Oldest determined record of a mesostigmatic mite (Acari: Mesostigmata: Sejidae) in Cretaceous Burmese amber
Joharchi, Omid  1
; Vorontsov, Dmitry D.2
and Walter, David Evans3
1
; Vorontsov, Dmitry D.2
and Walter, David Evans3
1✉ Institute of Environmental and Agricultural Biology (X-BIO), Tyumen State University, Tyumen, Russia.
2Institute of Developmental Biology, Russian Academy of Sciences, Moscow, Russia.
3School of Science, Technology and Engineering, University of the Sunshine Coast, 90 Sippy Downs Drive, Sippy Downs, Queensland 4556, Australia.
2021 - Volume: 61 Issue: 3 pages: 641-649
https://doi.org/10.24349/goj5-BZmsOriginal research
Keywords
Abstract
Introduction
The Sejida (= Sejina) can be subdivided into two superfamilies: Heterozerconoidea and Sejoidea, the latter comprising four families (Sejidae, Ichthyostomatogasteridae, Uropodellidae, Reginacharlottiidae), ten genera and over 60 described species of mites generally associated with woody substrates, and often with deutonymphs that are phoretic on insects (Walter 2013; Lekveishvili and Klompen 2004). The fossil record of Mesostigmata is surprisingly sparse. Only nine fossil families from the lineages Gamasina and Uropodina (Mesostigmata: Monogynaspida) have been recorded and no valid fossils of the seemingly more early derivative Sejida and Trigynaspida have been found (Dunlop et al. 2018; Sidorchuk 2018). Koch and Berendt (1854) reported a sejid fossil species from Baltic amber, but Dunlop et al. (2018) recently determined that it was a misinterpretation of an unknown species of cohort Anystina (Acariformes: Prostigmata). All recorded fossil mesostigmatans are from the Cenozoic period [from Palaeogene (Eocene, ca. 44–49 Ma) to Neogene (Miocene, ca. 16 Ma)] and among them only three species were named formally (Dunlop et al. 2013; Dunlop et al. 2014; Dunlop et al. 2018; Sidorchuk 2018). Mesostigmata (both Gamasina and Uropodina) are listed from a number of Cretaceous amber localities, beginning with the Lower Cretaceous Lebanese amber (Rasnitsyn et al. 2016), but neither of them was described. Here, we illustrate and describe two deutonymphs from mid-Cretaceous Burmese amber (Kachin) that belong to the Sejida (= Sejina) family Sejidae (superfamily Sejoidea). They thus become the oldest described mesostigmatic mites.
Material and methods
The piece of Cretaceous amber containing the fossil mites described herein was mined in the Hukawng Valley (Kachin State, northern Myanmar). The amber locality is radiometrically dated to 98.79 ± 0.62 Ma based on U–Pb zircon dating of the volcanoclastic matrix and shown to be earliest Cenomanian in age (Shi et al. 2012; Smith and Ross 2018; Zhang et al. 2018; Yu et al. 2019).
Amber preparation
The amber piece containing the mite was cut from a larger piece of amber and fine-polished following the protocols of Sidorchuk (2011, 2013) and tools described by Sidorchuk and Vorontsov (2018). The piece was shaped as a parallelepiped ca. 0.5 x 0.5 x 0.25 mm (Figure 1A). We polished the piece from six sides, trying to leave not more than 100 µm of amber between the mites and the surface in the dorso-ventral direction, as the thicker layer of semi-transparent amber did not let us see enough details while the close polishing allowed us to use high-resolution water and oil immersion optics. From the six available views, only three were found useful: dorsal, ventral and one of the lateral views.



Imaging
To image the mites two compound microscopes, a Nikon E-800 with water (40x and 60x) and oil (100x) immersion optics and a Zeiss AxioImager A2 with oil immersion optics (100x) were used, with brightfield illumination and differential interference contrast (DIC). To image deeper into the piece, in addition to the standard technique with immersion oil separated from the amber by a coverslip, we used a saturated fructose solution, as described in Khaustov et al. (2021). Stacks of images, comprising multiple focal planes (up to 200 in a single stack), were obtained with an Olympus OM-D E-M10-II digital camera. Figures herein contain images assembled from multiple focal planes using Helicon Focus 7.5 software, using algorithms A and B with subsequent manual addition of significant details from the individual focal planes. Some of the resulting original stacks are available through Figshare (https://doi.org/10.6084/m9.figshare.14707077 ![]() ). We also provide a link to the source image stacks of the type material studied by Dunlop et al., (2018): https://doi.org/10.6084/m9.figshare.c.4066367
). We also provide a link to the source image stacks of the type material studied by Dunlop et al., (2018): https://doi.org/10.6084/m9.figshare.c.4066367 ![]() .
