Acucapito hainanensis sp. nov., the first record of the family Acucapitidae Wiles, 1996 (Acari, Hydrachnidia) from China
Li, Hai-Tao1 ; Jin, Dao-Chao2 and Guo, Jian-Jun3
1Institute of Entomology, Guizhou University, Scientific Observing and Experimental Station of Crop Pests in Guiyang, Ministry of Agricultural and Rural Affairs of the P. R. China, Guiyang 550025, P. R. China.
2Institute of Entomology, Guizhou University, Scientific Observing and Experimental Station of Crop Pests in Guiyang, Ministry of Agricultural and Rural Affairs of the P. R. China, Guiyang 550025, P. R. China.
3✉ Institute of Entomology, Guizhou University, Scientific Observing and Experimental Station of Crop Pests in Guiyang, Ministry of Agricultural and Rural Affairs of the P. R. China, Guiyang 550025, P. R. China.
2022 - Volume: 62 Issue: 1 pages: 250-261
https://doi.org/10.24349/h7mp-06fxZooBank LSID: 3C0BE9F5-C4E0-4A3B-8E33-8D67C82294AF
Original research
Keywords
Abstract
Introduction
The family Acucapitidae Wiles, 1996 belongs to the superfamily Lebertioidea Thor, 1900 based on the sperchontid-type genital field of the nymph, and also bear similarities to the Oxidae K. Viets, 1926 and the Lebertiidae Thor, 1900. However, because of great differences (e.g. distinctive median suture of ventral shield, completely different gnathosoma and palp) between acucapitids and above three families, Wiles (1996) established Acucapitidae as a monotypic family for Acucapito naso Wiles, 1996 from Brunei. Another species, Acucapito vietnamensis Tuzovskij, 2009, was described from Vietnam, and until now these two species represented the only members of the Acucapitidae (Wiles 1996, Tuzovskij 2009, Smit 2020).
In a field survey of water mites from Hainan Province we found a new species of this rare family that we describe here as Acucapito hainanensis Li & Guo sp. nov. This adds another family of water mites to the Chinese fauna. Furthermore, some structures of acucapitids are distinctive, especially palp structures that are difficult to resolve using light microscopy because of its minute fifth segment. Consequently, we take SEM (scanning electron microscope) micrographs to learn more about these structures.
Material and methods
For details of collection and preservation of water mites and preparation of slides see Gu et al. (2021). A 30 cm in diameter net with fine mesh size (250 μm) is positioned in streams, then turning over the cobblestones at the bottom of the river, so that flowing water will carry mosses, aquatic plants and dislodged organisms into it. Next, two stacked sieves (mesh size 4 mm above, 250 µm below) are used to remove leaves and other large impurities. Finally, a 2-mL dropper and a white tray are used to capture water mites. Specimens are preserved in Koenike's fluid and mounted in gelatin mounting fluid.
Slide-mounted specimens were examined and illustrated using a Leica DM3000 microscope. In the illustrations, dashed lines of eye lenses and the fourth leg sockets mean these structures under the surface, short curved lines on cuticle indicate muscle attachment scars. All photographs and illustrations were edited using Adobe Photoshop CS6. Measurements were taken by Nikon Ni-E (with Nikon DS-Ri2 camera). All measurements are given in μm, the measurements outside parentheses belong to holotype and allotype, the ranges in parentheses are the measurements for all collected specimens.
For SEM, specimens were kept for 12 hours in 2% glutaraldehyde at 4°C. Afterwards, the specimens were dehydrated through a graded ethanol series of 30%, 50%, 75%, 85%, and 95% (for 30 min each), then fully dehydrated in absolute ethanol (seven times for 24 hours each). After drying (40°C) in an electric blast drying oven (WGLL-125BE) for 12 hours, the specimens were attached to a holder using electrical adhesive tape, sputter-coated with gold, examined and photographed with a JCM6000 Desktop SEM (at 15.0 kV).
