Re-descriptions of Amblyseius decolor (Westerboer) and Proprioseiopsis sororculus (Wainstein) (Acari: Phytoseiidae) based on the specimens collected in Turkey and France
Çobanoğlu, Sultan1 ; Faraji, Farid2 and Cılbırcıoğlu, Cihan3
1✉ Ankara University, Department of Plant Protection, Faculty of Agriculture, 06110 Ankara, Turkey.
2MITOX Consultants, P. O. Box 92260, 1090 AG Amsterdam, The Netherlands.
3University of Kastamonu, Taşköprü Vocational School Organic Agriculture Department, Turkey.
2018 - Volume: 58 Issue: 4 pages: 825-836
https://doi.org/10.24349/acarologia/20184272ZooBank LSID: 675A452A-38BB-4433-8A43-12439C746338
Keywords
Abstract
More than 2798 species of the family Phytoseiidae have been described and among them 2521 are known valid (Demite et al. 2018). Some members of the family Phytoseiidae are considered among the most important agents in pest biological control. These beneficial arthropods have been used successfully against the injurious mites and small insects like thrips and whiteflies (Gerson et al. 2003; Zhang 2003; Faraji et al. 2011, McMurtry et al. 2013). Considerable studies have been made in Turkey to record the species of Phytoseiidae and so far 97 phytoseiid species belonging to 19 genera have been recorded (Düzgüneş 1963, Moraes et al. 1986, 2004, Faraji et al. 2011, Döker et al. 2016, 2017; Demite et al. 2018). During a survey between 2014 and 2016 on determining the mites inhabiting garlic plants and those associated with garlic bulbs in storage, we have found two phytoseiid species new for the fauna of Turkey. The aim of this paper is to re-describe these two new species records and compare them morphologically with the species collected in France. We also present a key for the species in tribe Amblyseiini recorded from Turkey.
The mite samples were collected from garlic bulbs in storages or garlic plants in garlic fields of Kastamonu region, which is located North-eastern part of Turkey. The predatory mites were extracted using Berlese funnel set-up, cleared in a mixture of lactophenol: Nesbitt 1:1 and mounted in the Hoyer’s medium on microscope slides. The notations used for dorsal and ventral setations follow those of Lindquist and Evans (1965) as adapted by Rowell et al. (1978) and Chant and Yoshida-Shaul (1991), respectively. The notation for gland pores (solenostomes) or lyrifissures (poroids) is according to Athias-Henriot (1975). All measurements are given in micrometers (µm). The mean of the measurements is given first followed by the range in parentheses. The voucher specimens of species re-described here are deposited in the mite collection of the Department of Plant Protection (University of Ankara, Turkey) and MITOX consultants/Eurofins (Amsterdam, Netherlands).
Typhlodromus decolor Westerboer, in Hirschmann, 1962: 25.
Typhlodromus (Typhlodromus) decolor — Westerboer & Bernhard, 1963: 654.
Typhlodromips decolor — Moraes et al., 1986: 139, 2004: 211.
Amblyseius (Typhlodromips) decolor — Karg, 1991.
Amblyseius decolor — Chant & McMurtry, 2007: 78; Prasad, 2012: 393; Demite et al., 2018.
Amblyseius lutezhicus Wainstein, 1972: 1408. (new synonymy).



Idiosomal setal pattern: 10A:9B/JV–3:ZV.
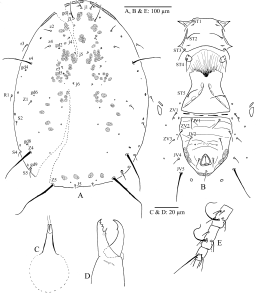
Dorsal idiosoma (Figure 1A) — Dorsal shield 340 long and 200 wide at j6 level, mostly smooth with some striae laterally; with 19 pairs of dorsal setae (r3 and R1 included), dorsal setae smooth, except for Z4 and Z5, slightly serrate; lengths j1 28, j3 48, j4 5, j5 5, j6 5, J2 13, J5 8, z2 10, z4 38, z5 5, Z1 8, Z4 90, Z5 98, s4 78, S2 25, S4 18, S5 13; setae r3 15 and R1 18 on lateral integument; dorsal shield with 6 pairs of large solenostomes (gd1, gd2, gd4, gd6, gd8 and gd9), and 10 pairs of poroids.
