New locality and host records of mites and ticks (Chelicerata: Acari) parasitizing lizards of Morocco
Er-Rguibi, Omar  1
; Laghzaoui, El-Mustapha
1
; Laghzaoui, El-Mustapha  2
; Aglagane, Abdessamad
2
; Aglagane, Abdessamad  3
; Kimdil, Latifa
3
; Kimdil, Latifa  4
; Stekolnikov, Alexandr A.
4
; Stekolnikov, Alexandr A.  5
; Abbad, Abdelaziz
5
; Abbad, Abdelaziz  6
and El Mouden, El Hassan
6
and El Mouden, El Hassan  7
7
1✉ Laboratory of Water, Biodiversity and Climate Change, Faculty of Sciences Semlalia, Cadi Ayyad University, Marrakech, Morocco.
2Laboratory of Water, Biodiversity and Climate Change, Faculty of Sciences Semlalia, Cadi Ayyad University, Marrakech, Morocco.
3Laboratory of Biodiversity and Ecosystem Functioning, Faculty of Sciences, Ibn Zohr University, Agadir, Morocco.
4Laboratory of Water, Biodiversity and Climate Change, Faculty of Sciences Semlalia, Cadi Ayyad University, Marrakech, Morocco.
5Laboratory of Parasitic Arthropods, Zoological Institute of the Russian Academy of Sciences, St. Petersburg, Russia.
6Laboratory of Microbial Biotechnologies, Agrosciences and Environment, Faculty of Sciences Semlalia, Cadi Ayyad University, Marrakech, Morocco.
7Laboratory of Water, Biodiversity and Climate Change, Faculty of Sciences Semlalia, Cadi Ayyad University, Marrakech, Morocco.
2023 - Volume: 63 Issue: 2 pages: 464-479
https://doi.org/10.24349/j4lz-jxdkOriginal research
Keywords
Abstract
Introduction
Information on host-arthropod associations and data on arthropods acting as vectors of pathogens is important for modeling and predicting zoonotic disease outbreaks (Benelli & Duggan 2018). In addition, studying parasite communities can help us better understand the structure and functioning of natural ecological systems. Such knowledge is of particular importance when host species are endemic, but of threatened conservation status (Er-Rguibi et al. 2021). Remarkably, more than 400 species of ectoparasites (mites and ticks of the subclass Acari) have been recorded parasitizing reptiles worldwide (Fajfer 2012). Reptilian ectoparasites belong to three Acari orders: Mesostigmata, Metastigmata and Prostigmata, and are represented by 15 families of temporary or permanent parasites (Fajfer 2012). Mites and ticks attached to the host skin extract blood and fluids from the host tissues, inducing local lesions, such as excoriation, papules, scaling, and inflammation (Lehmann 1993). Infestation with a high number of ectoparasites can reduce the density of blood cells, and consequently affect host physiology and behavior (Main & Bull 2000; Huyghe et al. 2010). These impacts depend in part on the intensity of infestation and where the ectoparasites are attached (Chilton et al. 1992; Bertrand et al. 1995). In addition, ectoparasites can be vectors of many other parasites and pathogens, such as haemogregarines, viruses, bacteria, and helminths, all of which can have strong negative effects on host body condition, survival and reproduction (Lehmann 1993; Smallridge & Bull 2000; Holmstad et al. 2008).
Morocco is a hot spot of reptilian biodiversity and endemism in North Africa and in the western Mediterranean region (Bons & Geniez 1996; Bouazza et al. 2021). The new updated checklist for Morocco showed the presence of 80 species of lizards, from 10 families including Agamidae (7 species), Anguidae (1 species), Gekkonidae (7 species), Lacertidae (25 species), Phyllodactylidae (8 species), Scincidae (19 species), and Sphaerodactylidae (9 species) (Bouazza et al. 2021). However, host-parasite interactions between Acari and lizards in Morocco have received limited attention, even if several families of Acari have been described as reptile ectoparasites worldwide (Fajfer 2012; Heath & Whitaker 2015; Mendoza-Roldan et al. 2020a). To date, two families of Trombidiformes were recorded on lizards in Morocco: Pterygosomatidae and Trombiculidae, with 17 species from 12 genera (Jack 1961, 1962; Jack & Girot 1965; Girot 1968; Stekolnikov 2018a, 2018b; Fajfer 2020). Moreover, one tick family has been reported, Ixodidae, with 3 species belonging to 2 genera (Bailly-Choumara et al. 1976). However, little information is currently available on their patterns of infestation (prevalence and intensity) and geographical distribution in Morocco.
The purpose of this article is therefore to: a) evaluate the overall prevalence and intensity of infestation by Acari in 27 lizard species in order to better understand host-ectoparasite relationships in these species; b) establish which ectoparasites most frequently infest lizards in Morocco, and evaluate their levels of co-infestation, c) examine parasitic niches and preferred attachment sites to better understand host exploitation, and d) evaluate the relationship between patterns of infestation (prevalence and intensity) and some specific host and environmental factors that may determine infestation (host species, sex, age class, host size, altitude and season).
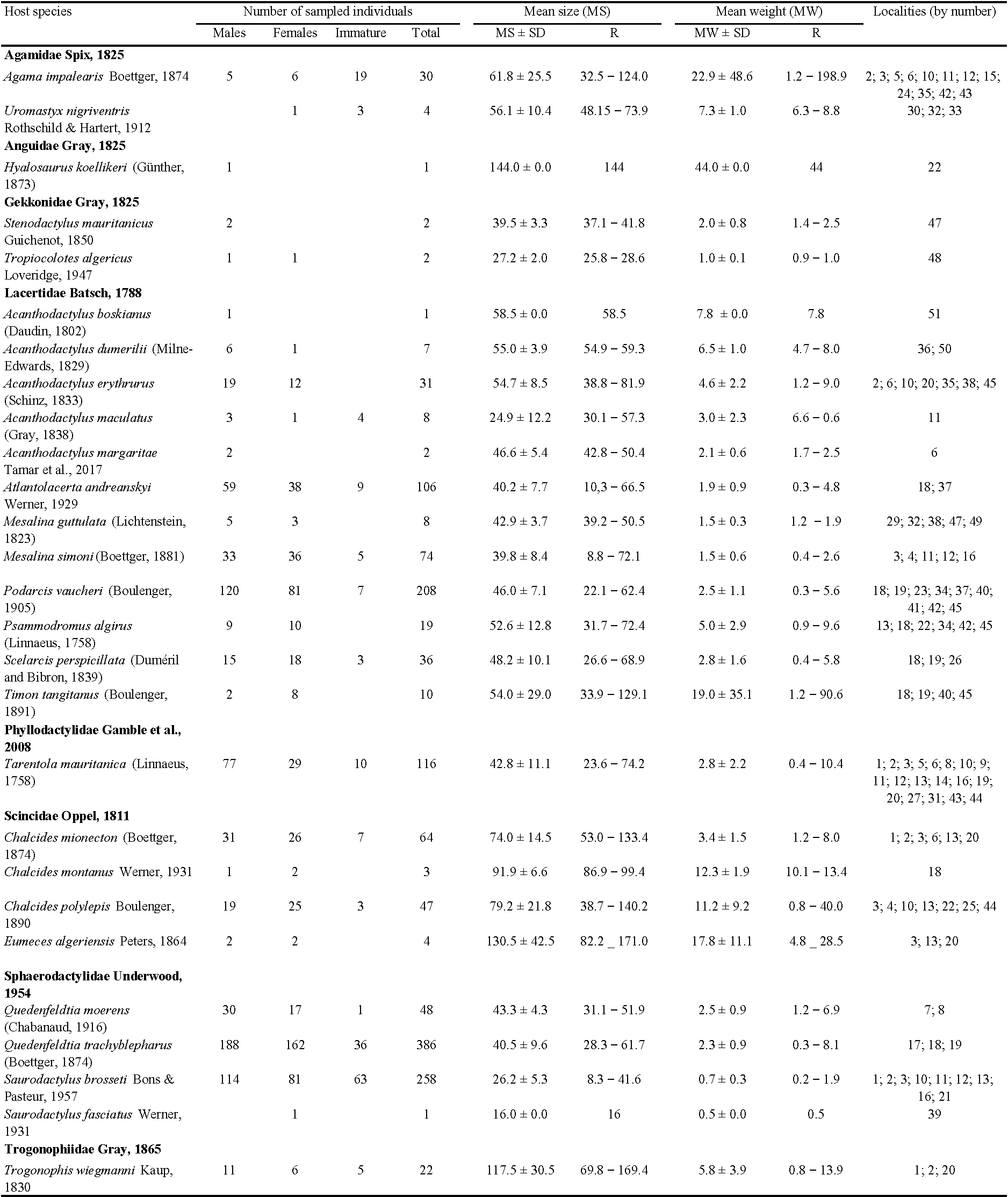
Material and methods
Sample collection



Lizard specimens were collected between 2019 and 2021 in Morocco. Individuals were randomly captured by hand or using a noose in 51 prospected localities (Figure 1). The study sites were visited twice per year (in spring and summer) for the three years of the study. A total of 1498 lizards were collected, comprising 27 species, 18 genera, and 8 families (Table 1). After capture, lizards were identified, weighted with a digital scale (± 0.01 g), measured (Snout-vent Length ''SVL'') using digital calipers (± 0.01mm), sexed according to morphological characteristics (Bons & Geniez 1996), and digitally photographed. Coordinates of the collection localities were registered using a GPS device. After measurements, lizards were immediately examined for mite and tick infestation. For the infested individuals, the number, the type of parasite (tick/mite), and the occupied niche were recorded (head, front and hind legs, back, vent, cloaca, and tail). The ectoparasites were then removed delicately using entomological tweezers and conserved in absolute ethanol (99.8%) for later identification. Lizards were released at the exact location of capture.


