Complementary description of Kuzinellus niloticus (El-Badry) (Acari, Mesostigmata) from Egypt
Abo-Shnaf, Reham  1
and Attia, Sahar Ali2
1
and Attia, Sahar Ali2
1✉ Vegetable and Aromatic Plant Mites Department, Plant Protection Research Institute, Agricultural Research Centre, 12611 Dokii, Giza, Egypt.
2Scale Insects and Mealy Bugs Department, Plant Protection Research Institute, Agricultural Research Centre, 12611 Dokii, Giza, Egypt.
2022 - Volume: 62 Issue: 1 pages: 143-147
https://doi.org/10.24349/rqg5-jj0iZooBank LSID: 07C5E8C0-9B4D-4E8B-AB0B-FA658D5F1445
Original research
Keywords
Abstract
Introduction
The predatory mite family Phytoseiidae is one of the most diverse mite families distributed world-wide with nearly 2,800 described species in 94 genera and three sub-families (Chant and McMurtry 2007; Demite et al. 2021). They mainly prey on spider mites but are also important predators of other mites, small arthropods and nematodes (McMurtry et al. 2013, 2015). Some species are commercially produced for controlling spider mites, thrips and whiteflies. Because many phytoseiids are specialized predators of phytophagous mites they are important agents for biological control.
The genus Kuzinellus Wainstein (Typhlodrominae) is known from all continents except Antarctica. Kuzinellus includes 53 nominal species so far (Demite et al. 2021). Most of the species have been reported from Africa while others have been collected in southern Russia, India, Pakistan, China, Japan, and the US and recently from Saudi Arabia (Alatawi et al. 2017; Kamran et al. 2017). Up to now, only one species of this genus has been reported from Egypt (El-Badry 1970).
Chant and McMurtry (1994) provided a detailed diagnosis of Kuzinellus and separated 27 nominal species in two species groups based on the morphology of most dorsal setae. Moraes et al. (2008) provided description, re-description, and illustrations of 22 species of Kuzinellus reported from sub-Saharan Africa along with a diagnostic key of these species, not considering the species groups of this genus made by Chant and McMurtry (1994). Later, two species were added in this genus by El-Banhawy and Knapp (2011).
The genus Kuzinellus can be distinguished by the absence of seta Z1 and the presence of setae z6 and JV2; fixed digit of chelicera with 2–4 teeth and seta z6 inserted halfway between setae j6 and s6 (Wainstein 1976; Chant and McMurtry 1994). In seven species in this genus setae R1 are placed on the dorsal shield while in the remaining species are located on the unsclerotized cuticle (Chant and McMurtry 1994; Moraes et al. 2008). Chant and Yoshida-Shaul (1986) and Chant and McMurtry (1994) clarified the identity of this seta as R1 in those species.
In this paper, we re-describe Kuzinellus niloticus (El-Badry) based on new specimens collected on citrus orchards at Sohage governorate, Egypt. To date, no other species of the genus has been registered for Egypt.
Material and Methods
Mite specimens were collected from leaves of mandarin orange orchards, Citrus reticulata Blanco (Rutaceae) at Sohag governorate, Egypt. The samples were kept in polyethylene bags and transferred to the laboratory for later extraction by modified Tullgren funnels (Krantz and Walter 2009). The mites were mounted on microscopic slides in Hoyer's medium and examined under a phase contrast (Olympus, BHA) research microscope. Taxonomically important structures were illustrated with the aid of an eyepiece attached to the phase contrast microscope, measured with a graded eyepiece. Measurements are given in micrometers. Setal nomenclature follows that of Rowell et al. (1978) and Chant and Yoshida-Shaul (1991) for dorsal and ventral surfaces of the idiosoma, respectively. The idiosomal setal pattern follows Chant and Yoshida-Shaul (1992). The notation for solenostomes and poroids is based on Athias-Henriot (1975). Coordinates provided are approximate, as these were not taken at the time the samples were collected.
Systematics
Kuzinellus niloticus (El-Badry)
Typhlodromus niloticus El-Badry, 1967: 464, 1970: 500; Chant & Yoshida-Shaul 1986: 464.
Paraseiulus (Hemiseiulus) niloticus, Wainstein 1976: 700.
Seiulus niloticus, Moraes et al. 1986: 231.
Kuzinellus niloticus, Moraes et al. 2004: 273, 2008: 18; Chant & McMurtry 2007: 143; Abo-Shnaf & Moraes 2014: 38.
Previous records from Egypt
Cairo governorate (El-Badry 1970).
