Functional and numerical responses of the predator Amblyseius swirskii to its prey Tetranychus turkestani in the laboratory
Rahmani Piyani, Atefeh1 ; Shishehbor, Parviz2 ; Kocheili, Farhan3 and Riddick, Eric W.4
1Department of Plant Protection, Faculty of Agriculture, Shahid Chamran University of Ahvaz, Ahvaz 61357-43311 Iran.
2Department of Plant Protection, Faculty of Agriculture, Shahid Chamran University of Ahvaz, Ahvaz 61357-43311 Iran.
3Department of Plant Protection, Faculty of Agriculture, Shahid Chamran University of Ahvaz, Ahvaz 61357-43311 Iran.
4National Biological Control Laboratory, Agricultural Research Service, USDA, Stoneville, MS 38776 USA.
2021 - Volume: 61 Issue: 4 pages: 901-909
https://doi.org/10.24349/r82w-YLJ1Original research
Keywords
Abstract
Introduction
Tetranychus turkestani Ugarov & Nikolskii (Acari: Tetranychidae) is an important pest of different field and glasshouse crops in the southwestern region of Iran (Mossadegh and Kocheili 2003), Russia (Popov 1981), USA (Jeppson et al. 1975), China (Li et al. 2014), India and some European countries (Zhang 2003). Characterized by frequent outbreaks coupled with acaricide resistance, this mite poses a threat to crop producers (Mossadegh and Kocheili 2003). The hosts of T. turkestani are species of Solanaceae, Cucurbitaceae and Fabaceae in Khuzestan province, southwestern of Iran (Mossadegh and Kocheili 2003; Sohrabi and Shishehbor 2008). Tetranychus turkestani is often found on field bindweed (Convolvulus arvensis L.), which may serve as a reservoir, and on several other weed species along road margins. This mite has several generations during the growing season, tolerates high temperatures and low humidity and has a short generation time of approximately 10 - 12 days (Sohrabi and Shishehbor 2008). Tetranychus turkestani initially feed on the lower leaf surface but can cover an entire plant as population density increases. Typical symptoms of T. turkestani feeding are small, light colored punctures which, in prolonged exposure, develop into irregularly shaped, white or greyish-colored spots. Changes in leaf color from yellowing to bronzing are often characteristics of attack. In some host plant species, leaf curling can be a result of feeding. Necrosis may also occur in young leaves, stems and even growing tips. Sometimes leaf burning and defoliation of the host plant has been observed (Jeppson et al. 1975).
Amblyseius swirskii (Athias-Henriot) (Acari: Phytoseiidae) is a generalist predatory mite which consumes insects, mites, plant pollen, plant exudates, and honeydew (McMurtry et al. 2013). More specifically, it can develop and reproduce on psyllids (Juan-Blasco et al. 2012), tetranychids (Riahi et al. 2017a, b), eriophyids (Abou-Awad et al. 2000; Momen and Abdel-Khalek 2008; Park et al. 2010), tenuipalpids (Peña et al. 2009), tarsonemids (Abou-Awad et al. 2014; Onzo et al. 2012), whiteflies (Nomikou et al. 2001; Medd and Greatrex 2014; Shahbaz et al. 2019), thrips (Messelink et al. 2006; Wimmer et al. 2008), and plant pollen (Swirskii et al. 1967; Nomikou et al. 2001; Park et al. 2010; Lee and Gillespie 2011; Nemati et al. 2019). The capacity to develop and reproduce on plant pollen permits the survival of A. swirskii populations on plants before insect or mite infestation occurs (Calvo et al. 2014; Riahi et al. 2017 a, b). Amblyseius swirskii is commercially available for augmentative biological control of plant pests in more than 50 countries (Park et al. 2010; Calvo et al. 2014; Knapp et al. 2018). Therefore, it is a potential candidate for augmentative biological control of T. turkestani in fields and greenhouses or glasshouses in Khuzestan province, southwestern Iran, and in countries where T. turkestani has become invasive.
Information on functional and numerical responses of A. swirskii to T. turkestani populations are necessary to predict the outcome of augmentative releases of A. swirskii against T. turkestani in the field. The functional response of a predator is a function of prey density and generally follows one of three mathematical models (Holling 1959 a, b, Holling 1961). With a type I functional response model, the number of prey killed increases linearly at constant rate as a function of prey density. With a type II response model, the number of prey killed increases up to a maximum (predator saturation) but the proportion of prey killed declines with prey density. With a type III response model, predation results in a sigmoidal curve. Predators with a type III response can regulate prey populations (Holling 1965). Predators with a type II response have proven to be effective under conditions of low prey density (Koehler 1999; Krebs 1978).