.
Terminology
The nomenclature used for anatomical structures follows Evans and Till (1979).
Material and repository
The piece containing the two mites is housed in the Öhm-Kühnle collection maintained permanently at the A.A. Borissiak Paleontological Institute, Russian Academy of Sciences (PIN RAS), Moscow. The piece of amber is stored in a plastic vial filled with water; thymol and myramistin (benzyl dimethyl [3-(myristoilamino) propyl] ammonium chloride) are added to the water to limit fungal and bacterial growth. Due to the minute size of the piece (0.5 mm), extreme care is needed when manipulating it.
Two mites (catalog number PIN 5620-26, inclusions E and F) lie very close to each other in a thin layer of darkened amber (probably, at the edge of amber layers) (Figure 1). Each mite is curved (L- shaped), with the dorsal side outwards (see Figure 1B, lateral view). Between the mites there is a structure of presumably plant origin, resembling stellate hairs (leaf trichomes) (Figure 1C). The amber is full of microscopic particles and bubbles that obscure the view of mites.
Arthropod syninclusions that were present in the original piece along with the mites:
PIN 5620-26 A Hymenoptera Scelionidae
PIN 5620-26 B, C, D Diptera Ceratopogonidae
PIN 5620-26 G Thysanoptera Adiheterothripidae
Systematic paleontology
Parasitiformes Reuter, 1909
Mesostigmata G. Canestrini, 1891
Sejida Kramer, 1885
Sejoidea Berlese, 1885
Sejidae Berlese, 1885
Description
Deutonymph (n= 2)
(Figures 1–5).
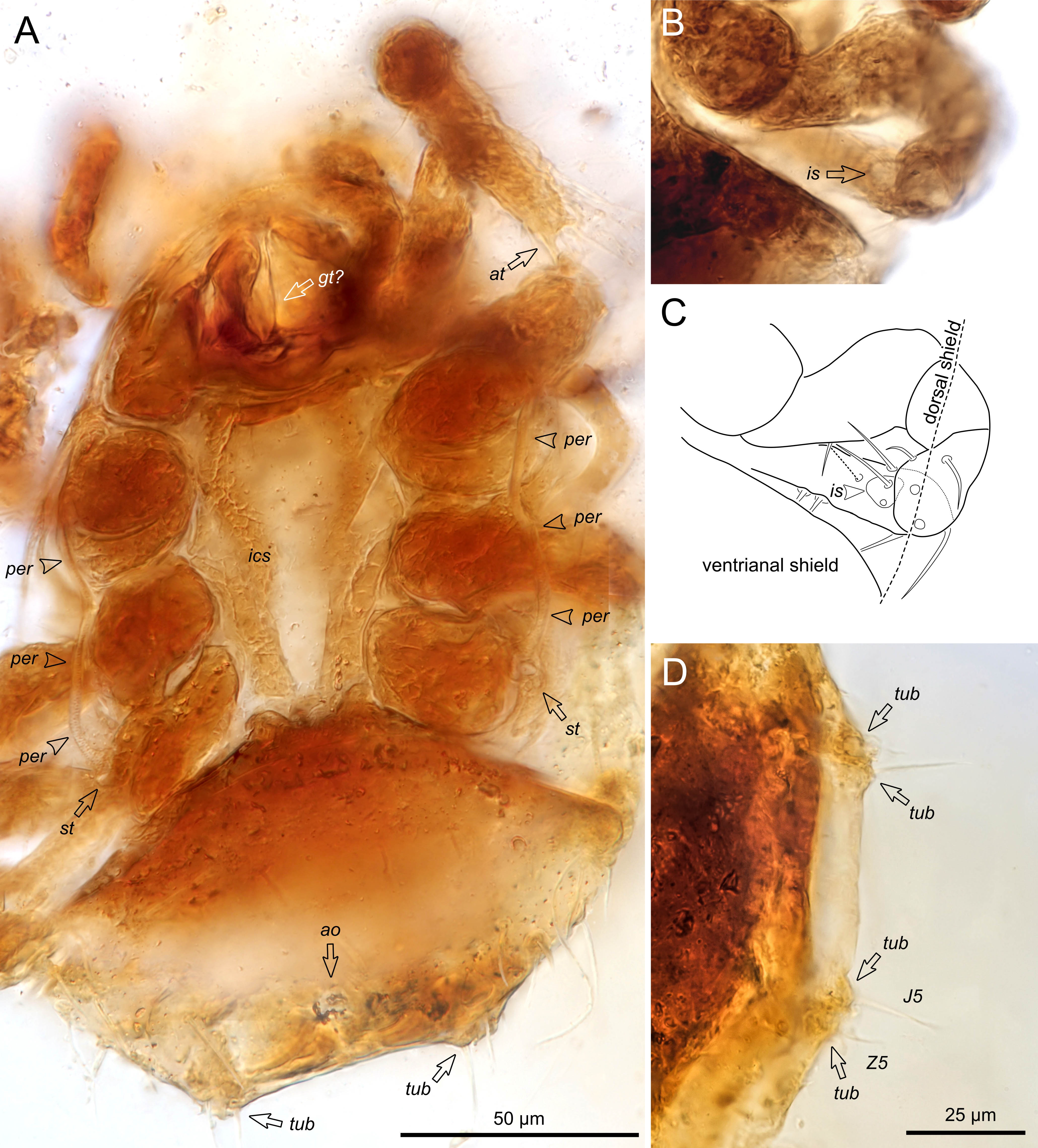











Dorsal Idiosoma — Oval in outline, dorsal surface smooth, without any distinct ornamentation; podonotal, mesonotal and pygidial shields indistinct, all dorsal setae smooth and relatively long, setae J5/Z5 insert on a small tubercles posteriorly (Figure 2A, D).
Ventral idiosoma — Tritosternal laciniae free, base expanded laterally, anchor-shaped (Figure 3A–D), presternal shields absent, intercoxal shield present, somewhat sclerotized, bearing sternal setae st1-4, ventrianal shield interlocks with intercoxal shield (Figure 4A, B), anal opening distinct posteriorly (Figure 2A), peritreme on ventral margin of shield extending from between coxae III–IV to mid-level of coxa II (Figures 2A and 4A), peritrematic shields absent.
