Diversity and abundance of Phytoseiidae (Acari: Mesostigmata) on horse chestnut (Aesculus hippocastanum L.) in an urban environment: a comparison between Greece and the Czech Republic
Kopačka, Michal1 ; Stathakis, Theodoros I.2 ; Broufas, Georgios3 ; Papadoulis, Georgios Th.4 and Zemek, Rostislav5
1University of South Bohemia, Faculty of Agriculture, Studentská 13, 37005 České Budějovice, Czech Republic.
2Agricultural University of Athens, Laboratory of Agricultural Zoology and Entomology, Iera Odos 75, 118 55 Athens, Greece.
3Democritus University of Thrace, Department of Agricultural Development, Pantazidou 193, 68 200 Orestiada, Greece.
4Agricultural University of Athens, Laboratory of Agricultural Zoology and Entomology, Iera Odos 75, 118 55 Athens, Greece.
5✉ Arthropod Ecology and Biological Control Research Group, Ton Duc Thang University, Ho Chi Minh City, Vietnam.
6Faculty of Applied Sciences, Ton Duc Thang University, Ho Chi Minh City, Vietnam.
7Biology Centre of the Czech Academy of Sciences, Institute of Entomology, Branišovská 31, 37005 České Budějovice, Czech Republic.
2018 - Volume: 58 Issue: Suppl pages: 83-90
https://doi.org/10.24349/acarologia/20184284Keywords
Abstract
Horse chestnut, Aesculus hippocastanum L. (Sapindaceae), is a large deciduous tree native to a small area in the Pindus Mountain mixed forests and the Balkan mixed forests of Southeast Europe. It was introduced to most countries of Europe for mainly ornamental purposes. Its ornamental function has been negatively affected by the horse chestnut leaf miner, Cameraria ohridella Deschka & Dimic (Lepidoptera: Gracillariidae), in the last two decades. This species was recorded at the Ohrid Lake in F.Y.R.O.M. in 1985 for the first time and became an invasive pest when it spread through Austria to the entire territory of Europe and Asia Minor. In the Czech Republic, this species appeared in 1993 (Liška, 1997). This species may damage up to 93% of the leaf area in an urban environment. Another cause of mostly aesthetic damage is the horse chestnut leaf blotch, Guignardia aesculi (Peck) V.B. Stewart (Botryosphaeriales: Botryosphaeriaceae). This fungal disease was uncovered in the Czech Republic in the 1950s (Scaramuzzi 1954) and has been reported in other European countries, the USA and Asia (Pastirčáková et al., 2009)
In addition to the ornamental function of A. hippocastanum, this tree might also serve as an important reservoir of predatory mites of the family Phytoseiidae (Acari: Mesostigmata) which spread to apple orchards or vineyards (Tuovinen and Rokx, 1991; Tuovinen, 1994). While a survey of phytoseiid mites on this tree species has already been conducted in the Czech Republic (Kabíček and Řeháková, 2004), and some data have also been reported for Finland (Tuovinen and Rokx, 1991), no such study has been conducted in a country where horse chestnut is an autochthonous tree. Therefore, the aim of the present study was to compare the occurrence, species composition and population densities of Phytoseiidae on introduced A. hippocastanum in Central Europe with that on the tree species in Greece. Since some phytoseiid species feed on fungi (Zemek and Prenerová, 1997; Duso et al., 2003; Zemek, 2005; Pozzebon et al., 2009), we hypothesize that the population density of phytoseiids might be positively correlated with the occurrence of the fungal pathogen G. aesculi. A higher infestation by C. ohridella might also support a phytoseiid population by providing more shelters similar to abandoned mines of the citrus leaf miner, Phyllocnistis citrella Stainton (Lepidoptera: Gracillariidae) (Villanueva and Childers, 2011). Therefore, we also tested whether G. aesculi and C. ohridella had any effect on the population density of phytoseiid mites.
The survey was conducted in the cities of České Budějovice, South Bohemia, Czech Republic (48° 59′ N, 14° 29′ E) and Orestiada, northeastern Greece (41° 30′ N, 26° 32′ E). České Budějovice has a population of approximately 100,000 inhabitants and an urban area of 55 km2 with the total number of A. hippocastanum trees in the town estimated to be 534. The trees are located mostly in city parks, green belts along rivers and streams and open space (Kopačka and Zemek, 2017). Orestiada is a smaller city with a population of approximately 20,000 inhabitants and an urban area of 39 km2 with approximately 83 A. hippocastanum trees, mostly found in alleys. Both towns are surrounded by agricultural fields, grasslands and woods.
