Recovery of Dermatophagoides pteronyssinus (Acariformes: Astigmata: Pyroglyphidae) is influenced by vacuuming protocol, fabric type and the viability of dust mites
Aljohani, Amal  1
; Burke, Daniel2
; Fleming, Gerard
1
; Burke, Daniel2
; Fleming, Gerard  3
; Gormally, Michael
3
; Gormally, Michael  4
and Byrne, Miriam
4
and Byrne, Miriam  5
5
1✉ Microbiology, School of Biological and Chemical Sciences, University of Galway, Ireland.
2Applied Ecology Unit, School of Natural Sciences, University of Galway, Ireland.
3Microbiology, School of Biological and Chemical Sciences, University of Galway, Ireland.
4Applied Ecology Unit, School of Natural Sciences, University of Galway, Ireland.
5Physics, School of Natural Sciences, University of Galway, Ireland.
2023 - Volume: 63 Issue: 1 pages: 136-147
https://doi.org/10.24349/ai0y-0w09Original research
Keywords
Abstract
Introduction
House dust mites (HDMs) are microscopic arachnids belonging to the sub-class Acari (Colloff, 2009). Dermatophagoides pteronyssinus (Trouessart, 1897) is commonly found in temperate regions of Western Europe whilst D. farinae Hughes, 1961 is more commonly found in North America and hotter climes of continental Europe (Tham et al., 2016). Studies carried out in Poland and other European countries indicated that D. pteronyssinus was found to be the dominant species in Bydgoszcz, Warsaw, Poznań and vicinity, whereas D. farinae was predominant in Gdansk and Gdynia (Solarz 2001; Solarz et al., 2021; Racewicz 2001; Colloff, 2009). Whilst mite populations thrive in environments with high humidity (\textgreater75%) and at temperatures of 25 °C (Colloff, 2009), these parameters influence the abundance and occurrence of the individual mite species. It is known that the optimal outdoor/indoor air temperature is higher (25 °C-30 °C) and the optimal humidity is lower (50%-75%) for D. farinae compared to D. pteronyssinus, (Arlian, 1992). In contrast, for D. pteronyssinus, Colloff (2009) has shown that higher relative humidity (75%-80%) and reduced temperature (15 °C-20 °C) is best for their growth in laboratory studies. The average relative humidity in the homes studied was below a critical equilibrium humidity (CEH) for Pyroglyphidae house dust mites but above 45% RH, the lowest threshold for mite survival in households (Hart, 1998; Arlian et al., 1998; Korsgaard, 1998). House dust mites inhabit soft furnishings including mattresses (Bischoff and Fischer, 1990), carpets (Svendsen et al., 2006) and upholstered couches (Mollet and Robinson, 1996). They have been isolated from fabrics used in the manufacture of clothing cotton, denim, fleece (Clarke et al., 2015a). Furthermore, cotton, denim and fleece are used for the manufacture of clothing and soft toys as well as covers for furniture (sofas, pillows, mattresses covers, cushions, pet beds); all potential reservoirs of house dust mites. The presence of house dust mites, their body parts or excrement in the home can lead to the production of allergens and sensitisation resulting in conditions including asthma, atopic eczema and rhinitis (Mumcuoglu et al., 1999).
