Survey of phytoseiid mites (Acari: Mesostigmata, Phytoseiidae) in citrus orchards and a key for Amblyseiinae in Vietnam
Fang, Xiao-Duan1 ; Nguyen, Van-Liem2 ; Ouyang, Ge-Cheng3 and Wu, Wei-Nan4
1Guangdong Key Laboratory of Animal Conservation and Resource Utilization, Guangdong Public Laboratory of Wild Animal Conservation and Utilization, Guangdong Institute of Applied Biological Resources, Guangzhou, 510260, Guangdong, China.
2Plant Protection Research Institute, Duc Thang, Bac Tu Liem, Ha Noi, Viet Nam.
3✉ Guangdong Key Laboratory of Animal Conservation and Resource Utilization, Guangdong Public Laboratory of Wild Animal Conservation and Utilization, Guangdong Institute of Applied Biological Resources, Guangzhou, 510260, Guangdong, China.
4Guangdong Key Laboratory of Animal Conservation and Resource Utilization, Guangdong Public Laboratory of Wild Animal Conservation and Utilization, Guangdong Institute of Applied Biological Resources, Guangzhou, 510260, Guangdong, China.
2020 - Volume: 60 Issue: 2 pages: 254-267
https://doi.org/10.24349/acarologia/20204366Original research
Keywords
Abstract
Introduction
Vietnam is long and narrow, located in the east of Indochina Peninsula. Vietnam shares its land borders with China to the north, Laos and Cambodia to the west, and surrounded by the south China sea to the east and south. Its climate is characterized by the interplay of the East Asian and South Asian Monsoons, cold surges in the winter months and a long rainy season which extend beyond summer (Nguyen et al. 2014). Latitude of the south (8°10′ N–10°48′ N) is more southern than Hainan Island (18°10′ N–20°10′ N) in China, and its temperatures vary less in the southern plains around the Mekong Delta. Vietnam is one of the 25 countries considered to possess a high level of biodiversity (Vietnam Environment Protection Agency, 2005, electronic source) (Sohn and Park, 2009). Until now, only 15 species of phytoseiid mites (Acari: Phytoseiidae) have been recorded in Vietnam (Nguyen et al. 2016; Nguyen and De Clercq, 2018; Thuan et al. 2019; Nguyen, 2019a, 2019b; Nguyen and Dao 2019; Nguyen et al. 2019a, 2019b).
Citrus is the second largest fruit crop in Vietnam, with Mekong Delta the main citrus production area. The sampling sites in this survey are all located in Mekong Delta. Phytoseiid mites are important natural enemies of pest mites and small insects in citrus orchards (Wu et al. 2009; Huang 2011). Phytoseiid mites have been mass-reared and widely used for biological control of pest mites and insects in citrus orchards in China (Zhang et al. 2002; Ouyang et al. 2007; Fang et al. 2013, 2018). It is important to survey the native species of phytoseiid mites in citrus orchard, which is supposed to be the most adapted bio-control agents can be used in local citrus orchards. This study proposes a new record A. obtuserellus Wainstein and Begljarov, 1971 and two other species, Amblyseius largoensis (Muma, 1955) and Euseius ovalis (Evans, 1953), from citrus orchards in Mekong Delta.
Material and methods
Mite specimens examined in this study were collected from three citrus orchards in Mekong Delta in April 2018. One citrus orchard in Chau Thanh district, Ben Tre Province is about 5 ha, 10-year-old Citrus reticulata × sinensis and Psidium guajava Linn. interplanted, surrounded by eucalyptus on edge; SK mineral oil was mainly used in this orchard. The citrus orchard in Phong Dien district, Can Tho Province is also about 5 ha, 3-year-old Citrus reticulata × sinensis and Psidium guajava Linn. interplanted, surrounded by a wild bush on one side; mineral oil and chemical pesticides are used in this orchard. The citrus orchard in Cao Lanh district, Dong Thap is about 7 ha, only 10-year-old Citrus reticulata × sinensis trees planted, surrounded by other citrus orchards; chemical pesticide abamectin was mainly used in this orchard.
Mites were mounted in Hoyer's medium and examined, measured and illustrated under a phase and DIC (differential interference contrast) microscope (Zeiss® Axio Imager A2) and picture pick-up system [ZEN 2.3 (Blue edition) © Carl Zeiss Microscopy GmbH, 2011]. Measurements are presented in μm, by their mean and range in parentheses. Morphological features of adult mites were measured as follows: dorsal shield length and width were taken from the anterior to posterior margins of the shield along the midline and from the lateral margins at s4 level; for all ventral shields, lengths were measured along their midline from the anterior to posterior margins; widths were taken from the lateral margins at st2, st5 and ZV2 level respectively; cheliceral fixed digit length was measured from dorsal poroid to anterior tip and movable digit length measured from the basal articulation to the tip. The general terminology used for morphological descriptions in this study follows that of Chant and McMurtry (2007), idiosomal setal terminology follows those by Rowell et al. (1978) and Chant and Yoshida-Shaul (1991, 1992), adenotaxy and poroidotaxy terminology followed that of Beard (2001), and chaetotaxy of legs follows that of Evans (1963).
All mites collected were measured. They were deposited in GIABR (Guangdong Institute of Applied Biological Resources, Guangzhou, China). The previous records of these species mainly referenced to the Phytoseiidae Database (Demite et al. 2019).
Results and discussion
Family Phytoseiidae Berlese
Subfamily Amblyseiinae Muma
Tribe Amblyseiini Muma
Subtribe Amblyseiina Muma
Genus Amblyseius Berlese
Amblyseius obtuserellus Wainstein & Begljarov
(Figs. 1 a – e)
Amblyseius obtuserellus Wainstein & Begljarov, 1971: 1806; Ehara & Yokogawa 1977: 54; Wainstein 1979: 14; Wu 1980: 44; Moraes et al. 1986: 24; Ryu & Lee 1992: 28; Wu et al. 1997: 50; Wu et al. 2009: 194.
Amblyseius (Multiseius) obtuserellus, Denmark & Muma 1989: 124.
