Scapulaseius asiaticus (Evans) and S. reptans (Blommers) (Mesostigmata: Phytoseiidae): one or two species?
Kreiter, Serge  1
and Ferragut, Francisco
1
and Ferragut, Francisco  2
2
1✉ Institut Agro-Montpellier SupAgro, UMR CBGP INRA/ IRD/ CIRAD/ SupAgro, 755 Avenue du Campus Agropolis (Baillarguet), CS 30016, 34988 Montferrier-sur-Lez cedex, France.
2Instituto Agroforestal Mediterráneo, Universitat Politècnica de València, Valencia, Spain.
2021 - Volume: 61 Issue: 3 pages: 747-757
https://doi.org/10.24349/tyea-u51QOriginal research
Keywords
Abstract
Introduction
Mites of the family Phytoseiidae are famous for their predatory behaviours on phytophagous mites and small insects on cultivated plants and wild vegetation. Several species are commercialised for the control of pest organisms in agricultural open fields and, above all, in protected crops all around the world (McMurtry and Croft 1997, McMurtry et al. 2013, Knapp et al. 2018). This family is widespread around the world, present on all inhabited continents and consists presently of 2,521 valid species belonging to 95 genera, 15 tribes and three sub-families (Chant and McMurtry 2007, Demite et al. 2021).
Biodiversity surveys in poorly investigated areas is still an urgent need and might result in the discovery of additional species potentially useful for biological control as well as having more information on the biodiversity of these areas and on taxonomy of species discovered. Most of the Indian Ocean constitutes one of the world's biodiversity hotspots (Myers 1988, Myers et al. 2000). Knowledge of the phytoseiid diversity in these areas may contribute to future establishment of conservation programs as well as biocontrol agents.
Mascareignes Archipelago is composed of three main islands: La Réunion, Mauritius and Rodrigues, all located in the East of Madagascar. Comoros Archipelago is composed of four main Islands: Mayotte, Anjouan, Mohéli and Grande Comore, located in the Northwest of Madagascar.
Ferragut and Baumann (2019) have investigated the Mauritian fauna of Phytoseiidae in April 2018. All main Islands of the two Archipelagos (except La Réunion which was investigated before, see Kreiter et al. 2020c) were investigated from October 25th to December 12th, 2018. Results of Phytoseiidae records during this survey were already published in seven papers: Kreiter and Abo-Shnaf 2020a & b for Rodrigues and Mauritius (in addition for Mauritius, see Kreiter et al. 2018a); Kreiter et al. 2020a, 2021b, c & d for Mayotte, Anjouan, Mohéli, and Grande Comore (in addition for Grande Comore, see Kreiter et al. 2018b), and Kreiter et al. (submitted) for description of six new species and six newly collected unknown males, respectively.
During these surveys in Mascareignes, two of the common species were Scapulaseius asiaticus (Evans) and S. reptans (Blommers). Authors have mentioned that these two species are morphologically very close (Ferragut and Baumann 2019; Kreiter and Abo-Shnaf 2020b).
The objective of this paper is to test the hypothesis of synonymy between these two species of the genus Scapulaseius, suspected by Ferragut and Baumann (2019) and Kreiter and Abo-Shnaf (2020b) after surveys in Mauritius Island.
Material and methods
The material examined comes from several surveys conducted by the senior author and from the type specimens of both species borrowed from the respective collections.
Surveys were done in La Réunion Island in 2016-2017 (see Kreiter et al. 2020c), in Vietnam in 2017 (Kreiter et al. 2020b), in Mauritius Island in 2017 (see Kreiter et al. 2018a) and in 2018 (see Ferragut and Baumann 2019 and Kreiter and Abo-Shnaf 2020b). Specimens were collected from cultivated and wild plants in several locations. Mites were directly collected on leaves with a fine brush or by beating the plants and collecting the mites in a black plastic rectangular saucer 45 x 30 cm (Ref. STR 45, BHR, 71370 Saint-Germain-du-Plain, France), depending on the plant investigated:
- large leaves of shrubs and trees with the direct collection method or by beating;
- very small leaves, spiny shrubs, trees and herbaceous plants with the beating method.
We transferred the mites with a brush into small plastic vials containing 1.5 ml of 70% ethanol. The mites were then all slide-mounted in Hoyer's medium (Walter and Krantz 2009), the slides were dried at 45-50oC for at least two weeks and then all examined and identified using a phase and interferential contrast microscope (DMLB, Leica Microsystèmes SAS, Nanterre, France). We used a graded eyepiece for measuring characters of specimens (Leica, see above).
Moreover, the following type and additional material have been borrowed and studied:
- The holotype, two paratype females and one paratype male of Scapulaseius asiaticus (Evans), from the reference collection of the National History Museum (NHM), Department of Life Sciences (Entomology), Cromwell Road, London SW7 5BD, United Kingdom;
- The holotype, four paratype females, two paratype males, 17 additional female and four additional male specimens as additional material of Scapulaseius reptans (Blommers), housed in the mites reference collection of the Naturalis Biodiversity Center, Darwinweg 2, 2333 CR Leiden, The Netherlands.