The predator's numerical response is an important factor that has been attributed the success of phytoseiids in pest control (Sabelis 1985). The numerical response is a progressive change in the number of offspring in relation to prey density (Solomon 1949), and female predatory mites can vary their progeny production at different prey densities. In phytoseiid predatory mites, the numerical response is linked to the density of prey mites (Cédola et al. 2001; Nomikou et al. 2010; Carrillo and Peña 2012; Fathipour et al. 2020). McMurtry and Rodriguez (1987) showed that the numerical response probably had a more important role in the adjustment of pest mite populations than the functional response because increments in the numerical response, with an increase of prey density, also involved an increment in the reproduction and other important components, such as the survival of progeny and the duration of development time. The analysis of both the functional and numerical responses is helpful and provides information regarding the prey-predator relationship, which is required for the successful usage of a biological control agent.
The functional responses of A. swirskii feeding on an eriophyid Aculops lycopersici (Massee) (Park et al. 2010), astigmatid Suidasia medanensis (Oudemans) (Acari: Suidasiidae) (Midthassel et al. 2014), tenuipalpid Cenopalpus irani Dosse, and the tetranychids Tetranychus urticae Koch (Xiao et al. 2013; Fathipour et al. 2020) and Eotetranychus frosti (McGregor) (Bazgir et al. 2020) have been reported previously. However, no published data are available on the functional response of A. swirskii feeding on T. turkestani (Rahmani Piyani 2021). In addition, only the numerical response of A. swirskii on T. urticae during 24 h is known (Fathipour et al. 2020). Therefore, the aim of this study was to determine the functional and numerical responses of A. swirskii feeding on T. turkestani. This information will provide baseline data necessary to predict the effectiveness of augmentative releases of A. swirskii to control T. turkestani on crop plants in greenhouses, glasshouses, or open fields.
Material and methods
Mite colonies
Tetranychus turkestani used in this study originated from a weed (Convolvulus arvensis L.) at Faculty of Agriculture, Shahid Chamran University of Ahvaz, Ahvaz, Iran. A stock colony of T. turkestani was maintained on cowpea (Vigna unguiculata (L.) Walp.) seedlings in compost in plastic pots (20 cm diam). Infested plants were held in wooden-framed rearing cages (120 × 60 × 60 cm) covered with nylon mesh (210 µm aperture). The cages were maintained in a laboratory at 25 ± 2 °C, 60 ± 5 % RH and a 16: 8 (L: D) h with illumination (4000 lux) provided by fluorescent lamps. New plants were introduced as required to maintain the colony.
The initial population of A. swirskii was obtained from laboratory colonies at Bu-Ali Sina University, Hamedan, Iran, which had been purchased from Koppert Biological Systems (Berkel en Rodenrijs, The Netherlands) in 2012 and reared on T. urticae. Each rearing unit consisted of a plastic sheet over a foam mat maintained under humid conditions with distilled water in a Plexiglas box (17 cm ×10 cm ×6 cm). To provide food, young cowpea leaves infested with T. turkestani were added to the units twice a week.
Experimental design
Experimental units were prepared according to the method described by Vantornhout et al. (2004) with some modifications. Each experimental unit was comprised of a green plastic sheet (3 cm x 3 cm) located on a thick foam pad, cut to a similar size (3 cm x 3 cm), in a plastic Petri dish (9 cm diam., 2 cm high) arena, half-filled with water. The edges of the plastic sheet were covered with moist tissue paper to prevent A. swirskii females from escaping. In addition, a few cotton threads were placed on each plastic sheet to provide shelter and oviposition substrates for A. swirskii females. Plant substrates were not used in the A. swirskii colony or in experimental units. A. swirskii prefers glabrous leaves (McMurtry et al. 2013), so we used a green plastic sheet as a substrate for experimental arenas. Besides several researchers also used plastic sheet instead of leaf to study the biology predatory behavior and rearing of phytoseiid mites (Vantornhout et al. 2004; Khanamani et al. 2016; Riahi et al. 2017c).