In České Budějovice, thirty horse chestnut trees were randomly selected at the Vltava estate site (area approx. 92 ha, geometric center at 48.9962106N, 14.4519647E) for the purpose of this study. The mean age of horse chestnut trees at this site, estimated from the trunk perimeter using the method described by Jura (2001), was 41.8 years (Kopačka and Zemek, 2017). In Orestiada, A. hippocastanum trees were randomly selected in an area of approx. 130 ha with a geometric center at 41.5071667N, 26.5295267E. The estimated mean age of the trees was 28.2 years.
The population of Phytoseiidae was assessed on thirty randomly selected compound leaves collected from tree branches up to 2.5 m above ground. Only one compound leaf from a single tree was collected. The sampling was conducted on 13th September 2013 and 30th September 2015 in České Budějovice and Orestiada, respectively.
First, the percentage of leaf area damaged by C. ohridella, G. aesculi or another cause, e.g., bird predation on C. ohridella larvae and pupae, or mechanical damage from weather-related events was estimated by visual method (Gilbert and Grégoire, 2003). Then, all mites were collected from individual leaves using a washing technique (Zacharda et al., 1988). This was done by splitting each compound leaf into individual leaflets, which were in turn placed in a sealable glass jar with 85% ethanol in which mites were washed off. The collected mites were stored in small glass vials (one vial per leaf sample) with 85% ethanol and then mounted on temporal microscope slides in lactic acid. The mites were identified using the keys of Beglyarov (1981a,b) and Miedema (1987).
All data were stored in a Microsoft Access 2010 database. The abundance of phytoseiid mites was expressed as the mean number of mites per composed A. hippocastanum leaf. The species diversity was quantified using Simpson's Diversity Index (Adams, 2009):
$D = 1 - ((Σ i ni (ni -1))/ N(N-1))$,
where n is the total number of organisms of a particular species, and N is the total number of organisms of all species; the value of the index ranges between 0 and 1; the greater the value, the greater the species diversity. The Sörensen's index (QS) or similarity coefficient (Southwood, 1968) was used to evaluate β-diversity: QS = 2j/(a+b), where a is the number of species in locality A, b the number of species in the locality B and j is the number of common species in both localities; the value of the index ranges between 0 and 1. The coefficient of constancy (C) (Dajoz, 1977) was used to indicate the frequency of different species in the studied localities: C(%) = Na / N × 100, where Na is the number of samples with the species a, and N is the total number of samples. The species were classified as accidental (C < 25%), accessory (C = 25–50%), constant (C = 50–75%) or euconstant (C > 75%) (Dajoz, 1977). The obtained leaf damage data were statistically analyzed using the Mann-Whitney U test (Siegel and Castellan, 1988). The significance was set at α = 0.05 with the Holm-Bonferroni correction applied for multiple comparisons (Holm, 1979). The abundance of phytoseiid mites was analyzed by ANCOVA with location as a main factor and the leaf damage inflicted by C. ohridella and G. aesculi and mechanical damage as continuous predictors. Two-tailed probabilities were used. All statistical analyses were performed by STATISTICA v. 12 software (StatSoft Inc., 2013).
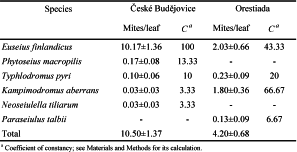
The estimated percentage of leaf area damaged by C. ohridella, G. aesculi or other causes in České Budějovice ranged from 0% to 10%, 2% to 25%, and 0% to 5%, respectively. The highest mean leaf damage (6.73%) was caused by the fungal pathogen (Table 1). The leaf damage in Orestiada ranged from 0% to 50%, 0% to 25% and 0% to 10% for C. ohridella, G. aesculi and other causes, respectively. On average, the damaged leaf area was 4.20%, 4.23% and 1.40%, respectively. The Mann-Whitney U test revealed statistically significant differences between localities in damage inflicted by G. aesculi (Table 1).