Chant and McMurtry's (1994, 2007) concepts of the taxonomy of the family Phytoseiidae and the world catalogue database of Demite et al. (2014, 2021) were used for identification and for distribution and information on descriptions and re-descriptions, respectively. The setal nomenclature system adopted was that of Lindquist and Evans (1965) and Lindquist (1994) as adapted by Rowell et al. (1978) and Chant and Yoshida-Shaul (1992) for the dorsum and by Chant and Yoshida-Shaul (1991) for the venter. Pore (= solenostome) and poroid (= lyrifissure) notations are that of Athias-Henriot (1975). Macrosetal notation (Sge = genual macroseta; Sti = tibial macroseta; St = tarsal macroseta) are that of Muma and Denmark (1970). Numbers of teeth on the fixed and movable cheliceral digits do not include the respective apical teeth. Setae not referred to in results section should be considered as absent. All measurements are given in micrometres (µm) and presented with the mean in bold followed by the range in parenthesis. Classification of plants follows the APG IV classification of 2016 (ex. Byng et al. 2018).
Specimens of each species are deposited in the mite collections of Montpellier SupAgro conserved in UMR CBGP INRA/IRD/CIRAD/IA-SupAgro/University of Montpellier.
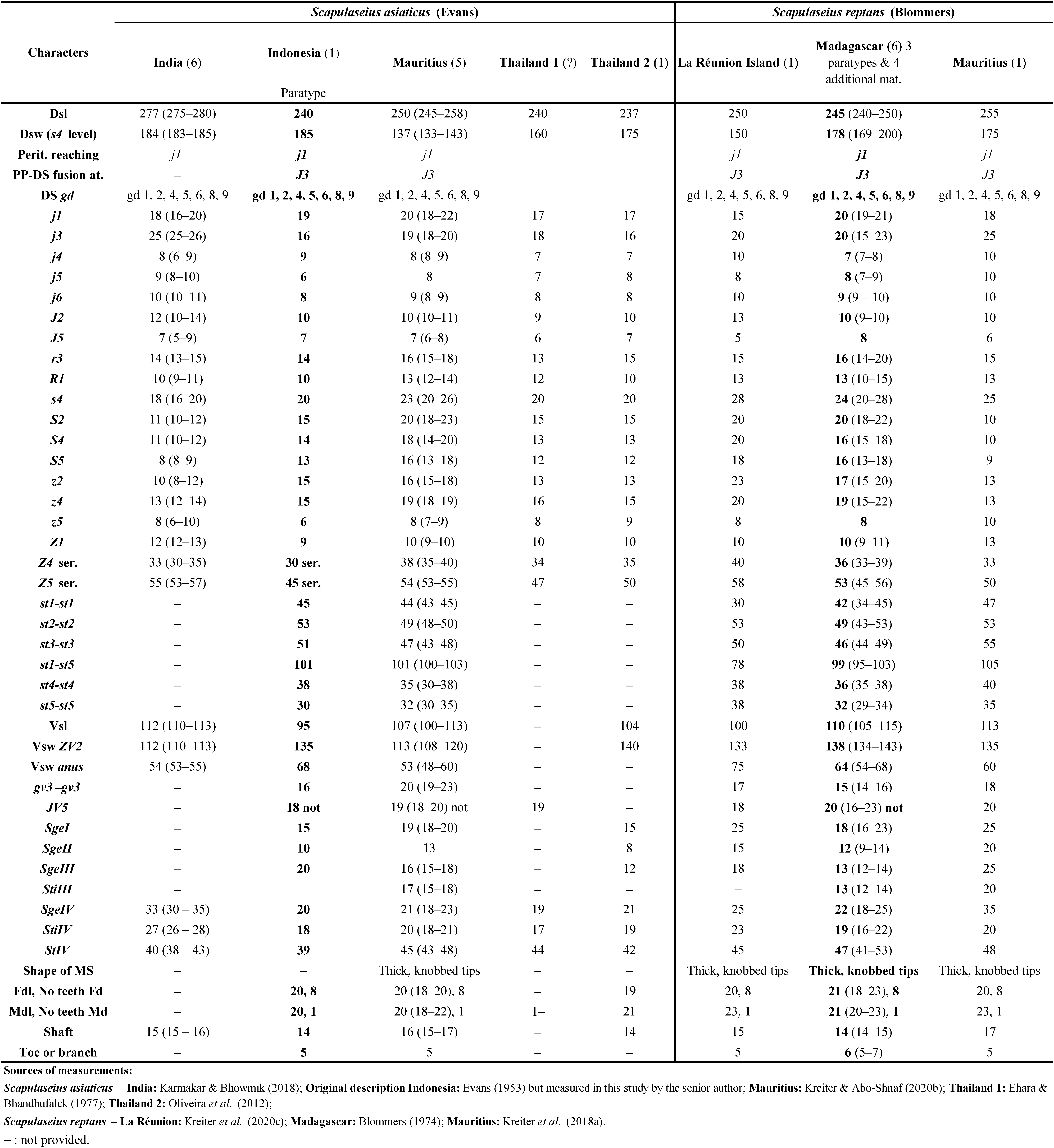
The following abbreviations are used in Tables 1 and 2 for morphological characters: dsl = dorsal shield length just anteriad of j1 to just posteriad of J5 in the middle line; Dsw (s4 level) = dorsal shield width at setae s4 level; Perit. Reaching: peritreme reaching the level of; gd: number of solenostomes; PP-DS fusion at. = Level of the fusion between peritremal plate and dorsal shield; DS gd: nature of solenostomes present on the dorsal shield; Z4 ser or not, Z5 ser. or not = Z4, Z5 serrated or not serrated; gensl = genital shield length; gensw post. cor. = genital shield width posteriorly; lisl = Largest inguinal sigilla (= ''metapodal plate″) length; lisw = Largest inguinal sigilla (= ''metapodal plate″) width; sisl = smallest inguinal sigilla (= ''metapodal plate″) length; sisw = smallest inguinal sigilla (= ''metapodal plate″) width; vsl = ventrianal shield length; gv3 - gv3 = distance between solenostomes gv3 on the ventrianal shield; vsw ZV2 & vsw anus = ventrianal shield width at ZV2 level and at paranal setae level; JV5 ser. or not = JV5 serrated or not; Shape of MS: shape of macrosetae; scl.: calyx length; scw = calyx widest width; Fdl = fixed digit length; Mdl = movable digit length; Nb teeth Fd = number of teeth on the fixed digit; Nb teeth Md = number of teeth on the movable digit; Shaft = length of the shaft of spermatodactyl; toe = length of the toe, the branch of spermatodactyl; BCA = Biological control agents; aasl = altitude above sea level; imm.: immature.
