Biodiversity of mites in mango orchards (Mangifera indica L.) and evaluation of some mineral and essential oils against Cisaberoptus kenyae Keifer (Acari: Eriophyidae) management
Abo-Shnaf, Reham  1
; Allam, Sally F.M.
1
; Allam, Sally F.M.  2
; El-Sobky, Menna L.3
; Abdul-Shafc, Ahmed F.4
and El-Tony, Aml G.5
2
; El-Sobky, Menna L.3
; Abdul-Shafc, Ahmed F.4
and El-Tony, Aml G.5
1✉ Vegetable and Aromatic Plant Mites Department, Plant Protection Research Institute, Agricultural Research Centre, 12611 Dokii, Giza, Egypt.
2Zoology and Agricultural Nematology Department, Faculty of Agriculture, Cairo University, Giza, Egypt.
3Undergraduate student, Faculty of Agriculture, Cairo University, Giza, Egypt.
4Undergraduate student, Faculty of Agriculture, Cairo University, Giza, Egypt.
5Undergraduate student, Faculty of Agriculture, Cairo University, Giza, Egypt.
2022 - Volume: 62 Issue: 1 pages: 130-142
https://doi.org/10.24349/7izc-dm2nOriginal research
Keywords
Abstract
Introduction
Mango (Mangifera indica L., Anacardiaceae) is one of the most consumed fresh fruits in the world, with production occurring in more than 100 countries including Egypt. There are over 1000 different cultivars of mango, giving rise to various shapes, sizes, colours, textures and nutritional properties (Siddiq 2017). Egypt accounted for more than half of the total production of tropical fruits in 2012 (Siddiq 2017). Mango is currently grown throughout the Nile Valley, the Nile Delta, and Fayoum governorate (Saad et al. 2007). It covers about 135,524 ha with a total production of 1,473,538 tons in 2019 (FAO 2019) (this number includes guava and mangosteen fruits). Mango is one of the most important export fruits in Egypt, with a total exportation valued at 34.6 million dollars in 2018 (73% fresh mango and 27% mango juice) (Gabr 2019).
Mango is attacked by different insect and mite pests causing serious economic lossess. Among them, four mite species are the most common pests on mango: broad mite, Polyphagotarsonemus latus (Banks); spider mite, Oligonychus mangiferus (Rhaman & Sapra); lychee erinose mite, Aceria litchii (Keifer) and mango bud mite, Aceria mangiferae Sayed. These mite pests prefer the upper surface of leaves causing chlorotic stippling on the leaves (Sarwar 2015). The infested leaves often become reddish or bronze in colour, may drop and yield can be greatly reduced. Damage caused by these mites to the leaves provides entry sites for fungi resulting in nutritional imbalances (Sarwar 2015). They cause huge economic losses which necessitates the search for effective, safe and eco-friendly control measures. The leaf coating mite, Cisaberoptus kenyae Keifer can be found under the epidermis of young leaves which resulted in a silvery appearance of the leaves. Leaf damage is concentrated at the base of the petiole and along the central veins. Narrow patches bordering the leaf margins and browning spots especially on ''Hindi'' and ''Alphonso'' cultivars can also occur (Abou-Awad et al. 2009).
Plant essential oils could be a potential alternative for mite control, as some are selective and biodegradable with low effects on non-target organisms and the environment, but effective on the mite pests (Isman and Machial 2006). Extracted essential oils apparently release fumes that can control the mites, as alternatives for pesticides.
The current study presents the biodiversity of different predatory, phytophagous, miscellaneous and soil mites and insects in the Giza governorate, and are evaluated for the efficacy of some essential oils to control Cisaberoptus kenya, that cause serious damage to mango orchards, economic importance and eco-friendliness.
Material and Methods
Biodiversity
Mango orchards comprising of five cultivars (''Hindi'', ''Zebda'', ''Alphonso'', ''Ewais'' and ''Sedika'') were sampled fortnightly at Abo Ruwaysh (29°40'07'' N, 31°13'28'' E), Baydaf, Al Ayat in the Giza governorate, Egypt for a full year from February 2020 to January 2021. Samples were taken from leaves, raceme, buds, debris and soil. A 5-acre block of mango trees was set apart for this study. Randomly 30 trees were selected but concentrating on trees showing symptoms or which seem under stress. Ten leaves, ten raceme and ten buds were taken from each tree (total 300 for each plant part) on each sampling date. Leaves, raceme, and buds were placed in polyethylene bags and transferred to the laboratory for direct examinations under a stereo-microscope (Olympus SZ-ST, SZ40). The mite density on leaves, raceme and buds from each tree was recorded according to Sarwar (2015). Debris and soil samples weighing about 0.25 Kg were collected underneath mango trees, placed in polyethelyne bags and then the mites were extracted using a modified Tullgren funnel (Krantz and Walter 2009). All mite specimens were then mounted on glass slides in Hoyer's medium and examined under a phase contrast (Olympus BHA) research microscope. The eriophyid mites were mounted in modified Hoyer's medium (10 ml of fresh prepared Hoyer's medium and 0.1 g Methylene blue under heating and Magnetic stirrer for 15 minutes, then put in a glass bottle after 2 hours) (modified in the current study). Mites were identified using Zaher 1984, 1986; Summers and Price 1970; Abo-Shnaf and Moraes 2014; Mašán 2017).
