New records of phytoseiid mites from Italy, with description of a new species and a redescription of other two (Parasitiformes, Phytoseiidae)
Tsolakis, Haralabos1 and Ragusa, Salvatore2
1✉ University of Palermo, Department of Agricultural Food and Forest Sciences, Laboratory of Applied Acarology "Eliahu Swirski", Edifice 5A, Viale delle Scienze, 13 – 90128 Palermo, Italy.
2University of Palermo, Department of Agricultural Food and Forest Sciences, Laboratory of Applied Acarology "Eliahu Swirski", Edifice 5A, Viale delle Scienze, 13 – 90128 Palermo, Italy.
2020 - Volume: 60 Issue: 4 pages: 735-752
https://doi.org/10.24349/acarologia/20204399ZooBank LSID: 7550FED8-09B9-4955-AFEC-1ABDAFBBA08F
Original research
Keywords
Abstract
Introduction
The family Phytoseiidae (Acari, Parasitiformes) being predatory, plays an important regulation role both in agricultural and natural ecosystems (McMurtry et al. 2013, 2015; Calvo et al. 2015; Lorenzon et al. 2018), and includes about 2,521 valid species (Demite et al. 2020). Many of them are considered important biocontrol agents of phytophagous mites and insects on crops, but despite more than a half century of intensive studies on this family and the great number of species described, only 20 species are presently reared in commercial insectaries (Knapp et al. 2018). It should be mentioned that this number has significantly grown in the last two decades with the increasing number of studies on bio-ethology, food preferences and ecology of less known or rare species (Quilici et al. 1997; Kreiter et al. 2002; Sznajder et al. 2011; Szabó et al. 2013; Tsolakis et al. 2016; Tsolakis and Ragusa 2017). The use of indigenous phytoseiid species, to control infestations of both endemic and invasive mite and insect pests, still represents the lower environmental impact strategy in agricultural ecosystems, and this should be the main scope of specialists in this field. It is commonly accepted that periodic surveys of phytoseiid mites on cultivated and spontaneous plants, are important to discover some rare species that could become future biocontrol agents of phytophagous mites on agricultural crops. As a matter of fact, it should be mentioned that the most common phytoseiids reared in insectaries world wide, i.e., Phytoseiulus persimilis Athias-Henriot, Neoseiulus cucumeris (Oudemans), Neoseiulus californicus (McGregor), Amblyseius swirskii Athias-Henriot, are rare species in natural ecosystems. Studies on phytoseiid mites in Italy, started in the 19th century by Canestrini and Fanzago (1876) and Berlese (1887, 1889), and continued by the latter author and Ribaga up to the first two decades of 20th century (Ribaga 1904; Berlese 1923). Afterwards, after a block of about 50 years, the studies on phytoseiid mites were resumed in a constant way starting from the 70s up to today (Ivancich-Gambaro 1975; Ragusa and Swirski 1976, 1978, 1982; Ragusa 1979, 1981; Ragusa and Paoletti 1985; Duso and Sbrissa 1990; Tsolakis and Ragusa Di Chiara 1993; Ragusa Di Chiara and Tsolakis 1996; Tsolakis and Ragusa 1999; Tsolakis et al. 2000, 2013; Duso et al. 2004; Tsolakis and Ragusa 2015, 2017; Lorenzon et al. 2018). At present, ninety-one valid phytoseiid species have been surveyed in Italy and 18 of them, were described as new species (Demite et al. 2020).
In the present paper we report nine new phytoseiid records from the Italian fauna, we describe a new species of Neoseiulus and redescribe two rare species, adding new information on them.
Materials and methods
Phytoseiid mites reported in the present paper were collected in different localities of Italy with the branch-shaking method (Tsolakis and Ragusa 1999). Specimens, fallen on a black plastic table, were collected using micro-aspirator and preserved in alcohol 70%. In laboratory they were cleared in Nesbitt solution and mounted on microscopic slides in Hoyer's medium. For the identification of the species and measurements, a differential interference contrast microscope (DIC) Zeiss Axioplan was used. Specimens of all species are kept in the Acari collection of the Laboratory of Agricultural and Applied Acarology ''Eliahu Swirski'', Department of Agricultural, Food and Forest Sciences, University of Palermo (Italy). All measurements are given in micrometres (μm), reporting mean (min-max) and were made using the Axiovision 40V 4.6.1.0 application (Zeiss, 2002–2007). In the description of the new species, and the redescriptions we followed the nomenclature proposed by Lindquist & Evans (1965) as adapted to the Phytoseiidae by Rowell et al. (1978) for the dorsal and Chant and Yoshida-Shaul (1991) for the ventral chaetotaxy. For the terminology of various sections of the insemination apparatus (spermathecal apparatus), we followed Beard (2001), with some additions and changes. For setal patterns of leg IV podomeres (genu, tibia, tarsus), the formulae proposed by Evans and Till (1979) were adopted. For the macrosetae, we considered the concept defined by Beard (2001). Nomenclature of the adenotaxy was the one suggested by Athias-Henriot (1975). The following descriptive terms were here adopted after Athias-Henriot (1977): Habitus = a cluster of characters that determine the appearance of a structure, i.e., the shape of the dorsal shield, its reticulation (strong, slight, absent), the position of the setae, the shape of some setae, and isotrichy or anisotrichy; Hoplochorous = situated on a shield; Tylochorous = situated on a microsclerite, a small platelet on interscutal membrane; Gymnochorous = situated on the interscutal membrane; Holoadeny = presence of all dorsal solenostomes reported up to now for the Phytoseiidae family; Meriadeny = absence of one or more solenostomes; Receptaculum = the proximal segment of the major duct after the opening between the 3rd and the 4th pair of coxae, it may be simple or differentiate, in the latter case it is defined receptaculum; Embolus = the dimple present in the atrium on which the minor duct is inserted. The identification of the host plants was based on Pignatti (1982) and on the direct contribution of different botanists of the University of Palermo. For some species, we referred to various recent papers on Sicilian flora (Gianguzzi and La Mantia 2009; Musarella et al. 2018).
Results
All the phytoseiid species of the present work belong to our Acari collection. Various slides were labelled with the generic name followed by sp. (i.e., Neoseiulus spp.). Reorganising the collection, we classified them in their correct taxon status and we described the new species reported below.
Subfamily Amblyseiinae Muma 1961
Tribe Neoseiulini Chant and McMurtry 2003
Genus Neoseiulus Hughes 1948
Neoseiulus mediterraneus Tsolakis & Ragusa sp. nov.