The following abbreviations are used in this paper for institutions: CBGP = Centre de Biologie pour la Gestion des Populations; CIRAD = Centre International de Recherche Agronomique pour le Développement; IA = Institut Agro; INRAE = Institut National de Recherche pour l'Agriculture, l'Alimentation et l'Environnement; IRD = Institut de Recherche pour le Développement; MSA = Montpellier SupAgro, France; UMR = Unité Mixte de Recherche.
Results and discussion
Taxonomy, biological characteristics and biogeography
Tribe Typhlodromipsini Chant & McMurtry
Typhlodromipsini Chant & McMurtry 2005: 318.
Genus Scapulaseius Karg & Oomen-Kalsbeek
Scapulaseius Karg & Oomen-Kalsbeek 1987: 132; Chant & McMurtry 2005: 331, 2007: 65.
Scapulaseius asiaticus (Evans)
Typhlodromus asiaticus Evans 1953: 461.
Typhlodromus (Amblyseius) asiaticus, Chant 1959: 80.
Amblyseius (Typhlodromopsis) asiaticus, Muma 1961: 289.
Amblyseius (Amblyseius) asiaticus, Ehara 1966: 20; Ehara & Bhandhufalck 1977: 58.
Amblyseius asiaticus, Carmona 1968: 280; Gupta, 1975: 32.
Amblyseius (Neoseiulus) asiaticus, Ehara 2002: 127.
Typhlodromips asiaticus, Moraes et al. 1986: 137, 2004b: 207.
Scapulaseius asiaticus, Chant & McMurtry 2005: 335, 2007: 67.
Scapulaseius linearis Corpuz & Rimando 1966: 125 (synonymy according to Schicha & Corpuz-Raros 1992).
Scapulaseius siaki Ehara & Lee 1971: 64 (synonymy according to Ehara & Bhandhufalck 1977).
According to the genus concept by Chant and McMurtry (2005), this species belongs to the asiaticus species group of the genus Scapulaseius as setae R1 are inserted on the lateral margin of the dorsal shield of the adult female. The species group comprises 24 species. Species of the genus Scapulaseius are supposed to belong to the life-type III (McMurtry and Croft 1997; McMurtry et al. 2013), i.e. a polyphagous generalist predator. Evans (1953) has indicated that the specimens collected were feeding in colonies of tetranychid mites. Despite these qualitative observations on the group, the biology of S. asiaticus itself remains totally unknown. The material examined had been collected in Java Island, Indonesia (Evans 1953), in Mauritius Island (Kreiter and Abo-Shnaf 2020b; Ferragut and Baumann 2019) and in Vietnam (Kreiter et al. 2020b).
World distribution Angola, China, Cyprus, Hong Kong, India, Indonesia, Malaysia, Mauritius Island, Philippines, Singapore, Sri Lanka, Thailand, Vietnam.
Specimens examined 33 ♀♀, 6 ♂♂ and 2 imm. in total. INDONESIA (3 ♀♀ and 1 ♂): Bogor (Java) (aasl 265 m on average, 6°21′36″ S, 106°28′48″ E), 1 ♀ (holotype, slide n° 1952-10-10.1), 1 ♂ (paratype male, slide n°1952-10-10-2) and 1 ♀ (paratype, slide n° 1952-10-10.3) on Gossypium hirsutum L. (Malvaceae), XI/1951; MALAYSIA: Kuala Lumpur (aasl 66 m on average, 101°41′36″ E, 3°8′27″ N), 1 ♀ (paratype, slide n° 1952-10-10.4) on an unknown host plant (collected by A. Newsam), 1952; MAURITIUS (23 ♀♀ and 5 ♂♂): Côte d'Or, Village (aasl 443 m, 57°32′21″ E, 20°15′26″ S), 2 ♀♀ on Clibadium surinamense L. (Asteraceae), 28/X/2018; Curepipe, Trou aux cerfs (aasl 593 m, 57°30′47″ E, 20°19′04″ S), 1 ♀ on Rubus apetalus Poiret (Rosaceae), 29/X/2018; Mare aux Vacoas (aasl 572 m, 57°29′59″ E, 20°21′40″ S), 11 ♀♀ on Tibouchina heteromalla Cogniaux (Melastomataceae) and 2 ♀♀ on Litsea monopetala (Roxburgh) Person (Lauraceae), 30/X/2018; Quartier Militaire (aasl 472 m, 57°36′05″ E, 20°19′11″ S), 1 ♀ on Clidemia hirta (L.) D. Don (Melastomataceae), 1/XI/2018; Curepipe, Bld Pasteur (aasl 510 m, 57°31′45″ E, 20°19′21″ S), 1 ♀ on Ageratum conyzoides L. (Asteraceae), 4/XI/2018; Curepipe, Anderson street (aasl 560 m, 57°31′52″ E, 20°19′11″ S), 5 ♀♀ and 3 ♂♂ on Erigeron canadensis (L.) Cronquist (Asteraceae) and 3 ♀♀ and 2 ♂♂ on Sonchus oleraceus L. (Asteraceae), 4/XI/2018; Mare aux Vacoas (aasl 581 m, 57°29′31″ E, 20°22′05″ S), 1 ♀ on Ludwigia octovalvis (Jacquemin) P.H.Raven (Onagraceae), 5/XI/2018; VIETNAM (7 ♀♀ and 2 imm.): Ma, in P1 plot (aasl 63 m, 105°1′28″ E, 21°45′53″ N), 1 ♀ on Chromolaena odorata (L.) King and Robinson (Asteraceae), 9/V/2017; Muoi, in P7 plot (aasl 66 m, 104°38′22″ E, 21°54′51″ N), 1 ♀ on Crassocephalum crepidioides (Bentham) Moore (Asteraceae), 16/V/2017; in P9 plot (aasl 66 m, 104°38′18″ E, 21°54′46″ N), 1 ♀ on an unknown plant support, 18/V/2017; in P11 plot (aasl 66 m, 104°38′14″ E, 21°54′30″ N), 1 imm. on C. odorata (Asteraceae), 31/V/2017; in P15 plot (aasl 66 m, 104°38′46″ E, 21°54′56″ N), 1 ♀ on C. odorata (Asteraceae), 31/V/2017; in P8 plot (aasl 66 m, 104°38′9″ E, 21°54′46″ N), 2 ♀♀ and 1 imm. on Xanthium strumarium L. (Asteraceae), 2/VI/2017; in P13 plot (aasl 66 m, 104°38′46″ E, 21°54′50″ N), 1 ♀ on X. strumarium (Asteraceae), 29/VII/2017.