To determine the functional response of A. swirskii, a newly emerged female (less than 24 h old) was exposed to seven densities (2, 4, 8, 16, 32, 64 and 128) of T. turkestani immature stages including larvae, protonymphs, and deutonymphs in similar proportions. Specifically, density 2 included 1 larva and 1 protonymph; density 4 included 1 larva, 1 deutonymph and 2 protonymphs; density 8 included 2 larvae, 2 deutonymphs and 4 protonymphs; density 16 included 5 larvae, 5 deutonymphs and 6 protonymphs; density 32 included 10 larvae, 10 deutonymphs and 12 protonymphs; density 64 included 20 larvae, 20 deutonymphs and 24 protonymphs; and density 128 included 40 larvae, 40 deutonymphs and 48 protonymphs. This procedure was used because our observations indicated that A. swirskii preferred the protonymph stage compared to other developmental stages of T. turkestani. Tetranychus turkestani immatures were transferred from cowpea plant foliage to Petri dish arenas using a fine paintbrush (no. 000). After 24 h, the number of dead larvae and nymphs were counted; all immature stages were combined in the final counts. Each prey density was replicated 10 times. The experiment was conducted in the laboratory at 25 ± 2 °C, 60 ± 5 % RH and a 16: 8 (L:D) h photoperiod.
In determining the numerical response, the same methods as described above were used to expose T. turkestani larvae, protonymphs, and deutonymphs to A. swirskii females. Note that A. swirskii mated females were transferred into new Petri dishes containing a fresh supply of T. turkestani prey, each day. Prey replacement was continued for the duration of the lifespan of each A. swirskii female. The number of eggs laid by each female and its longevity was recorded every day from the beginning of the numerical response experiment until the death of each female. Each prey density was replicated 10 times. The experiment was conducted in the laboratory at 25 ± 2 °C, 60 ± 5 % RH and a 16: 8 (L:D) h photoperiod.
Statistical analysis
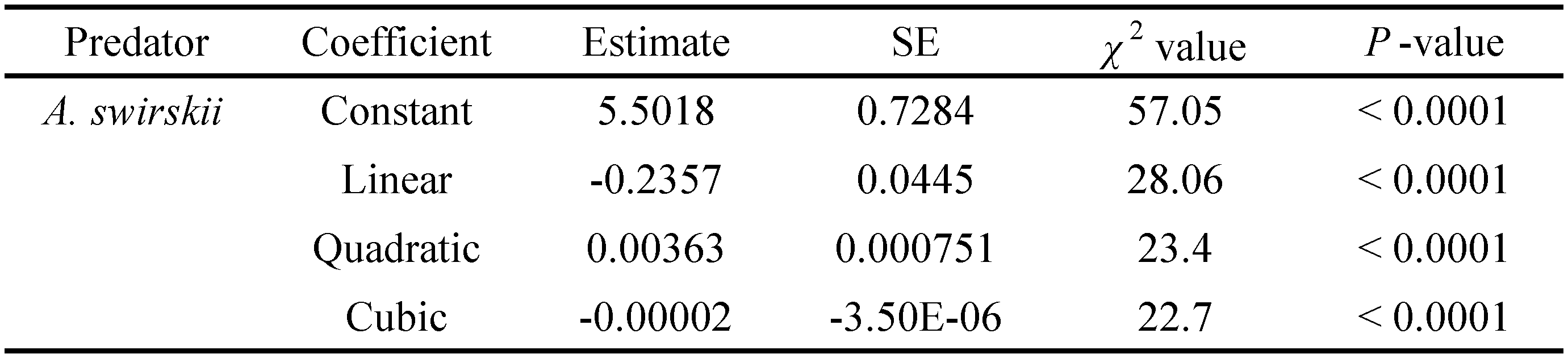
The data on functional response were analyzed in two steps (Juliano 2001). First, the type of functional response was determined by a logistic regression of the proportion of prey consumed (Na/N0) as a function of prey density (N0) (equation 1):

where Na is the number of prey attacked, N0 is the initial density of prey, P0, P1, P2 and P3 are the intercept, linear, quadratic, and cubic coefficients, respectively, estimated using the maximum likelihood method. The sign of the P1 from the above equation can be used to distinguish the type of the functional response curve. If P1 \textless0, it describes a type II functional response. If P1\textgreater0 and P2\textless0, it shows a type III functional response (Juliano 2001). In the next step, handling time (Th) and attack rate (a) coefficients were estimated using the Roger's random predator equation (Rogers 1972) (equation 2):

where Na and N0 are described above, T is 24 h, the total time available for the predator, a is attack rate and Th is the handling time. Nonlinear regression was used to estimate the attack rate and the handling time parameters (Proc NLIN, SAS Institute 2003), and the maximum predation rate was calculated from 24/Th.
Data on numerical response were analyzed using one-way analysis of variance (one-way ANOVA) to test whether the number of T. turkestani consumed and the number of eggs laid by A. swirskii were different in arenas with different prey density. Statistical analysis was conducted with SAS software (SAS Institute 2003). Differences between treatment means were compared by Tukey's HSD test (α = 0.05).