To record populations of two eriophyid mites: Cisaberoptus kenyae Keifer and Aceria mangiferae Sayed, leaves and buds were collected randomly form other trees during five months at irrigular intervals to check their numbers. Twenty leaves were selected, for each leaf about 3 inches were chossen and examined according to Elhalawany (2001) with some modifications. Ten buds were checked directly under a stereo-microscope.
Management study
Mango leaves of the ''Ewais'' cultivar infested with C. kenyae were collected from Abo Ruwaysh, Baydaf, Al Ayat in the Giza governorate, Egypt. The collected mites were directly used in the study in two assessments: 1) mineral oil, orange oil (a clear, mobile liquid, with a yellow-to-orange colour, having a high terpene monoterpene hydrocarbon content, the main constituent is (+)-limonene (93–96%), obtained from ''Top Chemical'' company, Egypt), and lastomide 24% (spirodiclofen); combinations of these oils with the pesticide; 2) essential oils of Geranium robertianum L. (Geraniaceae), Salvia rosmarinus Spenn., Thymus vulgaris L. (Lamiaceae), and Syzygium aromaticum (L.) Merr. & L.M. Perry (Myrtaceae), all were obtained from ''Ayurveda'' company, Egypt. In assessment (1), fifty (± 5) individuals of C. kenyae were placed on each leaf disc (3 inches/ disc), placed on wet cotton pads in foam trays. Six replicates were used for each application in addition to the control. In assessment (2), the same trend was followed except that 80 (± 5) individuals of eriophyid mite were used in four replicates. Three concentrations (2, 1, and 0.5%) were used. Each concentration was mixed with 6 surfacta rTA – 100 and applied by spraying the mites with a manual pump onto plant leaves. Discs were examined under a stereo-microscope in the laboratory to determine the mortalities. All discs were kept in an incubator (25°C ± 5). Twenty-four hours after the application, the mortality rate of the mites was recorded daily for four days.
Statistical analysis
Anova test with Randomized Complete Block Design (RCBD) with one factor was used for analysis of all data with different applications for each species. Relationship among studied traits was done according to Steel et al. (1997). The treatment means were compared with Least Significant Difference (L.S.D.) test as given by Snedecor and Cochran (1994) using Assistat program.
The LC50, LC95, chi square (X2), fiducial limits, and slope values were estimated according to probit analysis (Finney 1971) using Ldp line software according to Bakr (2000).
Results and discussion
Biodiversity of mites associated with mango orchards in Ayat, Giza governorate was carried out during the year 2020–2021, numbers of mites vary because of sampling time and micro agriculture eco-system. Our results agree with Peña et al. (2005) who mentioned that mite densities in mango orchards in Florida vary according to season and sampling unit. Based on previous studies, about 67 identified mite species in 52 genera and 30 families were previously reported from mango orchards in Egypt (Marei et al. 2020).
Our survey in the mango orchards resulted in 17 species and 16 genera in 12 families representing Mesostigmata, Prostigmata, Astigmatina and Oribatida groups (Table 1). Insects and woodlice were also found during this study (Table 2). Our results are in parallel with Marei et al. (2020) who collected the same mite families inhabiting mango orchards at Assiut governorate, Egypt.