Ectoparasite identification
In the laboratory, mites and ticks were identified according to morphological characteristics based on original descriptions and systematic reviews (Jack 1961, 1962; Girot 1968; Bertrand et al. 1995, 2000, 2013; Moraza et al. 2009; Walker et al. 2014; Estrada-Peña et al. 2017; Stekolnikov 2018a, 2018b; Fajfer 2020; Stekolnikov et al. 2021). Macronyssidae, Pterygosomatidae, and ticks (Ixodidae) were mounted on microscope slides using Berlese's medium (Walter and Krantz 2009). The slides were examined under a stereomicroscope (DM-143-FBLED-A5) and microscope (Euromex BScope series) using different lenses. Slides of chigger mites (Acariformes: Trombiculidae) were examined under a Leica DM 2500 compound microscope (Leica Microsystems GmbH, Wetzlar, Germany) using differential interference contrast (DIC). Microphotographs were taken by means of a Leica DMC 4500 digital camera. Identified mites and ticks were deposited in the collection of the Laboratory of Water, Biodiversity and Climate Changes (WaterBiodiCC), Faculty of Sciences, Semlalia, Cadi Ayyad University, Marrakech. Specimens of chigger mites were also deposited in the Zoological Institute of the Russian Academy of Sciences (ZIN, Saint Petersburg, Russia).
Data analysis
To evaluate the infestation data, two parasitological parameters were calculated using the Quantitative Parasitology software, version 3.0 (Rózsa et al. 2000): 1) infestation prevalence (%) (P) = 100 × (number of infested lizards/total number of lizards); 2) mean intensity (MI) = number of parasites/number of infested lizards. The values of P and MI are given with 95% confidence interval (CI) and the range of infestation values.
To examine the effect of different intrinsic [host species (infested species), sex (two levels), age class (two levels), body size ''SVL'' (continuous)], and extrinsic [season (two levels), altitude (continuous)] variables on ectoparasite prevalence a generalized linear model (GLZM) was used, with parasite prevalence as the dependent variable, following a binomial distribution, with size as a covariate, and including the interactions between different factors in the model. Likewise, the infestation intensity was studied using a general linear model (GLM), with parasite intensity as the dependent variable, size as a covariate, and with the aforementioned factors and their interactions. Some of the data concerning Quedenfeldtia trachyblepharus (n=227) were already published in Er-Rguibi et al. (2021). Separate tests were performed for each family of ectoparasites to investigate the impact of the six studied factors and their interactions on ectoparasitism patterns. We used a two-way ANOVA test to examine differences in parasitic niche use on the host among ectoparasite families. All statistical tests were performed using SPSS Statistics 19.0 software (SPSS, Chicago, IL, USA), and a p value \textless0.05 was considered statistically significant.
Results
Mite and tick diversity



From the 1498 lizards examined, 335 were found infested with ectoparasites, with an overall prevalence of 22.4% and mean intensity of 14.5 (95% confidence interval: 12.2 – 16.8). Three mite families (Macronyssidae, Pterygosomatidae, and Trombiculidae) and one tick family (Ixodidae) were found in the sampled lizards. Trombiculid mites presented the highest prevalence of infestation (17.2%), followed by Pterygosomatidae (4.1%), Ixodidae (1.5%), and Macronyssidae (0.4%). However, the latter showed the highest mean intensity 31.7 (14.3 – 54.2), followed by Pterygosomatidae 22.0 (16.2 – 30.5), Trombiculidae 12.7 (10.9 – 14.8), and Ixodidae 1.7 (1.3 – 2.2) (Table 2). The spatial distribution of ectoparasites infesting Moroccan lizards is shown in Figure 1.
