Factors influencing the level of infestation of Ixodes ricinus (Acari: Ixodidae) on Lacerta agilis and Zootoca vivipara (Squamata: Lacertidae)
Gwiazdowicz, Dariusz J.1 ; Gdula, Anna K.2 ; Kurczewski, Rafał3 and Zawieja, Bogna4
1✉ Poznan University of Life Sciences, Faculty of Forestry, Wojska Polskiego 71C, 60–625 Poznań, Poland.
2Poznan University of Life Sciences, Faculty of Forestry, Wojska Polskiego 71C, 60–625 Poznań, Poland.
3Wielkopolska National Park, Jeziory, 62-050 Mosina, Poland.
4Poznan University of Life Sciences, Department of Mathematical and Statistical Methods, Wojska Polskiego 28, 60–637 Poznań, Poland.
2020 - Volume: 60 Issue: 2 pages: 390-397
https://doi.org/10.24349/acarologia/20204372Original research
Keywords
Abstract
Introduction
Relations between ectoparasites, to which mites belong, and some species of lizards are the subject of numerous studies (Fain, 1962; Bauwens et al., 1983; Haitlinger, 1987; Gwiazdowicz and Filip, 2009a; Kurczewski, 2014). As a result of these studies, species of mites such as Dermacentor reticulatus, Haemaphysalis concinna, Ixodes ricinus, I. trianguliceps and Ophionyssus saurarum have been reported on the bodies of the sand lizard (Lacerta agilis) and the common (viviparous) lizard (Zootoca vivipara) (Siuda, 1993; Gwiazdowicz and Filip, 2009b). Most attention has been devoted to the castor bean tick (Ixodes ricinus) – a common blood reptile parasite worldwide (Barnard and Durden, 2000). This species has the widest dissemination range among European ticks and is frequently found on sand lizards (Bauwens et al., 1983; Matuschka et al., 1991; Gryczyńska-Siemiątkowska et al., 2007). Some studies suggest that infestation by this parasite might be a hindrance for lizards, e.g. European green lizards (Vacláv et al., 2007). Even though many studies take the form of short reports, the purpose of which is to draw attention to ticks that occur on lizards, it is worth noting a wide range of studies of an ecological nature (Oppliger and Clobert, 1997; Majláthová et al., 2010). Tälleklint-Eisen and Eisen (1999) examined the impact of environmental characteristics such as habitat type on the number of Ixodes pacificus ticks infesting the Western fence lizard Sceloporus occidentalis. On the other hand, Dudek et al. (2016) examined, among others, the correlation between the number of ticks on the body and a lizard's body size index (length measured from snout to tail).
Even though there are many publications on the subject, there is still a perceptible lack of knowledge in this respect; in particular when it comes to species of European lizards. Therefore, it is necessary to conduct further analyses of factors that influence the level of infestation of lizards in Central Europe with the castor bean tick.
The purpose of this study was to analyse infestation of two species of lizards, namely Lacerta agilis and Zootoca vivipara, which can be found in Central Europe, with I. ricinus, as well as to address the question of what determines levels of infestation. Therefore, both the effects of environmental (habitats within protected areas and areas transformed as a result of human activity) and morphological (age, sex and size of lizards, location of ticks on lizard's body) factors on tick prevalence were examined.
Understanding of the factors determining tick prevalence will allow for more effective protection of both species of lizard which in many countries are under legal protection and are on the red list of threatened species, such as the International Union for Conservation of Nature (IUCN) Red List (Agasyan et al. 2010).
Materials and methods
Study area and procedures
The research was carried out between April and June during the years 2010–2015 in Poznań city (52°24ʹ–52°28ʹ N, 16°50ʹ–16°56ʹ E) in urban green areas as well as in areas subject to anthropogenic impact, frequently visited by citizens, such as city parks, and in Wielkopolska National Park (52˚13ʹ39.4ʹʹ–52˚19ʹ08.0ʹʹ N, 16˚32ʹ00.0ʹʹ–16˚46ʹ08.0ʹʹ E), which is a nature protected area.