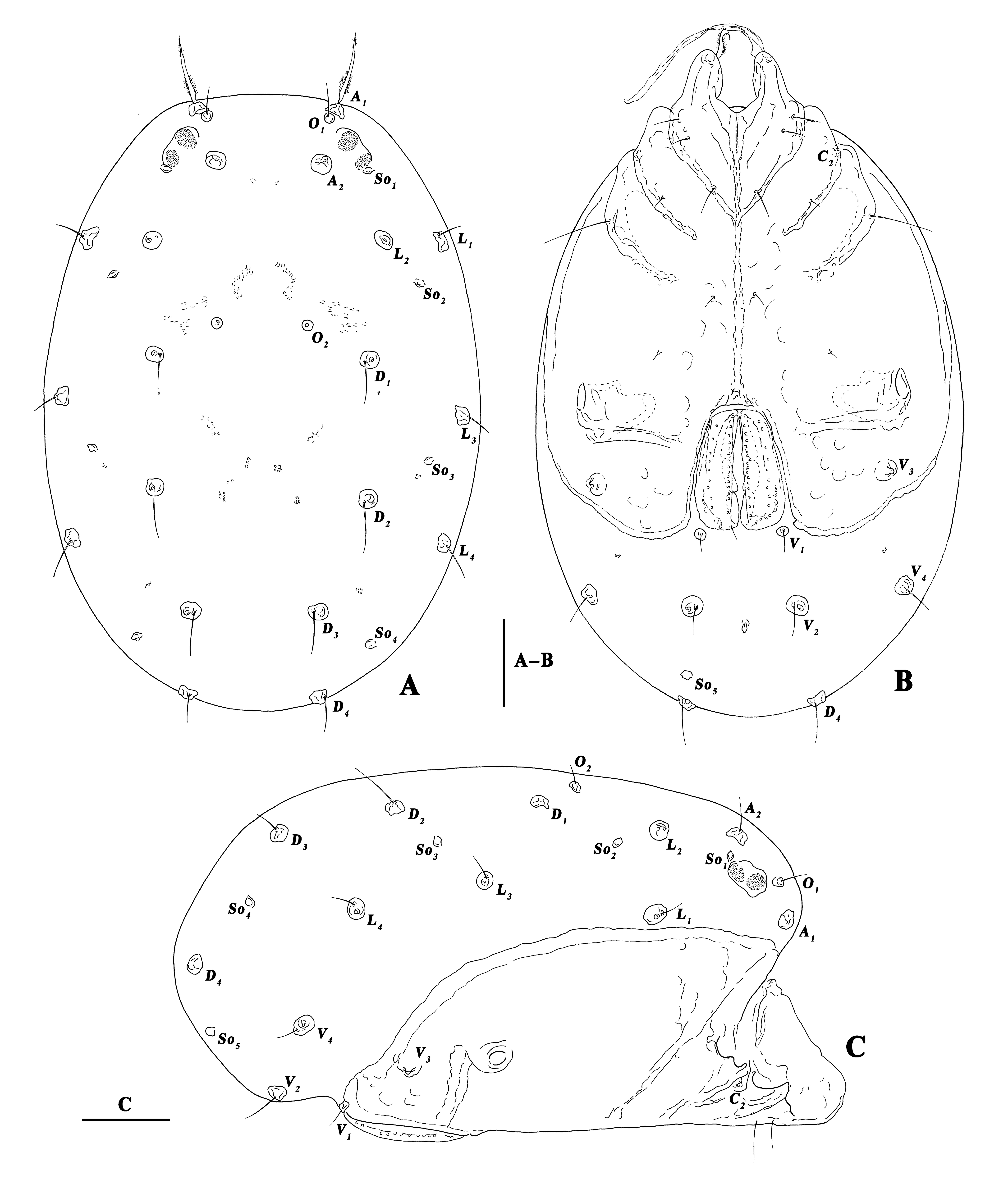
The terminology and abbreviations used are updated from Wiles (1996) and Jin (1997): a.s.l. = above sea level, A1 = preantennal glandularia, A2 = postantennal glandularia, C2 & C4 = coxoglandularia 2 & 4, Cx-I–IV = coxae I–IV, Cx-I/Cx-I = suture between median margins of first coxae, Cx-I/Cx-II = suture line between first and second coxae, D1 –D4 = dorsoglandularia 1–4, dL = dorsal length, I-L-1–6, etc. = the first–sixth segment of the first leg, etc., L = length, L1 –L4 = lateroglandularia 1–4, MS = median suture, O1 = preocularia, O2 = postocularia, P-1–P-5 = the first–fifth segment of palp (from most proximal to most distal), So1–So5 = slit organs 1–5, V1–V4 = ventroglandularia 1–4, W = width.
