Linked seasonality between a phoretic mite and its moth fly host (Parasitiformes: Mesostigmata and Diptera: Psychodidae)
Kvifte, Gunnar Mikalsen  1
; Kaczmarek, Sławomir
1
; Kaczmarek, Sławomir  2
; Marquardt, Tomasz3
and Seniczak, Anna4
2
; Marquardt, Tomasz3
and Seniczak, Anna4
1✉ Faculty of Biosciences and Aquaculture, Nord University, P.O. Box 2501, 7729 Steinkjer, Norway & Department of Natural History, University Museum of Bergen, University of Bergen, Allégt. 41, 5020 Bergen, Norway.
2Department of Evolutionary Biology, Faculty of Biological Sciences, Kazimierz Wielki University, Ossolińskich Av. 12, 85-093 Bydgoszcz, Poland.
3Department of Evolutionary Biology, Faculty of Biological Sciences, Kazimierz Wielki University, Ossolińskich Av. 12, 85-093 Bydgoszcz, Poland.
4Department of Natural History, University Museum of Bergen, University of Bergen, Allégt. 41, 5020 Bergen, Norway.
2022 - Volume: 62 Issue: 4 pages: 956-964
https://doi.org/10.24349/7gdm-suwvOriginal research
Keywords
Abstract
Introduction
The term ''phoresy'' was introduced by the French entomologist Pierre Lesne in 1896, referring to phenomena when a smaller animal (the phoretic) is transported by a larger one (the host or carrier) (Walter and Proctor, 2013). Many other definitions have been proposed since then, but none of them has yet been commonly accepted (Walter and Proctor, 2013). For example, Farish and Axtell (1971) restricted phoresy to the situations when the phoretic actively seeks the host, attaches to its outer surface for a limited period of time, and ceases feeding and ontogenesis during the attachment. Athias-Binche (1991) added to this definition that the phoretic must be quiescent when on the host. Houck and O'Connor (1991) disagreed with the opinion that the phoretic must be active when searching for the host, as some mites are not, and stated that phoresy takes place when the phoretic receives an ecological and evolutionary advantage when migrating to a new habitat, but cannot feed or develop when on the host. In turn, Walter and Proctor (2013) came back to a broader meaning of phoresy, defining it as a type of temporary symbiosis that allows a smaller individual to travel on a larger individual. In such understanding the phoretic can also feed or develop during the transportation. Phoresy might be facultative, depending on the population density or environmental perturbations, or obligatory, when it takes place in a relatively stable habitat, and the mites follow a seasonal migration cycle using traditional carriers (Krantz, 2009).
Phoresy can be found in all groups of Acariformes mites, but among Parasitiformes it is known only in Mesostigmata. Here, it is a widespread phenomenon, with over 1700 species associated with arthropods, mainly (95%) with insects (Hunter and Rosario, 1988). As a rule, mesostigmatid mites are associated with insects that live in nutritionally rich but ephemeral habitats, like mammal dung, decaying vegetation, carrion, or temporary ponds (Hunter and Rosario, 1988). The benefit of this relationship to the mites is obvious; they are small, wingless, and with sensory organs of a limited range, so they take advantage of travelling fast on larger animals that can detect a proper habitat from a greater distance (Walter and Proctor, 2013). For example, moth flies (Psychodidae) can easily detect fresh dung on which their larvae feed, and mesostigmatid mites travelling with them will achieve the same goal, finding there rich food sources: immature stages of insects or other mites and nematodes (Floate, 2011). For insects the presence of phoretic mites is usually neutral, but in few cases a mutualistic interaction between Mesostigmata and insects has been proved; the phoretic had a positive effect on host fitness by reducing the level of parasites (Walter and Proctor, 2013).
The relations between the mites and the insects are fascinating for many reasons, and draw attention of quite many acarologists, in particular to answer evolutionary and ecological questions (Walter and Proctor, 2013). Mites are considered one of the best, if not the best, group to study phoretic species associations (Klompen, 2009). Sometimes several different species (even 12) can be found on one insect (McGarry and Baker, 1997), but some mite species have a close association with a single host. In that case the phoretic has a life cycle synchronized with the life cycle of the host (Hunter and Rosario, 1988). Particularly in temporary habitats such as dungpats and carrion this is an advantage, because the mites dependent on such habitat must be moved from one place to another regularly and predictably in their life cycle. Usually, only one life-history stage is phoretic in a given species (Walter and Proctor, 2013), and in most cases phoresy is restricted to the deutonymphs or adult females (Krantz, 2009).
Among the Diptera, 19 families have been found to transport mites, and the most common is the Muscidae, followed by Sphaeroceridae and Psychodidae (Hunter and Rosario, 1988). Regarding the latest family, there is still a lack of information about the phoretic interactions with mites on the species level, so our aim was to fill this gap. We here present the first population-level data on Psychodidae and Mesostigmata interactions based on standardised collection throughout the field season, and use them to test the hypotheses that 1) interactions between Mesostigmata and Psychodidae are host specific, in which particular phoretic species are confined to particular hosts, and 2) the life cycles of the phoretics are synchronized with the life cycle of the host.