A total of 441 specimens of phytoseiid mites was collected from both countries. Phytoseiidae were found in all leaf samples in České Budějovice. The population density of mites ranged between 1 and 28, and the mean number of mites per leaf was 10.5. In contrast, in Orestiada, 10% of the samples were devoid of phytoseiid mites; the maximum number of mites in a sample was 16 mites, and on average, there were 4.2 phytoseiid mites per leaf. The ANCOVA revealed a highly significant effect of locality on phytoseiid mite abundance (F1,55=15.973, P=0.0002). The effects of covariates were, however, not statistically significant (F1,55=0.029, P=0.865; F1,55=0.853, P=0.360 and F1,55=0.367, P=0.547) for the leaf damage caused by C. ohridella, G. aesculi or other causes, respectively.
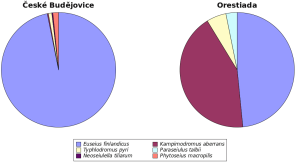
The following five species of phytoseiid mites were found in České Budějovice: Euseius finlandicus (Oudemans), Kampimodromus aberrans (Oudemans), Neoseiulella tiliarum (Oudemans), Phytoseius macropilis (Banks) and Typhlodromus (Typhlodromus) pyri Scheuten (Table 2). While E. finlandicus was present in all samples (C=100%), K. aberrans and N. tiliarum were found in only one sample (C=3.33%). In Orestiada, samples were devoid of N. tiliarum and P. macropilis, while another species, Paraseiulus talbii (Athias-Henriot), was found in two samples (C=6.67%). This species composition resulted in rather high similarity (QS=0.667) between studied sites. The predominant species in České Budějovice was E. finlandicus, representing 96.8% of all specimens (Figure 1). In Orestiada, the following two species were predominant: E. finlandicus (48.4%) and K. aberrans (42.9%). The Simpson’s Diversity Index was therefore higher for Greece (0.583) than for the Czech Republic (0.062).




Our study revealed a high population density of Phytoseiidae on A. hippocastanum, particularly in the Czech Republic where there were more than ten mites per composed leaf; i.e., approximately two mites per single leaflet were found. Kabíček and Řeháková (2004) reported the highest population density of phytoseiids on horse chestnut trees to be 3.3 mites per leaflet on average at the Prokopské údolí site, and the lowest to be 2.5 mites per leaflet at the Letenské sady site in Prague. The abundance of phytoseiid mites on A. hippocastanum in Orestiada was significantly lower, which might be attributed to prey availability, different abiotic conditions or less greenery in the city making the trees more isolated. In regard to prey associated with A. hippocastanum, Ripka and de Lillo (1997) reported Aculus hippocastani (Fockeu) and Shevtchenkella carinatus (Nalepa) (Acari: Eriophyidae), and Gyenis et al. (2005) reported Eotetranychus pruni (Oudemans) (Acari: Tetranychidae), infesting horse chestnut. Only a few phytophagous mites were collected in the samples from České Budějovice. Sampling efforts in the present study, however, did not focus on the eriophyid fauna. Availability of alternative food, e.g., pollen or fungi on leaves, might be another reason for the above differences. Aspects of the host plant are also considered to influence the behavior and distribution of phytoseiids independently of prey availability (Beard and Walter, 2001). The presence of leaf structures such as vein trichomes and leaf domatia is known to have a positive effect on the number of microarthropods, including phytoseiid mites (O’Dowd and Willson, 1989; Walter and O’Dowd, 1992; Karban et al., 1995; Kreiter et al., 2002; Schmidt, 2014), but no apparent differences in these leaf structures between the studied localities were seen by the naked eye.
