Contribution to the knowledge of the oribatid mite genus Discoppia (Acari, Oribatida, Oppiidae)
Ermilov, Sergey G.  1
and Makarova, Olga L.
1
and Makarova, Olga L.  2
2
1✉ Tyumen State University, Tyumen, Institute of Environmental and Agricultural Biology (X-BIO), Russia.
2Severtsov Institute of Ecology and Evolution, Russian Academy of Sciences, Moscow, Russia.
2023 - Volume: 63 Issue: 3 pages: 757-769
https://doi.org/10.24349/131f-vr0lZooBank LSID: 6B244B1D-D506-40AB-A69B-AF81458861A7
Original research
Keywords
Abstract
Introduction
The oribatid mite genus Discoppia (Acari, Oribatida, Oppiidae) was proposed by Balogh (1983), with Oppia limae Balogh and Mahunka, 1974 as type species. Subías and Rodríguez (1986) proposed a subgenus Discoppia (Cylindroppia), with Oppia minus cylindrica Pérez-Íñigo, 1965 as type species. Presently, the genus comprises two subgenera with five species (D. (Discoppia) limae (Balogh and Mahunka, 1974); D. (Cylindroppia) casuarina (Abd-el-Hamid, Hussein, Bayoumi and Al-Assiuty, 1983); D. (C.) cylindrica (Pérez-Íñigo, 1965); D. (C.) pentasetata Subías and Rodríguez, 1986; and D. (C.) rostroincisa Subías and Rodríguez, 1986), which are distributed collectively in tropics and subtropics.
The type species of Discoppia (Cylindroppia) was described by Pérez-Íñigo (1965). However, its description is brief and contains significant errors, e.g. incorrect arrangement of dorsal notogastral setae (see Fig. 3d in Pérez-Íñigo 1965). Later, this author (Pérez-Íñigo 1971) provided correct figures. Subías and Rodríguez (1986), Arillo and Subías (1996), Ay and Ayyildiz (2019) presented the supplementary descriptions of this species, however, this description remained incomplete (for example, morphology of exobothridial seta, gnathosoma, and ovipositor, as well as identification of leg setae and solenidia are missed) and rather briefly illustrated.
The main goals of the paper are: to present the supplementary description of D. (C.) cylindrica on the basis of mass materials from Caucasus; to summarize its main morphological traits, which will ensure the identification of this species in the future; to revise the generic diagnosis, which is short and incomplete so far; to discuss the systematic placement of Oppia agricola Fujikawa, 1982 and O. tenuis Hammer, 1958 which Subías (2022) included in Discoppia, as well as the synonymy of Oppia bifidus Bayoumi and Al-Khalifa, 1985 with D. (C.) casuarina (according to Subías 2022) and the taxonomic status of D. (C.) cylindrica rostroincisa Subías and Rodríguez, 1986; to provide an identification key to known species of Discoppia; to compile data concerning distribution and habitats of members of the genus. Additionally, some interesting findings of Oppiidae for Russia and Palaearctic are presented.
Material and methods
Specimens
Mites were found on the slope of the small sand drift (Fig. 1a) formed on the flat sandy beach of the Caspian Sea at the distance of 300 m from the shore line. The vegetation of small dune (two samples) and adjacent plane sandy surface (three samples) was dominated by the bindweed, Convolvulus persicus L. (Fig. 1d). Numerous individuals (231) of D. (C.) cylindrica were found only in two samples from the hill slope (Fig. 1b, c).



Samples (each of volume 250 ml) were taken from the depth of 20–30 cm and contained numerous small roots of the bindweed. Mites were extracted from cores in Moscow using laboratory eclectors during 10 days until their complete dryness without additional heating or lighting. The relative humidity of samples amounted 3.81%. The mites were fixed with 85% alcohol.
Observation and documentation
For measurement and illustration, specimens were mounted in lactic acid on temporary cavity slides. All measurements are in micrometers. Body length was measured in lateral view, from the tip of the rostrum to the posterior edge of the notogaster; other structures were oriented to avoid parallax errors. Notogastral width refers to the maximum in dorsal aspect. Setal lengths were measured perpendicular to their long axis, accounting for curvature. Formulas for leg solenidia are given in square brackets according to the sequence genu-tibia-tarsus. Drawings were made with a camera lucida using a Leica DM 2500 light microscope.
Terminology
Morphological terminology used in this paper follows that of F. Grandjean (see Travé and Vachon 1975) for references, Ermilov et al. (2021), Ermilov and Rybalov (2022); Norton (1977) for leg setal nomenclature; Norton and Behan-Pelletier (2009) for overview.