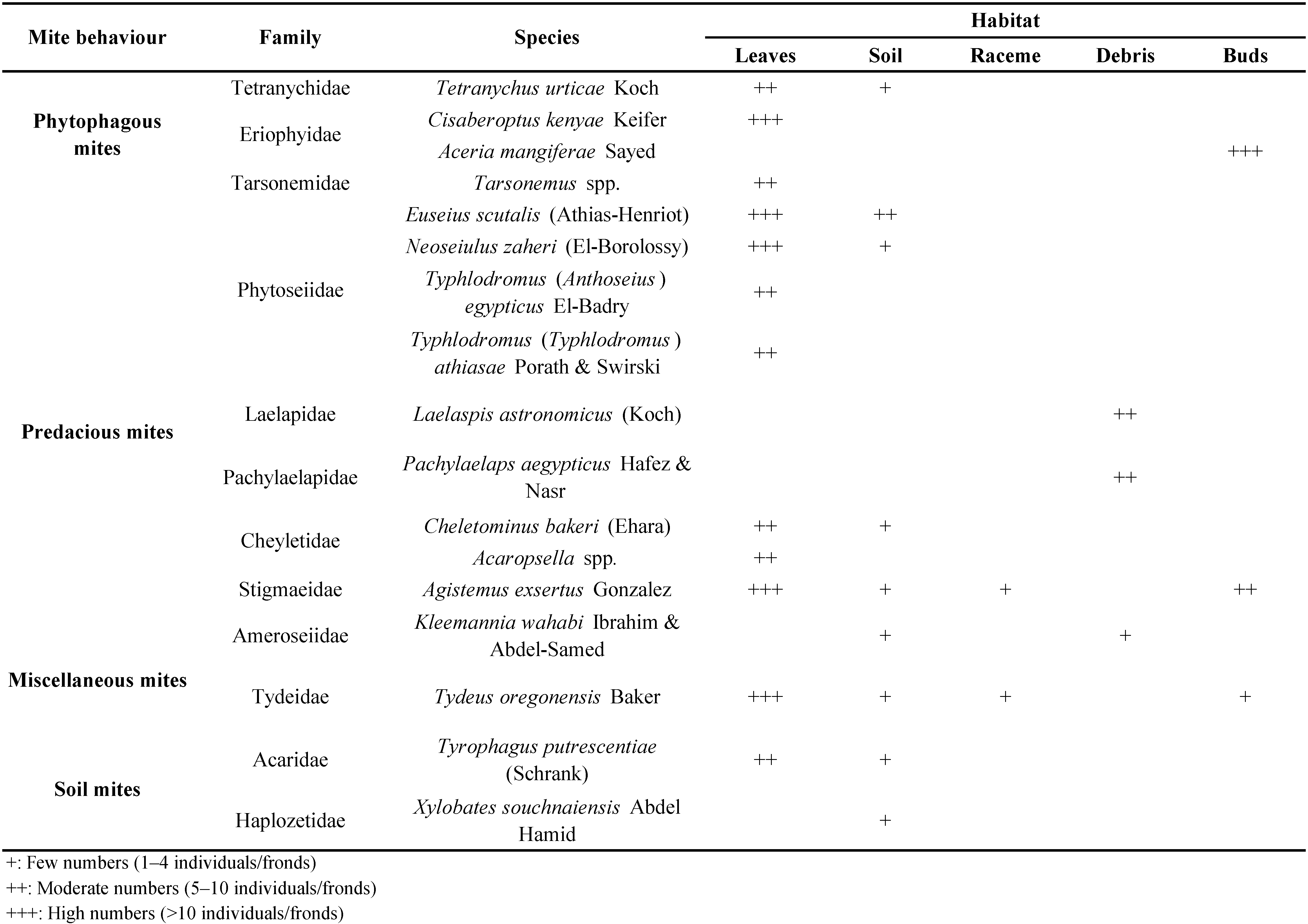





Statistical analysis confirm the significant differences in mite biodiversity (F value = 8.06, P value = 0.0000), these differences are due to biotic factors (food type) and abiotic factors (temperature, relative humidity and soil type) which reflect on agriculture eco-system in mango cultivation. These results agree with Manu (2013) who stated that development of mite communities is influenced by biotic and abiotic factors. The present results proved that numbers of ten species were homogenous at alpha (0.05) and critical value (2.048), except for Tydeus oregonensis Baker (Tydeidae) which reached 71% during the time of study (Figure 1).



Phytoseiid mites are important biological control candidates of mite pests, thrips, whiteflies, small arthropods and nematodes. They are implemented in biological control programs (Gerson et al. 2003). Phytoseiids are important natural enemies against phytophagous mites in agro-ecosystems, a total of 166 specimens were collected and identified to species level during this study, resulted in the absence of spider mites. Thus, phytoseiid mites play an important role in Integrated Pest Management programs.
McMurtry (2010) evaluated food habits of phytoseiids. Some are often found in homogeneous, while others in non-homogeneous microhabitats. Still others are commute between plant microhabitats looking for food or protection (McMurtry et al. 2013). Phytoseiids actually prefer feeding on egg and immature stages of thrips, whiteflies, scale insects and tetranychids (Muma 1971). Euseius scutalis (Athias-Henriot) is a promising control candidate for such pests (Al-Shammery 2010).
The importance of predaceous mites as biological control agents against phytophagus mites and economic important insects including the scale insect, Aspidiotus destructor Signoret (Diaspididae) and mealy bug, Drosicha mangiferae Stebbins (Margarodidae) benefits agriculture eco-systems. Our results concluded that E. scutalis feeds on egg and immature stages of A. destructor, apparently the dominant species in neighboring agro-ecosystem where it was found in high numbers. Our findings confirm that of Abou-Elella et al. (2013) that E. scutalis feeds also on eggs of scale insects. The thrips, Scirtothrips dorsalis Hood (Thripidae) was found as prey source for E. scutalis collected in this study (Chowdhury 2015). Strangly, Amblyseius swirskii Athias-Henriot was not collected during this study.
The abiotic factors in studied area affected the numbers of eriophyids and the phytoseiids in mango orchards. Abou-Awad et al. (2011) found the latter as important influences on population abundance of the eriophyids on mango, distribution and predation by Typhlodromus mangiferus Zaher & El-Borolossy (junior synonym of Typhlodromus (Anthoseius) egypticus El-Badry according to Abo-Shnaf and Moraes 2014).
Tarsonemidae, Tydeidae and Eriophyidae were found as prey for both phytoseiid mites, Typhlodromus (Typhlodromus) athiasae Porath & Swirski and Neoseiulus zaheri (El-Borolossy) collected in all our samples. This result agrees with McMurtry and Croft (1997) who reported that Typhlodromus species feed on tarsonemids, tydeids and eriophyoids. Momen (2009) found that T. (T.) athiasae females feed on nymphs of Tetranychus urticae Koch (Tetranychidae) and Eriophyes dioscoridis Soliman & Abou-Awad (Eriophyidae).