ZOOBANK: 2AAC66F1-7B31-406D-98EA-3789093D5470 ![]()
Diagnosis of female
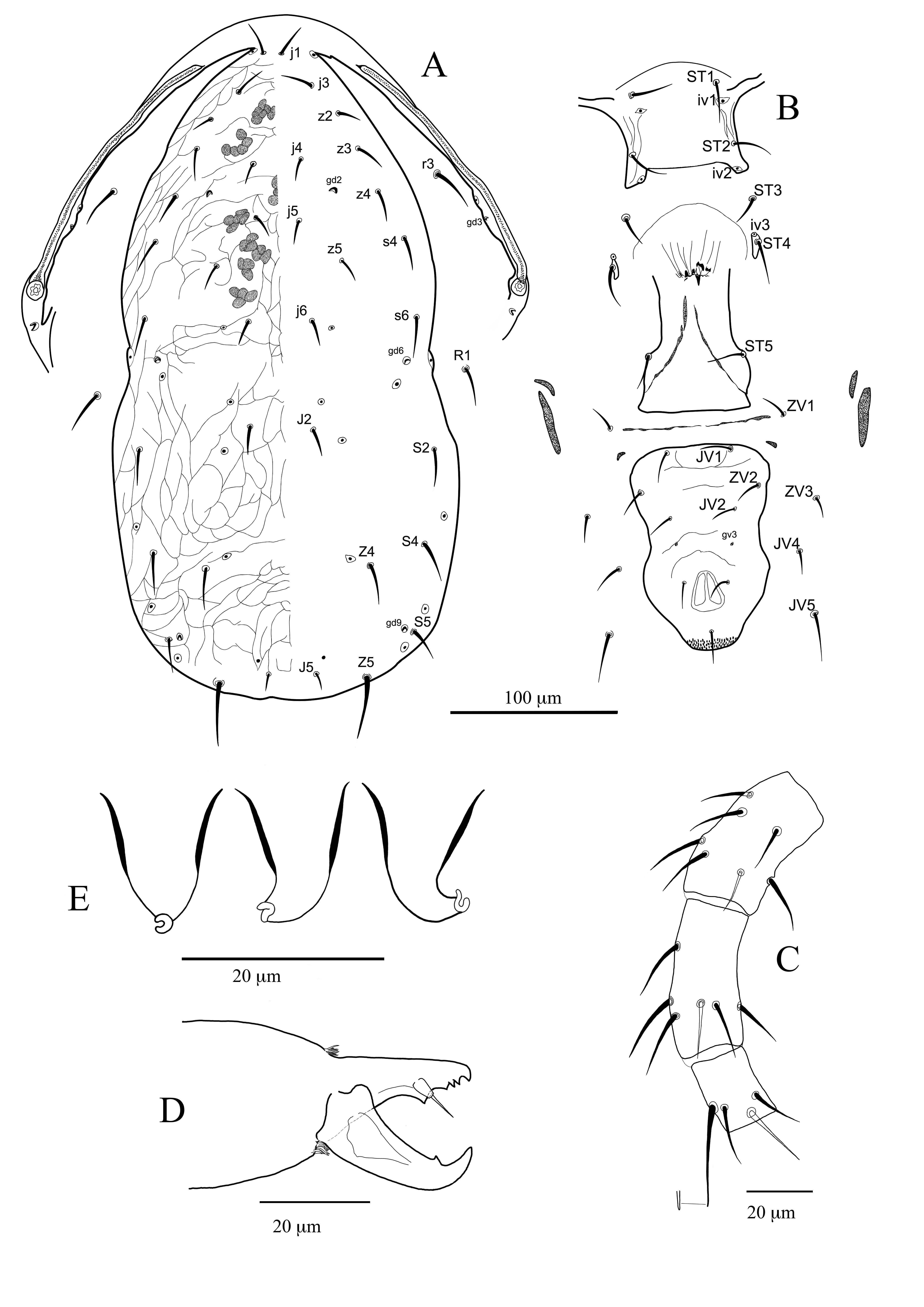
Dorsal shield slightly reticulated with a restriction at level of setae R1. Dorsal and peritrematal shields are fused at level of setae j1. All dorsal setae are slender and smooth except setae Z5 which are slightly serrated and the longest. Adenotaxy complete; solenostome gd3 well visible on peritrematal shield. Peritreme reaches the bases of setae j1. Sternal shield smooth with three pairs of setae (ST1–ST3) and two pairs of poroids (iv1 and iv2). Setae ST4 and poroids iv3 tylochorous. Genital shield with longitudinal striae; sigilla 1st–3rd pairs well visible. Sigilla of 4th and 5th pair well visible on the interscutal membrane between setae ZV1; sigilla of 6th pair posteroparaxial to setae ZV1. Ventrianal shield mostly reticulated with three pairs of preanal setae and a pair of small crateriform solenostomes (gv3). Setae JV5 smooth. Calyx of the insemination apparatus cup-shaped, without basal neck. Atrium nodular; minor duct well visible. Major duct cylindrical and narrow. Fixed digit of chelicera with seven teeth, movable digit with two teeth. Nine setae on genu II; three smooth and pointed macrosetae are present on leg IV, being of that on basitarsus the longest one.
Female
The description is based on 16 females.
(Figure 1 A–F)



Dorsum (Figure 1A) — Dorsal shield oval, waisted at level of setae R1, mostly reticulate. Seven pairs of small crescentic solenostomes are visible on the dorsal shield: gd1 posteroantiaxial to setae j3, gd2 posteroantiaxial to setae j4, gd4 posteroantiaxial to setae s4, gd5 posteroparaxial to setae z5, gd6 anteroparaxial to Z1, gd8 anteroantiaxial or anterior to Z4 and gd9 anteroparaxial to S5. Some poroids are visible on the dorsal shield. Some sigilla (muscle marks) are visible on the shield (Figure 1A). The dorsal setae s4, S2, Z4 almost of the same length. Setae Z5 is the longest one. Same length have also the dorsocentral setae j1, j4 and j6. Setae r3 and R1 on the interscutal membrane. All setae are slender and smooth, except for Z5, thicker and slightly serrated. Measurements of dorsal and sublateral setae are as follows: mean (min-max) (mean of setae of the holotype are given in square brackets after the measurements of other specimens): j1 21 (19–24) [21], j3 35 (31–38) [34], j4 20 (19–23) [19], j5 17 (15–18) [16], j6 21 (19–23) [19], J2 25 (22–27) [26], J5 11 (9–14) [10], z2 27 (25–30) [27], z4 34 (31–37) [32], z5 18 (17– 20) [18], s4 49 (44–54) [52], Z1 28 (24–31) [29], Z4 48 (43–52) [52], Z5 61 (52–65) [64], S2 44 (41–47) [45], S4 30 (25–32) [31], S5 31 (24–35) [34], r3 31 (27–35) [31], R1 25 (21–27) [25]. Length of dorsal shield (j1-end of shield) 325 (303–345) [319], width at level of setae s4 171 (162–184) [171], width at level of setae S2 191(172–199) [185] and width at level of setae S4 183 (170–200) [176].
Peritreme (Figure 1A) — Apex of peritreme reaches the bases of setae j1; 195 (183–202) [193] long. Solenostome gd3 sometimes visible on peritrematal shield at level of setae z4. Solenostome gdp crescentic, posterior to the opening of the stigma (Figure 1A).
Ventral idiosoma (Figure 1B) — Sternal shield almost smooth; a slight reticulation is visible in some specimens. Posterior margin straight, anterior arms visible. Setae ST1, ST2 and ST3 inserted on the shield. Setae ST4 tylochorous. Poroids iv1 and iv2 hoplochorus; poroids iv3 tylochorous (Figure 1B). Length of sternal shield (ST1-ST3) 60 (55–64) [61], width (ST1–ST1) 53 (49–56) [52], width (ST3–ST3) 68 (65–71) [70]. Epigynial shield with longitudinal striae, flask shaped with a straight posterior margin. Genital sigilla (1–3 pairs) well visible on majority of specimens. Poroids iv4 on the interscutal membrane posteroantiaxial to st5. Genital sigilla of 4th and 5th pair well visible between bases of setae ZV1. Sigilla of 6th pair (sgpa) on the interscutal membrane, posteroparaxial to setae ZV1. Ventrianal shield (VAS) subpentagonal, waisted at level of the solenostomes gv3, and allover reticulated; three pairs of setae (JV1, ZV2 and JV2), besides circumanal setae, are present on VAS. Solenostomes gv3 well visible posteroparaxially to setae JV2. Distance between solenostomes gv3 25 (18–30) [24]. Ratio of distances JV2-JV2/gv3-gv3 1.9 (1.7-2-2) [1.9]. (Length of VAS 113 (100–120) [113], widths: at level of ZV2 85 (76–89) [79], at level of JV2 80 (68–86) [75], at level of gv3 76 (67–82) [69], at level of paranal setae [70 (65–73) [65]. Ratio length/width VAS at level of ZV2 1.3 (1.2–1.4) [1.4], at level of JV2 1.4 (1.2–1.5) [1.5]. Four pairs of setae JV4, JV5, ZV1 and ZV3 on interscutal membrane. Setae JV5 smooth and the longest of the setae surrounding the VAS, 38 (35–42) [38]. Anterior inguinal sigilla (metapodal platelets), 15 (13–19) [15] long; posterior inguinal sigilla 23 (20–26) [23] long and 6 (4–6) [5] width (Figure 1B).
Insemination apparatus (Figure 1C) — Major duct cylindrical, narrow and slightly sclerotised. About 1.5 times the length of the calyx. Receptaculum slightly enlarged, not always visible. Atrium nodular, free and separate from the calyx. Embolus well visible; minor duct thin, clearly visible. Calyx broadbell shaped (cup-shaped), thick walled all over, without basal neck or stalk 14 (12–15) [13] long and 12 (9–15) [14] wide. The membranous vesicle is well visible.
Chelicerae (Figure 1D) — Fixed cheliceral digit, 25 (23–26) [24] long, commonly with seven teeth plus the apical tooth (80% of specimens); few specimens with six teeth and only one with eight teeth. Pilus dentilis evident. Movable digit with two teeth plus the apical one, 29 (26–31) [28] long.
Legs (Figure 1E, F) — Ten setae are presente on genu of leg I (2 2/1 2/1 2). Nine setae are present on genu of leg II (2 2/1 2/1 1). Seven setae are presente on genua of leg III (1 2/1 2/0 1) and IV (1 2/1 2/0 1). Three smooth and pointed macrosetae are present on leg IV: Sge IV 32 (26–38) [34], Sti IV 24 (20-27) [23] and St IV 40 (36-42) [40].