Abbreviations
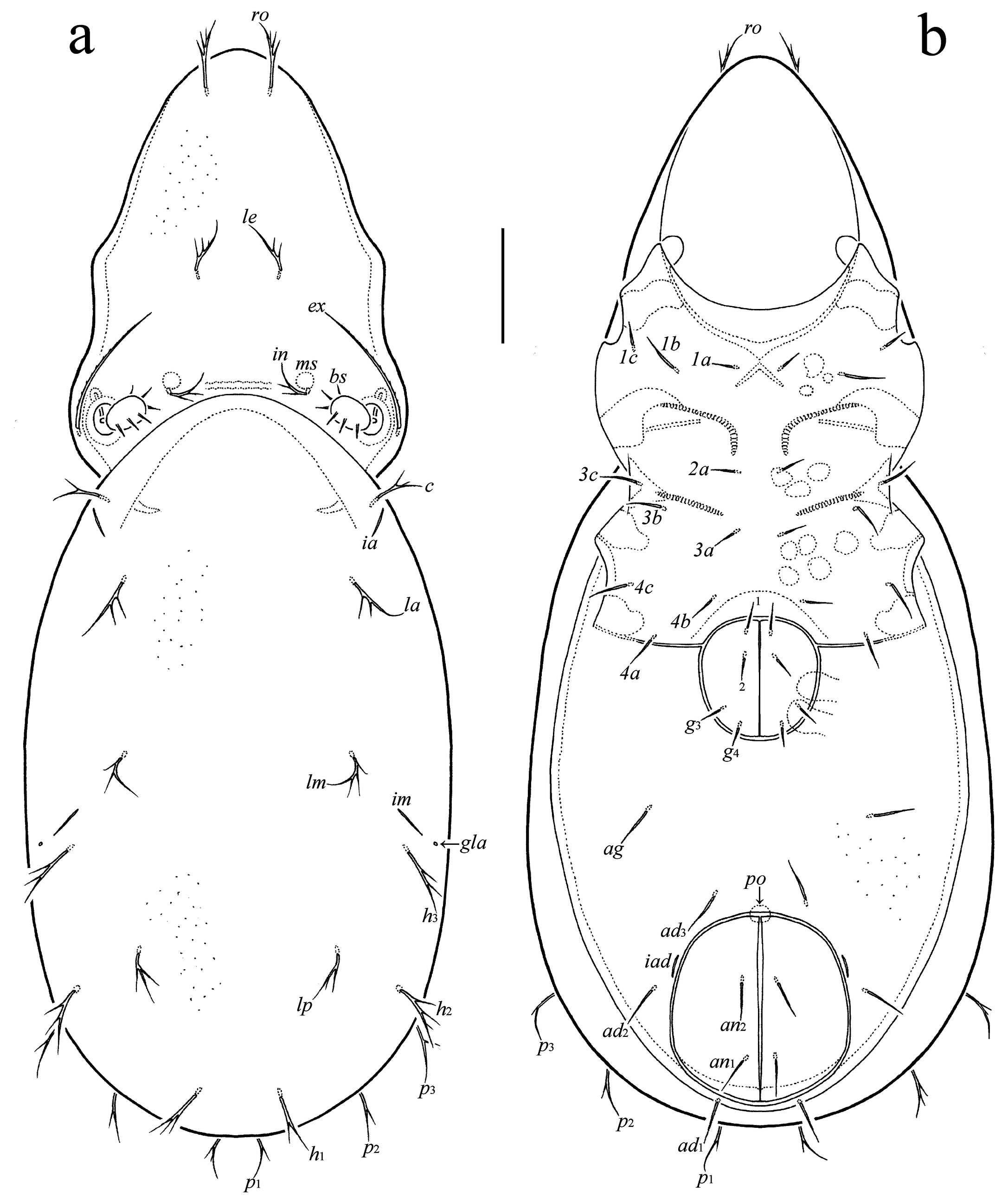
Prodorsum: ro, le, in, bs, ex = rostral, lamellar, interlamellar, bothridial, and exobothridial seta, respectively; exv = vestige of second exobothridial seta; ms = muscle sigillum. Notogaster: c, la, lm, lp, h, p = setae; ia, im, ip, ih, ips = lyrifissures; gla = opisthonotal gland opening. Gnathosoma: a, m, h = subcapitular setae; or = adoral seta; sup, inf, d, l, cm, acm, ul, su, vt = palp setae; ω = palp solenidion; cha, chb = cheliceral setae; Tg = Trägårdh's organ. Epimeral region: 1a–1c, 2a, 3a–3c, 4a–4c = epimeral setae. Anogenital region: g, ag, an, ad = genital, aggenital, anal, and adanal seta, respectively; iad = adanal lyrifissure; ψ, τ = setae of ovipositor; po = preanal organ. Legs: Tr, Fe, Ge, Ti, Ta = trochanter, femur, genu, tibia, and tarsus, respectively; ω, φ, σ = solenidia; ɛ = famulus; d, l, v, bv, ev, ft, tc, it, p, u, a, s, pv, pl = setae.
Taxonomy
Revised generic diagnosis of Discoppia Balogh, 1983
Adult — With character states of Oppiidae (Norton and Behan-Pelletier 2009). Measurements: Very small species with distinctly elongate body; length less than 300; ratio (length/width): about 2.0 or larger. Integument. Without heavy ornamentation and sculpturing. Prodorsum. Rostrum rounded or medially incised. Costula, transcostula and lateral ridge absent. Rostral, lamellar and interlamellar setae short, simple or branched; le inserted closer to in than to ro; exobothridial seta long, setiform; bothridial seta short or medium-sized, usually with unilaterally dilated head, nearly disk-like, rarely, with elongate fusiform head. Interbothridial tubercles absent or present; postbothridial tubercles absent. One pair of muscle sigilla in interbothridial region. Without lines, tubercles or sclerotized apophyses running from dorsosejugal scissure to basal part of prodorsum. Notogaster. Anterior margin convex or straight. Humeral region without tooth and crista. Ten pairs of setae short, roughly the same length, simple or branched. Gnathosoma. Subcapitulum diarthric; adoral setae present. Palp solenidion bacilliform, entirely pressed to surface. Chelicera chelate-dentate. Epimeral and lateral podosomal regions. Epimeral setal formula: 3–1–3–3; all setae short, simple. Epimeral border IV usually straight. Pedotectum I reduced. Discidium absent. Anogenital region. Four or five pairs of genital, one pair of aggenital, two pairs of anal, and three pairs of adanal setae short, simple; adanal seta ad1 posterior, ad2 lateral, ad3 anterior to anal plate; distance ad3–ad3 shorter than ag–ag and ad2–ad2. Adanal lyrifissure located close and parallel to anal aperture. Legs. Tarsus I with 20 setae (l″ and v′ present), tarsus II with 16 setae (l″ present). Tarsus II with two solenidia.
Subgeneric diagnosis of Discoppia (Discoppia) Balogh, 1983
Type species: Oppia limae Balogh and Mahunka, 1974
Adult — With character states of Discoppia (as above) but with straight anterior notogastral margin.
Subgeneric diagnosis of Discoppia (Cylindroppia) Subías and Rodríguez, 1986
Type species: Oppia minus cylindrica Pérez-Íñigo, 1965
Adult — With character states of Discoppia (as above) but with medially convex anterior notogastral margin.
Supplementary description
Discoppia (Cylindroppia) cylindrica (Pérez-Íñigo, 1965)
(Figures 2–4)


Material — Point 1: 127 specimens (all females): Russia, northeastern Caucasus, Dagestan Republic, 20 km N from Makhachkala City, vicinity of Shushiya village, sandy sea beach, slope of small dune, under Convolvulus persicus L. (layer 25–30 cm); 43.164938°N, 47.483112°E; 04.XI.2022 (leg. O.L. Makarova and K.V. Makarov) (Fig. 1b). Point 2: 104 specimens (all females): the same data, but about 10–15 meters distant from Point 1; 43.164902°N, 47.483042°E (Fig. 1c). All specimens (in 70% solution of ethanol with a drop of glycerol) are deposited in the collection of the Tyumen State University Museum of Zoology, Tyumen, Russia.