Our results showed that the predatory mite, Agistemus exsertus Gonzalez (Stigmaeidae) peaked during fall and winter seasons, preying on eriophyid mites and a scale insect, Parlatoria pergandii Comstock (Diaspididae) on mango leaves. This agrees with El-Sawi and Momen (2006) who found A. exsertus completing its life span feeding on eggs and crawlers of both scale insects, Chrysomphalus ficus Ashmead and Parlatoria blanchardi (Targioni Tozzetti) (Diaspididae).
Peña et al. (2005) repoted that Aceria mangiferae Sayed occured mostly in apical buds rather than in lateral buds. Our results showed that A. exsertus effectively controlled this mite in the lateral and apical buds. Osman and Zaki (1986) found that A. exsertus also effectively control the tomato rust mite, Aculops lycopersici (Massee).
The numbers of cheyletid mite, Cheletomimus bakeri (Ehara) increased from 5 to 23 individuals in November. The predator's increase was synchronized with the decrease in T. urticae numbers. This agrees with Ray and Hoy (2014) who found that T. urticae is a good food source for C. bakeri and Hemicheyletia wellsina (De Leon), suggesting that C. bakeri could be used in an augmentative or classical biological control programs, its use in biological control of crops or in orchards could be beneficial as well. Something that can also be followed up in the control of T. urticae in local mango orchards.
The present results showed that numbers of T. urticae was also very low because of the presence of the predatory thrips, Scolothrips longicornis Priesner (Thripidae). This result agrees with Masarovic et al. (2013) who reported S. longicornis in Slovakia is best predator for many spider mites including T. urticae.
Family Pachylaelapidae are found in many habitats including organic matter, soil and debris (Mašán 2007; Mašán and Halliday 2014), they are predators of a wide range of micro invertebrates (Lindquist et al. 2009). This may be the role they also played in the soil and debris underneath mango trees in this study.
According to Flechtmann (1985) and Mašán (1998) the ameroseiids we found in our mango orchards may also be fungivorous.
Laelaspis astronomicus (Koch) (Laelapidae) was collected from soil underneath mango trees in this study as did Zaher et al. (1986) and Rasmy et al. (1987) who collected this mite species from organic matter in Egypt.
Our results suggest that oribatids including Xylobates souchnaiensis Abdel Hamid (Haplozetidae) collected in this study may also feeds on fungi (Schneider et al. 2004).
The tydeid, T. oregonensis was collected on the mango leaves. It was previously reported by Elmoghazy (2014) from Egypt on date palm, olive and citrus trees.
Carrillo and Elanov (2004) and Uddin et al. (2019) reported the common green lacewing, Chrysoperla carnea (Stephens) (Chrysopidae) as a predator of many pests including aphids, white flies, thrips, mealy bugs, as well as eggs of different arthropods. This species was also recorded in our study.
Soil samples contained also woodlice identified as Porcellio scaber Latreille (Porcellionidae) feeding on organic matter. Hutchins (2003) and Zimmer et al. (2003) reported that P. scaber is detritivorous, saprophagous and coprophagous as they prefer decaying organic matter. Therefore, this species contributes towards a healthy soil.
Springtails or collembola are primitive, wingless micro-arthropods, closely related to insects were also collected in soil during the current study. They are supplemantary food for many predators, e.g., E. scutails. Walter and Lindquist (1989) reported that other mesostigmatid mites such as Lasioseius spp. (Blattisociidae) also feeds on springtails and other small arthropods.
Biodiversity of eriophyid mites on three cultivars of mango orchards
The abundance of eriophyid mites was noticeable only on three mango cultivars (''Hindi'', ''Alphonso'' and ''Ewais'') when temperature decreased because they prefer low temperature (Table 3).



No significant differences at 0.01 level were found between relative humidity and numbers of Cisaberoptus kenyae Keifer. In contrast, significant differences were found for A. mangiferae on ''Hindi'' and ''Alphonso'' cultivars, but in-significant on ''Ewais'' at 0.01 level (Table 3).
Statistical analysis showed that there were significant differences in temperature at 0.01 level between the two eriophyid mite species (A. mangiferae and C. kenyae) on the mango cultivars ''Ewais'', ''Hindi'' and ''Alphonso'', which resulted in the presence of the two eriophyids through the year; also minimum and maximum temperatures were significantly different at 0.01 level. This indicates that the mango cultivar ''Ewais'' affected by degrees of temperatures where the highest numbers of C. kenyae recorded (250 individuals/ 3 inches/ 20 leaves) during November when temperature reached 15°C (Tables 3 and 4).



Our results agree with Abdallah (2007) who recorded the maximum number of C. kenyae on ''Ewais'' cultivar, reaching 12000 and 14880 individuals/ 100 leaves, respectively at 25 and 27°C during November 2003 and 2004. Also the same author recorded the maximum number of A. mangiferae (405 and 375 individuals/ 100 leaves) on respectively ''Alphonso'' and ''Ewais'' cultivars. The highest number of A. mangiferae averaged (420 individuals/ 3 inches/ 20 leaves) on ''Alphonso'' in our study (Table 4).
Management study
This is the first report showing management of C. kenyae using combinations between different oils and a pesticide on mango leaves.
Toxicity of different oils, pesticide and their combinations. The current work demonstrated that A. mangiferae found on small leaves around the bud can be controlled by oils during spring before buds opening. Because the oils being hard to penetrate the buds to act, it is useful to control this mite on the leaves before it goes inside the buds.