We were also interested in whether there was any effect of C. ohridella or G. aesculi infestation on the abundance of Phytoseiidae. There could be either a negative effect due to leaf damage, thus reducing leaf area for herbivorous mites, or a positive effect when, for example, C. ohridella eggs could serve as an alternative food source. Grabenweger et al. (2005) studied whether E. finlandicus is able to utilize these eggs as a food source but observed no feeding. Mine-damaged A. hippocastanum leaves can also provide refugia to phytoseiid mites with more favorable microenvironment conditions than leaves without C. ohridella damage. In fact, they may function in a similar way to domatia. For example, in apples, Villanueva and Harmsen (1996) observed the phytoseiids Typhlodromus caudiglans Schuster and Neoseiulus fallacis (Garman) in old mines produced by the spotted tentiform leaf miner, Phyllonorycter blancardella Fabricius (Lepidoptera: Gracillariidae). A positive effect of the infestation by a leaf miner on the population of phytoseiid mites was reported by Villanueva and Childers (2011) who found that Phytoseiidae were significantly more abundant on mature grapefruit leaves mined by P. citrella than on mature leaves without mines. Our results, however, did not confirm such an effect. The leaf area damaged by the leaf blotch was significantly higher in České Budějovice than that in Orestiada, which is likely due to favorable abiotic conditions, mainly humidity. Since fungivory in some phytoseiid species was well-documented, for example T. pyri, E. finlandicus and K. aberrans readily feed on several mildew species (Kropczyńska-Linkiewicz, 1971; Zemek and Prenerová, 1997; Duso et al., 2003; Zemek, 2005; Pozzebon et al., 2009), we expected that they could utilize G. aesculi as a food source as well. No correlation between mite density and the leaf blotch damage was, however, confirmed in this study.
Five species were identified among specimens in České Budějovice and four in Orestiada, while three species were common in both sites. This resulted in a relatively high Sörensen's index indicating certain similarity between sites. Kabíček and Řeháková (2004) reported six phytoseiid species in total on A. hippocastanum found in Prague, but the species richness varied among investigated sites from one to four species. The authors claim that this variability may be determined by the different locality conditions in the city. Contrary to the present study, P. macropilis was not present; however, Galendromus longipilus (Nesbitt) and Neoseiulella aceri (Collyer) were found. Six species of the Phytoseidae were found on horse chestnut in Hungary (Ripka, 2006). They include two species which were not present in our samples: Amblyseius andersoni (Chant) and Paraseiulus triporus (Chant and Yoshida-Shaul). The most numerous species found in both České Budějovice and Orestiada was E. finlandicus. This species was found on 68% of surveyed trees in the park of the Czech Agricultural University, Prague (Kabíček and Koubková, 1998), while it represented approximately 96% of all specimens found on investigated deciduous trees in the Prague urban forest at Kunratice (Kabíček and Povondrová, 2004). E. finlandicus was also the predominant species on A. hippocastanum where it represented almost 98% of all phytoseiid specimens found (Kabíček and Řeháková, 2004), which is very close to our results. In Greece, however, another species, K. aberrans, is nearly as abundant as E. finlandicus; therefore, the Simpson’s diversity index, which takes into account both species richness and evenness, was higher in Greece than in the Czech Republic. We hypothesize that this is mainly due to different climate conditions.
We can conclude that (1) horse chestnut trees represent favorable host plants for phytoseiid mites with E. finlandicus as the predominant species in both countries, (2) higher abundance of Phytoseiidae on A. hippocastanum but lower Simpson’s diversity index was found in the Czech Republic compared to Greece, and (3) no significant effect of horse chestnut leaf miner or leaf blotch on population density of phytoseiid mites was confirmed.
This research was conducted with institutional support RVO: 60077344. The publication was supported by projects No. 8G15006 from the Ministry of Education, Youth and Sports of the Czech Republic and No. 063/2013/Z from the Grant Agency of the University of South Bohemia in České Budějovice. We would like to thank Prof. Jaroslav Boháč for the valuable advice during the study and Mrs. Barbora Kozelková for her technical assistance. We also appreciate the helpful comments and suggestions for improvement of the original version of this article by two anonymous referees. The authors would like to thank American Journal Experts (www.aje.com) for the English language review.
Adams J. 2009. Species richness: patterns in the diversity of life. Berlin, Heidlberg, New York: Springer. pp. 386. doi:10.1007/978-3-540-74278-4 ![]()
Beard J.J., Walter G.H. 2001. Host plant specificity in several species of generalist mite predators. Ecol. Entomol., 26: 562–570. doi:10.1046/j.1365-2311.2001.00367.x ![]()
Beglyarov G.A. 1981a. Opredelitel chiscnych klescej fitoseiid (Parasitiformes, Phytoseiidae) fauny SSSR. Leningrad: Infor. Bull EPS IOBC. pp. 1–95 (in Russian).