Lizards were captured in randomly selected locations from typical sand lizard and viviparous lizard habitats, e.g. a clearing in a pine forest, a pine coppice, southern side of a railway embankment, a verge of a tourist path. The lizards were captured with the use of herpetological nets or by hand. They were grouped according to sex (based on the presence of femoral pores in males – which are larger and more distinct than in females – and an expanded gonadal area in the tail base in males) and age (young – in the 1st year of life, adult – 2nd year or older). All lizards were measured (from snout to the tip of the tail) and assigned to size classes: I (0–5cm), II (5.1–10 cm), III (10.1–15 cm), IV (\textgreater 15.1 cm) and examined for the presence of I. ricinus. The locations of ticks on the lizards' bodies were recorded as: front limbs, front groins, hind limbs, hind groins, ears, neck, sides. The collected ticks were preserved in 70% ethanol, counted, and identified with the use of a stereoscopic microscope and taxonomical descriptions.
Statistics
A Poisson logistic regression model was used to investigate the impact of species, habitat, sex and age (combined into a single measure) and body size on the number of ticks found on individual lizards (Agresti, 2002). In this model body size was included as a covariate variable. As the design was not orthogonal, only some interactions could be included in the model. These were Body size x Species, Sex-age x Species, Body size x Habitat, Sex-age x Habitat, Body size x Sex-age. This model, including these 5 two-way interactions, was subsequently simplified by sequentially eliminating non-significant terms and interactions to establish a minimal model (Crawley, 2007). A Poisson logistic model was also used to investigate the impact of location on the body on the number of ticks. Because multiple locations were measured on the same lizard, these measures are not independent. In this situation, a general mixed model with unstructured covariance of repeated measures was used (Wolfinger, 1993). The response variable in this model was the number of ticks in each location. As multiple measures were taken from each individual, lizard was included in the model as a random effect. As there were no ticks found in the hind groin, this location was not taken into consideration in this model. It was not possible to make all post-hoc comparisons between locations, as such the number of ticks found in the front groin were compared with all other locations pooled. When terms were significant, contrasts analyses were used to detect differences among means. The statistical analyses were conducted in STATISTICA and SAS. In order to assess statistical significance of the examined factors, the empirical level of significance (p-value) is provided. All statistical testing was conducted at a significance level of 0.05. All mean values are given with standard errors (SE), unless otherwise stated.
Results