The type specimens are deposited in the Institute of Entomology, Guizhou University, Guiyang, P. R. China (GUGC).
Systematics
Family Acucapitidae Wiles, 1996
Genus Acucapito Wiles, 1996
Acucapito hainanensis Li & Guo sp. nov.
ZOOBANK: C8F2D52C-A622-4F56-9927-C54B505F5316 ![]()





















Habitat
Turbulent mountain streams with organic detritus and cobblestones as substrate.
Type series
Holotype: adult male, Bawangling National Nature Reserve, Hainan Province, P. R. China (19°05′49″ N, 109°06′40″ E, 240 m a.s.l.), collected by Xin-Yao Gu & Zhu-Hui Ding, 12-I-2018, slide No. HN-AC-2018011201. Allotype: adult female, Bawangling National Nature Reserve, Hainan Province, P. R. China (19°08′10″ N, 109°70′52″ E, 374 m a.s.l.), collected by Xin-Yao Gu & Zhu-Hui Ding, 09-I-2018, slide No. HN-AC-2018010901. Paratypes: two adult males, same data as holotype, slides No. HN-AC-2018011202–2018011203; two adult males and one female, same data as allotype, slides No. HN-AC-2018010902–2018010904.
Etymology
''hainan-'' is derived from the name of the Hainan Province, P. R. China, where the specimen was collected
Diagnosis
Both sexes — In dorsal view: O2 closer to D1 than to L2 ; D1 and D2 arranged a rectangle; P-3 very long and slender, dorsum with two fine setae, the sunken area of ventral side of P-3 more than half the total length and with two plumose setae at the terminal; P-4 with a minute stubby seta at the middle of dorsal side, a very fine seta at the distal end and a ventrolateral seta on the inner side. The numbers of swimming setae on legs: II-Leg-4, 2; II-Leg-5, 4–6; III-Leg-4, 5–6; III-Leg-5, 9–10; IV-Leg-4, 4–5; IV-Leg-5, 7–8; IV-Leg thicker than other legs.
Male — Genital flaps with 17–19 median setae and 9–10 lateral ones.
Female — V3 located near posterior margins of Cx-IV; genital flaps with 15–16 median setae, without lateral setae.
Description
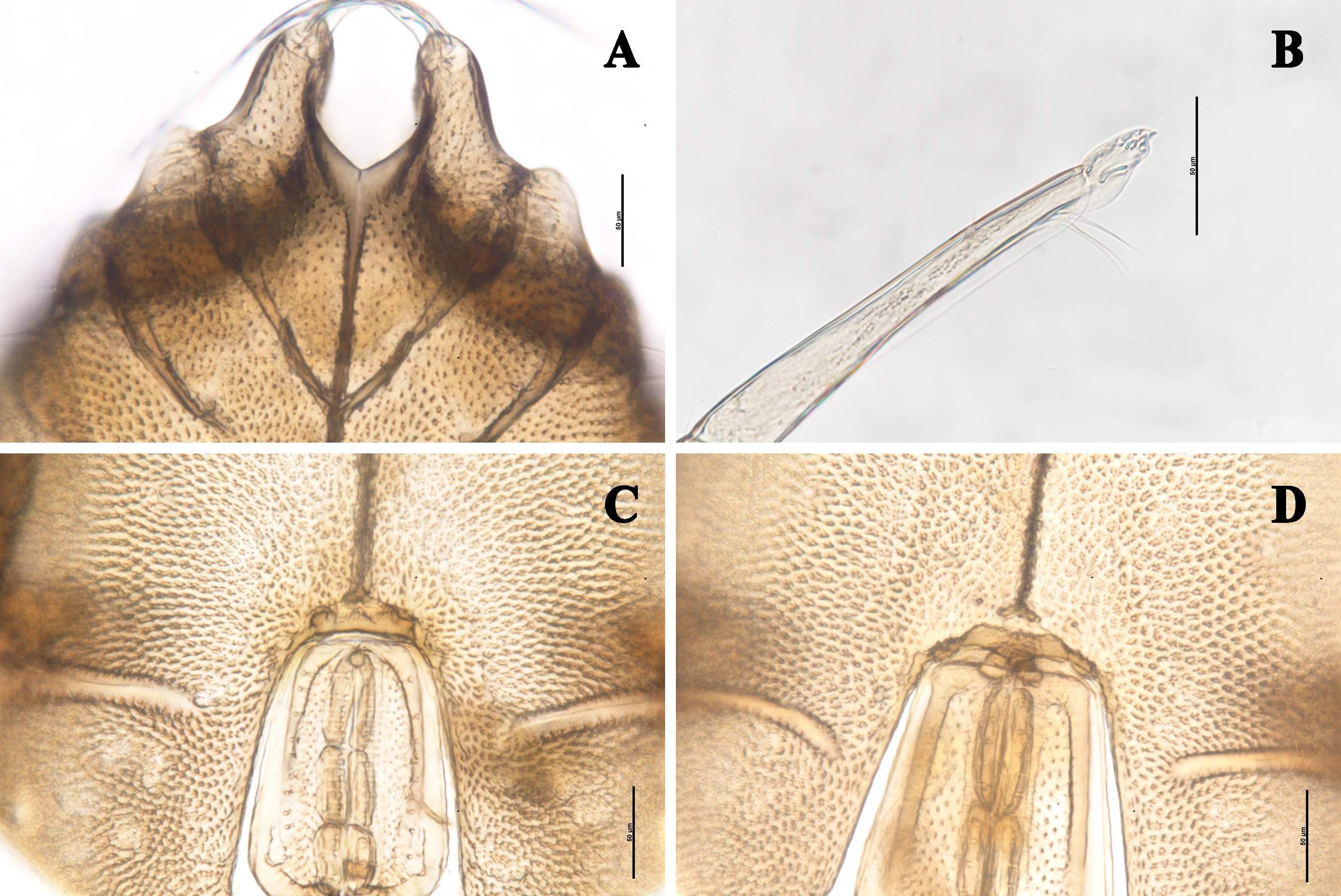