Distribution and location of types
Holotype female and one paratype female (No. 8656A) collected on Cirsium spp. (Asteraceae) at Dadia National Park (41°6'0.49'' N, 26°13'19.29'' E, m 124 a.s.l.), Evros (Greece) 16 Aug. 2016; one paratype female collected on the same data as the holotype. Ten paratype females collected on Cirsium spp. (Asteraceae) at Ficuzza (37°53'03.0'' N, 13°23'23.7'' E, m 708 a.s.l.), Palermo (Italy) 1 Nov. 1980. Two paratype females (No. S 544 A) collected on lichens at Ficuzza (37°53'00.4'' N, 13°23'05.1'' E, m 675 a.s.l.) Palermo (Italy) 24 Oct. 1980. One paratype female collected on Clinopodium nepeta (L.) Kuntze (Lamiaceae), at Vena di Maida (38°53'39.97'' N, 16°24'26.82'' E, m 157 a.s.l.), Catanzaro (Italy) 15 Sept. 1989. Type materials deposited in the Acari collection of the Laboratory of Applied Acarology ''Eliahu Swirski'', Department of Agricultural, Food and Forest Sciences, University of Palermo (Italy).
Etymology
The name ''mediterraneus'' refers to the Mediterranean countries where the type materials were found.
Remarks
Various slides with specimens of the new species were kept in the above mentioned Acari collection and labelled as Amblyseius sp. near umbraticus. Putting in order the collection and after the discovery of some specimens from Greece, we decided to describe the new species, considering the characters that differentiate it from the other nearer species.
Neoseiulus mediterraneus belongs to the cucumeris species group as defined by Chant and McMurtry (2003). Although it has been demonstrated that the above mentioned group is polyphyletic (Tsolakis et al. 2012), we adopted this taxonomy because no reorganization of this group has been proposed yet. Among the species included in the cucumeris species group, the new species resembles two other palearctic species, Neoseiulus umbraticus and N. pseudoumbraticus. Chant and Yoshida-Shaul (1982) described the latter species from specimens collected in Algeria, distinguishing it from N. umbraticus for some discrete characters: a different chaetotactic formula of genu II, presence/absence of pilus dentilis, different lengths of peritreme and number of macrosetae on leg IV (see Table 1). Neoseiulus mediterraneus shows the ancestral chaetotactic formula of genu II (2-2/1-2/1-1) among the above mentioned species, as well as for the whole cucumeris species group. Other ancestral characters are the distance between solenostomes gv3, greater in N. mediterraneus, the length of peritreme, longer in the new species, and a shorter macroseta on basitarsus IV.
Tribe Amblyseiini Muma 1961
Subtribe Proprioseiopsina Chant & McMurtry 2004
Genus Proprioseiopsis Muma 1961
Proprioseiopsis dacus (Wainstein 1973)
Amblyseius (Amblyseius) dacus Wainstein (1973) (original designation)
Amblyseius dacus Gilyarov et al. (1977)
Proprioseiopsis (Amblyseiulus) dacus Karg (1989)
Specimens examined — Two females at Ficuzza (Palermo), (37°53'06.9'' N, 13°23'33.2'' E, m 732 a.s.l.), on Rubus spp. (Rosaceae) 24 Oct. 1980.
Remarks — The species has been reported up to now only for Moldova (Wainstein 1973) and Iran (Faraji et al. 2007). The latter authors redescribed the species from one female collected on Citrus leaves. They reported some discordances with the original description regardings the lengths of various dorsal setae and the presence of a tooth on the movable digit that Wainstein (1973) considered edentate. In both our specimens a tooth is present on the movable digit, but in one of them it is vestigial. Wainstein described the species from only one specimen and probably he did not notice it or it was actually absent in the holotype. In the original description more than three teeth were reported on fixed digit, while Faraji et al. (2007) reported three teeth. On both our specimens we found four teeth (three between pilus dentilis and apical tooth and one beyond pilus dentilis). Waintein (1973) reported ''seven pairs of small and large pores'' on dorsal shield, while (Faraji et al. 2007) reported ten small visible pores. Evidently, the latter authors did not distinguish between solenostomes and poroids. In our specimens we verified the presence of seven small, but crataeriform solenostomes (gd1, gd2, gd4, gd5, gd6, gd8, gd9). Our measurements fit well with Waintein's original description, except for the setae j1 and z2, but also with Faraji's and co-authors' redescription (see Table 2). Proprioseiopsis dacus is reported in Italy for the first time.



Subfamily Typhlodrominae Wainstein 1962
Tribe Typhlodromini Wainstein 1962
Genus Typhloseiulus Chant & McMurtry 1994
Typhloseiulus subsimplex (Arutunjan, 1972)
Seiulus subsimplex Arutunjan (1972) (original designation)
Seiulus (Seiulus) subsimplex Beglyarov (1981)
Typhlodromus subsimplex Chant & Yoshida-Shaul (1983)
Specimens examined — One female at Palazzo Adriano (Palermo), (37°40'23.03'' N, 13°22'0.50'' E, m 671 a.s.l.), on Quercus gr. pubescens Willd. (Fagaceae) 29 Sept. 2007.
Remarks — The species have been reported up to now only for Armenia. This is the first record from Italy.
Genus Neoseiulella Muma 1961
Neoseiulella carmeli (Rivnay & Swirski 1980)
Typhloctonus carmeli Rivnay & Swirski (1980) (original designation)
Typhlodromus carmeli Chant & Yoshida-Shaul (1989)
Neoseiulella (Typhloctona) carmeli Denmark & Rather (1996)
Specimens examined — 36 ♀♀, 7 ♂♂ and 1 immature in total. All specimens were collected in the island of Pantelleria at the following places: Aeroporto (36°49'11.31'' N, 11°58'21.80'' E, m 158 a.s.l.), 4♀♀ and 1♂ on Vitis vinifera L. (Vitaceae) 29 May 1994; Contrada (C.da) Bue Marino (36°49'28.14'' N, 11°58'43.75'' E, m 77 a.s.l.), 1♀ on Capparis spinosa L. (Capparaceae) 28 May 1994; C.da Campobello (36°49'17.36'' N, 11°59'2.72'' E, m 51 a.s.l.), 1♀ on Mioporum spp. (Scrophulariaceae) 29 May 1994; C.da Cannachi (36°48'0.44'' N, 11°57'58.34'' E, m 230 a.s.l.), 1♀ on Parietaria spp. (Urticaceae) 28 May 1994; C.da Khufura (36°47'56.14'' N, 11°58'17.79'' E, m 241 a.s.l.), 2♀♀ on Malus domestica Borkh. (Rosaceae) 28 May 1994; C.da Scirafi (36°47'40.40'' N, 11°57'6.86'' E, m 149 a.s.l.), 20♀♀, 2♂♂ and 1 immature on Tamarix spp. (Tamaricaceae) 04 Oct. 1994; C.da Suachi (36°47'52.54'' N, 11°56'7.24'' E, m 13 a.s.l.), 1♀ on Lycopersicon esculentum Mill. (Solanaceae) 04 Oct. 1994; C.da Tracino (36°46'38.79'' N, 11°59'52.82'' E, m 738 a.s.l.) 1♀ on Quercus ilex L. (Fagaceae) 05 Oct. 1994; Calami (36°47'11.69'' N, 11°57'1.82'' E, m 63 a.s.l.), 1♀ on Vitis vinifera L. (Vitaceae) 04 Oct. 1994; Cuddia Attalora (36°47'14.44'' N, 11°59'44.78'' E, m 574 a.s.l.), 2♀♀ on Mentha spp. (Lamiaceae) 05 Oct. 1994; Cuddia Attalora (36°47'12.98'' N, 12°0'8.61'' E, m 648 a.s.l.), 1♀ and 2♂♂ on Lavandula stoechas L. (Lamiaceae) 05 Oct. 1994; Lago di Venere (36°48'59.93'' N, 11°59'0.05'' E, m 17 a.s.l.), 1♀ on Cynodon dactylon (L.) Pers. (Poaceae) 29 May 1994; Lago di Venere (36°48'56.95'' N, 11°59'21.25'' E, m 5 a.s.l.), 1♂ on Capparis spinosa L. (Capparaceae) 29 May 1994; Pineta Montagna grande (36°47'43.75'' N, 11°59'8.92'' E, m 346 a.s.l.), 1♂ on Pistacia lentiscus L. (Anacardiaceae) 28 May 1994.