Results
Functional response
The one-way ANOVA revealed a significant effect of prey density on predation rate (F = 14.87; df = 6, 63; P < 0.001). The consumption rate of A. swirskii females increased as T. turkestani density increased (Figure 1). The functional response was density-dependent and corresponded to a Holling's type II response curve (Table 1); the response increased on average from two individuals consumed a day when prey density was two T. turkestani immatures to 58 individuals when prey density was 128 T. turkestani immatures per arena.



The attack rate and handling time estimates from the random predator equation (equation 2) were 0.1148 h -1 and 0.3146 h, respectively (Table 2). The maximum number of T. turkestani immatures that one A. swirskii female could attack was 76.28 individuals in a day.





Numerical response and longevity
Fecundity of A. swirskii was greatly affected by T. turkestani density (F = 40.90; df = 6, 63; P < 0.001; Figure 2). Predators reared at the second highest prey density (64 prey per arena) produced the maximum number of eggs (33.10 eggs). Analysis of variance indicated that T. turkestani density had no significant effect on longevity of A. swirskii females (F = 0.54; df = 6, 63; P = 0.78; Table 3). Mean longevity was 21.26 days (18.86 – 24.76 days).






Discussion
The functional response displayed by A. swirskii was well-defined by the Roger's model. The functional response curve reached a plateau at approximately 64 prey individuals during 24 h, suggesting a type II functional response where predation rate decreased as prey density increased. This type of response has been reported for A. swirskii on tetranychid (Xiao et al. 2013; Fathipour et al. 2017; Fathipour et al. 2020), tenuipalpid (Bazgir et al. 2020), and eriophyid (Park et al. 2010) mites. However, Fathipour et al. (2020) reported that the functional response of A. swirskii feeding on T. urticae eggs and nymphs changed to a type III response when maize pollen was provided as an additional food source.
Attack rate and handling time are two parameters that describe the magnitude of functional responses (Pervez and Omkar 2005). Handling time is an important indicator of predator effectiveness and reflects the cumulative time required for identifying, chasing, capturing, killing, and eating prey (Holling 1959 a, b, Holling 1965, Holling 1966). Different factors can influence handling time such as the speed of predator, movement of prey, and the time needed for predators to overcome individual prey (Hassell 1978), which could be related to defensive behavior of prey (Ali et al. 2011). The handling time found in this study (0.3146 h) was lower than the values reported for A. swirskii feeding on T. urticae eggs (0.5184 h, Xiao et al. 2013), T. urticae deutonymphs (0.9959 h, Fathipour et al. 2020) and E. frosti deutonymphs (0.8052 h, Bazgir et al. 2020); suggesting that A. swirskii had a greater predation response when feeding on a mixture of T. turkestani immature stages. The shorter handling time in this study may be due to the presence of T. turkestani larvae, which are easier to capture and consume in comparison to other immature prey stages. Similarly, Ganjisafar and Perring (2015) reported that the predatory mite Galendromus flumentis (Chant) had the shortest handling time when consuming larvae of the bank grass mite, Oligonychus pratensis (Banks).
In this study, A. swirskii females produced an average maximum number of 33.10 eggs at a prey density of 64 T. turkestani immatures prey arena. Other laboratory studies with adequate densities of mature and immature stages of prey have reported a variety of fecundity (number of eggs) values for A. swirskii. For example, at 25 °C, Riahi et al. (2017 b) reported that A. swirskii produced an average of 40.85 eggs on T. urticae; Asgari et al. (2020) documented that A. swirskii produced an average of 82.17 eggs on Carpoglyphus lactis L. (Acari: Carpoglyphidae); Bazgir et al. (2018) found the average number of eggs produced by A. swirskii was 34.69 eggs and 25.96 eggs on E. frosti and Cenopalpaus irani Dosse (Acari: Tenuipalpidae), respectively. Hosseininia et al. (2020) reported an average of 25.21, 21.46, and 14.87 eggs produced by A. swirskii feeding on T. urticae, C. lactis and Trialeurodes vaporariorum Westwood (Hemiptera: Aleyrodidae), respectively. In comparison to other studies, A. swirskii fecundity was moderate when fed T. turkestani immatures.