In this study, 396 lizards were examined (328 individuals of L. agilis and 68 individuals of Z. vivipara). The percentage of lizards infested with ticks was 32.01 ± 2.6 % and 38.24 ± 5.9 % for Z. vivipara and L. agilis respectively. In total, 645 specimens of I. ricinus were found, including 339 nymphs and 306 larvae. The number of ticks found on one lizard ranged from 0 to 35 individuals and the mean number of ticks per lizard was 1.6 ± 3.99 SE in L. agilis and 1.76 ± 3.49 SE in Z. vivipara (Table 1).



Three interactions were significant, the Sex-age x Species, the Body size x Habitat and Sex-age x Habitat. This means that the effect of our sex-age category was not the same for each species, or in the different habitats. Moreover, the effect of body size differed across habitats. Below we discuss how the environmental variables measured impact levels of tick infestation on the different lizard species. As the effect of each variable measured was also included in a significant interaction, we discuss the results in light of the interactions shown in Table 2.
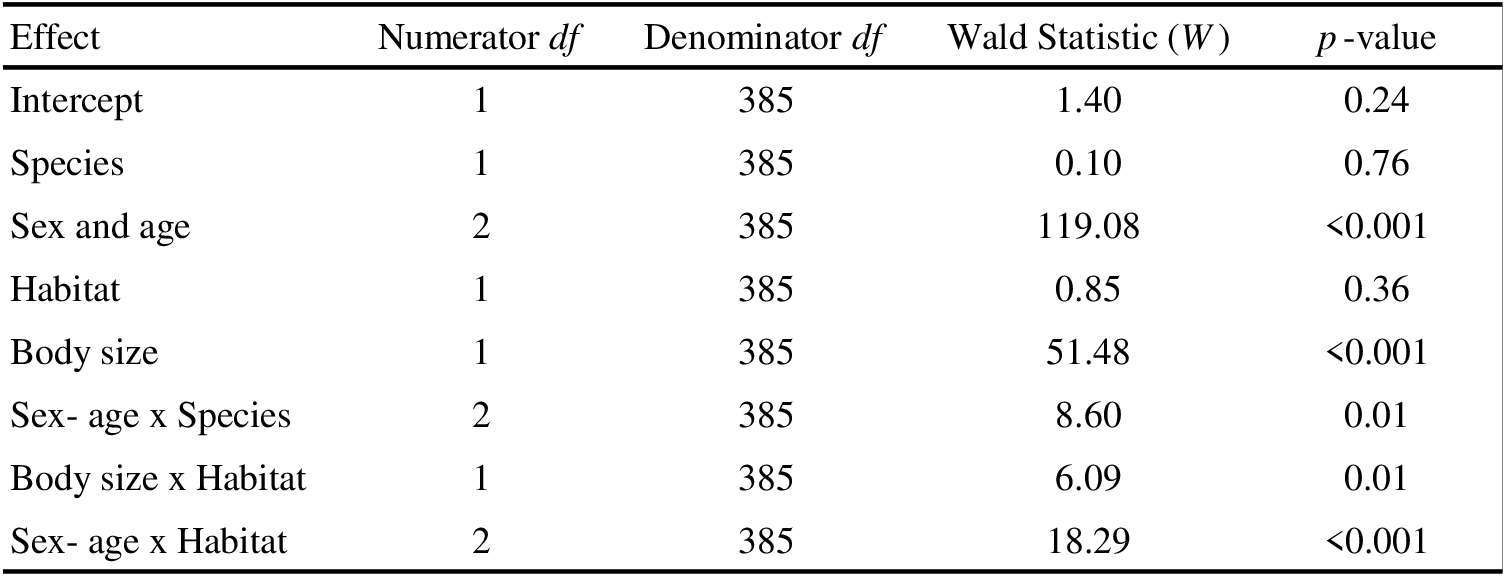


Effect of life-stage and lizard species on tick infestation
Does the level of infestation by I. ricinus depend on the species of lizard? The effect of species on the number of lizards was different according to the sex and age category as shown by the significant interaction.
The Sex-age x Species interaction was significant (W = 8.60, p = 0.01). Since not all post-hoc pairwise comparisons were possible, only the comparison between the species for each sex-age category were done. Thus, based on the contrasts analysis it was found, that there was no difference in the number of ticks infecting juveniles of L. agilis (predicted mean 0.52 ± 0.07 SE) and juveniles of Z. vivipara (predicted mean 0.43 ± 0.12 SE, p = 0.71, W = 0.13). However, infection of L. agilis females (predicted mean 1.80 ± 0.57 SE) was significantly lower (p = 0.01, W = 7.12) than Z. vivipara females (predicted mean 2.25 ± 0.99 SE). Although only these two contrasts could be estimated in the over-parameterized Poisson model, based on the estimated means it should be noted that the predicted number of ticks on males of L. agilis (2.38 ± 0.93 SE) was similar to the predicted mean for males of Z. vivipara (2.25 ± 1.10 SE); in the case of viviparous lizards more ticks were found on adults than juveniles (Fig. 1).
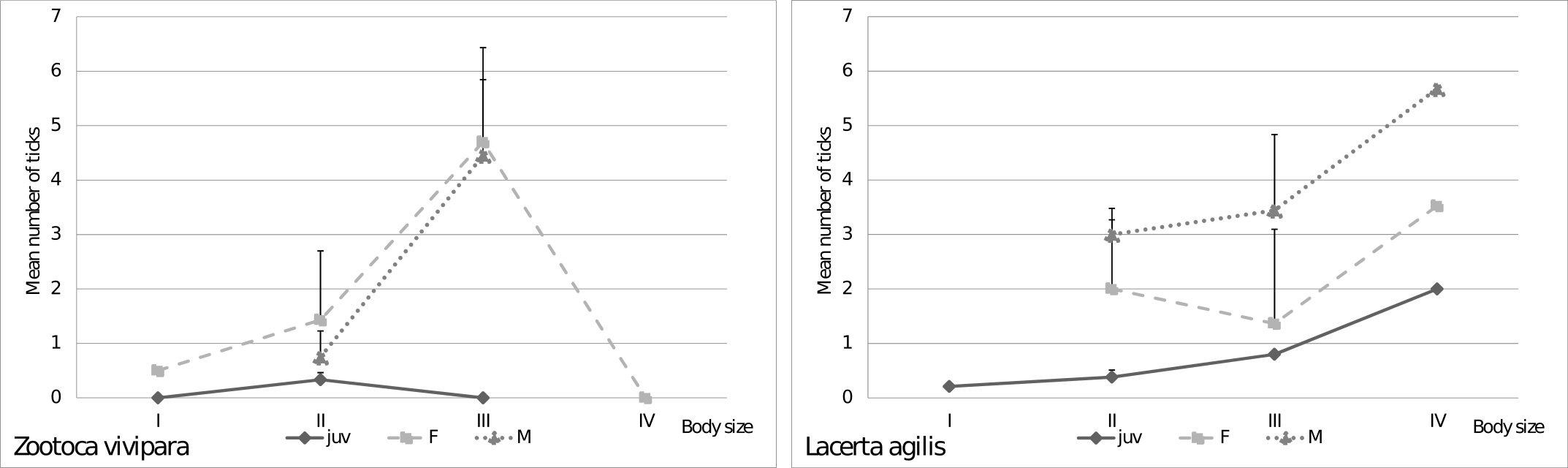


Effect of lizard habitat and interactions with other factors on tick infestation
Do environmental conditions distinguished in terms of a diversified form of anthropogenic activity have an influence on the level of infestation of lizards with Ixodes ricinus? In order to answer this question, we examined the significance of the differences between the number of ticks on lizards in protected areas (Wielkopolska National Park) with no direct anthropogenic interference and in areas under constant pressure from humans (urban green areas in Poznań) where grass was mowed in places where lizards occurred.
