First record of family Otopheidomenidae Treat (Acari: Mesostigmata) in Central Asia, with description of a new species of Nabiseius Chant & Lindquist
Joharchi, Omid  1
; Khaustov, Vladimir A.
1
; Khaustov, Vladimir A.  2
and Stanyukovich, Maria K.3
2
and Stanyukovich, Maria K.3
1✉ Institute of Environmental and Agricultural Biology (X-BIO), Tyumen State University, Semakova Str. 10, 625003 Tyumen, Russia.
2Institute of Environmental and Agricultural Biology (X-BIO), Tyumen State University, Semakova Str. 10, 625003 Tyumen, Russia.
3Zoological Institute of Russian Academy of Sciences, Saint Petersburg, Russia.
2022 - Volume: 62 Issue: 2 pages: 431-445
https://doi.org/10.24349/xo13-9sb4ZooBank LSID: 8028C439-31B3-46BB-AF59-E5297CDE8BE5
Original research
Keywords
Abstract
Introduction
Members of the family Otopheidomenidae (Acari: Mesostigmata) have been reported as ectoparasites on different orders of insects (e.g., Hemiptera, Orthoptera and Lepidoptera) (Costa 1968; Prasad 1970a, b, 1972, 1973, 1975; Fain and Lukoschus 1983; Zhang 1995). One of its genera, Nabiseius Chant & Lindquist, is known as a group of ectoparasite mites that are more intimate association with damsel bugs (Hemiptera: Nabidae) (Halliday 1994; Zhang 1995; Menon et al. 2011; Negm and Alatawi 2013). The genus comprises only four nominal species: Chant and Lindquist (1965) erected the genus Nabiseius to accommodate a new species N. duplicisetus, which was described as an ectoparasite on an unknown nabid bug found with grapes from Chile, intercepted in New York, USA. Amitai and Swirski (1980) described Nabiseius rivnayae from Israel where it was found on salt cedar, Tamarix sp. (Plantae: Tamaricaceae). Afterwards, Halliday (1994) extended the geographical range of the genus to Australia by describing a new species, N. melinae, in association with plant bugs, Creontiades sp. (Hemiptera: Miridae). Finally, Negm and Alatawi (2013) described N. arabicus on Bermuda grass, Cynodon dactylon (L.) Pers. (Plantae: Poaceae) in a date palm orchard from Saudi Arabia. Herein, we describe a new species belonging to the genus Nabiseius on the basis of specimens (including adult female, larva and deutonymphal stages) collected on Nabis palifer Seidenstücker (Hemiptera: Nabidae) from Tajikistan and Turkmenistan and extend the geographical range of the family Otopheidomenidae to include Central Asia.
Material and methods
The specimens documented in this study originate from the collection of the Zoological Institute of the Russian Academy of Sciences (ZIN, Saint Petersburg). The line drawings and examinations of the specimens were performed with Zeiss Axio Imager A2 and Leica DM 2500 compound microscopes equipped with drawing tubes and differential interference contrast and phase contrast optical systems, attached to cameras AxioCam ICc 5 and ICC50 HD, respectively. Most images were captured in stacks (with focal depth manually controlled). Selected images were combined using the Helicon Focus software (Version 7.6.4 Pro) by ©Helicon Soft Ltd., 2000 to produce final images with enhanced quality. Digital drawings were prepared using Adobe Photoshop CS2 software based on the original pencil line drawings. Images and morphological measurements were taken via ZEN 2012 software (version 8.0) and Leica Application Suite (LAS) software (version 4.2, Live and Interactive Measurements modules). Photomicrographs were taken with an AxioCam 506 camera (Carl Zeiss, Germany). Measurements of structures are expressed as ranges (minimum–maximum) in micrometers (μm). The length and width of the dorsal shield were taken from the anterior to the posterior margins along the midline, and at level of r3, respectively. Length and width of the sternal shield were measured at the maximum length and broadest points (at level between setae ST1, ST2 and posterior to ST2), respectively. The length of the genital shield was measured along the midline from the anterior margin of the hyaline extension to the posterior margin of the shield, and its width where maximal, posteriorly to genital setae ST5. The ventrianal shield length and width were measured along the midline from the anterior to posterior margins, including the cribrum, and at the broadest point (at level of JV2), narrowest (at level of JV1), respectively. The lengths of the second cheliceral segment were taken from their base to the apex of the fixed digit, and their widths at the broadest point. The lengths of the fixed cheliceral digits were measured from the dorsal poroid to the apex and that of the movable digits from the base to apex. Leg length was measured from the base of the coxa to the apex of the tarsus (excluding the pre-tarsus). The nomenclature used for the dorsal idiosomal chaetotaxy follows that of Chant and Yoshida-Shaul (1991, 1992), Chant (1993) and Prasad (2011). The notations for leg and palp setae follow those of Chant and Lindquist (1965) and Evans (1963a, b). Other anatomical structures mostly follow Evans and Till (1979). Notations for idiosomal pore-like structures (gland pores and poroids/lyrifissures) and peritrematal shield follow mostly Athias-Henriot (1971, 1975). The notations for pore-like structures on the sternal shield and for the peritrematal shield region also follow modifications and additions by Johnston and Moraza (1991). The holotype and paratypes are deposited at the ZIN.