Male — (n=5). Idiosoma oval. In dorsal view: integument soft, without sclerotized plates, nine places of muscle attachments clearly visible; lateral eyes in weakly developed capsules; A1 setae relatively long, thick and plumose; A2 and the second pair of eye lenses at the same level; O2 closer to D1 than to L2 ; D1 and D2 arranged a rectangle (Figure 1A). In ventral view: a well-developed ventral shield formed by the fused coxae, Cx-I–III narrow, Cx-IV wide and partially enclosing the genital field; under light microscope, Cx-I/Cx-I fused to the MS, the Y-shaped interspace between Cx-I/Cx-I smooth and different from the punctate ornamentation of Cx-I, Cx-I/Cx-II complete and fused to the MS (Figure 2A), other coxal sutures incomplete (Figure 1B), but under SEM, fusion of Cx-I/Cx-I and MS invisible, Cx-I/Cx-II incomplete (Figure 3A); a pair of long membranous extensions at the tip of Cx-I and a pair of plumose setae under them (Figure 3B–C); C2 located laterally between Cx-II/Cx-III (Figure 1B); V1 near the posterior margins of genital field, V3 located between IV-leg sockets and posterior margins of Cx-IV (Figures 1B, 3D); excretory pore slightly distal to V2 ; one place of muscle attachment well visible between V4 and the posterior margin of Cx-IV (Figure 1B). In lateral view: So1 dorsal to the second pair of eye lenses, So2 between O2 and L1 , So3 between D2 and L3 , So4 between D3 and D4 , So5 located ventrally; L2 directly above L1 , L2 –L4 in a straight line (Figure 1C).
Genital field with three pairs of acetabula under genital flaps (Figure 2C); acetabula porous (Figure 5D); pregenital sclerite fused to ventral shield, suture visible under light microscope (Figure 2C), but invisible under SEM (Figure 5C); genital flaps with 17–19 median setae and 9–10 lateral ones at each side, the interspace between two rows of setae porous under light microscope (Figures 4D, 5C).
Gnathosoma with exceptionally long, narrow rostrum; a pair of flabellate, sclerotized extensions on the dorsum of basal rostrum (Figures 4A, 5A). Chelicera long and narrow; chela with two rows of teeth (Figure 5B).