Beglyarov G.A. 1981b. Opredelitel chiscnych klescej fitoseiid (Parasitiformes, Phytoseiidae) fauny SSSR. Leningrad: Infor. Bull EPS IOBC. pp. 1–45 (in Russian).
Dajoz R. 1977. Introduction to ecology. London: Hodder & Stoughton.
Duso C., Pozzebon A., Capuzzo C., Bisol P.M., Otto S. 2003. Grape downy mildew spread and mite seasonal abundance in vineyards: evidence for the predatory mites Amblyseius andersoni and Typhlodromus pyri. Biol. Control, 27: 229–241. doi:10.1016/S1049-9644(03)00016-1 ![]()
Gilbert M., Grégoire J.C. 2003. Visual, semi-quantitative assessments allow accurate estimates of leafminer population densities: An example comparing image processing and visual evaluation of damage by the horse chestnut leafminer Cameraria ohridella (Lep., Gracillariidae). J. Appl. Entomol., 127: 354–359. doi:10.1046/j.1439-0418.2003.00757.x ![]()
Grabenweger G., Kehrli P., Schlick-Steiner B., Steiner F., Stolz M., Bacher S. 2005. Predator complex of the horse chestnut leafminer Cameraria ohridella: identification and impact assessment. J. Appl. Entomol., 129: 353–362. doi:10.1111/j.1439-0418.2005.00973.x ![]()
Gyenis K., Pénzes B., Hegyi T. 2005. Phytophagous and predatory mites on the horse chestnut tree. Novenyvedelem, 41: 143–148.
Holm S. 1979. A simple sequentially rejective multiple test procedure. Scand. J. Stat., 6: 65–70.
Jura S. 2001. Určování stáří stromů. Silva Bohemica, 1: 19–20 (in Slovak).
Kabíček J., Koubková Z. 1998. Phytoseiid mites on plants of a city park. Plant Protect. Sci., 34: 142–145.
Kabíček J., Povondrová K. 2004. Phytoseiid mite communities on urban deciduous trees. Acta Fytotech. Zootech., 7: 119–120.
Kabíček J., Řeháková M. 2004. Phytoseiid mite community on Aesculus hippocastanum in the parks. Acta Fytotech. Zootech., 7: 114–115.
Karban R., English-Loeb G., Walker M.A., Thaler J. 1995. Abundance of phytoseiid mites on Vitis species: effects of leaf hairs, domatia, prey abundance and plant phylogeny. Exp. Appl. Acarol., 19: 189–197. doi:10.1007/BF00130822 ![]()
Kopačka M., Zemek R. 2017. Spatial variability in the level of infestation of the leaves of horse chestnut by the horse chestnut leaf miner, Cameraria ohridella (Lepidoptera: Gracillariidae) and in the number of adult moths and parasitoids emerging from leaf litter in an urban environment. Eur. J. Entomol., 114: 42–52. doi:10.14411/eje.2017.007 ![]()
Kreiter S., Tixier M.S., Croft B.A., Auger P., Barret D. 2002. Plants and leaf characteristics influencing the predaceous mite, Kampimodromus aberrans (Oudemans), in habitats surrounding vineyards (Acari: Phytoseiidae). Environ. Entom., 31: 648–660. doi:10.1603/0046-225X-31.4.648 ![]()
Kropczyńska-Linkiewicz D. 1971. Studies on the feeding of four species of phytoseiid mites (Acarina: Phytoseiidae). In: Daniel R., Rosicky B. (Eds.), Proceedings of the Third International Congress on Acarology. Czechoslovak Academy of Sciences, Prague, August 31–September 6, 1971, pp. 225–227.
Liška J. 1997. Verbreitung Der Roβkastanienminiermotte in Tschechien. Forstschutz-Aktuell, 21: 5.