Peritreme — Extending beyond the insertions of setae j1 (Figure 1A).
Ventral idiosoma (Figure 1B) — Sternal shield mostly smooth with some slight striae anterior and laterally and with faint lines medially, 65 long and 75 wide at level of setae ST2, with two pairs of poroids (iv1 and iv2) and three pairs of setae, ST1 20, ST2 28, ST3 25; distances between ST1–ST3 60 and ST2–ST2 66; metasternal setae ST4 23 and a pair of poroids (iv3) on small platelets; genital shield smooth at middle and with a few striae laterally, width 70 at widest point, ST5 (18) distances between ST5–ST5 71; two pairs of metapodal shields, primary 25 and accessory 13 long; ventrianal shield subpentagonal, with some striae between JV1 and paranals; length 118, width at level of setae ZV2 95 and width at level of paranal setae 92; with three pairs of preanal setae (JV1 18, JV2 23, ZV2 18); four pairs of setae surrounding ventrianal shield on integument (JV4 13, JV5 50, ZV1 13, ZV3 13); ventrianal shield with a pair of non-aligned small round pores posteriad to JV2, and muscle marks posterolaterally, distance between these pores 38 and the distance between insertions of JV2–JV2 48.
Spermatheca — Calyx very long 60 snake-shaped, atrium incorporated in the calyx whose walls are thickened at the anterior tip, calyx gets Λ-shaped one-fourths of its length towards vesicle, minor duct not visible (Figure 1C).
Chelicera — Fixed digit 28 long with 6 small teeth and pilus dentilis; movable digit 25 long with two widely separated teeth (Figure1D).
Legs — Leg IV (Figure 1E) with three macrosetae, all pointed apically, SgeIV 60, StiIV 40, StIV 75; distance from base of macrosetae to slit like organ 58, chaetotatic formulae of genua and tibiae I–II–III–IV with 10(2-2/1, 2/1-2) – 8(2-2/1, 2/0-1) – 7(1-2/1, 2/0-1) – 7(1-2/1, 2/0-1) and 10(2-2/1, 2/1-2) – 7(1-1/1, 2/1-1) – 7(1-1/1, 2/1-1) – 6(1-1/1, 2/0-1) setae respectively.
Specimen examined — 1♀, 14 August 2015, stored garlic bulbs, Taşköprü-Kastamonu (41°31′00.94″N 34°11′46.74″E) collected by Cihan Cılbırcıoğlu.



Due to the similarities in features with the Turkish specimen, only the measurements are provided.
Dorsal idiosoma (Figure 2A) — Dorsal shield 332 (330–340) long and 210 (200–220) wide at j6 level; setal lengths: j1 23 (18–28), j3 34 (33–38), j4 4 (3–5), j5 4 (3–5), j6 4 (3–5), J2 7 (5–8), J5 10 (9–10), z2 8 (8–10), z4 19 (13–25), z5 3, Z1 9 (8–10), Z4 68 (63–70), Z5 73 (68–80), s4 64 (60–68), S2 18 (15–20), S4 12 (10–15), S5 11 (8–13); setae r3 13 (10–15) and R1 11 (8–15) on lateral integument.
Ventral idiosoma (Figure 2B) — Sternal shield 59 (58–63) long and 73 (68–75) wide at level of setae ST2; ST1 33 (33–35), ST2 25 (23–28), ST3 26 (23–28); distances between ST1-ST3 58–63 and ST2–ST2 66–70; ST4 29 (23–39); genital shield width at widest point 72 (68–75) and ST5 25 (20–28); distances between ST5-ST5 69–75; primary metapodal shields 23 (18–25) and accessory 19 (18–20) long; ventrianal shield length 109 (103–115), width at level of setae ZV2, 94 (93–105), and 94 (93–98) width at level of paranal setae; setae JV1 17 (15–20), JV2 21 (20–23), ZV2 16 (13–18), JV4 10 (8–10), JV5 46 (43–50), ZV1 17 (15–20), ZV3 11 (8–13); distance between preanal pores 29 (25–38) and JV2–JV2 45 (40–51).