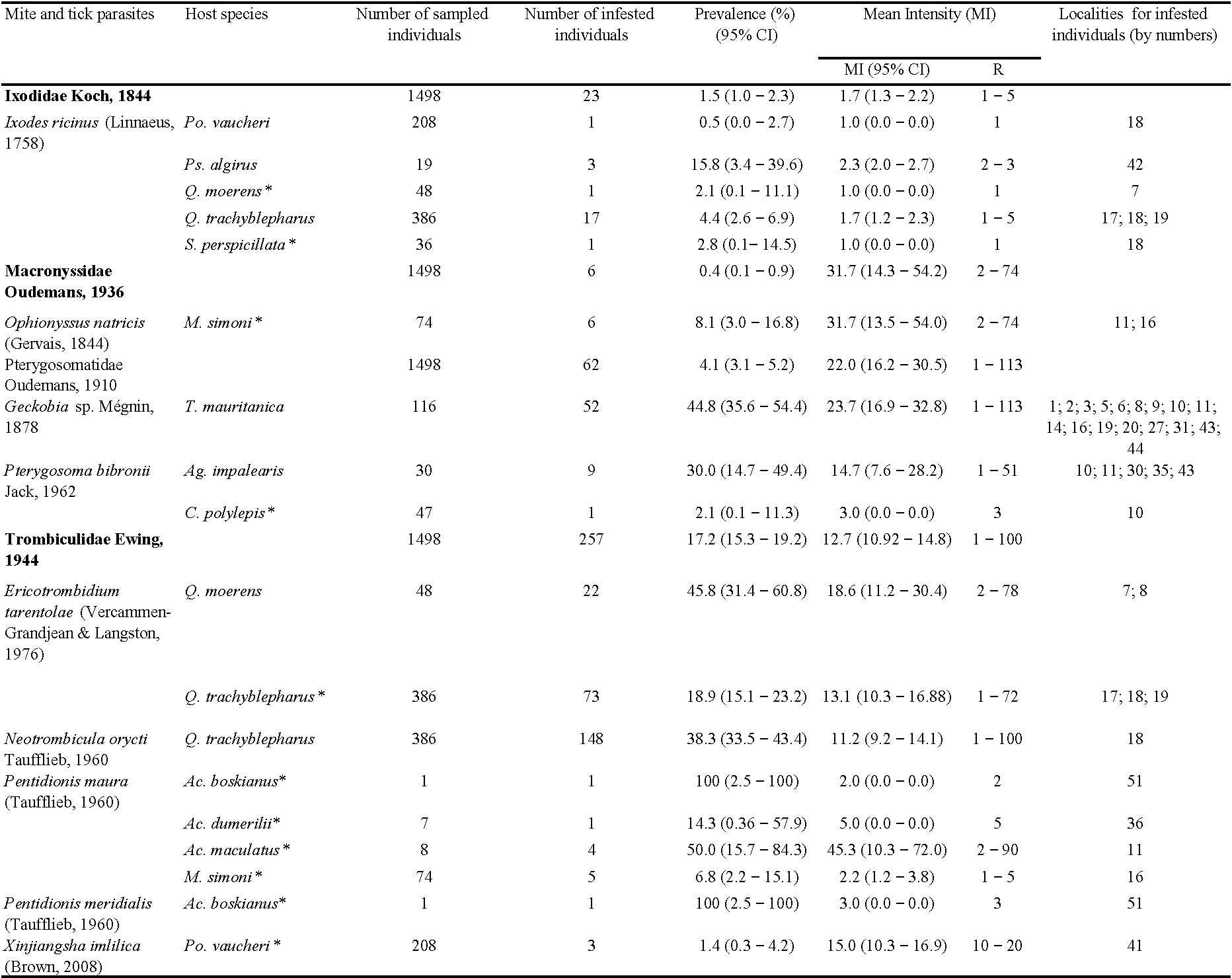


The family Ixodidae, represented by larvae and nymphs of Ixodes ricinus, were found infesting five lizard species: Podarcis vaucheri, Psammodromus algirus (Figure 2A), Scelarcis perspicillata, Quedenfeldtia moerens (Figure 2B), and Q. trachyblepharus (Figures 2C), with different patterns of infestation (Table 2). The Macronyssidae, represented by Ophionyssus natricis, was recorded for the first time in Morocco, and was found infesting Mesalina simoni. The Pterygosomatidae were represented by two species: an unidentified species of the genus Geckobia infesting Tarentola mauritanica (Figure 3A), and Pterygosoma bibronii that was found on Agama impalearis (Figure 3B) and Chalcides polylepis. The infestation of C. polylepis is the first record of scale mites on the genus Chalcides and for the family Scincidae. The Trombiculidae infesting lizards in Morocco (Figure 3C – D) were represented by five species – Ericotrombidium tarentolae, Neotrombicula orycti, Pentidionis maura, Pentidionis meridialis, and Xinjiangsha imlilica (Figure 4). Seven new host records for these species were reported (Table 2). In addition, co-infestation by two ectoparasites was recorded in three host species. Twelve of 386 examined individuals of the Atlas day gecko Q. trachyblepharus were simultaneously infested with the tick I. ricinus and one of the two chigger mites: E. tarentolae or N. orycti. One of 74 examined individuals of M. simoni was infested with O. natricis and P. maura. Finally, the one examined individual A. boskianus was parasitized by P. maura and P. meridialis.



The number of individuals and lizard species examined and infested by different Acari families, as well as their patterns of infestation, are summarized in Table 2. The highest prevalence of infestation was recorded in A. maculatus, where 50.0% of individuals were infested with the chigger species P. maura. In addition, notable cases of infestation were those of Q. moerens by E. tarentolae (45.8%) and T. mauritanica by Geckobia sp. (44.8%) (Table 2). The lowest infestation was recorded in P. vaucheri infested with the tick I. ricinus (0.5%). The maximum mean intensities were observed in A. maculatus infested with P. maura 45.3 (10.3 – 72.0), M. simoni infested with O. natricis 31.7 (13.5 – 54.0) and T. mauritanica infested with Geckobia sp. 23.7 (16.2 – 32.8). The lowest mean intensities were recorded for P. vaucheri, Q. moerens, and S. perspicillata infested with I. ricinus 1.0 (CI uncertain).



Effects of host species, host size, sex, age class, altitude, and season on ectoparasitism
The prevalence of infestation by Ixodidae was related to host species (GLZM, Wald's χ2 = 10.639, df = 4, p = 0.014) and altitude (Wald's χ2 = 34.166, df = 20, p = 0.018). No other factor nor their interactions were found to significantly affect the prevalence of Ixodidae infestations (Table 3). Likewise, the intensity of infestation by this tick family was linked only to host species (GLM, r2 = 0.101, F~(4, 692)~ = 4.574, p = 0.004) and altitude (F~(20, 676)~ = 1.904, p = 0.012) (Table 4).
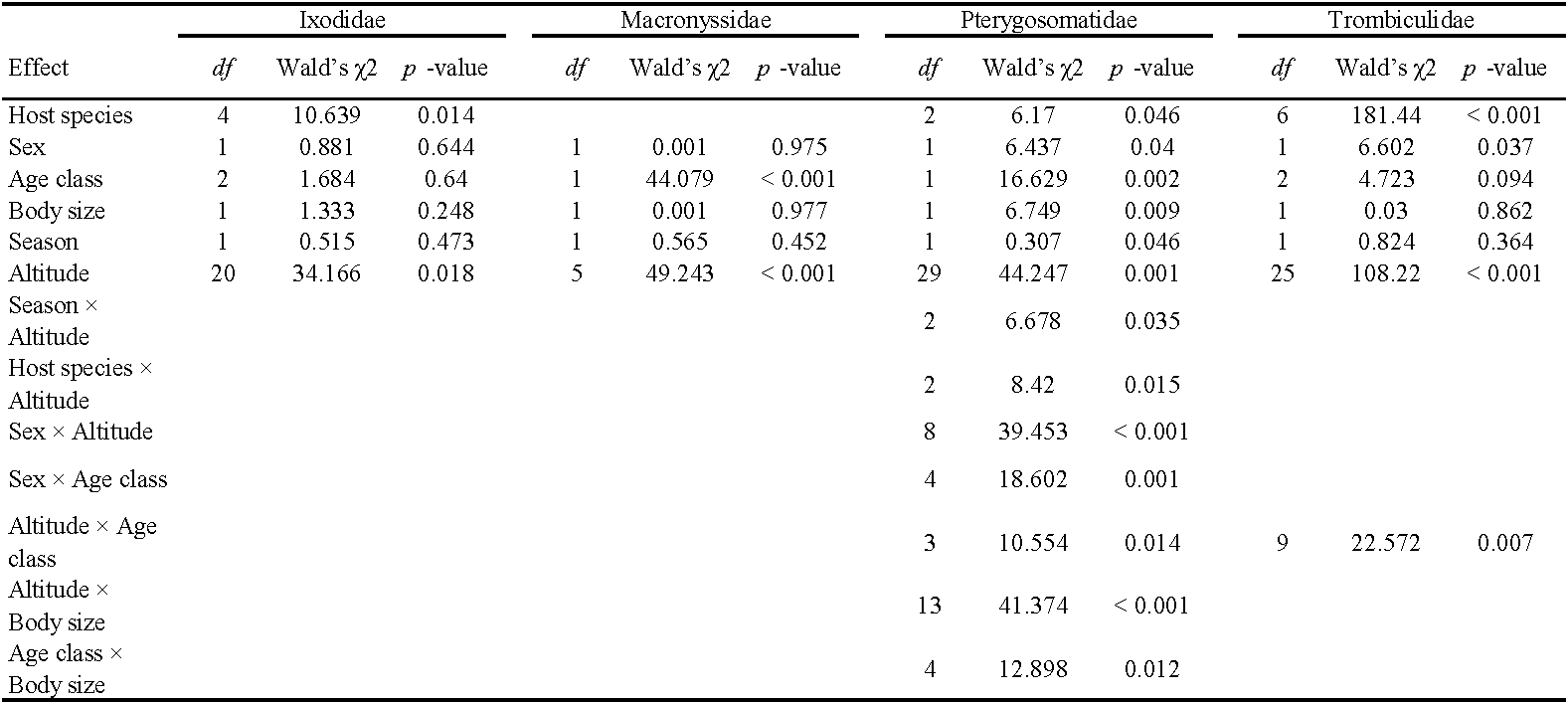


The prevalence of Macronyssidae infestation was related to age class (GLZM, Wald's χ2 = 44.079, df = 1, p < 0.001) and altitude (Wald's χ2 = 49.243, df = 5, p < 0.001), but was not related to the other studied factors (Table 3). The intensity of infestation by Macronyssid mites significantly differed between males and females (GLM, r2 = 0.930, F~(1, 72)~ = 4.718, p = 0.035), adults and juveniles (F~(1, 72)~ = 4.848, p = 0.033), altitude (F~(5, 68)~ = 10.539, p < 0.001), and season (F~(1, 72)~ = 6.478, p = 0.014). Likewise, the interactions between altitude × SVL, altitude × season, sex × SVL, and season × sex were statistically significant (Table 4). All the other non-statistically significant interactions were removed from the model.