Gnathosoma — (Figure 3A–D). Chelicerae short and robust, digits indistinct, corniculi appressed anteriorly and flattened, gnathotectum indistinct.
Legs — Leg setae smooth and simple, a distal subdivision (apicotarsus or acrotarsus) and ambulacrum of tarsus I present (Figure 5 A–D); tarsus IV with an intercalary sclerite bearing pair of ventral setae (av4, pv4) (Figure 2B, C).
Remarks
The mites are both deutonymphs because of the lack of genital shields and presence of an intercoxal shield bearing four pairs of setae. This species is assigned to the Sejidae (Mesostigmata: Sejida) by the following combination of characters: (1) a distal subdivision (apicotarsus or acrotarsus) and ambulacrum of tarsus I present (Figure 5 A–D), (2) tarsus IV with an intercalary sclerite that bears a pair of ventral setae (av4, pv4) (Figure 2B, C), (3) dorsal setae J5/Z5 insert on small tubercles (Figure 2A, D), (4) tritosternum with a broad base, anchor-shaped (Figure 3A, B). Unfortunately, we were unable to see the modification of anterior hypostomal setae (h1) as an apomorphic character (it is typically membranous and often highly reduced in Sejidae), nevertheless the above-mentioned diagnostic character states present enough arguments to consider these mites members of Sejidae. We are unable to claim a certain genus for two reasons: (1) modern genera are based mostly on adult females and immature stages have not been well studied, (2) sejid genera are not well studied or resolved. For example, here we would need adult females to check whether the sternal platelets bearing st1 and st2 and the posterior mesonotal shields are fused together or not (neither are fused in Sejus). The small posterior tubercles associated with setae J5/Z5 support an association with the genus Sejus, which usually have similar but larger tubercles in the adults, to which we tentatively assign these specimens. The genus Adenosejus Lekveishvili & Krantz is unlikely because of the absence of thorn-like leg setae; the bifurcate gnathotectum characteristic of Epicroseius Berlese cannot be seen; however, an unknown modern or ancient genus cannot be ruled out in this poorly studied group.