The corresponding LC50 values after 24 h of treatment (six replicates) against the C. kenyae adults were respectively 1.803, 2.033, 1.734, 1.415, 1.520 and 1.543%. The consequent LC95 values were respectively 17.427, 15.746, 22.488, 14.671, 17.87 and 15.304%. Mineral oil alone recorded the highest slope values after 24 and 48 h (1.850 & 1.702), respectively. The lowest slope was for spirodiclofen (1.478 & 1.618) after 24 and 48 h, respectively. On the other hand, the corresponding LC50 values after 72 h were 0.592, 0.671, 0.557, 0.395, 0.414 and 0.415%, respectively for orange oil, mineral oil, spirodiclofen, mineral oil + spirodiclofen, orange oil + spirodiclofen, and orang oil + mineral oil. The consequent LC95 values were respectively 4.226, 4.099, 3.723, 2.903, 3.021 and 3.926%. The slope values of regression line were 1.927, 2.092, 1.993, 1.899, 1.907 and 1.686, respectively for the same previous treatments.
Our results showed that the most effective compound is mineral oil + spirodiclofen that cause the highest toxicity, while the lowest one was for mineral oil alone (Table 5). The orange oil + spirodiclofen and orange oil + mineral oil gave the same results and was the second best effective. The orange oil and spirodiclofen alone gave the same results and was the third best effective.



Abou-Awad et al. (2009) tested the effect of some pesticides on C. kenyae during 2005 season. The application of Abamectin resulted in a promising control against this mite followed by Chlorfenapyr and Sulphur. Azadirachtin, Methoxyfenozide and Spinosad were less effective.
Therefore, we can report that mineral oil + spirodiclofen gave the best results, followed by orange oil + spirodiclofen or orange oil + mineral oil. Mineral oil was the least effective.
The minor difference between mineral or orange oils and spirodiclofen is due to the toxicity of mineral oil on mango leaves as it affected its texture, thus orange oil can be used instead. Our results indicated that plant oils are more recommended than mineral oils in the Integrated Pest Management programs as promising compounds against the eriophyid mite, C. kenyae in mango orchards in Egypt.
Toxicity of different essential oils. The corresponding LC50 values after 24 h of treatment (four replicates) against C. kenyae adults were respectively 2.213, 2.560, 1.605 and 2.311%. The consequent LC95 values were respectively 63.77, 38.74, 77.45 and 39.84%. Salvia rosmarinus Spenn. (Lamiaceae) oil recorded the highest slope values after 24 and 48 h (1.394 & 1.801), respectively. Whereas, the lowest slope was for Thymus vulgaris L. (Lamiaceae) oil (0.977 & 1.238) after 24 and 48 h, respectively (Table 6).



On the other hand, the corresponding LC50 values after 96 h were 0.269, 0.415, 0.234 and 0.364% and the consequent LC95 values were respectively 3.134, 4.90, 1.83 and 3.94%. The slope values of regression line were 1.542, 1.534, 1.842 and 1.5892 for respectively Geranium robertianum L. (Geraniaceae), S. rosmarinus, T. vulgaris and Syzygium aromaticum (L.) Merr. & L.M. Perry (Myrtaceae) oils.
The results proved that the highest toxicity was for T. vulgaris oil and the lowest toxicity was for S. rosmarinus oil (Table 6). The result agrees with Allam et al. (2020) who found that T. vulgaris is a highly toxic oil in controlling Phyllotetranychus aegypticus Sayed (Tetranychidae).
Our results indicated that LC50 and slope for S. rosmarinus oil after 24 and 48 h were about double in values of that of Elhalawany and Dewidar (2017) as the LC50 was 0.96 and 0.72, the slope was 0.92 and 0.97, respectively for the first and second day on T. urticae. The toxicity of S. rosmarinus oil is less on C. kenyae adults than on T. urticae, this could be due to C. kenyae living under a coating silvery layer. The sprayed oil stay on this layer then penetrates the mite, C. kenyae, while this mite needs a higher dose to be killed. Mossa et al. (2018) evaluated the affectivity of nano emulsion of garlic essential oil on two eriophyid olive mites, Aceria oleae (Nalepa) and Tegolophus hassani (Keifer) and showed high acaricidal activity against both mites, with LC50 298.225 and 309.634 μg/ml, respectively.
Patnaik et al. (2011) tested the efficiency of some aromatic and medicinal essential oils against Aceria guerreronis Keifer on coconut resulting in a significant control of this eriophyid mite. This agrees with our results except that the essential oils used in our study differ from theirs.
Elhalawany and Dewidar (2017) suggested that chamomile, coriander, spearmint and rosemary essential oils have the potential to control T. urticae, as it effectively decreased oviposition with increasing concentration, the highest decrease was observed on females treated with 4% coriander (4.7 eggs/ female compared with 44.3 eggs/ female in untreated females).
Using orange oil instead of T. vulgaris oil and avoiding using the mineral oils in controlling eriophyid mites in mango orchards is highly preferable for agriculture eco-system. The mango producers can choose the most suitable treatment according to their targets (i.e., organic farming, low cost products, safety control materials) in order to keep agriculture eco-friendly and pesticide-free products for both local and foreign markets.