The prevalence of Pterygosomatidae infestation was related to host species (GLZM, Wald's χ2 = 6.170, df = 2, p = 0.046), sex (Wald's χ2 = 6.437, df = 1, p = 0.040), age class (Wald's χ2 = 16.629, df = 1, p = 0.002), body size (Wald's χ2 = 6.749, df = 1, p = 0.009), and altitude (Wald's χ2 = 44.247, df = 29, p = 0.001). Furthermore, the interactions between season × altitude, host species × altitude, sex × altitude, sex × age classes, altitude × age classes, altitude × SVL, and age classes × SVL were statistically significant (Table 3). All the other non-statistically significant interactions were removed from the model. The intensity of infestation by Pterygosomatidae was linked to host species (GLM, r2 = 0.625, F~(2, 190)~ = 4.350, p = 0.016), sex (F~(1, 192)~ = 7.025, p = 0.002), age class (F~(1, 192)~ = 4.065, p = 0.005), and body size (F~(1, 192)~ = 11.983, p = 0.001). Only the interactions between host species × altitude and altitude × SVL were statistically significant (Table 4), all the other interactions were not significant and were removed from the model.
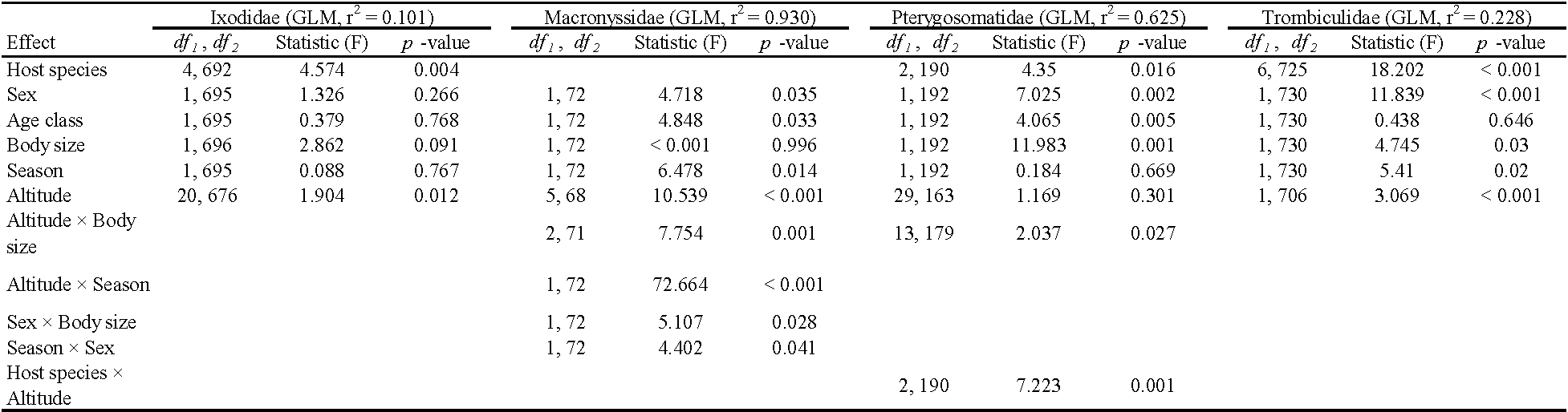


The prevalence of Trombiculidae infestation was significantly different between host species (GLZM, Wald's χ2 = 181.440, df = 6, p < 0.001), males and females (Wald's χ2 = 6.602, df = 1, p = 0.037), and altitude (Wald's χ2 = 108.220, df = 25, p < 0.001). Only the interaction between altitude × age classes was statistically significant; all other interactions were removed from the model (Table 3). The intensity of infestation by chigger mites was related to host species (GLM, r2 = 0.228, F~(6, 725)~ = 18.202, p < 0.001), sex (F~(1, 730)~ = 11.839, p < 0.001), body size (F~(1, 730)~ = 4.745, p = 0.030), altitude (F~(24, 706)~ = 3.069, p < 0.001), and season (F~(1, 730)~ = 5.410, p = 0.020) (Table 4). The interactions between analyzed factors were not statistically significant and were removed from the model.
Parasitic niches and preferred locations
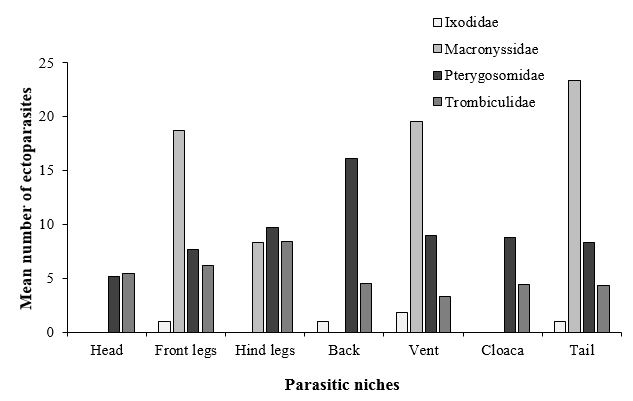
Concerning the parasitic niche on the animal (Figure 5), an important difference was observed in the attachment sites of the different ectoparasite families (two-way ANOVA test: Ectoparasite family F~(3, 720)~ = 10.51, p < 0.001; Parasitic niche F~(6, 717)~ = 3.336, p = 0.003; Interaction F~(18, 705)~ = 1.85, p = 0.017). The tick I. ricinus was found mainly attached on the ventral side of lizard hosts (vent) (Figure 2B). The Macronyssid mites O. natricis were found attached under the scales on the ventral side of front and hind legs, and on vent and tail of M. simoni. The Pterygosomatidae were found attached in different parts of the host body (head, front and hind legs, back, vent, cloaca, and tail). Mites of the genus Geckobia were found in different parts of T. mauritanica, especially on the dorsal side, between toes (Figure 3A), at the base of the claws, and in clusters near the cloaca. P. bibronii attached mainly under the scales of the vent, throat, and tail of A. impalearis (Figure 3B), and under the scales of the vent in C. polylepis. Attached chigger mites formed clusters on different body parts, but were especially frequent in skin fold regions at the base of the legs, eyes, ears, cloaca and between the fingers (Figure 3C – D).