Discussion
Fossil mesostigmatid mites are extremely rare. A few cases of Mesostigmata had been reported from the Cretaceous (e.g. Poinar 1998), but none of them have been formally confirmed as Mesostigmata. Our present discovery of the family Sejidae from Burmese amber is significant for representing the oldest and first determined example of the Sejida from the Cretaceous. Deutonymphs and adults of many families of Mesostigmata have established close phoretic relationships with other arthropods, and phoresy by deutonymphs is a common phenomenon among modern Sejidae, typically on beetles associated with dead wood, and it is possible that this pair of mites were dislodged from an insect that was ensnared by the amber. However, we were unable to see an anal disk or a remnant pedicel, and the only insects in this amber piece are very small, so how these mites became entrapped remains a mystery.
Acknowledgements
We cordially thank Christoph Öhm-Kühnle (Herrenberg, Germany) for donating the amber specimen from his collection to PIN RAS, which allowed us to study it. We are grateful to Alexander Rasnitsyn and Alexey Shmakov (PIN RAS) for identification of syninclusions. We express our sincere appreciation to Owen D. Seeman (Queensland Museum, South Brisbane, Queensland, Australia) and another anonymous reviewer for their invaluable comments on an earlier draft of this paper.
References
- Berlese, A. 1885. Acarorum systematis specimen. Bollettino della Società Entomologica Italiana, 17: 121-135.
- Canestrini G. 1891. Abbozzo del sistema Acarologico. Atti 1st. Ven, 38: 699-725.
- Evans G.O., Till W.M. 1979. Mesostigmatic mites of Britain and Ireland (Chelicerata: Acari-Parasitiformes). An introduction to their external morphology and classification. Transactions of the Zoological Society of London, 35: 145-270. https://doi.org/10.1111/j.1096-3642.1979.tb00059.x
- Dunlop J.A., Kontschán J., Zwanzig, M. 2013. Fossil mesostigmatid mites (Mesostigmata: Gamasina, Microgyniina, Uropodina), associated with longhorn beetles (Coleoptera: Cerambycidae) in Baltic amber. Naturwissenschaften, 100: 337-334. https://doi.org/10.1007/s00114-013-1031-8
- Dunlop J.A., Kontschán J., Walter D.E., Perrichot V. 2014. An ant associated mesostigmatid mite in Baltic amber. Biology Letters, 10(9): 20140531. https://doi.org/10.1098/rsbl.2014.0531
- Dunlop J.A., Walter D.E., Kontschán J. 2018. A putative fossil sejid mite (Parasitiformes: Mesostigmata) in Baltic amber re-identified as an anystine (Acariformes: Prostigmata). Acarologia, 58: 665-672. https://doi.org/10.24349/acarologia/20184263
- Khaustov A.A., Vorontsov D.D., Perkovsky E.E., Lindquist E.E. 2021. Review of fossil heterostigmatic mites (Acari: Heterostigmata) from late Eocene Rovno Amber. I. Families Tarsocheylidae, Dolichocybidae and Acarophenacidae. Systematic & Applied Acarology, 26(1): 33-61. https://doi.org/10.11158/saa.26.1.3
- Koch C.L., Berendt G.C. 1854. Die im Bernstein befindlichen Myriapoden, Arachniden und Apteren der Vorwelt. In: Berendt G.C. (Ed.) Die in Bernstein befindlichen organischen Reste der Vorwelt gesammelt in Verbindung mit mehreren bearbeitetet und herausgegeben 1. Berlin, Nicolai, 124 pp.
- Kramer P. 1885. Uber Halarachne Halichoeri Allm. Zeitschrift für die Gesammten Naturwissenschaften, 58: 46-74.