Spermatheca — Calyx very long 51 (48–58) (Figure 2C & 4A).
Chelicera — Fixed digit 32 (28–38) and movable digit 27 (25–30) long with two widely separated teeth (Figure 2D).
Legs — Leg IV (Figure 2E) SgeIV 54 (48–60), StiIV 40 (33–48), StIV 61 (60–65); distance from base of macrosetae to slit like organ 58 (53–60).
Specimens examined — 2♀♀, 21 August 2003; 3♀♀ 17 October 2003 Buzet, Lot-et-Garonne, Southwest France (44°15′53.6″N 0°17′57.5″E), ground weeds (apple orchard), Mitox, collected by F. Bakker.
Distribution — Armenia (Wainstein, 1977); France (Tixier et al., 2000); Moldova (Kolodochka, 1980), Portugal (Espinha et al., 1998); Spain (Hirschmann, 1962; Escudero & Ferragut, 1998); Turkey (this study) and Ukraine (Wainstein, 1972).
Remarks — This is the first report of A. decolor from Turkey. Most of the morphological features of the specimen found in Turkey are similar to those collected in France as well as in the original descriptions of A. decolor and A. lutezhicus. One of the noticeable differences is the length of z4 and its ratio to the distance between insertions of z4 and s4. Seta z4 reaching the insertion of s4 in the Turkish specimen while for the French specimens and specimen re-described by Ferragut et al. (2010) that is half and for the specimens collected in Ukraine (Wainstein, 1972) is almost one-thirds. The Turkish specimen has longer some dorsal shield setae as mentioned in Table 1. This observation shows to some extends variability in setal lengths in this species. The shape of spermatheca in A. decolor is very unique and does share it with two other closely related species, which are assigned in another genus (Transeius). Transeius infundibulatus (Athias-Henriot, 1961) and T. macrospermathecus Papadoulis, Emmanouel & Kapaxidi, 2009 described from Spain and Greece, respectively show a close affinity with A. decolor in many respects. Ignoring slightly longer S2 seta, the specimen of A. decolor collected in Turkey resembles T. macrospermathecus in all respects and the minor differences fall within intraspecific variation. It is astonishing enough to find that these two species suspected being conspecific classified in two different genera: Amblyseius and Transeius. In another study, Tsolakis et al. (2012) have shown that Transeius montdorensis (Schicha) is closely related to Amblyseius andersoni (Chant) and A. swirskii Athias-Henriot. Therefore, there would be a question about the integrity of the genus Amblyseius and the validity of the genus Transeius. A molecular study on A. decolor, T. macrospermathecus and T. infundibulatus or more collection of specimens from Europe might reveal these species conspecific. If these species turn to be conspecific then T. infundibulatus would be the valid name.



For other names and synonyms see Demite et al., 2018.


Idiosomal setal pattern: 10A:8E/JV–3:ZV.
Dorsal idiosoma (Figure 3A) — Dorsal shield smooth 407 (360–430) long, 264 (250–270) wide at j6 level, with 18 pairs of dorsal setae (r3 and R1 included); dorsal shield setae smooth, except for Z5, slightly serrated; lengths j1 26 (25–28), j3 40 (38–42), j4 5, j5 4 (3–5), j6 4 (3–5), J5 7 (5–8), z2 17 (15–18), z4 11 (10–12), z5 4 (3–5), Z1 7 (5–10), Z4 82 (78–85), Z5 100 (95–105), s4 67 (66–68), S2 9 (8–10), S4 8, S5 7 (5–8); setae r3 18 (16–20) and R1 13 (12–15) on lateral integument; Z4 and Z5 are the longest, dorsal shield with 7 pairs of solenostomes (gd1, gd2, gd4, gd5, gd6, gd8, gd9), gd9 larger than the others and 15 pairs small poroids. Setae z2 slightly longer than z4.