Remarks — Neoseiulella carmeli has been reported up to now only from Israel (Rivnay and Swirski 1980; Swirski and Amitai 1997). The species in the island of Pantelleria was wide spread both on cultivated and wild plants. The island is nearer to the African continent than to Sicily, where we never found this species in more than 50 years of constant surveys.
Genus Typhlodromus Scheuten 1857
Subgenus Anthoseius De Leon 1959
Typhlodromus (Anthoseius) jordanis Rivnay & Swirski 1980
Anthoseius jordanis Rivnay & Swirski (1980) (original designation)
Amblydromella jordanis Moraes et al. (1986)
Amblydromella (Aphanoseia) jordanis Denmark & Welbourn (2002)
Specimens examined — Two females and one immature collected in the island of Pantelleria (Trapani) (36°47'40.40'' N, 11°57'6.86'' E, m 149 a.s.l.), on Tamarix spp. (Tamaricaceae) 04 Oct. 1994.
Remarks — As the previous species, Typhlodromus (A.) jordanis has been reported up to now only from Israel (Rivnay and Swirski 1980). We found this species on Tamarix spp. at the island of Pantelleria, but we never found it in Sicily. No information is available on the bio-ecology of this species.
Typhlodromus (Anthoseius) singularis Chant 1957
Typhlodromus singularis Chant (1957) (original designation)
Typhlodromus (Typhlodromus) singularis Chant (1959)
Neoseiulus singularis Muma (1961)
Mumaseius singularis De Leon (1965)
Anthoseius singularis Charlet & McMurtry (1977)
Amblydromella singularis Moraes et al. (1986)
Amblydromella (Lindquistoseia) singularis Denmark & Welbourn (2002)
Neoseiulus polonicus (Willmann 1949) (synonymy by Athias-Henriot 1960)
Female
The redescription is based on seven females.
(Figure 2 A–E)


Dorsum (Figure 2A) — Dorsal shield almost ovoid, waisted at level of solenostome gd6 and weakly ornamented almost all over. Three pairs of crescentic solenostomes are visible on the dorsal shield: gd2 posteroantiaxial to setae j4 and posteroparaxial to setae z3, gd6 posteroparaxial to s6 and gd9 anteroantiaxial to S5. Some poroids are visible on the dorsal shield and sigilla on the podonotal region (Figure 2A). The mediolateral (z3, z4), lateral (s4-S5) and marginal (r3, R1) setae have similar lengths (Table 3). All setae are slender and smooth. Setae Z5 are the longest one 36 (32–40). Setae S4 anteriorly to the level of setae Z4. Solenostome gd9 near to the bases of setae S5 8 (7–10). Measurements of dorsal and sublateral setae are as follows: j1 20 (20–21), j3 20 (18–21), j4 14 (12–15), j5 15 (14–15), j6 16 (15–16), J2 17 (16–19), J5 10 (9–12), z2 14 (13–15), z3 19 (18–21), z4 19 (18–20), z5 15 (13–16), s4 21 (21–22), s6 24 (21–25), Z4 25 (23–27), Z5 36 (32–40), S2 22 (20–24), S4 23 (21–26), S5 19 (18–21), r3 21 (20–22), R1 20 (19–21). Length of dorsal shield (j1-end of shield) 381 (366–389), width at level of setae s4 174 (168–178), width at level of setae s6 180 (176–183), width at level of setae S2 194 (188–206) and width at level of setae S4 197 (190–207).



Peritreme (Figure 2A) — Apex of peritreme reaches the bases of setae j3, in some specimens between j3-j1. Solenostome gd3 and poroid id3 well visible on peritrematal shield. Solenostome gdp crescentic, posterior to the opening of the stigma. Length of peritreme 171 (156–188).
Ventral idiosoma (Figure 2B) — Sternal shield smooth with lateral margins slightly striated. Setae ST1 and ST2 inserted on the shield. Setae ST3 gymnochorous, setae ST4 tylochorous. Poroids iv1 and iv2 hoplochorus; the latter on the internal part of the projection of the shield. The posterior margin of the shield almost straight; poroids iv3 tylochorous (Figure 2B). Distances between setae ST1–ST1 52, ST2–ST2 63 and ST1–ST2 39. Epigynial shield smooth, with a straight posterior margin and lateral identations at the posterior end of genital sigilla (1–3rd pairs). Distance between setae st5, 58. Poroids iv4 on the interscutal membrane near the basis of the shield. Genital sigilla (4th and 5th pairs) well visible. Sigilla of 6th pair (sgpa) on the interscutal membrane, posteroparaxial to setae ZV1. In one specimen the right sigillum was on the anterior margin of the ventrianal shield. Ventrianal shield (VAS) almost sub-pentagonal, elongate, waisted at level of solenostomes gv3, and slightly striate; three pairs of setae (JV1, ZV2 and JV2), besides circumanal setae are present on VAS. Solenostomes gv3 small, posteriorly or light posteroparaxial to setae JV2. Length of VAS 121 (114–126), widths: at level of ZV2 87, at level of JV2 75 (71–78) and at level of paranal setae 76 (75–78). Ratio length/width VAS at level of ZV2 1.4, at level of JV2 1.6. Four pairs of setae, JV4, JV5, ZV1 and ZV3 on interscutal membrane. Setae JV5 smooth, 27 long. Anterior inguinal sigilla (metapodal platelets), 16 (15–18) long; posterior inguinal sigilla 37 (34–41) long and 5 (4–5) wide (Figure 2B).
Insemination apparatus (Figure 2E) — Major duct short (4 long) and membranous. Receptaculum not visible. Atrium differentiated, slightly larger in comparison to the major duct (Ø 2), inserted at the basis of the calyx. A prominent ring enfolds the embolus; minor duct rarely visible. Calyx campanulate, membranous at the basis for about 1/3 of its length, thick walled afterwards, 13 long, 11 wide at the distal part and 9 at the mean distance. The membranous vesicle is commonly visible.
Chelicerae (Figure 2D) — Fixed cheliceral digit 25 (24–25) long, with four teeth plus the apical one; three anteriorly and one posteriorly to pilus dentilis. Movable digit with one tooth plus the apical one, 27 long.
Legs (Figure 2C) — Ten setae are present on genu of leg I. Seven setae are present on genua of the remaining legs. A smooth, short and blunted macroseta is present on basitarsus of leg IV, St IV 29 (28–31). Lengths of genu IV 48 (46–50), tibia IV 48 (44–51) and basitarsus 25 (23–26).
Specimens examined
Cogne (Valle D'Aosta), (45°36'24.94'' N, 7°20'51.67'' E, m 1549 a.s.l.), 1♀ and 1♂ on Larix decidua Mill. (Pinaceae) 06 Aug. 1997; La Visaille (Valle D'Aosta), (45°47'31.29'' N, 6°55'9.67'' E, m 1696 a.s.l.), 2♀♀ on Larix decidua Mill. (Pinaceae), and 1♀ on Salix caprea L. (Salicaceae) 11 Aug. 1997; Pont Serrand (Valle D'Aosta), (45°42'40.43'' N, 6°55'29.60'' E, m 1684 a.s.l.), 1♀ and 1♂ on Larix decidua Mill. (Pinaceae) 14 Aug. 1997; Degioz (Valle D'Aosta), (45°35'28.09'' N, 7°12'21.01'' E, m 1570 a.s.l.), 2♀♀ on Sorbus aucuparia L. (Rosaceae), 09 Aug. 1997.