Scapulaseius reptans (Blommers)
Amblyseius (Amblyseius) reptans Blommers 1974: 145.
Typhlodromips reptans, Moraes et al. 1986: 146; Moraes et al. 2004b: 222.
Scapulaseius reptans, Chant & McMurtry 2005: 335; Chant & McMurtry 2007: 68.
Scapulaseius reptans appears to belong to the ficilocus species group of the genus Scapulaseius as the setae R1 are inserted on lateral integument of adult female and not on dorsal shield (Chant and McMurtry 2005). This species group contains 40 species.
This species is mentioned only from the Indian Ocean area, Madagascar (Blommers 1974), La Réunion (Quilici et al. 2000) and recently Mauritius (Kreiter et al. 2018a). Species of this genus Scapulaseius are supposed to be of type III (McMurtry and Croft 1997; McMurtry et al. 2013), i.e. polyphagous generalist predators. However, the biology of S. reptans remains unknown.
World distribution La Réunion Island, Madagascar, Mauritius Island.
Specimens examined 26 ♀♀ and 9 ♂♂ in total. LA RÉUNION ISLAND (2 ♀♀, 1 ♂ and 1 imm.): Ravine Langevin, Grand-Galet Waterfall (aasl 850 m, 55°21′33″ E, 21°17′47″ S), 1 ♀ and 1 ♂ on Desmodium incanum De Candolle (Fabaceae), 11/12/2016; Petite Île, Piton Bloc, Yébo Luguy farm (aasl 973 m, 55°34′64″ E, 21°18′64″ S), 1 ♀ and 1 imm. on Lantana camara L. (Verbenaceae), 9/1/2017. MADAGASCAR (22 ♀♀ and 7 ♂♂): Tamatave city, 1 ♀ (holotype), 4 ♀♀ and 3 ♂♂ (paratypes), and 8 ♀♀ and 4 ♂♂ (additional material) on Psidium guajava L. (Myrtaceae), 28/VII/1972; Tamatave Ivoloina, 3 ♀♀ (additional material) on Rubus sp., 25/VII/1972, 1 ♀ (additional material) on Pueraria phaseoloides (= javanica) (Roxburgh) Bentham (Fabaceae), 1/VIII/1972; 3 ♀♀ (additional material) on Passiflora foetida L. (Passifloraceae), 5/VII/1972 (2 ♀♀) and 24/VII/1972 (1 ♀), 2 ♀♀ (additional material) on Phaseolus lunatus L. (Fabaceae), 11/II/1972. MAURITIUS ISLAND (2 ♀♀ and 1 ♂): Chamouny (aasl 128 m, 57°27′58.00″ E, 20°28′55.99″ S), 2 ♀♀ and 1 ♂ on an unknown host plant, 21/IX/2017.
Discussion on the possible synonymy
Scapulaseius asiaticus was described by Evans (1953) under the name Typhlodromus asiaticus from specimens collected in Java Island, Indonesia, on cotton Gossypium hirsutum L. (Malvaceae) as indicated on slides of the type material. One female was added from Malaysia, Kwala Lumpur, to this type material (Evans 1953).
The closely related Scapulaseius reptans (Blommers) was described by Blommers in 1974 from Tamatave (Madagascar) under the name Amblyseius (Amblyseius) reptans from specimens collected on Psidium guajava L. (Myrtaceae).
Examination of specimens previously collected
Overall character measurements of females (Table 1) and males (Table 2) collected in various locations (by senior author) are compatible between populations, irrespective of whether they were previously identified as S. asiatcius or S. reptans. Consequently, the morphometrics strongly suggest synonymy.