Taxonomy
Family Otopheidomenidae Treat, 1955
Subfamily Treatiinae Wainstein, 1972
Genus Nabiseius Chant & Lindquist
Nabiseius Chant & Lindquist, 1965: 516.
Type species Nabiseius duplicisetus Chant & Lindquist, 1965: 516; by original designation.
Diagnosis — The concept of Nabiseius used here is based on that of Chant and Lindquist (1965) with modifications by Wainstein (1972) and Zhang (1995).
Nabiseius palifer Joharchi & Khaustov n. sp.
ZOOBANK: 90079BC8-08B2-4ADE-B345-97D0817C6C0C ![]()
(Figures 1–9)
Type material
Holotype, Kondara gorge, 1100 m. a. s. l., Varzob river valley, Tajikistan, 19 June 1937, Gusakovsky coll., under elytra of Nabis palifer Seidenstücker (Hemiptera: Nabidae). Paratypes, three females with same data as holotype; one female, three larvae, and eight deutonymphs, Chapaev (Niyazov) settlement, Turkmenistan, 05 June 1931, Frolova coll., under elytra of Nabis palifer Seidenstücker (Hemiptera: Nabidae) (in ZIN).
Diagnosis (adult female)
Dorsal shield entire, broad, reticulate throughout, smooth centrally (or faintly reticulate), bearing 18 pairs of moderately long setae, posterior margin of shield truncated; sternal shield smooth, with two pairs of setae and a median notch in its anterior margin, without distinguishable pore-like structures, shield with irregular posterior margin, ST3 and metasternal setae (ST4) located on soft integument; metasternal platelets absent. Genital shield wedge-shaped, faintly lineate, anterior hyaline margin of shield convex, extending beyond base of setae ST3, ventrianal shield sub-pentagonal, transversely lineate throughout (more distinct between JV1–2) and two pairs of very long pre-anal setae (JV1–2), in addition to circum-anals, anus large and located in posterior part of shield. Soft opisthogastric cuticle with 5–6 pairs of very long setae and a pair of incompletely divided metapodal plates; fixed digit of chelicera reduced to a small protuberance, proximally with membranous lobe, movable digit long, slender and bidentate.
Description adult female
(Figures 1–5)
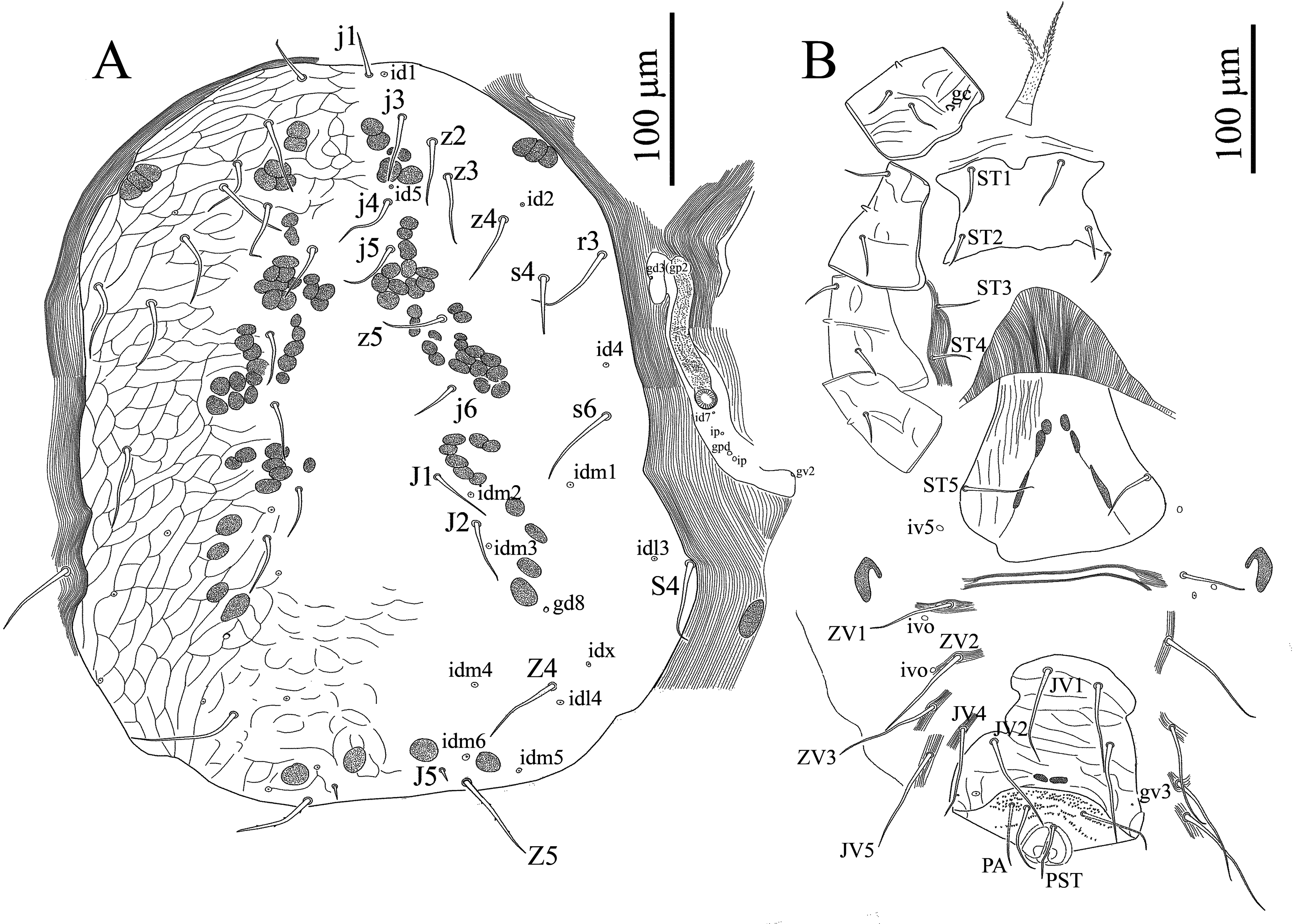













Five specimens measured.