Discussion
To the best of our knowledge, this work represents the most comprehensive investigation of mites and ticks parasitizing natural populations of Moroccan lizards, in terms of the number of lizard species (27), the number of individuals examined (1498), and the geographical coverage. Trombiculidae and Pterygosomatidae were the most prevalent families exploiting Moroccan lizards, probably due to the high diversity of hosts in the region. Indeed, the last taxonomic update showed that Morocco has one of the highest levels of lizard biodiversity and endemism in the Western Palaearctic, with Agamidae (7 species), Phyllodactylidae (8 species), Gekkonidae (7 species), Sphaerodactylidae (9 species), and Lacertidae (25 species) (Bouazza et al. 2021). The high prevalence of Trombiculd (chigger) mites is likely due to their low host specificity (Azad 1986), whereas Pterygosomatid mites spend their entire life cycle on the host, from larva to adult (Bertrand et al. 1995; Bertrand et al. 2000). In addition, the origin of examined lizards (captive or wild) can affect the diversity of ectoparasites (Castro et al. 2019; Mendoza-Roldan et al. 2020a), with the Trombiculidae and Pterygosomatidae families being most prevalent in natural populations. The same finding was also shown in the examination of Brazilian lizards (Mendoza-Roldan et al. 2020a).
Only one species of Ixodidae, I. ricinus, was found in our study, infesting five lizard species. Two additional tick species were found previously in Moroccan lizards, I. ventalloi and Haemaphysalis sulcata (Bailly-Choumara et al. 1976) (Supplementary Table 1). The absence of these species in the present study may be due to changes in the presence of other host species involved in the life cycle. It is well known that I. ricinus is a broad host generalist, with immature stages readily exploiting lizards, small mammals, and birds (Bauwens et al. 1983; Medlock et al. 2013; Nicholson et al. 2019).
Our results indicated the presence of mites from the genus Ophionyssus in Moroccan lizards for the first time; these mites were found in two localities in the Marrakech-Safi region (Guemassa and Central Jbilet) on M. simoni with a prevalence of infestation of 8.1% (6/74) and a high mean intensity of 31.7 mites/infested lizard (2 ̶ 74 mites/infested lizard). This finding suggests that this mite uses M. simoni as a host species and our observations do not represent an accidental infestation event. This genus has been previously found in different lizard families (Agamidae, Scincidae, and Lacertidae) (Fain & Bannert 2000; Fajfer 2012), and snakes are actually considered the preferred host of these mites. It has been suggested that these mites exploit lizards when the preferred host is not available or when a wide variety of reptiles share the same habitat (Castro et al. 2019). This mite parasite is a mechanical vector for several blood-borne infectious diseases in reptiles (e.g. Aeromonas hydrophila, Anaplasma sp. and Rickettsia sp.) (Mendoza-Roldan et al. 2020b; Mendoza-Roldan et al. 2021). Unfortunately, it may also accidentally infest humans and can cause severe mite-associated dermatitis (Schultz 1975; Amanatfard et al. 2014).
In our study, the Pterygosomatidae were represented by two genera: Geckobia and Pterygosoma. These two genera have been reported in the literature parasitizing T. mauritanica (Girot 1968), and A. impalearis (Jack 1961; Bertrand et al. 2000; Fajfer 2020), respectively. (Supplementary Table 1). Previously, Pterygosoma were found infesting only Agamidae; here we reported this genus from Scincidae for the first time. However, this finding may represent an accidental infestation. Further examinations for infestation of Scincidae by Pterygosoma are needed to evaluate this relationship.
The Trombiculidae are the most diverse lizard ectoparasites in Morocco. Previously, 10 chigger genera were reported from Moroccan lizards: Acomatacarus, Ericotrombidium, Eutrombicula, Hypotrombidium, Matacarus, Neoschoengastia, Neotrombicula, Ornithogastia, Pentidionis, and Schoutedenichia (Supplementary Table 1) (Taufflieb 1958; Stekolnikov 2018b, 2019; Stekolnikov et al. 2021). Here, we found five chigger mite species of four genera infesting nine lizard hosts, a difference likely explained by the number of examined hosts compared to existent lizard diversity in Morocco (27/86) (Bouazza et al. 2021).
Previously, a series of studies reported the co-infestation of lizards by different mite and tick species (Girot 1968; Tomassone et al. 2017; Mendoza-Roldan et al. 2019). Indeed, the high diversity of parasitic species may lead to regular co-infestation events. In the present work, we recorded four cases of co-infestation. For instance, the presence of two closely related chigger species, P. maura and P. meridialis, on the same host individual.
In addition to host specificity, patterns of infestation may be related to other biotic and abiotic factors, including differences between the environmental conditions in the habitats (Álvarez-Ruiz et al. 2018; Mendoza-Roldan et al. 2020a), and host behavior and immunity (Eisen & Wright 2001; Hawley & Altizer 2011). The behavior of the host can, for example, influence the degree of exposure to parasites, which could explain the observed differences in the patterns of infestation (Sol et al. 2000). Indeed, in agreement with the findings of Gwiazdowicz et al. (2020); Coêlho et al. (2021); and Er-Rguibi et al. (2021), infestations by Macronyssidae, Pterygosomatidae, and Trombiculidae were significantly different between males and females. We attribute these differences to differences in behavior (Er-Rguibi et al. 2021; Er-Rguibi et al. 2022); adult males are likely more active and can occupy more favorable areas where they are exposed to the ectoparasites. In addition, the maintenance of high concentrations of testosterone during the mating season, may act as an immune suppressor effect (Olsson et al. 2000; Foo et al. 2017) which indirectly affects the ectoparasite loads of male lizards (Halliday et al. 2014). This steroid hormone may also increase activity, competition, and aggression with other males to maintain a territory and a female mate, which can again lead to higher exposure risks to mite and tick infestations (Olsson et al. 2000).
The prevalence and intensity of Macronyssidae, Pterygosomatidae, and Trombiculidae infestations were related to age class, host body size or both. The same result was found in previous studies (e.g. Scali et al. 2001; Obi et al. 2013; Gwiazdowicz et al. 2020; Coêlho et al. 2021; Er-Rguibi et al. 2021). Higher infestations in adult hosts may be due to their long exposure time to the parasite's infective stages (Obi et al. 2013; Er-Rguibi et al. 2021). Furthermore, the adult lizards occupy favorable habitats, which may increase the probability of contact with the parasites; juveniles are often limited to marginal areas and are not physically associated with their parents or siblings (Amo et al. 2004; Er-Rguibi et al. 2021). Finally, older lizards have large body sizes that facilitate more place for mite and tick fixation (Amo et al. 2004; Gwiazdowicz et al. 2020; Coêlho et al. 2021).
The prevalence and intensity of Ixodidae, Macronyssidae, Pterygosomatidae, and Trombiculidae infestations were linked to altitude. This is in line with the results of Álvarez-Ruiz et al. (2018) and Er-Rguibi et al. (2021), which indicated that ectoparasite infestations varied with elevation. This finding could be explained by changes in environmental conditions, such as humidity and temperature that affect ectoparasite survival, and consequently lead to geographic variation of host-ectoparasite relationships (Medlock et al. 2013; Poisot et al. 2017). Variation in abiotic and biotic conditions in high elevations could also cause longer hibernation durations, resulting in shorter periods of activity for both host and parasite to reproduce and grow (Comas & Ribas 2015). Long periods of lizard hibernation likely contribute to increasing winter ectoparasite mortality (Postawa & Nagy 2016).