Redescription
Female (Two specimens measured, Figs. 1 a–f)
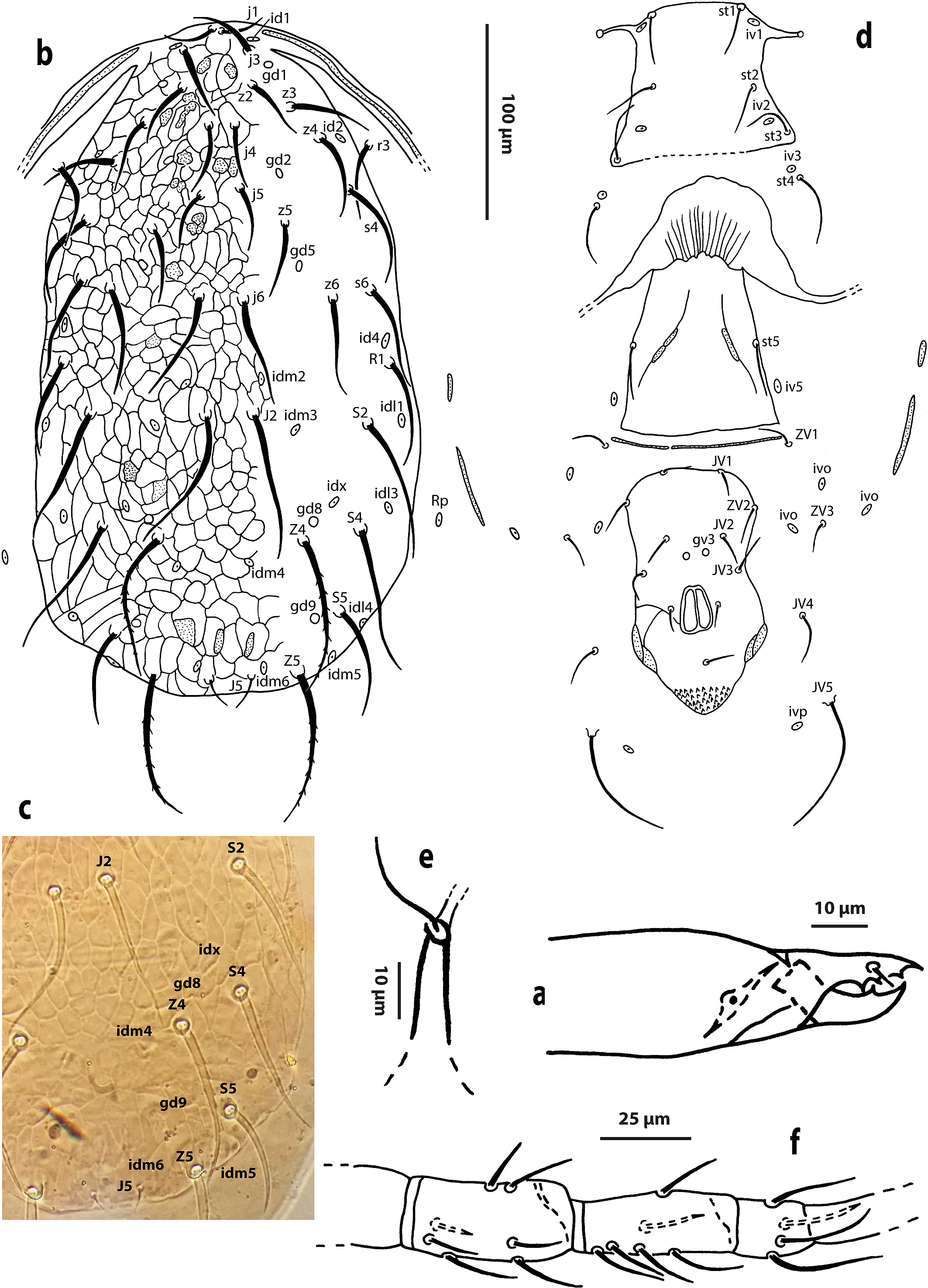


Gnathosoma — Corniculi parallel to each other; 18, 17 long, basal width of corniculus 5, 3; distance between tips of corniculi 7, 4. Fixed cheliceral digit 22, 21 long, with three teeth; movable digit 22, 20 long, with one tooth; dorsal and antiaxial lyrifissures are visible (Fig. 1a).
Dorsum (Figs. 1b and 1c) — Dorsal shield strongly reticulate; 339, 325 long and 166, 163 wide at s4 level; with 20 pairs of tuberculate setae, twelve pairs of poroids (id1, id2, id4, idx, idl1, idl3, idl4, idm2, idm3, idm4, idm5 and idm6) and five pairs of solenostomes (gd1, gd2, gd5, gd8 and gd9). Length of dorsal setae: j1 25; j3 36, 34; j4 31, 29; j5 29; j6 55, 52; J2 68, 62; J5 13; z2 31, 30; z3 39, 36; z4 39; z5 36, 35; z6 49, 46; Z4 73, 69; Z5 70, 69; s4 47, 43; s6 48, 47; S2 64, 62; S4 68, 66; S5 48, 47; r3 39, 31; R1 47, 44. All setae smooth, except Z4 and Z5, which are serrated. Peritreme extending forward to level of j3.