The longevity of A. swirskii females (21.26 d) fed T. turkestani in this study was comparable with that reported by Fadaei et al. (2018), namely 22.49 d at the same temperature (25 °C) but on different prey, T. urticae. Other researchers have reported a great deal of variation in the longevity of A. swirskii. At 25 °C, Riahi et al. (2017a) reported longevity of 34.2 d on T. urticae; Bazgir et al. (2018) discovered longevity of 39.81 and 32.41 d on E. frosti and C. irani, respectively, whereas Hosseininia et al. (2020) reported longevity of 25.21 d, 21.46 d, and 14.87 d on T. urticae, C. lactis, and T. vaporariorum, respectively. Considering previous works, A. swirskii longevity was moderate on T. turkestani immatures.
In conclusion, this study provides baseline knowledge on the association between A. swirskii and T. turkestani. The existing evidence proposes that A. swirskii can play an important role in reducing T. turkestani populations. However, results of the functional response experiment indicate that A. swirskii could be more effective at low T. turkestani population density. Nevertheless, it is necessary to consider the results with caution because these experiments have been conducted under simplified conditions using small arenas and no plant leaf substrates. In a whole plant system, A. swirskii adults can spread from one plant to the next and interact with other prey and predator populations, which could significantly affect its efficiency. Future research on prey stage preferences of A. swirskii and intraguild predation should add to the understanding of the capacity of A. swirskii as an effective biological control agent of T. turkestani. Moreover, glasshouse (greenhouse) and field studies are necessary to determine the effectiveness of A. swirskii against T. turkestani under more realistic conditions.
Acknowledgements
Authors thank the research deputy of Shahid Chamran University of Ahvaz for supporting this research. Two anonymous peer reviewers commented on a previous version of this manuscript. The U.S. Government has the right to retain a non-exclusive, royalty-free license in and to any copyright of this article. This article reports the results of research only. Mention of a commercial or proprietary product does not constitute an endorsement of the product by the U.S. Department of Agriculture or Shahid Chamran University of Ahvaz.
References
- Abou-Awad B.A., El-Sawaf B.M., Abdel-Khalek A.A. 2000. Impact of two eriophyoid fig mites, Aceria ficus and Rhyncaphytoptus ficifoliae, as prey on postembryonic development and oviposition rate of the predacious mite Amblyseius swirskii. Acarologia, 40: 367-371.
- Abou-Awad B.A., Hafez S.M., Farhat B.M. 2014. Biological studies of the predacious mite Amblyseius swirskii, a predator of the broad mite Polyphagotarsonemus latus on pepper plants (Acari: Phytoseiidae: Tarsonemidae). Arch. Phytopathol. Plant Prot., 47: 349-354. https://doi.org/10.1080/03235408.2013.809908
- Ali M., Naif A.A., Huang D. 2011. Prey consumption and functional response of a phytoseiid predator, Neoseiulus womersleyi, feeding on spider mite, Tetranychus macfarlanei. J. Insect Sci., 11: 167. https://doi.org/10.1093/jis/11.1.167
- Asgari F., Sarraf Moayeri H.R., Kavousi A., Enkegaard A., Chi H. 2020. Demography and mass rearing of Amblyseius swirskii (Acari: Phytoseiidae) fed on two species of stored-product mites and their mixture. J. Econ. Entomol., 113: 2604-2612. https://doi.org/10.1093/jee/toaa187
- Bazgir F., Shakarami J., Jafari S.H. 2018. Life table and predation rate of Amblyseius swirskii (Acari: Phytoseiidae) fed on Eotetranychus frosti (Tetranychidae) and Cenopalpus irani (Tenuipalpidae). Syst. Appl. Acarol., 23: 1614-1626. https://doi.org/10.11158/saa.23.8.11
- Bazgir F., Shakarami J., Jafari S.H. 2020. Functional response of the predatory mite Amblyseius swirskii (Acari: Phytoseiidae) to Eotetranychus frosti (Tetranychidae) and Cenopalpus irani (Tenuipalpidae). Acarologia, 60: 30-39. https://doi.org/10.24349/acarologia/20204359
- Calvo F.J., Knapp M., Yvonne M., Houten V., Hoogerbrugge H., Belda J.E. 2014. Amblyseius swirskii: What made this predatory mite such a successful biocontrol agent? Exp. Appl. Acarol., 65: 419-433. https://doi.org/10.1007/s10493-014-9873-0