Description — Female (n = 8)
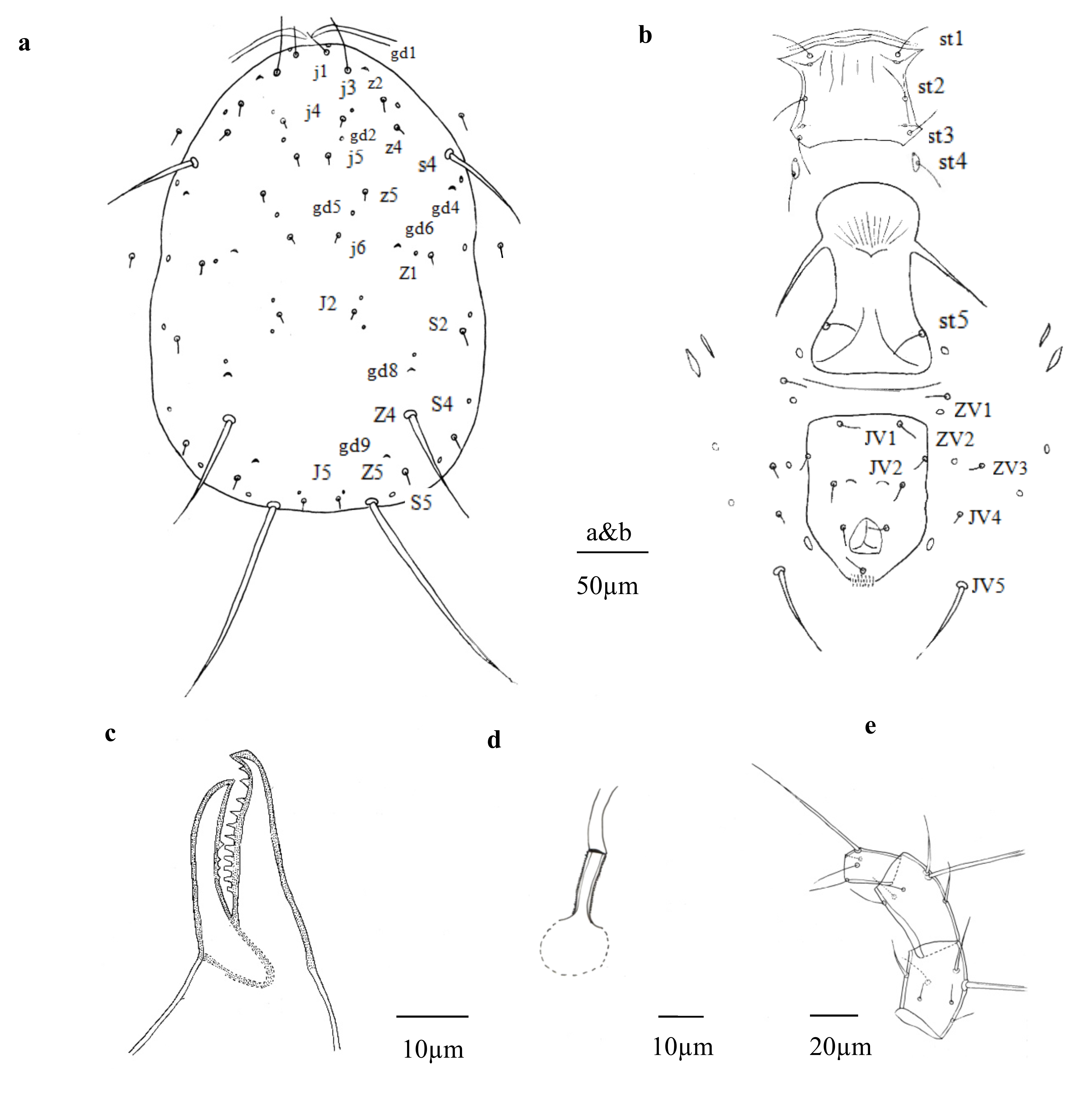


Dorsum — Idiosomal setal pattern: 10A: 9B/JV-3: ZV. Dorsal shield smooth (Fig. 1a), 317 (300–330) long and 200 (185–218) wide, distances between setae j1- J5 303 (289–316) and s4-s4 176 (164–186), shield nearly oval, constricted at level of R1. Setae r3 and R1 on soft membranous cuticle laterad dorsal shield, r3 at level of z4, R1 at level of shield incisions. All setae smooth, except Z4, Z5 with minute thorns. With seven pairs of solenostomes (gd1, gd2, gd4, gd5, gd6, gd8, gd9) on dorsal shield. Length of setae: j1 24 (23–26), j3 43 (40–46), j4 5 (4–6), j5 5 (4–6), j6 6 (4–8), J2 7 (5–9), J5 6 (4–7), z2 9 (7–11), z4 9 (7–11), z5 5 (4–6), Z1 7 (6–9), Z4 82 (75–88), Z5 161 (155–167), s4 68 (63–72), S2 8 (7–9), S4 7 (6–9), S5 7 (5–8), r3 12 (11–14), R1 8 (7–9).
Venter — (Fig. 1b). All ventral setae smooth. Sternal shield with sparse striation, anterior margin convex, posterior margin slightly concaved, 69 (68–71) long, 74 (73–75) wide, wider than long, with three pairs of setae st1 31 (29–33), st2 28 (25–32), st3 26 (24–30), and two pairs of lyrifissures (pst1-pst2), distance between st1-st3 55 (53–58) and st2-st2 68 (67–69). Metasternal platelets drop-shaped, each with one metasternal seta, st4 25 (24–26) and one lyrifissure (pst3). Genital shield smooth, with one pair of thin genital setae st5 27 (25–28), distance between st5-st5 59 (64–71); one pair of associated poroids on soft cuticle near posterior corners of shield. Ventrianal shield nearly pentagonal, 112 (105–116) long, 82 (76–88) wide at level of ZV2, 78 (73–84) wide at level of anus, with three pairs of thin pre-anal setae JV1 16 (14–19), JV2 14 (11–17), ZV2 12 (10–13); Pa 14 (12–17), Pst 16 (14–19) long. Pre-anal pores crescentic, posteromedian to JV2, distance between pores 21 (19–23). Opisthogastric soft cuticle with four pairs of setae, ZV1 14 (13–16), ZV3 9 (7–10), JV4 8 (6–9), JV5 59 (54–64) long. All ventral setae thin, except JV5, thicker. Two pairs of metapodal platelets, primary platelets 19 (17–21) long, 5 (3–5) wide, secondary ones 11 (10–12) long, 2 (1–2) wide.
Peritremes — Peritremes extending anteriorly between j1.
Chelicerae — (Fig. 1c). Fixed digit 32 (30–36) long, with 11 teeth, movable digit 28 (27–30) long, with four teeth.
Spermatheca — (Fig. 1d). Calyx of spermatheca tubular, 25 (22–28) long, the proximal part mostly distal part thicker, granular surface, 9 (8–10) long, 2 (2–2) wide at the junction with the vesicle, atrium and major duct indiscernible.
Legs — Genua formula for leg I 2 – 1/2, 2/1– 1, leg II 0 – 2/1, 0/2 – 2, leg III 1 – 1/2, 1/2 – 0, leg IV 2 – 2/1, 0/1 – 1. Genua I-III each with one macroseta, Sge I 32 (29–34), Sge II 31 (28–32), Sge III 42 (39–45). Leg III with one macroseta on tibia, Sti III 30 (27–32). Leg IV (Fig. 1e) with three macrosetae on genu, tibia and basitarsus, Sge IV 82 (76–86), Sti IV 46 (40–51) and St IV 59 (56–62).
Specimens examined — 8 ♀♀, Chau Thanh district, Ben Tre Province (accession no. BT-0190, BT-0200, BT-0211, BT-0212, BT-0220, BT-0231, BT-0232, BT-0240), on Citrus reticulata × sinensis, 10 IV 2018, Fang X.D. coll.
Previous records — China, Japan, Russia, South Korea.
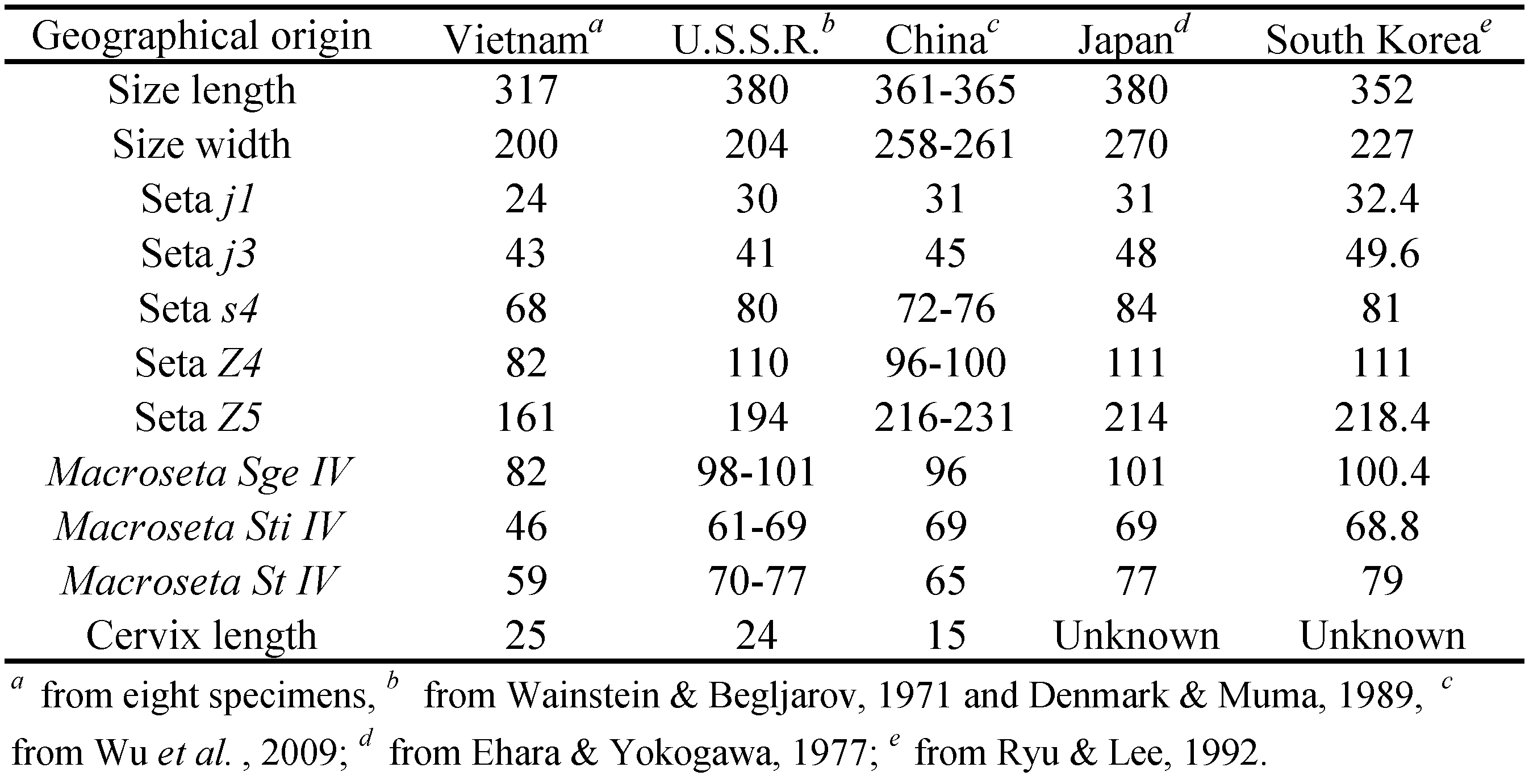
Remarks — Amblyseius obtuserellus holotype specimen was from the Primorsky Territory, U.S.S.R. (Wainstein and Begljarov, 1971; Denmark and Muma, 1989). This species was also recorded in Japan (Ehara and Yokogawa, 1977) and South Korea (Ryu and Lee, 1992). A. obtuserellus have been found in many Provinces in China, including Jiangsu, Zhejiang, Anhui, Fujian, Jiangxi, Hunan and Guangdong. Wu (1982) reported that the size of its genital shield, ventrianal shield and distance between pre-anal pores are variable, but the calyx shape of spermatheca does not present differences. The calyx of the spermatheca in this species is cylindrical and long and the distal part of calyx thickened and more sclerotized.