Material and methods
Study area
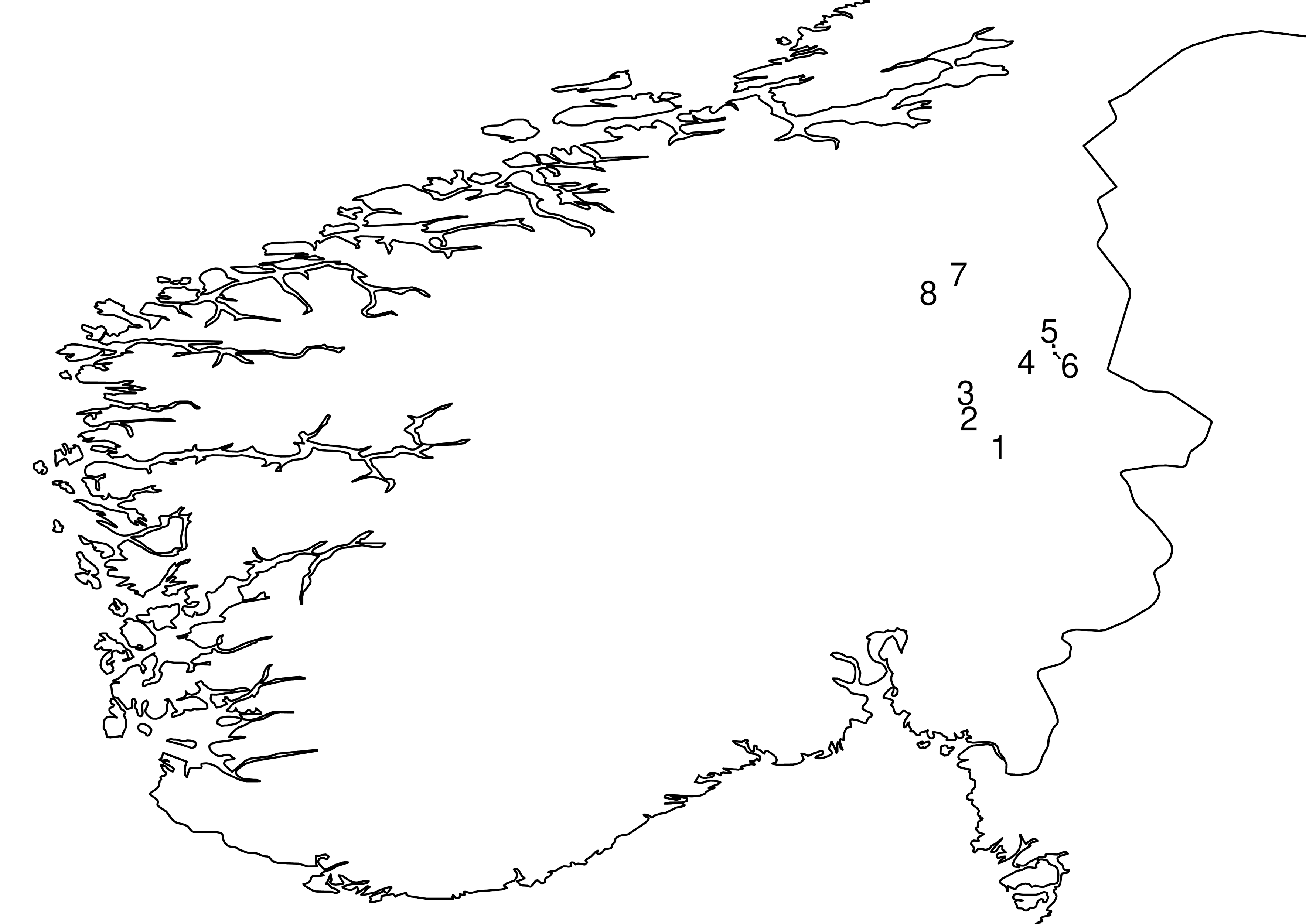
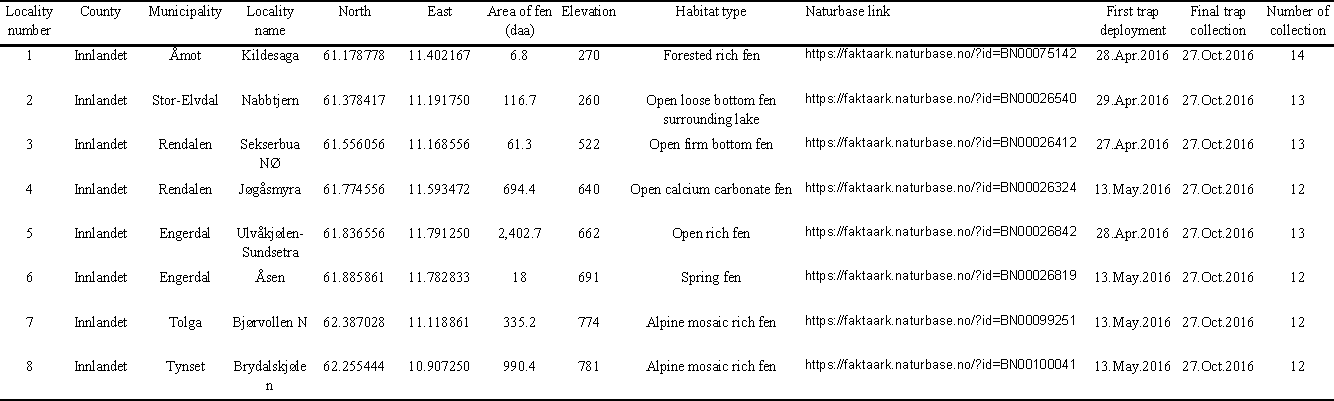
All material examined in this study was collected using Malaise traps on eight minerotrophic rich fens in Hedmark county, east Norway (Figure 1, Table 1). The study aimed at inventorying aquatic and semiaquatic insects of many different groups (analogous to Ekrem et al., 2012), but non-aquatic representatives of the families in question were also collected. Detailed descriptions of localities (in Norwegian) can be read at www.naturbase.no, see Table 1 for links.




Sampling and identification
Moth flies were identified using the reference collection in the University Museum of Bergen (most specimens are listed in Andersen and Håland, 1995, Kvifte et al., 2011, Kvifte and Andersen, 2012, Kvifte and Boumans, 2014 and Kvifte, 2019), as well as illustrations and descriptions in Withers (1989), Tonnoir (1922, 1940) and Ježek (1983, 1990). Some females were identified by associations with males and/or DNA barcoding following methods described in Ekrem et al. (2012). In the present paper we only present results from the species found to carry phoretic mites.
Mesostigmata mites were preserved in 70% ethyl alcohol, and mounted on permanent slides in Hoyer's medium. They were identified to species level, using universally applied keys (Ghilyarov and Bregetova, 1977; Hyatt, 1980; Karg, 1993; Kalúz and Fenda, 2005; Gwiazdowicz, 2007). The information on the Mesostigmata of Norway is based on the checklist (Gwiazdowicz and Gulvik, 2005a) and later references (Gwiazdowicz and Gulvik, 2007; Gwiazdowicz et al., 2013; Bolger et al., 2018; Seniczak et al., 2019, 2020, 2021a,b). Specimens in ethanol and on microscope slides are deposited in the entomological collections, University Museum of Bergen (ZMUB).
Statistical analysis
Statistical analyses were conducted in R version 3.4.3 (R Core Team, 2017). We used the base R functions cor.test to test for correlations in associations between flies and mites and glm to test for statistical significance of explanatory variables. All analyses were done individually for each locality and for all localities pooled together. For testing seasonality of associations, we treated fly and mite specimen counts as the poisson distributed response variable with site ID and the polynomial of sampling event number (collection event 1, collection event 2, etc.) as explanatory variable in a generalised linear model. All models were found to be overdispersed and we therefore assessed significance using an F-test in a quasi-poisson approach. To test seasonality of the association between mites and flies we treated a table of infested vs uninfested flies as a binomially distributed response variable with time and site ID as predictors.
Results
A total of 2593 specimens of Psychodidae belonging to an estimated 29 species were identified, of which 1198 belonged to the genus Psychoda; three species in this genus were found to carry mites, namely Psychoda phalaenoides (L., 1758), Psychoda grisescens Tonnoir, 1922 and Psychoda satchelli Quate, 1955. Together, these species made up over 50% of all Psychoda individuals collected in the samples, with counts of 354, 37 and 227 identified specimens, respectively. These species are all widespread and common in the Norwegian Psychodidae fauna (Kvifte et al., 2011).