Conclusion
The current study concluded that using mineral oil + spirodiclofen affected on the eriophyid mite, C. kenyae, while using only safty essential oils will be highly recommended instead in order to keep the environement clean and protect the predators for organic production activities followed in mango orchards. Thus, complementary investigations concerning field application of these oils to evaluate their efficacy on mite pests and their accociated predators is still needed. The GC-MS of the active ingredients of these oils is also necessary.
Acknowledgements
Grateful thanks are to Dr. A.S. Elhalawany (Fruit Tree Mite Department, Plant Protection Research Institute, Agricultural Research Centre; Egypt) for his invaluable help during the course of this work. Sincere thanks are to the Applied Centre of Entomonematodes, the Faculty of Agriculture, Cairo University, Giza governorate, Egypt for offering many facilities to finish the current study. Thanks also are to Dr. M.M. Abou-Setta (Scale Insects and Mealy Bugs Department, Plant Protection Research Institute, Agricultural Research Centre; Egypt) and Dr. A.Y. Zaki (Vegetable and Aromatic Plant Mites Department, Plant Protection Research Institute, Agricultural Research Centre; Egypt) for their help in data analysis of the control experiment.
References
- Abdallah A.A.M. 2007. Susceptibility of mango varieties for infestation with mites. Ph. D. Dissertation, Fac. Agric., Al-Azhar Univ., Egypt. 232 pp.
- Abo-Shnaf R.I.A., Moraes G.J.de. 2014. Phytoseiid mites (Acari: Phytoseiidae) from Egypt, with new records, descriptions of new species, and a key to species. Zootaxa, 3865 (1): 001-071. https://doi.org/10.11646/zootaxa.3865.1.1
- Abou-Awad B.A., Metwally A.M., Al-Azzazy M.M.A. 2009. Ecological, biological and control studies on the leaf coating and webbing mite Cisaberoptus kenyae Keifer (Eriophyoidea: Eriophyidae) in Egypt. Acarines: J. Egypt. Soc. Acarol., 3: 65-71. https://doi.org/10.21608/ajesa.2009.4968
- Abou-Awad B.A., Metwally A.M., Al-Azzazy M.M.A. 2011. Environmental management and biological aspects of two eriophyid mango mites in Egypt: Aceria mangiferae and Metaculus mangiferae. Acarologia, 51 (4): 481-497. https://doi.org/10.1051/acarologia/20112030
- Abou-Elella G.M., Saber S.A., El-Sawi S.A. 2013. Biological aspects and life tables of the predacious mites, Typhlodromips swirskii (Athias-Henriot) and Euseius scutalis (Athias-Henriot) feeding on two scale insect species and plant pollen. Arch. Phytopathol. Plant Prot., 46 (14): 1717-1725. https://doi.org/10.1080/03235408.2013.774715
- Allam S.F., Mahmoud M.A.E., Hassan M.F., Mabrouk A.H. 2020. Field application of six commercial essential oils against Date Palm mite, Phyllotetranychus aegypticus (Acari: Tenuipalpidae) in Egypt. Persian J. Acarol., 9 (4): 377-389.
- Al-Shammery K.A. 2010. Different biological aspects of the predacious mite Euseius scutalis (Acari: Gamasida: Phytoseiidae) and the effects due to feeding on three tetranychid mite species in Hail, Saudi Arabia. Asian J. Biol. Sci., 3: 77-80. https://doi.org/10.3923/ajbs.2010.77.84
- Bakr M.E. 2000. Ldp line3. (site of inter.). Avilable from: http://www.Ehab soft.com

- Carrillo M., Elanov P. 2004. The potential of Chrysoperla carnea as a biological control agent of Myzus persicae in glass houses. Ann. Appl. Biol., 32: 433-439.
- Chowdhury S.K. 2015. Diversity and nature of damage of mango insect pests at Kaliachak-II Block of Malda, West Bengal, India. J. Entomol. Zool. Studies, 3 (4): 307-311.
- Elhalawany A.S.H. 2001. Studies on some mite species infesting some fruit trees. M. Sc. Thesis, Fac. Agric., Moshtohor, Zagazig Univ., Benha Bran., Egypt. 163 pp.
- Elhalawany A.S.H., Dewidar A.A. 2017. Efficiency of some plant essential oils against the two-spotted spider mite, Tetranychus urticae Koch and the two predatory mites Phytoseiulus persimilis (A.-H.), and Neoseiulus californicus (McGregor). Egypt. Acad. J. Biolog. Sci., 10 (7): 135-147. https://doi.org/10.21608/eajb.2017.12101
- Elmoghazy M.M.E. 2014. Abundance and diversity of mites associated with date palm, olive and citrus trees in Sakaka, Kingdom of Saudi Arabia. Egypt. Acad. J. Biolog. Sci., 6 (2): 11-16. https://doi.org/10.21608/eajbsz.2014.13482
- El-Sawi S., Momen F. 2006. Agistemus exsertus Gonzalez (Acari: Stigmaeidae) as a predator of two scale insects of the family Diaspididae (Homoptera: Diaspididae). Arch. Phytopathol. Plant Prot., 39 (6): 421-427. https://doi.org/10.1080/03235400500321388
- FAO. 2019. Crop production. Available from: http://www.fao.org/faostat/en/#home

- Finney D.J. 1971. Probit Analysis. Cambridge Univ. Press, 3rd ed., London. 333 pp.