Dorsal idiosoma — (Figures 1A & 5A) — Length 590–604, width 365–375. Dorsal shield entire, broad, 500–510 long, 300–315 wide, truncate posteriorly, not covering entire dorsal idiosoma, leaving broad band of membrane exposed, and strongly expanded laterally surrounding dorsal shield, reticulate throughout, smooth centrally (or faintly reticulate) and 18 pairs of moderately long setae (39–50): 12 pairs of podonotal setae (j1, j3–j6, z2–z5, s4, s6, r3), six pairs of opisthonotal setae (J1, J2, J5, Z4, Z5, S4), almost uniform in length and smooth except Z4, Z5 and S4 (52–58) longer and Z5 with 2–4 small barbs, j1 (27–30) shorter and J5 (7–10) shortest. Cuticle around dorsal shield strongly striated; occasionally with setae S4 (in holotype and two paratypes). Muscle insertions prominent as desclerotized circular patches. Dorsal shield with 13 pairs of pore-like structures (id1, id2, id4, id5, idm1-idm6, idx, idl3, idl4), including one pair of gland opening (gd8).
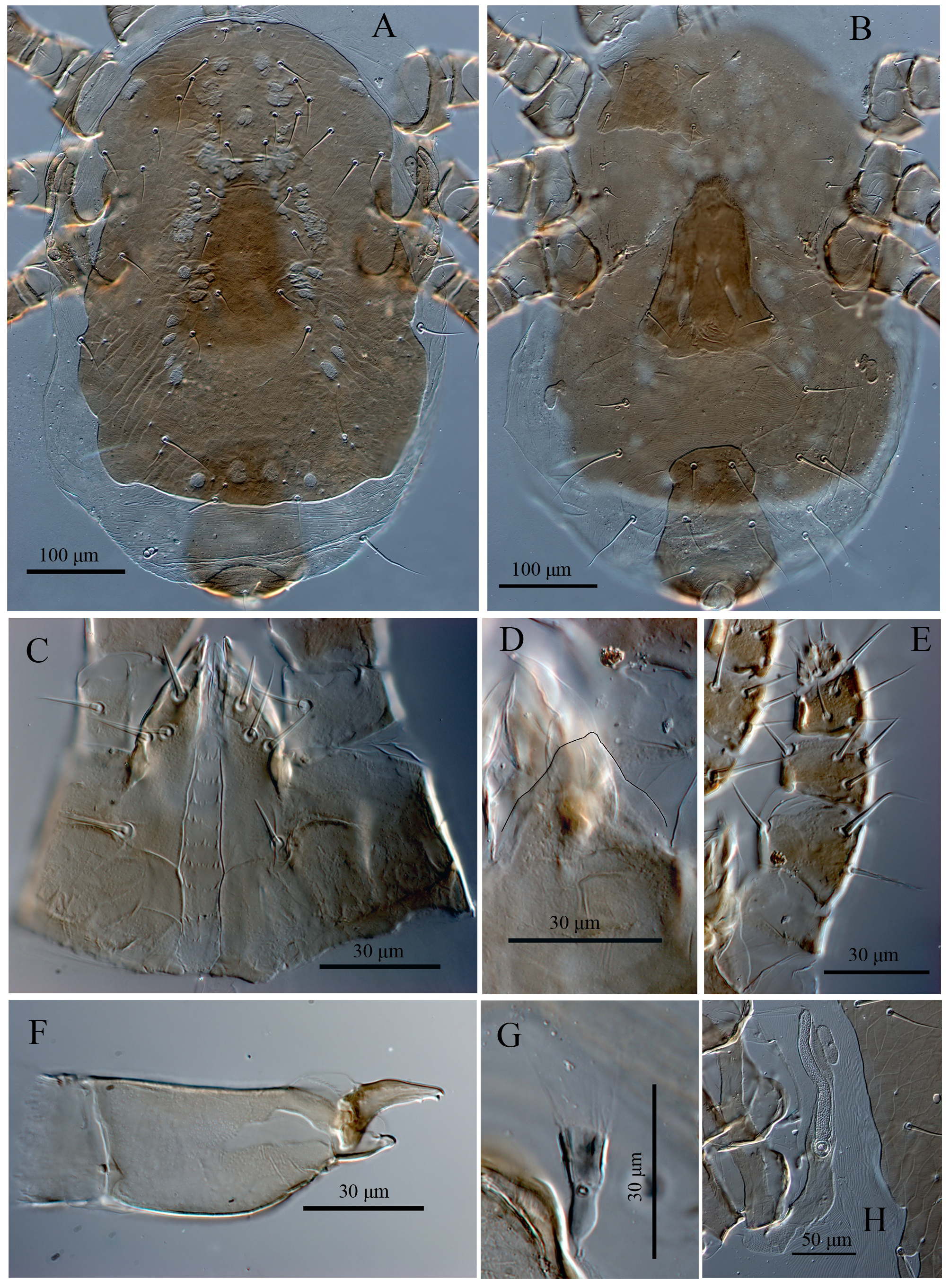
Ventral idiosoma — (Figures 1B, 5B & 5H) — Tritosternum with paired pilose laciniae (81–85), fused along basal third of their length (33–35), columnar base 15–17 × 19–21 wide; presternal area lightly sclerotized, with a few transverse curved lines. Sternal shield (length 71–96) narrowest (79–84), widest (106–110), smooth almost throughout (or faintly reticulate), with two pairs of smooth setae (ST1 32–34, ST2 25–27), and a median notch in its anterior margin, without distinguishable pore-like structures, with irregular posterior margin, endopodal extensions between coxae I and II, II and III united with sternal shield, those between III-IV absent. Sternal setae ST3 (26–28), ST4 (metasternal setae) (25–27) located on soft integument, metasternal poroids and platelets absent (Figures 1B & 5B). Genital shield somewhat wedge-shaped, length 203–209, width at broadest level 136–147, faintly lineate, anterior hyaline margin of shield convex, extending beyond base of setae ST3, poroids iv5 located on soft cuticle posterolaterad ST5, lateral margins relatively constricted anterior to genital setae and posterior margin irregularly rounded, postgenital platelets absent (Figures 1B & 5B). Ventrianal shield sub-pentagonal, narrower than genital shield, with irregular margins, length 205–220, maximum width 108–115 (at level of JV2), narrowest (at level of JV1) 84–86, transversely lineate throughout (more distinct between JV1–2) and two pairs of smooth and very long pre-anal