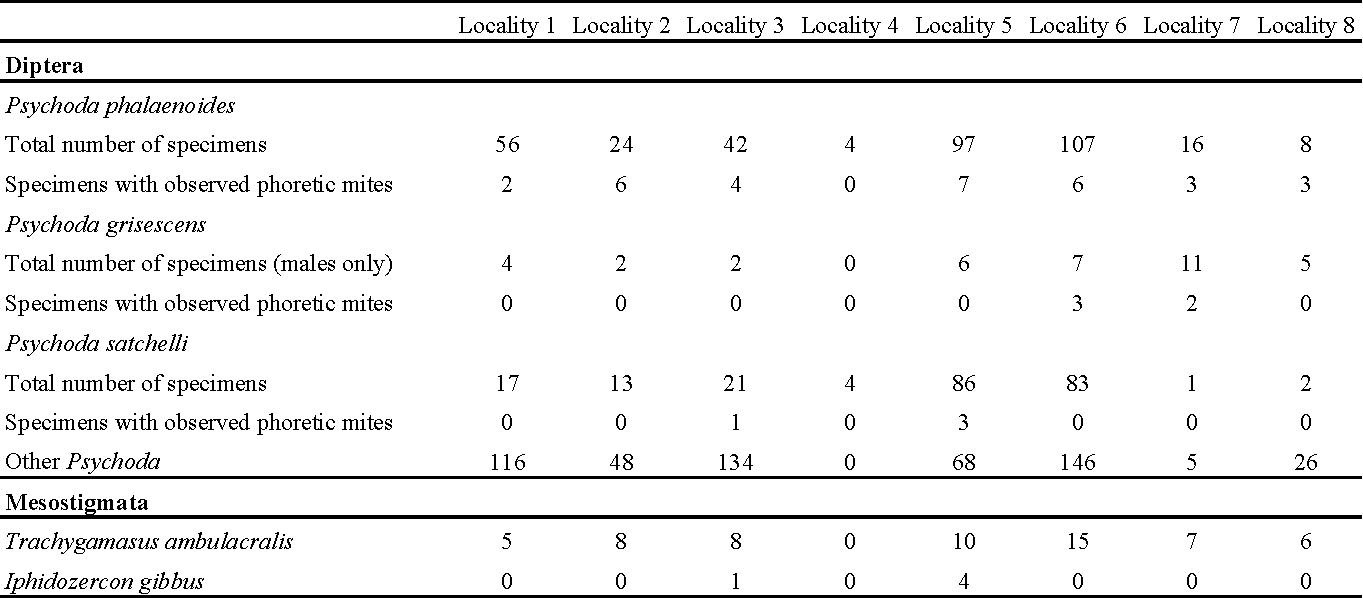


Two species of mites were identified: deutonymphs of Trachygamasus ambulacralis (Willmann, 1949) that made up over 90% of mites found in this study, and females of Iphidozercon gibbus (Berlese, 1903). Trachygamasus ambulacralis has not previously been recorded from Norway; I. gibbus was recorded by Gwiazdowicz and Gulvik (2005b) based on a male and three females from Sogn og Fjordane county. All identified mites were found to be phoretic, and those that were still hanging on to their hosts were attached by means of the chelicerae to the first three abdominal segments of the fly. For numbers of infested and uninfested flies throughout the seasons per locality, see Table 2.





The most frequent host of phoretic deutonymphs of Trachygamasus ambulacralis were Psychoda phalaenoides females (n=29). This mite species was also occasionally found on males of P. phalaenoides (n=2) and both sexes of P. grisescens (total number unknown due to ambiguous identifications of females). Females of Iphidozercon gibbus were phoretic on male P. satchelli (n=4) and female P. phalaenoides (n=1).



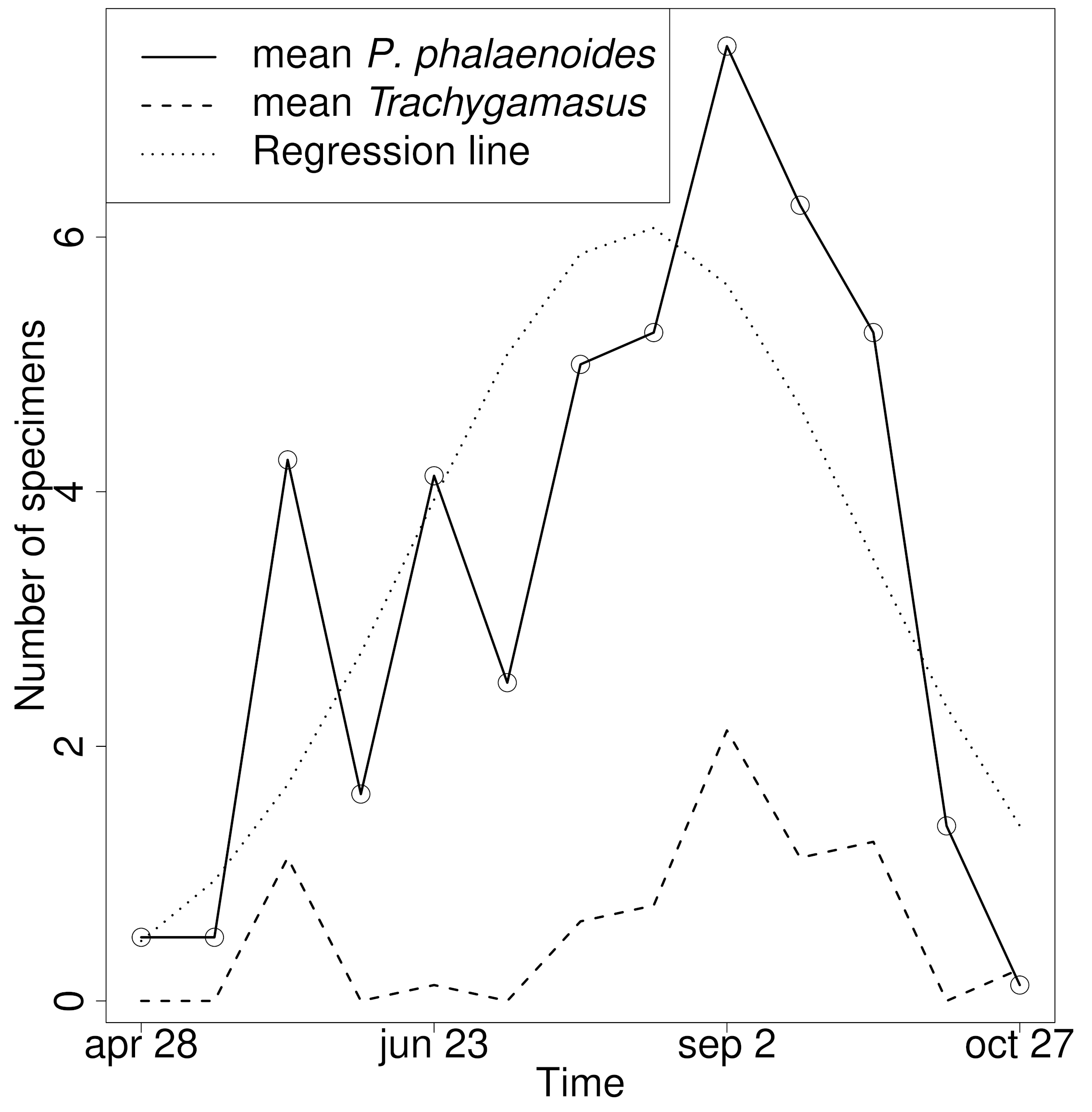
Due to low specimen numbers, I. gibbus, P. grisescens and P. satchelli could not be analysed quantitatively. Peaks of abundance in T. ambulacralis generally coincided with those of P. phalaenoides, although P. phalaenoides also displayed some peaks unassociated with T. ambulacralis (Figures 2, 3). The two abundances were strongly correlated when all sites and sampling intervals were pooled together (r2 = 0.445, P < 0.00001, Figure 2); significant correlations were also found on fens Bjørvollen (r2 = 0.706, P = 0.007), Brydalskjølen (r2 = 0.566, P = 0.043), Nabbtjern (r2=0.588, P = 0.034) and Sekserbua (r2 = 0.824, P < 0.001).