A significant interaction shows that the impact of habitat on the number of ticks depends on body size (W = 6.09, p = 0.01). It was shown that in natural conditions larger lizards had more ticks (coefficient of logistic regression: b1 = 0.60, p < 0.001), which was not observed in transformed conditions (coefficient of logistic regression: b1 = 0.13, p = 0.45) (Fig. 2). The Sex-age x Habitat interaction was also significant (W = 18.20, p < 0.001). Based on the analysis of contrasts it was found, that females in transformed conditions (predicted mean 3.14 ± 0.88 SE) had a significantly (W = 18.22, p < 0.001) larger number of ticks than females in natural conditions (predicted mean 1.76 ± 0.55 SE). Additionally, the number of ticks on juveniles in transformed conditions (predicted mean 0.49 ± 0.45 SE) did not differ significantly (W = 1.41, p = 0.24) from juveniles in natural conditions (predicted mean 0.46 ± 0.06 SE). Only these two contrasts could be estimated in an over-parameterized Poisson model, but based on estimated means it should be noted that the predicted number of ticks for males in transformed conditions was smaller than in natural conditions (1.78 ± 0.57 SE and 3.00 ± 1.00 SE respectively).
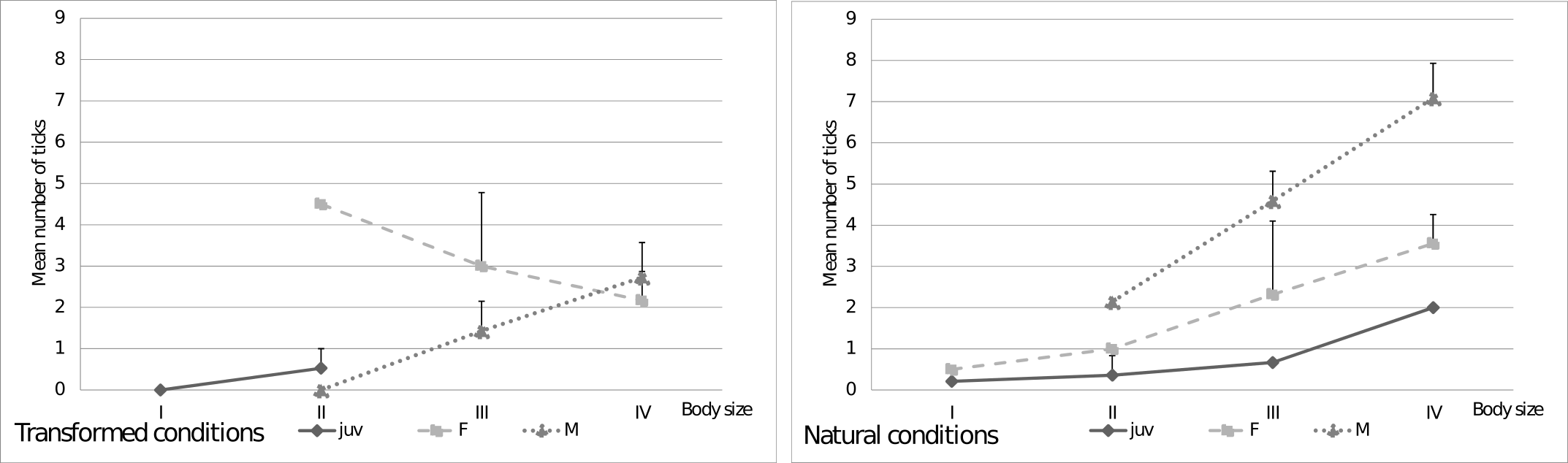


Effect of the location on the lizard's body on tick infestation
What part of a lizard's body is the most vulnerable to infestation by ticks? While examining the lizards, one of seven locations of ticks on a lizard's body was recorded (specified in the methodology). It turned out that ticks are most frequently found on the front groin (336 individual ticks) and on the front limbs (202 individual ticks), and less often on the neck – only 12 individuals had ticks here. No ticks were found on the hind groin.
