Four hundred and sixty-two mites for the ride: the phoretic companions of a historical Nicrophorus specimen (Coleoptera, Silphidae)
Saloña-Bordas, Marta I.  1
; Spooner, Amoret2
and Perotti, M. Alejandra
1
; Spooner, Amoret2
and Perotti, M. Alejandra  3
3
1University of the Basque Country UPV/EHU (retired), Apdo 644 Leioa, 48940 Bizkaia, Spain.
2Oxford University Museum Of Natural History, U.K.
3✉ Ecology and Evolutionary Biology Section, School of Biological Sciences, University of Reading, U.K.
2023 - Volume: 63 Issue: 4 pages: 1030-1038
https://doi.org/10.24349/p08h-853yOriginal research
Keywords
Abstract
Introduction
Mites of the families Parasitidae and Macrochelidae have established a close symbiotic relation with carrion beetles of the family Silphidae (Schwarz and Müller 1992, Schwarz et al. 1998, Andreev 1988, Athias-Binche et al. 1993, Baker and Schwarz 1997, Korn 1983, Wilson 1983, Wilson and Knollenberg 1987). The beetles are known to nest and feed on decaying organic matter, corpses and carcasses (Anderson 1982). Parasitidae and Macrochelidae mites will accompany the beetles patrolling their nests, consuming any blowfly maggots trying to eat their offspring food, and in return they will enjoy housing provided by the beetles and free transport to fresh carcasses (Perotti and Braig 2009, Braig and Perotti 2009). A detrimental association has been claimed for some Poecilochirus species (Parasitidae). However, the claims were based on experiments carried out on artificial arenas, micro-cosmos or enclosures where both beetles and mites were living in a very limited and sub-optimal habitat (Blackman 1997, Blackman and Evans 1994), quite far from their natural spacious grounds. This limitation led to overcrowding of the mites' population and predation of the beetles' eggs, a situation only seen under artificial rearing.
Phoresy or phoresis is the interaction between a host/carrier and a phoront for the sole purpose of dispersal (Lesne 1986), and is a wide spread transport mechanism adopted by mites and ticks (Acari) (Athias-Binche 1994, Petrova and Basikhin 1993, Mašán and Krištofík, 1992). On occasions, there are overcrowded masses of mites jumping on a host carrier making it impossible to take off (Perotti and Braig 2009), and the view of a few phoretic mite species becoming parasites or pseudoparasites has emerged (Athias-Binche 1994, Camerik 2010).
In general, the male carrion beetle is the first leaving the nest and consequently the first arriving to a new, fresh carcass, preferentially in early stages of decomposition, where it will start building a new nest underneath or in the surroundings of the cadaver (Schwarz and Koulianos 1998, Perotti et al. 2010). It is because of this habit that they were also coined ′burying′ beetles (Anderson and Peck 1985), and their occurrence around a corpse can be overlooked if the nearby soil to the body is not inspected in detail (Saloña-Bordas et al. 2010, Saloña-Bordas and Perotti 2014).
Silphidae beetles and most members of the Parasitidae interacting with the beetles will synchronise their life cycles in a symbiotic, beneficial manner. This topic has received much attention (Schwarz and Müller 1992, Schwarz et al. 1998, Houck and OConnor 1991, Neumann 1943), while the particulars of the interaction with other mites, like Macrochelidae, Eviphidae, Uropodidae, Melicharidae, Acaridae or Histiostomatidae among others, which are less numerous on silphids, is practically unknown. Still, thirteen (13) species of Macrochelidae seem to prefer these beetles as carriers (Perotti and Braig 2009, Knee 2017).
Parasitidae deutonymphs ride on top of the body surface of the beetles, over the elytra, while Macrocheles spp. prefer to hide under the elytra (Knee 2017). Parasitidae are very mobile, they hold onto the beetles using their special pulvilli that grab securely on the tegument. Upon arrival to the new carcass all mites disembark, commence chasing maggots, Parasitidae deutonynmphs moult into adulthood and reproduce. Most times, the beetle may not be seen on the surrounding soil, but the mites abundance will give it away (Perotti and Braig 2009, Perotti et al. 2010, Saloña-Bordas and Perotti 2014). Burying beetles directly compete with fly larvae for food resources (flesh), and therefore, predate them with the help of mites too, successfully eliminating maggots entering their nest (Balduf 1935, Clark 1895, Steele 1927).