Description — Measurements – Body length: 176–199 (n=100); body width: 67–90 (n=100).
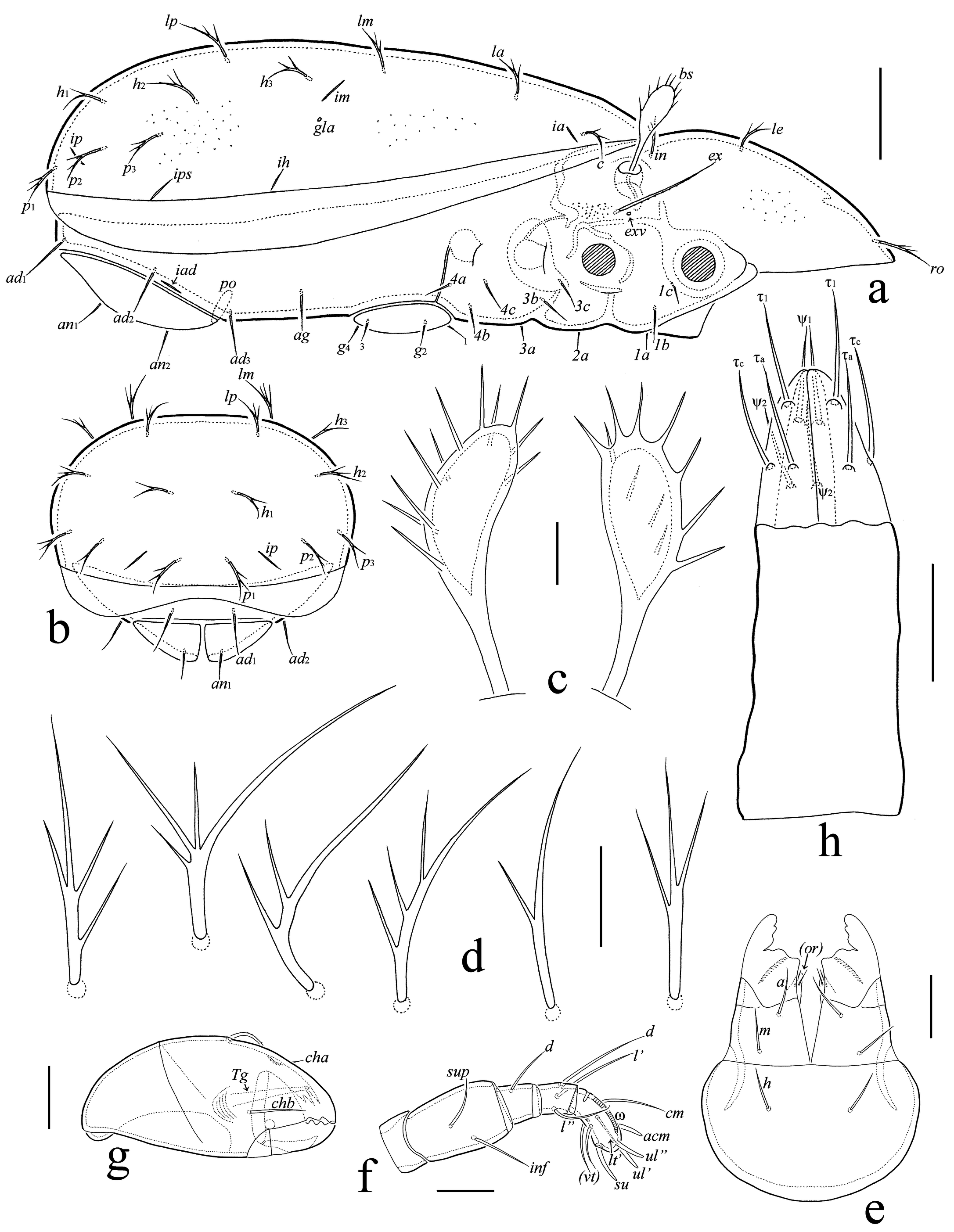


Integument (Fig. 3a) – Body color light yellowish to light brownish. Body surface with dense cuticular microgranules (visible in dissected specimens under high magnification) which well visible between bothridium and acetabula II and III. Dorsal side sometimes covered by sparse cerotegumental microgranules.
Prodorsum (Figs 2a, 3a) – Rostrum rounded. Rostral (15), lamellar (9–11) and interlamellar (9–11) setae branched; exobothridial seta (26–30) setiform, barbed, often pressed to prodorsal surface and therefore poorly observed in dorsal aspect; bothridial seta (19–22) with unilaterally dilated, nearly disk-like head bearing about 10–12 long ciliae (Fig. 3c). One pair of muscle sigilla well visible, located close and anteriorly to interlamellar setae. Slight transverse ridge located between insertions of interlamellar setae.
Notogaster (Figs 2a, 3a, 3b) – All notogastral setae (13–15) branched; number of ciliae variable, from one to four (Fig. 3d). Lyrifissures ia, im, ip, ih, and ips comparatively long, well visible.
Gnathosoma (Figs 3e–3g) – Subcapitulum size: 45–49 × 35–37; all subcapitular setae (9) setiform, slightly roughened; both adoral setae (2) setiform, smooth. Palp length: 24–26; setation: 0–2–1–3–8(+ω); tarsal seta l″ absent; solenidion long (2/3 of tarsus length); postpalpal seta (2) spiniform. Chelicera length: 45–49; setae (cha: 17–19; chb: 9–11) setiform, barbed.
Epimeral region (Figs 2b, 3a) – Epimeral border IV straight. Epimeral setae (1b, 3b, 3c, 4c: 9; others: 7) setiform, slightly roughened.