Peritreme — Extending beyond the insertions of setae j1 (Figure 3A).
Ventral idiosoma (Figure 3B) — Sternal shield wider than long, large, smooth, 63 (60–65) long, 81 (78–83) wide at level of setae ST2, with a lateral stria, three pairs of setae and two pairs of pores (iv1 and iv2), ST1 28, ST2 29 (25–35), ST3 22 (20–23); distances between ST1–ST3 64 (63–66) and ST2–ST2 72 (71–73); metasternal setae ST4 23 (18–30) and a pair of pores (iv3) on small platelets; genital shield smooth width at widest point 87 (80–90), ST5 32 (30–33); distances between ST5–ST5 80 (79–81) two pairs of metapodal shields, primary 25 (23–28) long and accessory 13 (13–15) long; ventrianal shield subpentagonal, slightly striated all over the shield between JV1 and paranals, with crosswise lines between preanal pores, length 119 (113–128), width at level of setae ZV2, 115 (113–118), and width at level of paranal setae 94 (93–98); with three pairs of preanal setae (JV1 23, JV2 22 (18–25), ZV2 21 (18–23)); four pairs of setae surrounding ventrianal shield on integument (JV4 16 (15–18), JV5 67 (65–69), ZV1 19 (18–20), ZV3 12 (10–15)), three pairs of pores and two pairs of small platelets posteriad to ZV1. Ventrianal shield with a pair of small round pores wide apart, posteriad to JV2, distance between these pores 51 (48–58) and muscle marks posterolaterally.
Spermatheca — Calyx conical, saccular, flared distally 23 (18–25) long and 12 (10–13) width, atrium large, nodular (Figure 3C & 4B).



Chelicera — Fixed digit 36 (35–38) long with four small teeth and pilus dentilis; movable digit 32 (30–33) long with one tooth (Figure 3D).
Legs — Leg IV (Figure 3E) with three macrosetae, all pointed apically, SgeIV 67 (63–70), StiIV 56 (53–58), StIV 67 (63–70) long; distance from base of macrosetae to slit like organ 51 (48–58); chaetotatic formulae of genua and tibiae I–II–III–IV with 10(2-2/1, 2/1-2) – 8(2-2/1, 2/0-1) – 7(1-2/1, 2/0-1) – 7(1-2/1, 2/0-1) and 10(2-2/1, 2/1-2) – 7(1-1/1, 2/1-1) – 7(1-1/1, 2/1-1) – 6(1-1/1, 2/0-1) setae respectively.
Specimens examined — 3♀♀, 28 April 2016 Taşköprü-Kastamonu (41°34′53.64″N 34°16′29.93″E) ground weeds (Cirsium arvense (L.) Scop., (Asteraceae) (Garlic growing areas), collected by Cihan Cılbırcıoğlu.
Due to the similarities in features with Turkish strain, only the measurements are provided.
Dorsal idiosoma — Dorsal shield 388 (370–400) long, 221 (200–230) wide at j6 level, setal lengths j1 32 (25–38), j3 41 (35–48), j4 5, j5 5, j6 5, J5 9 (8–10), z2 24 (20–25), z4 12 (10–13), z5 5, Z1 6 (5–10), Z4 81 (75–88), Z5 96 (85–110), s4 61 (55–70), S2 8 (8–10), S4 9 (8–10), S5 8 (8–10), r3 17 (13–23) and R1 13 (8–18) on lateral integument.
Ventral idiosoma — Sternal shield 65 (55–73) long, 83 (78–90) wide at level of setae ST2; ST1 29 (25–35), ST2 28 (25–30), ST3 26 (23–28), ST4 24 (23–30); distances between ST1–- ST3 62–64 and ST2– ST2 73–74; genital shield 84 (78–95) width at widest point, ST5 29 (28–30); distances between ST5–- ST5 80–88; primary metapodal shields 26 (23–30) long and accessory 13 long; ventrianal shield 117 (113–128) long, width at level of setae ZV2 115 (110–120), and width at level of paranal setae 107 (100–110); JV1 19 (15–23), JV2 20 (18–23), ZV2 19 (18–23); JV4 13 (10–18), JV5 57 (55–60), ZV1 19 (18–20), ZV3 11 (8–15); distance between preanal pores 47 (43–51).