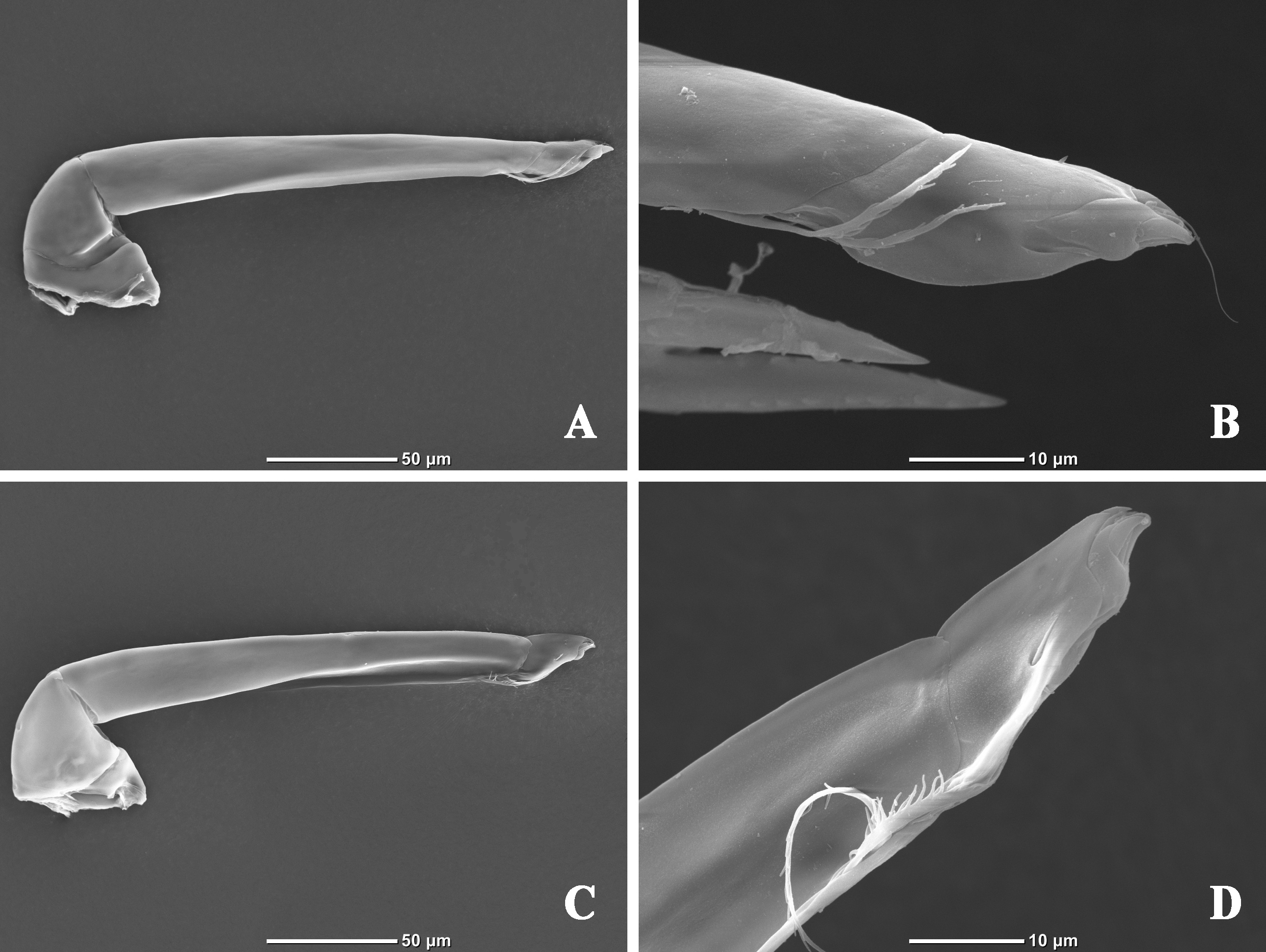
Palp five-segmented (Figures 4B, 6A, 6C); P-1 short, with a dorsal fine seta; P-2 with two dorsal fine setae; P-3 very long and slender, dorsum with two fine setae, the sunken area of venter more than half the total length and with two plumose setae at the terminal; P-4 with a minute, stubby seta at the middle of dorsum, a fine seta at the terminal and a ventrolateral seta on the inner side (Figure 6B, D); P-5 greatly reduced, the joint visible on the outer side (Figure 6B), only the front end visible on the inner side (Figure 6D).