Remarks
In the original description (Chant 1957) the author reported only measurements of dorsal and ventral shields, presence of a tooth on the movable digit and of three teeth on the fixed one and no macrosetae on leg IV. Afterwards, Schuster and Pritchard (1963) redescribed the species from a single specimen from California, reporting some measurements and a clear drawing of the insemination apparatus. The above authors noted longer anterolateral setae in their specimen, a 34 long macroseta on basitarsus IV and a short and thick peritreme (reaching the level of setae z4). About a decade after, Chant et al. (1974) redescribed the species, adding measurements of some dorsal setae and giving information of some other discrete characters: chaeotaxic formulae of genua II and III, presence of a short macroseta on basitarsus IV, of three pairs of solenostome on dorsal shield (from the description we individuated them as gd2, gd6 and gd9), giving a schematic drawing of the insemination apparatus and reporting the presence of solenostome gv3 (Table 3). The above authors reported also a significant variation on peritreme's length and lengths of macrosetae on basitarsus IV, among specimens collected in various Canadian regions. They noted that in specimens with a short peritreme (apex reached at level of setae z4), an indistinguishable macroseta on basitarsus IV is present, while in specimens with a longer peritreme (apex at level of setae j3) there was a conspicuous macroseta on basitarsus IV, but they did not report any measurements of it. Chant et al. (1974) also noted that this species was collected also in the United States, but no remarks on Schuster and Pritchard redescription are mentioned. Congdon (2002) measured a specimen found on Tsuga heterophylla (Raf.) Sarg. (Pinaceae) in western Washington, reporting that this specimen matches the redescription given by Chant et al. (1974) in most characters, but it is intermediate between the specimen described by Schuster and Pritchard (1963) and Chant et al. (1974) as far as lengths of various dorsal setae are concerned (Table 3). Similarly, our specimens are intermediate between specimens reported in the latter two references and well match the specimen measured by Congdon (2002), except for the peritreme's length (Table 3). The species was, up to now, reported only from the Nearctic region (Canada and USA). This is the first record from the Palearctic region. For its distribution see Demite et al. (2020).
Subgenus Typhlodromus Scheuten 1857
Typhlodromus (Typhlodromus) knisleyi Denmark 1992
Typhlodromus knisleyi Denmark (1992) (original designation)
Female
The redescription is based on the holotype with additions based on four specimens.
(Figure 3 A–E)
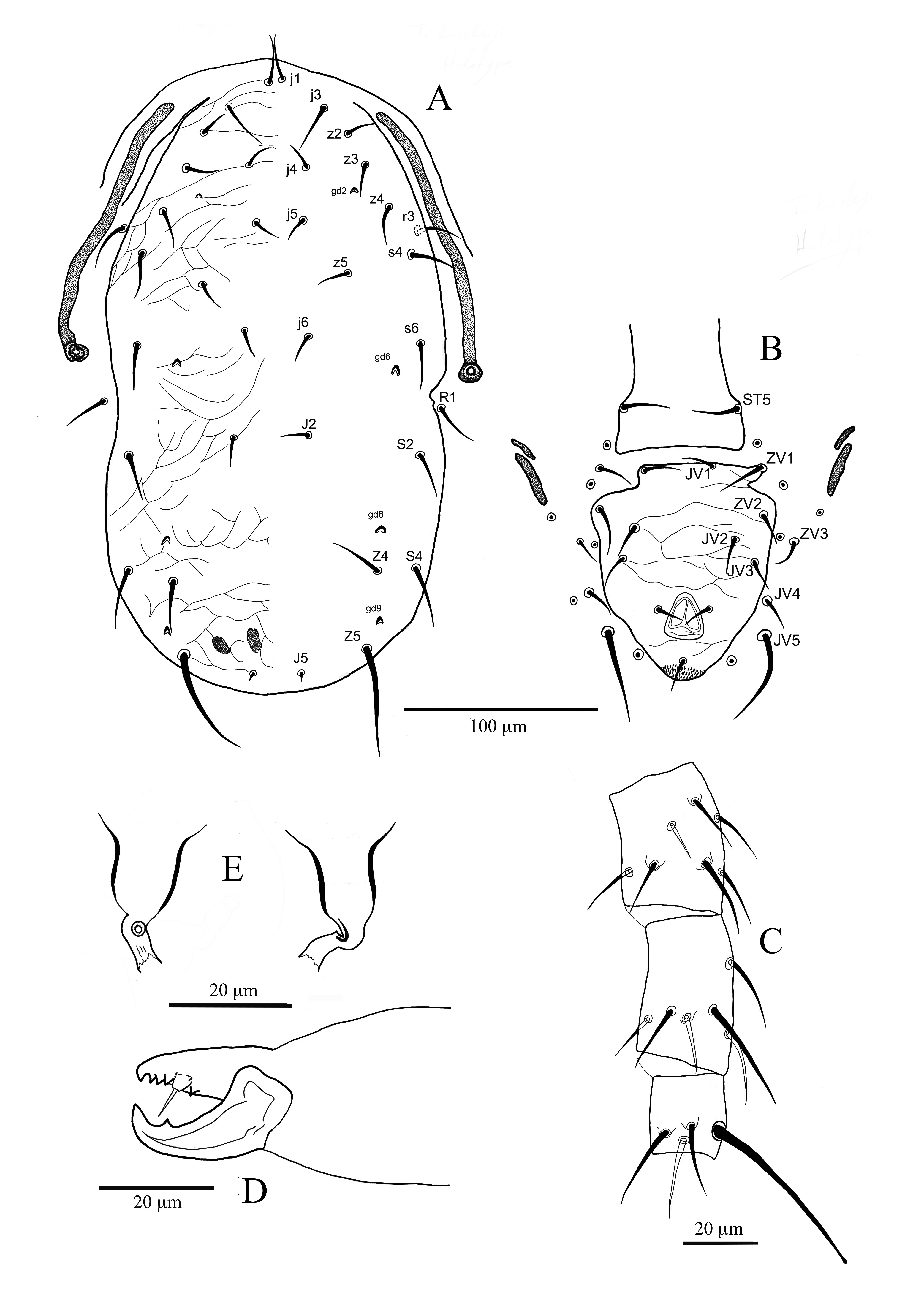


Dorsum (Figure 3A) — Dorsal shield almost oblong, rounded at the anterior and posterior levels, slightly waisted at level of setae R1 and weakly ornamented almost all over. Four pairs of crescentic solenostomes are visible on the dorsal shield: gd2 posteroantiaxial to setae j4 and posteroparaxial to setae z3, gd6 posteroparaxial to s6, gd8 anteroantiaxial or anterior to Z4 and gd9 anteroantiaxial to Z5. No poroids are visible on the dorsal shield and only sigilla saXIII and saXIV are visible on the shield (Figure 12). The dorsal setae j1 and j3 of the same length (23); the remaining dorsocentral setae, except J5, almost of the same length (14–17) (Table 3). Similar lengths have the series z setae (15–19), while the anterolateral series s4 and s6 are quite longer (22 and 25 respectively). The longest setae on the dorsal shield are Z5 (57). Setae r3 and R1 on the interscutal membrane, of equal length (21). All setae are slender and smooth. Measurements of dorsal and sublateral setae of Sicilian specimen are as follows (in parentheses our measurements of the holotype and in brackets measurements reported in the original description): j1 21 (23) [24], j3 29 (23) [27], j4 16 (14) [16], j5 17 (14) [14], j6 19 (15) [17], J2 20 (17) [17], J5 6 (6) [5], z2 15 (17) [17], z3 23 (19) [19], z4 23 (18) [20], z5 16 (15) [16], s4 27 (23) [24], s6 29 (22) [24], Z4 33 (28) [28], Z5 59 (57) [58], S2 30 (25) [25], S4 35 (32) [32], r3 24 (21) [21], R1 21 (21) [22]. Length of dorsal shield (j1-end of shield) 312 (318), width at level of setae s4 162 (171), width at level of setae s6 170 (164), width at level of setae S2 172 (164) and width at level of setae S4 159 (159).
Peritreme (Figure 3A) — Apex of peritreme reaches anteriorly setae z2 (between setae j3 and z2). Solenostome gd3 not visible on peritrematal shield. Solenostome gdp crescentic, posterior to the opening of the stigma. Peritreme 139 (140) long.
Ventral idiosoma (Figure 3B) — Sternal shield smooth with margins not well visible. Setae ST1 and ST2 inserted on the shield. Setae ST3 and ST4 tylochorous. Poroids iv1 and probably iv2 hoplochorus, the posterior margin of the shield is not well visible, but in our specimen iv2 is hoplochorus; poroids iv3 tylochorous. Length of sternal shield is not reported because of the bad position in the holotype. In our specimen distances between setae ST1–ST1, ST2–ST2 and ST1–ST2 are respectively 48, 57 and 30. Epigynial shield smooth, with a straight posterior margin. Genital sigilla not visible. Length between setae ST5, 55. Poroids iv4 on the interscutal membrane near the basis of the shield. Genital sigilla of 4th and 5th pair not visible in the holotype. Sigilla of 6th pair (sgpa) on the interscutal membrane, posteroparaxial to setae ZV1. The right ZV1 on a protrusion of the ventrianal shield. Ventrianal shield (VAS) almost triangular or sub-pentagonal, slightly waisted at level of setae JV2, and slightly reticulated; four pairs of setae (JV1-JV3 and ZV2), besides circumanal setae are present on VAS. Absence of the solenostomes gv3. The VAS of the holotype shows a lack of sclerotisation anteriorly to setae ZV2 in the left part of the scutum and a protrusion in the right part which included the seta ZV1. Length of VAS 106 (113), widths: at level of ZV2 97 (95), at level of JV2 89 (91), at level of JV3 83 (87), at level of paranal setae 70 (74). Ratio length/width VAS at level of ZV2 1.2, at level of JV2 1.2. Four pairs of setae, JV4, JV5, ZV1 and ZV3 on interscutal membrane. Setae JV5 smooth and the longest of the setae present on the ventral part, 48 (48). Anterior inguinal sigilla (metapodal platelets), 11 (15) long; posterior inguinal sigilla 26 (28) long and 5 (5) width (Figure 13).