Spermatheca — Calyx 19 (18–28) long and 11 (10–13) wide.
Chelicera — Fixed digit 34 (33–35) and movable digit 32 (30–33) long.
Legs — SgeIV 58 (50–65), StiIV 47 (40–53), StIV 61 (58–63) long; distance from base of macrosetae to slit like organ 55 (53–58).
Specimens examined — 4♀♀, 21 August 2003; 3♀♀ 17 October 2003 Buzet, Lot-et-Garonne, Southwest France (44°15′53.6″N 0°17′57.5″E), ground weeds (apple orchard), Mitox, collected by F. Bakker.
Distribution — Hungary (Ripka et al., 2005); Moldova (Kolodochka 1980); Russia (Wainstein, 1960; Meshkov, 1999); Serbia: (Kropczynska & Petanović, 1987); Spain (Escudero & Ferragut, 1998); Turkey (this study) and Ukraine (Kolodochka, 1981).
Remarks — This is the first report of P. sororculus from Turkey. All the measurement and morphological features are similar for the Turkish and French specimens.
1. Sternal shield broader, L/W ratio usually less than 1.0:1.0; female ventrianal shield usually broader, L/W ratio less than 1.0:1.1; all shields more strongly sclerotized; seta J2 present/absent; genital shield usually narrower than ventrianal shield, ratio width of genital shield to width of ventrianal shield usually 1.0:1.1–3.9; sternal and genital shields smooth or reticulate; peritremal shield ranging from narrow to extremely broad, ectal strip often present; legs II–IV with/without macrosetae; setae z2 and/or z4 often longer; setae j5, J2, S2, S4 and/or Z1 present/absent
...... subtribe Proprioseiopsina Chant & McMurtry, 2004 Proprioseiopsis Muma, 1961 2
— Sternal shield narrower, L/W ratio usually ca. 1.0:1.0; female ventrianal shield usually longer than wide, L/W ratio usually greater than 1.0:1.1; all shields lightly sclerotized; seta J2 present or, if absent, ventrianal shield narrow, L/W ratio > 1.5:1.0; genital shield approximately as wide as ventrianal shield, ratio width genital shield to width ventrianal shield usually approximately 1.0:1.0; sternal shield usually smooth, at most lightly reticulate; genital shield smooth; ventrianal shield usually smooth or at most lightly striate; peritremal shield narrow, ectal strip rarely present; legs II and III usually and leg I often with macrosetae; leg IV usually with three strong macrosetae; setae z2 and z4 usually short/minute; setae j5, S2 and S4 present, setae J2, S5 and Z1 present/absent
...... subtribe Amblyseiina Muma, 1961 5
2. Distance between preanal pores about 1/3 of distance between JV2 setae; sternal shield strongly reticulated
...... P. ovatus (Garman)
— Distance between preanal pores more than half distance between JV2 setae; sternal shield not reticulated
...... 3
3. Seta z4 longer than z2; S2 longer than Z1
...... P. okanagensis (Chant)
— Seta z4 shorter than z2; S2 and Z1 short and subequal
...... 4
4. Seta Z5 longer than the distance between their insertions; setae j1 and z2 subequal
...... P. messor (Wainstein)
— Seta Z5 shorter than the distance between their insertions; seta z2 shorter than j1
...... P. sororculus (Wainstein)
5. Spermatheca with atrium bifurcate/vacuolate at juncture with major duct; male spermatophoral process T-shaped, with both heel and toe elongate, approximately equal
...... Graminaseius Chant & McMurtry, 2004: G. graminis (Chant)
— Spermatheca with atrium not bifurcate/vacuolate at juncture with major duct; male spermatophoral process not T-shaped, heel and toe not both elongate, not approximately equal
...... 6
6. Ratio seta s4: S2>3.0:1.0
...... 7
— Ratio seta s4: S2<2.7:1.0
...... 17
7. Chelicera unusually large, robust with fixed digit longer than movable digit