setae (JV1–2 (68–78)), in addition to circum-anal setae (in three specimens (including holotype) a supernumerary seta between post-anal and para-anal setae present, Figure 1B), gland pores gv3 small, rounded, present on lateral margins of shield at level of para-anal setae (Figures 1B & 5B). Anterior margin of shield slightly convex, lateral margins concaved between levels of JV1–2, anus large and located in posterior part of shield, para-anal setae (54–58) slightly longer than post-anal seta (47–51), cribrum wide, limited to region posterior to post-anal seta. Soft opisthogastric cuticle surrounding genital, ventrianal shields, with one pair of suboval (sometimes incompletely divided) metapodal plates (29–31 long × 17–19 wide), two pairs of poroids (ivo) and 5–6 pairs of very long setae (JV4–JV5, ZV1–ZV3, JV3 occasionally present); setae relatively uniform in length (72–80) (Figures 1B & 5B). Peritremes short, anteriorly extending to level of coxae III (setae r3) (Figures 1B & 5H), peritrematal shield reduced to small platelet laterad peritremes, bearing one gland pores at level near coxae III (Figures 1B & 5H), fused posteriorly to expodal-parapodal curving behind coxae IV, with three pairs of poroids and two pair of gland pores (including gv2), free anteriorly, not extending beyond peritremes to connect anterolateral extensions of dorsal shield (Figures 1B & 5H).
Gnathosoma — (Figures 2A, 2B, 2D-F & 5C-F) — Epistome subtriangular, with entirely smooth margin (Figures 2B & 5D). Hypostomal groove with nine transverse rows of denticles, each row with 4–9 variously sized denticles, with smooth anterior convex transverse line, groove progressively narrowing from posterior to anterior. Corniculi (Figures 2A & 5C) considerably short and internal malae shorter than corniculi, comprising a pair of smooth median projections, hypostomal and capitular setae smooth, h3 (18–20) < h1, h2 (14–17) < pc (13–15); h1 and pc slightly thickened, with blunt tip. Chaetotaxy of palps: trochanter 1, femur 4, genu 5, tibia 12, tarsus 10, all setae smooth; palptarsus apotele weakly two-tined at apex (Figures 2D, 2E & 5E). Second segment of chelicera 76–79 long; cheliceral fixed digit reduced to small protuberance 17–20 long with membranous lobe proximally, pilus dentilis absent; movable digit 30–32 long, slender and bidentate, dorsal cheliceral seta short and thick, arthrodial membrane with a rounded flap and short filaments; cheliceral lyrifissures indistinct (Figures 2F & 5F).
