red palm mite Raoiella indica (Acari: Tenuipalpidae). Phytoparasitica, 49: 103-111. https://doi.org/10.1007/s12600-020-00875-8
- Mendes J.A., Lima D.B., Neto E.P.D.S., Gondim Jr. M.G.C., Melo J.W.S. 2018. Functional response of Amblyseius largoensis to Raoiella indica eggs is mediated by previous feeding experience. Syst. Appl. Acarol., 23(10): 1907-1914. https://doi.org/10.11158/saa.23.10.3
- Moffat H., Hainy M., Papanikolaou N.E., Drovandi C. 2020. Sequential experimental design for predator-prey functional response experiments. J. R. Soc. Interface, 17(166): 20200156. https://doi.org/10.1098/rsif.2020.0156
- Murdoch W.W. 1969. Switching in general predators: experiments on predator specificity and stability of prey populations. Ecol. Monogr., 39(4): 335-354. https://doi.org/10.2307/1942352
- Navia D., Morais E.G.F., Mendoça R.D., Gondim Jr. M.G.C. 2015. Ácaro-vermelho-das-palmeiras, Raoiella indica Hirst. In: Vilela, E.F., Zucchi R.A. (Eds). Pragas Introduzidas no Brasil: insetos e ácaros. Piracicaba: Fealq. p. 418-452.
- Pervez A., Omkar. 2005. Functional responses of coccinellid predators: an illustration of a logistic approach. J. Insect Sci., 5(1): 5. https://doi.org/10.1093/jis/5.1.5
- Puttaswamy, Rangaswamy, H.R. 1976. Stethorus keralicus Kapur, (Coleoptera: Coccinellidae)-a predator of arecanut palm mite. Curr. Res., 5(2): 27-28.
- Ragkou V.S., Athanassiou C.G., Kavallieratos N.G., Tomanović Ž. 2004. Daily consumption and predation rate of different Stethorus punctillum instars feeding on Tetranychus urticae. Phytoparasitica, 32: 154-159. https://doi.org/10.1007/BF02979781
- Sarmento R.A., Venzon M., Pallini A., Oliveira E.E., Janssen A. 2007. Use of odours by Cycloneda sanguinea to assess patch quality. Entomol. Exp. Appl., 124(3): 313-318. https://doi.org/10.1111/j.1570-7458.2007.00587.x
- SAS Institute. 2008. SAS/STAT User's guide, version 8.02, TS level 2 MO. SAS Institute Inc., Cary, North Carolina.
- Soares A.O., Coderre D., Schanderl H. 2004. Dietary self‐selection behaviour by the adults of the aphidophagous ladybeetle Harmonia axyridis (Coleoptera: Coccinellidae). J. Anim. Ecol., 73(3): 478-486. https://doi.org/10.1111/j.0021-8790.2004.00822.x
- Sohrabi F., Shishehbor P. 2007. Functional and numerical responses of Stethorus gilvifrons Mulsant feeding on strawberry spider mite, Tetranychus turkestani Ugarov and Nikolski. Pak. J. Biol. Sci., 10(24): 4563-4566. https://doi.org/10.3923/pjbs.2007.4563.4566
- Solomon M.E. 1949. The natural control of animal populations. J. Anim. Ecol., 18(1): 1-35. https://doi.org/10.2307/1578
- Song B., Yang Q., Li Q., Jiang C., Wang X., Wang H. 2018. Prey preferences of Orius munutus. Chin. J. Entomol., 55(1): 112-116.
- Sousa Neto E.P., Souza I.V., Guzzo E.C., Melo J.W.S. 2021. Carnaúba [Copernicia prunifera (Miller) HE Moore, Arecaceae], uma nova hospedeira de Raoiella indica Hirst, 1924 (Acari: Tenuipalpidae). Entomol. Commun., 3: ec03045-ec03045. https://doi.org/10.37486/2675-1305.ec03045
- Souza I.D., Lyra-Lemos R.P., Guzzo, E.C. 2020. Potential of native palm species in Northeast Brazil as hosts for the invasive mite Raoiella indica (Acari: Tenuipalpidae). Exp. Appl. Acarol., 80(4): 509-520. https://doi.org/10.1007/s10493-020-00484-4
- Souza I.V., Ferreira M.J.R., Lyra-Lemos R.P., Guzzo E.C. 2023. Plants harbouring the red palm mite Raoiella indica Hirst (Acari: Tenuipalpidae) in Alagoas State, Northeast Brazil, with novel host associations. Syst. Appl. Acarol., 28(6): 1179-1184. https://doi.org/10.11158/saa.28.6.12
- Stephens D.W., Krebs J.R. 1986. Foraging Theory. Princeton University Press, Princeton, NJ, pp. 247.
- Taghizadeh M., Irani-Nejad K.H., Iranipour S., Moghaddam M. 2018. Daily consumption and functional response of Stethorus gilvifrons (Coleoptera: Coccinellidae) and Orius albidipennis (Hemiptera: Anthocoridae) to Tetranychus urticae (Acari: Tetranychidae). Persian J. Acarol., 7(4): 363-380.
- Taylor B. 2022. [May 15 2022]. Raoiella indica (red palm mite) CABI Compendium, Datasheet. https://doi.org/10.1079/cabicompendium.46792
- Ullah I. 2000. Aspects of the biology of the ladybird beetle Stethorus vagans (Blackburn) (Coleoptera: Coccinellidae). Ph.D. Dissetation, University of Western Sydney, Australia, 180 pp.
- Vasconcelos G.J., Silva F.R.D., Gondim Jr. M.G., Barros R., Oliveira J.V. 2004. Efeito de diferentes temperaturas no desenvolvimento e reprodução de Tetranychus abacae Baker & Printchard (Acari: Tetranychidae) em bananeira Musa sp. cv. Prata. Neotrop. Entomol., 33(2): 149-154. https://doi.org/10.1590/S1519-566X2004000200004
- Zarghami S., Kocheili F., Mossadegh M.S., Allahyari H., Rasekh A. 2014. Prey preference and consumption capacity of Nephus arcuatus (Coleoptera: Coccinellidae): the influence of prey stage, prey size and feeding experience. Biocontrol Sci. Technol., 24(9): 1062-1072. https://doi.org/10.1080/09583157.2014.919376



2023-10-30
Date accepted:
2024-02-05
Date published:
2024-02-19
Edited by:
Tsolakis, Haralabos

This work is licensed under a Creative Commons Attribution 4.0 International License
2024 Monteiro, Vaneska Barbosa; Matheus, Maria Luíza Tavares; França-Beltrão, Girleide Vieira; Melo, José Wagner da Silva; de Lima, Debora Barbosa and Gondim-Júnior, Manoel Guedes Côrrea
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