House dust mites are routinely sampled from fabrics using a variety of methods, including the heat escape method (Hill, 1998), the mobility test method (Bischoff and Fischer, 1990) and by vacuum sampling (Lewis and Breysse, 2011). Sticky film is required for both the heat escape and the mobility test methods making these methods more time-consuming and cumbersome. Vacuum sampling is the most widely used sampling procedure for HDMs and their allergens. It is relatively simple to perform and rapid to carry out (Colloff, 2009). A review of the literature shows that a wide range of vacuum sampling methodologies have been reported but with variation in sampling areas and fabric types and strength of vacuuming sources applied (Zare et al., 2021; Mumcuoglu et al. 1999; Svendsen et al., 2006). Others have considered the efficiency of vacuum samplers for the purpose of dust collection (Platts-Mills et al., 1997; Zare et al., 2021). Tovey et al. (2003) recommend vacuum sampling (for reasons of practicality) as one of the preferred sampling options for measuring Der p 1 allergen exposure, whilst Stemeseder et al. (2017) used a nominal 30 seconds vacuuming period for the recovery of HDMs allergens from mattress, carpet and couches. The effect of torsion forces on the transfer of mites from child seats to fabrics was considered by Clarke et al. (2015a). Given the correlation between HDMs densities and HDMs allergen, specifically Der p 1 (Anasis et al., 2020), the levels of efficacy of HDMs recovery has implications for HDMs allergen sampling efficiencies. Furthermore, HDMs are also carriers of microorganisms (Hubert et al., 2021) which have the potential to negatively affect human health in cases where HDMs infest fabrics and bedding within homes. Such studies did not determine the efficiency of mite recovery from fabrics by vacuuming. Furthermore, the effect of the vacuuming protocol on HDMs recovery from fabrics is not standardised.
In this paper we report the effect of vacuuming duration, nature of fabric type and vacuuming direction (single or multiple pass) on the recovery hose dust mites seeded to fabrics. The efficiency of recovery of live mites (LM) and dead mites (DM) from fabrics are also considered. The seeding of live mites to fabric surfaces with subsequent euthanization prior to vacuuming (EM), facilitates the quantification of intact, fabric-embedded but dead mites, a fraction not considered previously.
Material and methods
Culture of mites
A laboratory strain of Dermatophagoides pteronyssinus was obtained from Fera Science Limited, UK and reared under constant hygrothermal laboratory conditions of 25 °C and 75% relative humidity (RH). Mites were reared on a diet composed of a fish food and brewery yeast (Holland and Barret, Ireland) at a 3:1 (w/w) ratio (Colloff, 2009).
Experimental design
Three experimental procedures were undertaken at room temperature using fabric squares (10 × 10 cm) each consisting of smooth cotton, denim or fleece. A stereo-binocular microscope (× 40 Olympus) was used to seed fabrics (using a fine brush) with HDMs (D. pteronyssinus) and to check for presence of HDMs on fabrics before and after seeding, and on filters after vacuuming. Each fabric was individually seeded with 25 HDMs at 2 cm spacing (Figure 1) as follows:



Experiment 1: Recovery of live mites from fabrics
Live mites (LM) were seeded onto fabrics and petroleum jelly (derived from cotton tree) was applied to the perimeter of the fabrics to prevent mite escape in accordance with protocols described by others (Reddy and Bautista, 2012). The fabrics were vacuumed after 10 minutes post seeding of mites. This facilitated the recovery of live mites from the surface of fabrics and from within the fabric weave (after mites burrowed/were entrapped within the fabric weave).
Experiment 2: Recovery of dead mites from fabrics
Dead mites (DM), euthanized by freezing prior to seeding, were added to the surface of fabrics and these were then vacuumed after 10 minutes. As such, this represented the recovery of dead mites from the surface of fabrics.
Experiment 3
Fabrics were seeded with live mites as described in Experiment 1. The mites were allowed to remain on the fabric for 10 minutes after which the fabrics were sealed in a plastic bags and frozen at (-20 °C) for 38 hours. Fabrics were then removed from the freezer and allowed to thaw fully before vacuuming took place. Mites recovered thus were called euthanized mites (EM) and represent mites which have died on the surface or within the fabric weave.
Sampling protocols
Samples were collected using a SKC Flite-2 air sampling pump (flow rate in l/min = 13.68 + 0.03 S.D.) fitted with a cassette containing a Whatman Nucleopore Polycarbonate membrane filter (diameter 37 mm, pore size 0.4µm). All cassettes were washed and dried between samples and new membrane filters fitted. Two sampling protocols (Figure 1) were adopted. In Protocol 1 (P1), each fabric was vacuumed only once in a unidirectional manner for 60, 90 or 120 seconds. The path begins at (A), and it ends at (B). In Protocol 2 (P2), fabrics were vacuumed every 30 seconds in a multi-directional manner; i.e. vacuumed twice in 60 seconds (A-B-A), thrice in 90 seconds (A-B-A-B), and four passes were applied for in 120 seconds of vacuuming (A-B-A-B-A).