A significant difference in infestation by Macronyssid and Trombiculid mites between seasons was observed. The same results were recorded in other studies (e.g. Huyghe et al. 2010; Er-Rguibi et al. 2021). This difference may be due to changes in lizard activity and behavior between seasons. For example, changes in testosterone concentrations during reproduction leads to strong variation in activity patterns which can modify exposure rates (Olsson et al. 2000; Foo et al. 2017). Moreover, as above, the modification of environmental conditions between seasons can alter lizard and ectoparasite interactions (Medlock et al. 2013; Poisot et al. 2017).
Regarding the on-host parasitic niches, the highest number of I. ricinus was found on the ventral side of lizards, where this tick could find an area protected from unfavorable conditions (temperature and wind) (Coêlho et al. 2021). The same result was found between the tick Amblyomma dissimile Koch, 1844 and its host Tropidurus oreadicus Rodrigues, 1987 in Brazil (Coêlho et al. 2021). Concerning the Macronyssidae, these mites were attached under the scales of the vent, legs, and tail. This result was also reported in previous studies, where Ophionyssus saurarum (Oudemans, 1901), infesting Lacerta viridis viridis (Laurenti, 1768), was found around the tail base and hind limb axillae (Masan et al. 2009). O. galloticolus Fain & Bannert, 2000, a parasite of Gallotia galloti eisentrauti Bischoff, 1982 from the Canary Island of Tenerife, was attached around the tail right behind the cloaca, and under the large scales of the belly (Bannert et al. 2000). The Pterygosomatidae were found in different parts of the host body. Thus, Geckobia sp. were found especially on back (dorsal side), between toes, at the base of the claws, and in clusters near the cloaca. The same attachment site was observed for G. loricata and G. latastei parasitizing T. mauritanica in Morocco (Girot 1968). Geckobia spp. infesting Hemidactylus frenatus Duméril & Bibron, 1836 in Colombia were observed mostly on the dorsum of the posterior legs and between the claws (Díaz et al. 2020). Concerning the scale-mite P. bibronii, they tended to be attached under the scales of the ventral side of hosts. However, other studies reported that species of this genus attached to multiple sites on the host body— under the scales of back, vent, tail, and legs (Mostafa 1974; Bertrand et al. 1995). This finding suggest that the body location of the mites of this group was not specified. Regarding the chigger mites, previous findings showed that they prefer small folds of skin in the cervical, axillary, and inguinal regions or the tail base of lizard hosts (Bertrand & Modrý 2004). In our study, chiggers were attached to different parts of the host body, mainly in clusters at the base of limbs, eyes, ears, cloaca and between fingers. For instance, P. maura was found attached in clusters at the tail base and at the leg base of A. maculatus. Thus, infesting these protected areas may help them either avoid being physically removed (either by host grooming or by physical contact with environmental substrates) or penetrate tissues more easily (because the host tissue is maybe more tender in these areas of the body).
In conclusion, Moroccan lizards were infested by nine species of ectoparasite of four mite families (Ixodidae, Macronyssidae, Pterygosomatidae, and Trombiculidae), with the dominance of Acari within the Trombidiformes order (Trombiculidae and Pterygosomatidae) in term of both prevalence and intensity of infestation. The diversity of ectoparasite species associated with lizards in Morocco remains low compared to Brazilian and New Zealand lizards (Heath & Whitaker 2015; Mendoza-Roldan et al. 2020a). This is likely linked to the higher diversity of hosts in these countries. Currently, much remains unknown concerning the impacts of the mite and tick parasites on lizard hosts, their determining factors, and the infectious agents they transmit. Thus, larger examination will be necessary to better understand the relationship between lizards and their ectoparasites.
Acknowledgements
We would like to thank ′Haut Commissariat aux Eaux et Forêts et à la Lutte Contre la Désertification (HCEFLCD)′ for the permit to work in the field. Financial support for fieldwork was provided by Cadi Ayyad University. Identification of chigger mites was supported by the Ministry of Science and Higher Education of the Russian Federation (Grant No. 122031100263-1, to AAS).
acarologia_4600_supplementary-table.xlsx
References
- Álvarez-Ruiz L., Megía-Palma R., Reguera S., Ruiz S., Zamora-Camacho F.J., Figuerola J., Moreno-Rueda G. 2018. Opposed elevational variation in prevalence and intensity of endoparasites and their vectors in a lizard. Curr. Zool., 64(2): 197-204. https://doi.org/10.1093/cz/zoy002
- Amanatfard E., Youssefi M.R., Barimani A. 2014. Human dermatitis caused by Ophionyssus natricis, a snake mite. Iran. J. Parasitol., 9(4): 594-596.
- Amo L, López P, Martín J. 2004. Prevalence and intensity of haemogregarinid blood parasites in a population of the Iberian rock lizard, Lacerta monticola. Parasitol. Res., 94(4): 290-293. https://doi.org/10.1007/s00436-004-1212-7
- Azad A. 1986. Vector control series training and information guide: XIII. Mites of public health importance and their control. World Health Organization, Vector Biology and Control Division, Genève.
- Bailly-Choumara H., Morel P.C., Rageau J. 1976. Sommaire des données actuelles sur les tiques du Maroc (Acari, Ixodoidea). Bull. l′Institut Sci., 1:101-117.
- Bannert B., Karaca H.Y., Wohltmann A. 2000. Life cycle and parasitic interaction of the lizard-parasitizing mite Ophionyssus galloticolus (Acari: Gamasida: Macronyssidae), with remarks about the evolutionary consequences of parasitism in mites. Exp. Appl. Acarol., 24(1): 597-613. https://doi.org/10.1023/A:1026504627926
- Bauwens D., Strijbosch H., Stumpel A.H.P. 1983. The lizards Lacerta agilis and L. vivipara as hosts to larvae and nymphs of the tick Ixodes ricinus. Holarct. Ecol., 6(1): 32-40. https://doi.org/10.1111/j.1600-0587.1983.tb01062.x
- Benelli G., Duggan M.F. 2018. Management of arthropod vector data - social and ecological dynamics facing the one health perspective. Acta Trop., 182:80-91. https://doi.org/10.1016/j.actatropica.2018.02.015
- Bertrand M., Modrý D. 2004. The role of mite pocket-like structures on Agama caudospinosa (Agamidae) infested by Pterygosoma livingstonei sp. n. (Acari: Prostigmata: Pterygosomatidae). Folia Parasitol., 51(1): 61-66. https://doi.org/10.14411/fp.2004.009
- Bertrand M., Paperna I., Finkelman S. 1995. Preliminary study on the genus Pterygosoma Peters, 1849 (Actinedida, Pterygosomidae): Complementary description of Pterygosoma bibronii Jack, 1962, and notes on evolutionary characters of Pterygosomidae. Acarologia, 36(2): 133-143.
- Bertrand M., Paperna I., Finkelman S. 2000. Descriptions and observations on the genera Ptergosoma, Geckobia and Hirstiella (Acari: Actinedida). Acarologia, 40(3): 275-304.
- Bertrand M., Kukushkin O., Pogrebnyak S. 2013. A new species of mites of the genus Geckobia (Prostigmata, Pterygosomatidae), parasitic on Mediodactylus kotschyi (Reptilia, Gekkota) from Crimea. Vestn. Zool., 47(2): 1-13. https://doi.org/10.2478/vzoo-2013-0009
- Bons J., Geniez P. 1996. Amphibiens et reptiles du Maroc (Sahara Occidental compris) Atlas biogéographique. In Barcelona: Asociación Herpetológica Española.