Venter (Fig. 1d) — Anterior region to st1 smooth; sternal shield smooth; with anterolateral corners fused with endopodal plates, with a pair of pores (gst1) on distal end of extension between coxae I-II; posterior margin indistinguishable; with three pairs of setae (st1-st3) and two pairs of poroids (iv1 and iv2). Distances between st1-st1 42, 31; st2-st2 53, 51; st3-st3 83, 73; st4-st4 100, 82; st1-st3 73, 65. Setae st3, st4 and poroid iv3 of each side on unsclerotized cuticle. Genital shield smooth with dense muscle scars and with lateral extensions; distance between st5-st5 60, 57. Ventrianal shield vase-shaped, smooth with a few striae lateral to the anal opening; 114, 112 long; 66, 62 wide at ZV2 level and 65, 64 wide at level of anus; with four pairs of pre-anal setae and a pair of pre-anal solenostomes (gv3) posteroparaxial to setae JV2. Seta JV5 tuberculate, 57, 53. Ventral setae smooth. With two pairs of filiform metapodal plates.
Spermatheca (Fig. 1e) — Calyx of spermatheca tubular, flaring distally, 13, 10 long; atrium distinct.
Legs (Fig. 1f) — Leg IV with one sharp-tipped macroseta on basitarsus IV: St IV 30, 29; chaetotaxy of genu II: 2, 2/0, 2/1, 1; genu III: 1, 2/1, 2/0, 1.
Specimens examined
Two females from mandarin orange orchards, Citrus reticulata Blanco (Rutaceae), collected by the junior author of this paper at Sohag governorate (26°44′38″ N, 32°15′9″ E), Egypt, December 04, 2018. Slides are deposited in the mite reference collection of the Egyptian Society of Acarology Museum (ESAM), Zoology and Agricultural Nematology Department, the Faculty of Agriculture, Cairo University, Giza governorate, Egypt.
Remarks
This species was originally described from the holotype female and one paratype female collected from Khartoum, Sudan. The original description was reasonably detailed, with illustrations and setal measurements.
Measurements of the females examined fit those in the original description and the re-description given by Chant and Yoshida-Shaul (1986), except for the longer setae J2, z3, Z4, Z5, s4, S4 and S5, which are about 1.1–1.3 in comparison to those reported in the two above mentioned publications and the shorter j5 and s6 setae (38 and 60 long according to those authors).
The dorsal shield bears two pairs of solenostomes (posterolaterad j4 and anteriad Z4) according to Chant and Yoshida-Shaul (1986) (five in the examined females in the current work).
Peritreme reaching to the level of j1 according to the original description, but to j3 in the examined females in the current work.
Fixed cheliceral digit with five teeth in the original description and the re-description given by Chant and Yoshida-Shaul (1986). We found three teeth in the examined females in the current work.
Shorter macroseta on St IV (23) according to the original description, instead to 30 and 29 in the examined females in the current work.
Biological investigations under laboratory condition to confirm the ability of feeding of Kuzinellus niloticus on some citrus pests would be desirable in order to use the phytoseiid predators in Integrated Pest Management approaches, thus reducing pesticide usage by growers.
The importance of systematics in biological control programs is well known. Mite pests and their associated predators must be correctly identified before adequate control measures can be considered.
References
- Abo-Shnaf R.I.A., Moraes G.J.de. 2014. Phytoseiid mites (Acari: Phytoseiidae) from Egypt, with new records, descriptions of new species, and a key to species. Zootaxa, 3865 (1): 1-71. https://doi.org/10.11646/zootaxa.3865.1.1
- Alatawi F.J., Basahih J., Kamran M. 2017. The superfamily Phytoseioidea (Acari: Mesostigmata) from Saudi Arabia: a new species, new records and a key to the reported species. Acarologia, 57 (2): 275-294. https://doi.org/10.1051/acarologia/20164154
- Athias-Henriot C. 1975. Nouvelles notes sur les Amblyseiini. II - Le relevé organotaxique de la face dorsale adulte (Gamasides protoadéniques, Phytoseiidae). Acarologia, 17 (1): 20-29.
- Chant D.A., McMurtry J.A. 1994. A review of the subfamilies Phytoseiinae and Typhlodrominae (Acari: Phytoseiidae). Int. J. Acarol., 20 (4): 223-310. https://doi.org/10.1080/01647959408684022
- Chant D.A., McMurtry J.A. 2007. Illustrated keys and diagnoses for the genera and subgenera of the Phytoseiidae of the world (Acari: Mesostigmata). Ind. Pub. House, West Bloomfield, USA. 220 pp.