Anogenital region (Figs 2b, 3a, 3b, 3h) – Genital (7), aggenital (9–11), anal (9), and adanal (11–13) setae setiform, slightly roughened. Adanal lyrifissure short, distinct. Ovipositor (Fig. 3h) elongated (39–45 × 11–13), blades (13–15) shorter than length of distal section (beyond middle fold; 26–30); setae of all blades (ψ1, τ1: 13; ψ2, τa, τc: 11) rod-like, smooth; first blade with four setae, second and third blades with three setae only (τb absent); coronal setae absent.
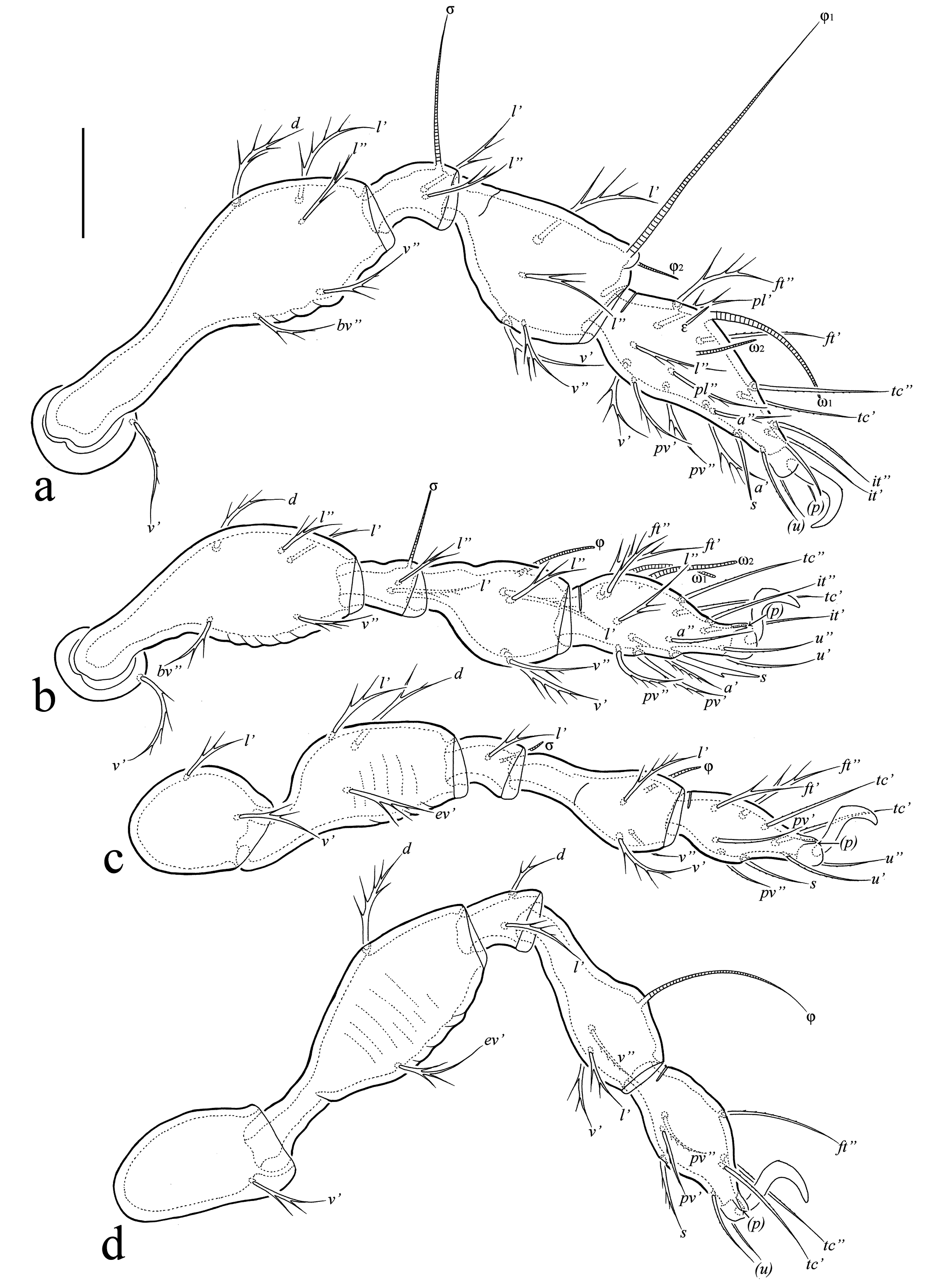


Legs (Figs 4a–4d) – Claw of each leg with slight barb on dorsal side. Porose area on all segments not observed. Formulas of leg setation and solenidia: I (1–5–2–4–20) [1–2–2], II (1–5–2–4–16) [1–1–2], III (2–3–1–3–11) [1–1–0], IV (1–2–2–3–10) [0–1–0]; homology of setae and solenidia indicated in Table 1. Setae p′ and p″ setiform on tarsus I versus very short, spiniform on tarsi II–IV; famulus short, rod-like, located laterally to solenidion ω1; seta s eupathidial on tarsus I; setae a′, a″, it′, and it″ absent on tarsus III, a′ and a″ absent on tarsus IV; many setae on segments branched; solenidia ω1 and ω2 on tarsi I and II, φ2 on tibia I, φ on tibiae II and III, and σ on genua II and III short or medium-sized, slightly or distinctly bacilliform; solenidia φ1 on tibia I, σ on genu I and φ on tibia IV long, subflagellate.