- Flechtmann C.A.H. 1985. On the biology of Ameroseius dendrovagans (Acari, Mesostigmata, Ameroseiidae). Revista. Bras. Zool., 2 (6): 397-399. https://doi.org/10.1590/S0101-81751984000200012
- Gabr R.H.A. 2019. Study the economic stability and competitiveness of Egyptian. Alex. J. Agric. Sci., 64 (6): 493-509.
- Gerson U., Smiley R.L., Ochoa R. 2003. Mites (Acari) for pest control. Blackwell Science, Oxford. 539 pp. https://doi.org/10.1002/9780470750995
- Hutchins M. 2003. Grzimek's animal life encyclopedia: insects. Gale Res. Inc. 496 pp.
- Isman M.B., Machial C.M. 2006. Pesticides based on plant essential oils: from traditional practice to commercialization. In: Carpinella R. (Ed). Naturally occurring bioactive compounds. Elsevier: 29-44. https://doi.org/10.1016/S1572-557X(06)03002-9
- Krantz G.W., Walter D.E. 2009. A manual of acarology. 3rd Ed. Texas Tech Univ. Press, Lubbock, Texas, USA. 807 pp.
- Lindquist E.E., Krantz G.W., Walter D.E. 2009. Order Mesostigmata. In: Krantz G.W., Walter D.E. (Eds). A manual of acarology, 3rd Ed. Texas Tech Univ. Press, Lubbock, Texas, USA: 124-232.
- Manu M. 2013. Diversity of soil mites (Acari: Mesostigmata: Gamasina) in various deciduous forest ecosystems of Muntenia region (southern Romania). Biol. Lett., 50 (1): 3-16. https://doi.org/10.2478/biolet-2013-0001
- Marei F.A., Negm M.W., Nasser M.A., Eraky S.A. 2020. Population dynamics of Oligonychus mangiferus and Aceria mangiferae (Acari: Tetranychidae, Eriophyidae) on two mango cultivars in Assiut governorate, with an annotated checklist of mango mites in Egypt. Int. J. Entomol. Nematol., 6 (1): 149-155.
- Mašán P. 1998. Ameroseius fungicolis sp. n. and A. callosus sp. n., two new ameroseiid species (Acarina, Mesostigmata) associated with wood-destroying fungi. Biologia, Bratislava, 53 (5): 645-649.
- Mašán P. 2007. A review of the family Pachylaelapidae in Slovakia, with systematics and ecology of European species (Acari: Mesostigmata: Eviphidoidea). Institute of Zoology, Slovak Academy of Sciences, Bratislava. 247 pp.
- Mašán P. 2017. A revision of the family Ameroseiidae (Acari, Mesostigmata) with some data on Slovak fauna. ZooKeys, 704: 1-228. https://doi.org/10.3897/zookeys.704.13304
- Mašán P., Halliday B. 2014. Review of the mite family Pachylaelapidae (Acari: Mesostigmata). Zootaxa, 3776 (1): 001-066. https://doi.org/10.11646/zootaxa.3776.1.1
- Masarovič R., Doričová M., Fedor P. 2013.The first record of predaceous Scolothrips longicornis Priesner 1926 (Thysanoptera: Thripidae) in Slovakia. J. Cent. Europ. Agric., 14 (2): 243-250. https://doi.org/10.5513/JCEA01/14.2.1253
- McMurtry J.A. 2010. Concepts of classification of the Phytoseiidae: Relevance to biological control of mites. In: Sabelis M.W., Bruin J. (Eds). Trends in acarology. Proc. 12th Int. Cong.: 393-397. https://doi.org/10.1007/978-90-481-9837-5_64
- McMurtry J.A., Croft B.A. 1997. Life-styles of phytoseiid mites and their roles in biological control. Annu. Rev. Entomol., 42 (1): 291-321. https://doi.org/10.1146/annurev.ento.42.1.291
- McMurtry J.A., Moraes G.J.de, Famah Sourassou N. 2013. Revision of the lifestyles of phytoseiid mites (Acari: Phytoseiidae) and implications for biological control strategies. Sys. Appl. Acarol., 18 (4): 297-320. https://doi.org/10.11158/saa.18.4.1
- Momen F.M. 2009. Life history of predatory mites Typhlodromus athiasae and Amblyseius cabonus (Acari: Phytoseiidae) on two pest mites as prey, with special reference to Eriophyes dioscoridis (Acari: Eriophyidae). Arch. Phytopathol. Plant Prot., 42 (11): 1088-1095. https://doi.org/10.1080/03235400701622204
- Mossa A.H., Afia S.I., Mohafrash S.M.M., Abou-Awad B.A. 2018. Formulation and characterization of garlic (Allium sativum L.) essential oil nanoemulsion and its acaricidal activity on eriophyid olive mites (Acari: Eriophyidae). Environ. Sci. Poll. Res., 25: 10526-10537. https://doi.org/10.1007/s11356-017-0752-1
- Muma M.H. 1971. Food habits of Phytoseiidae (Acarina: Mesostigmata) including common species on Florida citrus. Florida Entomol., 54 (1): 21-34. https://doi.org/10.2307/3493786
- Osman A.A., Zaki A.M. 1986. Studies on the predation efficiency ofAgistemus exsertus Gonzalez (Acarina, Stigmaeidae) on the eriophyid mite Aculops lycopersici (Massee). Anz. Schadl. Pflanz. Umweltsch., 59: 135-136. https://doi.org/10.1007/BF01903320
- Patnaik S., Rout K., Pal S., Panda P.K., Mukherjee P.S., Sahoo S. 2011. Essential oils of aromatic and medicinal plants as botanical biocide for management of coconut eriophyid mite (Aceria guerreronis Keifer). Psyche: 1-5. https://doi.org/10.1155/2011/710929
- Peña J.E., Palevsky E., Otero-Colina G., Ochoa R., Meister C.W. 2005. Mango bud mite, Aceria mangiferae bionomics and control under Florida conditions. Proc. Fla. State Hort. Soc., 118: 228-234.