There are however some discrepancies between our observations and previous descriptions of the two species. In the two descriptions, it is said that:
- dorsal shield is reticulated in the description of S. reptans in the anterior lateral margins and on all the posterior part of the dorsal shield except the central area. Reticulations were not included in the original description of S. asiaticus by Evans (1953), but were illustrated by Ehara and Bhandhulfalck (1977) and by Ferragut and Baumann (2019, using a photograph, their plate 1D);
- Ehara and Bhandhulfalck (1977) pointed out that seta R1 is inserted on a lateral projection of the dorsal shield, a feature that Ferragut and Baumann (2019) disagreed on. Scapulaseius reptans is morphologically very close to S. asiaticus, but with setae R1 indicated by the author as located off the dorsal shield in the description (Blommers 1974). Taking this trait into consideration as an apomorphic character, Chant and McMurtry (2005) placed the two species within different groups, S. asiaticus in the asiaticus species group characterized by having R1 on the dorsal shield and S. reptans in the ficilocus species group with species bearing R1 on the lateral integument. However, in S. asiaticus the position of this seta is variable even among individuals of the same population. Ehara and Bhandhufalck (1977) were the first to mention this variability. Ferragut and Baumann (2019) examined 19 females from Mauritius with the following results: eleven females (58%) had both R1 setae on the dorsal shield, four females (26%) had one setae of the pair on a lateral projection of the shield and the other on the soft integument, and three females (16%) had both setae R1 on the lateral integument. Although the majority of specimens have both or one seta on the dorsal shield, 16%, having both R1 on the soft integument, is not a negligible proportion;
- A peculiar trait in S. asiaticus not mentioned by previous authors and especially by Blommers (1974) for S. reptans is the position of the dorsal solenostome gd3. Ferragut and Baumann (2019) stated that while in females of the family Phytoseiidae, this solenostome is usually located on the peritremal plate, in S. asiaticus, it is on the soft lateral integument, between the peritremal and dorsal shields, posterior to setae r3 and close to the margin of dorsal shield.
Our examination of the material collected in Mauritius and Vietnam (Kreiter et al 2020b) and identified as S. asiaticus and of the material collected in Mauritius (Kreiter et al. 2018a) and in La Réunion (Kreiter et al. 2020c) and identified as S. reptans along with the original descriptions of S. asiaticus and S. reptans shows that:
- Re-examination of our specimens from La Réunion (Kreiter et al. 2020c) and of Mauritius (Kreiter et al. 2018a) point out that the dorsal shields of the two species present exactly the same reticulation as drawn by Blommers (1974) for the description of S. reptans and in Ehara and Bhandhulfalck (1977) for the redescription of S. asiaticus;
- In our 27 specimens females of Mauritius (Kreiter and Abo-Shnaf 2020b), we have 21 females out of 27 (77.8%) with R1 on the dorsal shield, four females / 27 (14.8%) with one of these setae on and the other one off shield and two females / 27 (7.4%) with setae R1 both off shield, compared to above mentioned data of Ferragut and Baumann (2019), respectively. In specimens from Vietnam (Kreiter et al. 2020b), we had four females out of seven (57.1%) with R1 on the dorsal shield, only one female / seven (14.3%) with one of these setae on and the other one off shield and two females / seven (28.6%) with setae R1 both off shield. In the two specimens from Mauritius (Kreiter et al. 2018a), both specimens have R1 on the dorsal shield, and in the two female specimens from La Réunion (Kreiter et al. 2020c), one specimen has both R1 on the dorsal shield, but the other specimens have one seta on and the other one off the dorsal shield.
- solenostomes gd3 are on integument in all our specimens from Mauritius (this study), from Vietnam (Kreiter et al. 2020b), but also on those from Mauritius (Kreiter et al. 2018a) and La Réunion (Kreiter et al. 2020c) previously identified as S. reptans.
Despite the confinement during the Covid-19 pandemic (2020-2021), as we were strongly suspecting that S. reptans is a junior synonym of S. asiaticus, just like Ferragut and Baumann (2019) did, we have however managed to borrow type material (and in case of S. reptans some additional material) of both species in 2021.
Examination of type material from both species
Results of the study of the type material of both sexes of both species (Tables 1 and 2) indicate that:
- Examination of respective female specimens belonging to S. asiaticus and S. reptans shows very little differences in character measurements: j3, z2, s4, S2, S4 and S5 are slightly longer in S. reptans and genital and ventrianal shields longer in S. asiaticus but these details have to be taken cautiously as we have only three specimens of S. asiaticus vs. 22 specimens of S. reptans. The dorsal shields of specimens of the two species present exactly the same reticulation as drawn by Blommers (1974) for the description of S. reptans and in Ehara and Bhandhulfalck (1977) for the redescription of S. asiaticus;
- Examination of respective male specimens belonging to S. asiaticus and S. reptans shows also very little differences in character measurements: the same setae than female, j3, z2, s4, S2, S4 and S5 with in addition Z4, Z5, StIV and the length of ventrianal shield are slightly longer in S. reptans. But these details have also to be taken very carefully as we have only one paratype specimen of S. asiaticus vs. 7 specimens of S. reptans;
- Examination of respective female and male specimens belonging to both species shows only very little differences with specimens previously collected and identified as one of the two species (Tables 1 and 2);
- While type specimen females of S. asiaticus have setae R1 on the dorsal shield, in type specimen females of S. reptans, we have 11 females out of 22 (50%) with R1 on the dorsal shield, six females / 22 (27%) with one of these setae on and the other one off shield and five females / 22 (20%) with setae R1 both off shield, compared to 58, 26 and 16% for Mauritius specimens (Ferragut and Baumann 2019), to 77.8%, 14.8% and 7.4% for other Mauritius specimens (Kreiter and Abo-Shnaf 2020b), and to 57.1%, 14.3%, and 28.6% for Vietnam specimens (Kreiter et al. 2020), respectively. In the two specimens from Mauritius (Kreiter et al. 2018a), both specimens have R1 on the dorsal shield. And in the two female specimens from La Réunion (Kreiter et al. 2020c), one specimen has both R1 on the dorsal shield, but the other specimens have one seta on and the other one off the dorsal shield.