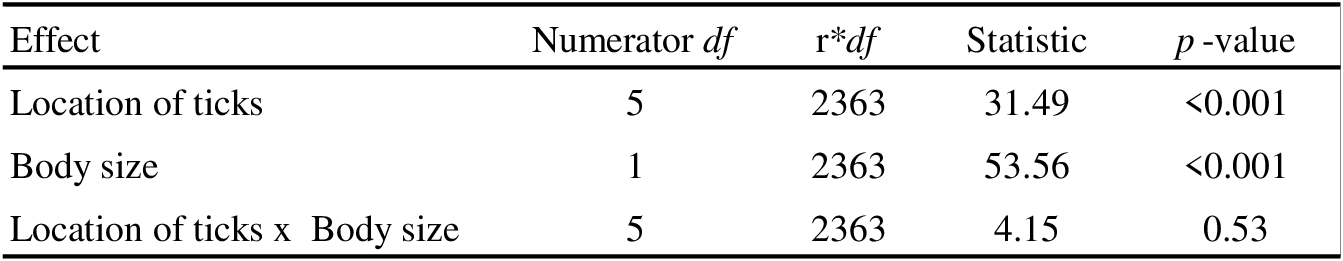


There was a significant difference between the number of ticks located in various parts of a lizard's body. Moreover, there was an effect of body size on the number of ticks found (Table 3, W = 53.56, p < 0.001), but the interaction between tick location and body was not significant (Table 3, W = 4.15, p = 0.53). This means that the number of ticks increases with body size, and certain locations of the body were associated with more ticks, but probability of infection on any particular location did not increase with body size. Substantial differences were found between the number of ticks in the front groin and the number of ticks in the remaining parts of body (p < 0.001) (Fig. 3).



Discussion and conclusions
We investigated several factors which could influence the level of infestation by I. ricinus ticks on two species of lizards – L. agilis and Z. vivipara were examined. We found that levels of infestation differed for females of the different species, and increases in tick infestation with size depended on the type of habitat. The influence of environmental factors (grassland, woodland) on the level of infestation by Ixodes pacificus on Western fence lizard Sceloporus occidentalis were examined by Tälleklint-Eisen and Eisen (1999). Although their results are not directly comparable with our study, due to different species and environmental conditions.
For both species of lizards significant differences were found between the number of ticks on young lizards and adult lizards, both in terms of their number as well as their location. The number of ticks on young lizards was low and this may be why in other studies ticks were not found on juveniles (e.g. Leeuwen and van de Hoef, 1976; House et al. 1980; Strijbosch et al. 1983; Bauwens et al., 1983). Furthermore, our results show differences between the level of infestation in adult individuals depending on sex and habitat as found in other studies (Nuland et al. 1981). Similar to research of other authors (Rahmel and Meyer, 1988; Kurczewski, 2014; Dudek et al., 2016), male lizards in natural conditions across both species (Figures 2) were more densely infested than in transformed conditions. This may result from a greater activity of males, during the mating season (Siuda, 1993).