...... Chelaseius Muma & Denmark 1968: C. valliculosus Kolodochka
— Chelicera of normal size, fixed digit not much longer than movable digit
...... Amblyseius Berlese, 1914 8
8. Ventrianal shield vase-shaped
...... A. largoensis (Muma)
— Ventrianal shield not vase-shaped
...... 9
9. Spermatheca with calyx annulated, flared distally
...... A. obtusus (Koch)
— Spermatheca with calyx not annulated
...... 10
10. Ventrianal shield with large elliptical (crescent-shaped) preanal solenostomes
...... 11
— Ventrianal shield with small round preanal solenostomes
...... 14
11. Dorsal shield strongly reticulated
...... A. bryophilus Karg
— Dorsal shield smooth
...... 12
12. Calyx of spermatheca about four times longer than width
...... A. adjaricus Wainstein & Vartapetov
— Calyx of spermatheca with about equal width and length
...... 13
13. Seta Z5 102–116 µm long; atrium of spermatheca relatively longer; StiIV at most reaching the insertion of StIV
...... A. swirskii Athias-Henriot
— Seta Z5 longer than 150 µm; atrium of spermatheca short and c-shaped; StiIV passing well behind the insertion of StIV
...... A. andersoni (Chant)
14. z4 short, less than 1/5 of its insertion to seta s4
...... 15
— z4 longer, at least half the distance to seta s4
...... 16
15. Z5 much longer than the distance between its insertions
...... A. meridionalis Berlese
— Z5 shorter or equal the distance between its insertions
...... A. kadzhajai Gomelauri
16. Spermatheca very long, calyx snake-shaped
...... A. decolor (Westerboer)
— Spermatheca not long, calyx saccular
...... A. armeniacus Arutunjan & Ohandjanian
17. Seta S5 absent
...... Amblyseiella Muma, 1955: A. setosa Muma
— Seta S5 present Transeius Chant & McMurtry, 2004
...... 18
18. Ventrianal shield without preanal solenostomes; seta Z5 shorter than distance between its bases
...... T. herbarius (Wainstein)
— Ventrianal shield with preanal solenostomes; seta Z5 longer than distance between its bases
...... 19
19. Calyx of spermatheca elongate about 25 µm
...... T. begljarovi (Abbasova)
— Calyx of spermatheca short about 8 µm long
...... T. wainsteini (Gomelauri)
The authors wish to thank Funding for this study through grants by the Turkish Council of Research (TUBITAK TOVAG grant no.: 114O416). We are grateful to Esra DAŞTAN for help in preparing the slides.
Athias-Henriot C. 1961. Mesostigmates (Urop. excl.) édaphiques Méditerranéens (Acaromorpha, Anactinotrichida). Acarologia, 3(4): 381-509.
Athias-Henriot C. 1975. Nouvelle notes sur lês Amblyseiini II. Le rélève organotaxique de la face dorsale adulte (Gamasides protoadeniques, Phytoseiidae). Acarologia, 17(1): 20-29.
Berlese A. 1914. Acari nuovi. Manipulus IX. Redia, 10: 113-150.
Chant D.A., McMurtry J.A. 2004. A review of the subfamily Amblyseiinae Muma (Acari: Phytoseiidae): Part III. The tribe Amblyseiini Wainstein, subtribe Amblyseiina n. subtribe. Int. J. Acarol., 30(3): 171-228. doi:10.1080/01647950408684388 ![]()
Chant D.A., McMurtry J.A. 2007. Illustrated keys and diagnoses for the genera and sub-genera of the Phytoseiidae of the World: Indira Publishing House, West Bloomfield, Michigan, USA. pp 220.
Chant D.A., Yoshida-Shaul E. 1991. Adult ventral setal patterns in the family Phytoseiidae (Acari: Gamasina). Int. J. Acarol., 17: 187-199. doi:10.1080/01647959108683906 ![]()
Demite P.R., Moraes G.J. de, McMurtry J.A., Denmark H.A., Castilho R.C. 2018. Phytoseiidae Database. Available from: www.lea.esalq.usp.br/phytoseiidae (accessed 04/02/2018).