Ehara and Yokogawa (1977), Denmark and Muma, 1989, Ryu and Lee (1992) and Wu et al. (2009) re-described this species. Geographical populations in different countries all have distinctive, granular spermathecal cervix, but they have differences in the individual size, length of some setae on dorsal shield and the macrosetae on Leg IV, and the length of cervix of spermatheca. Their measurements differences are listed in Table 1.
Amblyseius obtuserellus has 11 well-developed teeth on fixed digit and four teeth on movable digit of the chelicerae and this species may play an important role in bio-control. Its biology and bio-control application need to be further studied.


Amblyseius largoensis (Muma)
(Figs. 2 a – e, 3 a – c)
Amblyseiopsis largoensis Muma 1955: 266; Garman 1958: 76.
Typhlodromus (Amblyseius) largoensis, Chant 1959: 96.
Amblyseius largoensis, Ehara 1959: 293; De Leon 1966: 90, 1967: 23; Muma & Denmark 1970: 69; Wu 1980: 41; Schicha 1981: 105; McMurtry & Moraes 1984: 29; Chen et al. 1984: 332; Moraes et al. 1986: 17, 2000: 239, 2004a: 143, 2004b: 33; Denmark & Muma 1989: 55; Wu & Lan 1989: 449; Wu et al. 1997: 39; Gondim Jr & Moraes 2001: 72; Chant & McMurtry 2004: 208, 2007: 78; Zannou et al. 2007: 16; Ferragut et al. 2011: 40; Oliveira et al. 2012: 4; Nguyen & Dao 2019.
Amblyseius (Amblyseialus) largoensis, Muma 1961: 287.
Amblyseius magnoliae, Denmark & Evans 2011: 69.
Typhlodromus largoensis, Hirschmann 1962.
Amblyseius (Amblyseius) largoensis, Ehara 1966; Ehara & Bhandhufalck 1977: 67; Denmark & Evans 2011: 69.
Amblyseius amtalaensis, Gupta 1977: 53; 1986: 51.
Amblyseius neolargoensis, Van der Merwe 1965:59; Chant et al. 1978: 1347.
Amblyseius sakalava, Blommers 1976: 96; Ueckermann & Loot 1988: 70.
Description — Female (n = 25)
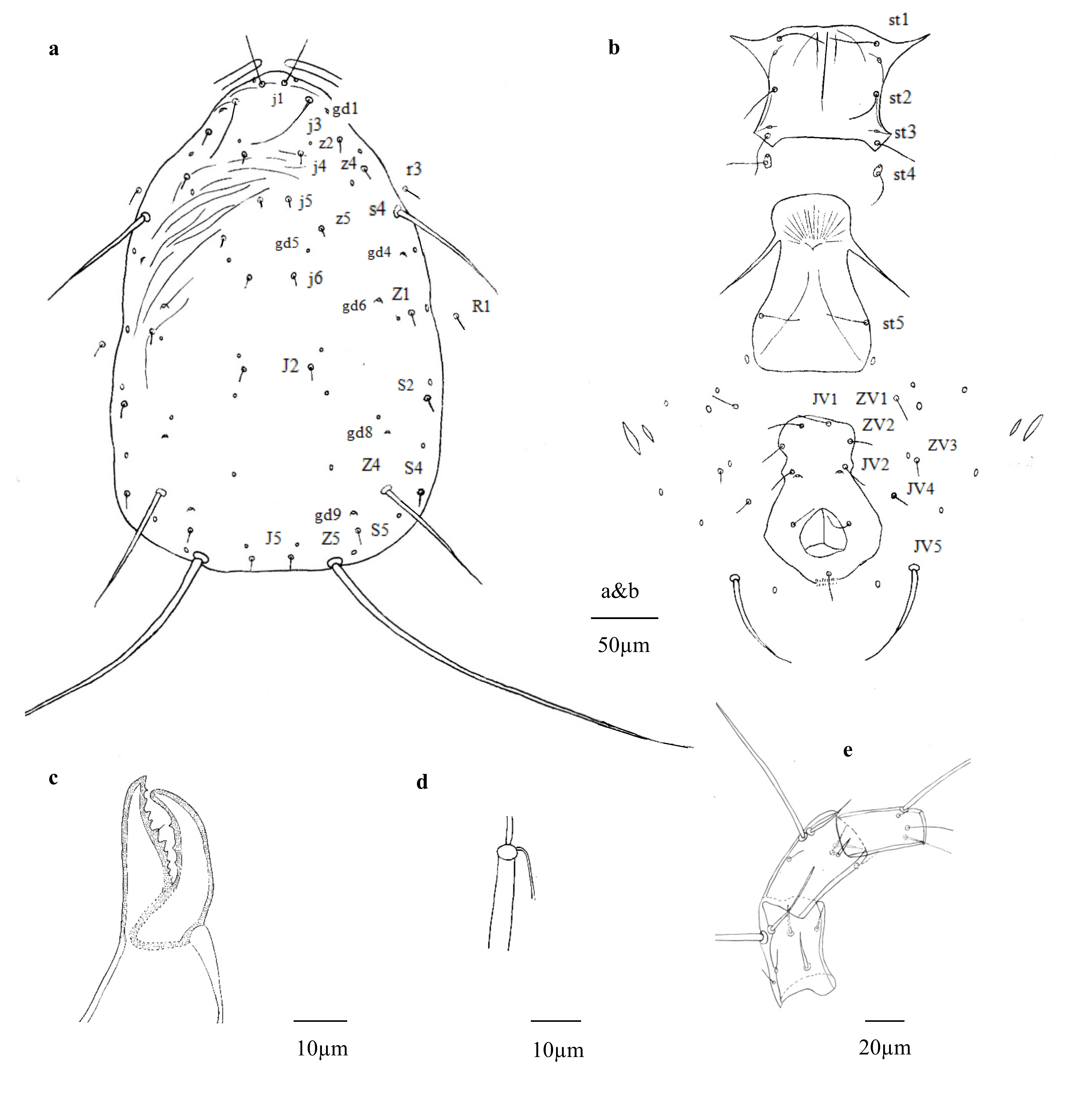


Dorsum — Idiosomal setal pattern: 10A: 9B/JV-3: ZV. Dorsal shield (Fig. 2a) mostly smooth except anteriorly striated. Dorsal shield 355 (341–376) long and 224 (212–253) wide, distances between setae j1- J5 338 (326–364) and s4-s4 194 (181–213), shield nearly oval, slightly constricted at level of R1. Setae r3 and R1 on soft membranous cuticle laterad dorsal shield, r3 at level of between z4 and s4, R1 at level of shield incisions. All setae smooth. With six pairs of solenostomes (gd1, gd4, gd5, gd6, gd8, gd9) on dorsal shield. Length of setae: j1 36 (30–40), j3 51 (41–59), j4 6 (4–8), j5 5 (3–6), j6 7 (4–10), J2 9 (6–12), J5 9 (6–12), z2 11 (8–14), z4 9 (7–13), z5 6 (4–8), Z1 10 (6–14), Z4 98 (89–105), Z5 277 (243–296), s4 95 (86–103), S2 13 (9–16), S4 12 (9–16), S5 11 (8–15), r3 12 (10–16), R1 10 (7–14).