On the contrary, our research shows that factors determining the overall mean level of infestation of females of both L. agilis and Z. vivipara by ticks increases with anthropogenic activity and environmental conditions created by humans (grass mowing, for instance). These results show that humans can indirectly impact tick infestation for different species of lizards and therefore possibly also the pathogenic agents transmitted by them. Fortunately, we found that in transformed conditions tick infestation per individual did not increase with body size, showing the potential for lower infestation levels. This is important because tick-borne diseases have shown a significant rise in the last decade (Jones et al. 2008). In Europe, Lyme borreliosis (LB) is maintained through complex interactions between the pathogen, Borrelia burgdorferi sensu lato (s.l.), different tick species and a large number of vertebrate hosts (Földvári et al. 2009). It is now known that over 230 animal species are implicated as hosts for Ixodes ricinus ticks (Gern 2008). A previous study shows that lizards can be important hosts for I. ricinus immatures (Földvári et al. 2009), thus may also be infected with B. burgdorferi. Our study compliments previous work showing ecological and morphological factors that influence tick infestation in 2 important lizard species currently on the ICUN list. We hope that the presented results, apart from broadening the knowledge on this subject, will also help in the more effective protection of these lizard species. The results presented in our study may also form the basis for further research.
Acknowledgments
We would like to thank anonymous reviewers who provided valuable contributions to improve to the manuscript. We would like to thank in particular Dr. Alison Duncan for the understanding and help during the editorial work.
References
Agasyan, A., Avci, A., Tuniyev, B., Lymberakis, P., Andrén, C., Cogalniceanu, D., Wilkinson, J., Ananjeva, N., Üzüm, N., Orlov, N., Podloucky, R., Tuniyev, S., Kaya, U., Crnobrnja Isailovic, J., Vogrin, M., Corti, C., Pérez Mellado, V., Sá-Sousa, P., Cheylan, M., Pleguezuelos, J., Kyek, M., Westerström, A., Nettmann, H.K., Borczyk, B., Sterijovski, B. & Schmidt, B. 2010 - Lacerta agilis. The IUCN Red List of Threatened Species 2010 - e.T157288A5071439. Downloaded on 13 June 2019. doi:10.2305/IUCN.UK.2010-4.RLTS.T157288A5071439.en ![]()
Agresti A. 2002 - Categorical data analysis - Willey and Sons: 117-118. doi:10.1002/0471249688 ![]()
Barnard S.M., Durden L.A. 2000 - A veterinary guide to the parasites of reptiles. Vol. 2. Arthropods (excluding mites) - Krieger, Malabar, Fla, pp. 288.
Bauwens D., Strijbosch H., Stumpel A.H.P. 1983 - The lizards Lacerta agilis and Lacerta vivipara as hosts to larvae and nymphs of the tick Ixodes ricinus - Holarctic Ecology, 6: 32-40. doi:10.1111/j.1600-0587.1983.tb01062.x ![]()
Crawley M.J., 2007 - The R Book. John Wiley & Sons, pp. 327. doi:10.1002/9780470515075 ![]()
Dudek K., Skórka P., Sajkowska Z.A., Ekner-Grzyb A., Dudek M., Tryjanowski P. 2016 - Distribution pattern and number of ticks on lizards - Ticks and Tick-borne Diseases, 7: 172-179. doi:10.1016/j.ttbdis.2015.10.014 ![]()
Fain A. 1962 - Les acariens mesostigmatiques ectoparasites des serpents - Bull. Inst. Roy. Sci. Nat. Belg., 38: 1-149.
Földvári G., Rigó K., Majláthová V., Majláth I., Farkas R., Pet'ko B. 2009 - Detection of Borrelia burgdorferi sensu lato in Lizards and Their Ticks from Hungary - Vec. Bor. Zoo. Dis., 9 (3): 331-336. doi:10.1089/vbz.2009.0021 ![]()
Gern L. 2008 - Borrelia burgdorferi sensu lato, the agent of Lyme borreliosis: life in the wilds - Parasite, 15: 244-247. doi:10.1051/parasite/2008153244 ![]()
Gryczyńska-Siemiątkowska A., Siedlecka A., Stańczak J., Barkowska M. 2007 - Infestation of sand lizard (Lacerta agilis) resident in North eastern Poland by Ixodes ricinus (L.) ticks and their infection with Borrelia burgdorferi sensu lato - Acta Parasitol., 52: 165-170. doi:10.2478/s11686-007-0015-2 ![]()
Gwiazdowicz D.J., Filip K.P. 2009a - Ophionyssus saurarum (Acari, Mesostigmata) infecting Lacerta agilis (Reptilia, Lacertidae) - Wiad. Parazytol., 51(1): 61-62.
Gwiazdowicz D.J., Filip K.P. 2009b - Ixodes ricinus (L.) (Acari, Ixodida) parasitic on lizards (Reptilia, Lacertidae) - Nauka Przyroda Technologia, 3(3): 76.