Döker I., Kazak C., Karut K. 2016. Contributions to the Phytoseiidae (Acari: Mesostigmata) fauna of Turkey: morphological variations, twelve new records, re-description of some species and a revised key to the Turkish species. Syst. Appl. Acarol., 21(4): 505-527. doi:10.11158/saa.21.4.10 ![]()
Döker I., Kazak C., Karut K. 2017. Three new species of the family Phytoseiidae (Acari: Mesostigmata) from Turkey. Zootaxa, 4243(3): 565-576. doi:10.11646/zootaxa.4243.3.8 ![]()
Düzgüneş Z. 1963. The mite species newly found in Turkey. Plant Protection Bull. 3, 237-246. [in Turkish]
Escudero L.A., Ferragut F. 1998. Comunidad de ácaros del ecosistema hortícola mediterrâneo: composición y distribución geográfica. Bol. San. Veg. Plagas, 24(4): 749-762.
Espinha I.G., Ferragut F., Fereira J.A., Torres L.M. 1998. Ácaros fitoseideos (Acari: Phytoseiidae) del cultivo de manzano em el nordeste de Portugal. Bol. San. Veg. Plagas, 24, 383-390.
Faraji F., Çobanoğlu S., Çakmak I. 2011. A checklist and a key for the Phytoseiidae species of Turkey with two new species records (Acari: Mesostigmata). Int. J. Acarol., 37(Supl. 1): 221-243. doi:10.1080/01647954.2011.558851 ![]()
Ferragut F., Pérez M.I., Iraola V., Escudero A. 2010. Ácaros depredadores em las plantas cultivadas. Família Phytoseiidae. Ediciones Agrotécnicas, Madrid, 202 pp.
Gerson U., Smiley R.L., Ochoa R. 2003. Mites (Acari) for pest control. Blackwell Science, Oxford, 539 pp. doi:10.1002/9780470750995 ![]()
Hirschmann W. 1962. Gangystematik der Parasitiformes. Acarologie Schriftenreihe furVergleichende Milbenkunde, Hirschmann-Verlag, Furth/Bay, Germany, 5(5–6), 80 pp.+ 32 plates
Karg W. 1991. Die Raubmilbenarten der Phytoseiidae Berlese (Acarina) Mitteleuropas sowie angrenzender Gebiete. Zoologische Jahrbucher Systematik, Germany, 118(1), 1-64.
Kolodochka L.A. 1980. New phytoseiid mites (Parasitiformes: Phytoseiidae) from Moldavia, USSR. Vestn. Zool., (4): 39-45 [in Russian].
Kolodochka L.A. 1981. New phytoseiid mites from Crimea (Parasitiformes: Phytoseiidae). I. Vestn. Zool., (1): 18-22 [in Russian].
Kropczynska D., Petanović R. 1987. Contribution to the knowledge of the predacious mites (Acarida, Phytoseiidae) of Yugoslavia. Biosistematika, 13(1): 81-86.
Lindquist E.E., Evans G.O. 1965. Taxonomic concepts in the Ascidae, with a modified setal nomenclature for the idiosoma of the Gamasina (Acarina: Mesostigmata). Mem. ent. Soc. Canada, 47: 1-64. doi:10.4039/entm9747fv ![]()
McMurtry J.A., de Moraes G.J, Sourassou N.F. 2013. Revision of the life styles of phytoseiid mites (Acari: Phytoseiidae) and implications for biological control strategies. Systematic and Applied Acarology 18(4):297. doi:10.11158/saa.18.4.1 ![]()
Meshkov Y.I. 1999. Contribution to phytoseiid fauna (Parasitiformes, Phytoseiidae) of Moscow District. Zool. Zh. (78): 426-431.