- Bouazza A., El Mouden E.H., Rihane A. 2021. Checklist of amphibians and reptiles of Morocco\,: A taxonomic update and standard Arabic names. Herpetol. Notes., 14(January): 1-14.
- Castro P.D.J., Pietzsch M., Pantchev N. 2019. Ectoparasites in captive reptiles. Vet. Nurse., 10(1): 33-41. https://doi.org/10.12968/vetn.2019.10.1.33
- Chilton N.B., Bull C.M., Andrews R.H. 1992. Differences in attachment site of the Australian reptile tick Amblyomma limbatum (Acari: Ixodidae) on two host species. Int. J. Parasitol., 22(6): 783-787. https://doi.org/10.1016/0020-7519(92)90128-8
- Coêlho T.A., Vasconcelos-Neto L.B.D., Ishiguro F.L., Chalkidis H.M., de Souza D.C. 2021. Ticks (Acari: Ixodidae) on Tropidurus oreadicus Rodrigues, 1987 in Monte Alegre, State of Pará, central-eastern Amazon. Int. J. Acarol., 47(1): 74-78. https://doi.org/10.1080/01647954.2020.1870550
- Comas M., Ribas A. 2015. Why are the prevalence and diversity of helminths in the endemic Pyrenean brook newt Calotriton asper (Amphibia, Salamandridae) so low? J. Helminthol., 89(2): 175-181. https://doi.org/10.1017/S0022149X13000710
- Díaz J.A., Torres R.A., Paternina L.E., Santana D.J., Miranda R.J. 2020. Traveling with an invader: ectoparasitic mites of Hemidactylus frenatus (Squamata: Gekkonidae) in Colombia. Cuad. Herpetol., 34(1): 79-82. https://doi.org/10.31017/CdH.2020.(2019-027)
- Eisen R.J., Wright N.M. 2001. Landscape features associated with infection by a malaria parasite (Plasmodium mexicanum) and the importance of multiple scale studies. Parasitology, 122(5): 507-513. https://doi.org/10.1017/S0031182001007636
- Er-Rguibi O., Laghzaoui E., Aglagane A., Kimdil L., Abbad A., El Mouden E.H. 2021. Determinants of prevalence and co-infestation by ecto- and endoparasites in the Atlas day gecko, Quedenfeldtia trachyblepharus, an endemic species of Morocco. Parasitol. Res., 120: 2543-2556 https://doi.org/10.1007/s00436-021-07120-z
- Er-Rguibi O., Bursey C.R., Laghzaoui E.M., Aglagane A., Kimdil L., Abbad A, El Mouden E.H. 2022. New host and locality records of helminths' infection of seven lizards from Morocco. Parasitol. Res., 1-10. https://doi.org/10.1007/s00436-022-07588-3
- Estrada-Peña A., Mihalca A.D., Petney T.N. 2017. Ticks of Europe and North Africa a guide to species identification. In Springer, Cham. Edited by Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge. pp. 1-30. https://doi.org/10.1017/CBO9781107415324.004
- Fain A., Bannert B. 2000. Two new species of Ophionyssus Mégnin (Acari: Macronyssidae) parasitic on lizards of the genus Gallotia Boulenger (Reptilia: Lacertidae) from the Canary Islands. Int. J. Acarol., 26(1): 41-50. https://doi.org/10.1080/01647950008683634
- Fajfer M. 2012. Acari (Chelicerata) - Parasites of reptiles. Acarina, 20(2): 108-129.
- Fajfer M. 2020. A systematic revision of the scale mite genus Pterygosoma Peters, 1849 (Acariformes: Pterygosomatidae). Zootaxa, 4805(1): 1-147. https://doi.org/10.11646/zootaxa.4805.1.1
- Foo Y.Z., Nakagawa S., Rhodes G., Simmons L.W. 2017. The effects of sex hormones on immune function: a meta-analysis. Biol. Rev., 92:551-571. https://doi.org/10.1111/brv.12243
- Girot B. 1968. Etude du cycle de Geckobia latastei et Geckobia loricata, acariens parasites du gecko Tarentola mauritanica. Vie Milieu, 19: 63-142.
- Gwiazdowicz D.J., Gdula A.K., Kurczewski R., Zawieja B. 2020. Factors influencing the level of infestation of Ixodes ricinus (Acari: Ixodidae) on Lacerta agilis and Zootoca vivipara (Squamata: Lacertidae). Acarologia, 60(2): 390-397. https://doi.org/10.24349/acarologia/20204372
- Gyawali P., Khanal S., Shrestha B. 2017. Intestinal helminth fauna in sleepy lizard (Tiliqua rugosa) in Australia. Int. J. Vet. Sci., 2(1): 17-20.
- Halliday W.D., Paterson J.E., Patterson L.D., Cooke S.J., Blouin-Demers G. 2014. Testosterone, body size, and sexual signals predict parasite load in Yarrow's Spiny Lizards (Sceloporus jarrovii). Can. J. Zool., 92(12): 1075-1082. https://doi.org/10.1139/cjz-2014-0256
- Hawley D.M., Altizer S.M. 2011. Disease ecology meets ecological immunology: understanding the links between organismal immunity and infection dynamics in natural populations. Funct. Ecol., 25(1): 48-60. https://doi.org/10.1111/j.1365-2435.2010.01753.x
- Heath A.C.H., Whitaker A.H. 2015. Mites (Acari: Pterygosomatidae, Macronyssidae) taken from lizards intercepted at the New Zealand border. Syst. Appl. Acarol., 20(7): 739-756. https://doi.org/10.11158/saa.20.7.3
- Holmstad P.R., Jensen K.H., Skorping A. 2008. Ectoparasite intensities are correlated with endoparasite infection loads in willow ptarmigan. Oikos, 117(4): 515-520. https://doi.org/10.1111/j.0030-1299.2008.16219.x
- Huyghe K., Van Oystaeyen A., Pasmans F., Tadić Z., Vanhooydonck B., Van Damme R. 2010. Seasonal changes in parasite load and a cellular immune response in a colour polymorphic lizard. Oecologia, 163(4): 867-874. https://doi.org/10.1007/s00442-010-1646-9
- Jack K.M. 1961. New species of Pterygosoma (Acari, Pterygosomidae). Parasitology, 138(4): 579-603. https://doi.org/10.1111/j.1469-7998.1962.tb05333.x
- Jack K.M. 1962. Observations on the genus Pterygosoma (Acari: Pterygosomidae). Parasitology, 52(3-4): 261-295. https://doi.org/10.1017/S0031182000027177
- Jack K., Girot B. 1965. Notes on the development of Hirstiella insignis (Berlese) 1892. Acarologia, 7(2): 286-293.
- Lehmann T. 1993. Ectoparasites: direct impact on host fitness. Parasitol. Today, 9(1): 13-17. https://doi.org/10.1016/0169-4758(93)90154-8
- Main A.R., Bull C.M. 2000. The impact of tick parasites on the behaviour of the lizard Tiliqua rugosa. Oecologia, 122 (4): 574-581. https://doi.org/10.1007/s004420050981
- Masan P., Václav R., Prokop P. 2009. First record of the lizard-parasitizing mite, Ophionyssus saurarum (Acari\,: Macronyssidae) in Slovakia. Entomofauna Carpathica, 21(10).