- Carrillo D., Peña JE. 2012. Prey-stage preference and functional and numerical responses of Amblyseius largoensis (Acari: Phytoseiidae) to Raoiella indica (Acari: Tenuipalpidae). Exp. App. Acarol., 57: 361-372. https://doi.org/10.1007/s10493-011-9488-7
- Cédola C., Sánchez N., Liljesthröm G. 2001. Effect of tomato leaf hairiness on functional and numerical response of Neoseiulus californicus (Acari: Phytoseiidae). Exp. App. Acarol., 5: 819-831. https://doi.org/10.1023/A:1020499624661
- Fadaei E., Hakimitabar M., Seiedy M., Sarraf Moaieri H.R. 2018. Effects of different diets on biological parameters of the predatory mite Amblyseius swirskii (Acari: Phytoseiidae). Int. J. Acarol., 44: 341-346. https://doi.org/10.1080/01647954.2018.1525428
- Fathipour Y., Karimi M., Farazmand A., Talebi A.S. 2017. Age-specific functional response and predation rate of Amblyseius swirskii (Phytoseiidae) on two-spotted spider mite. Sys. Appl. Acarol., 22: 159-169. https://doi.org/10.11158/saa.22.2.1
- Fathipour Y., Maleknia B., Bagheri A., Soufbaf M., Reddy G.V.P. 2020. Functional and numerical responses, mutual interference, and resource switching of Amblyseius swirskii (Phytoseiidae) on two-spotted spider mite. Biol. Control, 146: 104266. https://10.1016/j.biocontrol2020.104266
- Ganjisafar F., Perring T.M. 2015. Prey stage preference and functional response of the predatory mite Galendromus flumentis to Oligonychus pratensis. Biol. Control, 82: 40-45. https://doi.org/10.1016/j.biocontrol.2014.12.004
- Hassell M.P. 1978. The dynamics of arthropod predator-prey systems. Princeton University Press, 248 pp. https://doi.org/10.2307/3280305
- Holling C.S. 1959a. Some characteristics of simple types of predation and parasitism. Can. Entomol., 91: 385-398. https://doi.org/10.4039/Ent91385-7
- Holling C.S. 1959b. The components of predation as revealed by a study of small-mammal predation of European pine sawfly. Can. Entomol., 91: 293-320. https://doi.org/10.4039/Ent91293-5
- Holling C.S. 1961. Principles of insect predation. Annu. Rev. Entomol., 6: 163-182. https://doi.org/10.1146/annurev.en.06.010161.001115
- Holling C.S. 1965. The functional response of predators to prey density and its role in mimicry and population regulation. Mem. Entomol. Soc. Can., 45:1-60. https://doi.org/10.4039/entm9745fv
- Holling C.S. 1966. The functional response of insect predators to prey density. Mem. Entomol. Soc. Can., 48: 1-86. https://doi.org/10.4039/entm9848fv
- Hosseininia A., Khanjani M., Asadi M., Soltani J. 2020. Life-history of the predatory mite Amblyseius swirskii (Athias-Henriot) (Acari: Phytoseiidae) on Tetranychus urticae Koch (Acari: Tetranychidae), Carpoglyphus lactis Linnaeus (Acari: Carpoglyphidae) and Trialeurodes vaporariorum (Westwood) (Hemiptera: Aleyrodidae). J. Ornamental Plants, 10: 155-166.
- Jeppson L.R., Keifer H.H., Buker E.W. 1975. Mites injurious to economic plants. Univ. Cal. Press. Berkeley, pp. 614.
- Juan-Blasco M., Oureshi J.A., Urbaneja A., Stansly P.A. 2012. Predatory mite, Amblyseius swirskii (Acari: Phytoseiidae), for biological control of Asian citrus psyllid, Diaphorina citri (Hemiptera: Psyllidae). Fla. Entomol., 95: 543-551. https://doi.org/10.1653/024.095.0302
- Juliano S.A. 2001. Non-linear curve fitting: predation and functional response curve. Design and analysis of ecological experiments. In: Scheiner S. M., Gurevitch J. (Eds). New York, Oxford U. Press. pp. 178-196.
- Khanamani M., Fathipour Y., Talebi AA., Mehrabadi M. 2016. Linking pollen quality and performance of Neoseiulus californicus (Acari: Phytoseiidae) in two-spotted spider mite management programs. Pest Manag. Sci., 73: 452-461.
- Knapp M., van Houten Y., van Ball E., Groot T. 2018. Use of the predatory mites in commercial biocontrol: current status and future prospects. Acarologia, 58 (Suppl): 72-82.
- Koehler H.H. 1999. Predatory mites (Gamasina: Mesostigmata). Agric. Ecosys. Environ., 74: 395-410. https://doi.org/10.1016/S0167-8809(99)00045-6
- Krebs C.J. 1978. Ecology: the experimental analysis of distribution and abundance. Second edition. Harper and Row, New York.