Haitlinger R. 1987 - Roztocze (Acari) występujące w Polsce na Lacertidae Bonaparte, 1838 (Reptilia) - Wiad. Parazytol., 33(2): 229-230.
House S.M., Taylor P.J., Spellerberg I.F. 1980 - Patterns of Daily Behaviour in Two Lizard Species Lacerta agilis L. and Lacerta vivipara Jacquin. Oecologia, 44: 396-402. doi:10.1007/BF00545244 ![]()
Jones K.E., Patel N.G., Levy M.A, Storeygard A., Balk D., Gittleman J.L., Daszak P. 2008 - Global trends in emerging infectious diseases - Nature Letters, 451: 990-994. doi:10.1038/nature06536 ![]()
Kurczewski R. 2014 - Jaszczurka zwinka (Lacerta agilisLinnaeus, 1758) w Wielkopolskim Parku Narodowym - Bogucki Wydawnictwo Naukowe, Poznań, pp. 85.
Majláthová V., Majláth I., Haklová B., Hromada M., Ekner A., Antczak M., Tryjanowski P. 2010 - Blood parasites in two co-existing species of lizards (Zootoca vivipara and Lacerta agilis) - Parasitol. Res., 107: 1121-1127. doi:10.1007/s00436-010-1981-0 ![]()
Matuschka F.R., Fischer P., Musgrave K., Richter D., Spielman A. 1991 - Hosts on which nymphal Ixodes ricinus most abundantly feed - Am. J. Trop. Med. Hyg., 44: 100-107. doi:10.4269/ajtmh.1991.44.100 ![]()
Nuland G.J. van, Strijbosch H. 1981 - Annual Rhythmics of Lacerta vivipara Jacquin and Lacerta agilis agilis L. (Sauria, Lacertidae) in the Netherlands. Amphibia-Reptilia, 2(1): 83-95. doi:10.1163/156853881X00302 ![]()
Oppliger A., Clobert J. 1997 - Reduced tai regeneration in the Common Lizard, Lacerta vivipara, parasitized by blood parasites - Functional Ecology, 11: 652-655. doi:10.1046/j.1365-2435.1997.00134.x ![]()
Rahmel U., Meyer S. 1988 - Populationsökologische Daten von Lacerta agilis argus (Laurenti, 1786) aus Niederöoserreich - In: Glandt D., Bischof W. (Eds.). Biologie und Schutz der Zauneidechse (Lacerta agilis). Martensiella 1, p. 220-234.
Siuda K. 1993 - Kleszcze Polski (Acari: Ixodida). Część 2. Systematyka i rozmieszczenie - Polskie Towarzystwo Parazytologiczne, Warszawa, pp. 381.
Strijbosch H., Rooy P.Th.J.C. van, Voesenek L.A.C.J. 1983 - Homing behaviour of Lacerta agilis and Lacerta vivipara (Sauria, Lacertidae). Amphibia-Reptilia, 4(1): 43-47. doi:10.1163/156853883X00256 ![]()
Tälleklint-Eisen L., Eisen R.J. 1999 - Abundance of ticks (Acari: Ixodidae) infesting the western fence lizard, Sceloporus occidentalis, in relation to environmental factors - Exp. Appl. Acarol., 23(9): 731-740.
Vacláv R., Prokop P., Fekiač V. 2007 - Expression of breeding coloration in European Green Lizard (Lacerta viridis): variation with morphology and tick infestation - Can. J. Zool., 85: 1199-1206. doi:10.1139/Z07-102 ![]()
Weallers W.W., Marganeidge K.R. 1971 - Cutaneous vascular responses to temperature change in the spiny-tailes Iguana, Ctenosaura cemilopha - Copeia, 3: 548-551. doi:10.2307/1442454 ![]()
White F.N. 1976 - Circulation - In: Gans C., Dawson W.R. (Eds.). Biology of the Reptilia, vol. 5, Academic Press, p. 275-334.
Wolfinger R.D. 1993 - Covariance Structure Selection in General Mixed Models - Commun. Stat. - Simul. Comput., 22(4): 1079-1106. doi:10.1080/03610919308813143 ![]()



2018-01-15
Date accepted:
2020-04-24
Date published:
2020-04-30
Edited by:
Duncan, Alison

This work is licensed under a Creative Commons Attribution 4.0 International License
2020 Gwiazdowicz, Dariusz J.; Gdula, Anna K.; Kurczewski, Rafał and Zawieja, Bogna
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