Insemination apparatus (Figure 3E) — Major duct short and slightly sclerotised, 6 long and 3 wide. Receptaculum of the same width of the major duct. Atrium differentiated, slightly larger in comparison to the major duct (Ø 4); a light incidence distinguishes it from the calyx. A prominent ring enfolds the embolus; minor duct thin, poorly visible. Calyx campanulate, membranous at the base, thick walled afterwards, 16 long, 15 wide at the distal part and 11 at the mean distance. The membranous vesicle is always visible.
Chelicerae (Figure 3D) — Fixed cheliceral digit 25 long, with four teeth plus the apical tooth; three anteriorly and one posteriorly to pilus dentilis. Movable digit with one tooth plus the apical one, 29 long.
Legs (Figure 3C) — Ten setae are present on genu of leg I. Seven setae are present on genua of the remaining legs. Three smooth and pointed macrosetae are present on leg IV; on Sge and Sti macrosetae are pointed, on basitarsus slightly knobbed: Sge IV 24 (22), Sti IV 27 (33) and St IV 49 (51).
Specimens examined
One female at Piano Zucchi (Palermo), (37°53'54.1'' N, 13°59'38.4'' E, m 1066 a.s.l.), on Quercus spp. (Fagaceae) 28 Oct. 1975; Holotype, St. Forest, N.J. 21 Aug. 1972, C.B. Knisley on Tsuga canadensis (L.) Carrière (Pinaceae).
Remarks
Typhlodromus (Typhlodromus) knisleyi was kept for a long time in the Acari collection of our laboratory and labelled as Typhlodromus sp. After its description from material collected at New Jersey (USA), only one specimen of T. knisley was reported from the Fraktò virgin forest (Greece) (Tsolakis and Ragusa 2009). This is the first record from Italy. It is certainly, a rare species mainly associated with forest plants. We have had the type material of T. (T.) knisleyi in loan by Harold Denmark and here we redescribe the species adding some characters not reported in the original description. Our measurements of the holotype are almost the same as those reported in the original description except for the setae j3 (27 in the original description and 23 measured by us) and the number of teeth on the fixid digit (two reported in the original description, but four, plus the apical one, in the present work: three anteriorly to pilus dentilis and one posteriorly).
Typhlodromus (Typhlodromus) kykladiticus Papadoulis & Emmanouel 1993
Typhlodromus kykladiticus Papadoulis & Emmanouel (1993) (original designation)
Specimens examined — 35 ♀♀ in total. Mussomeli (Caltanissetta) (37°34'59.19'' N, 13°43'32.32'' E, m 803 a.s.l.), 1♀ on Sideritis italica (Mill.) Greuter & Burdet (Lamiaceae) 02 Jul. 1980; Palazzo Adriano (Palermo), (37°40'18.50'' N, 13°22'23.91'' E, m 707 a.s.l.), 6♀♀ on Artemisia arborescens L. (Asteraceae) 16 Jul. 2007; S. Stefano di Camastra (Messina), (38°0'42.48'' N, 14°20'27.53'' E, m 22 a.s.l.), 2♀♀ on Vitis vinifera L. (Vitaceae) 14 Oct. 1986; Selinunte (Trapani), (37°35'7.30'' N, 12°49'38.26'' E, m 5 a.s.l.), 1♀ on Senecio vulgaris L. (Asteraceae) 22 Jul. 1980; Termini Imerese (Palermo), (37°59'25.02'' N, 13°41'22.68'' E, m 31 a.s.l.), 10♀♀ on Cycas revoluta Thunb. (Cycadaceae) 11 Oct. 1978; Termini Imerese (Palermo), (37°59'29.41'' N, 13°40'58.95'' E, m 5 a.s.l.), 15♀♀ on Citrus limon (L.) Osbeck
(Rutaceae) 02 May 1979.
Remarks — As above, also this species was collected in Sicily about a decade before its first description and kept in the Acari colection of our laboratory, labelled as Typhlodromus sp. The species has been reported up to now only from Greece.
Typhlodromus (Typhlodromus) pentelicus Papadoulis & Emmanouel 1990
Typhlodromus pentelicus Papadoulis & Emmanouel (1990) (original designation)
Specimens examined — Eight females and two males at Palazzo Adriano (Palermo), (37°40'49.81'' N, 13°21'32.91'' E, m 652 a.s.l.), on Cupressus sempervirens L. (Cupressaceae) 10 Jul. 2007.
Remarks — As the previous species, Typhlodromus (T.) pentelicus was reported up to now only for Greece.
Acknowledgements
Authors are deeply indebted to Mrs Eleonora Chiavetta for the revision of the English text, and to Dr. Harold Denmark who lent us the type material of Typhlodromus (T.) knisleyi, and the two anonymous reviewers for their constructive comments.
References
Arutunjan E.S. 1972. New species of mites of the genus Seiulus Berlese, 1887 (Parasitiformes: Phytoseiidae). Doklady Akademii Nauk Armyanskoi SSR, 54: 237-240. [in Russian].
Athias-Henriot C. 1960. Phytoseiidae et Aceosejidae (Acarina: Gamasina) d' Algerie. IV. Genre Typhlodromus Scheuten, 1857. Bulletin de la Societe d'Histoire Naturelle de l'Afrique du Nord, 51: 62-107.
Athias-Henriot C. 1975. Nouvelles notes sur les Amblyseiini II- Le relevé organotaxique de la face dorsale adulte (Gamasides Protoadeniques, Phytoseiidae). Acarologia, 17(1): 20-29.
Athias-Henriot C. 1977. Nouvelles notes sur les Amblyseiini III. Sur le genre Cydnodromus : Redéfinition, Composition (Parasitiformes, Phytoseiidae). Entomophaga, 22(1): 61-73. doi:10.1007/BF02372991
Beard J.J. 2001. A review of Australian Neoseiulus Hughes and Typhlodromips De Leon (Acari: Phytoseiidae: Amblyseiinae). Invertebrate Taxonomy 15: 73-158. doi:10.1071/IT99017
Beglyarov G.A. 1981. Keys to the determination of phytoseiid mites of the USSR. Information Bulletin International Organization for Biological Control of Noxious Animals and Plants, East Palaearctic Section, Leningrad, Russia, 2, 1-97. [in Russian].
Berlese A. 1887. Acari, Myriopoda et Scorpiones hucusque in Italia reperta. Tipografia del Seminario, Padova, Italy, 4(38): 6-7.
Berlese A. 1889. Acari, Myriopoda et Scorpiones hucusque in Italia reperta. Tipografia del Seminario, Padova, Italy, 6(54): 7-9.
Berlese A. 1923. Centuria sesta di Acari nuovi. Redia, 15: 237-262.
Calvo J.F., Knapp M., van Houten Y.M., Hoogerbrugge H., Belda J.E.2015. Amblyseius swirskii: What made this predatory mite such a successful biocontrol agent? Exp. Appl. Acarol., 65: 419-433. doi:10.1007/s10493-014-9873-0
Canestrini G., Fanzago F. 1876. Nuovi acari italiani (Seconda Serie). Atti Società Veneto-Trentina di Scienze Naturali, Italy, 5: 130-142.