The beetles offer a large surface on their bodies, enabling transport of many phoretic mites at once. Post-breeder beetles leaving a corpse or a carcass are the busiest carriers, as most mites in the area want to get transported away. Hundreds of them will crowd themselves on the beetle dorsum, which does not stop the strong Nicrophorus from taking off (Schwarz and Müller 1992, Springett 1968). The highest documented number was reported by Hyatt (1980), who counted 1,607 mites on a Nicrophorus humator male found around a rodent carcass (North Hill, Cornwall, UK). The most predominant mite species were from the Parasitidae, Poecilochirus carabi, with 1,486, followed by 107 of P. subterraneus, all deutonymphs. Mites from another two families were also passengers on this same beetle, 11 of them were females of Macrocheles glaber (Macrochelidae), while the remaining three were Alliphis halleri females (Eviphididae).
In this work, we studied a specimen of Nicrophorus vespillo kept at the historical Silphidae collection in the Oxford University Museum of Natural History (OUMNH), which was stored and preserved with its hundreds of phoretic mites. We revealed the identity of 462 mites that once attempted to travel on this single N. vespillo beetle back in the summer of 1912, on a hill between the marshes that surrounded Oxford, South England.
Materials and Methods
A beetle voucher specimen, over 100-year-old, with the accession number 1912-5580 was examined under a LeicaR stereo microscope with a camera attached, to detect the presence of different morpho-species of mites glued to the cardboard slides (Fig. 1). A handful of mite morphotypes were carefully detached from the cardboard with an entomological pin and mounted with Hoyer mounting medium onto microscope slides after softening for 1 day in Lactic Acid 50% (V/V) (Krantz, 1978). Slides were labeled accordingly and form part of the specimen voucher, at the OUMNH. The slides were then inspected with different magnifications using a LeicaR Phase Contrast compound microscope, attached to a MoticR 10 MP camera.



Results and Discussion
In 1912, insect collector A. H. Hamm found a specimen of Nicrophorus on Lye Hill, from Cowley Marsh towards Hogley Bog, in an area now known as the Lye Valley and Cowley Marsh Nature Reserves, Oxford (according to the labels, see Fig. 2). He removed the 456 plus mites from the single beetle, and painstakingly glued each of them to the cardboard mounts, preserving beetle and mites together (Fig. 1). Specimens have been carefully prepared and photographed, managing to preserve their integrity despite their morphological fragility.



One of the labels attached to the beetle specimen stated its identity as Nicrophorus vespillo. We confirmed this using a key to British Silphidae (Lane et al., 2020). Distinguishing characters included transverse reddish markings on the elytra, partly orange club of the antenna, and curved hind tibia (see beetle in Fig. 1).
Regarding the total number of mites, a hand-written note by Hamm -also preserved in the box (Fig. 1), mentions: ''Lye Hill, July. 1912... 456, (some lost)'', suggesting that perhaps some went missing during collection, extraction and gluing. This high density of mites on such small surface (beetle tegument) once again suggests that the beetle might have ended its breeding duties and was departing towards a new fresh carcass (Perotti and Braig 2009, Schwarz and Koulianos 1998, Perotti et al., 2010).
There is no information about the specific body locations where the mites were originally attached to it when collected in 1912, perhaps they detached when the beetle was euthanatised. Five different mite morphotypes were identified after inspection of each cardboard slide under the stereo microscope. Two of them corresponded to Parasitidae, which are the largest in size and the most numerous, while the remaining smaller three types belonged to two Macrochelidae females, and one Histiostomatidae in its hypopial stage (phoretic forms). These hypopi were observed as hyperphoretic on 4 of the Parasitidae, one on each, as these are very minute and fragile (200-300 µm) and again, on fragile specimens, it was not possible to mount them. Two more hypopi were found on the cardboard slides. The Histiostomatidae hypopi (identified to family using Hughes and Jackson 1958, Fig. 3) bring the total number of mites detected in the voucher to 462. Hyperphoresy is a well-known phenomenon in the Acari, especially used by Uropodidae mites (Mesostigmata) (Bajerlein and Bloszyk 2003), but also reported for Astigmatina and Heterostigmata (Paraschiv and Isaia 2020, Baumann 2018).