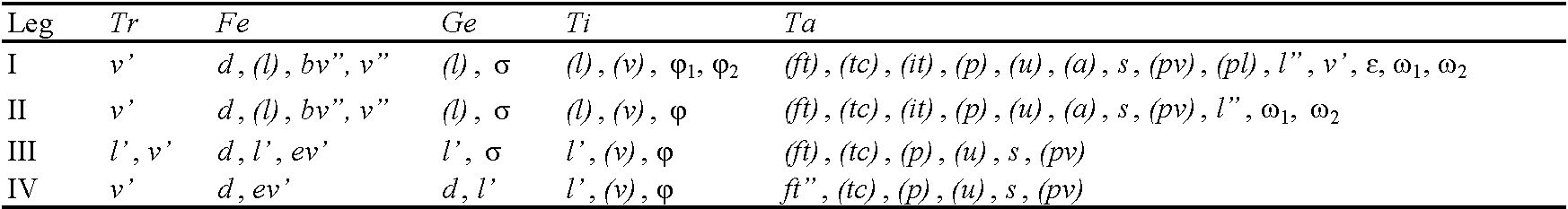


Remarks — Based on our supplementary description and on the available data (e.g., Pérez-Íñigo 1971; Subías and Rodríguez 1986) about adult D. (C.) cylindrica, we propose the following diagnostic morphological traits for this species: body length: 176–225; rostrum rounded; rostral, lamellar, interlamellar, and notogastral setae branched; exobothridial seta setiform, barbed; bothridial seta with unilaterally dilated, nearly disk-like head; epimeral border IV straight; epimeral and anogenital setae setiform, slightly roughened; leg setae a′, a″, it′, and it″ absent on tarsus III, a′ and a″ absent on tarsus IV; many leg setae branched.
Taxonomical proposals
- Oppia bifidus was described by Bayoumi and Al-Khalifa (1985). The distinctive traits of the species are morphologically variable (branches of rostral and notogastral setae; barbulation of bothridial seta) or were not correct (presence of costulae), therefore, Subías (2004) rightly synonymized it with D. (C.) cylindrica cylindrica. However, later, Subías (2022) changed his opinion and synonymized O. bifidus with D. (C.) casuarina. Discoppia (C.) cylindrica and D. (C.) casuarina are morphologically very similar differing only by the morphology of head of the bothridial seta (unilaterally dilated, nearly disk-like versus elongate fusiform). Oppia bifidus has unilaterally dilated, nearly disk-like head of bothridial seta (Bayoumi and Al-Khalifa 1985), therefore, we maintain the initial synonymy (O. bifidus with D. (C.) cylindrica) of Subías in 2004.
- Subías (2004, 2022) considered the species Oppia agricola Fujikawa, 1982 as a junior synonym of D. (C.) cylindrica. However, all morphological traits of O. agricola clearly correspond to the generic diagnosis of Microppia Balogh, 1983 (Balogh 1983; Subías and Shtanchaeva 2023); for example, this species has one pair of small sclerotized tubercles running from dorsosejugal scissure to basal part of prodorsum (versus absent in Discoppia) and differing localization of notogastral seta lm to la (medially versus mostly posteriorly in Discoppia). Therefore, we: 1) do not support the synonymy proposed by Subías (2004), 2) propose a transfer of O. agricola in Microppia: Microppia agricola (Fujikawa, 1982) n. comb.
- Subías (2004, 2022) included the species Oppia tenuis Hammer, 1958 in D. (Cylindroppia). However, some important morphological traits of this species clearly do not correspond to the generic diagnosis of Discoppia; for example, O. tenius has differing localization of notogastral seta lm to la (medially versus mostly posteriorly in Discoppia) and seta c (distanced from humeral region versus in humeral region). Therefore, we do not support the placement of O. tenuis in Discoppia. The problem is that O. tenuis was described briefly by Hammer (1958), and its ventral side, containing important diagnostic characters (e.g., epimeral formula; number of genital setae; localization of adanal setae and adanal lyrifissure), is completely unknown, therefore, an objectively correct placement of O. tenuis in any known oppiid genus seems almost impossible at the moment.
- Discoppia (Cylindroppia) cylindrica rostroincisa was described by Subías and Rodríguez (1986). This subspecies differs from D. (C.) cylindrica cylindrica only by one morphological trait (rostrum incised medially versus rounded). The morphology of the rostrum is not variable in specimens of the same oppiid species and is widely used in taxonomy of the family at the species and generic levels. Therefore, in our opinion, this difference is sufficient to support the insularity of Discoppia rostroincisa on species level, and we propose a species status (instead of the existing subspecies status) for this species: Discoppia (Cylindroppia) rostroincisa Subías and Rodríguez, 1986 n. stat.
Key to known species of Discoppia
1. Anterior notogastral margin straight; one pair of interbothridial tubercles present; body length: 228
...... Subgenus Discoppia (Discoppia); single representative: Discoppia (Discoppia) limae (Balogh and Mahunka, 1974)
— Anterior notogastral margin medially clearly convex; interbothridial tubercles absent
...... 2. Subgenus Discoppia (Cylindroppia)
2. All notogastral setae simple; five pairs of genital setae present; body length: 230
...... Discoppia (Cylindroppia) pentasetata Subías and Rodríguez, 1986 (see also Hammer 1979)
— All notogastral setae branched; four pairs of genital setae present
...... 3
3. Rostrum medially incised; body length: 205–225
...... Discoppia (Cylindroppia) rostroincisa Subías and Rodríguez, 1986 (see also Subías and Arillo 2001)
— Rostrum entire, rounded
...... 4
4. Head of bothridial seta elongate fusiform; body length: 230
...... Discoppia (Cylindroppia) casuarina (Abd-el-Hamid, Hussein, Bayoumi and Al-Assiuty, 1983) (replacing the name for Oppia sitnikovae Shereef, 1976 due the name preoccupation by Kulijev (1962))
— Head of bothridial seta unilaterally dilated, nearly disk-like; body length: 176–225
...... Discoppia (Cylindroppia) cylindrica (Pérez-Íñigo, 1965) (= Oppia bifidus Bayoumi and Al-Khalifa, 1985) (see also Pérez-Íñigo 1971; Subías and Rodríguez 1986; Subías and Arillo 2001; Arillo and Subías 1996)
Distribution and habitats of Discoppia
At present, representatives of Discoppia are known collectively from the tropical and subtropical areas (Subías 2022).
Discoppia (D.) limae is distributed in the Oriental region. It was described from the hill forest litter in Malaysia (Balogh and Mahunka 1974) and also was found in the soil-litter from the base of low limestone hill with coconut trees as well as in litter of the typical beach plants and secondary forest in the Philippines (Ermilov and Corpuz-Raros 2017).
Discoppia (C.) casuarina and D. (C.) pentasetata have a highly confined geographic distribution, i.e. are conventional endemics to one locality: the former is known from soil of the cabbage plantation on the farm (Shereef 1976; as Oppia sitnikovae) and from litter of the date-palm and Casuarina trees (Abd-el-Hamid et al. 1983) in Egypt; the later species was found in the grassland with ferns, mosses, and dead leaves in Java, Indonesia (Hammer 1979; as Oppia sp.).