Legs robust and swimming setae only on the inner side; I-Leg without swimming setae (Figure 7A–B); the numbers of swimming setae on other legs: II-Leg-4, 2; II-Leg-5, 4–6 (Figure 7C–D); III-Leg-4, 5–6; III-Leg-5, 9–10 (Figure 8A–B); IV-Leg-4, 4–5; IV-Leg-5, 7–8; IV-Leg thicker than other legs (Figure 8C–D).
Female — (n=2). Similar to the male (Figure 9); V3 located near posterior margins of Cx-IV (Figure 9B); genital flaps with 15–16 median setae, without lateral setae, porous area also existed under light microscope (Figures 2D, 4C).



Measurements
Male — (n=5). Idiosoma L 717 (717–748), W 521 (521–545); MS L (from the gnathosoma bay to the genital plates) 353 (347–353); genital plates L 148 (148–150), W 108 (108–111); gnathosoma L 348 (342–348), rostrum L 149 (145–149), flabellate sclerotized extensions L 94 (89–94); chelicera L 374 (374–377); Palp dL: P-1 9 (8–9), P-2 43 (40–43), P-3 167 (167–171), P-4 28 (24–28), P-5 24 (24–25), ventral sunken area of P-3 L 106 (105–106), distal plumose seta of P-3 L 29 (29–30), second plumose seta of P-3 L 31 (31–33); Leg segments dL: I-L-1 54 (53–54), I-L-2 88 (86–88), I-L-3 65 (65–67), I-L-4 95 (93–95), I-L-5 103 (103–104), I-L-6 98 (96–98); II-L-1 63 (63–67), II-L-2 95 (89–95), II-L-3 64 (63–65), II-L-4 87 (85–87), II-L-5 104 (104–108), II-L-6 112 (110–112); III-L-1 68 (61–68), III-L-2 109 (106–109), III-L-3 81 (80–81), III-L-4 110 (106–110), III-L-5 134 (134–140), III-L-6 130 (130–132); IV-L-2 97 (97–100), IV-L-3 129 (126–129), IV-L-4 159 (159–165), IV-L-5 162 (162–172), IV-L-6 142 (142–153).
Female — (n=2). Idiosoma L 769 (653–769), W 569 (482–569); MS L (from the gnathosoma bay to the genital plates) 376 (365–376); genital plates L 203 (201–203), W 143 (137–143); gnathosoma L 387 (387–398), rostrum L 162 (162–171), flabellate sclerotized extensions L 115 (111–115); chelicera L 422 (422–438); Palp dL: P-1 12 (12–13), P-2 50 (50–53), P-3 188 (188–192), P-4 30 (30–32), P-5 31 (29–31), ventral sunken area of P-3 L 104 (104–115), distal plumose seta of P-3 L 28 (28–30), second plumose seta of P-3 L 33 (33–34); Leg segments dL: I-L-1 70 (65–70), I-L-2 98 (86–98), I-L-3 72 (71–72), I-L-4 105 (105–106), I-L-5 111 (111–113), I-L-6 105 (101–105); II-L-1 79 (68–79), II-L-2 95 (94–95), II-L-3 65 (63–65), II-L-4 110 (95–110), II-L-5 113 (113–116), II-L-6 117 (117–127); III-L-1 74 (74–77), III-L-2 116 (109–116), III-L-3 88 (77–88), III-L-4 129 (124–129), III-L-5 155 (150–155), III-L-6 137 (137–146); IV-L-2 111 (109–111), IV-L-3 138 (134–138), IV-L-4 183 (175–183), IV-L-5 188 (188–190), IV-L-6 167 (167–171).