Seasonal variance in abundance was found to be significant for both P. phalaenoides (F-test of quasi-poisson model with site ID and 2-polynomial time since start of sampling, P = 5.30 x 10-9) and T. ambulacralis (Chi-test of poisson model with 2-polynomial time since start of sampling, P = 6.90 x 10-6). There were also significant differences between sites both for abundance of P. phalaenoides (P = 2.45 x 10-12) and T. ambulacralis (P = 0.0017). No evidence was found for the association between the two species varying throughout the season, but there were significant differences in infestation rates between sites (p=0.017). Also, no significant relationships were found between latitude and abundance for any of the species. For further details of the GLMs, see Table 3.
Discussion
These are to our knowledge the first phoretic associations of identified Psychodidae with identified Mesostigmata from Europe, however, several previous associations have been documented from other regions or with lower taxonomic resolution.
Whitsel and Schoeppner (1973) identified two species of Iphidozercon (I. californicus Chan, 1963 and Iphidozercon sp.) as phoretic on Psychoda satchelli in California; in our study we identified I. gibbus from the same nominal species. Withers (1988, 1989) considered P. satchelli as a synonym of P. albipennis Zetterstedt, 1850 and listed sewage beds, rotten vegetation, bird and bat guano, carnivore and ruminant dung and polyporous fungi as recorded breeding habitats for their saprophagous larvae. Iphidozercon gibbus also occurs in many different decaying habitats including bird and rodent nests (Kalúz and Fenda, 2005; Gwiazdowicz, 2007) from or to which it probably migrates, at least with P. satchelli.
Lundqvist (1998) identified deutonymphs of Trachygamasus gracilis Karg, 1965 as phoretic on unidentified Psychoda sp. in Southern Sweden, and further identified T. ambulacralis deutonymphs from fly trap residues. We deem it likely that his Trachygamasus records predominantly are from P. phalaenoides and other dung-breeding psychodids, as these are very common in Southern Sweden (Svensson, 2009) and furthermore overlap with Trachygamasus in habitat.