- Solenostomes gd3 are on integument in all specimens of S. asiaticus and all specimens of S. reptans;
- Except for specimens from India and La Réunion (Table 1), specimens of both species have the same number of teeth and the same shape of terminal tips of macrosetae;
- A double-blind identification by one of us with all label hidden conducted to exactly half S. asiaticus and half S. reptans using all available type and collected specimens.
Conclusion on the synonymy
Given this variability in S. asiaticus and S. reptans, we agree with Ferragut and Baumann (2019) that S. reptans represent a junior synonym of S. asiaticus. The holotype of S. reptans has setae R1 on the dorsal shield, whereas many type specimens and additional specimens have setae R1 on or off the shield, or both (asymetry) as indicated before. This character is thus quite variable, even if the majority (< 50%) of specimens of both described taxa have setae R1 on the dorsal shield.
Setal measurements and other morphological features of the specimens collected from Indonesia, Malaysia, Madagascar, La Réunion Island, Mauritius and Vietnam agree well with both, with those of the original description and subsequent redescriptions of S. asiaticus by Ehara and Bhandhufalck (1977), Moraes et al. (2004a), Oliveira et al. (2012), Karmakar and Bhowmik (2018) and Ferragut and Baumann (2019); as well as with the morphological data provided in the original description and the redescriptions of S. reptans given by Kreiter et al. (2018a, 2020c).
Considering all this information and new examination of type and additional material, we can conclude that our specimens from Mauritius (Kreiter et al. 2018a, Kreiter and Abo-Shnaf 2020b) and from La Réunion (Kreiter et al. 2020c) must be all named S. asiaticus. Previous specimens collected in La Réunion Island (Kreiter et al. 2020d) and in Mauritius Island (Kreiter et al. 2018a, Kreiter and Abo-Shnaf 2020b) and previously identified as S. reptans are thus consequently belonging all to the unique valid species, S. asiaticus.
The logical outcome of our findings is that the division of the genus Scapulaseius into two species groups based on the position of setae R1 (on vs off the shield) is not supported. We have only examined two of the species in the genus, but our observations reveal a wide variability in the placement of R1, which is evident in a reduced number of individuals and even on the same specimen. This variability does not seem to be local or restricted to a part of the geographical range of the species. Instead, we have observed the three possible patterns (R1 on the dorsal shield, R1 off the shield, one member of the pair on and the other one off the shield) on females collected in the extremes of the geographical area of distribution, Mauritius Island and Vietnam, and in the type series of S. reptans collected by Blommers in Eastern Madagascar.
Furthermore, in those females with R1 on the shield, the insertion of these setae is marginal and small deviations in the development of the dorsal shield may lead indistinctly to one of the three patterns observed or, as Chant and McMurtry (2005) mentioned for other Scapulaseius species, to the setal placement on a lateral projection of the dorsal shield.
Acknowledgements
Acknowledgements are first due to the Department to which the senior author belongs for his research activities that have funded this work for travels and accommodations in Mauritius: UMR CBGP (Internal call for proposals 2018). I would like to warmly thank the National History Museum (NHM), Department of Life Sciences (Entomology), Cromwell Road, London SW7 5BD, United Kingdom and especially Dr Anne Baker for the loan of the type material of Scapulaseius asiaticus; and the Naturalis Biodiversity Center, Darwinweg 2, 2333 CR Leiden, The Netherlands, especially Wendy van Bohemen for the loan of the type and the additional materials of Scapulaseius reptans. Many thanks to Mrs Marie Anne Edouard for hosting me and to his son for helping me with car problems. I am very grateful to Mrs Claudia Baider, responsible for the Mauritius Herbarium, who has identified some plants and advice on Island locations and Mauritius biodiversity. I would like to thank National Authorities of Mauritius for the signature of a Memorandum of agreement for the supply of biological material by the Government of Mauritius and a Phytosanitary certificate. I must also thank Le Vélo Vert Association, especially Mrs Géraldine d'Unienville for e-mail exchanges and advices.
References
- Athias-Henriot C. 1975. Nouvelles notes sur les Amblyseiini. II. Le relevé organotaxique de la face dorsale adulte (Gamasides protoadéniques, Phytoseiidae). Acarologia, 17(1): 20-29.
- Byng J.W., Smets E.F., van Vugt R., Bidault E., Davidson C., Kenicer G., Chase M.W., Christenhusz M.J.M. 2018. The phylogeny of angiosperms poster: a visual summary of APG IV family relationships and floral diversity. The Global Flora, 4-7.
- Carmona M.M. 1968. Contribuicao para o estudo de alguns acaros fitofagos e depredadores, de Angola. Agron. Lusit., 29: 267-288 + 12 plates.
- Chant D.A. 1959. Phytoseiid mites (Acarina: Phytoseiidae). Part I. Bionomics of seven species in southeastern England. Part II. A taxonomic review of the family Phytoseiidae, with descriptions of thirty-eight new species. Can. Entomol., 61(12): 1-166. https://doi.org/10.4039/entm9112fv
- Chant D.A., McMurtry J.A. 1994. A review of the subfamilies Phytoseiinae and Typhlodrominae (Acari: Phytoseiidae). Intern. J. Acarol., 20(4): 223-310. https://doi.org/10.1080/01647959408684022
- Chant D.A., McMurtry J.A. 2005. A review of the subfamily Amblyseiinae Muma (Acari: Phytoseiidae) Part VII. Typhlodromipsini n. tribe. Intern. J. Acarol., 31(4): 315-340. https://doi.org/10.1080/01647950508683673
- Chant D.A., McMurtry J.A. 2007. Illustrated keys and diagnoses for the genera and subgenera of the Phytoseiidae of the world (Acari: Mesostigmata). Indira Publishing House, West Bloomfield, 219 pp.