- Lee H., Gillespie D. 2011. Life table and development of Amblyseius swirskii (Acari: Phytoseiidae) at different temperatures. Exp. Appl. Acarol., 53: 17-27. https://doi.org/10.1007/s10493-010-9385-5
- Li G.Y., Li J.J., Xia W., Qu H.L., Yang S., Zhang J.P. 2014. Effects of Bt+ Cp TI transgenic cotton on the performance of Tetranychus turkestani (Acari: Tetranychidae). Syst. Appl. Acarol., 19: 236-247. https://doi.org/10.11158/saa.19.2.14
- McMurtry J.A., Rodriguez J.G. 1987. Nutritional ecology of phytoseiid mites. In: Slansky F., Rodriguez J.G. (Eds). Nutritional ecology of insects, mites, spiders and related invertebrates. J. Wiley and Sons, New York, pp: 609-644.
- McMurtry J.A., Moraes G.J., Famah Sourassou N. 2013. Revision of the lifestyle of phytoseiid mites (Acari: Phytoseiidae) and implications for biological control strategies. Syst. Appl. Acarol., 18: 297-320. https://doi.org/10.11158/saa.18.4.1
- Medd N.C., Greatrex R.M. 2014. An evaluation of three predatory mite species for the control of greenhouse whitefly (Trialeurodes vaporariorum). Pest Manag. Sci., 70: 1492-1496. https://doi.org/10.1002/ps.3794
- Messelink G.J., Van Steenpaal E.F., Ramakers P.M.J. 2006. Evaluation of phytoseiid predators for control of western flower thrips on greenhouse cucumber. BioControl, 51: 753-768. https://doi.org/10.1007/s10526-006-9013-9
- Midthassel A., Leather S.R., Wright DJ., Baxter I.H. 2014. The functional and numerical response of Typhlodromips swirskii (Acari: Phytoseiidae) to the factitious prey Suidasia medanensis (Acari: Suidasidae) in the context of a breeding sachet. Biocontrol Sci. Technol., 24: 361-374. https://doi.org/10.1080/09583157.2013.863270
- Momen F.M., Abdel-Khalek A. 2008. Effect of the tomato rust mite Aculops lycopersici (Acari: Eriophyidae) on the development and reproduction of three predatory phytoseiid mites. Int. J. Trop. Insect Sci., 28: 53-57. https://doi.org/10.1017/S1742758408942594
- Mossadegh M.S., Kocheili F. 2003. A semi descriptive checklist of identified species of arthropods (agricultural, medical) and other pests from Khuzestan, Iran. Shahid Chamran U., Ahvaz Press, Ahvaz, Iran.
- Nemati A., Riahi E., Khalili-Moghadam A., Gwiazdowicz D.J., Bahari M.R., Amini P. 2019. Comparison of different pollen grains and a factitious prey as food sources for Amblyseius swirskii (Acari: Phytoseiidae). Syst. Appl. Acarol., 24: 2427-2438. https://doi.org/10.11158/saa.24.12.10
- Nomikou M., Janssen A., Schraag R., Sabelis M.W. 2001. Phytoseiid predators as potential biological control agents for Bemisia tabaci. Exp. Appl. Acarol., 25: 271-291. https://doi.org/10.1023/A:1017976725685
- Nomikou AJ., Sabelis MW., Janssen A. 2010. Pollen subsidies promote whitefly control through numerical response of predatory mites. BioControl, 55: 253-260. https://doi.org/10.1007/s10526-009-9233-x
- Onzo A., Houedokoho A.F., Hanna R. 2012. Potential of the predatory mite, Amblyseius swirskii to suppress the broad mite, Polyphagotarsonemus latus on the gboma eggplant, Solanum macrocarpon. J. Insect Sci., 12: 7. https://doi.org/10.1673/031.012.0701
- Park H., Shipp L., Buitenhuis R. 2010. Predation, development, and oviposition by the predatory mite Amblyseius swirskii (Acari: Phytoseiidae) on tomato russet mite (Acari: Eriophyidae). J. Econ. Entomol., 103: 563-569. https://doi.org/10.1603/EC09161
- Peña J.E., Rodrigues J.C.V., Roda A., Carrillo D., Osborne L.S. 2009. Predator-prey dynamics and strategies for control of the red palm mite (Raoiella indica) (Acari: Tenuipalpidae) in areas of invasion in the Neotropics. IOBC WPRS Bull, 50: 69-79.