The two forms of Parasitidae belong both to the same species, Poecilochirus carabi Canestrini & Canestrini, 1882 (Fig. 4). Four hundred and forty-five (445) P. carabi s.s. were transported by the beetle. All were deutonymphs. Both morphotypes presented the same morphological characters defining the species, the dorsal shields shape and setae, sternal shields, their setae and its sculpture or transverse band at the anterior part, as well as palp chaetotaxis (Hyatt 1980). The two populations differ only in size, confirmed by measuring the podosomal (Fig. 4 C and D) and some of the ophistosomal shields. The length of the podosomal shields measured: 625 - 655µm long for C, and 545 - 600µm long for D (3 specimens were measured for each morphotype). Interestingly, the hypopi of Histiostomatidae were observed only on the population of P. carabi of larger size. While the remaining eleven (11) Mesostigmata, both types were again assigned to one same species, Macrocheles nataliae Bregetova & Koroleva, 1960 (Fig. 5) (Mašán 2003). For these, the two M. nataliae populations only differ in the degree of sclerotization.



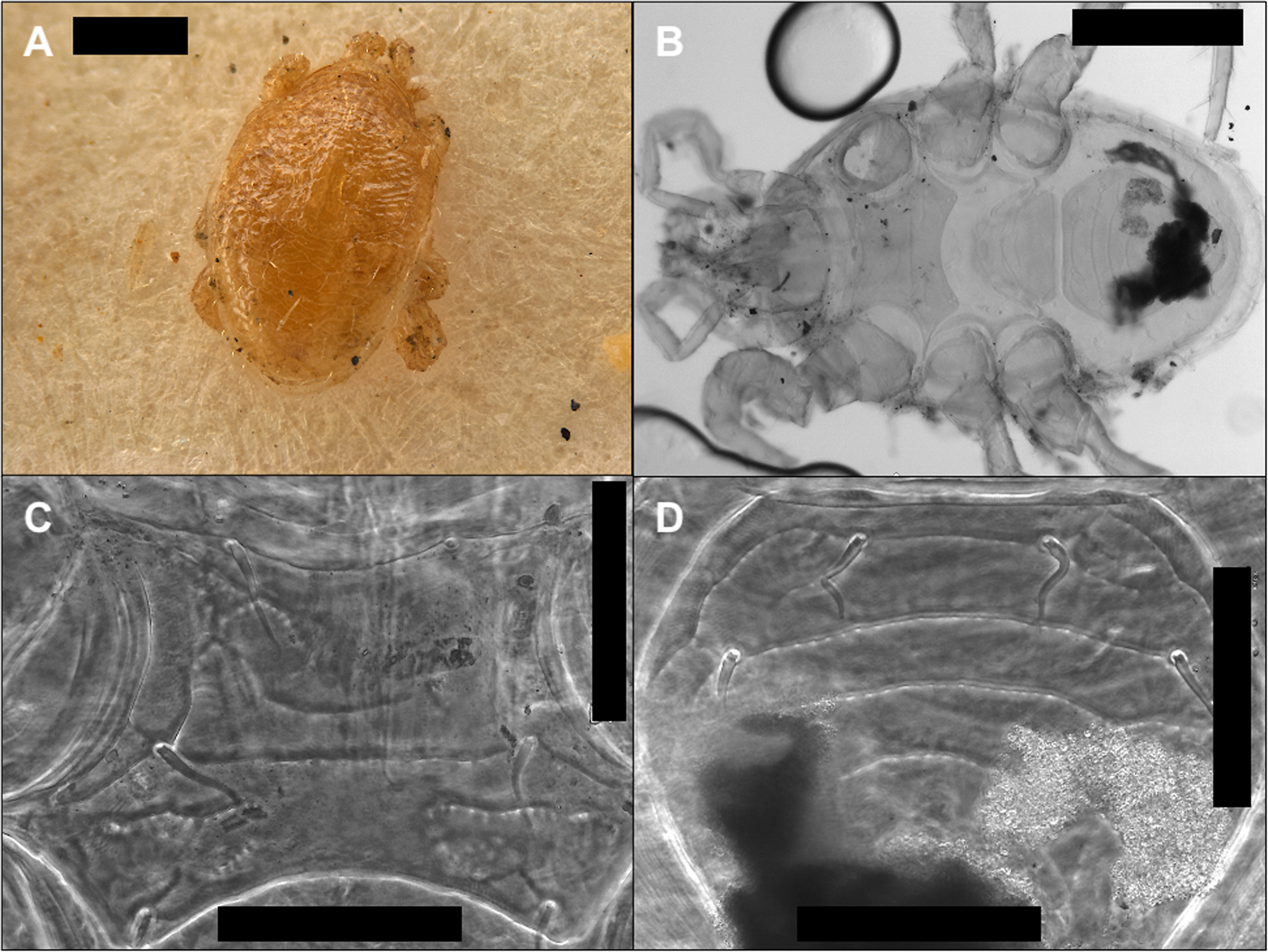


It is not surprising to find two different populations of P. carabi s.s. riding simultaneously on the same beetle. These mites switch carriers, perhaps to avoid inbreeding at arrival, as roughly the same number of male and female nymphs travel on a single carrier and they moult into adulthood and start breeding shortly after disembarking (Schwarz and Müller 1992, Schwarz et al. 1998, Baker and Schwarz 1997, Korn 1983, Perotti and Braig 2009, Schwarz and Koulianos 1998, Perotti et al. 2010, Springett 1968, Brown and Wilson 1994). This might suggest that the small rodent carcass where the beetle originated was colonised by more than one silphid that could be the same or a different Nicrophorus species (Schwarz and Müller 1992, Baker and Schwarz 1997, Saloña-Bordas and Perotti 2014, Brown and Wilson 1994, Mašán 1999). P. carabi s.s. has been recorded from a number of Nicrophorus, Necrodes and Oiceoptoma silphid species in Europe and the British Islands, including the beetle species inspected here, N. vespillo (Hyatt 1980, Mašán 1999). An interesting outcome of the size differences of the populations was the finding of the astigmatid hypopi attached only to the larger morphotypes. This might relate to phoretic preferences of this unknown species of Histiostomatidae.