- Chant D.A., Yoshida-Shaul E. 1991. Adult ventral setal patterns in the family Phytoseiidae (Acari: Gamasina). Intern. J. Acarol., 17(3): 187-199. https://doi.org/10.1080/01647959108683906
- Chant D.A., Yoshida-Shaul E. 1992. Adult idiosomal setal patterns in the family Phytoseiidae (Acari: Gamasina). Intern. J. Acarol., 18(3): 177-193. https://doi.org/10.1080/01647959208683949
- Corpuz L.A., Rimando L. 1966. Some Philippine Amblyseiinae (Phytoseiidae: Acarina). Philip. Agric., 50: 114-136.
- Demite P.R., Moraes G.J. de, McMurtry J.A., Denmark H.A., Castilho R.C. 2021. Phytoseiidae Database. Available from: www.lea.esalq.usp.br/phytoseiidae (last access 30/VI/2020).
- Demite P.R., McMurtry J.A., Moraes G.J. de. 2014. Phytoseiidae Database: a website for taxonomic and distributional information on phytoseiid mites (Acari). Zootaxa, 3795 (5): 571-577. https://doi.org/10.11646/zootaxa.3795.5.6
- Denmark H.A., Evans G.A. 2011. Phytoseiidae of North America and Hawaii (Acari: Mesostigmata). Indira Publishing House, West Bloomfield, USA, 451 pp.
- Ehara S. 1966. A tentative catalogue of predatory mites of Phytoseiidae known from Asia, with descriptions of five new species from Japan. Mushi, 39: 9-30.
- Ehara S. 2002. Some phytoseiid mites (Arachnida: Acari: Phytoseiidae) from west Malaysia. Species Div., 7: 29-46. https://doi.org/10.12782/specdiv.7.29
- Ehara S., Bhandhufalck A. 1977. Phytoseiid mites of Thailand (Acarina: Mesotigmata). J. Fac. Educ. Tottori University, Nat. Sc., 27(2): 43-82.
- Ehara S., Lee L.H.Y. 1971. Mites associated with plants in Hong Kong. J. Fac. Educ., Tottori Univ., Natur. Sci., 22(2): 61-78.
- Evans G.O. 1953. On some mites of the genus Typhlodromus Scheuten, 1857, from S. E. Asia. Ann. Mag. Nat. Hist., 6: 449-467. https://doi.org/10.1080/00222935308654444
- Ferragut F., Baumann J. 2019. New phytoseiid mites (Mesostigmata: Phytoseiidae) of Mauritius, with the description of two new species. Syst. Appl. Acarol., 24(5): 825-856. https://doi.org/10.11158/saa.24.5.8
- Gupta S.K. 1975. Mites of the genus Amblyseius (Acarina: Phytoseiidae) from India with descriptions of eight new species. Intern. J. Acarol., 1(2): 26-45. https://doi.org/10.1080/01647957508683746
- Karg W., Oomen-Kalsbeek F. 1987. Neue Raubmilbenarten der Gattung Amblyseius Berlese (Acarina, Parasitiformes, Phytoseiidae) Antagonisten der unechten Spinnmilbe Brevipalpus phoenicis Geijskes. Zool. Jahrb. Syst., 114(1): 131-140.
- Karmakar K., Bhowmik S. 2018. Description of eight new species and of four species belonging to the family Phytoseiidae (Acari: Mesostigmata) from West Bengal, India. Zootaxa, 4422(1), 41-77. https://doi.org/10.11646/zootaxa.4422.1.3
- Knapp M., Van Houten Y., Van Baal E., Groot T. 2018. Use of predatory mites in commercial biocontrol: current status and future prospects. Acarologia, 58 (Suppl):72-82. https://doi.org/10.24349/acarologia/20184275
- Kreiter S., Abo-Shnaf R.I.A. 2020a. Phytoseiid mites of Rodrigues Island. Acarologia, 60(2): 449-468. https://doi.org/10.24349/acarologia/20204376
- Kreiter S., Abo-Shnaf R.I.A. 2020b. New records of phytoseiid mites from Mauritius Island (Acari: Mesostigmata). Acarologia 60(3): 520-545. https://doi.org/10.24349/acarologia/20204382
- Kreiter S., Abo-Shnaf R.I.A., Payet R.-M. 2020a. Phytoseiid mites of Mayotte Island (Acari: Mesostigmata). Acarologia, 60(3): 622-642. https://doi.org/10.24349/acarologia/20204391
- Kreiter S., Bopp M.-C., Douin M., Nguyen D.T., Wyckhuys K. 2020b. Phytoseiidae of Vietnam (Acari: Mesostigmata) with description of a new species. Acarologia 60(1): 75-110. https://doi.org/10.24349/acarologia/20204362
- Kreiter S., Fontaine O., Payet R.-M. 2018a. New records of Phytoseiidae (Acari: Mesostigmata) from Mauritius. Acarologia, 58(4): 773-785. https://doi.org/10.24349/acarologia/20184273
- Kreiter S., Payet R.-M., Fillâtre J., Abdou Azali H. 2018b. First records of Phytoseiidae from one island of the Comoros Archipelago. Acarologia, 58(3): 529-545. https://doi.org/10.24349/acarologia/20184256
- Kreiter S., Payet R.-M., Abdou Azali H. 2021a. Phytoseiid mites of Anjouan Island (Acari: Mesostigmata). Acarologia, 61(1): 62-83. https://doi.org/10.24349/acarologia/20214418
- Kreiter S., Payet R.-M., Abdou Azali H. 2021b. Phytoseiid mites of Mohéli Island (Acari: Mesostigmata). Acarologia, 61(1): 94-114. https://doi.org/10.24349/acarologia/20214419
- Kreiter S., Payet R.-M., Mouigni H., Douin M., Tixier M.-S., Abdou Azali H. 2021c. New records of phytoseiid mites (Acari: Mesostigmata) from Grande Comore Island (Comoros Archipelago). Acarologia, 61(2): 241-273. https://doi.org/10.24349/acarologia/20214429
- Kreiter S., Payet R.-M., Abo-Shnaf R.I.A., Douin M. 2021d. New Phytoseiidae (Acari: Mesostigmata) of Mascareignes and Comoros Archipelagos (Indian Ocean): one new record, three new species groups and description of six new species and of six unknown males. Acarologia in press. https://doi.org/10.24349/Krky-e23s
- Kreiter S., Payet R.-M., Douin M., Fontaine O., Fillâtre J., Le Bellec F. 2020c. Phytoseiidae of La Réunion Island (Acari: Mesostigmata): three new species and two males described, new synonymies, and new records. Acarologia 60(1): 111-195. https://doi.org/10.24349/acarologia/20204361
- Lindquist E.E. 1994. Some observations on the chaetotaxy of the caudal body region of gamasine mites (Acari: Mesostigmata), with a modified notation for some ventrolateral body setae. Acarologia, 35: 323-326.