Miedema E. 1987. Survey of phytoseiid mites (Acari: Phytoseiidae) in orchards and surrounding vegetation of northwestern Europe, especially in the Netherlands. Keys, descriptions and figures. Neth. J. Pl. Path., 93: 1–64. doi:10.1007/BF01984462 ![]()
O'Dowd D.J., Willson M.F. 1989. Leaf domatia and mites on Australasian plants: ecological and evolutionary implications. Biol. J. Linnean Soc., 37: 191–236. doi:10.1111/j.1095-8312.1989.tb01901.x ![]()
Pastirčáková K., Pastirčák M., Celar F., Shin H. 2009. Guignardia aesculi on species of Aesculus: new records from Europe and Asia. Mycotaxon, 108: 287–296.
Pozzebon A., Loeb G.M., Duso C. 2009. Grape powdery mildew as a food source for generalist predatory mites occurring in vineyards: effects on life-history traits. Ann. Appl. Biol., 155: 81–89. doi:10.1111/j.1744-7348.2009.00323.x ![]()
Ripka G., de Lillo E. 1997. New data to the knowledge on the eriophyoid fauna in Hungary (Acari: Eriophyoidea). Folia Ent. Hung., 58: 147–157.
Ripka G. 2006. Checklist of the Phytoseiidae of Hungary (Acari: Mesostigmata). Folia Ent. Hung., 67: 229–260.
Scaramuzzi, G. 1954. Sul seccume delle foglie d'Ippocastano. Ann. Sper. Agr., 8: 1265–1281.
Schmidt, R.A. 2014. Leaf structures affect predatory mites (Acari: Phytoseiidae) and biological control: a review. Exp. Appl. Acarol., 62: 1–17. doi:10.1007/s10493-013-9730-6 ![]()
Siegel S., Castellan N. J. J. 1988. Nonparametric statistics for the behavioral sciences. New York: McGraw-Hill. pp. 399.
Southwood T.R.E. 1968. Ecological methods with particular references to study of insect populations. Methuen: London.
StatSoft Inc. 2013: STATISTICA v. 12.0. Data Analysis Software System. StatSoft Inc., Tulsa, OK, USA.
Tuovinen T., Rokx J.A.H. 1991. Phytoseiid mites (Acari: Phytoseiidae) on apple trees and in surrounding vegetation in southern Finland. Densities and species composition. Exp. Appl. Acarol., 12: 35–46. doi:10.1007/BF01204398 ![]()
Tuovinen T. 1994. Influence of surrounding trees and bushes on the phytoseiid mite fauna on apple orchard trees in Finland. Agr. Ecosyst. Environ., 50: 39–47. doi:10.1016/0167-8809(94)90123-6 ![]()
Villanueva R.T., Harmsen R. 1996. Ecological interactions of tarsonemid mites in apple orchards: predation of apple rust mite and use of Phyllonorycter blancardella mines. Proc. Entomol. Soc. Ontario, 127: 99–106.
Villanueva R.T., Childers C.C. 2011. Mine-damaged leaves by Phyllocnistis citrella Stainton provide refuge for phytoseiids on grapefruit in Florida and Texas. In: Moraes G.J. de, Proctor H. (eds) Acarology XIII: Proceedings of the International Congress. Zoosymposia, 6: 118–123.
Walter D.E., O'Dowd D.J. 1992. Leaves with domatia have more mites. Ecology, 73: 1514–1518.
Zacharda M., Pultar O., Muška J. 1988. Washing technique for monitoring mites in apple orchards. Exp. Appl. Acarol., 5: 181–183. doi:10.1007/BF02053827 ![]()
Zemek R. 2005. The effect of powdery mildew on the number of prey consumed by Typhlodromus pyri (Acari : Phytoseiidae). J. Appl. Entomol., 129: 211–216. doi:10.1111/j.1439-0418.2005.00947.x ![]()
Zemek R., Prenerová E. 1997. Powdery mildew (Ascomycotina: Erysiphales) – an alternative food for the predatory mite Typhlodromus pyri Scheuten (Acari: Phytoseiidae). Exp. Appl. Acarol., 21: 405–414. doi:10.1023/A:1018427812075 ![]()



2016-10-26
Date accepted:
2018-01-23
Date published:
2018-09-28
Edited by:
Tixier, Marie-Stéphane

This work is licensed under a Creative Commons Attribution 4.0 International License
2018 Kopačka, Michal; Stathakis, Theodoros I.; Broufas, Georgios; Papadoulis, Georgios Th. and Zemek, Rostislav
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