Insemination structures — (Figures 2C & 5G) — Phytoseiid type, calyx of spermatheca funnel-shaped, atrium incorporated in the calyx, minor duct thin and prominent, major duct visible.
Legs — (Figures 3 & 4) — Legs II and III short (323–355, 333–346), I and IV longer (369–397, 355–372) (excluding pre-tarsus). Chaetotaxy normal for genus Nabiseius (sensu Chant and Lindquist 1965): Leg I (Figure 3A): coxa 0-0/1, 0/1-0, trochanter 1-0/1, 1/2-1, femur 2-2/1, 2/2-2 (ad1 and pd1 longer 53–58), genu 2-2/1, 2/1-2 (dorsals slightly thicker and longer 39–44), tibia 2-2/1, 2/1-2. Leg II (Figure 3B): coxa 0-0/1, 0/1-0, trochanter 1-0/1, 0/2-1, femur 1-2/1, 2/1-1 (ad1 and pd1 longer 53–58), genu 1-2/0, 2/0-1 (dorsals slightly thicker and longer 39–44), tibia 1-1/1, 2/1-1. Leg III (Figure 3C): coxa 0-0/1, 0/1-0, trochanter 1-1/2, 0/1-0, femur 1-2/1, 1/0-0 (ad1 thicker and longer 56–58), genu 1-2/1, 2/0-1 (dorsals slightly thicker and longer 40–44, av present), tibia: 1-1/1, 2/1-1. Leg IV (Figure 3D): coxa 0-0/1, 0/0-0, trochanter 1-1/2, 0/1-0 (ad slightly longer), femur 1-2/1, 1/0-0 (ad1 thicker and longer 55–57), genu 1-2/1, 2/0-0 (dorsals slightly thicker and longer 43–48), tibia 1-1/1, 1/0-1. Tarsus I (Figure 4A) with an irregular shallow pit on dorso-distal edge of telotarsus discernible, with six short and modified setae. Tarsi II-IV with 18 setae (3-3/2, 3/2-3 + mv, md), all pretarsi with a well-developed membranous ambulacrum and claws indiscernible (Figure 4A-D).
Description larva
(Figures 6 & 7)






Three specimens measured.
Dorsal idiosoma — (Figure 6A) — Whitish, oval (254–259 long × 190–202 wide), with podonotal (155–160 long × 150–155 wide) and pygidial (50–55 long × 106–111 wide) shields, mesonotal scutellae absent. Podonotal shield with nine pairs of simple setae (j1, j3–6, z2, z4, z5, s4) and two pairs of discernible pore-like structures (id4, id5). All setae similar in length (9–12) except j1 (8–10) shortest and j6 (27–30) longest. Pygidial shield without setae, 4–5 pairs of rounded structures in alveoli shape (incipient setae) present on shield and four pairs located on soft integument between podonotal and pygidial shields.
Ventral idiosoma — (Figure 6B) — Sternal shield indistinct, intercoxal area with three pairs of smooth setae (ST1 (13–15), ST2 and ST3 (8–11)), related poroids, metasternal (ST4) and genital setae (ST5) absent. Anal shield small, suboval, weakly sclerotized, margins hardly conspicuous, circumanal setae smooth, postanal seta (14–17) shorter than para-anal setae (25–28). Soft opisthogastric cuticle with three pairs of setae (JV1, JV2, ZV2) and three pairs of rounded structures in alveoli shape (incipient setae); setae relatively uniform in length (8–11), except JV2 (17–21) longest. Metapodal plates and peritremes absent.
Gnathosoma — (Figure 6C-E) — Similar to the adult except hypostome with two pairs of setae (h1 and h2), h3 and palpcoxal setae absent and deutosternal denticles indiscernible (Figure 6C). Chaetotaxy of palps: trochanter 0, femur 4, genu 5, tibiotarsus 19, all setae smooth (Figure 6D). Cheliceral fixed digit reduced to small protuberance as in adult female; movable digit long, slender with one tooth, dorsal cheliceral seta, arthrodial membrane and cheliceral lyrifissures indistinct (Figure 6E).
Legs — (Figure 7) — Legs II and III short (147–151, 154–158), I longer (162–170) (excluding pre-tarsus). Chaetotaxy: Leg I (Figure 7A): coxa 0-0/1, 0/1-0, trochanter 1-0/1, 0/1-1, femur 2-2/1, 2/1-2, genu 1-2/1, 2/1-1, tibia 1-2/1, 2/1-1. Leg II (Figure 7B): coxa 0-0/1, 0/1-0, trochanter 1-0/1, 0/1-1, femur 1-2/1, 2/0-1, genu 1-2/0, 2/0-1, tibia 1-1/1, 2/1-1. Leg III (Figure 7C): coxa 0-0/1, 0/1-0, trochanter 1-1/2, 0/1-0, femur 1-2/1, 1/0-0, genu 1-2/0, 1/0-1, tibia: 1-1/1, 2/1-1. Tarsi II-III with 16 setae (3-3/2, 3/2-3, mv, md absent). All pretarsi with a well-developed membranous ambulacrum and claws indiscernible.