Data analysis
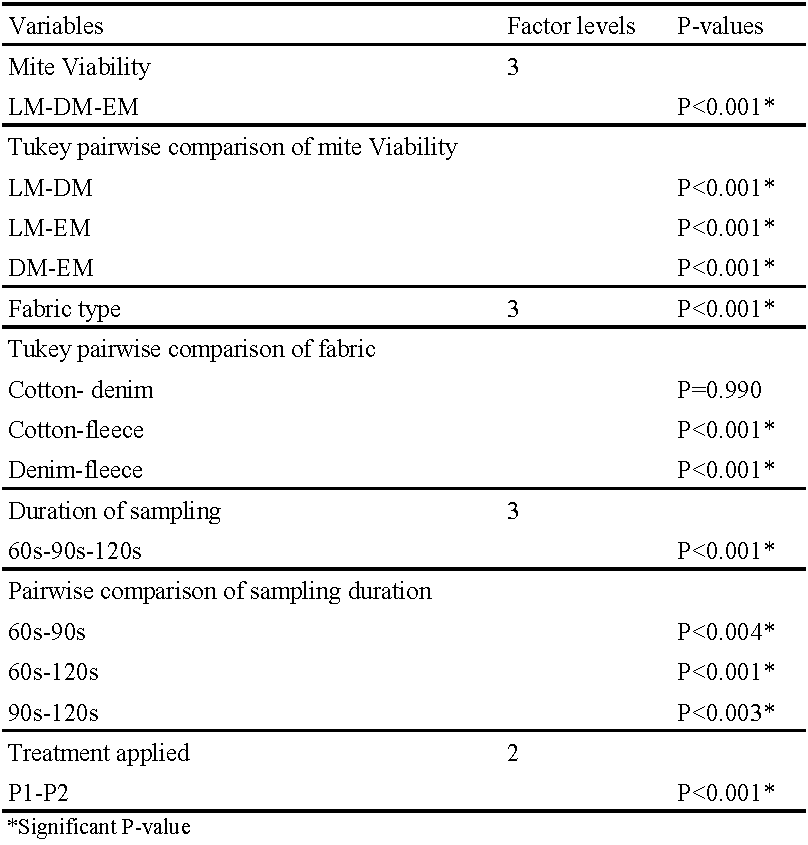
Basic descriptive statistics were calculated using Minitab 19 statistical software (2019). The General Linear Model (GLM) is an ANOVA procedure performing a least squares regression approach to describe statistical correlations. Here the GLM analysis was used to test the effect of several factors by using a coding scheme for time, protocols, fabric and viability of mites. The response variable was the number of mites recovered. The input variables were mite viability: live (LM), dead (DM) and euthanized (EM), sampling protocol used (P1 and P2), fabric type (cotton, denim and fleece) and duration of sampling (60 seconds, 90 seconds and 120 seconds). The GLM model requires normally-distributed data, therefore all data (number of mites recovered was transformed first to root squared values). A one-way ANOVA test was also carried out to compare the three different sampling times in each time category as well as comparing the same sampling times between the three different fabrics. Each one-way ANOVA test was followed by a Tukey post-hoc test to determine whether there were significant differences between the groups tested.
Scanning electron microscopy
Scanning electron microscopy images were taken of fabrics inoculated with live and dead mites. Fabric samples were taken 10 minutes after the mites were seeded to the surface of the fabrics. Iodine stained samples were imaged using an Electron Microscope (Hitachi S2600N), University of Galway.