Interestingly, the psychodid species that T. ambulacralis attach to are also the ones most commonly found within inflorescences of the plant Arum maculatum L., 1753 (Espíndola et al., 2011). This plant attracts P. phalaenoides females and P. grisescens of both sexes using chemical mimicry of these insects' larval habitats, and relies on these flies for pollination. The sex ratios of these pollinators seem to follow the same pattern as the hosts of T. ambulacralis– predominantly P. phalaenoides females and P. grisescens of both sexes. The difference in sex ratios between these two species could possibly reflect different mating systems in the two species: P. phalaenoides males have been inferred to locate females with species-specific pheromones (Yeargan and Quate, 1996), meaning mating can take place anywhere and the only individuals who will actively seek out larval development substrates will be females. No behavioral data is available for P. grisescens, but their males may well locate females directly on the larval habitats.
In Mesostigmata some of species use the deutonymphs (e.g. in Sejida, Uropodina and Parasitiae) for the phoretic dispersal while in others (Trigynaspida and derived Dermanyssiae) females are typical phoretics (Walter and Proctor, 2013). The use of the last nymphs could be explained with their greater resistance to harsh environmental circumstances and ability to delay the last moult until appropriate conditions are available (Rapp, 1959; Walter and Proctor, 2013). It is likely that it also prevents inbreeding during transport which would be possible with both males and females present on the same host individual. That could be true for T. ambulacralis. In case of I. gibbus, in which males are very rare when compared to females (e.g. Gwiazdowicz, 2005, 2007), phoresy of females could be more effective in terms of reproduction when transported to the new habitat, however, biology of both mite species mentioned in this work is virtually unknown.
The phoretic associations between Mesostigmata and Psychodidae are in our opinion likely to be phylogenetically recent ecological associations rather than co-cladogenetic macroevolutionary relationships. The Psychoda species encountered as phoretic hosts in this study are phylogenetically distant from each other (Cordeiro, 2013; Kvifte, unpubl. data; see also genetic distances in Kvifte and Andersen, 2012) and both T. ambulacralis and I. gibbus have been previously found in phoretic association with other coprophagous Diptera such as the ceratopogonid Culicoides obsoletus (Meigen, 1818) (Mašán and Országh, 1994). Interestingly many dung-associated Psychoda have not been observed with mites; and psychodids breeding in other habitats than dung have not to our knowledge been associated with Mesostigmata. Nevertheless, the T. ambulacralis / P. phalaenoides relationship shown herein may be indicative of the latter species being the ''primary'' phoresy host of T. ambulacralis, and it is worthy to emphasize that I. gibbus females mainly infested other hosts (P. satchelli) than those used by T. ambulacralis.
For both mite species found associated with Psychodidae in this study, the association is likely motivated by their need to colonise the patchy and ephemeral habitats that the host fly actively seeks out. Association studies such as the present one are, however, only the first step in understanding the fascinating evolutionary phenomenon of phoresy, and future studies should address the short and long term benefits and disadvantages for both parts of the association, its dynamics and the behavioral triggers for mounting and dismounting.
Acknowledgements
The present contribution was funded by two grants to the University Museum of Bergen from the Norwegian Taxonomy Initiative, as part of the projects ''Insects on rich fens in Hedmark, east Norway'' (Grant No. 50-15), and ''Mites of Atlantic Raised Bogs'' (Grant No. 6-20, 70184243). We are grateful to Trond Andersen and Linn K. Hagenlund (University Museum of Bergen, Norway) for tirelessly sorting out psychodids from the many Malaise trap samples originating from the Hedmark project, and to Frode Næstad, Inland Norway University of Applied Sciences, for managing the traps during most of the 2016 field season. Finally we are indepted to Dr. Lise Roy and two anonymous reviewers for their insightful comments on an earlier version of this manuscript.
Author contributions
GMK planned fieldwork with colleagues at the University Museum of Bergen. GMK conceived of the study, identified psychodids, performed statistical analyses and prepared figures. AS coordinated identification of mites and conducted background research in phoresy. SK and TM identified mites and performed observations on their phoretic modes. GMK and AS wrote the paper, all authors approved the final version.
References
- Andersen T., Håland, Ø. 1995. Norwegian moth flies (Diptera: Psychodidae). Fauna Norvegica Series B, 42: 125-130.
- Athias-Binche F. 1991. Evolutionary ecology of dispersal in mites. In: Dusbabek F., Bukva V. (Eds). Modern acarology 1. Prague: SPB Academic. p. 27-41.
- Bolger T., Devlin M., Seniczak A. 2018. First records of ten species of Mesostigmata (Acari, Mesostigmata) added to the published Norwegian species list. Norwegian Journal of Entomology, 65: 94-100.