Description deutonymph
(Figures 8 & 9)
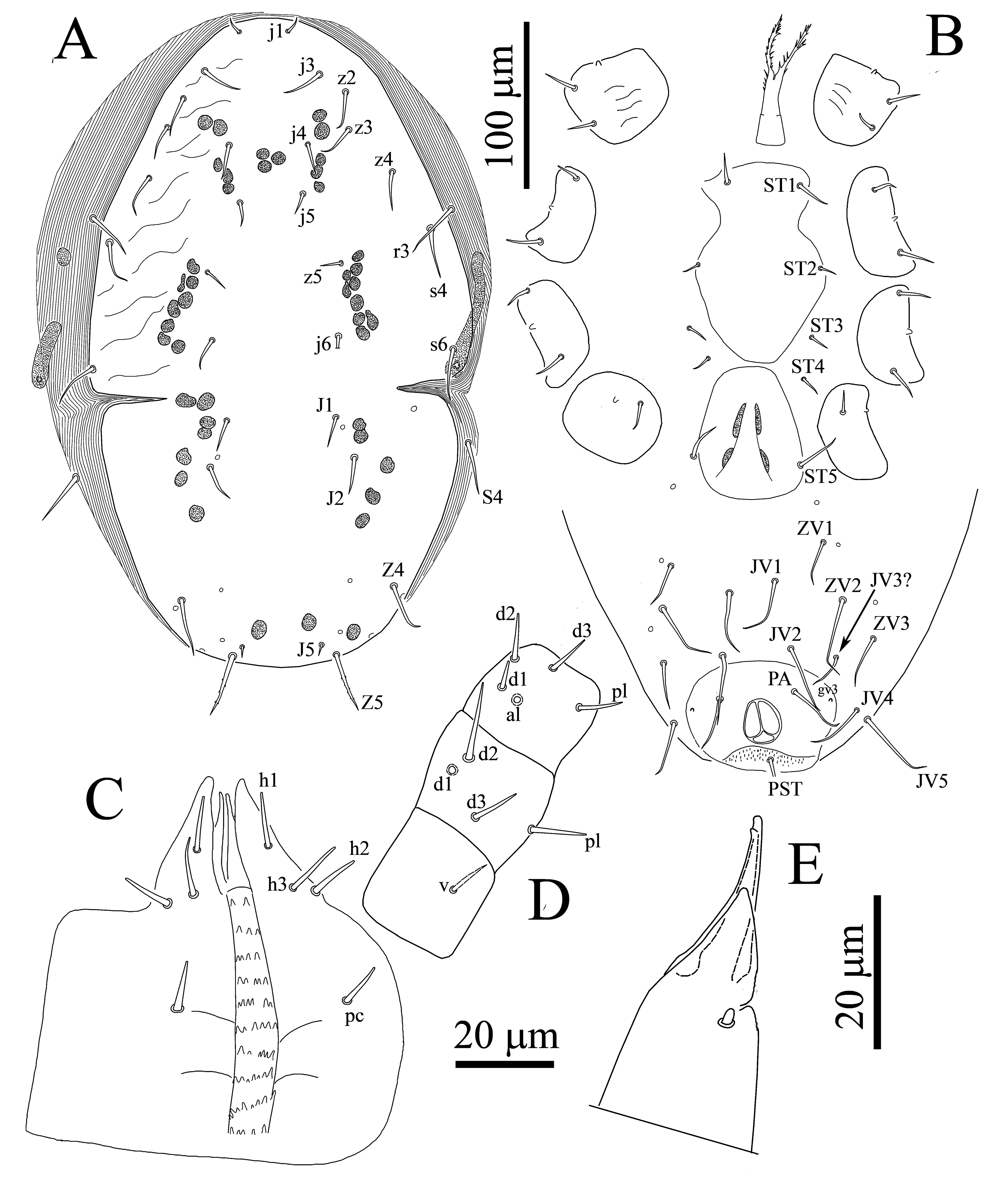





Seven specimens measured
Dorsal idiosoma — (Figure 8A) — Dorsal shield entire, oval, 378–385 long, 200–271 wide, not covering entire dorsal idiosoma, leaving broad band of membrane exposed, and strongly expanded laterally surrounding dorsal shield, shield with weak reticulation, podonotal and opisthonotal sections separated by lateral incisions at a level between j6 and J1. Podonotal region with 11 pairs of setae (j1, j3–j6, z2–z5, s4, s6) plus r3 on lateral soft cuticle. Opisthonotal region with five pairs of setae (J1, J2, J5, Z4, Z5), S4 on lateral soft cuticle. Almost all setae uniform in length (20–28) and smooth except r3, Z4, Z5 (36–41) and s6, S4 (33–36) longer and Z5 with 2–4 small barbs, j1 (7–10) shorter and J5 (5–8) shortest.