Macrochelidae are quite ubiquitous on Nicrophorus species (Knee 2017, Mašán 2003, Hyatt 1990, Hyatt and Emberson 1988, Acs et al. 2017, Ciccolani et al. 1981, Evans and Browning 1956, Halliday 2000, Kamaruzaman et al. 2018, Krantz 1998, Krantz and Whitaker 1988, Olivo 1966, Özbek 2017), however, in much less numbers (abundance) than Parasitidae. Despite the finding of just 11 M. nataliae, two populations were riding the beetle. Macrochelids might use a similar tactic than parasitids by travelling together as mixed phoretic female founders (populations), with the purpose of preventing future inbreeding. The two mites analysed, one of each population were virgin females, as there was no spermatheca inserted in the slits of coxae IV. This might suggest that M. nataliae starts reproducing at arrival, parthenogenetically, producing their (F1) haploid males (Kamaruzaman et al. 2018). Availability of mates from different genetic pools will favour successful outbreeding. If M. nataliae females travel as virgin this has also utility in forensic analyses, in estimations of time. The absence of males and the occurrence of females-only indicates recent arrival of the beetles to the carcass or corpse (Perotti and Braig 2009, Perotti et al 2010, Kamaruzaman et al. 2018). M. nataliae has already been reported from the British Isles on N. vespillo in 1988 (Hyatt and Emberson 1988). M. nataliae belongs to the M. glaber species group (Hyatt and Emberson 1988). This group of species is commonly found in association with decomposition (Halliday 2000, Kamaruzaman et al. 2018). The higher occurrence of M. nataliae in early findings (Evans and Browning 1956), such as this historical case, might suggest that M. glaber, which is very common at present, could be a recent replacement of M. nataliae. Perhaps M. glaber was a rare species for the British Isles in the past, as the species was originally described from continental Europe, Italy (Filipponi and Pegazzano 1962).
Author Contributions
MAP and MISB designed the study. AS separated the mites from the cardboard for identification and investigated and photographed the voucher specimen. MISB and AS confirmed the beetle species. MAP prepared and identified the mite species. MISB and MAP analysed the findings and wrote a first draft of the manuscript. All authors contributed to the final version.
Funding
No funding.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Acs A., Sutak A., Kontschan J. 2017. New records of macrochelid mites and description of a new phoretic species (Acari: Mesostigmata: Macrochelidae) from Greece. Acarologia, 56: 63-71. https://doi.org/10.1051/acarologia/20162188
- Anderson R.S. 1982. Resource partitioning in the carrion beetle (Coleoptera:Silphidae) fauna of southern Ontario: ecological and evolutionary considerations. Can. J. Zool., 60: 1314-1325. https://doi.org/10.1139/z82-178
- Anderson R.S., Peck, S.B. 1985. The carrion beetles of Canada and Alaska: Coleoptera: Silphidae and Agyrtidae. Ottawa: Agriculture Canada. pp. 121.