Venter — (Fig. 2b). Ventral setae smooth. Sternal shield with sparse striation, anterior margin convex, posterior margin straight, 80 (72–90) long, 77 (68–82) wide, with three pairs of setae st1 34 (29–39), st2 31 (27–34), st3 30 (26–33), and two pairs of lyrifissures (pst1-pst2), distance between st1-st3 63 (57–67) and st2-st2 68 (60–73). Metasternal platelets drop-shaped, each with one metasternal seta, st4 29 (26–34) and one lyrifissure (pst3). Genital shield smooth, with one pair of thin genital setae st5 27 (23–33), distance between st5-st5 74 (68–81); one pair of associated poroids on soft cuticle near posterior corners of shield. Ventrianal shield vase-shaped, 109 (99–117) long, 48 (45–53) wide at level of ZV2, 72 (64–84) wide at level of anus, with three pairs of thin pre-anal setae JV1 20 (15–23), JV2 17 (15–20), ZV2 15 (12–18); Pa 18 (15–21), Pst 18 (14–21) long. Pre-anal pores crescentic, posteromediad to JV2, distance between pores 26 (23–28). Opisthogastric soft cuticle with four pairs of setae, ZV1 17 (14–21), ZV3 10 (8–13), JV4 10 (6–13), JV5 64 (53–74) long. All ventral setae thin, except JV5, thicker. Two pairs of metapodal platelets, primary platelets 23(18–27) long, 4 (3–6) wide, secondary ones 13 (9–16) long, 2 (1–4) wide.
Peritremes — Peritremes extending anteriorly between j1.
Chelicerae — (Fig. 2c). Fixed digit 32 (29–35) long, with nine teeth and pilus dentilis, movable digit 29 (26–31) long, with three teeth.
Spermatheca — (Fig. 2d). Calyx of spermatheca tubular, 21 (18–24) long, 4 (2–5) wide at the opening; atrium 4 (2–5) long, 4 (3–5) wide, massive atrium as wide as cervix, connected with calyx; major duct broad, short, directly attached to atrium, and minor duct visible.
Legs — Genua formula for leg I 0 – 1/2, 2/2– 2, leg II 0 – 1/2, 1/1 – 2, leg III 2 – 2/1, 1/1 – 0, leg IV 2 – 2/1, 0/1 – 1. Genua I-III each with one macroseta, Sge I 41 (37–45), Sge II 38 (32–41), Sge III 50 (42–55). Leg III with one macroseta on tibia, Sti III 41 (35–45). Leg IV (Fig. 2e) with three long-whipped macrosetae on genu, tibia and basitarsus, Sge IV 128 (111–138), Sti IV 101 (91–113) and St IV 65 (56–73).
Male — (n=7)



Dorsum — (Fig. 3a). Idiosomal setal pattern: 10A: 9B/JV-3: ZV. Dorsal shield mostly smooth except anteriorly striated, same as on female, 271 (264–291) long and 184 (173–201) wide at level of setae s4, shield nearly oval, nineteen pairs of dorsal setae, all smooth. Same adenotaxy and poroidotaxy as in females. Length of setae: j1 29 (24–31), j3 47 (42–50), j4 6 (4–7), j5 5 (4–6), j6 6 (5–8), J2 8 (7–9), J5 7 (5–8), z2 9 (7–11), z4 9 (7–10), z5 5 (4–7), Z1 9 (6–10), Z4 73 (65–81), Z5 215 (194–236), s4 79 (75–82), S2 11 (8–13), S4 10 (9–11), S5 10 (9–11), r3 11 (9–13), R1 8 (7–10).
Venter — (Fig. 3b). Ventral setae smooth. Sternogenital shield smooth, 131 (125–143) long, wider 81 (77–87) between coxae II–III than at posterior corners 41 (38–44). Five pairs of sterogenital setae (st1–st5), st1 27 (24–30), st2 23 (21–24), st3 22 (20–26), st4 21 (19–24), st5 22 (19–24), and three pairs of lyrifissures (pst1-pst3). Chaetotaxy of genua similar to female. Ventrianal shield subtriangular, 111 (106–116) long, 155 (147–161) wide at level anterior corners; transversally striated; striation on anterior part of the shield more obvious, with three pairs of thin pre-anal setae JV1 17 (14–20), JV2 15 (13–17), ZV2 13 (11–17); Pa 14 (11–17), Pst 17 (15–18) long. Preanal pores crescentic, posteromedian to JV2, distance between pores 19 (18–21). Opisthogastric soft cuticle with one pairs of setae, JV5 37 (33–40) long.
Peritremes — Peritremes extending to between j1. Peritrematal shield fused with dorsal shield.
Chelicerae and spermatodactyl — (Fig. 3c). Cheliceral dentition not discernible in the examined specimens. Fixed digit 22 (20–23) long, movable digit 20 (18–22) long. Spermatodactyl L-shaped; shaft 16 (15–18), foot 8 (7–10) long.
Legs — Chaetotaxy of genua similar to female. Macrosetae on genu I 31 (29–35), genu II 31 (30–33), genu III 37 (35–40), tibia III 35 (32–38), genu IV 81 (78–85), tibia IV 69 (65–72), tarsus IV 56 (52–59).
Specimens examined – 9 ♀♀, Chau Thanh district, Ben Tre Province (accession no. BT-0121, BT-0130, BT-0140, BT-0180, BT-0250, BT-0260, BT-0270, BT-0281, BT-0282), on Citrus reticulata × sinensis, 10 IV 2018, Fang X.D. coll.; 2 ♂♂ (accession no. BT-0122, BT-0160) same locality, host, date and collector; 16 ♀♀, Phong Dien district, Can Tho Province (accession no. CT-0301, CT-0302, CT-0321, CT-0322, CT-0341, CT-0342, CT-0350, CT-0360, CT-0371, CT-0372, CT-0401, CT-0402, CT-0430, CT-0441, CT-0450, CT-0481), and 2 ♂♂ (accession no. CT-0442, CT-0482), 12 IV 2018, same host and collector; 4 ♀♀, Cao Lanh district, Dong Thap Province (accession no. DT-0490, DT-0500, DT-0521, DT-0540) and 3 ♂♂ (accession no. DT-0522, DT-0532, DT-0550), 13 IV 2018, same host and collector.
Previous Records — Nearctic, Neotropical, Oriental, Australasian, Ethiopian, West Palaearctic and East Palaearctic, almost including all biogeographic realms except Antarctica (Hernandes et al. 2011).
Remarks — This species is very similar to A. herbicolus (Chant, 1959) and A. eharai Amitai and Swirski, 1981, but having differences in sternal shield with weak sclerotization and posterior margin straight, and calyx of spermatheca tubular. This species prefers high humidity environment, and it is the dominant species of brushes in Hainan islands in China. This species is important natural enemies of agriculture and forestry pest mites (Wu et al. 2009). In this survey, A. largoensis was found the dominant native species in surveyed citrus orchards in Vietnam.
Tribe Euseiini Chant and McMurtry
Subtribe Euseiina Chant and McMurtry
Genus Euseius Wainstein
Euseius ovalis (Evans)
(Figs. 4 a – e)
Typhlodromus ovalis Evans 1953: 458.
Typhlodromus (Amblyseius) ovalis, Chant 1959: 68; Narayanan, Kaur and Ghai 1960: 388.
Amblyseius (Typhlodromalus) ovalis, Muma 1961.
Amblyseius ovalis, Collyer 1964: 634; Gupta 1970: 98; Prasad 1974: 168; Wu 1980: 46; Ray and Gupta 1981: 279; Chen et al. 1984: 327.
Amblyseius (Amblyseius) ovalis, Ehara 1966: 24.
Euseius ovalis, Gupta 1978: 335; Moraes et al. 1986: 49; Wu et al. 1997: 117; Wu et al. 2009: 235; Liao et al. 2017: 220.
Amblyseius (Euseius) ovalis, Gupta 1985; Ehara and Amano 1998: 43.