Ventral idiosoma — (Figure 8B) — Tritosternum with paired pilose laciniae (52–57), fused along basal third of their length (21–23), columnar base 14–18 × 19–21 wide. Sternal shield somewhat pandurate shape (length 116–123) widest (72–80), smooth throughout, with two pairs of smooth setae (ST1 20–24, ST2 13–16), without distinguishable pore-like structures, sternal setae ST3 and ST4 (12–16) present, located on soft integument, metasternal poroids, related platelets and endopodal extensions absent. Genital shield completely divided from sternal shield, not developed, somewhat obovate, length 73–84, width at broadest level 58–71, poroids iv5 located on soft cuticle posterolaterad st5. Anal shield large, smooth, subcircular, length 72–82, maximum width 80–90 (at level of gv3), bearing only circum-anal setae, anal gland pores (gv3) on anterolateral margin of shield, anus large, para-anal setae (30–37) slightly longer than post-anal seta (23–28), cribrum wide, limited to region posterior to post-anal seta. Soft opisthogastric cuticle with 7–8 pairs of very long setae (JV1–JV5, ZV1–ZV3); in some specimens JV3 absent, setae relatively uniform in length (37–52). Peritremes slightly shorter than as in adult female, peritrematal shield and metapodal plates absent.
Gnathosoma — (Figure 8C-E) — Similar to adult female, except cheliceral movable digit with one tooth, dorsal cheliceral seta distinct, arthrodial membrane and cheliceral lyrifissures indistinct (Figure 8E).
Legs — (Figure 9) — Legs II (Figure 9B) and III (Figure 9C) short (250–263), I (Figure 9A) and IV (Figure 9D) longer (265–275, 278–281) (excluding pre-tarsus). Chaetotaxy similar to adult female.
Male and protonymph
Unknown.
Etymology
The name of this species refers to its occurrence on damsel bug of the species Nabis palifer Seidenstücker (Hemiptera: Nabidae).
Remarks
Nabiseius palifer Joharchi & Khaustov most resembles the type species of the genus, N. duplicisetus Chant & Lindquist, 1965 (couplet 2 in the identification key to Nabiseius species provided by Negm and Alatawi 2013), due to the entire dorsal shield, genital setae (ST5) present, anus located in posterior part of ventrianal shield (terminal) and one pair of metapodal plates present. Nabiseius palifer differs from N. duplicisetus by having 18 pairs of dorsal setae (versus 19 in N. duplicisetus, a supernumerary pair setae between z2 and z3 present), sternal and metasternal setae (ST3 and ST4) located on soft integument (versus ST4 absent in N. duplicisetus), pre-anal and opisthogastric setae long enough to reach well past the base of next posterior seta (versus in N. duplicisetus both pre-anal and opisthogastric setae short), genu III with seven setae (1-2/1, 2/0-1) (versus in N. duplicisetus genu III with six setae, av absent). Nabiseius palifer Joharchi & Khaustov is also similar to N. rivnayae Amitai & Swirski, 1980, due to the dorsal shield having 18 pairs of setae (three pairs of setae present in J series and a supernumerary pair setae absent) and incompletely divided metapodal plates, however, the new species can easily be distinguished from N. rivnayae by having much longer dorsal setae (especially S4, Z4 and Z5, more than six times the length of J5), sternal and metasternal setae (ST3 and ST4) located on soft integument and metasternal platelets absent, while in N. rivnayae dorsal setae much shorter (S4, Z4 and Z5 about four times the length of J5), ST4 absent and ST3 located on metasternal platelets.
Acknowledgements
The present research was supported by the cooperative agreement No. FEWZ-2021–0004 from the Russian Ministry of Science and Higher Education; and for Maria K. Stanyukovich was supported by the state task for ZIN RAS АААА-А19-1021051603202-7. We are deeply grateful to Zoological Institute of the Russian Academy of Sciences (ZIN, Saint Petersburg) for their hospitality of the first author during his visit to the Museum.
References
- Amitai S., Swirski E. 1980. Two new species of phytoseiid mites (Mesostigmata: Phytoseiidae) from Israel. Isr. J. Entomol., 14: 1-7.
- Athias-Henriot C. 1971. La divergence néotaxique des Gamasides (Arachnides). Bulletin Sc Bourgogne, 28: 93-106. [in French].
- Athias-Henriot C. 1975. Nouvelles notes sur les Amblyseiini. 2. Le relevé organotaxique de la face dorsale adulte (gamasides, protoadéniques, Phytoseiidae). Acarologia, 17: 20-29. [in French].