- Lindquist E.E., Evans G.W. 1965. Taxonomic concepts in the Ascidae, with a modified setal nomenclature for the idiosoma of the Gamasina (Acarina: Mesostigmata). Mem. Entomol. Soc. Canada, 47: 1-64. https://doi.org/10.4039/entm9747fv
- McMurtry J.A., Croft B.A. 1997. Life-styles of phytoseiid mites and their roles in biological control. Ann. Rev. Entomol., 42: 291-321. https://doi.org/10.1146/annurev.ento.42.1.291
- McMurtry J.A., Moraes G.J. de, Sourassou N.F. 2013. Revision of the life styles of phytoseiid mites (Acari: Phytoseiidae) and implications for biological control strategies. Syst. Appl. Acarol., 18: 297-320. https://doi.org/10.11158/saa.18.4.1
- Moraes G.J. de, McMurtry J.A., Denmark H.A. 1986. A catalog of the mite family Phytoseiidae. References to taxonomy, synonymy, distribution and habitat. EMBRAPA-DDT, Brasilia, Brazil, 353 pp.
- Moraes G.J. de, Lopes P.C., Fernando C.P. 2004a. Phytoseiid mite (Acari: Phytoseiidae) of coconut growing areas in Sri Lanka, with descriptions of three new species. J. Acarol. Soc. Japan, 13(2): 141-160. https://doi.org/10.2300/acari.13.141
- Moraes G.J. de, McMurtry J.A., Denmark H.A., Campos C.B. 2004b. A revised catalog of the mite family Phytoseiidae. Zootaxa, 434: 1-494. https://doi.org/10.11646/zootaxa.434.1.1
- Muma M.H. 1961. Subfamiles, genera, and species of Phytoseiidae (Acarina: Mesostigmata). Fla St. Mus. Bul., 5(7): 267-302
- Muma M.H., Denmark H.A. 1970. Phytoseiidae of Florida. Arthropods of Florida and neighboring land areas, 6. Florida Department of Agriculture and Consumer Services, Division of Plant Industry, Gainesville, USA, 150 pp.
- Myers N. 1988. Threatened biotas: hostspots in tropical forests. Environmentalist, 8: 187-208. https://doi.org/10.1007/BF02240252
- Myers N., Mittermeier R.A., Mittermeier C.G., Da Fonseca G.A., Kent J. 2000. Biodiversity hotspots for conservation priorities. Nature, 403: 853-858. https://doi.org/10.1038/35002501
- Oliveira D.C., Charanasri V., Kongchuensin M., Konvipasruang P., Chandrapatya A., Moraes G.J. de. 2012. Phytoseiidae of Thailand (Acari: Mesostigmata), with a key for their identification. Zootaxa, 3453: 1-24. https://doi.org/10.11646/zootaxa.3453.1.1
- Quilici S., Ueckermann E. A., Kreiter S., Vayssières J.-F. 2000. Phytoseiidae (Acari) of La Réunion Island. Acarologia, 41(1-2): 97-108.
- Rowell H.J., Chant D.A., Hansell R.I.C. 1978. The determination of setal homologies and setal patterns on the dorsal shield in the family Phytoseiidae (Acarina: Mesostigmata). Can. Entomol., 110: 859-876. https://doi.org/10.4039/Ent110859-8
- Schicha E., Corpuz-Raros L.A. 1992. Phytoseiidae of the Philippines. Indira Publishing House, West Bloomfield, Michigan, USA, 190 pp.
- Walter D.E., Krantz G.W. 2009. Collecting, rearing and preparing specimens. In: Krantz G.W., Walter D.E. (eds) A manual of Acarology, 3rd ed. Texas Tech University Press, Lubbock. 807 pp.



2021-07-26
Date accepted:
2021-09-29
Date published:
2021-09-30
Edited by:
Tsolakis, Haralabos

This work is licensed under a Creative Commons Attribution 4.0 International License
2021 Kreiter, Serge and Ferragut, Francisco
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