- Cordeiro D.P. 2013. Filogenia de Psychoda sensu lato (Diptera, Psychodidae, Psychodinae) e o uso de marcadores moleculares na associação de sexos e identifição de espécies no Brasil. [Phd Thesis] Curitiba: Universidade Federal do Paraná. pp. 169
- Ekrem T., Roth S., Andersen T., Stur E., Søli G., Halvorsen G.A. 2012. Insects inhabiting freshwater and humid habitats in Finnmark, northern Norway. Norwegian Journal of Entomology, 59: 91-107.
- Espíndola A., Pellissier L., Alvarez N. 2011. Variation in the proportion of flower visitors of Arum maculatum along its distributional range in relation with community-based climatic niche analyses. Oikos, 120: 728-734. https://doi.org/10.1111/j.1600-0706.2010.18937.x
- Farish D.J., Axtell R.C. 1971. Phoresy redefined and examined in Macrocheles muscaedomesticae (Acarina: Macrochelidae). Acarologia, 13: 16-29.
- Floate K.D. 2011. Arthropods in Cattle Dung on Canada's Grasslands. In: Floate K.D. (Ed.) Arthropods of Canadian Grasslands (Volume 2): Inhabitants of a Changing Landscape. Biological Survey of Canada. p. 71-88. https://doi.org/10.3752/9780968932155
- Ghilyarov M.S., Bregetova N.G. 1977. (Eds): Key to the Soil Inhabiting Mites. Mesostigmata. Leningrad: Nauka Press.
- Gwiazdowicz D.J. 2005. Description of the male of Iphidozercon gibbus (BERLESE, 1903) (Acari: Mesostigmata). Genus, 16: 463-467.
- Gwiazdowicz D.J. 2007. Ascid mites (Acari, Mesostigmata) from selected forest ecosystems and microhabitats in Poland. Poznan: Wydawnictwo Akademii Rolniczej.
- Gwiazdowicz D.J., Gulvik M.E. 2005a. Checklist of Norwegian mesostigmatid mites (Acari, Mesostigmata). Norwegian Journal of Entomology, 52: 117-125.
- Gwiazdowicz D.J., Gulvik M.E. 2005b. Mesostigmatid mites (Acari, Mesostigmata) new to the fauna of Norway. Norwegian Journal of Entomology, 52: 103-109.
- Gwiazdowicz D.J., Gulvik M.E. 2007. The first records of five mite species (Acari, Mesostigmata) in Norway. Norwegian Journal of Entomology, 54: 125-127.
- Gwiazdowicz D.J., Solhøy T., Kaasa K. 2013. Five mesostigmatid mites (Acari, Mesostigmata) new to the Norwegian fauna. Norwegian Journal of Entomology, 60: 8-10.
- Houck M.A., O'Connor B.M. 1991. Ecological and Evolutionary Significance of Phoresy in the Astigmata. Annual Review of Entomology, 36: 611-636. https://doi.org/10.1146/annurev.en.36.010191.003143
- Hunter P.E., Rosario R.M.T. 1988. Associations of Mesostigmata with other arthropods. Annual Review of Entomology, 33: 393-417. https://doi.org/10.1146/annurev.en.33.010188.002141
- Hyatt K.H. 1980. Mites of the subfamily Parasitinae (Mesostigmata: Parasitidae) in the British Isles. Bulletin of the British Museum (Natural History), Zoology Series, 38: 344-347. https://doi.org/10.5962/p.12620
- Ježek J. 1983. Contribution to the taxonomy of the genus Logima Eat. (Diptera, Psychodidae). Acta Entomologica Musei Nationalis Pragae, 41: 213-259.
- Ježek J. 1990. Redescriptions of nine common Palearctic and Holarctic species of Psychodini End. (Diptera: Psychodidae). Acta Entomologica Musei Nationalis Pragae, 43: 33-83.
- Kalúz S., Fenda P. 2005. Mites (Acari: Mesostigmata) of the family Ascidae of Slovakia. Bratislava: Institute of Zoology Slovak Academy of Sciences.
- Karg W. 1993. Acari (Acarina), Milben, Unterordnung Anactinochaeta (Parasitiformes). Die freilebenden Gamasina (Gamasides), Raubmilben. Die Tierwelt Deutschlands, 59: 1-475.
- Klompen H. 2009. From sequence to phoresy - molecular biology in acarology. In: Sabelis M.W., Bruin J. (Eds.) Trends in Acarology. pp. 1-6. https://doi.org/10.1007/978-90-481-9837-5_1
- Krantz G.W. 2009. Habits and habitats. In: Krantz G.W., Walter, D.E. (Eds.) A manual of Acarology, 3rd ed. Lubbock: Texas Tech. University Press. pp. 64-82.
- Kvifte G.M. 2019. New records of Norwegian Psychodidae, with the first description of the female of Trichosepedon balkanicum (Krek, 1970) comb.nov. Norwegian Journal of Entomology, 66: 1-10.
- Kvifte G.M., Andersen T. 2012. Moth flies (Diptera, Psychodidae) from Finnmark, northern Norway. Norwegian Journal of Entomology, 59: 108-119.