- Lekveishvili M., Klompen H. 2004. Phylogeny of infraorder Sejina (Acari, Mesostigmata). Zootaxa, 629: 1-19. https://doi.org/10.11646/zootaxa.629.1.1
- Poinar Jr G.O. 1998. Fossils explained 22: Palaeontology of amber. Geology Today, July-August 1998: 154-160. https://doi.org/10.1046/j.1365-2451.1998.014004154.x
- Rasnitsyn A.P., Bashkuev A.S., Kopylov D.S., Lukashevich E.D., Ponomarenko A.G., Popov Y.A., Rasnitsyn D.A., Ryzhkova O.V., Sidorchuk E.A., Sukatsheva I.D. 2016. Sequence and scale of changes in the terrestrial biota during the Cretaceous (based on materials from fossil resins). Cretaceous Research, 61: 234-255. https://doi.org/10.1016/j.cretres.2015.12.025
- Reuter E. 1909. Zur Morphologie und Ontogenie der Acariden mit besonderer Berücksichtigung von Pediculopsis graminum. Acta Soc. Sci. Fenn., 36: 1-288. https://doi.org/10.5962/bhl.title.4705
- Shi G., Grimaldi D.A., Harlow G.E., Wang J., Wang J., Yang M., Lei W., Li Q., Li X., 2012. Age constraint on Burmese amber based on U-Pb dating of zircons. Cretaceous Research 37, 155 e163. https://doi.org/10.1016/j.cretres.2012.03.014
- Sidorchuk E.A. 2011. Preparation of six-sided micro-samples of Baltic amber for study of organismal inclu¬sions. In: Kostyashova, Z.V. (Eds.), Amber Mining and Processing in Sambia, International Symposium Materials,12-14 May 2010, Kaliningrad, pp. 47-53.
- Sidorchuk, E.A. 2013. New technique for p reparation of small-sized amber samples with application to mites. In: Azar D., Engel M.S., Jarzembowski E., Krogmann L., Nel A., Santiago-Blay J.A. (Eds.), Insect evolution in an Amberiferous and Stone Alphabet. Proceedings of the 6th International Congress on Fos¬sil Insects, Arthropods and Amber; Brill: Leiden-Boston. pp. 189-201.
- Sidorchuck, E.A. 2018. Mites as fossils: forever small?. International Journal of Acarology, 44(8): 349-359. https://doi.org/10.1080/01647954.2018.1497085
- Sidorchuk E.A., Vorontsov D.D. 2018. Preparation of small-sized 3D amber samples: state of the technique. Palaeoentomology, 1: 80-90. https://doi.org/10.11646/palaeoentomology.1.1.10
- Smith R.D.A., Ross A.J., 2018. Amberground pholadid bivalve borings and inclusions in Burmese amber: implications for proximity of resin-producing forests to brackish waters, and the age of the amber. Earth and Environmental Science Transactions of the Royal Society of Edinburgh, 107 (2-3): 239-247. https://doi.org/10.1017/S1755691017000287
- Walter D.E. 2013. A new genus and family of sejine mites (Acari, Parasitiformes, Mesostigmata, Sejoidea) based on new species from Lord Howe Island and Brazil, and a redescription of Sejus americanus (Banks, 1902). Zootaxa, 3691: 301-323. https://doi.org/10.11646/zootaxa.3691.3.1
- Yu T.T., Kelly R., Mu L., Ross A., Kennedy J., Broly P., Xia F.Y., Zhang H.C., Wang B., Dilcher D. 2019. An ammonite trapped in Burmese amber. Proceeding of the National Academy of Sciences of Unites States of America, 116 (23): 11345-11350. https://doi.org/10.1073/pnas.1821292116
- Zhang Q., Rasnitsyn A.P., Wang B., Zhang H.C. 2018. Hymenoptera (wasps, bees and ants) in mid-Cretaceous Burmese amber: A review of the fauna. Proceedings of the Geologists' Association, 129: 736-747. https://doi.org/10.1016/j.pgeola.2018.06.004



2021-06-10
Date accepted:
2021-08-18
Date published:
2021-09-14
Edited by:
Faraji, Farid

This work is licensed under a Creative Commons Attribution 4.0 International License
2021 Joharchi, Omid; Vorontsov, Dmitry D. and Walter, David Evans
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