- Rasmy A.H., Nasr A.K., Reda A.S. 1987. Reproductive response and development of three soil predaceous mites utilizing the acarid mite Tyrophagus casei Oud. as an alternate diet. Anz. Schadl. Pflanz. Umweltsch., 60: 92-94. https://doi.org/10.1007/BF01906036
- Ray H.A., Hoy M.A. 2014. Evaluation of the predacious mite Hemicheyletia wellsina (Acari: Cheyletidae) as a predator of arthropod pests of orchids. Exp. Appl. Acarol., 64 (3): 287-298. https://doi.org/10.1007/s10493-014-9833-8
- Saad R.I., Said G.A., Abdalla M.Y. 2007. Evaluation of mango trees production grown between date palms. Alex. Sci. Ex. J., 28 (1): 25-35. https://doi.org/10.21608/asejaiqjsae.2007.1842
- Sarwar M. 2015. Mite pests (Acari) in mango (Mangifera indica L.) plantations and implementation of control strategy. Biosci. Bioeng., 1 (3): 41-47.
- Schneider K., Migge S., Norton R.A., Scheu S., Langel R., Reineking A., Maraun M. 2004. Trophic niche differentiation in soil microarthropods (Oribatida, Acari): evidence from stable isotope ratios (15N/14N). Soil Biol. Biochem., 36 (11): 1769-1774. https://doi.org/10.1016/j.soilbio.2004.04.033
- Siddiq M., Brecht J.K., Sidhu J.S. 2017. Handbook of mango fruit: production, postharvest science, processing technology and nutrition. Wiley-Blackwell. 320 pp. https://doi.org/10.1002/9781119014362
- Snedecor G.W., Cochran W.G. 1994. Statistical methods, 8th edition. In: Jones D.H. (Ed). Book review. Iowa State Univ. Press, Ames, Iowa, USA. 1989. xix + pp. 491. J. Educ. Behav. Stat., 19 (3): 304-307. https://doi.org/10.3102/10769986019003304
- Steel R.G.D., Torri J.H., Dickey D.A. 1997. Principles and procedures of statistics: a biometrical approach. McGraw-Hill International, New York. 633 pp.
- Summers F.M., Price D.W. 1970. Review of the mite family Cheyletidae. Univ. Ca. Pub. Entomol., 61: 1-153.
- Uddin A., Ahmed S., Ali A., Khoso A.G., Khan M., Asghar F., Asghar K. 2019. Functional response of green lacewing, Chrysoperla carnea (Neuroptera: Chrysopidae) larvae on different insects pests. Int. J. Acad. Multidis. Res., 3 (6): 49-56.
- Walter D.E., Lindquist E.E. 1989. Life history and behavior of mites in the genus Lasioseius (Acari: Mesostigmata) from grassland soils in Colorado, with taxonomic notes and description of a new species. Can. J. Zool., 67 (11): 2797-2813. https://doi.org/10.1139/z89-396
- Zaher M.A. 1984. Survey and ecological studies on phytophagous, predaceous and soil mites in Egypt. I. Phytophagous mites in Egypt (Nile Valley and Delta). PL 480 Programme U.S.A., Project No. EG-ARS-30, Grant No. FG-EG-139. 228 pp.
- Zaher M.A. 1986. Survey and ecological studies on phytophagous, predaceous and soil mites in Egypt. II-A: predaceous and nonphytophagous mites (Nile Valley and Delta). Text. PL 480 Programme U.S.A., Project No. EG-ARS-30, Grant No. FG-EG-139. 567 pp.
- Zimmer M., Kautz G., Werner T. 2003. Leaf litter-colonizing microbiota: supplementary food source or indicator of food quality for Porcellio scaber (Isopoda: Oniscidea)?. Europ. J. Soil Biol., 39: 209-216. https://doi.org/10.1016/j.ejsobi.2003.07.001



2021-03-09
Date accepted:
2021-12-18
Date published:
2022-01-31
Edited by:
Kreiter, Serge

This work is licensed under a Creative Commons Attribution 4.0 International License
2022 Abo-Shnaf, Reham; Allam, Sally F.M.; El-Sobky, Menna L.; Abdul-Shafc, Ahmed F. and El-Tony, Aml G.
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