- Medlock J.M., Hansford K.M., Bormane A., Derdakova M., Estrada-Peña A., George J., Golovljova I., Jaenson T.G.T., Jensen J.-K., Jensen P.M., Kazimirova M., Oteo J.A., Papa A., Pfister K., Plantard O., Randolph S.E., Rizzoli A., Santos-Silva M.M., Sprong H., Vial L., Hendrickx G., Zeller H., Van Bortel W. 2013. Driving forces for changes in geographical distribution of Ixodes ricinus ticks in Europe. Parasit. Vectors., 6(1): 1-11. https://doi.org/10.1186/1756-3305-6-1
- Mendoza-Roldan J.A., Colella V., Lia R.P., Nguyen V.L., Barros-Battesti D.M., Iatta R., Dantas-Torres F., Otranto D. 2019. Borrelia burgdorferi (sensu lato) in ectoparasites and reptiles in southern Italy. Parasit. Vectors., 12(1): 1-9. https://doi.org/10.1186/s13071-019-3286-1
- Mendoza-Roldan J., Ribeiro S.R., Castilho-Onofrio V., Grazziotin F.G., Rocha B., Ferreto-Fiorillo B., Pereira J.S., Benelli G., Otranto D., Barros-Battesti D.M. 2020a. Mites and ticks of reptiles and amphibians in Brazil. Acta Trop., 208(March): 105515. https://doi.org/10.1016/j.actatropica.2020.105515
- Mendoza-Roldan J.A., Modry D., Otranto D. 2020b. Zoonotic parasites of reptiles: a crawling threat. Trends Parasitol., 36(8): 677-687. https://doi.org/10.1016/j.pt.2020.04.014
- Mendoza-Roldan J.A., Ribeiro S.R., Castilho-Onofrio V., Marcili A., Simonato B.B., Latrofa M.S., Benelli G., Otranto D., Barros-Battesti D.M. 2021. Molecular detection of vector-borne agents in ectoparasites and reptiles from Brazil. Ticks. Tick. Borne. Dis., 12(1): 101585. https://doi.org/10.1016/j.ttbdis.2020.101585
- Moraza M.L., Irwin N.R., Godinho R., Baird S.J.E., Bellocq J.G. 2009. A new species of Ophionyssus Mégnin (Acari: Mesostigmata: Macronyssidae) parasitic on Lacerta schreiberi Bedriaga (Reptilia: Lacertidae) from the Iberian Peninsula, and a world key to species. Zootaxa., 2007(1): 58-68. https://doi.org/10.11646/zootaxa.2007.1.3
- Mostafa A.R. 1974. Biological and behavioral aspects of the lizard mite Pterygosoma mutabilis Jack, 1961 (Acarina: Pterygosomidae). Acarologia., 16(4): 100-105.
- Nicholson W.L., Sonenshine D.E., Noden B.H., Brown R.N. 2019. Ticks (Ixodida). In Medical and Veterinary Entomology. Academic Press. pp. 603-672. https://doi.org/10.1016/B978-0-12-814043-7.00027-3
- Obi Z.C., Anyaegbunam L.C., Igboanugo N.A. 2013. The house gecko (Hemidactylus frenatus) and parasitaemia. Int. J. Fauna. Biol. Stud., 1(2): 13-15.
- Olsson M., Wapstra E., Madsen T., Silverin B. 2000. Testosterone, ticks and travels: a test ofthe immunocompetence-handicap hypothesis in free-ranging male sand lizards. Proc. R. Soc. B. Biol. Sci., 267(1459): 2339-2343. https://doi.org/10.1098/rspb.2000.1289
- Paredes-León R., Klompen H., Pérez T.M. 2012. Systematic revision of the genera Geckobiella Hirst, 1917 and Hirstiella Berlese, 1920 (Acari: Prostigmata: Pterygosomatidae) with description of a new genus for American species parasites on geckos formerly placed in Hirstiella. Zootaxa, 40(1): 1-40. https://doi.org/10.11646/zootaxa.3510.1.1
- Poisot T., Guéveneux-Julien C., Fortin M.J., Gravel D., Legendre P. 2017. Hosts, parasites and their interactions respond to different climatic variables. Glob. Ecol. Biogeogr., 26(8): 942-951. https://doi.org/10.1111/geb.12602
- Postawa T., Nagy Z. 2016. Variation of parasitism patterns in bats during hibernation: the effect of host species, resources, health status, and hibernation period. Parasitol. Res., 115(10): 3767-3778. https://doi.org/10.1007/s00436-016-5138-7
- Rózsa L., Reiczigel J., Majoros G. 2000. Quantifying parasites in samples of hosts. J. Parasitol., 86(2): 228-232. https://doi.org/10.1645/0022-3395(2000)086%5B0228:QPISOH]2.0.CO;2
- Schultz H. 1975. Human infestation by Ophionyssus natricis snake mite. Br. J. Dermatol., 93(6): 695-697. https://doi.org/10.1111/j.1365-2133.1975.tb05120.x
- Smallridge C.J., Bull C.M. 2000. Prevalence and intensity of the blood parasite Hemolivia mariae in a field population of the skink Tiliqua rugosa. Parasitol. Res., 86(1): 655-660. https://doi.org/10.1007/PL00008547
- Sol D., Jovani R., Torres J. 2000. Geographical variation in blood parasites in feral pigeons: The role of vectors. Ecography, 23(3): 307-314. https://doi.org/10.1111/j.1600-0587.2000.tb00286.x
- Stekolnikov A.A. 2018a. African chiggers (Acariformes: Trombiculidae) in the collection of Alex Fain, with a description of a new genus and three new species. Acarologia, 58(2): 265-286. https://doi.org/10.24349/acarologia/20184240
- Stekolnikov A.A. 2018b. Taxonomy and distribution of African chiggers (Acariformes, Trombiculidae). Eur. J. Taxon., 395(1): 1-233. https://doi.org/10.5852/ejt.2018.395
- Stekolnikov A.A. 2019. A catalogue of the holotypes of chigger mites (Acariformes: Trombiculidae) at the Natural History Museum of Geneva. Zootaxa, 4620(1): 1-71. https://doi.org/10.11646/zootaxa.4620.1.1
- Stekolnikov A.A., Er-Rguibi O., Laghzaoui E.M., Aglagane A., El Mouden E.H. 2021. New locality and host records of two chigger mite species (Acariformes\,: Trombiculidae) from Morocco. Acarologia, 61(3): 538-547. https://doi.org/10.24349/acarologia/20214448
- Taufflieb R. 1958. Contribution a l′étude des Trombiculidae Marocains. Desription de cinq espèces nouvelles. Arch. Inst. Pasteur Maroc, 5(9): 616-634.
- Tomassone L., Ceballos L.A., Ragagli C., Martello E., De Sousa R., Stella M.C., Mannelli A. 2017. Importance of common wall lizards in the transmission dynamics of tick-borne pathogens in the Northern Apennine Mountains, Italy. Microb. Ecol., 74(4): 961-968. https://doi.org/10.1007/s00248-017-0994-y
- Walter D.E., Krantz G.W. 2009. Collecting, rearing, and preparing specimens, pp. 83-96. In Krantz G.W., Walter D.E. (eds.). A manual of acarology. Texas Tech University Press, Lubbock, TX.
- Walker A.R., Bouattor A., Camicas J.L., Estrada-Pena A., Horak I.G., Latiff A.A., Pegram R.G. 2014. Ticks of domestic animals in Africa: a guide to identification of species. In The University of Edinburgh.



2022-02-14
Date accepted:
2022-12-20
Date published:
2023-04-12
Edited by:
McCoy, Karen

This work is licensed under a Creative Commons Attribution 4.0 International License
2023 Er-Rguibi, Omar; Laghzaoui, El-Mustapha ; Aglagane, Abdessamad ; Kimdil, Latifa; Stekolnikov, Alexandr A.; Abbad, Abdelaziz and El Mouden, El Hassan
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