Discoppia (C.) cylindrica is the most widespread and often occurring member of the genus. It is known from different countries (mostly in western, central and southern Europe and in Central Asia) of the Palaearctic (e.g., Bayoumi and Al-Khalifa 1985; Noto la Diega et al. 2004; Starý 2006; Shtanchaeva and Subías 2010; Mahunka et al. 2013; Subías et al. 2013; Akrami 2015; Murvanidze and Mumladze 2016; Ay and Ayyildiz 2019), Neotropical (Subías et al. 2004; Schatz 2006) and Oriental (Ermilov et al. 2012) regions. This species is distributed in the upper and deep layers of the soil profile in different biotopes in flat and mountain areas; e.g., from the litter and mineral layer of agricultural soils, grasslands, littoral sandy soils, soils of dry and mixed grass steppes, deciduous forests and plantations as well as coniferous forests (e.g., Al-Khalifa and Bayoumi 1983; Vasiliu and Ivan 1996; Ivan 2007; Smelyansky 2010; Abdurakhmanov et al. 2013; Akrami and Behmanesh 2015; Movahedzade et al. 2016; Murvanidze and Mumladze 2016; Akrami and Salehi Sarbizhan 2020; Akrami and Shahedi 2020) including both natural and anthropogenic habitats. Also, the species was registered from soil-litter of the tropical moist forest in Panama (Subías et al. 2004; Schatz 2006) and from the ground substratum of the cave in Vietnam (Ermilov et al. 2012).
Discoppia (C.) rostroincisa is recorded mostly from mineral layer of different soils, including agricultural soils of Spain (e.g. Subías and Rodríguez 1986; Subías et al. 2013); also, the species was found from soil-litter under the Pinus trees in Hungary (Mahunka and Mahunka-Papp 1999), in litter of the mixed forest in Tanzania (Ermilov and Khaustov 2018) and without designation of biotope in Dominican Republic (Subías and Arillo 2001).
Thus, one can conclude that D. (C.) cylindrica is the northernmost member of its genus.
Accompanying interesting findings of Oppiidae
In total, the biotope where the numerous specimens of D. (C.) cylindrica were found (sandy soils dominated by C. persicus on the Caspian Sea coast) was populated by seven oribatid species from which only solitary Microppia minus (Paoli, 1908) is the semicosmopolitan polyzonal species. All other mites are either south European/Palaearctic species (Epilohmannia styriaca Schuster, 1960; Lalmoppia krivolutskyi (Poltavskaya, 1994); Oxyoppia (Oxyoppiella) minuscula Hugo-Coetzee, Lotfollahi, Abbasi-Kalo and Movahedzade, 1917) or the broader distributed species that in the Palaearctic inhabit only southern areas (Sellnickochthonius furcatus (Weis-Fogh, 1948); D. cylindrica; Graptoppia (Stenoppia) boucheri Ermilov and Frolov, 2021).
The discovery of two recently described oppiid species are of particular interest. Oxyoppia (Oxyoppiella) minuscula till now was known only from Iran (Hugo-Coetzee et al. 2017) where this species resides also open biotopes (wheat fields and grassland). Our finding in the seaboard Dagestan is the first record of O. minuscula in Russia and the northernmost record of the semicosmopolitan subgenus O. (Oxyoppiella) Subías and Rodríguez, 1986 (Hugo-Coetzee et al. 2017). Graptoppia (Stenoppia) boucheri was known only from passalid beetles Erionomus planiceps Eschscholtz collected in Ghana and Gabon, West Africa (Ermilov and Frolov 2021). So, our finding of this species in sandy soil of maritime Dagestan is its first record in the Palaearctic and one of a few northernmost records of the semicosmopolitan subgenus G. (Stenoppia) Balogh, 1983 (Subías 2022). Distributional range of the mainly tropical beetle family Passalidae in total is far away from Caucasus (Boucher 2005), that indicates the need for the further investigation of habits and habitats of G. boucheri.
Thus, the oribatid species composition revealed in the sandy coastal soil of middle Caspian Sea can be determined as thermophilous consisting mainly of small-size species. The size of most species found is smaller than 300 μm (only the length of E. styriaca exсeeds 475 μm). The specific habitat of deep sandy soil (where the vertical migration of small arthropod is enhanced) may serve as a stable concentration site of the tropical-subtropical oribatid fauna.
Notably, that the most species (five from seven) and individuals (283 from 285) in studied biotope were parthenogenetic (thelytokous), i.e. reproducing without males (so far presumably for O. minuscula). This community feature also argues the stable resource ability that was demonstrated recently in the comprehensive analysis of very different oribatids assemblages (Maraun et al. 2019). Possibly, this tendency is common for the free-living mites in general (see Manning and Halliday1994; Walter and Lindquist 1995).
Acknowledgements
We are thankful to L.S. Subías and U.Ya. Shtanchaeva for consultations and two anonymous reviewers for valuable comments. This research was partially supported by the grant from the Russian Foundation for Basic Research (No. 20-54-56054 Iran_t).
References
- Abd-el-Hamid M.E., Hussein M.A., Bayoumi B.M, Al-Assiuty A.I. 1983. The distribution of oribatid mites (Acari: Oribatida) in Gharbia governorate, Egypt. Delta J. Sci., 7(2): 734-755.
- Abdurakhmanov G.M., Grikurova A.A., Subías L.S., Shtanchaeva U.Ya., Kuramagomedov B.M. 2013. Species composition and zoogeographical characteristics of the armored mites (Acariformes, Oribatida) of coast and islands of north-western part of the Caspian Sea. Ecology of animals. The South of Russia: Ecol., Develop., 1: 16-21. https://doi.org/10.18470/1992-1098-2013-1-16-21
- Akrami M.A. 2015. An annotated checklist of oribatid mites (Acari: Oribatida) of Iran. Zootaxa, 3963(4): 451-501. https://doi.org/10.11646/zootaxa.3963.4.1
- Akrami M.A., Behmanesh M. 2015. Oribatid mites (Acari: Oribatida) of Shiraz township, Fars province, Iran. Entomofauna, 36: 377-396.