- Chant D.A., Yoshida-Shaul E. 1986. A world review of the ecclesiasticus species group in the genus Typhlodromus Scheuten (Acarina: Phytoseiidae). Can. J. Zool., 64 (2): 447-466. https://doi.org/10.1139/z86-069
- Chant D.A., Yoshida-Shaul E. 1991. Adult ventral setal patterns in the family Phytoseiidae (Acari: Gamasina). Int. J. Acarol., 17 (3): 187-199. https://doi.org/10.1080/01647959108683906
- Chant D.A., Yoshida-Shaul E. 1992. Adult idiosomal setal patterns in the family Phytoseiidae (Acari: Gamasina). Int. J. Acarol., 18 (3): 177-193. https://doi.org/10.1080/01647959208683949
- Demite P.R., Moraes G.J.de, McMurtry J.A., Denmark H.A., Castilho R.C. 2021. Phytoseiidae Database. Available from: www.lea.esalq.usp.br/phytoseiidae (Accessed September 09, 2021).
- El-Badry E.A. 1967. New species of the genus Typhlodromus from Sudan (Acari: Phytoseiidae). J. Zool., 153: 463-474. https://doi.org/10.1111/j.1469-7998.1967.tb04978.x
- El-Badry E.A. 1970. Taxonomic review of the phytoseiid mites of Egypt (Acarina: Phytoseiidae). Bull. Soc. Entomol. Egypt., 54: 495-510.
- El-Banhawy E.M., Knapp M. 2011. Mites of the family Phytoseiidae Berlese from Kenya (Acari: Mesostigmata). Zootaxa, 2945: 1-176. https://doi.org/10.11646/zootaxa.2945.1.1
- Kamran M., Basahih J.S., Alatawi F.J. 2017. A new species of Kuzinellus Wainstein, 1976 (Acari: Mesostigmata: Phytoseiidae) from Saudi Arabia, with a key to the world species. Int. J. Acarol., 43 (7): 545-551. https://doi.org/10.1080/01647954.2017.1360937
- Krantz G.W., Walter D.E. 2009. A manual of acarology. 3rd Ed. Texas Tech Univ. Press, Lubbock, Texas, USA. 807 pp.
- McMurtry J.A., Famah-Sourassou N., Demite P.R. 2015. The Phytoseiidae (Acari: Mesostigmata) as biological control agents. In: Carrilo D., Moraes G.J.de, Peña J.E. (Eds). Prospects for biological control of plant feeding mites and other harmful organisms. Cham (CH): Springer Int. Pub. p. 133-149. https://doi.org/10.1007/978-3-319-15042-0_5
- McMurtry J.A., Moraes G.J.de, Famah-Sourassou N. 2013. Revision of the lifestyles of phytoseiid mites (Acari: Phytoseiidae) and implications for biological control strategies. Sys. Appl. Acarol., 18 (4): 297-320. https://doi.org/10.11158/saa.18.4.1
- Moraes G.J.de, McMurtry J.A., Denmark H.A. 1986. A catalog of the mite family Phytoseiidae. References to taxonomy, synonymy, distribution and habitat. EMBRAPA - DDT, Brasilia, Brazil. 353 pp.
- Moraes G.J.de, McMurtry J.A., Denmark H.A., Campos C.B. 2004. A revised catalog of the mite family Phytoseiidae. Zootaxa, 434: 1-494. https://doi.org/10.11646/zootaxa.434.1.1
- Moraes G.J.de, Zannou I.D., Ueckermann E.A., Oliveira A.R., Hanna R., Yaninek J.S. 2008. Phytoseiid mites of the tribe Paraseiulini Wainstein (Acari: Phytoseiidae) from sub-Saharan Africa. Zootaxa, 1687: 1-34. https://doi.org/10.11646/zootaxa.1687.1.1
- Rowell H.J., Chant D.A., Hansell R.I.C. 1978. The determination of setal homologies and setal patterns on the dorsal shield in the family Phytoseiidae (Acarina: Mesostigmata). The Can. Entomol., 110 (8): 859-876. https://doi.org/10.4039/Ent110859-8
- Wainstein B.A. 1976. A new tribe of the family Phytoseiidae (Parasitiformes). Zool. Zh., 55: 696-700. [in Russian].



2021-09-18
Date accepted:
2021-12-18
Date published:
2022-02-01
Edited by:
Kreiter, Serge

This work is licensed under a Creative Commons Attribution 4.0 International License
2022 Abo-Shnaf, Reham and Attia, Sahar Ali
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