Results
Descriptive analysis
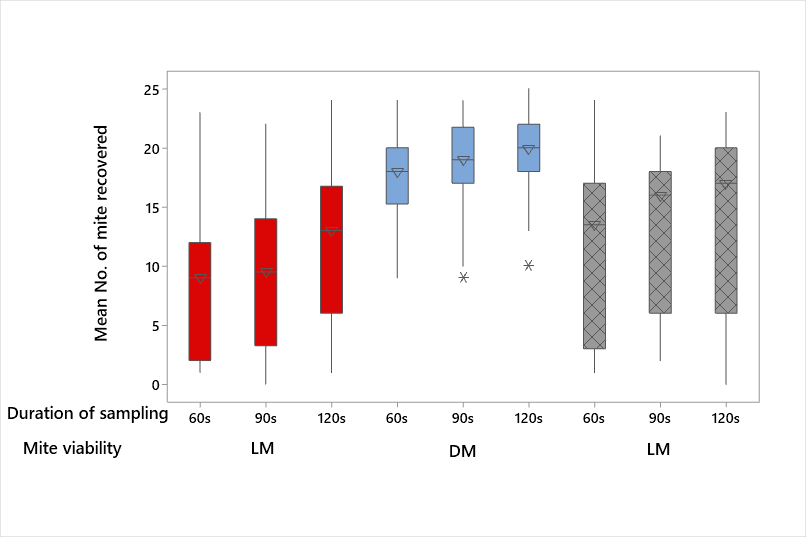
The percentage recovery of HDMs from fabrics (Table 1- Experiments 1, 2 and 3), pooled data (Table 2), and box plots are shown in plots 2, 3 and 4. The efficiency of recovery ranged from 7.2% (fleece, LM, protocol 2, 60s) to 91.2% (fleece, DM, protocol 2, 120s). The recovery (irrespective of treatment) was consistently lowest for fleece when LM and EM were deployed. In contrast, no discernible patterns were observed with DM recovery across fabrics (Table 1; Figure 2). The application of the P2 protocol resulted in slightly greater mean numbers of HDMs being recovered from all fabrics when compared with P1 (Figure 3). However, increasing the duration of sampling time on all fabrics combined, resulted in more efficient recovery rates of all groups i.e. LM, DM and EM (Figure 4).











General linear model outputs
The GLM was used to determine the statistical effect of every possible combination of the experimental treatments on the response variable i.e. number of mites recovered (Table 3). All treatments (mite viability, fabric type, protocol applied and duration of sampling) had a significant effect on the response variable. Furthermore, Tukey pairwise comparisons indicated significant differences in mite recovery between the mite categories (LM, DM and EM). While no significant difference in mite recovery was detected between cotton and denim, there were significant differences between fleece and denim/cotton. Furthermore, as presented in Table 3, there were significant effects on the mean number of mites recovered from fabrics between all sampling durations (60 seconds, 90 seconds and 120 seconds).




Scanning electron microscopy images
Figure 5 shows the weave structures of fabrics used in this study. Cotton (A) and denim (B) are both tightly woven fabrics making them structurally similar while fleece (C) is a non-woven fabric consisting of a deep pile (3- 4mm) of polyester material. Dead mites generally remained on the surfaces of textiles while live mites frequently showed evidence of burrowing into the fabric (Figure 6).






Discussion
The variability of vacuuming protocols employed in the recovery of mites from household fabrics (Svendsen et al., 2006; Byrne, 2000; Lewis and Breysse, 2011) can present challenges to the interpretation of mite density results between studies. Furthermore, the efficiency of recovery of mites which entered the fabric weave and subsequently died within the fabric (without active attachment by tarsi) has not been reported in the published scientific literature. Data presented here shows that vacuuming protocol and the type of fabric under test influenced the efficiency of mite recovery. We have evaluated vacuuming protocols for the recovery of live (LM), dead (DM) and mites euthanized post-seeding (EM). These represent mites that actively cling to the fabric weave (LM), thus resisting removal by vacuuming whilst dead mites (DM) are mainly surface based. Finally, we have determined the efficiency of recovery of live mites that are on the fabric surface or embedded in the weave but have died in-situ. These embedded mites (EM) do not actively resist the vacuum force and the efficiency of their recovery has not been reported previously.