Description — Female (n = 2)
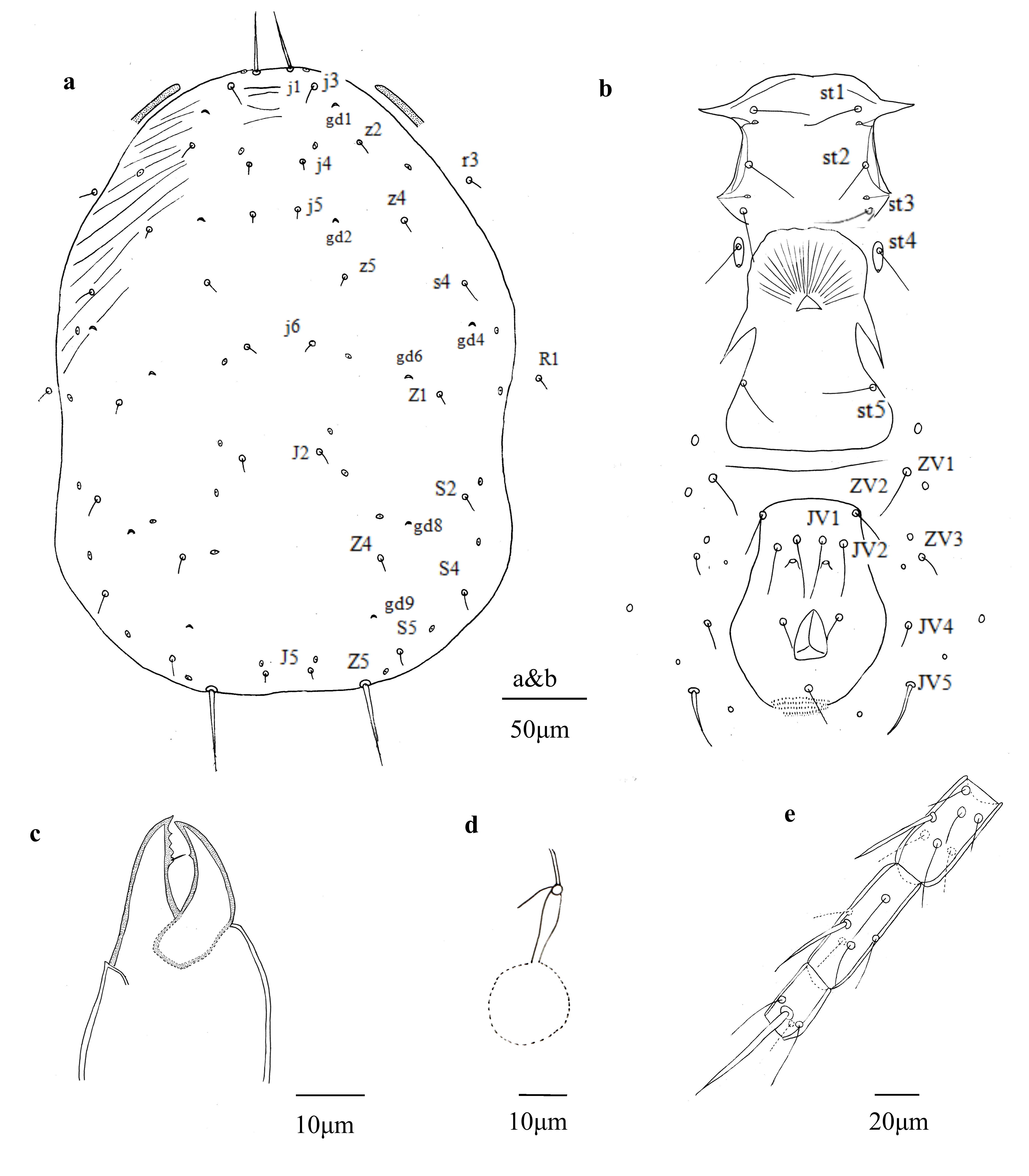


Dorsum — Idiosomal setal pattern: 10A: 9B/JV-3: ZV. Dorsal shield (Fig. 4a) smooth, with anterolateral striated. Dorsal shield 359 (348–365) long and 238 (230–248) wide, distances between setae j1- J5 344 (331–353) and s4-s4 206 (198–213), shield nearly oval, constricted at level of R1. Setae r3 and R1 on soft membranous cuticle laterad dorsal shield, r3 at level of z4, R1 at level of shield incisions. All setae smooth, except Z5 slightly serrate. With six pairs of solenostomes on dorsal shield (gd1, gd2, gd4, gd6, gd8, gd9). Length of setae: j1 33 (30–35), j3 14 (13–15), j4 8 (7–9), j5 7 (5–8), j6 8 (7–8), J2 12 (10–13), J5 7 (5–8), z2 10 (9–11), z4 10 (8–12), z5 8 (7–9), Z1 11 (9–12), Z4 13 (11–14), Z5 56 (53–58), s4 14 (13–16), S2 14 (12–15), S4 14 (13–16), S5 12 (11–14), r3 13 (12–15), R1 10 (9–11).
Venter —(Fig. 4b). All ventral setae smooth. Sternal shield smooth, posterior margin not discernible in the examined specimens, 79 (78–81) wide, with three pairs of setae st1 33 (32–35), st2 29 (27–32), st3 30 (28–32), and two pairs of lyrifissures (pst1-pst2), distance between st1-st3 59 (57–60) and st2-st2 66 (65–68). Metasternal platelets drop-shaped, each with one metasternal seta, st4 27 (25–29) and one lyrifissure (pst3). Genital shield smooth, with one pair of thin genital setae st5 30 (29–31), distance between st5-st5 78 (76–80); one pair of associated poroids on soft cuticle near posterior corners of shield. Ventrianal shield smooth, nearly oval, 105 (102–109) long, 49 (47–52) wide at level of ZV2, 81 (79–83) wide at level of anus, three pairs of thin pre-anal setae, JV1 migrate next to JV2, JV1 29 (27–32), JV2 24 (23–26), ZV2 16 (14–19); Pa 15 (14–16), Pst 16 (15–17) long. Pre-anal pores crescentic, posteromedian to JV2, distance between pores 33 (31–34). Opisthogastric soft cuticle with four pairs of setae, ZV1 22 (20–23), ZV3 10 (8–11), JV4 13 (13–14), JV5 28 (27–30) long. All ventral setae thin, except JV5, thicker. Two pairs of metapodal platelets, primary platelets 22 (21–23) long, 5 (4–6) wide, secondary ones 13 (12–14) long, 2 (1–2) wide.
Peritremes — Peritremes extending anteriorly between j3 and z2.
Chelicerae — (Fig. 4c). Fixed digit 25 (24–27) long, with three teeth and pilus dentilis, movable digit 23 (21–24) long, unidentate.
Spermatheca — (Fig. 4d). Calyx of spermatheca 12 (10–13) long, base swollen, tubular, 3 (3–4) wide at inflated part, 1 (1–1) at distal part, atrium 1 1 (1–2) wide, small-knobbed, incorporated with calyx; major duct narrow, directly attached to calyx, without neck, and minor duct visible.
Legs — Genua formula for leg I 2 – 2/1, 2/1 – 2, leg II 2 – 2/0, 2/0 – 1, leg III 1 – 2/1, 2/0 – 1, leg IV 1 – 2/1, 2/1 – 0. Genua and tibia III each with one macroseta, Sge III 27 (25–28), tibia III 23 (21–24). Leg IV with three macrosetae on genu, tibia and basitarsus (Fig. 4e), Sge IV 36 (35–38), Sti IV 35 (33–36) and St IV 55 (52–57).
Material examined — 2 ♀♀, Cao Lanh district, Dong Thap Provine (accession no. DT-0532, DT-0560), on Citrus reticulata × sinensis, 13 IV 2018, Fang X.D. coll.
Previous records — Australia, China, Cook Islands, Fiji, Hawaii, India, Indonesia, Japan, Malaysia, Mauritius, Mexico, New Zealand, Papua New Guinea, Philippines, Sri Lanka.
Remarks — The calyx of the examined specimens is tubular, similar to illustration in Wu et al. (2009), but not distal flaring as illustration in Gupta (1986). Fixed digit of the examined specimens with three teeth is similar to illustration in Liao et al. (2017), but not only with one tooth as the original description (Evans, 1953).
Euseius ovalis can be mass-reared on pollen and it is the dominant natural enemy in controlling Aceria litchi (Keifer) (Wu et al. 1991). This species also had potentiality in controlling Tetranychus kanzawai Kishida (Shih et al. 1993) and Oligonychus mangiferus (Rahman and Sapra) (Nguyen and Shih, 2010). Liao et al. (2017) reported E. ovalis rarely located on the leaves with Tetranychus colonies and only located close to Oligonychus and Panonychus colonies.