- Chant D.A. 1993. Adaptive radiation in the family Phytoseiidae (Acari: Gamasina) as reflected by adult idiosomal setation. Internat. J. Acarol., 19: 203-231. https://doi.org/10.1080/01647959308683548
- Chant D.A., Lindquist E.E. 1965. Nabiseius duplicisetus, a new genus and species of Otopheidomenidae (Acarina: Phytoseiidae) from nabid bugs. Can. Entomol., 97: 515-521. https://doi.org/10.4039/Ent97515-5
- Chant D.A., Yoshida-Shaul E. 1991. Adult ventral setal patterns in the family Phytoseiidae (Acari: Gamasina). Internat. J. Acarol., 17: 187-200. https://doi.org/10.1080/01647959108683906
- Chant D.A., Yoshida-Shaul E. 1992. Adult idiosomal setal patterns in the family Phytoseiidae (Acari: Gamasina). Internat. J. Acarol., 18(3): 177-199. https://doi.org/10.1080/01647959208683949
- Costa M. 1968. Notes on the genus Hemipteroseius Evans (Acari: Mesostigmata) with the description of a new species from Israel. J. Nat. Hist., 2: 1-15. https://doi.org/10.1080/00222936800770581
- Evans G.O. 1963a. Observations on the classification of the family Otopheidomenidae (Acari: Mesostigmata) with description of two new species. Ann. Mag. Nat. Hist., Ser. 13 (5): 609-620. https://doi.org/10.1080/00222936208651292
- Evans G.O. 1963b. Some observations on the chaetotaxy of the pedipalps in the Mesostigmata (Acari). Ann. Mag. Nat. Hist., Ser. 13 (6): 513-527. https://doi.org/10.1080/00222936308651393
- Evans G.O., Till W.M. 1979. Mesostigmatic mites of Britain and Ireland (Chelicerata: Acari Parasitiformes). An introduction to their external morphology and classification. Transactions of the Zoological Society of London, 35: 145-270. https://doi.org/10.1111/j.1096-3642.1979.tb00059.x
- Fain A., Lukoschus F.S. 1983. Katydiseius nadchatrami n.g., n. sp., (Acari: Otopheidomenidae) from the trachea of a Malaysian katydid Chloracris brullei Pictet & Saussure, 1892 (Orthoptera, Pseudophyllidae). Internat. J. Acarol., 9: 173-178. https://doi.org/10.1080/01647958308683333
- Johnston D.E., Moraza M.L. 1991. The idiosomal adenotaxy and poroidotaxy of Zerconidae (Mesostigmata: Zerconina). In: Dusbábek F., Bukva V. (Eds). Modern Acarology. Vol. 2. Academia: Prague. p. 349-356.
- Halliday R.B. 1994. First record of the family Otopheidomenidae (Acarina: Mesostigmata) in Australia, with description of Nabiseius melinae sp. n. J. Aust. ent. Soc., 33: 347-350. https://doi.org/10.1111/j.1440-6055.1994.tb01243.x
- Menon P., Joshi S., Hussain M., Ramamurthy V.V. 2011. A new species of Hemipteroseius (Acari: Otopheidomenidae) parasitic on Dysdercus (Hemiptera: Pyrrhocoridae) in India. Zootaxa, 2800: 53-63. https://doi.org/10.11646/zootaxa.2800.1.4
- Negm M.W., Alatawi F.J. 2013. First record of Otopheidomenidae Treat, 1955 (Acari: Mesostigmata) in Saudi Arabia, with description of Nabiseius arabicus sp. nov. Turk. J. Zool., 37(2): 184 -187.
- Prasad V. 1970a. Two new species of Otopheidomenis mites from South America (Acarina: Phytoseiidae). Acarologia, 12(1): 28-33.
- Prasad V. 1970b. Two new species of Otopheidomenis (Acarina: Phytoseiidae) ectoparasitic on sphingid moths with a note on Noctuiseius., Can. Entomol., 102(10): 1209-1215. doi:10.4039/Ent1021209-10 https://doi.org/10.4039/Ent1021209-10
- Prasad V. 1972. New species of Otopheidomenis (Acarina: Phytoseiidae) ectoparasitic on sphingid moths from Uganda. Acarologia, 14(3): 345-249.
- Prasad V. 1973. A new species of Otopheidomenis (Acarina: Otopheidomenidae) from India. Acarologia, 15(2): 193-196.
- Prasad V. 1975. Ectoparasitic mites. (Acarina: Otopheidomenidae) on sphingid moths. Acarologia, 17: 375-383.
- Prasad V. 2011. Proposed nomenclature for idiosomal setae in otopheidomenid mites (Acari: Otopheidomenidae) known from sphingid moths (Lepidoptera: Sphingidae). Internat. J. Acarol., 37(1): 11-30. https://doi.org/10.1080/01647954.2010.489055
- Treat A.E. 1955. An ectoparasite (Acarina: Mesostigmata) from moths of the genus Zale. J. Parasitol., 41: 555-561. https://doi.org/10.2307/3274134
- Wainstein B.A. 1972. On the system of ectoparasitic mite of the family Otopheidomenidae Treat, 1955 (Parasitiformes). Parazit., 5: 451-456. [in Russian].
- Zhang Z.-Q. 1995. Review of the systematics and biology of the Otopheidomenidae (Acari: Mesostigmata) with a description of Eickwortius gen. n. from a termite (Isoptera: Termitidae). Syst. Entomol., 20: 239-246. https://doi.org/10.1111/j.1365-3113.1995.tb00095.x



2022-03-18
Date accepted:
2022-04-14
Date published:
2022-04-15
Edited by:
Faraji, Farid

This work is licensed under a Creative Commons Attribution 4.0 International License
2022 Joharchi, Omid; Khaustov, Vladimir A. and Stanyukovich, Maria K.
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