Differential diagnosis
There are six differences between Acucapito hainanensis Li & Guo sp. nov. and the type species A. naso Wiles, 1996: (1) through measurement comparison of both sexes, all structures of A. hainanensis Li & Guo sp. nov. are smaller than A. naso; (2) in both sexes, O2 of A. hainanensis Li & Guo sp. nov. is near D1 , while O2 of A. naso is at the middle between L2 and D1 ; (3) in both sexes, D1 and D2 are composed a rectangle in A. hainanensis Li & Guo sp. nov., but a trapezoid in A. naso; (4) in female, V3 of A. hainanensis Li & Guo sp. nov. is at the margins of Cx-IV, while it is further from the margins of Cx-IV in A. naso; (5) as for palps of both sexes, the sunken area of P-3 at the ventral side is more than half the total length in A. hainanensis Li & Guo sp. nov., but less than half the total length in A. naso; (6) as for legs, the swimming seta counts (both sexes are same) of II-L-4, III-L-4, III-L-5, IV-L-4 and IV-L-5 are 2, 4–6, 9–10, 4–5 and 7–8 in A. hainanensis Li & Guo sp. nov., while in A. naso, II-L-4 and III-L-4 have 3 and 8 which are same in both sexes, III-L-5 of male has 12, IV-L-4 and IV-L-5 of female have 8 and 11 (Wiles 1996).
With regard to differences between Acucapito hainanensis Li & Guo sp. nov. and the remaining described species A. vietnamensis Tuzovskij, 2009, although less information of glandular position could be obtained from original description and illustration due to the bad condition of the specimen, five differences are still found obviously between these two species: (1) in both sexes, seta of A1 is plumose in A. hainanensis Li & Guo sp. nov., but smooth in A. vietnamensis; (2) in dorsal view of both sexes, So4 of A. hainanensis Li & Guo sp. nov. is behind D3 , but So4 and D3 are at the same level in A. vietnamensis; (3) in both sexes, a pair of membranous extensions and a pair of plumose setae are existed at the tip of Cx-I in A. hainanensis Li & Guo sp. nov., but only a pair of short plumose setae at the tip of Cx-I in A. vietnamensis; (4) in males, genital flaps have 17–19 median setae and 9–10 lateral ones at each side in A. hainanensis Li & Guo sp. nov., while they have 14–15 median setae and 11–12 lateral ones in A. vietnamensis; (5) as for palp of both sexes, P-3 has a sunken area at the ventral side, P-4 only has one distal seta and P-5 is extended out of P-4 in A. hainanensis Li & Guo sp. nov., while P-3 is flat, P-4 has three distal setae and P-5 is extremely reduced in A. vietnamensis (Tuzovskij 2009).
In addition to the geographical separation between localities of the previously described species (Borneo, Vietnam), the above morphological differences demonstrate that the specimen we collected from Hainan Province, P. R. China represents the third acucapitid mite, Acucapito hainanensis Li & Guo sp. nov. Molecular data (e.g., the barcoding region of COI) would provide valuable additional information about the distinctiveness of these three species and their placement within the Lebertioidea.
Acknowledgements
We are indebted to Dr. Harry Smit (Naturalis Biodiversity Center, Leiden) for providing literature. This research was supported by National Natural Science Foundation of China (31772421, 31750002).
References
- Gu X.Y., Li H.T., Jin D.C., Guo J.J. 2021. Collection and Preservation of Water Mites and Preparation of Slide Specimens. Bio-101 e1010676. [in Chinese]
- Jin D.C. 1997. Hydrachnellae-morphology systematics a primary study of Chinese fauna. Guiyang: Guizhou Science and Technology Publishing House. pp. 356. [in Chinese]
- Smit H. 2020. Water mites of the world, with keys to the families, subfamilies, genera and subgenera (Acari: Hydrachnidia). Leiden: Monogr. Nederl. Entomol. Ver. pp. 765.
- Tuzovskij P.V. 2009. A new water mite species of the genus Acucapito (Acariformes: Acucapitidae) from Vietnam. Zoosyst. Ross., 18(2): 212-217. https://doi.org/10.31610/zsr/2009.18.2.212
- Wiles P.R. 1996. A new family, genus and species of watermite (Acari: Hydrachnidia, Lebertioidea) from Brunei. Quekett J. Microsc., 37: 692-695.



2022-01-06
Date accepted:
2022-02-07
Date published:
2022-02-10
Edited by:
Mąkol, Joanna

This work is licensed under a Creative Commons Attribution 4.0 International License
2022 Li, Hai-Tao; Jin, Dao-Chao and Guo, Jian-Jun
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