The above three species are all found in citrus orchards in Vietnam, and they are associated with spider mites and small pests. Now, there are fifteen species reported in Vietnam in total. Nguyen et al. (2016) first reported eight phytoseiid mites were found in Ho Chi Minh city, including A. matinikus Schicha and Corpuz-Raros, 1992, Graminaseius polisensis (Schicha and Corpuz-Raros, 1992), Neoseiulus longispinosus (Evans, 1952), Paraphytoseius orientalis (Narayanan, Kaur and Ghai, 1960) (synonym of Paraphytoseius multidentatus Swirski and Shechter according to Chant and McMurtry 2003, Kreiter et al. 2018), Scapulaseius asiaticus (Evans, 1953), Amblyseius tamatavensis Blommers, 1974, Proprioseiulus dahonagnas (Schicha and Corpuz-Raros, 1992) and an undetermined Typhlodromus sp. Furthermore, Paraphytoseius cracentis (Corpuz and Rimando, 1966), Neoseiulus californicus (McGregor, 1954), Proprioseiopsis lenis (Corpuz and Rimando, 1966), E. aizawai (Ehara and Bhandhufalck, 1977), E. ovalis (Evans, 1953) and A. largoensis were reported to be found in the Red River Delta respectively (Nguyen and De Clercq, 2018; Thuan et al. 2019; Nguyen, 2019a, 2019b; Nguyen and Dao 2019; Nguyen et al. 2019a, 2019b). In this paper, A. obtuserelus was first recorded in Vietnamese fauna. Fourteen identified species all belong to the subfamily Amblyseiinae. A key to the Vietnamese Amblyseiinae species is given as below.
Key to the Vietnamese Amblyseiinae species
1. Ventrianal shield oval, JV1 migrate next to JV2
...... 2
— Ventrianal shield non-oval, JV1 not migrate next to JV2
...... 3
2. Spermatheca with calyx slender, tubular
...... Euseius ovalis
— Spermatheca with calyx short, saccular
...... Euseius aizawai
3. Setae S4 absent
...... 4
—Setae S4 present
...... 5
4. Setae S5 absent, with seven macrosetae on Leg IV
...... Paraphytoseius orientalis
— Setae S5 present, with five macrosetae on Leg IV
...... Paraphytoseius cracentis
5. J2 absent
...... 6
— J2 present
...... 7
6. S2 absent, Leg I with one or more macrosetae on tibia
...... Proprioseiulus dahonagnas
— S2 present, Leg I without elongate macrosetae on tibia
...... Proprioseiopsis lenis
7. Ratio seta s4: Z1 < 3.0: 1: 0
...... 8
— Ratio seta s4: Z1 \textgreater 3.0: 1: 0
...... 10
8. GeII without a macroseta
...... 9
— GeII with a macroseta
...... Scapulaseius asiaticus
9. Most dorsal setae very long
...... Neoseiulus longispinosus
— Dorsal setae general long
...... Neoseiulus californicus
10. Ventrianal shield vase-shaped
...... Amblyseius largoensis
— Ventrianal shield pentagonal, sub-pentagonal
...... 11
11. Cervix bladder-like, with spines
...... Amblyseius matinikus
— Cervix tubular or funnel-like
...... 12
12. Leg IV with two macrosetae on genu and basitarsus
...... Graminaseius polisensis
— Leg with three macrosetae on genu, tibia and basitarsus
...... 13
13. The calyx of spermatheca with an granular surface on the distal part
...... Amblyseius obtuserellus
— The calyx of spermatheca without an granular surface
...... Amblyseius tamatavensis
Acknowledgements
We thank to Professors-Drs Serge Kreiter (Institut Agro Montpellier), Francisco Ferragut (UPV) and Antonio Carlos Lofego (SPSU) for revisions of an earlier version of the manuscript. This paper was supported by the Special Project of Guangdong Academy of Sciences (Grant No. 2019GDASYL-0503004), and GDAS Special Project of Science and Technology Development (2018GDASCX-0107).
References
Amitai S., Swirski E. 1981. A new species of Amblyseius (Acarina: Phytoseiidae) from the Far East. Isr. J. Entomol., 15: 59-66.
Beard J.J. 2001. A review of Australian Neoseiulus Hughes and Typhlodromips de Leon (Acari: Phytoseiidae: Amblyseiinae). Invert. Taxon., 15: 73-58. doi:10.1071/IT99017 ![]()
Blommers L. 1974. Species of the genus Amblyseius Berlese, 1914, from Tamatave, east Madagascar (Acarina: Phytoseiidae). Bull. Zool. Mus. Univ. Amsterdam, 3: 143-55.
Blommers L. 1976. Some Phytoseiidae (Acarina: Mesostigmata) from Madagascar, with descriptions of eight new species and notes on their biology. Bijd. tot. Dierk., 46(1): 80 -106. doi:10.1163/26660644-04601005 ![]()
Chant D.A. 1959. Phytoseiid mites (Acarina: Phytoseiidae). Part I. Bionomics of seven species in southeastern England. Part II. A taxonomic review of the family Phytoseiidae, with descriptions of thirty-eight new species. Canad. Entomol., 61(12): 1-166.
Chant D.A., McMurtry J.-A. 2003. A review of the subfamily Amblyseiinae Muma (Acari: Phytoseiidae): Part ii. The tribe Kampimodromini Kolodochka. Int. J. Acarol., 29(3): 179-224. doi:10.1080/01647950308684331 ![]()
Chant D.A., McMurtry J.A. 2004. A review of the subfamily Amblyseiinae Muma (Acari: Phytoseiidae): Part III. The tribe Amblyseiini Wainstein, subtribe Amblyseiina n. subtribe. Int. J. Acarol., 30(3): 171-228. doi:10.1080/01647950408684388 ![]()
Chant D.A., McMurtry J.A. 2007. Illustrated keys and diagnoses for the genera and subgenera of the Phytoseiidae of the world (Acari: Mesostigmata). Indira Publishing House, West Bloomfield, 219 pp.
Chant D.A., Yoshida-Shaul E., 1991. Adult ventral setal patterns in the family Phytoseiidae (Acari: Gama sina). Int. J. Acarol., 17: 187-199. doi:10.1080/01647959108683906 ![]()
Chant D.A., Yoshida-Shaul E. 1992. Adult idiosomal setal patterns in the family Phytoseiidae (Acari: Gamasina). Int. J. Acarol., 18: 177-193. doi:10.1080/01647959208683949 ![]()
Chant D.A., Hansell R.I.C., Rowell H.J., Yoshida-Shaul E. 1978. A study of the Family Phytoseiidae (Acarina: Mesostigmata) using the methods of numerical taxonomy. Canad. J. Zool., 56: 1330-1347. doi:10.1139/z78-185 ![]()
Chen S., Zhu Z., Liang L. 1984. Phytoseiidae. In: Jiangxi University (Eds). Agricultural Mites of China. Shanghai: Shanghai Science & Technology Press: 306-363 [in Chinese].
Collyer E. 1964. The occurrence of some mites of the family Phytoseiidae in New Zealand, and descriptions of seven new species. Acarologia, 6: 632-646.
Corpuz L.A., Rimando L. 1966. Some Philippine Amblyseiinae (Phytoseiidae: Acarina). The Philipp. Agric., 50: 114-136.
De Leon D. 1966. Phytoseiidae of British Guyana with keys to species (Acarina: Mesostigmata). Studies on the Fauna of Suriname and other Guyanas, 8: 81-102.
De Leon D. 1967. Some mites of the Caribbean Area. Part I. Acarina on plants in Trinidad, West Indies. Lawrence: Allen Press Inc. 66 pp.
Demite P.R., Gondim Jr.M.G.C., Lofego A.C., Moraes G.J. de 2014. A new species of Galendromimus Muma from Brazil (Acari: Phytoseiidae) with a review of the tribe Galendromimini Chant and McMurtry. Zootaxa, 3835(4): 593-599. doi:10.11646/zootaxa.3835.4.10 ![]()
Demite P.R., Moraes G.J. de, McMurtry J.A., Denmark H.A., Castilho R.C. 2019. Phytoseiidae Database. Available from: http://www.lea.esalq.usp.br/phytoseiidae/ ![]() (accessed 10 VIII 2019).
(accessed 10 VIII 2019).