- Andreev E.A. 1988. Ecology of mites phoretic on beetles of the genus Necrophorus (Silphidae). Nauchnye. Doki. Vyss. Shkoly. Biol. Nauki., 5: 44-47.
- Athias-Binche F. 1994. La phorésie chez les acariens - aspects adaptatifs et evolutifs. Editions du Castillet: Perpignan. pp. 178.
- Athias-Binche F., Schwarz H.H., Meierhofer I. 1993. Phoretic association of Neoseius novus (Ouds., 1902) (Acari: Uropodina) with Nicrophorus spp. (Coleoptera: Silphidae): a case of sympatric speciation? Int. J. Acarol., 19: 75-86. https://doi.org/10.1080/01647959308683542
- Baker A.S., Schwarz H.H. 1997. Morphological differences between sympatric populations of the Poecilochirus carabi complex (Acari: Mesostigmata: Parasitidae) associated with burying beetles (Silphidae: Nicrophorus). Syst. Parasitol., 37: 179-185. https://doi.org/10.1023/A:1005822702267
- Balduf W.V. 1935. Bioeconomics of entomophagous Coleoptera. New York: John S. Swift Co. pp. 220.
- Bajerlein D., Błoszyk J. 2003. Two cases of hyperphoresy in mesostigmatic mites (Acari: Gamasida: Uropodidae, Macrochelidae). Biol. Letters, 40: 135-136.
- Baumann J. 2018. Tiny mites on a great journey - a review on scutacarid mites as phoronts and inquilines (Heterostigmatina, Pygmephoroidea, Scutacaridae). Acarologia, 58: 192-251. https://doi.org/10.24349/acarologia/20184238
- Blackman S.W. 1997. Experimental evidence that the mite Poecilochirus davydovae (Mesostigmata: Parasitidae) eats the eggs of its beetle host. J. Zool., 242: 63-67. https://doi.org/10.1111/j.1469-7998.1997.tb02929.x
- Blackman S.W., Evans G.O. 1994. Observations on a mite (Poecilochirus davydovae) predatory on the eggs of burying beetles (Nicrophorus vespilloides) with a review of its taxonomic status. J. Zool., 234: 217-227. https://doi.org/10.1111/j.1469-7998.1994.tb06070.x
- Braig H.R., Perotti M.A. 2009. Carcasses and mites. Exp. Appl. Acarol., 49: 45-84. https://doi.org/10.1007/s10493-009-9287-6
- Brown J.M., Wilson D.S. 1994. Poecilochirus carabi: Behavioral and life-history adaptations to different hosts and the consequences of geographical shifts in host communities. In: Houck M.A., (Ed). Mites. Ecological and evolutionary analyses of life history patterns. New York: Chapman and Hall. p. 1-22. https://doi.org/10.1007/978-1-4615-2389-5_1
- Camerik, A.M. 2010. Phoresy revisited. In: Sabelis M.W., Bruin J. (Eds).Trends in acarology: Proceedings of the 12th International Congress of Acarology. Amsterdam: Springer, p. 333-336. https://doi.org/10.1007/978-90-481-9837-5_53
- Ciccolani B., Bianchi Bullini A.L., Bullini L. 1981. Morphological and genetic differentiation between Macrocheles glaber and Macrocheles perglaber (Acarina: Mesostigmata). Int. J. Acarol., 7: 221-224. https://doi.org/10.1080/01647958108683264
- Clark C.U. 1895. On the food habits of certain dung and carrion beetles. J. N. Y. Entomol. Soc., 3: 61.
- Evans G.O., Browning E. 1956. British mites of the subfamily Macrochelinae Trägårdh (Gamasina, Macrochelidae). Bull. Brit. Mus. (Nat. Hist.) Zool., 4: 1-55.
- Filipponi A., Pegazzano F. 1962. Specie italiane del gruppo-glaber (Acarina, Mesostigmata, Macrochelidae, Macrocheles). Redia, 47: 211-238.
- Halliday, R.B. 2000. The Australian species of Macrocheles (Acarina: Macrochelidae). Invertebr. Syst., 14: 273-326. https://doi.org/10.1071/IT99009
- Houck M.A., OConnor B.M. 1991. Ecological and evolutionary significance of phoresy in the Astigmata. Annu. Rev. Entomol., 36: 611-636. https://doi.org/10.1146/annurev.en.36.010191.003143
- Hughes R.D., Jackson C.G. 1958. A review of the Anoetidae (Acari). Virg. J. Sci. 8: 5-198.