Of the three fabrics used in this investigation (Figure 5), data presented in Table 1 (and summarised in Table 2), demonstrate that the recovery of DM (from the surface of fabrics) was significantly greater for denim (76%), cotton (75%) and fleece (72%) than the same fabrics seeded with live mites (11.6-58%). Dead mites remain on the surface of the fabric and are more amenable to recovery by vacuuming. On the other hand, the smallest number of LM (11.6%) and EM (15.2%) were recovered from fleece and this recovery was significantly different to the recovery from denim and cotton (p=0.000: Figure 2 and Table 2). It is probable that mites burrowed and were entrapped in the fleece weave (which acted akin to a depth filter), thereby reducing recovery rates of HDMs (Figure 6). There was no significant difference (P=0.990) in the recovery of HDMs between denim and cotton (Table 3). Denim and cotton have a structured and low pile-depth weave which acts as a barrier for the penetrability of mites (Figure 5). It is likely that LM adhere to fabrics by clawing which reduces their recovery by vacuuming (Colloff, 2009). It is interesting to note that Clarke et al. (2015a) observed greater transfer efficiencies between fabrics using dead rather than live mites. This was also observed for inert particles deposited on fabrics in surface to surface contact experiments (McDonagh et al., 2012).
Hay (1995) has indicated the efficiency of recovery of mites from natural habitats was in the region of 0.5 – 1% of recovery rates. However, the vacuuming protocols employed in the present study yielded a greater recovery (41%-80%) of live, dead or euthanized mites. It is evident that the protocols described here enhance the recovery of mites from natural fabrics.
Generally, a greater recovery was achieved using Protocol 2 (Figure 3) and vacuuming for 120s (Figure 4) under all conditions employed in this study (Table 2). There was a direct relationship between the time of vacuuming and the number of the mites recovered irrespective of the of the type of fabric tested where 120 seconds < 90 seconds < 60 seconds (Table 2). These results would support the use of 120 seconds of vacuuming as a standardised procedure for the recovery of HDMs from fabrics.
A novel aspect of this study was that comparisons were made between a single pass (Protocol 1) of the vacuum sampler and repeated sampling (Protocol 2) for mite-incurred fabrics. Regardless of fabric or sampling duration, Protocol 2 yielded a significantly greater recovery of mites than Protocol 1 (p\textless0.001) in 50 of the 54 tests carried out (Table 3). Thus, the use Protocol 2 in tandem with 120 seconds of vacuuming is the protocol of choice for the recovery of HDMs from the fabrics under investigation. The methodologies used in in this study may be useful in providing some insight to the standardisation of sampling/collection of HDMs by the vacuuming protocol.
Mite-associated allergies are influenced by the air quality of the indoor environment. Allergic determinants contained on clothing (Tovey et al., 1995; Lucca et al., 2000) and within carpets (Becher et al., 2018; Salo et al., 2018) can manifest an allergic response in humans and pets. Der p 1 is associated with the faecal pellets of Dermatophagoides pteronyssinus and is a significant mite allergen leading to skin and lung-associated allergic responses. Clarke and co-authors (2015b) have shown a correlation between HDMs densities and HDM-derived Der p 1 allergen concentrations. The recovery of live, dead and euthanized HDMs (which represent mites that have died within the fabric weave) vary significantly with the sampling protocol. Furthermore, as shown in the present study, weave-bound mites resist vacuuming and recovery. The levels of allergens may thus be underestimated if inefficient vacuuming protocols are employed or if mites are embedded in the fabric weave. Future studies might consider a wider range of fabrics with varying weave densities and varying the vacuuming force applied to fabrics.
Acknowledgements
This study has been funded by a Ph.D. studentship awarded by the Kingdom of Saudi Arabia. Special thanks to Dr Emma McDermott in the electron microscopy unit in the University of Galway, who generated the scanning electron microscopy images for this study.