- Pervez A., Omkar. 2005. Functional response of coccinellid predators: an illustration of a logistic approach. J. Insect Sci., 5: 1-6. https://doi.org/10.1093/jis/5.1.5
- Popov S.Y. 1981. Survival tables and biological parameters of the spider mite Tetranychus turkestani. Izvestia Timiryazeskoi Selesokhozyaistvonnoi Akademii, 1: 124-133.
- Rahmani Piyani, A. 2021. Biological characteristics and functional and numerical responses of predatory mite Amblyseius swirskii Athias-Henriot (Acari: Phytoseiidae) on strawberry spider mite, Tetranychus turkestani Ugarov & Nikolskii (Acari: Tetranychidae). M. Sc. Thesis. Shahid Chamran U. Ahvaz, Ahvaz, Iran.
- Riahi E., Fathipour Y., Talebi A.A., Mehrabadi M. 2017a. Linking life table and consumption rate of Amblyseius swirskii (Acari: Phytoseiidae) in presence and absence of different pollens. Ann. Entomol. Soc. Am., 110: 244-253. https://doi.org/10.1093/aesa/saw091
- Riahi E., Fathipour Y., Talebi A.A., Mehrabadi M. 2017b. Natural diets versus factitious prey: Comparative effects on development, fecundity and life table of Amblyseius swirskii (Acari: Phytoseiidae). Syst. Appl. Acarol., 22: 711-723. https://doi.org/10.11158/saa.22.5.10
- Riahi E., Fathipour Y., Talebi AA., Mehrabadi M. 2017c. Attempt to develop cost-effective rearing of Amblyseius swirskii (Acari: Phytoseiidae): assessment of different artificial diets. J. Econ. Entomol., 112: 1- 8. https://doi.org/10.1093/jee/tox172
- Rogers D. 1972. Random search and insect population models. J. Anim. Ecol., 41: 369-383. https://doi.org/10.2307/3474
- Sabelis MW. 1985. Predation on spider mites. In Helle W., Sabelis M. W. (Eds). World crop pests: Spider mites, their biology, natural enemies and control. Elsevier, Amsterdam. Pp: 103- 129.
- SAS Institute. 2003. SAS/STAT User's guide, V. 9.1., 8th ed., SAS Institute, Cary, NC. 943 pp.
- Shahbaz M., Khoobdel M., Khanjani M., Hosseininia A., Javadi Khederi S. 2019. Sublethal effects of acetamiprid on biological aspects and life table of Amblyseius swirskii (Acari: Phytoseiidae) fed on Aleuroclava jasmini (Hemiptera: Aleyrodidae). Syst. Appl. Acarol., 24: 814-824. https://doi.org/10.11158/saa.24.5.7
- Sohrabi F., Shishehbor P. 2008. Effects of host plant and temperature on growth and reproduction of the strawberry spider mite Tetranychus turkestani Ugarov & Nikolskii (Acari: Tetranychidae). Syst. Appl. Acarol., 13: 26-32. https://doi.org/10.11158/saa.13.1.2
- Swirski E., Amitai S.Y., Dorzia N. 1967. Laboratory studies on feeding development and reproduction of the predaceous mites Amblyseius rubini and Amblyseius swirskii Athias (Acarina: Phytoseiidae) on various kind of food substances. Isr. J. Agric. Res., 17: 101-119.
- Vantornhout I., Minnaert H. L., Tirry L., De Clercq P. 2004. Effect of pollen, natural prey and factitious prey on the development of Iphiseius degenerans. BioControl, 49: 627-644. https://doi.org/10.1007/s10526-004-5280-5
- Wimmer D., Hoffmann D., Schausberger P. 2008. Prey suitability of western flower thrips, Frankliniella occidentalis, and onion thrips, Thrips tabaci, for the predatory mite Amblyseius swirskii. Biocontrol Sci. Technol., 18: 533-542. https://doi.org/10.1080/09583150802029784
- Xiao Y., Osborne L.S., Chen J., McKenzie C.L. 2013. Functional responses and prey-stage preferences of a predatory gall midge and two predacious mites with two spotted spier mite, Tetranychus urticae, as a host. J. Insect Sci., 13, 8. https://doi.org/10.1673/031.013.0801
- Zhang Z.Q. 2003. Mites of greenhouses: identification, biology and control. CABI Publ., 244 pp. https://doi.org/10.1079/9780851995908.0000



2021-07-22
Date accepted:
2021-10-12
Date published:
2021-10-21
Edited by:
Palevsky, Eric

This work is licensed under a Creative Commons Attribution 4.0 International License
2021 Rahmani Piyani, Atefeh; Shishehbor, Parviz; Kocheili, Farhan and Riddick, Eric W.
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