Denmark H.A., Evans G.A. 2011. Phytoseiidae of North America and Hawaii (Acari: Mesostigmata). Indira Publishing House, West Bloomfield, USA, 451 pp.
Denmark H.A., Muma M.H. 1989. A revision of the genus Amblyseius Berlese, 1914 (Acari: Phytoseiidae). Occasional Papers of the Florida State Collection of Arthropods, 4, pp. 149.
Ehara S. 1959. Some predatory mites of the genera Typhlodromus and Amblyseius from Japan (Phytoseiidae). Acarologia, 1: 285-295.
Ehara S. 1966. A tentative catalogue of predatory mites of Phytoseiidae known from Asia, with descriptions of five new species from Japan. Mushi, 39: 9-30.
Ehara S., Amano H. 1998. A revision of the mite family Phytoseiidae in Japan (Acari: Gamasida), with remarks on its biology. Sp. Divers., 3: 25-73. doi:10.12782/specdiv.3.25 ![]()
Ehara S., Bhandhufalck A. 1977. Phytoseiid mites of Thailand (Acarina: Mesotigmata). J. Fac. Educ., Tottori University, Natural Science, 27(2): 43-82.
Ehara S., Yokogawa M. 1977. Two new Amblyseius from Japan with notes on three other species (Acarina: Phytoseiidae). Proc. Jpn. Soc. Syst. Zool., 13: 50-58.
Evans G.O. 1952. A new typhlodromid mite predaceous on Tetranychus bimaculatus Harvey in Indonesia. Annu. Mag. Nat. Hist., 5: 413-416. doi:10.1080/00222935208654311 ![]()
Evans G.O. 1953. On some mites of the genus Typhlodromus Scheuten, 1857, from S.E. Asia. Annu. Mag. Nat. Hist., 6: 449-467. doi:10.1080/00222935308654444 ![]()
Evans G.O. 1963. Observations on the chaetotaxy of the legs in the free-living Gamasina (Acari: Mesostigmata). Bull. Br. Mus. (Nat. Hist.) Zool., 10: 277-303. doi:10.5962/bhl.part.20528 ![]()
Fang X.D., Lu H.L., Ouyang G.C., Xia Y.L., Guo M.F., Wu W.N. 2013. Effectiveness of two predatory mite species (Acari: Phytoseiidae) in controlling Diaphorina citri (Hemiptera: Liviidae). Fla. Entomol., 96(4): 1325-1333. doi:10.1653/024.096.0411 ![]()
Fang X.D., Ouyang G.C., Lu H.L., Guo M.F., Wu W.N. 2018. Ecological control of citrus pests primarily using predatory mites and the bio-rational pesticide matrine. Int. J. Pest Manage., 64(3): 262-270. doi:10.1080/09670874.2017.1394507 ![]()
Ferragut F., Moraes G.J. de, Návia D. 2011. Phytoseiid mites (Acari: Phytoseiidae) of the Dominican Republic, with a re-definition of the genus Typhloseiopsis De Leon. Zootaxa, 2997: 37-53. doi:10.1186/1742-9994-8-19 ![]()
Garman P. 1958. New species belonging to the genera Amblyseius and Amblyseiopsis with keys to Amblyseius, Amblyseiopsis, and Phytoseiulus. Ann. Entomol. Soc. Am., 51: 69-79. doi:10.1093/aesa/51.1.69 ![]()
Gondim Jr. M.G.C., Moraes G.J. de 2001. Phytoseiid mites (Acari: Phytoseiidae) associated with palm trees (Arecaceae) in Brazil. Syst. Appl. Acarol., 6: 65-94. doi:10.11158/saa.6.1.11 ![]()
Gupta S.K. 1970. Preliminary note on plant mites (Acarina) from West Bengal. Sci. Cul., 36: 98-99.
Gupta S.K. 1977. Description of four species of Amblyseius Berlese (Acarina: hytoseiidae) from India. Entomol. Month. Mag., 112: 53-58.
Gupta S.K. 1978. Some Phytoseiidae from south India with descriptions of five new species. Orient. Insects, 12: 327-338. doi:10.1080/00305316.1978.10432093 ![]()
Gupta S.K. 1985. Plant mites of India. Zooogical Survey of India Handbook Series, Calcutta, 5: 520 pp.
Gupta S.K. 1986. Fauna of India (Acari: Mesostigmata) Family Phytoseiidae. Zoological Survey of India, Calcutta, India, 350 pp.
Hernandes F.A., Kreiter S., Tixier M.-S. 2011. Biogeographical analysis within the Family Phytoseiidae Berlese (Acari: Mesostigmata): an example from the large sub-genus Typhlodromus (Anthoseius) De Leon. Acarologia, 51(4): 431-448. doi:10.1051/acarologia/20112026 ![]()
Hirschmann W. 1962. Gangystematik der Parasitiformes. Acarologie Schriftenreihe fur Vergleichende Milbenkunde, Hirschmann-Verlag, Furth/Bay, 5(5-6), pp.80+ plates 32.
Huang M.D. 2011. Studies and application of Phytoseiids in China. Guangzhou: Sun Yat-sen University Press. 104 pp.
Kreiter S., Fontaine O., Payet R.-M. 2018. New records of Phytoseiidae (Acari: Mesostigmata) from Mauritius. Acarologia, 58(4): 773-785. doi:10.24349/acarologia/20184273 ![]()
Liao J.-R., Ho C.C., Ko C.C. 2017. Species of the genus Euseius Wainstein (Acari: Phytoseiidae: Amblyseiinae) from Taiwan. Zootaxa, 4226(2): 205-228. doi:10.11646/zootaxa.4226.2.3 ![]()
McGregor E.A. 1954. Two new mites in the genus Typhlodromus (Acarina: Phytoseiidae). Bull. South. Calif. Acad. Sci., 53: 89-92.
McMurtry J.A., Moraes G.J. de. 1984. Some phytoseiid mites from the South Pacific, with descriptions of new species and a definition of the Amblyseius largoensis species group. Int. J. Acarol., 10: 27-37. doi:10.1080/01647958408683347 ![]()
Moraes G.J. de, McMurtry J.A., Denmark H.A. 1986. A catalog of the mite family Phytoseiidae. References to taxonomy, synonymy, distribution and habitat. EMBRAPA - DDT, Brasilia, Brazil, 353 pp.
Moraes G.J. de, Kreiter S., Lofego A.C. 2000. Plant mites (Acari) of the French Antilles. 3. Phytoseiidae (Gamasida). Acarologia, 40(3): 237-264. doi:10.1016/j.jval.2014.08.1890 ![]()
Moraes G.J. de, McMurtry J.A., Denmark H.A., Campos C.B. 2004a. A revised catalog of the mite family Phytoseiidae. Zootaxa, 434: 1-494. doi:10.11646/zootaxa.434.1.1 ![]()
Moraes G.J. de, Lopes P.C., Fernando C.P. 2004b. Phytoseiid mite (Acari: Phytoseiidae) of coconut growing areas in Sri Lanka, with descriptions of three new species. J. Acarol. Soc. Jpn., 13(2): 141-160. doi:10.2300/acari.13.141 ![]()
Muma M.H. 1955. Phytoseiidae (Acarina) associated with citrus in Florida. Ann. Entomol. Soc. Am., 48: 262-272. doi:10.1093/aesa/48.4.262 ![]()
Muma M.H. 1961. Subfamiles, genera, and species of Phytoseiidae (Acarina: Mesostigmata). Bull. Fla. State Mus., 5(7): 267-302.
Muma M.H., Denmark H.A. 1970. Phytoseiidae of Florida. Arthropods of Florida and neighboring land areas, 6. Florida Department of Agriculture and Consumer Services, Division of Plant Industry, Gainesville, USA, 150 pp.
Narayanan E.S., Kaur R.B., Ghai S. 1960. Importance of some taxonomic characters in the family Phytoseiidae Berl., 1916, (predatory mites) with new records and descriptions of species. Proc. Nat. Inst. Sci. India, 26B: 384-394.
Nguyen D.Q., Renwick J., McGregor J. 2014. Variations of surface temperature and rainfall in Vietnam from 1971 to 2010. Int. J. Climatol., 34: 249-264. doi:10.1002/joc.3684 ![]()
Nguyen D.T. 2019a. Study on biological characteristics of predatory mite Euseius aizawai and Amblyseius swirskii (Acari: Phytoseiidae) to control broad mite Polyphagotarsonemus latus. Sci. Technol. J. Agric. Rural Develop. (Ministry of agriculture and Rural development, Vietnam), 8: 11-17 [In Vietnamese].