Chant D.A. 1957. Note on the status of some genera in the family Phytoseiidae (Acarina). Can. Entomol., 89(11): 528-532. doi:10.4039/Ent89528-11
Chant D.A. 1959. Phytoseiid mites (Acarina: Phytoseiidae). Part I. Bionomics of seven species in southeastern England. Part II. A taxonomic review of the family Phytoseiidae, withdescriptions of thirty-eight new species. Can. Entomol., Supplement 12, 166 pp. doi:10.4039/entm9112fv
Chant D.A., McMurtry J.A. 1994. A review of the subfamilies Phytoseiinae and Typhlodrominae (Acari: Phytoseiidae). Int. J. Acarol., 20: 223-310. doi:10.1080/01647959408684022
Chant D.A., McMurtry J.A. 2003. A review of the subfamilies Amblyseiinae (Acari: Phytoseiidae): Part II. Neoseiulini new tribe. Int. J. Acarol., 29: 3-46. doi:10.1080/01647950308684319
Chant D.A., McMurtry J.A. 2004. A review of the subfamily Amblyseiinae Muma (Acari: Phytoseiidae): Part III. The tribe Amblyseiini Wainstein, subtribe Amblyseiina N. subtribe. Int. J. Acarol. 30: 171-228. doi:10.1080/01647950408684388
Chant D.A., Yoshida-Shaul E. 1982. On the identity of Amblyseius umbraticus (Chant) (Acarina: Phytoseiidae). Can. J. Zool., 60(8): 1998-2005. doi:10.1139/z82-257
Chant D.A., Yoshida-Shaul E. 1983. A world review of the simplex species group in the genus Typhlodromus Scheuten (Acarina: Phytoseiidae). Can. J. Zool., 61(5): 1142-1151. doi:10.1139/z83-151
Chant D.A., Yoshida-Shaul E. 1989. Adult dorsal setal patterns in the family Phytoseiidae (Acari: Gamasina). Int. J. Acarol., 15: 219-233. doi:10.1080/01647958908683852
Chant D.A., Yoshida-Shaul E. 1991. Adult ventral setal patterns in the family Phytoseiidae (Acari: Gamasina). Int. J. Acarol., 17: 187-199. doi:10.1080/01647959108683906
Chant D.A., Hansell R.I.C., Yoshida-Shaul E. 1974. The genus Typhlodromus Scheuten (Acarina: Phytoseiidae) in Canada and Alaska. Can. J. Zool., 52: 1265-1291. doi:10.1139/z74-168
Charlet L.D., McMurtry J.A. 1977. Systematics and bionomics of predaceous and phytophagous mites associated with pine foliage in California. Hilgardia, 45(7), 173-236. doi:10.3733/hilg.v45n07p173
Congdon B.D. 2002. The family Phytoseiidae (Acari) in western Washington State with descriptions of three new species. Int. J. Acarol., 28(1): 3-27. doi:10.1080/01647950208684275
De Leon D. 1959. Two new genera of phytoseiid mites with a note on Proprioseius meridionalis Chant (Acarina: Phytoseiidae). Entomological News, Philadelphia, 70(10): 257-262.
De Leon D. 1965. A note on Neoseiulus Hughes 1948 and new synonymy (Acarina: Phytoseiidae). Proceedings of the Entomological Society of Washington, 67(1), 23.
Demite P.R., Moraes G.J. de, McMurtry J.A., Denmark H.A., Castilho R.C. 2020. Phytoseiidae Database. Available from: www.lea.esalq.usp.br/phytoseiidae (accessed 2/03/2020).
Denmark H.A. 1992. A revision of the genus Typhlodromus Scheuten (Acari: Phytoseiidae). Occasional Papers of the Florida State Collection of Arthropods, 7: 1-43.
Denmark H.A., Rather A.Q. 1996. Revision of the genus Neoseiulella Muma (Acari: Phytoseiidae). Int. J. Acarol., 22(1): 43-77. doi:10.1080/01647959608684080
Denmark H.A., Welbourn W.C. 2002. Revision of the genera Amblydromella Muma and Anthoseius De Leon (Acari: Phytoseiidae). Int. J. Acarol., 28(4): 291-316. doi:10.1080/01647950208684308
Duso C., Sbrissa F. 1990. Gli Acari fitoseidi (Acari Phytoseiidae) del melo nell'Italia settentrionale: distribuzione, biologia, ecologia ed importanza economica. Boll. Zool. Agr. Bachic., Serie II, 22(1): 53-89.
Duso C., Malagnini V., Paganelli A., Aldegheri L., Bottini M., Otto S. 2004. Pollen availability and abundance of predatory phytoseiid mites on natural and secondary hedgerows. BioControl, 49: 397-415. doi:10.1023/B:BICO.0000034601.95956.89
Evans G.O., Till W.M. 1979. Mesostigmatic mites of Britain and Ireland (Chelicerata: Acari-Parasitiformes). An introduction to their external morphology and classification. Trans. Zool. Soc., 35: 139-270. doi:10.1111/j.1096-3642.1979.tb00059.x
Faraji A.R., Shiroodbakhshi M., Ostovan H., McMurtry J.A. 2007. Redescription of the female of Paraseiulus triporus and Proprioseiopsis dacus (Acari: Phytoseiidae) based on material collected from citrus in northern Iran. Syst. Appl. Acarol., 12: 199-204. doi:10.11158/saa.12.3.4
Gianguzzi L., La Mantia A. 2009. Contributo alla conoscenza della vegetazione e del paesaggio vegetale della Riserva Naturale "Monte Cofano" (Sicilia occidentale) (con allegata Carta sinfitosociologica della vegetazione, scala 1:20.000). Fitosociologia, 45(1) Suppl. 1: 1-55.
Gilyarov M.S., Bregetova N.G., Wainstein B.A., Kadite B.A., Koroleva E.V., Petrova A.D., Tikhomirov S.I., Shcherbak G.I. 1977. Manual of edaphic mites (Mesostigmata). Akademiya Nauk SSSR, Nauka Publishing House, Leningrad, Russia: 1-718 pp. [in Russian].
Hughes A.M. 1948. The mites associated with stored food products. Ministry of Agriculture and Fisheries, H.M. Stationary Office, London, 1-168 pp.
Ivancich-Gambaro P. 1975. Observations on the biology and behaviour of the predacious mite Typhlodromus italicus (Acarina: Phytoseiidae) in peach orchards. Entomophaga, 20: 171-177. doi:10.1007/BF02371657
Karg W. 1989. Neue Raubmilbenarten der Gattuig Proprioseiopsis Muma, 1961 (Acarina, Parasitiformes) mit Bestimmungsschlusseln. Zoologische Jahrbucher Systematik, 116(2), 199-216.
Knapp M., van Houten Y., van Baal E., Groot T. 2018. Use of predatory mites in commercial biocontrol: current status and future prospects. Acarologia, 58(Suppl): 72-82. doi:10.24349/acarologia/20184275
Kreiter S., Ueckermann E.A., Quilici S. 2002. Seven new phytoseiid species, with a new generic assignement and a key to the species of La Reunion Island (Acari: Mesostigmata). Acarologia, 42(4): 335-350.
Lindquist E, Evans GW. 1965. Taxonomic concepts in the Ascidae, with a modified setal nomenclature for the idiosoma of the Gamasina (Acarina: Mesostigmata). Mem. Ent. Soc. Can., 47: 1-64. doi:10.4039/entm9747fv
Lorenzon M., Pozzebon A., Duso C. 2018. Biological control of spider mites in North-Italian vineyards using pesticide resistant predatory mites. Acarologia, 58(Suppl): 98-118. doi:10.24349/acarologia/20184277
McMurtry J.A., Famah Sourassou N., Demite P.R. 2015. The Phytoseiidae (Acari: Mesostigmata) as Biological Control Agents. In: Carrillo et al. (Eds.), Prospects for Biological Control of Plant Feeding Mites and Other Harmful Organisms, Progress in Biological Control, 19, pp. 133- 149. doi:10.1007/978-3-319-15042-0_5
McMurtry J.A., Moraes G.J.de, Famah Sourassou N. 2013. Revision of the lifestyles of phytoseiid mites (Acari: Phytoseiidae) and implications for biological control strategies. Syst. Appl. Acarol., 18(4): 297-320. doi:10.11158/saa.18.4.1
Moraes G.J.de, McMurtry J.A., Denmark H.A. 1986. A catalog of the mite family Phytoseiidae. References to taxonomy, synonymy, distribution and habitat. EMBRAPA - DDT, Brasilia, Brazil: 1-353.
Muma M.H. 1961. Subfamilies, genera and species of Phytoseiidae (Acarina: Mesostigmata). Bull. Fl St. Mus., 5: 267-302.