- Hyatt, K.H. 1980. Mites of the subfamily Parasitinae (Mesostigmata: Parasitidae) in the British Isles. Bull. Brit. Mus. (Nat. Hist.) Zool., 38: 237-378. https://doi.org/10.5962/p.12620
- Hyatt K.H. 1990. Mites associated with terrestrial beetles in the British Isles. Entomol. Mon. Mag. 126: 1512-1515.
- Hyatt K.H., Emberson R.M. 1988. A review of the Macrochelidae (Acari: Mesostigmata) of the British Isles. Bull. Brit. Mus. (Nat. Hist.) Zool., 54: 63-125. https://doi.org/10.5962/bhl.part.17598
- Kamaruzaman N.A.C., Masan P., Velasquez Y., Gonzalez-Medina A., Lindstrom A., Braig H.R., Perotti M.A. 2018. Macrocheles species (Acari: Macrochelidae) associated with human corpses in Europe. Exp. Appl. Acarol., 76: 453-471. https://doi.org/10.1007/s10493-018-0321-4
- Knee, W. 2017. New Macrocheles species (Acari, Mesostigmata, Macrochelidae) associated with burying beetles (Silphidae, Nicrophorus) in North America. Zookeys, 721: 1-32. https://doi.org/10.3897/zookeys.721.21747
- Korn W. 1983. Zur Vergesellschaftung der Gamasidenarten Poecilochirus carabi G. u. R. Canestrini 1882 (= P. necrophori Vitzthum 1930), P. austroasiaticus Vitzthum 1930 und P. subterraneus Müller 1859 mit Aaskäfern aus der Familie der Silphidae [On the association between gamasid mites, Poecilochirus carabi G. u. R. Canestrini 1882 (= P. necrophori Vitzthum 1930), P. austroasiaticus Vitzthum 1930 and P. subterraneus Müller 1859 with carrion beetles (Silphidae)]. Spixiana, 6: 251-280.
- Krantz, G.W. 1978. A manual of acarology. USA: Oregon State University Book Stores. pp. 509.
- Krantz G.W. 1998. Reflections on the biology, morphology and ecology of the Macrochelidae. Exp. Appl. Acarol., 22: 125-137. https://doi.org/10.1023/A:1006097811592
- Krantz G.W., Whitaker J.O. Jr. 1988. Mites of the genus Macrocheles (Acari: Macrochelidae) associated with small mammals in North America. Acarologia, 29: 225-259.
- Lane S.A., Lucas C.B.H., Whiffin A.L. 2020. The Histeridae, Sphaeritidae and Silphidae of Britain and Ireland. Great Britain, Shrewsbury: Field Studies Council. pp. 310
- Lesne P. 1896. Moeurs du Limosina sacra Meig. Phénomènes de transport mutuel chez les animaux articulés. Origine du parasitisme chez les insectes diptères. Bulletin de la Société Entomologique de France, 1896: 162-165. https://doi.org/10.3406/bsef.1896.21803
- Mašán, P. 1999. Mites (Acarina) associated with burying beetles and carrion beetles (Coleoptera: Silphidae) and description of Poecilochirus mrciaki sp.n. (Mesostigmata, Gamasina). Biologia (Bratislava), 54: 515-524.
- Mašán, P. 2003. Macrochelid mites of Slovakia (Acari, Mesostigmata, Macrochelidae). Bratislava: Slovak Academy of Sciences, Institute of Zoology. pp. 149.
- Mašán P., Krištofík J. 1992. Phoresy of some arachnids (Acarina and Pseudoscorpionidea) on synanthropic flies (Diptera) in the South Slovakia. Biologia (Bratislava), 47: 87-96.
- Neuman K.W. 1943. Die Lebensgeschichte der Käfermilbe Poecilochirus necrophori Vitzt. Nebst Beschreibung aller Entwicklungsstufen. Zool Anz, 142: 1-21.
- Olivo C.M. 1966. Notas sobre Macrocheles de México (Acari: Macrochelidae) [Notes about Mexican Macrocheles (Acari: Macrochelidae)]. Anales de la Escuela Nacional de Ciencias Biológicas, 15: 135-183.