References
- Anasis A. M., Rozaliyani A., Wibowo H. 2020. Density of Dermatophagoides spp. and its relationship with House-dust Mite specific serum IgE in persistent Asthma. Mol. Cell Biomed. Sci., 4(2): 61. https://doi.org/10.21705/mcbs.v4i2.92
- Arlian L. G. 1992. Water balance and humidity requirements of house dust mites. Exp. Appl. Acarol., 16: 15-35. https://doi.org/10.1007/BF01201490
- Arlian L. G., Confer P.D., Rapp C.M., Vyszenski-Moher D. L., Chang J. C. 1998. Population dynamics of the house dust mites Dermatophagoides farinae, D. pteronyssinus, and Euroglyphus maynei (Acari: Pyroglyphidae) at specific relative humidities. J. Med. Entomol., 35(1): 46-53. https://doi.org/10.1093/jmedent/35.1.46
- Becher R., Øvrevik J., Schwarze P. E., Nilsen S., Hongslo J. K., Bakke J. V. 2018. Do carpets impair indoor air quality and cause adverse health outcomes: A review. Int. J. Environ. Res. Public. Health, 15(2): 184. https://doi.org/10.3390/ijerph15020184
- Bischoff E., Fischer A. 1990. New methods for the assessment of mite numbers and results obtained for several textile objects. Aerobiologia, 6(1): 23-27. https://doi.org/10.1007/BF02539037
- Byrne M. A. 2000. Suction methods for assessing contamination on surfaces. The Ann. Occup. Hyg. 44(7): 523-28. https://doi.org/10.1016/S0003-4878(00)00038-7
- Clarke D., Burke D., Gormally M., Byrne M. 2015a. Dynamics of house dust mite transfer in modern clothing fabrics. Ann. Allergy Asthma Immunol., 114(4): 335-340. https://doi.org/10.1016/j.anai.2014.12.021
- Clarke D., Gormally M., Sheahan J., Byrne M. 2015b. Child car seats - a habitat for house dust mites and reservoir for harmful allergens. Ann. Agric. Environ. Med., 22(1): 17-22. https://doi.org/10.5604/12321966.1141362
- Colloff M. J. 2009. Dust mites. CSIRO Publishing. Australia. pp.600. https://doi.org/10.1007/978-90-481-2224-0
- Hart B. J. 1998. Life cycle and reproduction of house-dust mites: Environmental factors influencing mite populations. Allergy: Eur. J. Allergy Clin. Immuonol., 53(48): 13-17. https://doi.org/10.1111/j.1398-9995.1998.tb04990.x
- Hay D.B. 1995. An in Situ coring technique for estimating the population size of house dust mites in their natural habitat. Acarologia, 36(4): 341-345. https://www1.montpellier.inra.fr/CBGP/acarologia/article.php?id=2257.
- Hill M. R. 1998. Quantification of house-dust-mite populations. Allergy, 53(48): 18-23. https://doi.org/10.1111/j.1398-9995.1998.tb04991.x
- Hubert J., Marta N., Stefan J. G., Pavel B. K. 2021. Microbial communities of stored product mites: variation by species and population. Microb. Ecol., 81(2): 506-522. https://doi.org/10.1007/s00248-020-01581-y
- Korsgaard J. 1998. House-Dust Mites and Asthma. A Review on house-dust mites as a domestic risk factor for mite asthma. Allergy, 53(48 Suppl): 77-83. https://doi.org/10.1111/j.1398-9995.1998.tb05003.x
- Lewis R.D., Breysse P.N. 2011. A Comparison of the sampling characteristics of two vacuum surface samplers for the collection of dust mite allergen. Appl. Occu. Environ. Hyg., 13(7): 536-541. https://doi.org/10.1080/1047322X.1998.10390102
- Lucca S. D., O'Meara T. J., Tovey E. R. 2000. Exposure to mite and cat allergens on a range of clothing items at home and the transfer of cat allergen in the workplace. J. Allergy Clin. Immunol., 106(5): 874-879. https://doi.org/10.1067/mai.2000.110804
- McDonagh A., Sextro R., Byrne M. 2012. Mass Transport of Deposited Particles by Surface-to-Surface Contact. J. Hazard. Mater., 227-228: 370-377. https://doi.org/10.1016/j.jhazmat.2012.05.070
- Mollet J. A., Robinson W. H. 1996. Dispersal of American house dust mites (Acari: Pyroglyphidae) in a residence. J. Med. Entomol., 33(5): 844-847. https://doi.org/10.1093/jmedent/33.5.844
- Mumcuoglu K. Y., Gat Z., Horowitz T., Miller J., Bar-Tana R., Ben-Zvi A., Naparstek Y. 1999. Abundance of house-dust mites in relation to climate in contrasting agricultural settlements in Israel. Med. Vet. Entomol., 13(3): 252-258. https://doi.org/10.1046/j.1365-2915.1999.00149.x
- Platts-Mills T. A. E., Vervloet D., Thomas W. R., Aalberse R. C., Chapman M. D. 1997. Indoor allergens and asthma: report of the third international workshop. J. Allergy Clin. Immunol., 100(6): S2-24. https://doi.org/10.1016/S0091-6749(97)70292-6
- Racewicz M. 2001. House dust mites (Acari: Pyroglyphidae) in the cities of Gdańsk and Gdynia (northern Poland). Ann. Agric. Environ. Med., 8(1): 33-38.