- Akrami M.A., Salehi Sarbizhan A.R. 2020. Oribatid mites (Acari: Oribatida) of Jiroft County, Kerman province, Iran: Introduction of fifty-six species, new records for the fauna of the province. Munis Ent. Zool., 15(1): 73-84.
- Akrami M.A., Shahedi A. 2020. Oribatid mites (Acari: Oribatida) of Taft county, Yazd province of Iran, with new records. Pers. J. Acarol., 9(2): 141-160. https://doi.org/10.22073/pja.v9i2.58955
- Al-Khalifa M.S., Bayoumi B.M. 1983. New records of soil Acarina and Collembola in Riyadh region. J. Coll. Sci. King Saud Univ., 14(1): 57-61.
- Arillo A., Subías L.S. 1996. La subfamilia Medioppiinae en el Sur de España. Descripción de Medioppia pinsapi sp. nov. (Oppiidae). Estud. Mus. Cien. Nat. Álava (1995-1996), 10-11: 211-221.
- Ay Y., Ayyildiz N. 2019. Investigations on oppioid oribatid mites of Amanos Mountains (Turkey). Plant Prot. Bul., 59(3): 15-23. https://doi.org/10.16955/bitkorb.503754
- Balogh J. 1983. A partial revision of the Oppiidae Grandjean, 1954 (Acari: Oribatei). Acta Zool. Acad. Sci. Hung., 29(1-3): 1-79.
- Balogh J., Mahunka S. 1974. Oribatid species (Acari) from Malaysian soils. Acta Zool. Acad. Sci. Hung., 20(3-4): 243-264.
- Bayoumi B.M., Al-Khalifa M.S. 1985. Three new oribatid mites (Acari, Oribatei) from Saudi Arabia. Bul. Soc. Amis Sci. Let. Poznan, Ser. D., Poznan, 25: 119-126.
- Boucher S. 2005. Évolution et phylogénie des Coléoptères Passalidae (Scarabaeoidea). Ann. Soc. Ent. Fr., 41: 239-604. https://doi.org/10.1080/00379271.2005.10697444
- Ermilov S.G., Corpuz-Raros L.A. 2017. New data on oribatid mites of the family Oppiidae (Acari: Oribatida) from the Philippines. Acta Zool. Bulg., 69(1): 9-16. https://doi.org/10.11646/zootaxa.4171.1.3
- Ermilov S.G., Frolov A.V. 2021. New data on oribatid mites (Acari, Oribatida) phoretic on passalid beetles (Coleoptera, Passalidae) from the Afrotropical and Oriental regions, with descriptions of three new species from Congo, Gabon and Ghana. Syst. Appl. Acarol., 26(4): 769-787. https://doi.org/10.11158/saa.26.4.8
- Ermilov S.G., Khaustov A.A. 2018. A contribution to the knowledge of oribatid mites (Acari, Oribatida) of Zanzibar. Acarina, 26(2): 151-159. https://doi.org/10.21684/0132-8077-2018-26-2-151-159
- Ermilov S.G., Rybalov L.B. 2022. Taxonomic contribution to the knowledge of the oribatid mite genus Arcoppia (Acari, Oribatida, Oppiidae). Int. J. Acarol., 48(7): 588-593. https://doi.org/10.1080/01647954.2022.2149854
- Ermilov S.G., Anichkin A.E., Wu D. 2012. Two new species of the genus Papillacarus (Acari: Oribatida: Lohmanniidae) from caves of Southern Vietnam. Zootaxa, 3593: 75-88. https://doi.org/10.11646/zootaxa.3593.1.4
- Ermilov S.G., Hugo-Coetzee E.A., Khaustov A.A. 2021. Contribution to the knowledge of Geminoppia (Acari, Oribatida, Oppiidae), with description of a new species from South Africa. Acta Zool. Acad. Sci. Hung., 67(3): 211-222. https://doi.org/10.17109/AZH.67.3.211.2021
- Fujikawa T. 1982. Oribatid fauna from nature farm in Nayoro (5). Edaphologia, 27: 1-4.
- Hammer M. 1958. Investigations on the oribatid fauna of the Andes Mountains. I. The Argentine and Bolivia. Biol. Skr. Kong. Dansk. Vidensk. Selsk., 10: 1-129.
- Hammer M. 1979. Investigations on the oribatid fauna of Java. Biol. Skr. Kong. Dansk. Vidensk. Selsk., 22 (9): 1-78.
- Hugo-Coetzee E.A., Lotfollahi P., Abbasi-Kalo A., Movahedzade E. 1917. A new species of Oxyoppia (Oxyoppiella) (Acari: Oribatida: Oppiidae) from Iran. Turk. J. Zool., 41: 630-637. https://doi.org/10.3906/zoo-1611-32
- Ivan O. 2007. Diversity and distribution of the oribatid mites (Acari, Oribatida) in some lawn ecosystems from Moldavian Plain (Romania). Studii si Comunicari, Complexul Muzeal de Stiintele Naturii "Ion Borcea» Bacau, 21: 236-243.
- Kulijev K.A. 1962. Fifteen new representatives of oribatid mites (Acariformes, Oribatei) of the genera Oppia and Ceratozetes. Trudy Azerbaijan NIVI, 13: 250-268.
- Mahunka S., Mahunka-Papp L. 1999. Oribatids from the Szarvas Arboretum (SE Hungary) (Acari: Oribatida). Folia Ent. Hung., 60: 83-107. https://doi.org/10.5962/bhl.part.80144
- Mahunka S., Murányi D., Kontschán J. 2013. The role of the Balkan Peninsula in the origin and genesis of the soil fauna of the Carpathian Basin: history, aims and results. Opusc. Zool. Budapest, 44 (suppl. 1): 5-10.
- Manning M., Halliday R.B. 1994. Biology and reproduction of some Australian species of Macrochelidae (Acarina). Aus. Ent., 21: 89-94.