- Özbek H.H. 2017. A review of the macrochelid mites of Turkey (Acari: Macrochelidae), with new records and descriptions of three new species. Zootaxa, 4317: 559-572. https://doi.org/10.11646/zootaxa.4317.3.7
- Paraschiv M., Isaia G. 2020. Disparity of phoresy in mesostigmatid mites upon their specific carrier Ips typographus (Coleoptera: Scolytinae). Insects, 11: 771. https://doi.org/10.3390/insects11110771
- Perotti M.A., Braig, H.R. 2009. Phoretic mites associated with animal and human decomposition. Exp. Appl. Acarol., 49: 85-124 https://doi.org/10.1007/s10493-009-9280-0
- Perotti M.A., Braig H.R. 2011. Eukaryotic ectosymbionts of Acari. J. Appl. Entomol., 135: 514-523. https://doi.org/10.1111/j.1439-0418.2011.01639.x
- Perotti M.A., Braig H.R., Goff M.L. 2010. Phoretic mites and carcasses: Acari Transported by Organisms Associated with Animal and Human Decomposition. In: Amendt J., Campobasso C.P., Grassberger M., Goff M.L. (Eds). Current Concepts in Forensic Entomology: Novel Arthropods, Environments and Geographical Regions. Amsterdam: Springer. p. 69-91. https://doi.org/10.1007/978-1-4020-9684-6_5
- Petrova A.D., Basikhin P.V. 1993. The finding of larvae of the taiga tick Ixodes persulcatus (Parasitiformes: Ixodidae) on necrophagous piophilid flies (Diptera: Piophilidae) in the southern Yamal. Parasitology, 27: 427-429.
- Polak, M. 2003. Heritability of resistance against ectoparasitism in the Drosophila-Macrocheles system. J. Evol. Biol., 16: 74-82. https://doi.org/10.1046/j.1420-9101.2003.00500.x
- Saloña-Bordas M.I., Moraza M.L., Carles-Tolrá M., Iraola V., Bahillo P., Yélamos T., Outerelo R., Alcaraz R. 2010. Searching the soil: Forensic importance of edaphic fauna after the removal of a corpse. J. Forensic Sci., 55: 1652-1655. https://doi.org/10.1111/j.1556-4029.2010.01506.x
- Saloña-Bordas M.I., Perotti M.A. 2014. First contribution of mites (Acari) to the forensic analysis of hanged corpses: A case study from Spain. Forensic Sci. Int., 244: 6-11. https://doi.org/10.1016/j.forsciint.2014.08.005
- Schwarz H.H., Koulianos S. 1998. Review.When to leave the brood chamber? Routes of dispersal in mites associated with burying beetles. Exp. Appl. Acarol., 22: 621-631. https://doi.org/10.1023/A:1006054604520
- Schwarz H.H., Müller J.K. 1992. The dispersal behaviour of the phoretic mite Poecilochirus carabi (Mesostigmata, Parasitidae): adaptation to the breeding biology of its carrier Necrophorus vespilloides (Coleoptera, Silphidae). Oecologia, 89: 487-493.
- Schwarz H.H., Starrach M., Koulianos, S. 1998. Host specificity and permanence of associations between mesostigmatic mites (Acari: Anactinotrichida) and burying beetles (Coleoptera: Silphidae: Nicrophorus). J. Nat. Hist., 32: 159-172. https://doi.org/10.1080/00222939800770101
- Springett, B.P. 1968. Aspects of the relationship between burying beetles, Necrophorus spp. and the mite, Poecilochirus necrophori. Vitz. J. Anim. Ecol., 37: 417-424. https://doi.org/10.2307/2957
- Steele B.F. 1927. Notes on the feeding habits of carrion beetles. J. N. Y. Entomol. Soc., 35: 77-81.
- Wilson, D.S. 1983. The effect of population structure on the evolution of mutualism: a field test involving burying beetles and their phoretic mites. Am. Nat., 121: 851870. https://doi.org/10.1086/284108
- Wilson D.S., Knollenberg W.G. 1987. Adaptive indirect effects: the fitness of burying beetles with and without their phoretic mites. Evol. Ecol., 1: 139-159. https://doi.org/10.1007/BF02067397



2023-02-21
Date accepted:
2023-08-16
Date published:
2023-09-28
Edited by:
Baumann, Julia

This work is licensed under a Creative Commons Attribution 4.0 International License
2023 Saloña-Bordas, Marta I.; Spooner, Amoret and Perotti, M. Alejandra
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