- Reddy G. V. P., Bautista J. R. 2012. Integration of the predatory mite Neoseiulus californicus with petroleum spray oil treatments for control of Tetranychus marianae on eggplant. Biocontrol Sci. and Technol., 22(10): 1211-1220. https://doi.org/10.1080/09583157.2012.719152
- Salo P. M., Cohn R. D., Zeldin D. C. 2018. Bedroom allergen exposure beyond house dust mites. Curr. Allergy Asthma Rep., 18(10): 52. https://doi.org/10.1007/s11882-018-0805-7
- Solarz K. 2001. Risk of exposure to house dust Pyroglyphidae mites in Poland. Ann. Agric. Environ. Med., 8(1): 11-24.
- Solarz K., Obuchowicz A., Asman M., Nowak W., Witecka J., Pietrzak J., Marek M., Łonak A., Stadnicka I., Hajduga-Staśko B. 2021. Abundance of domestic mites in dwellings of children and adolescents with Asthma in relation to environmental factors and allergy symptoms. Scientific Rep., 11(1). https://doi.org/10.1038/s41598-021-97936-7
- Stemeseder T., Schweidler B., Doppler P., Klinglmayr E., Moser S., Lueftenegger L., Himly M., Lang R., Zumbach J., Oostingh G., Hawranek T., Bathke A., Gadermaier G. 2017. Exposure to indoor allergens in different residential settings and its influence on IgE sensitization in a geographically confined Austrian Cohort. PLOS ONE, 12(1). https://doi.org/10.1371/journal.pone.0168686
- Svendsen E., Thorne P., O'Shaughnessy P., Zimmerman D., Reynolds S. 2006. House dust collection efficiency of the high volume small surface sampler on worn carpets. J. Occup. Environ. Hyg. 3(6): 334-341. https://doi.org/10.1080/15459620600700651
- Tham E. H., Lee A. J., van Bever H. 2016. Aeroallergen sensitization and allergic disease phenotypes in Asia. Asian Pac. J. Allergy Immunol., 34: 181-189 https://doi.org/10.12932/AP0770
- Tovey E. R., Mahmic A., McDonald L. G. 1995. Clothing-an important source of mite allergen exposure. J. Allergy Clin. Immunol., 96(6): 999-1001. https://doi.org/10.1016/S0091-6749(95)70239-3
- Tovey E. R., Mitakakis T.Z., Sercombe J.K., Vanlaar C.H., Marks G.B. 2003. Four methods of sampling for dust mite allergen: differences in dust. Allergy. 58(8): 790-794. https://doi.org/10.1034/j.1398-9995.2003.00228.x
- Zare M., Hassani-Azad M., Soleimani-Ahmadi M., Majnoon R. 2021. The influence of indoor environmental factors on the prevalence of house dust mites as aeroallergens in Bandar Abbas student dormitories, South of Iran. J. Med. Entomol., 58(4): 1865-1873. https://doi.org/10.1093/jme/tjab032



2022-06-29
Date accepted:
2022-12-19
Date published:
2023-01-26
Edited by:
Akashi Hernandes, Fabio

This work is licensed under a Creative Commons Attribution 4.0 International License
2023 Aljohani, Amal; Burke, Daniel; Fleming, Gerard; Gormally, Michael and Byrne, Miriam
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