- Maraun M., Caruso T., Hense J., Lehmitz L., Murvanidze M., Nae I., Schulz J., Seniczak A., Scheu S. 2019. Parthenogenetic vs. sexual reproduction in oribatid mite communities. Ecol. Evol. 9(12): 7324-7332. https://doi.org/10.1002/ece3.5303
- Movahedzade E., Lotfollahi P., Abbasi A. 2016. Some oppiid mites (Acari: Oribatida) of East Azerbaijan province, with two new records for the fauna of Iran. Pers. J. Acarol., 5 (2): 153-158.
- Murvanidze M., Mumladze L. 2016. Annotated checklist of Georgian oribatid mites. Zootaxa, 4089 (1): 1-81. https://doi.org/10.11646/zootaxa.4089.1.1
- Norton R.A. 1977. A review of F. Grandjean's system of leg chaetotaxy in the Oribatei (Acari) and its application to the family Damaeidae. In: Dindal D.L. (Ed.). Biology of oribatid mites. Syracuse: SUNY College of Environmental Science and Forestry. pp. 33-61.
- Norton R.A., Behan-Pelletier V.M. 2009. Oribatida. Chapter 15. In: Krantz G.W., Walter D.E. (Eds.). A Manual of Acarology. Lubbock: Texas Tech University Press. pp. 430-564.
- Noto la Diega R., Caruso N., Bernini F. 2004. Oribatid mites of the Pelagian Islands in the southern Mediterranean Sea: a faunistic and biogeographical study. Phytophaga, XIV: 513-518.
- Paoli G. 1908. Monografia del genere Dameosoma Berl. e generi affini. Redia, 5: 31-91.
- Pérez-Íñigo C. 1965. Especies españolas del género Oppia C.L. Koch (Acari, Oribatei). Bol. Real Soc. Esp. Hist. Nat., 62: 385-416.
- Pérez-Íñigo С. 1971. Acaros oribátidos de suelos de España peninsular e islas Baleares (Acari, Oribatei). Parte III. Eos, 46: 263-350.
- Poltavskaya M.P. 1994. A new genus and new species of oribatid mites from the virgin steppes of the Palaearctic (Acariformes, Oribatei). Zool. Zh., 73(3): 138-141.
- Schatz H. 2006. Catalogue of known oribatid mite species (Acari: Oribatida) from the Central American landbridge (First part). Trop. Zool., 19(2): 209-288.
- Schuster R. 1960. Über die Morphologie und Artengliederung der Gattung Epilohmannia Berlese 1917 (Acari, Oribatei). Zool. Anz., 165(5/6): 197-213.
- Shereef G.M. 1976. Oppia sitnikovae, a new species of oribatid mite in Giza region, Egypt. Bul. Zool. Soc. Egypt, 28: 8-9.
- Shtanchaeva U.Ya., Subías L.S. 2010. Catalogue of oribatid mites of the Caucasus. Makhachkala: Dagestan Sci. Center, Rus. Acad. Sci. 276 p. [In Russian]
- Smelyansky I.E. 2010. Structure of oribatid mite (Acariformes, Oribatida) assemblages along a vertical soil gradient in the steppe habitats of the Trans-Volga Region (Samara Province, Russia). Euroasian Ent. J., 9(2): 206-222.
- Stary J. 2006. Oribatid mites (Acari: Oribatida) of some localities in Kokořínsko Protected Landscape Area, Czech Republic. Bohemia Centralis, 27: 143-160.
- Subías L.S. 2004. Listado sistemático, sinonímico y biogeográfico de los ácaros oribátidos (Acariformes, Oribatida) del mundo (1758-2002). Graellsia, 60: 3-305. https://doi.org/10.3989/graellsia.2004.v60.iExtra.218
- Subías L.S. 2022. Listado sistemático, sinonímico y biogeográfico de los ácaros oribátidos (Acariformes, Oribatida) del mundo (excepto fósiles). Monogr. Electr. S.E.A., 12: 1-539.
- Subías L.S., Arillo A. 2001. Acari, Oribatei, Gymnonota II. Oppioidea. In: Ramos A. et al. (Eds.). Fauna Iberica. Vol. 15. Madrid: Museo de Ciencias Naturales, 289 pp.
- Subías L.S., Rodríguez P. 1986. Oppiidae (Acari, Oribatida) de los Sabinares (Juniperus thurifera) de España. IX. Subiasella (Lalmoppia) n. subgen. y Discoppia (Cylindroppia) n. subgen. Rev. Biol. Univ., 4: 111-121.
- Subías L.S., Shtanchaeva U.Ya. 2023. Claves de familias, géneros y subgéneros de ácaros oribátidos del mundo (Acari, Oribatida). Monogr. Electr. S.E.A., 13: 1-290.
- Subías L.S., Shtanchaeva U.Ya., Arillo A. 2013. Oribátidos (Acari, Oribatida) de España peninsular e islas Baleares. Distribución. Monogr. Electr. S.E.A., 5: 1-255.
- Subias L.S., Zaballos J.P., Banda E., Fontal-Cazalla F., Nieves-Aldrey J.L. 2004. Ácaros oribátidos (Acari, Oribatei) del Parque Nacional de la isla de Coiba, Panamá. Rev. Biol. Trop., 52(1): 85-95. https://doi.org/10.15517/rbt.v52i1.14755
- Travé J., Vachon M. 1975. François Grandjean. 1882-1975 (Notice biographique et bibliographique). Acarologia, 17(1): 1-19.
- Vasiliu N., Ivan O. 1996. Oribatids, bioindicators of the pollution of agricultural soils. Lucrarile Stiintifice ale celei de a 2-a conferinte Nat. Pentru Prot. Mediului: 370-376.
- Walter D.E., Lindquist E.E. 1995. The distribution of parthenogenetic ascid mites (Acari: Parasitiformes) do not support the biotic uncertainty hypothesis. Exp. Appl. Acarol., 19: 423-442. https://doi.org/10.1007/BF00048262
- Weis-Fogh T. 1948. Ecological investigations on mites and Collemboles in the soil (Appendix: Description of some new mites (Acari)). Nat. Jutland., Aarhus, 1: 135-270.



2023-04-23
Date accepted:
2023-06-16
Date published:
2023-06-26
Edited by:
Pfingstl, Tobias

This work is licensed under a Creative Commons Attribution 4.0 International License
2023 Ermilov, Sergey G. and Makarova, Olga L.
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