Musarella C.M., Mendoza-Fernández A.J., Mota J.F., Alessandrini A., Bacchetta G., Brullo S., Caldarella O., Ciaschetti G., Conti F., Di Martino L., Falci A., Gianguzzi L., Guarino R., Manzi A., Minissale P., Montanari S., Pasta S., Peruzzi L., Podda L., Sciandrello S., Scuderi L., Troìa A., Spampinato G. 2018. Checklist of the gypsophilous vascular flora in Italy. PhytoKeys, 103: 61-82. doi:10.3897/phytokeys.103.25690
Papadoulis G.Th., Emmanouel N.G. 1990. Two new species of the genus Typhlodromus Scheuten (Acari: Phytoseiidae) from Greece. Entomologia Hellenica, 8: 11-19. doi:10.12681/eh.13974
Papadoulis G.Th., Emmanouel N.G. 1993. New records of phytoseiid mites from Greece with descriptions of two new species of Typhlodromus Scheuten (Acarina: Phytoseiidae). Int. J. Acarol., 19(4): 321-328. doi:10.1080/01647959308683986
Pignatti S. 1982. Flora d'Italia, 1-3. Edagricole, Bologna.
Quilici S., Kreiter S., Ueckermann E.A., Vincenot D. 1997. Predatory mites (Acari) from various crops on Réunion Island. Int. J. Acarol., 23(4): 283-291. doi:10.1080/01647959708683578
Ragusa Di Chiara S., Tsolakis H. 1996. A survey of phytoseiid mites (Parasitiformes, Phytoseiidae) associated with various plants in Sicily (Italy). In: Mitchell R., Horn D.J., Needham G.R., Welbourn W.C. (Eds). Acarology IX, 1 Proceedings, Xvi +718 pp Ohio Biological Survey, Columbus, Ohio: 253-256.
Ragusa S. 1979. Laboratory studies on the food habits of the predaceous mite Typhlodromus exhilaratus. Advances in Acarology, 1: 485-490. doi:10.1016/B978-0-12-592201-2.50068-3
Ragusa S. 1981. Influence of different kinds of foood substances on the developmental time in young stage of the predacious mite Typhlodromus exhilaratus Ragusa (Acarina: Phytoseiidae). Redia, 64: 237-243.
Ragusa S., Paoletti M.G. 1985. Phytoseiid mites (Parasitiformes: Phytoseiidae) of corn and soybean agroecosystem in the low-layng plain of Veneto (N-E Italy.). Redia, 68: 69-89.
Ragusa S., Swirski E. 1976. Notes on predacious mites of Italy, with a description of two new species and of an unknown male (Acarina: Phytoseiidae). Redia, 59: 179-196.
Ragusa S., Swirski E. 1978. Description of three new species of Typhlodromus Scheuten from Italy with redescription of Typhlodromus baccettii Lombardini (Acari: Phytoseiidae). Int. J. Acarol., 4(3): 211-220. doi:10.1080/01647957808683118
Ragusa S., Swirski E. 1982. A new species of Phytoseius (Acarina: Phytoseiidee) from Italy. Redia, 65: 293-301.
Ribaga C. 1904. Gamasidi planticoli. Rivista di Patologia Vegetale, 10: 175-178.
Rivnay T., Swirski E. 1980. Four new species of phytoseiid mites (Acarina: Mesostigmata) from Israel. Phytoparasitica, 8: 173-187. doi:10.1007/BF03158314
Rowell H.J., Chant D.A., Hansell R.I.C. 1978. The determination of setal homologies and setal patterns on the dorsal shield in the family Phytoseiidae (Acarina: Mesostigmata). Can. Ent., 110: 859-876. doi:10.4039/Ent110859-8
Scheuten A. 1857. Einiges uber Milben. Archiv fur Naturgeschichte, 23: 104-112.
Schuster R.O., Pritchard A.E. 1963. Phytoseiid mites of California. Hilgardia, 34(7): 191-285. doi:10.3733/hilg.v34n07p191
Swirski E., Amitai S. 1997. Notes on phytoseiid mites (Mesostigmata: Phytoseiidae) of Mt. Carmel (Israel), with descriptions of two new species. Israel J. Entomol., 31: 1-20.
Szabó A., Pénzes B., Sipos P., Hegyi T., Hajdú Z., Markó V. 2013. Pest management systems affect composition but not abundance of phytoseiid mites (Acari: Phytoseiidae) in apple orchards. Exp. Appl. Acarol., 62:525-537. doi:10.1007/s10493-013-9752-0
Sznajder B., Sabelis M.W., Egas M. 2011. Innate responses of the predatory mite Phytoseiulus persimilis to a herbivore-induced plant volatile. Exp. Appl. Acarol., 54: 125-138. doi:10.1007/s10493-011-9430-z
Tsolakis H., Ragusa Di Chiara S. 1993. Effetti collaterali di alcuni fitofarmaci nei confronti di Amblyseius andersoni (Chant) (Parasitiformes, Phytoseiidae). Boll. Zool. agr. Bachic. Ser. II, 25(2): 205-212.
Tsolakis H., Ragusa E. 2017. Phytoseiid mites from Basilicata region (Southern Italy): species diversity and redescription of Typhloseiulus arzakanicus (Arutunjan) with a key of the species of Typhloseiulus Chant and McMurtry 1994 (Parasitiformes: Phytoseiidae). Acarologia, 57(4): 805-821. doi:10.24349/acarologia/20174195
Tsolakis H., Ragusa S. 1999. Overwintering of phytoseiid mites (Parasitiformes, Phytoseiidae) on hazelnut (Corylus avellanae L.) in Sicily (Italy). In: Bruin, van der Geest, Sabelis (Eds). Ecology and Evolution of the Acari, Kluwer Academic Publishers. pp. 625-635. doi:10.1007/978-94-017-1343-6_54
Tsolakis H., Ragusa S. 2009. Phytoseiid mites associated with forest trees in two "Natura 2000" location: S. Adriano woods (Sicily) and Fraktò virgin forest (Greece). IOBC/WPRS, work group Integrated control of plant-feeding mites, Florence 9-12 march 2009, 24 (abstract).
Tsolakis H., Ragusa S. 2015. Considerations on systematics of the Phytoseiidae (Acari: Mesostigmata), with definition of a new species group and description of a new species. Zootaxa, 3926 (2): 229-243. doi:10.11646/zootaxa.3926.2.4
Tsolakis H., Ragusa E., Ragusa Di Chiara S. 2000. Distribution of phytoseiid mites (Parasitiformes, Phytoseiidae) on hazelnut at two different altitudes in Sicily. Env. Entomol., 29(6): 1251-1257. doi:10.1603/0046-225X-29.6.1251
Tsolakis H., Tixier M.-S., Kreiter S., Ragusa S. 2012. The concept of genus within the family Phytoseiidae (Acari: Parasitiformes): historical review and phylogenetic analyses of the genus Neoseiulus Hughes. Zool. J. Linnean Soc., 165: 253-273. doi:10.1111/j.1096-3642.2011.00809.x
Tsolakis H., Sinacori M., Ragusa S. 2013. Predation of Typhlodromus longilaterus Athias-Henriot (Parasitiformes, Phytoseiidae) females on eggs and juveniles of the tetranychid mites Tetranychus urticae (Koch) and Panonychus citri (McGregor) (Acariformes, Tetranychidae). IOBC-WPRS Bulletin 93: 129-132.
Tsolakis H., Principato D., Jordà Palomero R., Lombardo A. 2016. Biological and life table parameters of Typhlodromus laurentii and Iphiseius degenerans (Acari, Phytoseiidae) fed on Panonychus citri and pollen of Oxalis pes-caprae under laboratory conditions. Exp. Appl. Acarol., 70(2): 205-218. doi:10.1007/s10493-016-0076-8
Wainstein BA.1962. Révision du genre Typhlodromus Scheuten,1857 et sistématique de la famille des phytoseiidae (Berlese, 1916) (Acarina: Parasitiformes). Acarologia, 4(1): 5-30.
Wainstein B.A. 1973. Predatory mites of the family Phytoseiidae (Parasitiformes) of the fauna of the Moldavian SSR. Fauna i Biologiya Nasekomykh Moldavii, Akademiya Nauk Moldavskoy SSR, Institut Zoologii, 12: 176-180 [in Russian].
Willmann C. 1949. Beitrage zur Kenntnis des Salzgebietes von Ciechocinek. 1. Milben aus den Salzwiesen und Salzmooren von Ciechocinek an der Weichsel. Veröffentlichungen Museum Bremen, 14A, 106-135 + 141-142.



2020-07-13
Date accepted:
2020-10-13
Date published:
2020-10-19
Edited by:
Kreiter, Serge

This work is licensed under a Creative Commons Attribution 4.0 International License
2020 Tsolakis, Haralabos and Ragusa, Salvatore
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)