- Kvifte, G.M., Boumans L. 2014. Further records and DNA barcodes of Norwegian moth flies (Diptera, Psychodidae). Norwegian Journal of Entomology, 61; 11-14.
- Kvifte G.M., Håland Ø., Andersen T. 2011. A revised checklist of Norwegian moth flies (Diptera, Psychodidae). Norwegian Journal of Entomology, 58: 180-188.
- Lundqvist L. 1998. Phoretic Gamasina (Acari) from Southern Sweden: Taxonomy, host preferences and seasonality. Acarologia, 39: 111-114.
- Mašán P., Országh I. 1994. Records of phoretic mites (Acarina, Mesostigmata) on biting midge Culicoides obsoletus (Meigen, 1818) (Diptera, Ceratopogonidae). Biologia (Bratislava), 492: 207-210.
- McGarry J.W., Baker A.S. 1997. Observations on the mite fauna associated with adult Stomoxys calcitrans in the U.K. Medical and Veterinary Entomology, 11: 159-164. https://doi.org/10.1111/j.1365-2915.1997.tb00307.x
- R Core Team. 2017. R: a language and environment for statistical computing. R foundation for statistical computing, Vienna. http://www.r-project.org

- Rapp A. 1959. Zur Biologie und Ethologie der Kafermilbe Parasitus coleoptratorum L. 1758 (Ein Beitrag zum Phoresie Problem). Zoologische Jahrbücher. Abteilung für Systematik, Ökologie und Geographie der Tiere, 86: 303-366.
- Seniczak A., Bolger T., Roth S., Seniczak S., Djursvoll P., Jordal B.H. 2019. Diverse mite communities (Acari: Oribatida, Mesostigmata) from a broadleaf forest in western Norway. Annales Zoologici Fennici, 56: 121-136. https://doi.org/10.5735/086.056.0111
- Seniczak A., Seniczak S., Schwarzfeld M.D., Coulson S.J., Gwiazdowicz D.J. 2020. Diversity and Distribution of Mites (Acari: Ixodida, Mesostigmata, Trombidiformes, Sarcoptiformes) in the Svalbard Archipelago. Diversity, 12: 323. https://doi.org/10.3390/d12090323
- Seniczak A., Seniczak S., Graczyk R., Kaczmarek S., Jordal B.H., Kowalski J., Djursvoll P., Roth S., Bolger T. 2021a. A forest pool as a habitat island for mites in a limestone forest in Southern Norway. Diversity, 13: 578. https://doi.org/10.3390/d13110578
- Seniczak A., Seniczak S., Starý J., Kaczmarek S., Jordal B.H., Kowalski J., Roth S., Djursvoll P., Bolger T. 2021b. High diversity of mites (Acari: Oribatida, Mesostigmata) supports the high conservation value of a broadleaf forest in Eastern Norway. Forests, 12: 1098. https://doi.org/10.3390/f12081098
- Svensson B.W. 2009. Fjärilsmyggfaunan i ett hagmarksområde och en ladugård i östra Blekinges skogsland. Med en översikt av familjen Psychodidae:s morfologi, systematik och utforskande, samt särskilt de svenska Psychoda s.l.-arternas biologi. Entomologisk Tidskrift, 130: 185-208.
- Tonnoir A.L. 1922. Synopsis des espèces Européennes du genre Psychoda (Diptères). Annales de la Société Entomologique de Belgique, 62: 49-88.
- Tonnoir A.L. 1940. A synopsis of the British Psychodidae (Dipt.) with descriptions of new species. Transactions of the Society for British Entomology, 7: 21-64.
- Walter D.E., Proctor H.C. 2013. Mites: Ecology, Evolution & Behaviour. Life at a Microscale. 2nd Edition. Dordrecht: Springer. https://doi.org/10.1007/978-94-007-7164-2
- Whitsel R.M., Schoeppner R.F. 1973. Mites associated with aquatic and semi-aquatic Diptera from San Mateo County, California. Proceedings of the Entomological Society of Washington, 75: 71-77.
- Withers P. 1988. Revisionary notes on British species of Psychoda Latreille (Diptera, Psychodidae) including new synonyms and a species new to science. British Journal of Entomology and Natural History, 1: 69-76.
- Withers P. 1989. Moth flies. Diptera: Psychodidae. Dipterists Digest, 4: 1-83.
- Yeargan K.V., Quate L.W. 1996. Juvenile Bolas spiders attract Psychodid flies. Oecologia, 106: 266-271. https://doi.org/10.1007/BF00328607



2021-01-11
Date accepted:
2022-08-18
Date published:
2022-09-23
Edited by:
Roy, Lise

This work is licensed under a Creative Commons Attribution 4.0 International License
2022 Kvifte, Gunnar Mikalsen; Kaczmarek, Sławomir; Marquardt, Tomasz and Seniczak, Anna
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