Moraes G.J. de, McMurtry J.A., Denmark H.A. 1986. A catalog of the mite family Phytoseiidae. References to taxonomy, synonymy, distribution and habitat. EMBRAPA – DDT, Brasilia, Brazil, 353 pp.
Moraes G.J., McMurtry J.A., Denmark H.A., Campos C.B. 2004. A revised catalog of the mite family Phytoseiidae. Zootaxa, 434: 1-494. doi:10.11646/zootaxa.434.1.1 ![]()
Muma M.H. 1955. Phytoseiidae (Acarina) associated with citrus in Florida. Ann. Entomol. Soc. Amer., 48: 262-272. doi:10.1093/aesa/48.4.262 ![]()
Muma M.H. 1961. Subfamilies, genera, and species of Phytoseiidae (Acarina: Mesostigmata). Florida State Mus. Bull., 5(7): 267-302.
Muma, M.H., Denmark H.A. 1968. Some generic descriptions and name changes in the family Phytoseiidae (Acarina: Mesostigmata). Florida Ent., 51(4): 229-240. doi:10.2307/3493424 ![]()
Papadoulis G.Th., Emmanouel N.G., Kapaxidi E.V. 2009. Phytoseiidae of Greece and Cyprus (Acari: Mesostigmata). West Bloomfield, MI, USA: Indira Publishing House 200 pp.
Prasad V. 2012. Checklist of Phytoseiidae of the World (Acari: Mesostigmata): Indira Publishing House, West Bloomfield, Michigan, USA. 1020 pp.
Ripka G., Fain A., Kazmierski A., Kreiter S., Magowski W.L. 2005. New data to the knowledge of the mite fauna of Hungary (Acari: Mesostigmata, Prostigmata and Astigmata). Acta Phytopathol. Entomol. Hung., 40 (1-2): 159-176. doi:10.1556/APhyt.40.2005.1-2.13 ![]()
Rowell H.J., Chant D.A., Hansell R.I.C. 1978. The determination of setal homologies and setal patterns on the dorsal shield in the family Phytoseiidae (Acarina: Mesostigmata). Can. J. Zool., (110): 859-876. doi:10.4039/Ent110859-8 ![]()
Tixier M.S., Kreiter S., Auger P., Sentenac G., Salva G., Weber M. 2000. Phytoseiidae mite species located in uncultivated areas surrounding vineyards in three French regions. Acarologia, 41(1-2): 127-140.
Tsolakis H., Tixier M.-S., Kreiter S., Ragusa S. 2012. The concept of genus within the family Phytoseiidae (Acari: Parasitiformes): historical review and phylogenetic analyses of the genus Neoseiulus Hughes. Zool. J. Linn. Soc., 165: 253-273. doi:10.1111/j.1096-3642.2011.00809.x ![]()
Wainstein B.A. 1960. New species and subspecies of the genus Typhlodromus Scheuten (Parasitiformes, Phytoseiidae) of the USSR fauna Zool. Zh., 39: 683-690 [in Russian].
Wainstein B.A. 1972. New species of the family Phytoseiidae (Parasitiformes). Zool. Zh., 51: 1407-1411 [in Russian].
Wainstein B.A. 1977. Family Phytoseiidae Berlese, 1916. In: Ghilarov, B.M. (Ed.), Determination of mites inhabiting soil (Mesostigmata). Ed. Nauka, Leningrad, Russia, 226-244.
Westerboer I., Bernhard F. 1963. Die Familie Phytoseiidae Berlese 1916. In: Stammer H. (Ed.), Beitrage zur Systematik und Okologie mitteleuropaischer Acarina. Band II, Mesostigmata I, Germany, pp. 451-791.
Zhang Z-Q. 2003. Mites of greenhouses: identification, biology and control. CABI Publishing, Wallingford, 244 pp. doi:10.1079/9780851995908.0000 ![]()



2018-04-09
Date accepted:
2018-07-07
Date published:
2018-09-14
Edited by:
Kreiter, Serge

This work is licensed under a Creative Commons Attribution 4.0 International License
2018 Çobanoğlu, Sultan; Faraji, Farid and Cılbırcıoğlu, Cihan
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