Nguyen D.T. 2019b. Evaluating using pollen in rearing predatory mite Euseius ovalis (Evans) (Acari: Phytoseiidae). Sci. Technol. J. Agric. Rural Develop. (Ministry of agriculture and Rural development, Vietnam), 9: 35-40 [In Vietnamese].
Nguyen D.T., Dao T.L. 2019. Effect of temperatures and diets on biological characteristics of predatory mite Amblyseius largoensis (Acari: Phytoseiidae). Sci. Technol. J. Agric. Rural Develop. (Ministry of agriculture and Rural development, Vietnam), 11: 66-72 [In Vietnamese].
Nguyen D.T., De Clercq P. 2018. Some morphological and biological characteristics of predatory mite Paraphytoseius cracentis (Acari: Phytoseiidae) reared on two different preys. Vietnam J. Agri. Sci., 16(2): 95-104 [In Vietnamese].
Nguyen D.T., Than A.T., Jonckheere W., Nguyen V.H., Leeuwen T.V., De Clercq P. 2019a. Life tables and feeding habits of Proprioseiopsis lenis (Acari: Phytoseiidae) and implications for its biological control potential in Southeast Asia. Syst. Appl. Acarol. 24(5): 857-865. doi:10.11158/saa.24.5.9 ![]()
Nguyen T.P.T., Tran N.V., Tran T.T.A., 2016. Species occurrence of phytophagous and predatory mites (Acari: Tetranychidae, Phytoseiidae) on fruit vegetables in Ho Chi Minh City, Vietnam. J. Acarol. Soc. Jpn., 25(S1): 133-136. doi:10.2300/acari.25.Suppl\_133 ![]()
Nguyen T.V., Shih C.I.T. 2010. Development of Neoseiulus womersleyi (Schicha) and Euseius ovalis (Evans) feeding on four tetranychid mites (Acari: Phytoseiidae, Tetranychidae) and pollen. J. Asia-Pac. Entomol, 13: 289-296. doi:10.1016/j.aspen.2010.06.010 ![]()
Nguyen V.H., Jonckheere W., Nguyen D.T., Moraes G.J. de, Leeuwen T. van., De Clercq P. 2019b. Phytoseiid mites prey effectively on thrips eggs: Evidence from predation trials and molecular analyses. Biol. Control. 137: 1-7. doi:10.1016/j.biocontrol.2019.104012 ![]()
Oliveira D.C., Charanasri V., Kongchuensin M., Konvipasruang P., Chandrapatya A., Moraes G.J. de 2012. Phytoseiidae of Thailand (Acari: Mesostigmata), with a key for their identification. Zootaxa, 3453: 1-24. doi:10.1186/1742-9994-9-22 ![]()
Ouyang C.H., Tang Y.Z., Zhong L., Zhong A.P. 2007. Report on the effects of releasing Amblyseius barkeri (Hughes) to control Panonychus citri McGregor in Anyuan County. Jiangxi Plant Prot., 30: 101-104. [In Chinese].
Prasad V. 1974. A catalogue of mites of India. Indira Acarology Publishing House, Ludhiana, Punjab, India, 320 pp.
Ray S., Gupta S.K. 1981. A new species of Platyseiella Muma (Acari: Phytoseiidae), with collection records of twelve other species. Bull. Zool. Surv. India, (3): 277-281.
Rowell H.J., Chant D.A., Hansell R.I.C.1978. The determination of setal homologies and setal patterns on the dorsal shield in the family Phytoseiidae (Acarina: Mesostigmata). Canad. Entomol., 110: 859-876. doi:10.4039/Ent110859-8 ![]()
Ryu M.O., Lee W.K. 1992. Ten newly recorded phytoseiid mites (Acarina: Phytoseiidae) from Korea. Korean J. Entomol., 22(1): 23-42. doi:10.2476/asjaa.40.23 ![]()
Schicha E. 1981. Two new species of Amblyseius Berlese from Queensland and New Caledonia compared with A. largoensis (Muma) from the South Pacific and A. deleoni Muma and Denmark from New South Wales (Acari: Phytoseiidae). J. Aust. Entomol. Soc., 20: 101-109. doi:10.1111/j.1440-6055.1981.tb01008.x ![]()
Schicha E., Corpuz-Raros L.A. 1992. Phytoseiidae of the Philippines. Indira Publishing House, West Bloomfield, Michigan, USA, 190 pp.
Shih C.I.T., Chang H.Y., Hsu P.H., Hwang Y.F. 1993. Responses of Amblyseius ovalis (Evans) (Acarina: Phytoseiidae) to natural food resources and two artificial diets. Exp. Appl. Acarol., 17(7): 503-519. doi:10.1007/BF00058894 ![]()
Sohn J.C., Park K.T. 2009. A new species of Wockia Heinemann, 1870 (Lepidoptera: Urodidae) from Vietnam. Trop. Lepid. Res., 19(2): 62-63. doi:10.4289/07-071.1 ![]()
Tran T.T., Bui T.T.M., Luong T.H., Cao V.C., Nguyen D.T., Nguyen V.L., Nguyen V.D. 2019. Effect of relative humidity on population growth of predatory mite. J. Vietnam Agric. Sci. Technol., 5(102): 105-110.
Ueckermann E.A., Loots G.C. 1988. The African species of the subgenera Anthoseius De Leon and Amblyseius Berlese (Acari: Phytoseiidae). Entomology Memoir, Department of Agriculture and Water Supply, Republic of South Africa,73, 168 pp.
van der Merwe G.G. 1965. South African Phytoseiidae (Acarina) I. Nine new species of the genus Amblyseius Berlese. J. Entomol. Soc. South. Afr., 28(1): 57-76.
Wainstein B.A. 1979. Predatory mites of the family Phytoseiidae (Parasitiformes) of the Primorsky Territory. Nazemnye Chlenistonogie Dal'nego Vostoka, Vladivostok, Russia. p. 137-144 [in Russian].
Wainstein B.A., Beglyarov G.A. 1971. New species of the genus Amblyseius (Parasitiformes: Phtyoseiidae) from the Primorsky Territory. Zool. Zhur., 50: 1803-1812 [in Russian].
Wu W.N. 1980. Notes on the genus Amblyseius Berlese from China (Acarina: Phytoseiidae). Natural Enem. Insects, 2(3): 39-50.
Wu W.N. 1982. Notes on the genus Amblyseius Berlese with descriptions of two new species from citrus orchards in South China (Acarina: Phytoseiidae). Acta Ent. Sinica, 25(1): 96-101.
Wu W.N., Lan W.M. 1989. Notes on the largoensis group of the genus Amblyseius from China (Acari: Phytoseiidae). Acta Zootaxon. Sinica, 14 (4): 447-452 [in Chinese].
Wu W.N., Lan W.M., Liu Y.H. 1991. Phytoseiid mites of lychee from China, and their value for application. Natural Enem. Insects, 13(2): 82-91[in Chinese].
Wu W.N., Liang L.R., Lan W.M. 1997. Acari: Phytoseiidae. Economic Insect Fauna of China, 53. Beijing: Science Press. 227 pp. [in Chinese].
Wu W.N., Ou J.F., Huang J.L. 2009. Fauna Sinica, Invertebrate Vol. 47, Arachnida, Acari, Phytoseiidae. Beijing: Science Press. 511 pp. [in Chinese].
Zannou I.D., Moraes G.J. de, Ueckermann E.A., Oliveira A.R., Yaninek J.S., Hanna R. 2007. Phytoseiid mites of the subtribe Amblyseiina (Acari: Phytoseiidae: Amblyseiini) from sub-Saharan Africa. Zootaxa, 1550: 1-47. doi: 10.11646/zootaxa.1550.1.1 doi:10.11646/zootaxa.1550.1.1 ![]()
Zhang, Y.X., Lin, J.Z., Ji, J. 2002. Controlling Panonychus citri (McGregor) with Amblyseius cucumeris produced in factory. Plant Prot. Tech. Ext., 22: 25-28 [In Chinese].



2019-09-23
Date accepted:
2020-02-27
Date published:
2020-03-06
Edited by:
Kreiter, Serge

This work is licensed under a Creative Commons Attribution 4.0 International License
2020 Fang, Xiao-Duan; Nguyen, Van-Liem; Ouyang, Ge-Cheng and Wu, Wei-Nan
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)




